Gracilaria parva sp. nov. (Gracilariales, Rhodophyta) a Diminutive Species from the Tropical Eastern Pacific
Abstract
:1. Introduction
2. Materials and Methods
3. Results
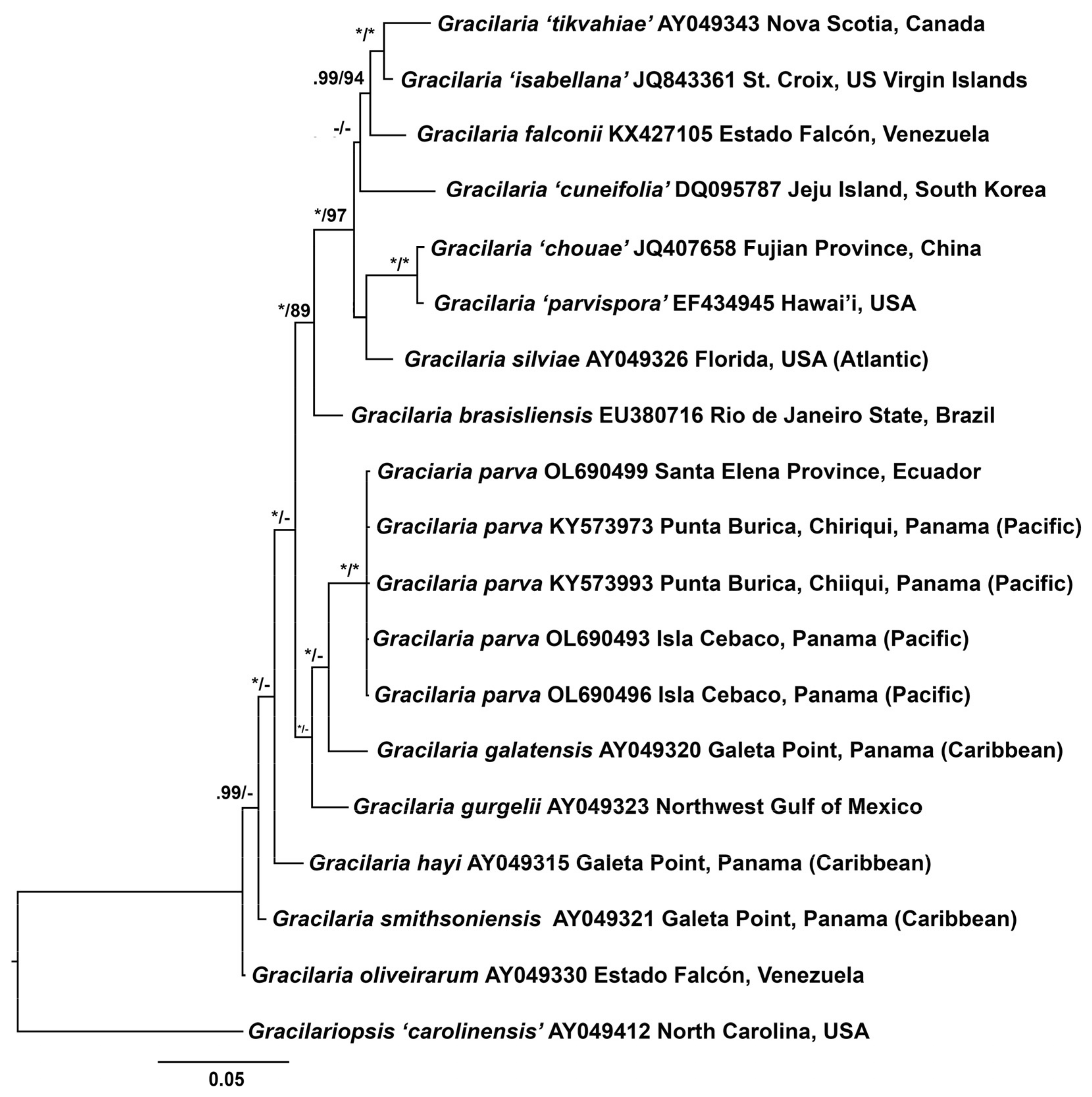
3.1. Molecular Analyses
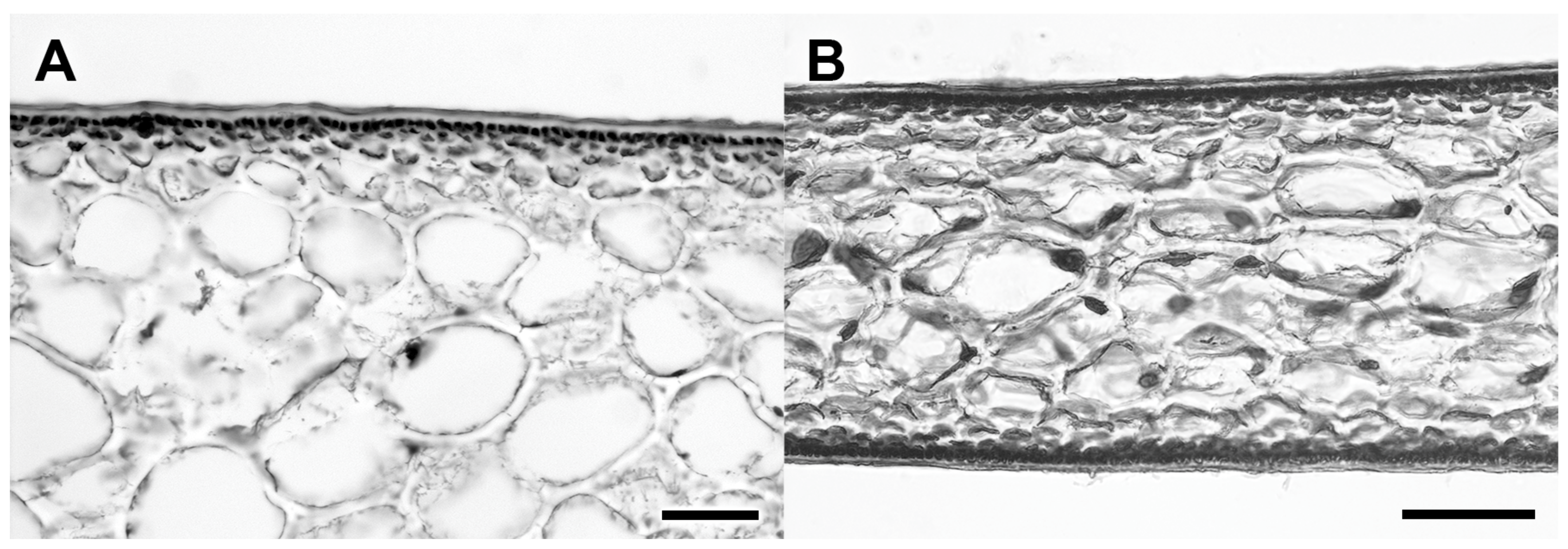
3.2. New Species Description
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Armisen, R. World-wide use and importance of Gracilaria. J. Appl. Psychol. 1995, 7, 231–243. [Google Scholar] [CrossRef]
- Guiry, M.D.; Guiry, G.M. AlgaeBase; World-Wide Electronic Publication; National University of Ireland: Galway, Ireland, 2017; Available online: www.algaebase.org (accessed on 31 October 2021).
- Gargiulo, G.M.; Morabito, M.; Genovese, G.; De Masi, F. Molecular systematics and phylogenetics of Gracilariacean species from the Mediterranean Sea. J. Appl. Phycol. 2006, 18, 497–504. [Google Scholar] [CrossRef]
- Hommersand, M.H.; Freshwater, D.W. Gracilaria hummii sp. nov. (Gracilariales, Rhodophyta), a new name for the agarophyte “Gracilaria confervoides” harvested in North Carolina during World War II. J. Phycol. 2009, 45, 503–516. [Google Scholar] [CrossRef]
- Hardesty, D.M.; Freshwater, D.W. Studies of North Carolina marine algae XIV. Increased diversity of flattened Gracilaria (Gracilariales, Rhodophyta) species revealed by DNA sequences of contemporary specimens and the G. mammillaris type. Bot. Mar. 2018, 61, 407–413. [Google Scholar] [CrossRef]
- Dawes, C.J.; Mathieson, A.C. The Seaweeds of Florida; University Press of Florida: Gainesville, FL, USA, 2008; pp. 1–591. [Google Scholar]
- Norris, J.N. Marine Algae of the Northern Gulf of California, II: Rhodophyta; Smithsonian Contributions to Botany, no. 96; Smithsonian Institution Scholarly Press: Washington, DC, USA, 2014; pp. i–xvi, 1–555. [Google Scholar]
- Gurgel, C.F.; Fredericq, S.; Norris, J.N. Molecular systematics and taxonomy of flattened species of Gracilaria Greville (Gracilariaceae, Gracilariales, Rhodophyta) from the western Atlantic. In Taxonomy of Economic Seaweeds with Reference to the Pacific and Other Locations; Abbott, I.A., McDermid, K.J., Eds.; Hawaii Sea Grant College Program: Honolulu, Hawaii, 2004; Volume IX, pp. 159–199. [Google Scholar]
- Gurgel, C.F.; Fredericq, S.; Norris, J.N.; Yoneshigue-Valentin, Y. Two new flat species of Gracilaria (Gracilariales, Rhodophyta) from Brazil: G. abyssalis sp. nov. and G. brasiliensis sp. nov. Phycologia 2008, 47, 249–264. [Google Scholar] [CrossRef]
- Lyra, G.D.M.; Gurgel, C.F.D.; Costa, E.D.S.; De Jesus, P.B.; Caires, T.A.; Gama de Matos, J.C.; Oliveira, M.C.; Oliveira, E.C.; Nunes, J.M.C. A new tropical species of Gracilariaceae (Rhodophyta, Gracilariales): Gracilaria silviae sp. nov. Phytotaxa 2015, 222, 199–210. [Google Scholar] [CrossRef]
- Soares, L.P.; Gurgel, C.F.D.; Fujii, M.T. Taxonomic reassessment of Gracilaria cearensis (Rhodophyta, Gracilariales), a poorly defined yet common flattened species based on morphological and molecular analysis including topotype collections. Phytotaxa 2015, 201, 241–255. [Google Scholar] [CrossRef]
- Soares, L.P.; Gurgel, C.F.D.; Fujii, M.T. Gracilaria suzanneae sp nov. (Gracilariales, Rhodophyta), a new flattened species from northeast Brazil based on morphological and molecular evidence. Phycologia 2018, 57, 345–353. [Google Scholar] [CrossRef]
- Ardito, M.S.; Núñez-Resendiz, M.L.; Dreckman, K.M.; Sentíes, A. Gracilaria falconii sp. nov. (Gracilariales, Rhodophyta): A new species with flat axes from Venezuela. Phytotaxa 2017, 292, 271–278. [Google Scholar]
- Taylor, W.R. Pacific Marine Algae of the Allan Hancock Expeditions to the Galapagos Islands; The University of Southern California Press: Los Angeles, CA, USA, 1945; p. 528. [Google Scholar]
- Dawson, E.Y. Studies of northeast Pacific Gracilariaceae. A. Hancock Found. Publ. Occas. Pap. 1949, 9, 1–105. [Google Scholar]
- Dawson, E.Y. Marine algae of the Pacific Costa Rican gulfs. Los Angeles County Museum Cont. Sci. 1957, 15, 1–28. [Google Scholar] [CrossRef]
- Dawson, E.Y. Marine red algae of Pacific Mexico, Part 4. Gigartinales. Pac. Nat. 1961, 2, 191–343. [Google Scholar]
- Dawson, E.Y. Plantas marinas de la zona de las mareas de El Salvador. Pac. Nat. 1961, 2, 389–461. [Google Scholar]
- Littler, D.S.; Littler, M.M. Marine Plants of Pacific Panama. 2010. Available online: http://biogeodb.stri.si.edu/pacificalgae (accessed on 25 November 2021).
- Müller-Gelenik, H.; Salazar, M. Algas Marinas del Ecuador; Instituto Nacional de Pesca: Guayaquil, Ecuador, 1996; p. 187.
- Bernecker, A.; Wehrtmann, I.S. New records of benthic marine algae and Cyanobacteria for Costa Rica, and a comparison with other Central American countries. Helgol. Mar. Res. 2009, 63, 219–229. [Google Scholar] [CrossRef]
- Schnetter, R.; Bula Meyer, G. Algas Marinas del Litoral Pacífico de Colombia, Chlorophyceae, Phaeophyceae, Rhodophyceae; Lubrecht & Cramer Limited: Stuttgart, Germany, 1982; p. 287. [Google Scholar]
- Freshwater, D.W.; Idol, J.N.; Parham, S.L.; Fernández-García, C.; León, N.; Gabrielson, P.W.; Wysor, B.L. Molecular assisted identification reveals hidden red algae diversity from the Burica Peninsula, Pacific Panama. Diversity 2017, 9, 19. [Google Scholar] [CrossRef]
- Thiers, B. Index Herbariorum: A Global Directory of Public Herbaria and Associated Staff. 2021 (Continuously Updated). New York Botanical Garden’s Virtual Herbarium. Available online: https://sweetgum.nybg.org/ih/ (accessed on 31 October 2021).
- Millar, A.J.K.; Wynne, M.J. Patulophycus eclipes gen. et sp. nov. (Delesseriaceae, Rhodophyta) from the southwestern Pacific. Syst. Bot. 1992, 17, 409–416. [Google Scholar] [CrossRef]
- Hughey, J.R.; Silva, P.C.; Hommersand, M.H. Solving taxonomic and nomenclatural problems in Pacific Gigartinaceae (Rhodophyta) using DNA from type material. J. Phycol. 2001, 37, 1091–1109. [Google Scholar] [CrossRef]
- Freshwater, D.W.; Rueness, J. Phylogenetic relationships of some European Gelidium (Gelidiales, Rhodophyta) species based on rbcL nucleotide sequence analysis. Phycologia 1994, 33, 187–194. [Google Scholar] [CrossRef]
- Taylor, R.L.; Bailey, J.C.; Freshwater, D.W. Systematics of Cladophora spp. (Chlorophyta) from North Carolina, USA, based upon morphology and DNA sequence data with a description of Cladophora subtilissima sp. nov. J. Phycol. 2017, 53, 541–556. [Google Scholar] [CrossRef]
- Freshwater, D.W.; Scott, S.; Tronchin, E.M.; Saunders, G.W. Reassessment of Tristan da Cunha Gelidium (Gelidiales, Rhodophyta) species. Bot. Mar. 2020, 63, 455–462. [Google Scholar] [CrossRef]
- Le Gall, L.; Saunders, G.W. DNA barcoding is a powerful tool to uncover algal diversity: A case study of the Phyllophoraceae (Gigartinales, Rhodophyta) in the Canadian flora. J. Phycol. 2010, 46, 374–389. [Google Scholar] [CrossRef]
- Saunders, G.W.; McDevit, D.C. Methods for DNA barcoding photosynthetic protists emphasizing the macroalgae and diatoms. Methods Mol. Biol. 2012, 858, 207–222. [Google Scholar] [PubMed]
- Sherwood, A.R.; Presting, G.G. Universal primers amplify a 23S rDNA plastid marker in eukaryotic algae and cyanobacteria. J. Phycol. 2007, 43, 605–608. [Google Scholar] [CrossRef]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [Green Version]
- Stamatakis, A. RAxML Version 8: A tool for phylogenetic analysis and post-analysis of large phylogenies. Bioinformatics 2014, 30, 1312–1313. [Google Scholar] [CrossRef]
- Huelsenbeck, J.P.; Ronquist, F. MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 2001, 17, 754–775. [Google Scholar] [CrossRef] [Green Version]
- Bernecker, A. Marine benthic algae. In Marine Biodiversity of Costa Rica; Wehrtmann, I.S., Cortés, J., Eds.; Springer Science: Berlin, Germany, 2009; pp. 109–117. [Google Scholar]
- Setchell, W.A.; Gardner, N.L. New marine algae from the Gulf of California. Proc. Cal. Acad. Sci. Iv 1924, 12, 695–949. [Google Scholar]
- Dawson, E.Y. The marine algae of the Gulf of California. A. Hancock Pac. Exped. 1944, 3, 189–464. [Google Scholar]
- Howe, M.A. Report on a botanical visit to the Isthmus of Panama. J. N. Y. Bot. Gard. 1910, 11, 30–44. [Google Scholar]
- Dawson, E.Y. Additions to the marine flora of Costa Rica and Nicaragua. Pac. Nat. 1962, 3, 375–395. [Google Scholar]
- Glynn, P.W. Observations on the ecology of the Caribbean and Pacific coasts of Panamá. Bull. Biol. Soc. Wash. 1972, 2, 13–30. [Google Scholar]
- Hay, M.E.; Gaines, S.D. Geographic differences in herbivore impact: Do Pacific herbivores prevent Caribbean seaweeds from colonizing via the Panama Canal? Biotropica 1984, 16, 24–30. [Google Scholar] [CrossRef]
- Wysor, B.L. An annotated list of marine Chlorophyta from the Pacific coast of the Republic of Panama with a comparison to Caribbean Panama species. Nova Hedwigia 2004, 78, 209–241. [Google Scholar] [CrossRef]
- Grusz, A.L.; Freshwater, D.W. Studies of Costa Rican Gelidiales (Florideophyceae): II. Two Pacific taxa including Gelidium microglossum sp. nov. Pac. Sci. 2014, 68, 97–110. [Google Scholar] [CrossRef]
- Quiroz-González, N.; Ponce-Márquez, M.E.; Fernández-García, C.; Rodríguez, D. Gelidium gonzalezii sp. nov. (Gelidiales, Rhodophyta) from the Mexican tropical Pacific based on molecular and morphological evidence. Phytotaxa 2020, 459, 124–138. [Google Scholar] [CrossRef]
- Quiroz-González, N.; Ponce-Márquez, M.E.; López-Gómez, N.; Rodríguez, D. Morphological and molecular characterization of two species of Gelidium (Gelidiales, Rhodophyta) from Mexico: G. nayaritense sp. nov. and the new record of G. sanyaense. Phytotaxa 2021, 527, 161–176. [Google Scholar] [CrossRef]
- Freshwater, D.W.; University of North Carolina, Wilmington, NC, USA. Personal communication, 2021.
- Fernández-García, C.; Brenes-Mesen, L.; González, K.; Rojas-Angulo, M.; Quesada-Pérez, F.; Tejada, O.L. Unravelling marine macroalgae from the Pacific coast of Central America. Phycologia 2021, 60 (Suppl. 1), 70. [Google Scholar] [CrossRef]


| Species | Mexico | El Salvador | Costa Rica | Panama | Colombia | Ecuador |
|---|---|---|---|---|---|---|
| G. brevis W.R. Taylor | [19] | [14] | ||||
| G. crispata Setchell and N.L. Gardner | [17] | [18] | [16] | |||
| G. domingensis (Kützing) Sonder | [20], p. 187 | |||||
| G. ecuadoreana (W.R. Taylor) E.Y. Dawson | [18] | |||||
| G. johnstonii Setchell and N.L. Gardner | [14] | |||||
| G. linearis Kylin | [14] | |||||
| G. pinnata Setchell and N.L. Gardner | [14] | |||||
| G. spinigera E.Y. Dawson | [21] | |||||
| G. symmetrica E.Y. Dawson | [17] | [22] | ||||
| G. tepocensis (E.Y. Dawson) E.Y. Dawson | [18] | [16] a | ||||
| G. veleroae E.Y. Dawson | [17] |
| Collection No. | Voucher | Collection Information | GenBank Accession No. | ||
|---|---|---|---|---|---|
| rbcL | COI-5P | UPA | |||
| PHYKOS-4630 | WNC-34055 | Southwest side of Isla Burica, Chiriqui, Panama, 08.01865° N 082.88390° W, ca. 6 m depth, 10.i.11, collected by D.W. Freshwater. | KY573993 | KY656553 | KY573953 |
| PHYKOS-4630 | NCU-675206 | Southwest side of Isla Burica, Chiriqui, Panama, 08.01865° N 082.88390° W, ca. 6 m depth, 10.i.11, collected by D.W. Freshwater. | - | - | - |
| PHYKOS-4541 | WNC-34186 | Near Mono Feliz, Punta Burica, Chiriqui, Panama, 08.03042° N 082.87574° W, 3 m depth, 08.i.11, collected by B.L. Wysor and D.W. Freshwater. | KY573973 | KY656537 | KY573932 |
| PHYKOS-4519 | WNC-34181 | Near Mono Feliz, Punta Burica, Chiriqui, Panama, 08.03042° N 082.87574° W, 3 m depth, 08.i.11, collected by B.L. Wysor and D.W. Freshwater. | - | KY656532 | KY573927 |
| PHYKOS-6833 | WNC-34185 | Northwest side of Sombrero Rock Cove, Isla Cebaco, Veraguas, Panama, 07.48229° N 081.25705° W, 17 m depth, 20.i.12, collected by D.W. Freshwater. | OL690493 | OL690495 | OL690494 |
| PHYKOS-6870 | WNC-34184 | Cebaco Bay rock pile, Isla Cebaco, Veraguas, Panama, 07.49173° N 081.22262° W, 14 m depth, 20.i.12, collected by D.W. Freshwater. | OL690496 | OL690498 | OL690497 |
| E438 | NCU-675207 | La Chocolatera, Santa Elena Province, Ecuador, 02.18667° S 081.00583° W, 21.i.2021, epilithic low intertidal, collected by A. Eguiguren and M. Brandt. | OL690499 | - | - |
| E438 | USFQ #438 | La Chocolatera, Santa Elena Province, Ecuador, 02.18667° S 081.00583° W, 21.i.2021, epilithic low intertidal, collected by A. Eguiguren and M. Brandt. | - | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Freshwater, D.W.; Williamson, B.; Gabrielson, P.W.; Brandt, M. Gracilaria parva sp. nov. (Gracilariales, Rhodophyta) a Diminutive Species from the Tropical Eastern Pacific. Taxonomy 2022, 2, 48-56. https://doi.org/10.3390/taxonomy2010004
Freshwater DW, Williamson B, Gabrielson PW, Brandt M. Gracilaria parva sp. nov. (Gracilariales, Rhodophyta) a Diminutive Species from the Tropical Eastern Pacific. Taxonomy. 2022; 2(1):48-56. https://doi.org/10.3390/taxonomy2010004
Chicago/Turabian StyleFreshwater, D. Wilson, Bo Williamson, Paul W. Gabrielson, and Margarita Brandt. 2022. "Gracilaria parva sp. nov. (Gracilariales, Rhodophyta) a Diminutive Species from the Tropical Eastern Pacific" Taxonomy 2, no. 1: 48-56. https://doi.org/10.3390/taxonomy2010004
APA StyleFreshwater, D. W., Williamson, B., Gabrielson, P. W., & Brandt, M. (2022). Gracilaria parva sp. nov. (Gracilariales, Rhodophyta) a Diminutive Species from the Tropical Eastern Pacific. Taxonomy, 2(1), 48-56. https://doi.org/10.3390/taxonomy2010004






