An Extinct New Rail (Gallirallus, Aves: Rallidae) Species from Rapa Island, French Polynesia †
Abstract
1. Introduction
2. Material & Methods
3. Systematics
| Family Rallidae GenusGallirallus Lafresnaye,1841 Gallirallus astolfoisp. nov. (Figure 1) |
| rail (cf. Gallirallus): Tennyson & Anderson, 2012: 108. |
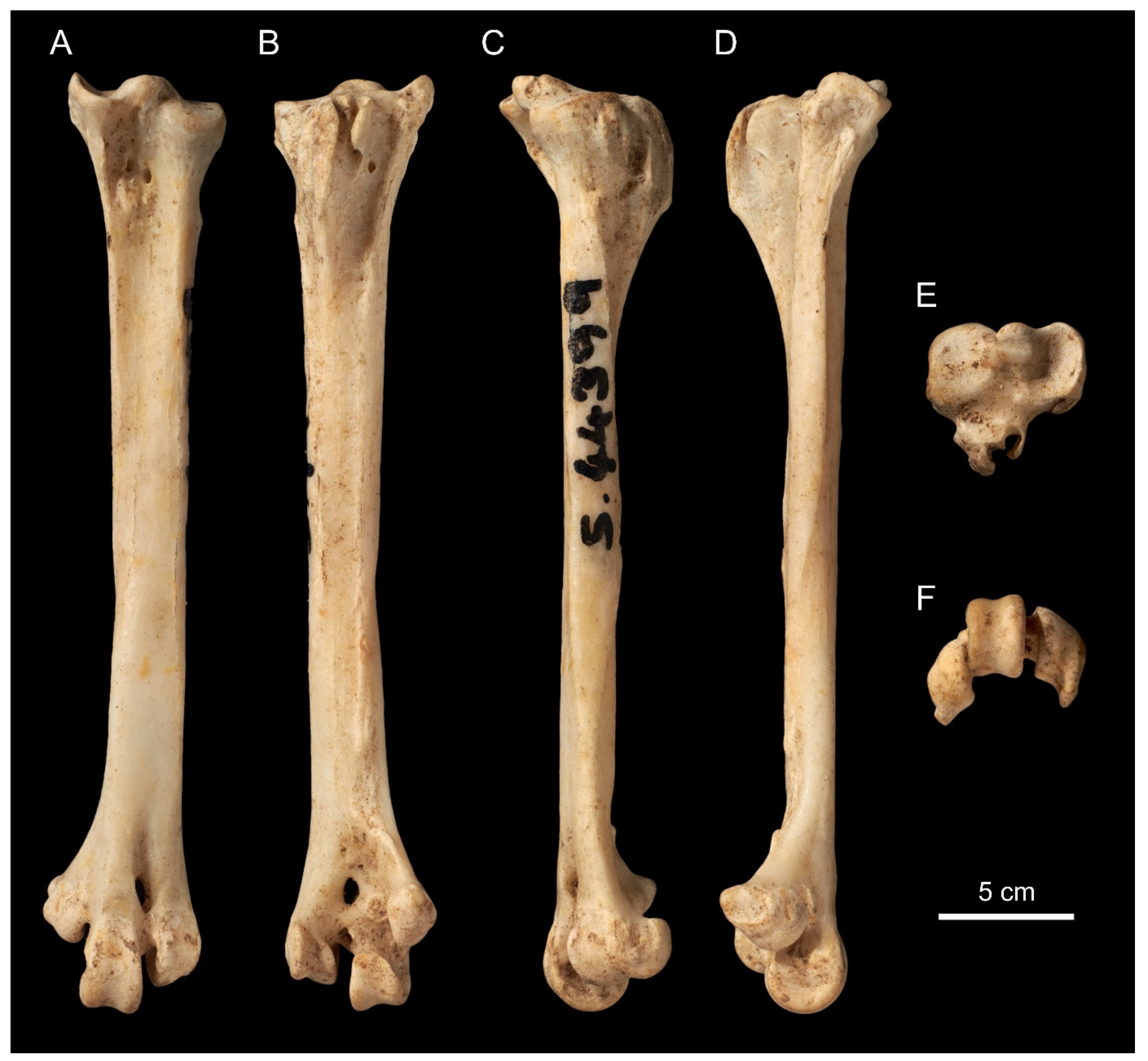
4. Discussion
5. Conclusions
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
Appendix A
References
- Anderson, A.; Kennett, D.J.; Conte, E. Archaeological research on Rapa Island, French Polynesia. Terra Aust. 2012, 37, 7–23. [Google Scholar]
- Anderson, A.; Kennett, D.J.; Conte, E. The prehistory of Rapa Island. Terra Aust. 2012, 37, 247–256. [Google Scholar]
- Kennett, D.; Anderson, A.; Prebble, M.; Conte, E.; Southon, J. Prehistoric human impacts on Rapa, French Polynesia. Antiquity 2006, 80, 340–354. [Google Scholar] [CrossRef]
- Richards, R. The earliest foreign visitors and their massive depopulation of Rapa-iti from 1824 to 1830. J. Société Océanistes 2004, 118, 3–10. [Google Scholar] [CrossRef]
- Tennyson, A.; Anderson, A. Bird, reptile and mammal remains from archaeological sites on Rapa Island. Terra Aust. 2012, 37, 105–114. [Google Scholar]
- Alcover, J.A.; Pieper, H.; Pereira, F.; Rando, J.C. Five new extinct species of rails (Aves: Gruiformes: Rallidae) from the Macaronesian Islands (North Atlantic Ocean). Zootaxa 2015, 4057, 151–190. [Google Scholar] [CrossRef] [PubMed]
- Bourne, W.R.P.; Ashmole, N.P.; Simmons, K.E.L. A new subfossil night heron and a new genus for the extinct rail from Ascension Island, central tropical Atlantic Ocean. Ardea 2003, 91, 45–51. [Google Scholar]
- Kirchman, J.J.; Steadman, D.W. Rails (Rallidae: Gallirallus) from prehistoric archaeological sites in Western Oceania. Zootaxa 2006, 1316, 1–31. [Google Scholar] [CrossRef]
- Livezey, B.C. Evolution of flightlessness in rails (Gruiformes: Rallidae): Phylogenetic, ecomorphological, and ontogenetic perspectives. Ornithol. Monogr. 2003, 53, 1–654. [Google Scholar] [CrossRef]
- Matsuoka, H. The Late Pleistocene fossil birds of the central and southern Ryukyu Islands, and their zoogeographical implications for the recent avifauna of the archipelago. Tropics 2000, 10, 165–188. [Google Scholar] [CrossRef]
- Olson, S.L. A classification of the Rallidae. Wilson Bull. 1973, 85, 381–416. [Google Scholar]
- Taylor, B.; van Perlo, B. Rails. A Guide to the Rails, Crakes, Gallinules and Coots of the World; Pica Press: Sussex, UK, 1998; 600p. [Google Scholar]
- Kirchman, J.J.; Steadman, D.W. Rails (Aves: Rallidae: Gallirallus) from prehistoric sites in the Kingdom of Tonga, including a description of a new species. Proc. Biol. Soc. Wash. 2005, 118, 465–477. [Google Scholar] [CrossRef]
- Olson, S.L.; Wingate, D.B. Two new species of flightless rails (Aves: Rallidae) from the Middle Pleistocene “crane fauna” of Bermuda. Proc. Biol. Soc. Wash. 2000, 113, 356–368. [Google Scholar]
- Olson, S.L.; Wingate, D.B. A new species of large flightless rail of the Rallus longirostris/elegans complex (Aves: Rallidae) from the Late Pleistocene of Bermuda. Proc. Biol. Soc. Wash. 2001, 114, 509–516. [Google Scholar]
- Takano, O.M.; Steadman, D.W. Another new species of flightless rail (Aves: Rallidae: Rallus) from Abaco, The Bahamas. Zootaxa 2018, 4407, 376–382. [Google Scholar] [CrossRef] [PubMed]
- Anderson, A. Archaeology of the coastal sites on Rapa Island. Terra Aust. 2012, 37, 47–76. [Google Scholar]
- Baumel, J.J.; Witmer, L.M. Osteologia. In Handbook of Avian Anatomy: Nomina Anatomica Avium; Baumel, J.J., King, A.S., Breazile, J.E., Evans, H.E., Vanden Berge, J.C., Eds.; Nuttall Ornithological Club: Cambridge, UK, 1993; pp. 45–132. [Google Scholar]
- Gilbert, B.M.; Martin, L.D.; Savage, H.G. Avian Osteology; Modern Printing: Laramie, WY, USA, 1981; p. 252. [Google Scholar]
- Livezey, B.C.; Zusi, R.L. Higher-order phylogeny of modern birds (Theropoda, Aves: Neornithes) based on comparative anatomy. II. Analysis and discussion. Zool. J. Linn. Soc. 2007, 149, 1–95. [Google Scholar] [CrossRef]
- Winkler, D.W.; Billerman, S.M.; Lovette, I.J. Rails, gallinules, and coots (Rallidae), version 1.0. In Birds of the World; Billerman, S.M., Keeney, B.K., Rodewald, P.G., Schulenberg, T.S., Eds.; Cornell Lab of Ornithology: Ithaca, NY, USA, 2020. [Google Scholar] [CrossRef]
- Kirchman, J.J.; McInerney, N.R.; Giarla, T.C.; Olson, S.L.; Slikas, E.; Fleischer, R.C. Phylogeny based on ultra-conserved elements clarifies the evolution of rails and allies (Ralloidea) and is the basis for a revised classification. Ornithology 2021, 138, 1–21. [Google Scholar] [CrossRef]
- Kirchman, J.J.; McInerney, N.R.; Giarla, T.C.; Olson, S.L.; Slikas, E.; Fleischer, R.C. Corrigendum to: Phylogeny based on ultra-conserved elements clarifies the evolution of rails and allies (Ralloidea) and is the basis for a revised classification. Ornithology 2021, 138, ukab065. [Google Scholar] [CrossRef]
- Kirchman, J.J.; Steadman, D.W. New species of extinct rails (Aves: Rallidae) from archaeological sites in the Marquesas Islands, French Polynesia. Pac. Sci. 2007, 61, 145–163. [Google Scholar] [CrossRef]
- Worthy, T.H.; Burley, D.V. Prehistoric avifaunas from the Kingdom of Tonga. Zool. J. Linn. Soc. 2020, 189, 998–1045. [Google Scholar] [CrossRef]
- Steadman, D.W.; Bollt, R. Prehistoric Birds from Rurutu, Austral Islands, East Polynesia. Pac. Sci. 2010, 64, 315–325. [Google Scholar] [CrossRef]
- Worthy, T.H.; Bollt, R. Prehistoric birds and bats from the Atiahara Site, Tubuai, Austral Islands, East Polynesia. Pac. Sci. 2011, 65, 69–85. [Google Scholar] [CrossRef]
- Kirchman, J.J.; Steadman, D.W. New species of rails (Aves: Rallidae) from an archaeological site on Huahine, Society Islands. Pac. Sci. 2006, 60, 281–297. [Google Scholar] [CrossRef]
- Steadman, D.W. Extinction of birds in Eastern Polynesia: A review of the record, and comparisons with other Pacific Island groups. J. Archaeol. Sci. 1989, 16, 177–205. [Google Scholar] [CrossRef]
- Steadman, D.W. Prehistoric extinctions of Pacific island birds: Biodiversity meets zooarchaeology. Science 1995, 267, 1123–1131. [Google Scholar] [CrossRef] [PubMed]
- Seitre, R.; Seitre, J. Causes of land-bird extinctions in French Polynesia. Oryx 1992, 26, 215–222. [Google Scholar]
- Thibault, J.-C.; Varney, A. Numbers and habitat of the Rapa fruit-dove, Ptilinopus huttoni. Bird Conserv. Int. 1991, 1, 75–81. [Google Scholar] [CrossRef]
- Holyoak, D.T.; Thibault, J.-C. Contribution à L’étude des Oiseaux de Polynésie Orientale. Mémoires du Muséum National D’Histoire Naturelle Série A Zoologie 1984, 127, 1–209. [Google Scholar]
- Thibault, J.-C.; Varney, A. Breeding seabirds of Rapa (Polynesia): Numbers and changes during the 20th century. Bull. Br. Ornithol. Club 1991, 111, 70–77. [Google Scholar]
- Olson, S.L. Evolution of the rails of the South Atlantic islands (Aves: Rallidae). Smithson. Contrib. Zool. 1973, 152, 1–53. [Google Scholar] [CrossRef]
- García-R, J.C.; Gibb, G.C.; Trewick, S.A. Deep global evolutionary radiation in birds: Diversification and trait evolution in the cosmopolitan bird family Rallidae. Mol. Phylogenet. Evol. 2014, 81, 96–108. [Google Scholar] [CrossRef] [PubMed]
- García-R, J.C.; Gibb, G.C.; Trewick, S.A. Eocene diversification of crown group rails (Aves: Gruiformes: Rallidae). PLoS ONE 2014, 9, e109635. [Google Scholar] [CrossRef] [PubMed]
- Kirchman, J.J. Speciation of flightless rails on islands: A DNA-based phylogeny of the typical rails of the Pacific. Auk 2012, 129, 56–69. [Google Scholar]
| Gallirallus spp. | Geographic Distribution | Flight | Status | Total Length | Distal Width | Proximal Width | Mid-Shaft Width | Mid-Shaft Depth | Reference |
|---|---|---|---|---|---|---|---|---|---|
| G. astolfoi sp. nov. | Fench Polynesia (Rapa Iti) | flightless | extinct | †: 34.6 (1) | †: 5.8 (1) | †: 5.8 (1) | †: 2.7 (1) | †: 2.1 (1) | this paper |
| G. australis (Sparrman, 1786) | New Zealand | flightless | living | †: 60.8–70.7 (7) | †: 11.1–12.8 (7) | †: 9.7–11.9 (7) | †: 4.7–6.0 (7) | †: 4.1–5.0 (7) | this paper |
| G. calayanensis Allen et al., 2004 | Philippines (Calayan Is.) | flightless | living | — | — | — | — | — | — |
| G. dieffenbachii (G.R. Gray, 1843) | New Zealand (Chatham Is.) | flightless | extinct | †: 42.0–48.0 (10) | †: 7.8–8.8 (10) | †: 7.3–8.3 (10) | †: 3.4–4.3 (10) | †: 3.2–4.0 (10) | this paper |
| G. epulare Kirchman & Steadman, 2007 | French Polynesia (Nuku Hiva) | flightless | extinct | — | †: 6.5 (1) | — | †: 3.5 (1) | †: 2.4 (1) | [24] |
| G. ernstmayri Kirchman & Steadman, 2006 | Papua New Guinea (New Ireland) | flightless | extinct | — | — | — | — | — | [8] |
| G. gracilitibia Kirchman & Steadman, 2007 | French Polynesia (Ua Huka) | flightless | extinct | — | — | — | — | — | [8] |
| G. huiatua Steadman et al., 2000 | Niue | flightless | extinct | †: 39.2 (1) | †: 6.5 (1) | †: 5.4 (1) | †: 2.7 (1) | †: 2.5 (1) | this paper |
| G. insignis (P.L. Sclater, 1880) | Papua New Guinea (New Britain) | flightless | living | — | — | — | — | — | — |
| G. lafresnayanus Verreaux & Des Murs, 1860 | New Caledonia | flightless | extinct | — | — | — | — | — | — |
| G. modestus (Hutton, 1872) | New Zealand (Chatham Is.) | flightless | extinct | †: 26.4–32.1 (4) | †: 4.8–5.4 (4) | †: 4.4–5.0 (4) | †: 2.1–2.4 (4) | †: 1.7–2.1 (4) | this paper |
| G. okinawae (Yamashina & Mano, 1981) | Japan (Okinawa) | flightless | living | — | — | — | — | — | — |
| G. owstoni (Rothschild, 1895) | Guam, Mariana Islands | flightless | living | F: 49.6 (1) M: 49.6–53.7 (2) | — | F: 7.1 (1) M: 7.1–8.2 (2) | — | — | [13] |
| G. pacificus (Gmelin, 1789) | French Polynesia (Tahiti) | flightless? | extinct | — | — | — | — | — | [12] |
| G. pendiculentus Kirchman & Steadman, 2006 | Northern Mariana Is. (Tinian) | flightless | extinct | †: 38.4–46.5 (7) | †: 6.1–6.8 (14) | †: 5.6–7.3 (17) | †: 2.7–3.7 (17) | — | [8] |
| G. philippensis (Linnaeus, 1766) | Oceania | volant | living | F: 40.8–45.2 (6) M: 45.3–49.0 (6) †: 36.7–43.9 (4) | F: 6.0–6.9 (6) M: 6.7–7.5 (6) †: 6.2–7.5 (4) | F: 5.8–6.6 (6) M: 6.7–7.2 (6) †: 5.6–7.1 (4) | F: 2.8–3.3 (6) M: 3.1–3.6 (6) †: 3.1–3.9 (4) | †: 2.5–3.3 (4) | F/M: [8] †: this paper |
| G. pisonii Kirchman & Steadman, 2006 | Northern Mariana Is. (Aguiguan) | flightless | extinct | — | †: 6.1 (1) | †: 5.6–5.7 (2) | †: 3.0–3.1 (2) | — | [8] |
| G. poecilopterus (Hartlaub, 1866) | Fiji | flightless | extinct | †: 66.6–70.0 (2) | †: 10.0 (1) | †: 9.5 (1) | †: 4.0 (1) | — | [25] |
| G. ripleyi Steadman, 1986 | Cook Is. (Mangaia) | flightless | extinct | †: 33.1 (1) | †: 6.3–6.5 (3) | †: 5.7–5.9 (2) | †: 2.8–3.1 (2) | — | [8,13] |
| G. roletti Kirchman & Steadman, 2007 | French Polynesia (Tahuata) | flightless | extinct | — | †: 8.7 (1) | †: 7.9 (1) | †: 3.5–3.9 (2) | †: 2.5–2.8 (2) | [24] |
| G. rovianae Diamond, 1991 | Solomon Is. | flightless | living | — | — | — | — | — | — |
| G. sp. sensu Steadman & Bollt, 2010 | Fench Polynesia (Rurutu) | flightless | extinct | — | — | — | — | — | [26] |
| G. steadmani Worthy & Bollt, 2011 | Fench Polynesia (Tubuai) | flightless | extinct | †: 40.0 (1) | †: 6.2 (1) | †: 5.9–6.4 (3) | †: 2.9–3.1 (3) | — | [27] |
| G. storrsolsoni Kirchman & Steadman, 2006 | Fench Polynesia (Huahine) | flightless | extinct | — | — | †: 7.4 (1) | †: 4.1 (1) | †: 2.8 (1) | [28] |
| G. sylvestris (P.L. Sclater, 1870) | Australia (Lord Howe Is.) | flightless | living | M: 49.6 (1) | M: 8.9 (1) | M: 8.7 (1) | M: 4.4 (1) | M: 3.4 (1) | this paper |
| G. temptatus Kirchman & Steadman, 2006 | Northern Mariana Is. (Rota) | flightless | extinct | †: 47.4 (1) | †: 7.4 (1) | †: 6.4–6.8 (2) | †: 3.3–3.5 (2) | — | [8] |
| G. torquatus (Linnaeus, 1766) | Indonesia, Papua New Guinea, Philippines | volant | living | M: 54.2 (1) | — | M: 7.4 (1) | — | — | [13] |
| G. vavauensis (Worthy & Burley, 2020) | Tonga (Vava’u) | flightless | extinct | †: 70.9–73.0 (2) | †: 12.2–13.1 (2) | †: 12.9–13.2 (3) | †: 5.9–6.7 (6) | †: 5.4–6.0 (6) | [25] |
| G. vekamatolu Kirchman & Steadman, 2005 | Tonga (’Eua) | flightless | extinct | †: 45.1 (1) | — | †: 7.7–8.6 (2) | — | — | [13] |
| G. wakensis (Rothschild, 1903) | USA (Wake Is.) | flightless | extinct | F: 33.3 (1) M: 35.6 (1) | F: 5.9 (1) M: 6.2 (1) | F: 5.5 (1) M: 6.0 (1) | F: 2.5 (1) M: 2.9 (1) | — | [8] |
| G. wallacii (G.R. Gray, 1861) | Indonesia (Halmahera) | flightless | living | — | — | — | — | — | — |
| G. woodfordi (Ogilvie-Grant, 1889) | Solomon Is. | flightless | living | F: 67.7–72.0 (3) M: 69.8–72.3 (3) | F: 10.5–10.6 (3) M: 10.7–11.0 (3) | F: 9.9–10.1 (3) M: 10.2–10.7 (3) | F: 4.4–4.7 (3) M: 4.8–5.0 (3) | — | [8] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Salvador, R.B.; Anderson, A.; Tennyson, A.J.D. An Extinct New Rail (Gallirallus, Aves: Rallidae) Species from Rapa Island, French Polynesia. Taxonomy 2021, 1, 448-457. https://doi.org/10.3390/taxonomy1040032
Salvador RB, Anderson A, Tennyson AJD. An Extinct New Rail (Gallirallus, Aves: Rallidae) Species from Rapa Island, French Polynesia. Taxonomy. 2021; 1(4):448-457. https://doi.org/10.3390/taxonomy1040032
Chicago/Turabian StyleSalvador, Rodrigo B., Atholl Anderson, and Alan J. D. Tennyson. 2021. "An Extinct New Rail (Gallirallus, Aves: Rallidae) Species from Rapa Island, French Polynesia" Taxonomy 1, no. 4: 448-457. https://doi.org/10.3390/taxonomy1040032
APA StyleSalvador, R. B., Anderson, A., & Tennyson, A. J. D. (2021). An Extinct New Rail (Gallirallus, Aves: Rallidae) Species from Rapa Island, French Polynesia. Taxonomy, 1(4), 448-457. https://doi.org/10.3390/taxonomy1040032







