Twelve Weeks Rest–Pause and Traditional Resistance Training: Effects on Myokines and Performance Adaptations among Recreationally Trained Men
Abstract
:1. Introduction
2. Results
3. Discussion
4. Materials and Methods
4.1. Research Design
4.2. Procedure
4.3. TRT Program
4.4. RP Resistance Training Program
4.5. Assessing the 1RM
4.6. Assessment of Muscle Hypertrophy
4.7. Nutrient Intake and Dietary Analysis
4.8. Assessment of Biochemical Indicators
4.9. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Prestes, J.; Tibana, R.A.; de Araujo Sousa, E.; da Cunha Nascimento, D.; de Oliveira Rocha, P.; Camarço, N.F.; de Sousa, N.M.; Willardson, J.M. Strength and muscular adaptations after 6 weeks of rest-pause vs. traditional multiple-sets resistance training in trained subjects. J. Strength Cond. Res. 2019, 33, S113–S121. [Google Scholar] [CrossRef] [PubMed]
- Marshall, P.; Robbins, D.; Wrightson, A.; Siegler, J. Acute neuromuscular and fatigue responses to the rest-pause method. J. Sci. Med. Sport. Med. Aust. 2012, 15, 153–158. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A.; Moro, T.; Marcolin, G.; Neri, M.; Bianco, A.; Palma, A.; Grimaldi, K. High-Intensity Interval Resistance Training (HIRT) influences resting energy expenditure and respiratory ratio in non-dieting individuals. J. Transl. Med. 2012, 10, 237. [Google Scholar] [CrossRef] [PubMed]
- Shimano, T.; Kraemer, W.J.; Spiering, B.A.; Volek, J.S.; Hatfield, D.L.; Silvestre, R.; Vingren, J.L.; Fragala, M.S.; Maresh, C.M.; Fleck, S.J.; et al. Relationship between the number of repetitions and selected percentages of one repetition maximum in free weight exercises in trained and untrained men. J. Strength Cond. Res. 2006, 20, 819–823. [Google Scholar] [CrossRef]
- Stull, G.A.; Clarke, D.H. Patterns of recovery following isometric and isotonic strength decrement. Med. Sci. Sports 1971, 3, 135–139. [Google Scholar] [CrossRef]
- Schoenfeld, B.J. Potential mechanisms for a role of metabolic stress in hypertrophic adaptations to resistance training. Sports Med. 2013, 43, 179–194. [Google Scholar] [CrossRef]
- Grgic, J.; Mcllvenna, L.C.; Fyfe, J.J.; Sabol, F.; Bishop, D.J.; Schoenfeld, B.J.; Pedisic, Z. Does aerobic training promote the same skeletal muscle hypertrophy as resistance training? A systematic review and meta-analysis. Sports Med. 2018, 49, 1–22. [Google Scholar] [CrossRef]
- Schoenfeld, B.J.; Ogborn, D.; Krieger, J.W. Effects of resistance training frequency on measures of muscle hypertrophy: A systematic review and meta-analysis. Sports Med. 2016, 46, 1689–1697. [Google Scholar] [CrossRef]
- Krieger, J.W. Single vs. multiple sets of resistance exercise for muscle hypertrophy: A meta-analysis. J. Strength Cond. Res. 2010, 24, 1150–1159. [Google Scholar] [CrossRef]
- Taniguchi, Y. Lateral specificity in resistance training: The effect of bilateral and unilateral training. Eur. J. Appl. Physiol. 1997, 75, 144–150. [Google Scholar] [CrossRef]
- Taniguchi, Y. Relationship between the modifications of bilateral deficit in upper and lower limbs by resistance training in humans. Eur. J. Appl. Physiol. 1998, 78, 226–230. [Google Scholar] [CrossRef]
- Lee, S.-J.; Lee, Y.-S.; Zimmers, T.A.; Soleimani, A.; Matzuk, M.M.; Tsuchida, K.; Cohn, R.D.; Barton, E.R. Barton, Regulation of muscle mass by follistatin and activins. Mol. Endocrinol. 2010, 24, 1998–2008. [Google Scholar] [CrossRef]
- Hiroki, E.; Abe, S.; Iwanuma, O.; Sakiyama, K.; Yanagisawa, N.; Shiozaki, K.; Ide, Y. A comparative study of myostatin, follistatin and decorin expression in muscle of different origin. Anat. Sci. Int. 2011, 86, 151–159. [Google Scholar] [CrossRef]
- Motevalli, M.S.; Dalbo, V.J.; Attarzadeh, R.S.; Rashidlamir, A.; Tucker, P.S.; Scanlan, A.T. The Effect of Rate of Weight Reduction on Serum Myostatin and Follistatin Concentrations in Competitive Wrestlers. Int. J. Sports. Physiol. Perform. 2015, 10, 139–146. [Google Scholar] [CrossRef]
- Sandri, M. Signaling in Muscle Atrophy and Hypertrophy. Physiology 2008, 23, 160–170. [Google Scholar] [CrossRef]
- Murach, K.; Bagley, J. Skeletal Muscle Hypertrophy with Concurrent Exercise Training: Contrary Evidence for an Interference Effect. Sports Med. 2016, 46, 1029–1039. [Google Scholar] [CrossRef]
- Schoenfeld, B. The Mechanisms of Muscle Hypertrophy and Their Application to Resistance Training. J. Strength Cond. Res. 2010, 24, 2857–2872. [Google Scholar] [CrossRef]
- Schoenfeld, B. The use of specialized training techniques to maximize muscle hypertrophy. Strength Cond. J. 2011, 33, 60–65. [Google Scholar] [CrossRef]
- Fink, J.; Schoenfeld, B.J.; Kikuchi, N.; Nakazato, K. Effects of drop set resistance training on acute stress indicators and long-term muscle hypertrophy and strength. J. Sports Med. Phys. Fitness 2018, 58, 597–605. [Google Scholar] [CrossRef]
- Ozaki, H.; Kubota, A.; Natsume, T.; Loenneke, J.P.; Abe, T.; Machida, S.; Naito, H. Effects of drop sets with resistance training on increases in muscle CSA, strength, and endurance: A pilot study. J. Sports Sci. 2018, 36, 691–696. [Google Scholar] [CrossRef]
- Girman, J.C.; Jones, M.T.; Matthews, T.D.; Wood, R.J. Acute effects of a cluster-set protocol on hormonal, metabolic and performance measures in resistance-trained males. Eur. J. Sport Sci. 2014, 14, 151–159. [Google Scholar] [CrossRef] [PubMed]
- Goto, K.; Ishii, N.; Kizuka, T.; Kraemer, R.R.; Honda, Y.; Takamatsu, K. Hormonal and metabolic responses to slow movement resistance exercise with different durations of concentric and eccentric actions. Eur. J. Appl. Physiol. 2009, 106, 731–739. [Google Scholar] [CrossRef] [PubMed]
- Kilian, Y.; Engel, F.; Wahl, P.; Achtzehn, S.; Sperlich, B.; Mester, J. Markers of biological stress in response to a single session of high-intensity interval training and high-volume training in young athletes. Eur. J. Appl. Physiol. 2016, 116, 2177–2186. [Google Scholar] [CrossRef] [PubMed]
- Peake, J.M.; Tan, S.J.; Markworth, J.F.; Broadbent, J.A.; Skinner, T.L.; Cameron-Smith, D. Metabolic and hormonal responses to isoenergetic high-intensity interval exercise and continuous moderate intensity exercise. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E539–E552. [Google Scholar] [CrossRef] [PubMed]
- Wahl, P.; Mathes, S.; Achtzehn, S.; Bloch, W.; Mester, J. Active vs. passive recovery during high-intensity training influences hormonal response. Int. J. Sports Med. 2014, 35, 583–589. [Google Scholar] [CrossRef] [PubMed]
- Urhausen, A.; Gabriel, H.; Kindermann, W. Blood hormones as markers of training stress and overtraining. Sports Med. 1995, 20, 251–276. [Google Scholar] [CrossRef]
- Elliott, B.T.; Herbert, P.; Sculthorpe, N.; Grace, F.M.; Stratton, D.; Hayes, L.D. Lifelong exercise, but not short-term high-intensity interval training, increases GDF11, a marker of successful aging: A preliminary investigation. Physiol. Rep. 2017, 5, e13343. [Google Scholar] [CrossRef]
- Walker, K.S.; Kambadur, R.; Sharma, M.; Smith, H.K. Resistance training alters plasma myostatin but not IGF-1 in healthy men. Med. Sci. Sports Exerc. 2004, 36, 787–793. [Google Scholar] [CrossRef]
- Negaresh, R.; Ranjbar, R.; Habibi, A.; Mokhtarzade, M.; Fokin, A.; Gharibvand, M. The effect of resistance training on quadriceps muscle volume and some growth factors in elderly and young men. Adv. Gerontol. Uspekhi Gerontol. 2017, 30, 880–887. [Google Scholar]
- Hofmann, M.; Schober-Halper, B.; Oesen, S.; Franzke, B.; Tschan, H.; Bachl, N.; Strasser, E.M.; Quittan, M.; Wagner, K.H.; Wessner, B. Effects of elastic band resistance training and nutritional supplementati on muscle quality and circulating muscle growth and degradation factors of institutionalized elderly women: The Vienna Active Ageing Study (VAAS). Eur. J. Appl. Physiol. 2016, 116, 885–897. [Google Scholar] [CrossRef]
- Goto, K.; Nagasawa, M.; Yanagisawa, O.; Kizuka, T.; Ishii, N.; Takamatsu, K. Muscular adaptations to combinations of highand low-intensity resistance exercises. J. Strength Cond. Res. 2004, 18, 730–737. [Google Scholar]
- Gotshalk, L.A.; Loebel, C.C.; Nindl, B.C.; Putukian, M.; Sebastianelli, W.; Newton, R.U.; Häkkinen, K.; Kraemer, W.J. Hormonal responses of multiset versus single-set heavy-resistance exercise protocols. Can. J. Appl. Physiol. 1997, 22, 244–255. [Google Scholar] [CrossRef]
- Mourkioti, F.; Kratsios, P.; Luedde, T.; Song, Y.-H.; Delafontaine, P.; Adami, R.; Parente, V.; Bottinelli, R.; Pasparakis, M.; Rosenthal, N. Targeted ablation of IKK2 improves skeletal muscle strength, maintains mass, and promotes regeneration. J. Clin. Investig. 2006, 116, 2945–2954. [Google Scholar] [CrossRef]
- Wenz, T.; Rossi, S.G.; Rotundo, R.L.; Spiegelman, B.M.; Moraes, C.T. Increased muscle PGC-1alpha expression protects from sarcopenia and metabolic disease during aging. Proc. Natl. Acad. Sci. USA 2009, 106, 20405–20410. [Google Scholar] [CrossRef]
- Gilson, H.; Schakman, O.; Kalista, S.; Lause, P.; Tsuchida, K.; Thissen, J.P. Follistatin induces muscle hypertrophy through satellite cell proliferation and inhibition of both myostatin and activin. Am. J. Physiol. Endocrinol. Metab. 2009, 297, E157–E164. [Google Scholar] [CrossRef]
- Walsh, S.; Metter, E.J.; Ferrucci, L.; Roth, S.M. Activin-type II receptor B (ACVR2B) and follistatin haplotype associations with muscle mass and strength in humans. J. Appl. Physiol. 2007, 102, 2142–2148. [Google Scholar] [CrossRef]
- Pedersen, B.; Febbraio, M. Muscles, exercise and obesity: Skeletal muscle as a secretory organ. Nat. Rev. Endocrinol. 2012, 8, 457–465. [Google Scholar] [CrossRef]
- El Shafey, N.; Guesnon, M.; Simon, F.; Deprez, E.; Cosette, J.; Stockholm, D.; Scherman, D.; Bigey, P.; Kichler, A. Inhibition of the myostatin/Smad signaling pathway by short decorin-derived peptides. Exp. Cell Res. 2016, 341, 187–195. [Google Scholar] [CrossRef]
- Barbé, C.; Kalista, S.; Loumaye, A.; Ritvos, O.; Lause, P.; Ferracin, B.; Thissen, J.P. Role of IGF-I in follistatin-induced skeletal muscle hypertrophy. Am. J. Physiol. Endocrinol. Metab. 2015, 309, E557–E567. [Google Scholar] [CrossRef]
- Enes, A.; Alves, R.C.; Schoenfeld, B.J.; Oneda, G.; Perin, S.C.; Trindade, T.B.; Prestes, J.; Souza-Junior, T.P. Rest-pause and drop-set training elicit similar strength and hypertrophy adaptations compared with traditional sets in resistance-trained males. Appl. Physiol. Nutr. Metab. 2021, 46, 1417–1424. [Google Scholar] [CrossRef]
- Oliver, J.M.; Kreutzer, A.; Jenke, S.; Phillips, M.D.; Mitchell, J.B.; Jones, M.T. Acute response to cluster sets in trained and untrained men. Eur. J. Appl. Physiol. 2015, 115, 2383–2393. [Google Scholar] [CrossRef]
- Korak, J.A.; Paquette, M.R.; Brooks, J.; Fuller, D.K.; Coons, J.M. Effect of rest-pause vs. traditional bench press training on muscle strength, electromyography, and lifting volume in randomized trial protocols. Eur. J. Appl. Physiol. 2017, 117, 1891–1896. [Google Scholar] [CrossRef]
- Peterson, M.D.; Rhea, M.R.; Alvar, B.A. Applications of the dose-response for muscular strength development: A review of meta-analytic efficacy and reliability for designing training prescription. J. Strength Cond. Res. 2005, 19, 950–958. [Google Scholar] [CrossRef]
- Rhea, M.R.; Alvar, B.A.; Burkett, L.N.; Ball, S.D. A meta-analysis to determine the dose response for strength development. Med. Sci. Sports Exerc. 2003, 35, 456–464. [Google Scholar] [CrossRef]
- Gantois, P.; Fonseca, F.D.S.; de Lima-Júnior, D.; Costa, M.D.C.; Costa, B.D.D.V.; Cyrino, E.S.; Fortes, L.D.S. Acute effects of muscle failure and training system (traditional vs. rest-pause) in resistance exercise on countermovement jump performance in trained adults. Isokinet. Exerc. Sci. 2021, 29, 11–20. [Google Scholar] [CrossRef]
- Seynnes, O.R.; Boer, M.D.; Narici, M.V. Early skeletal muscle hypertrophy and architectural changes in response to high-intensity resistance training. J. Appl. Physiol. 2007, 102, 368–373. [Google Scholar] [CrossRef] [PubMed]
- Hammarström, D.; Øfsteng, S.; Koll, L.; Hanestadhaugen, M.; Hollan, I.; Apró, W.; Whist, J.E.; Blomstrand, E.; Rønnestad, B.R.; Ellefsen, S. Benefits of higher resistance-training volume are related to ribosome biogenesis. J. Physiol. 2020, 598, 543–565. [Google Scholar] [CrossRef] [PubMed]
- Figueiredo, V.C.; de Salles, B.F.; Trajano, G.S. Volume for muscle hypertrophy and health outcomes: The most effective variable in resistance training. Sports Med. 2018, 48, 499–505. [Google Scholar] [CrossRef] [PubMed]
- Atherton, P.J.; Babraj, J.A.; Smith, K.; Singh, J.; Rennie, M.J.; Wackerhage, H. Selective activation of AMPK-PGC-1α or PKB-TSC2-mTOR signaling can explain specific adaptive responses to endurance or resistance training-like electrical muscle stimulation. FASEB J. 2005, 19, 1–23. [Google Scholar] [CrossRef]
- Sword, D.O.; Carolina, S. Exercise as a Management Strategy for the Overweight and Obese: Where Does Resistance Exercise Fit in? Strength Cond. J. 2013, 35, 47–55. [Google Scholar] [CrossRef]
- Wang, Z.; Ying, Z.; Bosy-Westphal, A.; Zhang, J.; Heller, M.; Later, W.; Heymsfield, S.B.; Müller, M.J. Evaluation of specific metabolic rates of major organs and tissues: Comparison between men and women. Am. J. Hum. Biol. 2011, 23, 333–338. [Google Scholar] [CrossRef]
- Allman, B.; Morrissey, M.C.; Kim, J.-S.; Panton, L.B.; Contreras, R.J.; Hickner, R.; Ormsbee, M.J. Fat metabolism and acute resistance exercise in trained women. J. Appl. Physiol. 2019, 126, 739–745. [Google Scholar] [CrossRef]
- Niewiadomski, W.; Laskowska, D.; Gąsiorowska, A.; Cybulski, G.; Strasz, A.; Langfort, J. Determination and Prediction of One Repetition Maximum (1RM): Safety Considerations. J. Hum. Kinet. 2008, 19, 109–120. [Google Scholar] [CrossRef]
- Barbalho, M.; Gentil, P.; Raiol, R.; Del Vecchio, F.B.; Ramirez-Campillo, R.; Coswig, V.S. High 1RM Tests Reproducibility and Validity are not Dependent on Training Experience, Muscle Group Tested or Strength Level in Older Women. Sports 2018, 6, 171. [Google Scholar] [CrossRef]
- Knapik, J.J.; Staab, J.S.; Harman, E.A. Validity of an anthropometric estimate of thigh muscle cross-sectional area. Med. Sci. Sports Exerc. 1996, 28, 1523–1530. [Google Scholar] [CrossRef]
- Heymsfield, S.B.; McManus, C.; Smith, J.; Stevens, V.; Nixon, D.W. Anthropometric measurement of muscle mass: Revised equations for calculating bone-free arm muscle area. Am. J. Clin. Nutr. 1982, 3, 680–690. [Google Scholar] [CrossRef]
- Cohen, J. Statistical Power Analysis for the Behavioral Sciences; Lawrence Erlbaum Associates: Hillsdale, NJ, USA, 1988; pp. 284–288. [Google Scholar] [CrossRef]
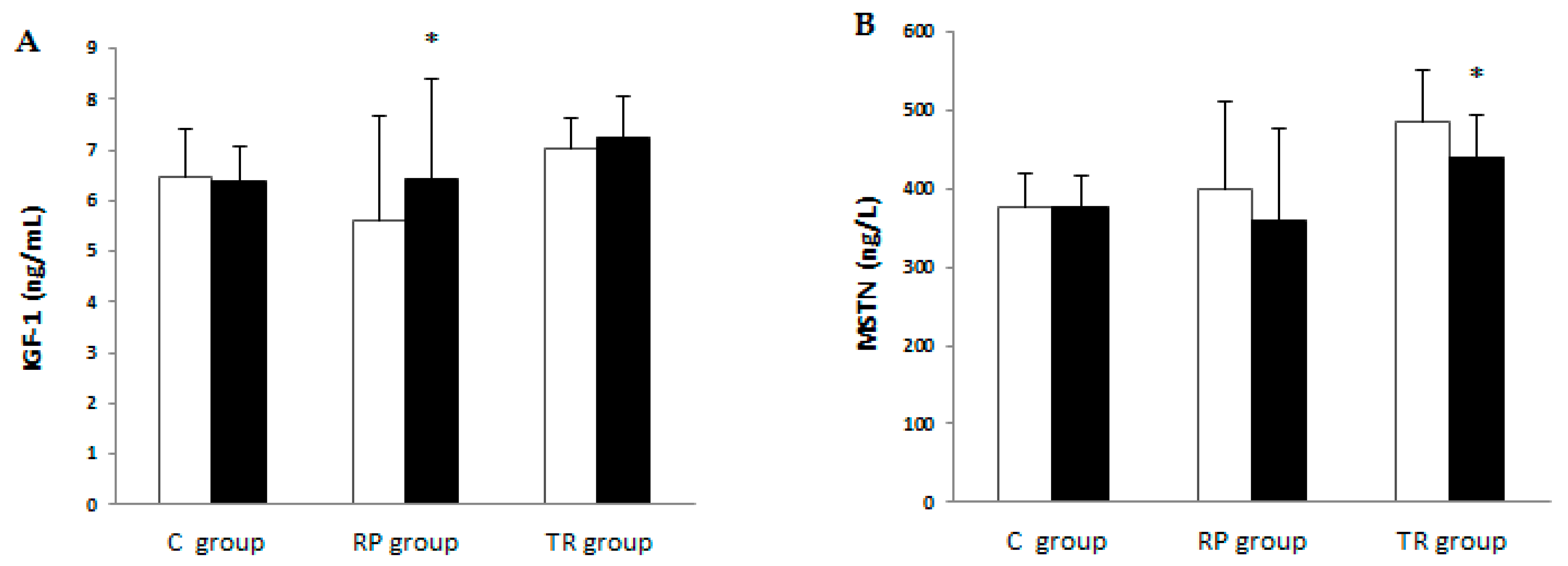
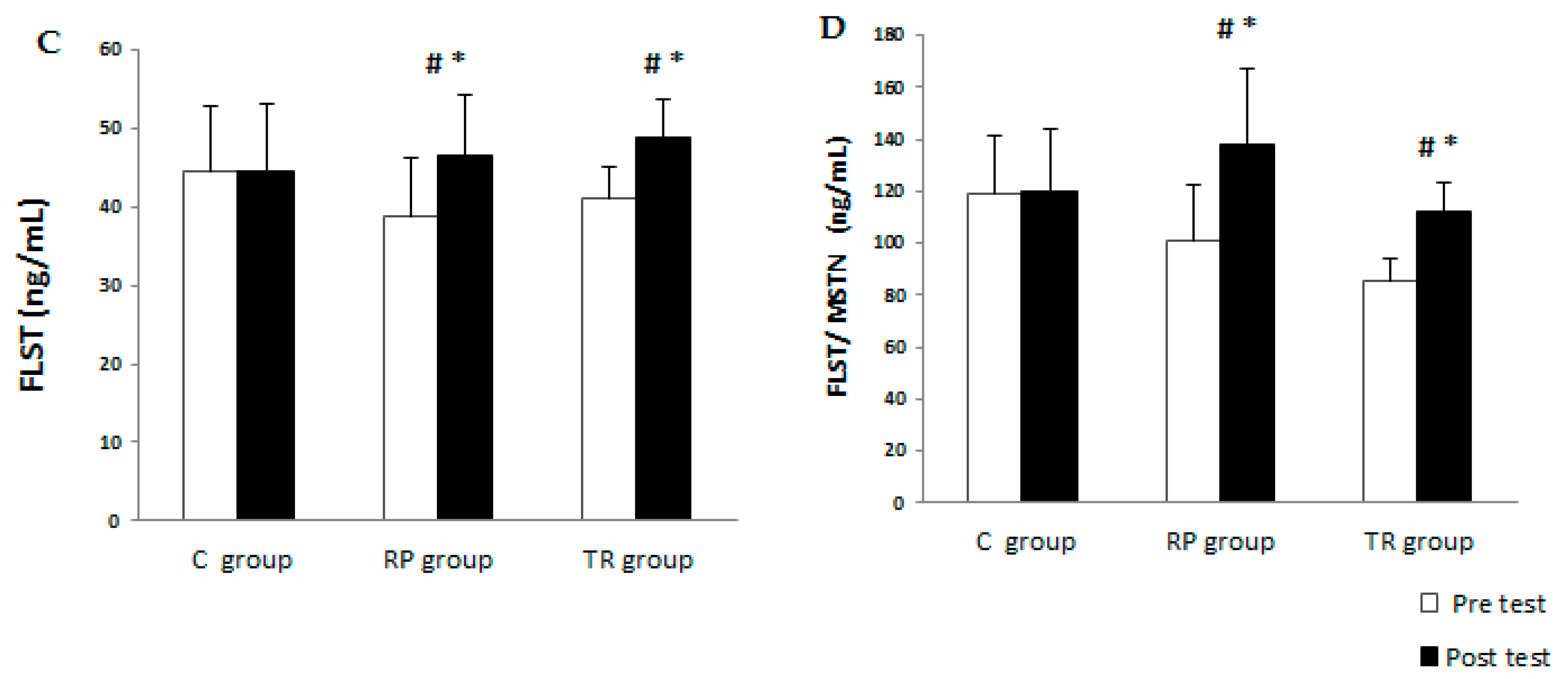

| Variable | Group | Pre-Training | Post-Training | p | ES | %Change | |
|---|---|---|---|---|---|---|---|
| Mean ± SD | Mean ± SD | Within Group | Between Group | ||||
| Leg press 1RM (kg) | Rest–Pause | 91.50 ± 16.33 | 150 ± 31.09 | <0.001 * | <0.001 * | 2.35 | 63.79 |
| Traditional training | 96.50 ± 29.44 | 140.50 ± 36.70 | <0.001 * | 1.32 | 51.19 | ||
| Control | 111.11 ± 31.69 | 113.33 ± 34.18 | 0.27 | 0.06 | 1.78 | ||
| Chest press 1RM (kg) | Rest–Pause | 57.75 ± 19.16 | 95.25 ± 17.73 | <0.001 * | <0.001 * | 2.03 | 74.90 |
| Traditional training | 42.50 ± 12.30 | 68 ± 16.19 | <0.001 * | 1.77 | 63.54 | ||
| Control | 51.66 ± 23.94 | 51.38 ± 25.52 | 0.87 | 0.01 | −0.69 | ||
| Thigh MCSA (cm2) | Rest–Pause | 130.90 ± 26.64 | 173.19 ± 33.94 | <0.001 * | <0.001 * | 1.38 | 32.86 |
| Traditional training | 135.95 ± 31.99 | 159.32 ± 34.33 | <0.001 * | 0.70 | 17.72 | ||
| Control | 136.58 ± 34.85 | 136.82 ± 39.17 | 0.93 | 0.005 | −0.25 | ||
| Arm MCSA (cm2) | Rest–Pause | 44.13 ± 6.29 | 59.26 ± 12.48 | <0.001 * | <0.001 * | 1.53 | 33.80 |
| Traditional training | 50.02 ± 9.27 | 59.61 ± 11.07 | <0.001 * | 0.93 | 19.36 | ||
| Control | 48.47 ± 14.86 | 48.38 ± 17.03 | 0.96 | 0.006 | −0.21 | ||
| Body mass (kg) | Rest–Pause | 81.42 ± 12.53 | 81.33 ± 12.80 | 0.95 | 0.52 | 0.007 | 0.04 |
| Traditional training | 80.57 ± 13.33 | 82.51 ± 11.19 | 0.13 | 0.15 | 2.93 | ||
| Control | 80.17 ± 19.56 | 80.57 ± 20.22 | 0.61 | 0.02 | 0.35 | ||
| BMI (kg/m2) | Rest–Pause | 26.14 ± 2.87 | 26.07 ± 2.38 | 0.90 | 0.67 | 0.02 | 0.06 |
| Traditional training | 24.59 ± 3.41 | 25.24 ± 2.89 | 0.09 | 0.20 | 3.05 | ||
| Control | 25.87 ± 4.21 | 25.99 ± 4.29 | 0.64 | 0.02 | 0.42 | ||
| SMM (kg) | Rest–Pause | 37.18 ± 4.80 | 38.43 ± 4.88 | 0.02 * | 0.05 * | 0.25 | 3.48 |
| Traditional training | 38.83 ± 6.25 | 40.46 ± 5.76 | 0.01 * | 0.27 | 4.58 | ||
| Control | 34.95 ± 4.93 | 34.87 ± 5.65 | 0.85 | 0.01 | −0.48 | ||
| BF (%) | Rest–Pause | 22.59 ± 4.88 | 20.54 ± 4.44 | 0.02 * | 0.01 * | 0.43 | −8.57 |
| Traditional training | 15.66 ± 3.26 | 13.79 ± 3.51 | 0.001 * | 0.55 | −12.38 | ||
| Control | 26.91 ± 4.13 | 27.31 ± 4.44 | 0.50 | 0.09 | 1.5 | ||
| Week 1–3 | Week 4–6 | Week 7–9 | Week 10–12 | ||
|---|---|---|---|---|---|
| Rest–pause resistance training | Set | 1 | 1 | 1 | 1 |
| Rep | 30 | 24 | 18 | 12 | |
| Rest intra-set (s) | 30 | 25 | 20 | 15 | |
| Rest between exercises (min) | 3 | 3 | 3 | 3 | |
| Intensity (% 1RM) | 70 | 75 | 80 | 85 | |
| Total repetitions for each exercise | 30 | 24 | 18 | 12 | |
| Traditional resistance training | Set | 3 | 3 | 3 | 3 |
| Rep | 10 | 8 | 6 | 4 | |
| Rest inter-sets (min) | 2 | 2 | 2 | 2 | |
| Rest between exercises (min) | 3 | 3 | 3 | 3 | |
| Intensity (% 1RM) | 70 | 75 | 80 | 85 | |
| Total repetitions for each exercise | 30 | 24 | 18 | 12 | |
| Variable | Group | Pre-Training | Post-Training | p |
|---|---|---|---|---|
| Mean ± SD | Mean ± SD | |||
| Calories (Kcal/day) | Rest–Pause | 2369.28 ± 76.10 | 2351.37 ± 205.38 | 0.79 |
| Traditional training | 2335.48 ± 97.20 | 2228.22 ± 95.34 | 0.07 | |
| Control | 2368.06 ± 134.59 | 2347.49 ± 131.58 | 0.74 | |
| Carbohydrate (gr/day) | Rest–Pause | 326.83 ± 43.63 | 322.85 ± 38.37 | 0.83 |
| Traditional training | 307.35 ± 29.07 | 293.46 ± 42.44 | 0.30 | |
| Control | 319.05 ± 30.16 | 309.58 ± 30.25 | 0.49 | |
| Protein (gr/day) | Rest–Pause | 119.58 ± 17.62 | 115.79 ± 12.79 | 0.51 |
| Traditional training | 129.12 ± 35.63 | 130.27 ± 32.27 | 0.93 | |
| Control | 120.43 ± 26.94 | 125.83 ± 26.44 | 0.60 | |
| Fat (gr/day) | Rest–Pause | 64.84 ± 14.19 | 66.31 ± 16.14 | 0.67 |
| Traditional training | 65.50 ± 12.06 | 59.26 ± 13.45 | 0.20 | |
| Control | 67.79 ± 14.97 | 67.31 ± 21.85 | 0.93 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Karimifard, M.; Arazi, H.; Mehrabani, J. Twelve Weeks Rest–Pause and Traditional Resistance Training: Effects on Myokines and Performance Adaptations among Recreationally Trained Men. Stresses 2023, 3, 302-315. https://doi.org/10.3390/stresses3010022
Karimifard M, Arazi H, Mehrabani J. Twelve Weeks Rest–Pause and Traditional Resistance Training: Effects on Myokines and Performance Adaptations among Recreationally Trained Men. Stresses. 2023; 3(1):302-315. https://doi.org/10.3390/stresses3010022
Chicago/Turabian StyleKarimifard, Mahmood, Hamid Arazi, and Javad Mehrabani. 2023. "Twelve Weeks Rest–Pause and Traditional Resistance Training: Effects on Myokines and Performance Adaptations among Recreationally Trained Men" Stresses 3, no. 1: 302-315. https://doi.org/10.3390/stresses3010022







