The Evolving Role for Zinc and Zinc Transporters in Cadmium Tolerance and Urothelial Cancer
Abstract
1. Introduction
2. Zinc, Zinc Transporters, and Metallothionein
2.1. Zinc Transporters, Deranged Zinc Homeostasis and Cancer
2.2. Putative Zinc Transporters Involved in Tolerance to Cadmium Toxicity
2.3. Metallothionein-Metal Complexes
2.4. Zinc (Cadmium) as a Concomitant Inducer of MT and ZnT1
2.5. Metallothionein as a ‘Double-Edged Sword’
3. Cadmium as a Risk Factor for Bladder Cancer
3.1. Cadmium-Induced Cell Transformation: An In Vitro Carcinogenicity Test
3.2. UROtsa Cell Line as a Cell Model to Dissect the Carcinogenicity of Cadmium
3.3. Zinc Transporters Expressed by Parental UROtsa Cells
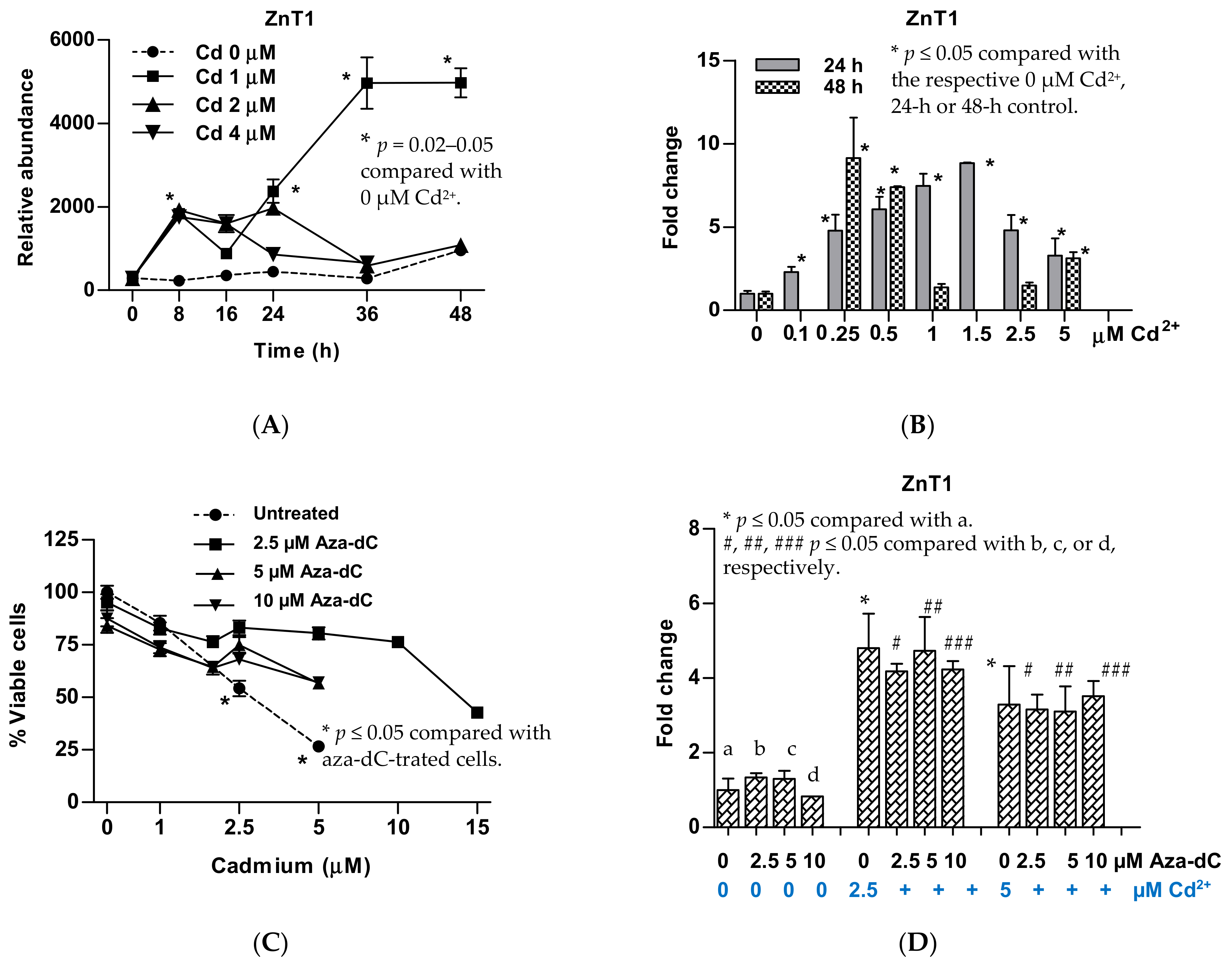
3.4. Upregulation of ZnT1 and Acquired Resistance to Cadmium
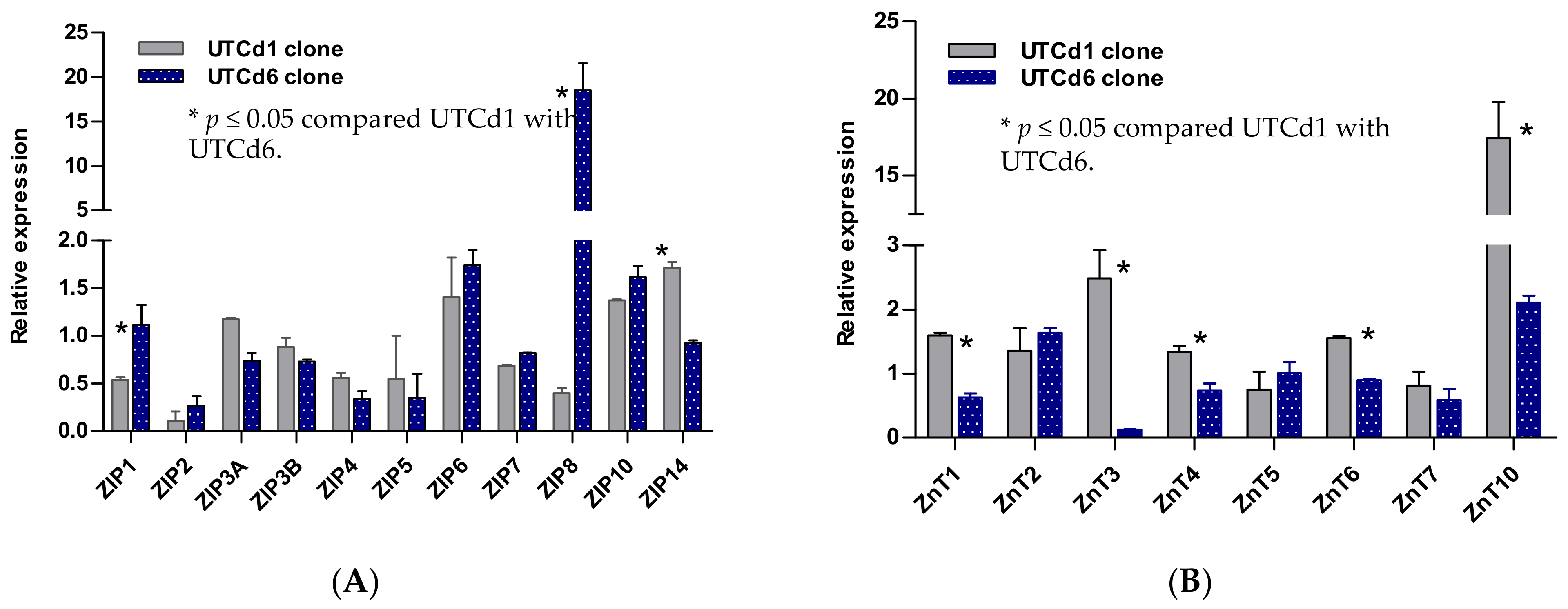
4. Insights from Transformed UROtsa Cells
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Satarug, S.; Vesey, D.A.; Gobe, G.C. Health risk assessment of dietary cadmium intake: Do current guidelines indicate how much is safe? Environ. Health Perspect. 2017, 125, 284–288. [Google Scholar] [CrossRef]
- Satarug, S.; Vesey, D.A.; Gobe, G.C. Current health risk assessment practice for dietary cadmium: Data from different countries. Food Chem. Toxicol. 2017, 106, 430–445. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S. Dietary Cadmium Intake and Its Effects on Kidneys. Toxics 2018, 6, 15. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Phelps, K.R. Cadmium Exposure and Toxicity. In Metal Toxicology Handbook; Bagchi, D., Bagchi, M., Eds.; CRC Press: Boca Raton, FL, USA, 2021; pp. 219–274. [Google Scholar]
- Pappas, R.S.; Fresquez, M.R.; Watson, C.H. Cigarette Smoke Cadmium Breakthrough from Traditional Filters: Implications for Exposure. J. Anal. Toxicol. 2015, 39, 45–51. [Google Scholar] [CrossRef] [PubMed]
- Thévenod, F.; Fels, J.; Lee, W.-K.; Zarbock, R. Channels, transporters and receptors for cadmium and cadmium complexes in eukaryotic cells: Myths and facts. BioMetals 2019, 32, 469–489. [Google Scholar] [CrossRef]
- Flanagan, P.R.; McLellan, J.S.; Haist, J.; Cherian, M.G.; Chamberlain, M.J.; Valberg, L.S. Increased dietary cadmium absorption in mice and human subjects with iron deficiency. Gastroenterology 1978, 46, 609–623. [Google Scholar] [CrossRef]
- Finley, J.W. Manganese absorption and retention by young women is associated with serum ferritin concentration. Am. J. Clin. Nutr. 1999, 70, 37–43. [Google Scholar] [CrossRef]
- Fujishiro, H.; Hamao, S.; Tanaka, R.; Kambe, T.; Himeno, S. Concentration-dependent roles of DMT1 and ZIP14 in cadmium absorption in Caco-2 cells. J. Toxicol. Sci. 2017, 42, 559–567. [Google Scholar] [CrossRef]
- Aydemir, T.B.; Cousins, R.J. The Multiple Faces of the Metal Transporter ZIP14 (SLC39A14). J. Nutr. 2018, 148, 174–184. [Google Scholar] [CrossRef]
- Jenkitkasemwong, S.; Wang, C.-Y.; MacKenzie, B.; Knutson, M.D. Physiologic implications of metal-ion transport by ZIP14 and ZIP8. BioMetals 2012, 25, 643–655. [Google Scholar] [CrossRef]
- Rentschler, G.; Kippler, M.; Axmon, A.; Raqib, R.; Skerfving, S.; Vahter, M.; Broberg, K. Cadmium concentrations in human blood and urine are associated with polymorphisms in zinc transporter genes. Metallomics 2014, 6, 885–891. [Google Scholar] [CrossRef]
- Moulis, J.M.; Bourguinon, J.; Catty, P. Chapter 23 Cadmium. In RSC Metallobiology Series No. 2, Binding, Transport and Storage of Metal. Ions in Biological Cells; Wolfgang, M., Anthony, W., Eds.; The Royal Society of Chemistry: London, UK, 2014; pp. 695–746. [Google Scholar]
- Carpenter, M.C.; Shah, A.S.; DeSilva, S.; Gleaton, A.; Su, A.; Goundie, B.; Croteau, M.L.; Stevenson, M.J.; Wilcox, D.E.; Austin, R.N. Thermodynamics of Pb(ii) and Zn(ii) binding to MT-3, a neurologically important metallothionein. Metallomics 2016, 8, 605–617. [Google Scholar] [CrossRef]
- King, J.C.; Shames, D.M.; Woodhouse, L.R. Zinc Homeostasis in Humans. J. Nutr. 2000, 130, 1360S–1366S. [Google Scholar] [CrossRef] [PubMed]
- Lichten, L.A.; Cousins, R.J. Mammalian Zinc Transporters: Nutritional and Physiologic Regulation. Annu. Rev. Nutr. 2009, 29, 153–176. [Google Scholar] [CrossRef] [PubMed]
- King, J.C. Zinc: An essential but elusive nutrient. Am. J. Clin. Nutr. 2011, 94, 679S–684S. [Google Scholar] [CrossRef]
- Kambe, T.; Tsuji, T.; Hashimoto, A.; Itsumura, N. The Physiological, Biochemical, and Molecular Roles of Zinc Transporters in Zinc Homeostasis and Metabolism. Physiol. Rev. 2015, 95, 749–784. [Google Scholar] [CrossRef] [PubMed]
- Polykretis, P.; Cencetti, F.; Donati, C.; Luchinat, E.; Banci, L. Cadmium effects on superoxide dismutase 1 in human cells revealed by NMR. Redox Biol. 2019, 21, 101102. [Google Scholar] [CrossRef] [PubMed]
- Nishito, Y.; Kambe, T. Absorption Mechanisms of Iron, Copper, and Zinc: An Overview. J. Nutr. Sci. Vitaminol. 2018, 64, 1–7. [Google Scholar] [CrossRef]
- Kambe, T.; Taylor, K.M.; Fu, D. Zinc transporters and their functional integration in mammalian cells. J. Biol. Chem. 2021, 296, 100320. [Google Scholar] [CrossRef]
- Nishito, Y.; Kambe, T. Zinc transporter 1 (ZNT1) expression on the cell surface is elaborately controlled by cellular zinc levels. J. Biol. Chem. 2019, 294, 15686–15697. [Google Scholar] [CrossRef]
- Huang, L.; Tepaamorndech, S. The SLC30 family of zinc transporters—A review of current understanding of their biological and pathophysiological roles. Mol. Asp. Med. 2013, 34, 548–560. [Google Scholar] [CrossRef]
- Jeong, J.; Eide, D.J. The SLC39 family of zinc transporters. Mol. Asp. Med. 2013, 34, 612–619. [Google Scholar] [CrossRef] [PubMed]
- Lehvy, A.I.; Horev, G.; Golan, Y.; Glaser, F.; Shammai, Y.; Assaraf, Y.G. Alterations in ZnT1 expression and function lead to impaired intracellular zinc homeostasis in cancer. Cell Death Discov. 2019, 5, 144. [Google Scholar] [CrossRef]
- Bafaro, E.; Liu, Y.; Xu, Y.; Dempski, R.E. The emerging role of zinc transporters in cellular homeostasis and cancer. Signal Transduct. Target. Ther. 2017, 2, 17029. [Google Scholar] [CrossRef]
- Pan, Z.; Choi, S.; Ouadid-Ahidouch, H.; Yang, J.-M.; Beattie, J.H.; Korichneva, I. Zinc transporters and dysregulated channels in cancers. Front. Biosci. 2017, 22, 623–643. [Google Scholar] [CrossRef] [PubMed]
- Takatani-Nakase, T. Zinc Transporters and the Progression of Breast Cancers. Biol. Pharm. Bull. 2018, 41, 1517–1522. [Google Scholar] [CrossRef]
- Zhu, B.; Huo, R.; Zhi, Q.; Zhan, M.; Chen, X.; Hua, Z.-C. Increased expression of zinc transporter ZIP4, ZIP11, ZnT1, and ZnT6 predicts poor prognosis in pancreatic cancer. J. Trace Elem. Med. Biol. 2021, 65, 126734. [Google Scholar] [CrossRef]
- Satarug, S.; Garrett, S.; Somji, S.; Sens, M.; Sens, D. Aberrant Expression of ZIP and ZnT Zinc Transporters in UROtsa Cells Transformed to Malignant Cells by Cadmium. Stresses 2021, 1, 78–89. [Google Scholar] [CrossRef]
- Ohana, E.; Sekler, I.; Kaisman, T.; Kahn, N.; Cove, J.; Silverman, W.F.; Amsterdam, A.; Hershfinkel, M. Silencing of ZnT-1 expression enhances heavy metal influx and toxicity. J. Mol. Med. 2006, 84, 753–763. [Google Scholar] [CrossRef] [PubMed]
- Takiguchi, M.; Cherrington, N.J.; Hartley, D.P.; Klaassen, C.D.; Waalkes, M.P. Cyproterone acetate induces a cellular tolerance to cadmium in rat liver epithelial cells involving reduced cadmium accumulation. Toxicology 2001, 165, 13–25. [Google Scholar] [CrossRef]
- Fujishiro, H.; Okugaki, S.; Yasumitsu, S.; Enomoto, S.; Himeno, S. Involvement of DNA hypermethylation in down-regulation of the zinc transporter ZIP8 in cadmium-resistant metallothionein-null cells. Toxicol. Appl. Pharmacol. 2009, 241, 195–201. [Google Scholar] [CrossRef] [PubMed]
- Fujishiro, H.; Ohashi, T.; Takuma, M.; Himeno, S. Suppression of ZIP8 expression is a common feature of cadmium-resistant and manganese-resistant RBL-2H3 cells. Metallomics 2013, 5, 437–444. [Google Scholar] [CrossRef] [PubMed]
- Aiba, I.; Hossain, A.; Kuo, M.T. Elevated GSH Level Increases Cadmium Resistance through Down-Regulation of Sp1-Dependent Expression of the Cadmium Transporter ZIP8. Mol. Pharmacol. 2008, 74, 823–833. [Google Scholar] [CrossRef]
- Vašák, M.; Meloni, G. Chemistry and biology of mammalian metallothioneins. JBIC J. Biol. Inorg. Chem. 2011, 16, 1067–1078. [Google Scholar] [CrossRef]
- Petering, D.H. Reactions of the Zn Proteome with Cd2+ and Other Xenobiotics: Trafficking and Toxicity. Chem. Res. Toxicol. 2016, 30, 189–202. [Google Scholar] [CrossRef]
- Krężel, A.; Maret, W. The Functions of Metamorphic Metallothioneins in Zinc and Copper Metabolism. Int. J. Mol. Sci. 2017, 18, 1237. [Google Scholar] [CrossRef]
- Boonprasert, K.; Satarug, S.; Morais, C.; Gobe, G.C.; Johnson, D.W.; Na-Bangchang, K.; Vesey, D.A. The stress response of human proximal tubule cells to cadmium involves up-regulation of haemoxygenase 1 and metallothionein but not cytochrome P450 enzymes. Toxicol. Lett. 2016, 249, 5–14. [Google Scholar] [CrossRef]
- Boonprasert, K.; Ruengweerayut, R.; Aunpad, R.; Satarug, S.; Na-Bangchang, K. Expression of metallothionein isoforms in peripheral blood leukocytes from Thai population residing in cadmium-contaminated areas. Environ. Toxicol. Pharmacol. 2012, 34, 935–940. [Google Scholar] [CrossRef]
- Hennigar, S.R.; Kelley, A.M.; McClung, J.P. Metallothionein and Zinc Transporter Expression in Circulating Human Blood Cells as Biomarkers of Zinc Status: A Systematic Review. Adv. Nutr. 2016, 7, 735–746. [Google Scholar] [CrossRef]
- Garrett, S.H.; Sens, M.A.; Todd, J.H.; Somji, S.; Sens, D.A. Expression of MT-3 protein in the human kidney. Toxicol. Lett. 1999, 105, 207–214. [Google Scholar] [CrossRef]
- Vašák, M.; Meloni, G. Mammalian Metallothionein-3: New Functional and Structural Insights. Int. J. Mol. Sci. 2017, 18, 1117. [Google Scholar] [CrossRef]
- Sabolić, I.; Skarica, M.; Ljubojevic, M.; Breljak, D.; Herak-Kramberger, C.M.; Crljen, V.; Ljubešić, N. Expression and immunolocalization of metallothioneins MT1, MT2 and MT3 in rat nephron. J. Trace Elem. Med. Biol. 2018, 46, 62–75. [Google Scholar] [CrossRef] [PubMed]
- Adams, S.V.; Barrick, B.; Christopher, E.P.; Shafer, M.M.; Makar, K.W.; Song, X.; Lampe, J.W.; Vilchis, H.; Ulery, A.; Newcomb, P.A. Genetic variation in metallothionein and metal-regulatory transcription factor 1 in relation to urinary cadmium, copper, and zinc. Toxicol. Appl. Pharmacol. 2015, 289, 381–388. [Google Scholar] [CrossRef] [PubMed]
- Sato, M.; Mehra, R.K.; Bremner, I. Measurement of Plasma Metallothionein-I in the Assessment of the Zinc Status of Zinc-Deficient and Stressed Rats. J. Nutr. 1984, 114, 1683–1689. [Google Scholar] [CrossRef] [PubMed]
- Sullivan, V.K.; Burnett, F.R.; Cousins, R.J. Metallothionein Expression Is Increased in Monocytes and Erythrocytes of Young Men during Zinc Supplementation. J. Nutr. 1998, 128, 707–713. [Google Scholar] [CrossRef]
- Takeda, K.; Fujita, H.; Shibahara, S. Differential Control of the Metal-Mediated Activation of the Human Heme Oxygenase-1 and Metallothionein IIa Genes. Biochem. Biophys. Res. Commun. 1995, 207, 160–167. [Google Scholar] [CrossRef]
- Katakai, K.; Liu, J.; Nakajima, K.; Keefer, L.K.; Waalkes, M.P. Nitric oxide induces metallothionein (MT) gene expression apparently by displacing zinc bound to MT. Toxicol. Lett. 2001, 119, 103–108. [Google Scholar] [CrossRef]
- McNeill, R.V.; Mason, A.S.; Hodson, M.E.; Catto, J.W.F.; Southgate, J. Specificity of the metallothionein-1 response by cadmium-exposed normal human urothelial cells. Int. J. Mol. Sci. 2019, 20, 1344. [Google Scholar] [CrossRef]
- Satarug, S.; Garrett, S.; Somji, S.; Sens, M.; Sens, D. Zinc, Zinc Transporters, and Cadmium Cytotoxicity in a Cell Culture Model of Human Urothelium. Toxics 2021, 9, 94. [Google Scholar] [CrossRef]
- Misra, R.R.; Hochadel, J.F.; Smith, G.T.; Cook, J.C.; Waalkes, M.P.; Wink, D.A. Evidence That Nitric Oxide Enhances Cadmium Toxicity by Displacing the Metal from Metallothionein. Chem. Res. Toxicol. 1996, 9, 326–332. [Google Scholar] [CrossRef]
- Satarug, S.; Baker, J.R.; Reilly, P.E.; Esumi, H.; Moore, M.R. Evidence for a Synergistic Interaction between Cadmium and Endotoxin Toxicity and for Nitric Oxide and Cadmium Displacement of Metals in the Kidney. Nitric Oxide 2000, 4, 431–440. [Google Scholar] [CrossRef]
- Satarug, S.; Gobe, G.C.; Vesey, D.A.; Phelps, K.R. Cadmium and Lead Exposure, Nephrotoxicity, and Mortality. Toxics 2020, 8, 86. [Google Scholar] [CrossRef]
- Sabolić, I.; Breljak, D.; Skarica, M.; Herak-Kramberger, C.M. Role of metallothionein in cadmium traffic and toxicity in kidneys and other mammalian organs. Biometals 2010, 23, 897–926. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Vesey, D.A.; Ruangyuttikarn, W.; Nishijo, M.; Gobe, G.C.; Phelps, K.R. The source and pathophysiologic significance of excreted cadmium. Toxics 2019, 7, 55. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Vesey, D.; Nishijo, M.; Ruangyuttikarn, W.; Gobe, G.; Phelps, K. The Effect of Cadmium on GFR Is Clarified by Normalization of Excretion Rates to Creatinine Clearance. Int. J. Mol. Sci. 2021, 22, 1762. [Google Scholar] [CrossRef]
- Knoell, D.L.; Smith, D.; Bao, S.; Sapkota, M.; Wyatt, T.A.; Zweier, J.L.; Flury, J.; Borchers, M.T.; Knutson, M. Imbalance in zinc homeostasis enhances lung Tissue Loss following cigarette smoke exposure. J. Trace Elem. Med. Biol. 2020, 60, 126483. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Baker, J.R.; Reilly, P.E.; Moore, M.R.; Williams, D.J. Changes in zinc and copper homeostasis in human livers and kidneys associated with exposure to environmental cadmium. Hum. Exp. Toxicol. 2001, 20, 205–213. [Google Scholar] [CrossRef] [PubMed]
- Satarug, S.; Nishijo, M.; Ujjin, P.; Moore, M.R. Chronic exposure to low-level cadmium induced zinc-copper dysregulation. J. Trace Elem. Med. Biol. 2018, 46, 32–38. [Google Scholar] [CrossRef]
- Satarug, S.; Gobe, G.C.; Ujjin, P.; Vesey, D.A. Gender differences in zinc and copper excretion in response to co-exposure to low environmental concentrations of cadmium and dead. Stresses 2021, 1, 3–15. [Google Scholar] [CrossRef]
- Eom, S.Y.; Yim, D.H.; Huang, M.; Park, C.H.; Kim, G.B.; Yu, S.D.; Choi, B.S.; Park, J.D.; Kim, Y.D.; Kim, H. Copper-zinc imbalance induces kidney tubule damage and oxidative stress in a population exposed to chronic environmental cadmium. Int. Arch. Occup. Environ. Health 2020, 93, 337–344. [Google Scholar] [CrossRef]
- Pasin, E.; Josephson, D.Y.; Mitra, A.P.; Cote, R.J.; Stein, J.P. Superficial Bladder Cancer: An Update on Etiology, Molecular Development, Classification, and Natural History. Rev. Urol. 2008, 10, 31–43. [Google Scholar]
- Hong, Y.M.; Loughlin, K.R. Economic Impact of Tumor Markers in Bladder Cancer Surveillance. Urology 2008, 71, 131–135. [Google Scholar] [CrossRef]
- Cumberbatch, M.G.; Cox, A.; Teare, D.; Catto, J.W. Contemporary occupational carcinogen exposure and bladder cancer: A systematic review and meta-analysis. JAMA Oncol. 2015, 1, 1282–1290. [Google Scholar] [CrossRef] [PubMed]
- Kwan, M.L.; Garren, B.; Nielsen, M.E.; Tang, L. Lifestyle and nutritional modifiable factors in the prevention and treatment of bladder cancer. Urol. Oncol. Semin. Orig. Investig. 2019, 37, 380–386. [Google Scholar] [CrossRef] [PubMed]
- Dy, G.W.; Gore, J.L.; Forouzanfar, M.H.; Naghavi, M.; Fitzmaurice, C. Global Burden of Urologic Cancers, 1990–2013. Eur. Urol. 2017, 71, 437–446. [Google Scholar] [CrossRef] [PubMed]
- Miyazaki, J.; Nishiyama, H. Epidemiology of urothelial carcinoma. Int. J. Urol. 2017, 24, 730–734. [Google Scholar] [CrossRef]
- Samanic, C.; Kogevinas, M.; Dosemeci, M.; Malats, N.; Real, F.X.; Garcia-Closas, M.; Real, F.X.; Garcia-Closas, M.; Serra, C.; Carrato, A.; et al. Smoking and bladder cancer in Spain: Effects of tobacco type, timing, environmental tobacco smoke, and gender. Cancer Epidemiol. Prev. Biomark. 2006, 15, 1348–1354. [Google Scholar] [CrossRef]
- Sciannameo, V.; Carta, A.; D’Errico, A.; Giraudo, M.T.; Fasanelli, F.; Arici, C.; Maule, M.; Carnà, P.; Destefanis, P.; Rolle, L.; et al. New insights on occupational exposure and bladder cancer risk: A pooled analysis of two Italian case-control studies. Int. Arch. Occup. Environ. Health 2018, 92, 347–359. [Google Scholar] [CrossRef]
- Kellen, E.; Zeegers, M.P.; Hond, E.D.; Buntinx, F. Blood cadmium may be associated with bladder carcinogenesis: The Belgian case-control study on bladder cancer. Cancer Detect. Prev. 2007, 31, 77–82. [Google Scholar] [CrossRef]
- Wolf, C.; Strenziok, R.; Kyriakopoulos, A. Elevated metallothionein-bound cadmium concentrations in urine from bladder carcinoma patients, investigated by size exclusion chromatography-inductively coupled plasma mass spectrometry. Anal. Chim. Acta 2009, 631, 218–222. [Google Scholar] [CrossRef]
- Achanzar, W.E.; Diwan, B.A.; Liu, J.; Quader, S.T.; Webber, M.M.; Waalkes, M.P. Cadmium-induced malignant transformation of human prostate epithelial cells. Cancer Res. 2001, 61, 455–458. [Google Scholar] [PubMed]
- Sens, D.A.; Park, S.; Gurel, V.; Sens, M.A.; Garrett, S.H.; Somji, S. Inorganic cadmium- and arsenite-induced malignant transformation of human bladder urothelial cells. Toxicol. Sci. 2004, 79, 56–63. [Google Scholar] [CrossRef]
- Hoggarth, Z.E.; Osowski, D.B.; Freeberg, B.A.; Garrett, S.H.; Sens, D.A.; Sens, M.A.; Zhou, X.D.; Zhang, K.K.; Somji, S. The urothelial cell line UROtsa transformed by arsenite and cadmium display basal characteristics associated with muscle invasive urothelial cancers. PLoS ONE 2018, 13, e0207877. [Google Scholar] [CrossRef]
- Benbrahim-Tallaa, L.; Tokar, E.J.; Diwan, B.A.; Dill, A.L.; Coppin, J.-F.; Waalkes, M.P. Cadmium Malignantly Transforms Normal Human Breast Epithelial Cells into a Basal-like Phenotype. Environ. Health Perspect. 2009, 117, 1847–1852. [Google Scholar] [CrossRef]
- Soh, M.A.; Garrett, S.H.; Somji, S.; Dunlevy, J.R.; Zhou, X.D.; Sens, M.A.; Bathula, C.S.; Allen, C.; Sens, D.A. Arsenic, cadmium and neuron specific enolase (ENO2, γ-enolase) expression in breast cancer. Cancer Cell Int. 2011, 11, 41. [Google Scholar] [CrossRef] [PubMed]
- Blommel, K.; Knudsen, C.S.; Wegner, K.; Shrestha, S.; Singhal, S.K.; Mehus, A.A.; Garrett, S.H.; Singhal, S.; Zhou, X.; Voels, B.; et al. Meta-analysis of gene expression profiling reveals novel basal gene signatures in MCF-10A cells transformed with cadmium. Oncotarget 2020, 11, 3601–3617. [Google Scholar] [CrossRef]
- Jing, Y.; Liu, L.Z.; Jiang, Y.; Zhu, Y.; Guo, N.L.; Barnett, J.; Rojanasakul, Y.; Agani, F.; Jiang, B.H. Cadmium increases HIF-1 and VEGF expression through ROS, ERK, and AKT signaling pathways and induces malignant transformation of human bronchial epithelial cells. Toxicol. Sci. 2012, 125, 10–19. [Google Scholar] [CrossRef] [PubMed]
- Qu, W.; Tokar, E.J.; Kim, A.J.; Bell, M.W.; Waalkes, M.P. Chronic Cadmium Exposure In Vitro Causes Acquisition of Multiple Tumor Cell Characteristics in Human Pancreatic Epithelial Cells. Environ. Health Perspect. 2012, 120, 1265–1271. [Google Scholar] [CrossRef] [PubMed]
- Takiguchi, M.; Achanzar, W.E.; Qu, W.; Li, G.; Waalkes, M.P. Effects of cadmium on DNA-(Cytosine-5) methyltransferase activity and DNA methylation status during cadmium-induced cellular transformation. Exp. Cell Res. 2003, 286, 355–365. [Google Scholar] [CrossRef]
- Qu, W.; Diwan, B.A.; Reece, J.M.; Bortner, C.D.; Pi, J.; Liu, J.; Waalkes, M.P. Cadmium-induced malignant transformation in rat liver cells: Role of aberrant oncogene expression and minimal role of oxidative stress. Int. J. Cancer 2005, 114, 346–355. [Google Scholar] [CrossRef]
- Garrett, S.H.; Somji, S.; Sens, N.A.; Zhang, K.K. Prediction of the Number of Activated Genes in Multiple Independent Cd+2- and As+3-Induced Malignant Transformations of Human Urothelial Cells (UROtsa). PLoS ONE 2014, 9, e85614. [Google Scholar] [CrossRef] [PubMed]
- Petzoldt, J.L.; Leigh, I.M.; Duffy, P.G.; Sexton, C.; Masters, J.R.W. Immortalisation of human urothelial cells. Urol. Res. 1995, 23, 377–380. [Google Scholar] [CrossRef] [PubMed]
- Rossi, M.R.; Masters, J.R.W.; Park, S.; Todd, J.H.; Garrett, S.H.; Sens, M.A.; Somji, S.; Nath, J.; Sens, D.A. The immortalized UROtsa cell line as a potential cell culture model of human urothelium. Environ. Health Perspect. 2001, 109, 801–808. [Google Scholar] [CrossRef]
- Sens, D.; Rossi, M.; Park, S.; Gurel, V.; Nath, J.; Garrett, S.; Sens, M.A.; Somji, S. Metallothionein isoform 1 and 2 gene expression in a human urothelial cell line (UROtsa) exposed to CdCl2 and NaAsO2. J. Toxicol. Environ. Health A 2003, 66, 2031–2046. [Google Scholar] [CrossRef]
- Sens, M.A.; Somji, S.; Lamm, D.L.; Garrett, S.H.; Slovinsky, F.; Todd, J.H.; Sens, D.A. Metallothionein isoform 3 as a potential biomarker for human bladder cancer. Environ. Health Perspect. 2000, 108, 413–418. [Google Scholar] [CrossRef] [PubMed]
- Zhou, X.D.; Sens, M.A.; Garrett, S.H.; Somji, S.; Park, S.; Gurel, V.; Sens, D.A. Enhanced expression of metallothionein isoform 3 protein in tumor heterotransplants derived from As+3- and Cd+2-transformed human urothelial cells. Toxicol. Sci. 2006, 93, 322–330. [Google Scholar] [CrossRef]
- Somji, S.; Garrett, S.H.; Toni, C.; Zhou, X.D.; Zheng, Y.; Ajjimaporn, A.; Sens, M.A.; Sens, D.A. Differences in the epigenetic regulation of MT-3 gene expression between parental and Cd+2 or As+3 transformed human urothelial cells. Cancer Cell Int. 2011, 11, 2. [Google Scholar] [CrossRef] [PubMed]
- Ajjimaporn, A.; Botsford, T.; Garrett, S.H.; Sens, M.A.; Zhou, X.D.; Dunlevy, J.R.; Sens, D.A.; Somji, S. ZIP8 expression in human proximal tubule cells, human urothelial cells transformed by Cd+2 and As+3 and in specimens of normal human urothelium and urothelial cancer. Cancer Cell Int. 2012, 12, 16. [Google Scholar] [CrossRef]
- Audenet, F.; Attalla, K.; Sfakianos, J.P. The evolution of bladder cancer genomics: What have we learned and how can we use it? Urol. Oncol. Semin. Orig. Investig. 2018, 36, 313–320. [Google Scholar] [CrossRef]
- Kagara, N.; Tanaka, N.; Noguchi, S.; Hirano, T. Zinc and its transporter ZIP10 are involved in invasive behavior of breast cancer cells. Cancer Sci. 2007, 98, 692–697. [Google Scholar] [CrossRef]
- Mathews, W.R.; Wang, F.; Eide, D.J.; Van Doren, M. Drosophila fear of intimacy Encodes a Zrt/IRT-like Protein (ZIP) Family Zinc Transporter Functionally Related to Mammalian ZIP Proteins. J. Biol. Chem. 2005, 280, 787–795. [Google Scholar] [CrossRef] [PubMed]
- Taylor, K.M.; Muraina, I.A.; Brethour, D.; Schmitt-Ulms, G.; Nimmanon, T.; Ziliotto, S.; Kille, P.; Hogstrand, C. Zinc Transporter ZIP10 forms a heteromer with ZIP6 which regulates embryonic development and cell migration. Biochem. J. 2016, 473, 2531–2544. [Google Scholar] [CrossRef]
- Golovine, K.; Makhov, P.; Uzzo, R.G.; Shaw, T.; Kunkle, D.; Kolenko, V.M. Overexpression of the zinc uptake transporter hZIP1 inhibits nuclear factor-κB and reduces the malignant potential of prostate cancer cells in vitro and in vivo. Clin. Cancer Res. 2008, 14, 5376–5384. [Google Scholar] [CrossRef]
- Zhao, L.; Chen, W.; Taylor, K.M.; Cai, B.; Li, X. LIV-1 suppression inhibits HeLa cell invasion by targeting ERK1/2-Snail/Slug pathway. Biochem. Biophys. Res. Commun. 2007, 363, 82–88. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Liu, T.; Jin, H.; Yin, L.; Yu, H.; Bi, J. MiR-411 suppresses the development of bladder cancer by regulating ZnT1. OncoTargets Ther. 2018, 11, 8695–8704. [Google Scholar] [CrossRef]
- Chen, Q.Y.; Desmarais, T.; Costa, M. Metals and Mechanisms of Carcinogenesis. Annu. Rev. Pharmacol. Toxicol. 2019, 59, 537–554. [Google Scholar] [CrossRef]
- Xu, Y.-M.; Gao, Y.-M.; Wu, D.-D.; Yu, F.-Y.; Zang, Z.-S.; Yang, L.; Yao, Y.; Cai, N.-L.; Zhou, Y.; Chiu, J.-F.; et al. Aberrant cytokine secretion and zinc uptake in chronic cadmium-exposed lung epithelial cells. Proteom. Clin. Appl. 2017, 11, 1600059. [Google Scholar] [CrossRef] [PubMed]
- Person, R.J.; Tokar, E.J.; Xu, Y.; Orihuela, R.; Ngalame, N.N.O.; Waalkes, M.P. Chronic cadmium exposure in vitro induces cancer cell characteristics in human lung cells. Toxicol. Appl. Pharmacol. 2013, 273, 281–288. [Google Scholar] [CrossRef] [PubMed]
| Zinc Transporters | Number of Transcripts in 1000 β-Actin | ||||
|---|---|---|---|---|---|
| Batch I, 0 µM Cd2+ | Batch II, 0 µM Cd2+ | 1 µM Cd2+ | 2 µM Cd2+ | 4 µM Cd2+ | |
| SLC30A family | |||||
| ZnT1 | 181 ± 23 | 365 ± 38 | 3007 ± 465 | 1434 ± 146 | 1216 ± 153 *** |
| ZnT2 | 0.01 ± 0.001 | 0.06 ± 0.01 | 73 ± 15 | 16 ± 1.9 | 11 ± 1.5 *** |
| ZnT3 | 0.03 ± 0.007 | 0.15 ± 0.01 | 0.24 ± 0.05 | 0.10 ± 0.02 | 0.10 ± 0.02 * |
| ZnT4 | 1.6 ± 0.26 | 11.4 ± 0.8 | 10 ± 1 | 8.7 ± 1 | 6.2 ± 0.5 ** |
| ZnT5 | 150 ± 19 | 510 ± 30 | 1038 ± 132 | 495 ± 54 | 568 ± 91 ** |
| ZnT6 | 4.5 ± 0.15 | 65 ± 8 | 77 ± 6 | 63 ± 13 | 57 ± 12 |
| ZnT7 | 734 ± 28 | 758 ± 76 | 1007 ± 136 | 706 ± 44 | 488 ± 63 * |
| ZnT10 | 0.04 ± 0.005 | 1.1 ± 0.2 | 2.4 ± 0.2 | 1.7 ± 0.2 | 1.1 ± 0.1 *** |
| SLC39A family | |||||
| ZIP1 | 19.5 ± 2.0 | 82 ± 9 | 99 ± 15 | 55 ± 10 | 59 ± 12 * |
| ZIP2 | 0.02 ± 0.004 | 1.2 ± 0.1 | 0.8 ± 0.2 | 0.4 ± 0.1 | 0.2 ± 0.03 *** |
| ZIP3A | 9.1 ± 0.4 | 19 ± 1 | 23 ± 2.3 | 17 ± 1.7 | 14 ± 1.3 * |
| ZIP3B | 0.48 ± 0.05 | 4.1 ± 0.2 | 6.2 ± 0.7 | 4.4 ± 0.2 | 4.2 ± 0.4 * |
| ZIP4 | 0.18 ± 0.04 | 0.06 ± 0.01 | 0.06 ± 0.01 | 0.04 ± 0.01 | 0.06 ± 0.01 |
| ZIP5 | 0.01 ± 0.003 | 0.01 ± 0.001 | 0.01 ± 0.003 | 0.01 ± 0.002 | 0.01 ± 0.002 |
| ZIP6 | 18.3 ± 2.6 | 92 ± 8 | 133 ± 12 | 80 ± 9 | 75 ± 10 ** |
| ZIP7 | 121 ± 9.5 | 204 ± 25 | 342 ± 69 | 149 ± 32 | 94 ± 21 *** |
| ZIP8 | 0.09 ± 0.01 | 2.1 ± 0.2 | 2.6 ± 0.3 | 2.0 ± 0.2 | 2.7 ± 0.4 |
| ZIP10 | 5 ± 0.3 | 54 ± 4 | 30 ± 8 | 14 ± 3 | 14 ± 3 *** |
| ZIP14 | 83.4 ± 10.5 | 146 ± 19 | 218 ± 24 | 158 ± 26 | 128 ± 19 * |
| Gene Expression/Phenotypes | Cadmium-Transformed UROtsa Clones | ||||||
|---|---|---|---|---|---|---|---|
| UTCd1 | UTCd2 | UTCd3 | UTCd4 | UTCd5 | UTCd6 | UTCd7 | |
| SLC30A gene family | |||||||
| ZnT1 | ↑ | ↓ | ↓ | ↓ | ↓ | ↓ | ↓ |
| ZnT2 | ↑ | ↓ | ↓ | ↔ | ↑ | ↑ | ↓ |
| ZnT3 | ↑ | ↓ | ↓ | ↓ | ↓ | ↓ | ↓ |
| ZnT4 | ↑ | ↓ | ↓ | ↔ | ↑ | ↓ | ↓ |
| ZnT5 | ↔ | ↓ | ↓ | ↔ | ↔ | ↔ | ↓ |
| ZnT6 | ↑ | ↓ | ↓ | ↓ | ↓ | ↔ | ↓ |
| ZnT7 | ↔ | ↓ | ↓ | ↓ | ↔ | ↓ | ↓ |
| ZnT10 | ↑ | ↔ | ↔ | ↔ | ↑ | ↑ | ↑ |
| SLC39A gene family | |||||||
| ZIP1 | ↓ | ↓ | ↓ | ↔ | ↔ | ↔ | ↓ |
| ZIP2 | ↓ | ↓ | ↓ | ↓ | ↓ | ↓ | ↓ |
| ZIP3A | ↔ | ↓ | ↓ | ↓ | ↔ | ↔ | ↓ |
| ZIP3B | ↔ | ↔ | ↓ | ↔ | ↔ | ↔ | ↓ |
| ZIP4 | ↓ | ↓ | ↓ | ↓ | ↓ | ↓ | ↓ |
| ZIP5 | ↓ | ↓ | ↓ | ↓ | ↔ | ↓ | ↔ |
| ZIP6 | ↑ | ↔ | ↓ | ↔ | ↔ | ↑ | ↓ |
| ZIP7 | ↔ | ↔ | ↓ | ↔ | ↔ | ↔ | ↓ |
| ZIP8 | ↓ | ↑ | ↑ | ↑ | ↑ | ↑ | ↑ |
| ZIP10 | ↑ | ↔ | ↓ | ↔ | ↑ | ↑ | ↓ |
| ZIP14 | ↑ | ↓ | ↓ | ↔ | ↔ | ↔ | ↓ |
| Phenotypes | |||||||
| Doubling time (h) | 27.8 | 18.8 * | 16.4 * | 20.7 * | 18.2 * | 16.9 * | 27.1 |
| § Heterotransplant (sc) | 5/5 | 2/5 | 4/5 | 2/5 | 3/5 | 2/5 | 4/5 |
| Histology | TCC | TCC | TCC | TCC | TCC | TCC | TCC |
| § Heterotransplant (ip) | + | − | − | − | − | − | − |
| Histology | TCC | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Satarug, S.; Vesey, D.A.; Gobe, G.C. The Evolving Role for Zinc and Zinc Transporters in Cadmium Tolerance and Urothelial Cancer. Stresses 2021, 1, 105-118. https://doi.org/10.3390/stresses1020009
Satarug S, Vesey DA, Gobe GC. The Evolving Role for Zinc and Zinc Transporters in Cadmium Tolerance and Urothelial Cancer. Stresses. 2021; 1(2):105-118. https://doi.org/10.3390/stresses1020009
Chicago/Turabian StyleSatarug, Soisungwan, David A. Vesey, and Glenda C. Gobe. 2021. "The Evolving Role for Zinc and Zinc Transporters in Cadmium Tolerance and Urothelial Cancer" Stresses 1, no. 2: 105-118. https://doi.org/10.3390/stresses1020009
APA StyleSatarug, S., Vesey, D. A., & Gobe, G. C. (2021). The Evolving Role for Zinc and Zinc Transporters in Cadmium Tolerance and Urothelial Cancer. Stresses, 1(2), 105-118. https://doi.org/10.3390/stresses1020009








