Molecular Survey of Piroplasmids and Hemosporidians in Vampire Bats, with Evidence of Distinct Piroplasmida Lineages Parasitizing Desmodus rotundus from the Brazilian Amazon
Abstract
:1. Introduction
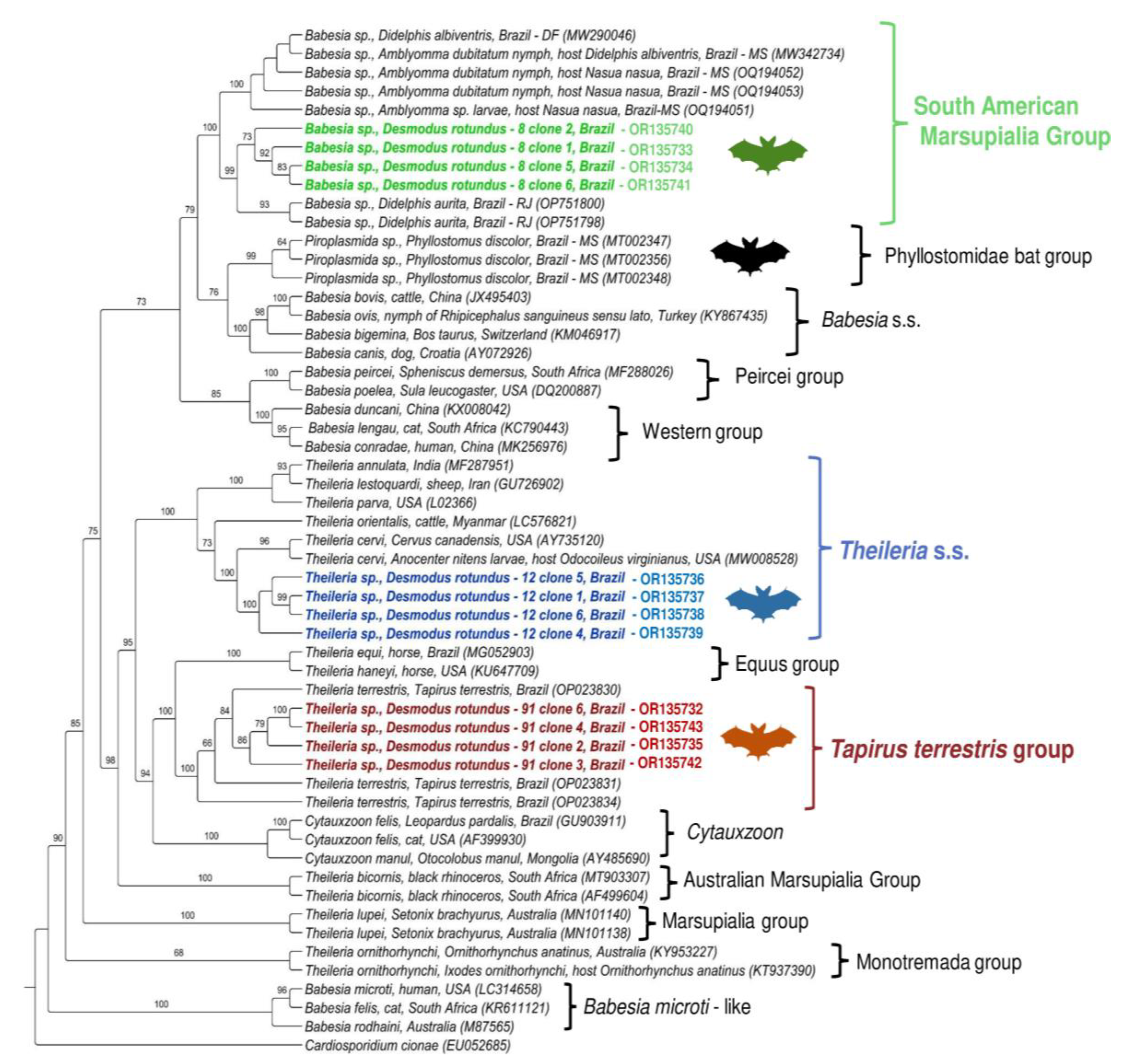
2. Results
3. Discussion
4. Materials and Methods
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Reis, N.R.; Peracchi, A.L.; Pedro, W.A.; Lima, I.P. Morcegos do Brasil; NR Reis: Londrina, Brazil, 2007. [Google Scholar]
- Laurindo, R.S.; Novaes, R.L.M. Desmistificando os Morcegos. Monte Belo: Núcleo de Pesquisas Integradas—NUPEI/ISMECN/Instituto Sul Mineiro de Estudos e Conservação da Natureza. 2015. Available online: https://www.researchgate.net/publication/303837222_Desmitificando_os_morcegos (accessed on 20 April 2023).
- Stoner-Duncan, B.; Streicker, D.G.; Tedeschi, C.M. Vampire Bats and Rabies: Toward an Ecological Solution to a Public Health Problem. PLoS Negl. Trop Dis. 2014, 8, e2867. [Google Scholar] [CrossRef]
- Zepeda Mendoza, M.L.; Xiong, Z.; Escalera-Zamudio, M.; Runge, A.K.; Thézé, J.; Streicker, D.; Frank, H.K.; Loza-Rubio, E.; Liu, S.; Ryder, O.A.; et al. Hologenomic adaptations underlying the evolution of sanguivory in the common vampire bat. Nat. Ecol. Evol. 2018, 2, 659–668. [Google Scholar] [CrossRef] [Green Version]
- Muhldorfer, K. Bats and bacterial pathogens: A review. Zoonoses Public Health 2013, 60, 93–103. [Google Scholar] [CrossRef]
- Jalovecka, M.; Hajdusek, O.; Sojka, D.; Kopacek, P.; Malandrin, L. The complexity of piroplasms life cycles. Front. Cell. Infect. 2018, 248, 8. [Google Scholar] [CrossRef] [Green Version]
- Jalovecka, M.; Sojka, D.; Ascencio, M.; Schnittger, L. Babesia life cycle–when phylogeny meets biology. Trends Parasitol. 2019, 13, 356–368. [Google Scholar] [CrossRef]
- Schnittger, L.; Ganzinelli, S.; Bhoora, R. The Piroplasmida Babesia, Cytauxzoon, and Theileria in farm and companion animals: Species compilation, molecular phylogeny, and evolutionary insights. Parasitol. Res. 2022, 121, 1207–1245. [Google Scholar] [CrossRef]
- Alvarado-Rybak, M.; Gallego, L.S.; Millán, J. A review of piroplasmid infections in wild carnivores worldwide: Importance for domestic animal health and wildlife conservation. Parasit. Vectors 2016, 9, 538. [Google Scholar] [CrossRef] [Green Version]
- Yabsley, M.J.; Vanstreels, R.E.T.; Shock, B.C.; Purdee, M.; Horne, E.C.; Peirce, M.A.; Parsons, N.J. Molecular characterization of Babesia peircei and Babesia ugwidiensis provides insight into the evolution and host specificity of avian piroplasmids. Int. J. Parasitol. Parasites Wildl. 2017, 6, 257–264. [Google Scholar] [CrossRef]
- Dionisi, A. La malaria di alcuni specie di pipistrelli. Atti Della Soc. Per Glistudi Malar. 1899, 1, 133–173. [Google Scholar]
- Marinkelle, C.J. Babesia sp. in Colombian bats (Microchiroptera). J. Wildl. Dis. 1996, 32, 534–535. [Google Scholar] [CrossRef]
- Concannon, R.; Wynn-Owen, K.; Simpson, V.R.; Birtles, R.J. Molecular characterization of haemoparasites infecting bats (Microchiroptera) in Cornwall, UK. Parasitol 2005, 131, 489–496. [Google Scholar] [CrossRef]
- Hornok, S.; Estók, P.; Kováts, D.; Flaisz, B.; Takács, N.; Szőke, K.; Krawczyk, A.; Kontschán, J.; Gyuranecz, M.; Fedák, A.; et al. Screening of bat faeces for arthropod-borne apicomplexan protozoa: Babesia canis and Besnoitia besnoiti-like sequences from Chiroptera. Parasit. Vectors 2015, 28, 441. [Google Scholar] [CrossRef] [Green Version]
- Hornok, S.; Szőke, K.; Kováts, D.; Estók, P.; Görföl, T.; Boldogh, S.A.; Takács, N.; Kontschán, J.; Földvári, G.; Barti, L.; et al. DNA of Piroplasms of Ruminants and Dogs in Ixodid Bat Ticks. PLoS ONE 2016, 11, e0167735. [Google Scholar] [CrossRef] [Green Version]
- Hornok, S.; Kovács, R.; Meli, M.L.; Gönczi, E.; Hofmann-Lehmann, R.; Kontschán, J.; Gyuranecz, M.; Dán, A.; Molnár, V. First detection of bartonellae in a broad range of bat ectoparasites. Vet. Microbiol. 2012, 159, 541–543. [Google Scholar] [CrossRef]
- Corduneanu, A.; Hrazdilová, K.; Sándor, A.D.; Matei, I.A.; Ionică, A.M.; Barti, L.; Ciocănău, M.A.; Măntoiu, D.Ș.; Coroiu, I.; Hornok, S.; et al. Babesia vesperuginis, a neglected piroplasmid: New host and geographical records, and phylogenetic relations. Parasit. Vectors 2017, 10, 598. [Google Scholar] [CrossRef] [Green Version]
- Han, H.J.; Liu, J.W.; Wen, H.L.; Qin, X.R.; Zhao, M.; Wang, L.J.; Zhou, C.M.; Qi, R.; Yu, H.; Yu, X.J. Babesia vesperuginis in insectivorous bats from China. Parasit. Vectors 2018, 11, 317. [Google Scholar] [CrossRef] [Green Version]
- Liu, X.; Yan, B.; Wang, Q.; Jiang, M.; Tu, C.; Chen, C.; Hornok, S.; Wang, Y. Babesia vesperuginis in Common Pipistrelle (Pipistrellus pipistrellus) and the Bat Soft Tick Argas vespertilionis in the People’s Republic of China. J. Wildl. Dis. 2018, 54, 419–421. [Google Scholar] [CrossRef] [Green Version]
- Lv, J.; Fernández de Marco, M.; Goharriz, H.; Phipps, L.P.; McElhinney, L.M.; Hernández-Triana, L.M.; Wu, S.; Lin, X.; Fooks, A.R.; Johnson, N. Detection of tick-borne bacteria and babesia with zoonotic potential in Argas (Carios) vespertilionis (Latreille, 1802) ticks from British bats. Sci. Rep. 2018, 8, 1865. [Google Scholar] [CrossRef] [Green Version]
- Ranaivoson, H.C.; Héraud, J.M.; Goethert, H.K.; Telford, S.R.; Rabetafika, L.; Brook, C.E. Babesial infection in the Madagascan flying fox, Pteropus rufus É Geoffroy, 1803. Parasit. Vectors 2019, 12, 51. [Google Scholar] [CrossRef] [Green Version]
- Ikeda, P.; Menezes, T.R.; Torres, J.M.; de Oliveira, C.E.; Lourenço, E.C.; Herrera, H.M.; Machado, R.Z.; André, M.R. First molecular detection of piroplasmids in non-hematophagous bats from Brazil, with evidence of putative novel species. Parasitol. Res. 2021, 120, 301–310. [Google Scholar] [CrossRef]
- Linhart, P.; Bandouchova, H.; Zukal, J.; Votýpka, J.; Baláž, V.; Heger, T.; Kalocsanyiova, V.; Kubickova, A.; Nemcova, M.; Sedlackova, J.; et al. Blood Parasites and Health Status of Hibernating and Non-Hibernating Noctule Bats (Nyctalus noctula). Microorganisms 2022, 10, 1028. [Google Scholar] [CrossRef]
- O’Donoghue, P. Haemoprotozoa: Making biological sense of molecular phylogenies. Int. J. Parasitol. Parasites Wildl. 2017, 6, 241–256. [Google Scholar] [CrossRef]
- Lee, J.J.; Leedale, G.F.; Bradbury, P. The Illustrated Guide to the Protozoa, 2nd ed.; vols. I and II. Society of Protozoologists; Allen Press: Lawrence, KS, USA, 2000; 1432p. [Google Scholar]
- Tsague, K.J.A.; Bakwo Fils, E.M.; Atagana, J.P.; Dongue, N.V.; Mbeng, D.W.; Schaer, J.; Tchuinkam, T. Hepatocystis and Nycteria (Haemosporida) parasite infections of bats in the Central Region of Cameroon. Parasitology 2022, 149, 51–58. [Google Scholar] [CrossRef]
- Balasubramaniam, S.; Mulder, R.A.; Sunnucks, P.; Pavlova, A.; Amos, J.N.; Melville, J. Prevalence and diversity of avian haematozoa in three species of Australian passerine. Emu 2013, 113, 353–358. [Google Scholar] [CrossRef]
- Schaer, J.; Perkins, S.L.; Decher, J.; Leendertz, F.H.; Fahr, J.; Weber, N.; Matuschewski, K. High diversity of West African bat malaria parasites and a tight link with rodent Plasmodium taxa. Proc. Natl. Acad. Sci. USA 2013, 110, 17415–17419. [Google Scholar] [CrossRef]
- Schaer, J.; Reeder, D.M.; Vodzak, M.E.; Olival, K.J.; Weber, N.; Mayer, F.; Matuschewski, K.; Perkins, S.L. Nycteria parasites of Afrotropical insectivorous bats. Int. J. Parasitol. 2015, 45, 375–384. [Google Scholar] [CrossRef] [Green Version]
- Duval, L.; Robert, V.; Csorba, G.; Hassanin, A.; Randrianarivelojosia, M.; Walston, J.; Nhim, T.; Goodman, S.M.; Ariey, F. Multiple host-switching of Haemosporidia parasites in bats. Malar. J. 2007, 6, 157. [Google Scholar] [CrossRef] [Green Version]
- Adam, J.P.; Landau, I. Developmental stages of Polychromophilus sp., a parasite of insectivorous bats from the Congo-Brazzaville, in the nycteribiid fly Penicillidia fulvida Bigot. Trans. R. Soc. Trop. Med. Hyg. 1973, 67, 5–6. [Google Scholar] [CrossRef]
- Landau, I.; Chavatte, J.M.; Karadjian, G.; Chabaud, A.; Beveridge, I. The haemosporidian parasites of bats, with description of Sprattiella alecto gen. nov., sp. nov. Parasite 2012, 19, 137–146. [Google Scholar] [CrossRef] [Green Version]
- Megali, A.; Yannic, G.; Christe, P. Disease in the dark: Molecular characterization of Polychromophilus murinus in temperate zone bats revealed a worldwide distribution of this malaria-like disease. Mol. Ecol. 2011, 20, 1039–1048. [Google Scholar] [CrossRef]
- Landau, I.; Chabaud, A.G. Description de Plasmodium cyclopsi n. sp. parasite du microchiroptère Hipposideros cyclops à Makokou (Gabon). APHCA 1978, 53, 247–253. [Google Scholar] [CrossRef] [Green Version]
- Ramasindrazana, B.; Goodman, S.M.; Dsouli, N.; Gomard, Y.; Lagadec, E.; Randrianarivelojosia, M.; Dellagi, K.; Tortosa, P. Polychromophilus spp. (Haemosporida) in Malagasy bats: Host specificity and insights on invertebrate vectors. Malar. J. 2018, 17, 318. [Google Scholar] [CrossRef] [Green Version]
- Witsenburg, F.; Salamin, N.; Christe, P. The evolutionary host switches of Polychromophilus: A multi-gene phylogeny of the bat malaria genus suggests a second invasion of mammals by a haemosporidian parasite. Malar. J. 2012, 11, 53. [Google Scholar] [CrossRef]
- Minozzo, G.A.; da Silva Mathias, B.; Riediger, I.N.; de Oliveira Guimarães, L.; Dos Anjos, C.C.; Monteiro, E.F.; Dos Santos, A.P.; Biondo, A.W.; Kirchgatter, K. First molecular detection of Polychromophilus parasites in Brazilian Bat Species. Microorganisms 2021, 9, 1240. [Google Scholar] [CrossRef]
- Mathias, B.d.S.; Minozzo, G.A.; Biondo, A.W.; Costa, J.d.O.J.; Soares, H.S.; Marcili, A.; Guimarães, L.d.O.; dos Anjos, C.C.; Santos, A.P.D.; Riediger, I.N.; et al. Molecular investigation confirms Myotis Genus Bats as Common Hosts of Polychromophilus in Brazil. Microorganisms 2023, 11, 1531. [Google Scholar] [CrossRef]
- de Mello, V.V.C.; Placa, A.J.V.; Lee, D.A.B.; Franco, E.O.; Lima, L.; Teixeira, M.M.G.; Hemsley, C.; Titball, R.W.; Machado, R.Z.; André, M.R. Molecular detection of blood-borne agents in vampire bats from Brazil, with the first molecular evidence of Neorickettsia sp. in Desmodus rotundus and Diphylla ecaudata. Acta Trop. 2023, 244, 106945. [Google Scholar] [CrossRef]
- Trifinopoulos, J.; Nguyen, L.T.; von Haeseler, A.; Minh, B.Q. W-IQ-TREE: A fast online phylogenetic tool for maximum likelihood analysis. Nucleic Acids Res. 2016, 44, 232–235. [Google Scholar] [CrossRef] [Green Version]
- Mongruel, A.C.B.; Medici, E.P.; Canena, A.D.C.; Calchi, A.C.; Machado, R.Z.; André, M.R. Expanding the Universe of Hemoplasmas: Multi-Locus Sequencing Reveals Putative Novel Hemoplasmas in Lowland Tapirs (Tapirus terrestris), the Largest Land Mammals in Brazil. Microorganisms 2022, 10, 614. [Google Scholar] [CrossRef]
- Oliveira, Á.F.X.; Calchi, A.C.; Stocco, A.V.; Stocco, N.V.; Costa, A.C.; Mureb, E.N.; Pires, J.R.; Guimarães, A.; Raimundo, J.M.; de Almeida Balthazar, D.; et al. Expanding the universe of Piroplasmids: Morphological detection and phylogenetic positioning of putative novel piroplasmids in black-eared opossums (Didelphis aurita) from southeastern Brazil, with description of “South American Marsupialia Group” of Piroplasmida. Parasitol. Res. 2023, 122, 1519–1530. [Google Scholar] [CrossRef]
- Hornok, S.; Szőke, K.; Görföl, T.; Földvári, G.; Tu, V.T.; Takács, N.; Kontschán, J.; Sándor, A.D.; Estók, P.; Epis, S.; et al. Molecular investigations of the bat tick Argas vespertilionis (Ixodida: Argasidae) and Babesia vesperuginis (Apicomplexa: Piroplasmida) reflect “bat connection” between Central Europe and Central Asia. Exp. Appl. Acarol. 2017, 72, 69–77. [Google Scholar] [CrossRef]
- Gonçalves, L.R.; Paludo, G.; Bisol, T.B.; Perles, L.; de Oliveira, L.B.; de Oliveira, C.M.; da Silva, T.M.V.; Nantes, W.A.G.; Duarte, M.A.; Santos, F.M.; et al. Molecular detection of piroplasmids in synanthropic rodents, marsupials, and associated ticks from Brazil, with phylogenetic inference of a putative novel Babesia sp. from white-eared opossum (Didelphis albiventris). Parasitol. Res. 2021, 120, 3537–3546. [Google Scholar] [CrossRef]
- Hornok, S.; Szőke, K.; Estók, P.; Krawczyk, A.; Haarsma, A.J.; Kováts, D.; Boldogh, S.A.; Morandini, P.; Szekeres, S.; Takács, N.; et al. Assessing bat droppings and predatory bird pellets for vector-borne bacteria: Molecular evidence of bat-associated Neorickettsia sp. in Europe. Antonie Van Leeuwenhoek 2018, 111, 1707–1717. [Google Scholar] [CrossRef]
- Schnittger, L.; Rodriguez, A.E.; Florin-Christensen, M.; Morrison, D.A. Babesia: A world emerging. Infect. Genet. Evol. 2012, 12, 1788–1809. [Google Scholar] [CrossRef]
- Kerth, G. Causes and Consequences of Sociality in Bats. BioScience 2008, 58, 737–746. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Li, Y.; Learn, G.H.; Rudicell, R.S.; Robertson, J.D.; Keele, B.F.; Ndjango, J.B.; Sanz, C.M.; Morgan, D.B.; Locatelli, S.; et al. Origin of the human malaria parasite Plasmodium falciparum in gorillas. Nature 2010, 467, 420–425. [Google Scholar] [CrossRef] [Green Version]
- Kamani, J.; Atuman, Y.J.; Oche, D.A.; Shekaro, A.; Werb, O.; Ejotre, I.; Schaer, J. Molecular detection of Trypanosoma spp. and Hepatocystis parasite infections of bats in Northern Nigeria. Parasitology 2022, 149, 1460–1467. [Google Scholar] [CrossRef]
- Rasoanoro, M.; Goodman, S.M.; Randrianarivelojosia, M.; Rakotondratsimba, M.; Dellagi, K.; Tortosa, P.; Ramasindrazana, B. Diversity, distribution, and drivers of Polychromophilus infection in Malagasy bats. Malar. J. 2021, 20, 157. [Google Scholar] [CrossRef]
- Rosyadi, I.; Shimoda, H.; Takano, A.; Yanagida, T.; Sato, H. Isolation and molecular characterization of Polychromophilus spp. (Haemosporida: Plasmodiidae) from the Asian long-fingered bat (Miniopterus fuliginosus) and Japanese large-footed bat (Myotis macrodactylus) in Japan. Parasitol. Res. 2022, 121, 2547–2559. [Google Scholar] [CrossRef]
- Birkenheuer, A.J.; Levy, M.G.; Breitschwerdt, E.B. Development and evaluation of a seminested PCR for detection and differentiation of Babesia gibsoni (Asian genotype) and B. canis DNA in canine blood samples. J. Clin. Microbiol. 2003, 41, 4172–4177. [Google Scholar] [CrossRef] [Green Version]
- Jefferies, R.; Ryan, U.M.; Irwin, P.J. PCR-RFLP for the detection and differentiation of the canine piroplasm species and its use with filter paper-based technologies. Vet. Parasitol. 2007, 144, 20–27. [Google Scholar] [CrossRef]
- Merino, S.; Martínez, J.; Martínez-de la Puente, J.; Criado-Fornelio, Á.; Tomás, G.; Morales, J.; Lobato, E.; García-Fraile, S. Molecular characterization of the 18S rDNA gene of an avian Hepatozoon reveals that it is closely related to Lankesterella. J. Parasitol. 2006, 92, 1330–1335. [Google Scholar] [CrossRef]
- Quillfeldt, P.; Martinez, J.; Bugoni, L.; Mancini, P.L.; Merino, S. Blood parasites in noddies and boobies from Brazilian offshore islands–differences between species and influence of nesting habitat. Parasitology 2014, 141, 399–410. [Google Scholar] [CrossRef] [Green Version]
- Greay, T.L.; Zahedi, A.; Krige, A.; Owens, J.M.; Rees, R.L.; Ryan, U.M.; Oskam, C.L.; Irwin, P.J. Endemic, exotic and novel apicomplexan parasites detected during a national study of ticks from companion animals in Australia. Parasites Vectors 2018, 11, 197. [Google Scholar] [CrossRef] [Green Version]
- Barbosa, A.D.; Austen, J.; Portas, T.J.; Amigo, J.Á.; Ahlstrom, L.A.; Oskam, C.L.; Irwin, P.J. Sequence analyses at mitochondrial and nuclear loci reveal a novel Theileria sp. and AID in the phylogenetic resolution of piroplasms from Australian marsupials and ticks. PLoS ONE 2019, 12, e0225822. [Google Scholar] [CrossRef] [Green Version]
- Shock, B.C.; Birkenheuer, A.J.; Patton, L.L.; Olfenbuttel, C.; Beringer, J.; Grove, D.M.; Peek, M.; Butfiloski, J.W.; Hughes, D.W.; Lockhart, J.M.; et al. Variation in the ITS-1 and ITS-2 rRNA genomic regions of Cytauxzoon felis from bobcats and pumas in the eastern United States and comparison with sequences from domestic cats. Vet. Parasitol. 2012, 190, 29–35. [Google Scholar] [CrossRef]
- Soares, J.F.; Girotto, A.; Brandão, P.E.; Da Silva, A.S.; França, R.T.; Lopes, S.T.A.; Labruna, M.B. Detection and molecular characterization of a canine piroplasm from Brazil. Vet. Parasitol. 2011, 180, 203–208. [Google Scholar] [CrossRef]
- Furuta, P.I.; Oliveira, T.M.; Theixeira, M.C.; Rocha, A.G.; Machado, R.Z.; Tinucci-Costa, M.G. Comparison between a soluble antigen-based ELISA and IFAT in detecting antibodies against Babesia canis in dogs. Rev. Bras. Parasitol. Vet. 2009, 18, 41–45. [Google Scholar] [CrossRef]
- Werther, K.; Luzzi, M.C.; Gonçalves, L.R.; de Oliveira, J.P.; Alves Junior, J.R.F.; Machado, R.Z.; André, M.R. Arthropod-borne agents in wild Orinoco geese (Neochen jubata) in Brazil. Comp. Immunol. Microbiol. Infect. Dis. 2017, 55, 5530–5541. [Google Scholar] [CrossRef] [Green Version]
- Kohn, M.A.; Senyak, J. Sample Size Calculators. UCSF CTSI. 20 December 2021. Available online: https://www.sample-size.net/ (accessed on 1 August 2023).
- Sanger, F.; Nicklen, S.; Coulson, A.R. DNA sequencing with chain-terminating inhibitors. Proc. Natl. Acad. Sci. USA 1977, 74, 5463–5467. [Google Scholar] [CrossRef]
- Lau, A.O.; Cereceres, K.; Palmer, G.H.; Fretwell, D.L.; Pedroni, M.J.; Mosqueda, J.; McElwain, T.F. Genotypic diversity of merozoite surface antigen 1 of Babesia bovis within an endemic population. Mol. Biochem. Parasitol. 2010, 172, 107–112. [Google Scholar] [CrossRef]
- Ewing, B.; Green, P. Base-calling of automated sequencer traces using phred. II. Error probabilities. Genome. Res. 1998, 8, 186–194. [Google Scholar] [CrossRef] [Green Version]
- Ewing, B.; Hillier, L.; Wendl, M.C.; Green, P. Base-calling of automated sequencer traces using phred. I. Accuracy assessment. Genome. Res. 1998, 8, 175–185. [Google Scholar] [CrossRef] [Green Version]
- Altschul, S.F.; Gish, W.; Miller, W.; Myers, E.W.; Lipman, D.J. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- GenBank Overview. Available online: https://www.ncbi.nlm.nih.gov/genbank/ (accessed on 10 June 2023).
- Katoh, K.; Toh, H. PartTree: An algorithm to build an approximate tree from a large number of unaligned sequences. Bioinformatics 2007, 23, 372–374. [Google Scholar] [CrossRef] [Green Version]
- Hall, T.A. BioEdit: A User-Friendly Biological Sequence Alignment Editor and Analysis Program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Minh, B.Q.; Schmidt, H.A.; Chernomor, O.; Schrempf, D.; Woodhams, M.D.; von Haeseler, A.; Lanfear, R. IQ-TREE 2: New models and efficient methods for phylogenetic inference in the genomic era. Mol. Biol. Evol. 2020, 37, 1530–1534. [Google Scholar] [CrossRef] [Green Version]
- Hoang, D.T.; Chernomor, O.; von Haeseler, A.; Minh, B.Q.; Vinh, L.S. UFBoot2: Improving the Ultrafast Bootstrap Approximation. Mol. Biol. Evol. 2018, 35, 518–522. [Google Scholar] [CrossRef]
- Stover, B.C.; Muller, K.F. TreeGraph 2: Combining and visualizing evidence from different phylogenetic analyses. BMC Bioinform. 2010, 11, 7. [Google Scholar] [CrossRef] [Green Version]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Stecher, G.; Tamura, K.; Kumar, S. Molecular Evolutionary Genetics Analysis (MEGA) for macOS. Mol. Biol. Evol. 2020, 37, 1237–1239. [Google Scholar] [CrossRef]

| Sample (GenBank Accession Number—State) | Size (bp) | Percent Identity | Query Cover (%)/E-Value | Sequence Best Match (GenBank Accession Number, Locality) |
|---|---|---|---|---|
| Sample 8 OR127024 Pará | 633 | 100% | 100%/0.0 | Piroplasmida sp. from Didelphis aurita—Brazil (OP751800) |
| Sample 12 OR127025 Pará | 725 | 97.66% | 100%/0.0 | Theileria cervi from Odocoileus virginianus—USA (MW008528) |
| Sample 13 OR127026 Pará | 769 | 97.27% | 100%/0.0 | Theileria sp. from Tapirus terrestris—Brazil (OP023835) |
| Sample 65 OR127027 Pará | 737 | 99.59% | 100%/0.0 | Piroplasmida sp. from Didelphis aurita—Brazil (OP751799) |
| Sample 82 OR127028 Pará | 828 | 99.88% | 100%/0.0 | Piroplasmida sp. from Didelphis aurita—Brazil (OP751800) |
| Sample 90 OR127030 Pará | 747 | 96.12% | 100%/0.0 | Theileria sp. from Tapirus terrestris—Brazil (OP023835) |
| Sample 92 OR127031 Pará | 670 | 100% | 100%/0.0 | Theileria sp. from Tapirus terrestris—Brazil (OP023835) |
| Sample 179 OR127032 Pará | 497 | 100% | 99%/0.0 | Piroplasmida sp. from Didelphis aurita—Brazil (OP751800) |
| Sample 182 OR127033 Pará | 625 | 100% | 100%/0.0 | Theileria sp. from Tapirus terrestris—Brazil (OP023835) |
| Sample 205 OR127034 Pará | 474 | 91.04% | 98%/0.0 | Theileria sp. from domestic cats—Brazil (KP402164) |
| Sample 215 OR127035 Pará | 533 | 99.62% | 100%/0.0 | Piroplasmida sp. from Didelphis aurita—Brazil (OP751800) |
| Sample 231 OR127036 Pará | 758 | 99.87% | 100%/0.0 | Babesia sp. from Procyon cancrivorus—Uruguay (MG682489) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
de Mello, V.V.C.; Calchi, A.C.; de Oliveira, L.B.; Coelho, T.F.S.B.; Lee, D.A.B.; Franco, E.O.; Machado, R.Z.; André, M.R. Molecular Survey of Piroplasmids and Hemosporidians in Vampire Bats, with Evidence of Distinct Piroplasmida Lineages Parasitizing Desmodus rotundus from the Brazilian Amazon. Parasitologia 2023, 3, 248-259. https://doi.org/10.3390/parasitologia3030026
de Mello VVC, Calchi AC, de Oliveira LB, Coelho TFSB, Lee DAB, Franco EO, Machado RZ, André MR. Molecular Survey of Piroplasmids and Hemosporidians in Vampire Bats, with Evidence of Distinct Piroplasmida Lineages Parasitizing Desmodus rotundus from the Brazilian Amazon. Parasitologia. 2023; 3(3):248-259. https://doi.org/10.3390/parasitologia3030026
Chicago/Turabian Stylede Mello, Victória Valente Califre, Ana Cláudia Calchi, Laryssa Borges de Oliveira, Taciana Fernandes Souza Barbosa Coelho, Daniel Antônio Braga Lee, Eliz Oliveira Franco, Rosangela Zacarias Machado, and Marcos Rogério André. 2023. "Molecular Survey of Piroplasmids and Hemosporidians in Vampire Bats, with Evidence of Distinct Piroplasmida Lineages Parasitizing Desmodus rotundus from the Brazilian Amazon" Parasitologia 3, no. 3: 248-259. https://doi.org/10.3390/parasitologia3030026
APA Stylede Mello, V. V. C., Calchi, A. C., de Oliveira, L. B., Coelho, T. F. S. B., Lee, D. A. B., Franco, E. O., Machado, R. Z., & André, M. R. (2023). Molecular Survey of Piroplasmids and Hemosporidians in Vampire Bats, with Evidence of Distinct Piroplasmida Lineages Parasitizing Desmodus rotundus from the Brazilian Amazon. Parasitologia, 3(3), 248-259. https://doi.org/10.3390/parasitologia3030026





