Wet vs. Dry Dentin Bonding: A Systematic Review and Meta-Analysis of Adhesive Performance and Hybrid Layer Integrity
Abstract
1. Introduction
2. Materials and Methods
3. Results
3.1. Selection of the Included Studies
3.2. Characteristics of Included Studies
3.3. Methodological Quality Assessment
3.4. Certainty of Evidence (GRADE)
3.5. Microtensile Bond Strength
3.6. Long-Term Bond Durability
3.7. Interface Morphology
3.8. Effect of Acetone Wet Bonding and Ethanol Wet Bonding
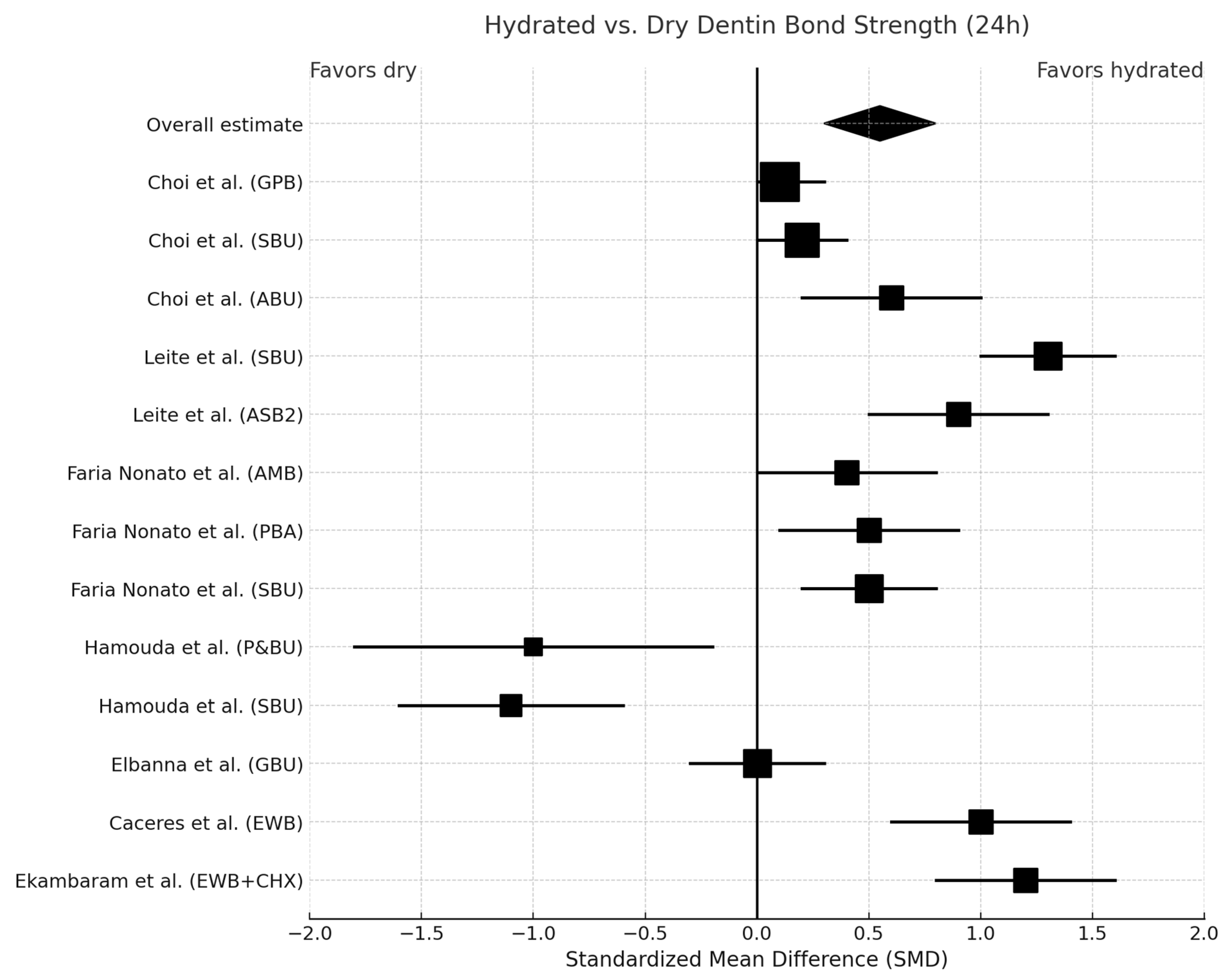
3.9. Meta-Analysis Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| 10-MDP | 10-methacryloyloxydecyl dihydrogen phosphate |
| MMP | Matrix Metalloproteinase |
| μTBS | Microtensile Bond Strength |
| MPa | Megapascal |
| PRISMA | Preferred Reporting Items for Systematic reviews and Meta-Analyses |
| DMSO | Dimethyl Sulfoxide |
| HEMA | 2-Hydroxyethyl Methacrylate |
| UDMA | Urethane Dimethacrylate |
| TEGDMA | Triethylene Glycol Dimethacrylate |
| Bis-GMA | Bisphenol A Glycidyl Methacrylate |
| SBU | Scotchbond Universal |
| PBU | Prime&Bond Universal |
| BBU | BeautiBond Universal |
| UBQ | Universal Bond Quick |
| UBQexp | Universal Bond Quick Experimental |
| AMB | Ambar Universal APS |
| PBA | Prime&Bond Active |
| FTIR | Fourier-Transform Infrared Spectroscopy |
| CLSM | Confocal Laser Scanning Microscopy |
| SEM | Scanning Electron Microscopy |
| ESEM | Environmental Scanning Electron Microscopy |
| WWB | Water Wet Bonding |
| EWB | Ethanol Wet Bonding |
| AWB | Acetone Wet Bonding |
| 50%AWB | 50% Acetone Wet Bonding |
| ER | Etch-and-Rinse |
| SE | Self-Etch |
| SMD | Standardized Mean Difference |
| CI | Confidence Interval |
| ROB | Risk of Bias |
| GRADE | Grading of Recommendations, Assessment, Development, and Evaluations |
| MINORS | Methodological Index for Non-Randomized Studies |
| SNU | Silver Nitrate Uptake |
References
- Pashley, D.H.; Tay, F.R.; Carvalho, R.M.; Rueggeberg, F.A.; Agee, K.A.; Carrilho, M.; Donnelly, A.; García-Godoy, F. From dry bonding to water-wet bonding to ethanol-wet bonding. A review of the interactions between dentin matrix and solvated resins using a macromodel of the hybrid layer. Am. J. Dent. 2007, 20, 7–20. [Google Scholar]
- Tay, F.R.; Pashley, D.H. Dental adhesives of the future. J. Adhes. Dent. 2002, 4, 91–103. [Google Scholar] [PubMed]
- Mokeem, L.S.; Garcia, I.M.; Melo, M.A. Degradation and Failure Phenomena at the Dentin Bonding Interface. Biomedicines 2023, 11, 1256. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Amin, F.; Fareed, M.A.; Zafar, M.S.; Khurshid, Z.; Palma, P.J.; Kumar, N. Degradation and Stabilization of Resin-Dentine Interfaces in Polymeric Dental Adhesives: An Updated Review. Coatings 2022, 12, 1094. [Google Scholar] [CrossRef]
- Franz, A.; König, F.; Skolka, A.; Sperr, W.; Bauer, P.; Lucas, T.; Watts, D.C.; Schedle, A. Cytotoxicity of resin composites as a function of interface area. Dent. Mater. 2007, 23, 1438–1446. [Google Scholar] [CrossRef]
- Frassetto, A.; Breschi, L.; Turco, G.; Marchesi, G.; Di Lenarda, R.; Tay, F.R.; Pashley, D.H.; Cadenaro, M. Mechanisms of degradation of the hybrid layer in adhesive dentistry and therapeutic agents to improve bond durability—A literature review. Dent. Mater. 2016, 32, e41–e53. [Google Scholar] [CrossRef]
- Pioch, T.; Staehle, H.; Wurst, M.; Duschner, H.; Dörfer, C. The nanoleakage phenomenon: Influence of moist vs dry bonding. J. Adhes. Dent. 2002, 4, 23. [Google Scholar]
- Isolan, C.P.; Valente, L.L.; Münchow, E.A.; Basso, G.R. Bond strength of a universal bonding agent and other adhesives applied to multiple substrates. Appl. Adhes. Sci. 2014, 2, 25. [Google Scholar] [CrossRef]
- Chuang, S.F.; Chang, L.T.; Chang, C.H.; Yaman, P.; Liu, J.K. Influence of enamel wetness on composite restorations using various dentin bonding agents. J. Dent. 2006, 34, 682–691. [Google Scholar] [CrossRef]
- Kwon, J.; Kim, J.; Choi, D.; Kim, D.-S. The Influence of Drying Time, Application Mode, and Agitation on the Dentin Bond Strength of a Novel Mesoporous Bioactive Glass-Containing Universal Dentin Adhesive. J. Funct. Biomater. 2025, 16, 247. [Google Scholar] [CrossRef] [PubMed]
- Carrilho, E.; Cardoso, M.; Marques Ferreira, M.; Marto, C.M.; Paula, A.; Coelho, A.S. 10-MDP Based Dental Adhesives: Adhesive Interface Characterization and Adhesive Stability-A Systematic Review. Materials 2019, 12, 790. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhao, S.K.; Zhu, Z.Y.; Yu, J.; Yao, C.M.; Yu, M.Y.; Yang, H.Y.; Huang, C. Enhancing dentin bonding quality through Acetone wet-bonding technique: A promising approach. Front. Bioeng. Biotechnol. 2023, 11, 1309503. [Google Scholar] [CrossRef]
- Reis, A.; Pellizzaro, A.; Dal-Bianco, K. Impact of adhesive application to wet and dry dentin on long-term resin-dentin bond strengths. Oper. Dent. 2007, 32, 380–387. [Google Scholar] [CrossRef]
- Manso, A.P.; Marquezini, L.; Silva, S.M.A.; Pashley, D.H.; Tay, F.R.; Carvalho, R.M. Stability of wet versus dry bonding with different solvent-based adhesives. Dent. Mater. 2008, 24, 476–482. [Google Scholar] [CrossRef]
- Hari, K.; Sujatha, V.; Mahalaxmi, S.; Mathias, J.; Sagir, V. Shear Bond Strength of Composite to Dentin after Various Drying Techniques and Its Micro Morphological Analysis under SEM—An In-vitro Study. J. Conserv. Dent. 2012, 15, 258–262. [Google Scholar]
- Mahdan, M.H.A.; Talibe, A.; Shamsuddin, A.S.S.; Maideen, M.M. Ultrastructural Comparison between Wet and Dry Bonding Techniques of Two Different Solvent-Based Adhesives. J. Int. Dent. Med. Res. 2021, 14, 136–140. [Google Scholar]
- Perdigao, J.; Muñoz, M.; Sezinando, A.; Luque-Martinez, I.; Staichak, R.; Reis, A.; Loguercio, A. Immediate Adhesive Properties to Dentin and Enamel of a Universal Adhesive Associated With a Hydrophobic Resin Coat. Oper. Dent. 2013, 39, 489–499. [Google Scholar] [CrossRef] [PubMed]
- Muñoz, M.A.; Luque, I.; Hass, V.; Reis, A.; Loguercio, A.D.; Bombarda, N.H. Immediate bonding properties of universal adhesives to dentine. J. Dent. 2013, 41, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Chen, H.; Feng, S.; Jin, Y.; Hou, Y.; Zhu, S. Comparison of bond strength of universal adhesives using different etching modes: A systematic review and meta-analysis. Dent. Mater. J. 2022, 41, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Castro, A.S.; Maran, B.M.; Gutierrez, M.F.; Chemin, K.; Mendez-Bauer, M.L.; Bermúdez, J.P.; Reis, A.; Loguercio, A.D. Effect of Dentin Moisture in Posterior Restorations Performed with Universal Adhesive: A Randomized Clinical Trial. Oper. Dent. 2022, 47, e91–e105. [Google Scholar] [CrossRef] [PubMed]
- Hongye, Y.; Jingmei, G.; Deng, D.; Zhiyong, C.; Cui, H. Effect of adjunctive application of epigallocatechin-3-gallate and ethanol-wet bonding on adhesive-dentin bonds. J. Dent. 2016, 44, 44–49. [Google Scholar] [CrossRef]
- Caceres, S.; Ayala, G.; Alvítez-Temoche, D.; Suarez, D.; Watanabe, R.; Mayta-Tovalino, F. Bond Strength to Microtraction and Nanofiltration Using Ethanol Wet Bonding Technique in Fresh Extracted Teeth: An Ex Vivo Study. J. Int. Soc. Prev. Community Dent. 2020, 10, 466–472. [Google Scholar] [CrossRef]
- Angelo, E.; Guerra, A.; Vitti, R.; Fuentes, L.R.M.M.d. The Effect of Ethanol Associated with Universal Adhesive on Resin Composite Adhesion to Different Dentin Depths: A Long-Term In Vitro Study. Eur. J. Prosthodont. Restor. Dent. 2023, 31, 254–261. [Google Scholar] [CrossRef]
- Marcov, E.-C.; Burlibașa, M.; Marcov, N.; Căminișteanu, F.; Ștețiu, A.A.; Popescu, M.; Costea, R.-C.; Costea, R.M.; Burlibașa, L.; Drăguș, A.C.; et al. The Evaluation of Restored Proximal Contact Areas with Four Direct Adherent Biomaterials: An In Vitro Study. J. Funct. Biomater. 2025, 16, 128. [Google Scholar] [CrossRef]
- Anovazzi, G.; Mendes Soares, I.P.; Fernandes, L.d.O.; Gomes, L.N.; Scheffel, D.L.S.; Soares, D.G.; de Souza Costa, C.A.; Hebling, J. Water/ethanol and water/acetone proanthocyanidin solutions enhance the mechanical stability and sealing ability of resin-dentin bonds produced by the cross-linked dry bonding technique. Int. J. Adhes. Adhes. 2024, 132, 103736. [Google Scholar] [CrossRef]
- Furtado, C.; Baroudi, K.; Amaral, M.; Amorim, T.N.; Devito, K.L.; de Goes, M.F.; Salvio, L.A. Microtensile Bond Strength of Self-Etch Adhesives on Dentin Treated with Chlorhexidine. Eur. J. Prosthodont. Restor. Dent. 2024, 32, 133–141. [Google Scholar] [CrossRef] [PubMed]
- Kanca, J. Resin bonding to wet substrate I: Bonding to dentin. Quintessence Int. 1992, 23, 39–41. [Google Scholar] [PubMed]
- Burrer, P.; Dang, H.; Par, M.; Attin, T.; Tauböck, T.T. Effect of Over-Etching and Prolonged Application Time of a Universal Adhesive on Dentin Bond Strength. Polymers 2020, 12, 2902. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Breschi, L.; Mazzoni, A.; Ruggeri, A.; Cadenaro, M.; Di Lenarda, R.; De Stefano Dorigo, E. Dental adhesion review: Aging and stability of the bonded interface. Dent. Mater. Off. Publ. Acad. Dent. Mater. 2008, 24, 90–101. [Google Scholar] [CrossRef] [PubMed]
- De Munck, J.; Van Landuyt, K.; Peumans, M.; Poitevin, A.; Lambrechts, P.; Braem, M.; Van Meerbeek, B. A critical review of the durability of adhesion to tooth tissue: Methods and results. J. Dent. Res. 2005, 84, 118–132. [Google Scholar] [CrossRef] [PubMed]
- Van Meerbeek, B.; Yoshihara, K.; Yoshida, Y.; Mine, A.; De Munck, J.; Van Landuyt, K.L. State of the art of self-etch adhesives. Dent. Mater. Off. Publ. Acad. Dent. Mater. 2011, 27, 17–28. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y.; Yoshihara, K.; Nagaoka, N.; Hayakawa, S.; Torii, Y.; Ogawa, T.; Osaka, A.; Meerbeek, B.V. Self-assembled Nano-layering at the Adhesive interface. J. Dent. Res. 2012, 91, 376–381. [Google Scholar] [CrossRef] [PubMed]
- Hebling, J.; Pashley, D.H.; Tjäderhane, L.; Tay, F.R. Chlorhexidine arrests subclinical degradation of dentin hybrid layers in vivo. J. Dent. Res. 2005, 84, 741–746. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.; Tan, J.; Chen, L.; Li, D.; Tan, Y. The incorporation of chlorhexidine in a two-step self-etching adhesive preserves dentin bond in vitro. J. Dent. 2009, 37, 807–812. [Google Scholar] [CrossRef]
- Alomran, W.K.; Nizami, M.Z.I.; Xu, H.H.K.; Sun, J. Evolution of Dental Resin Adhesives-A Comprehensive Review. J. Funct. Biomater. 2025, 16, 104. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Zhou, H.; Yuan, Y.; Luo, C.; Wang, Q.; Li, Z.; Chen, M.; Gong, B.; Li, Z. The role of oral environmental factors in the degradation of resin-dentin interfaces: A comprehensive review. J. Dent. 2025, 159, 105839. [Google Scholar] [CrossRef] [PubMed]
- Jayaprakash, T.; Srinivasan, M.R.; Indira, R. Evaluation of the effect of surface moisture on dentinal tensile bond strength to dentine adhesive: An in vitro study. J. Conserv. Dent. Endod. 2010, 13, 116–118. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Stape, T.H.S.; Uctasli, M.; Cibelik, H.S.; Tjäderhane, L.; Tezvergil-Mutluay, A. Dry bonding to dentin: Broadening the moisture spectrum and increasing wettability of etch-and-rinse adhesives. Dent. Mater. 2021, 37, 1676–1687. [Google Scholar] [CrossRef] [PubMed]
- Choi, A.N.; Lee, J.H.; Son, S.A.; Jung, K.H.; Kwon, Y.H.; Park, J.K. Effect of Dentin Wetness on the Bond Strength of Universal Adhesives. Materials 2017, 10, 1224. [Google Scholar] [CrossRef]
- Kumagai, R.Y.; Hirata, R.; Pereira, P.N.R.; Reis, A.F. Moist vs over-dried etched dentin: FE-SEM/TEM and bond strength evaluation of resin-dentin interfaces produced by universal adhesives. J. Esthet. Restor. Dent. 2020, 32, 325–332. [Google Scholar] [CrossRef]
- Page, M.J.; McKenzie, J.E.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E.; et al. The PRISMA 2020 statement: An updated guideline for reporting systematic reviews. BMJ (Clin. Res. ed.) 2021, 372, n71. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Slim, K.; Nini, E.; Forestier, D.; Kwiatkowski, F.; Panis, Y.; Chipponi, J. Methodological index for non-randomized studies (minors): Development and validation of a new instrument. ANZ J. Surg. 2003, 73, 712–716. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Huang, L.; Wang, D.; Ren, P.; Hong, Q.; Kang, D. The ROBINS-I and the NOS had similar reliability but differed in applicability: A random sampling observational studies of systematic reviews/meta-analysis. J. Evid. Based Med. 2021, 14, 112–122. [Google Scholar] [CrossRef] [PubMed]
- Cumpston, M.; Li, T.; Page, M.J.; Chandler, J.; Welch, V.A.; Higgins, J.P.; Thomas, J. Updated guidance for trusted systematic reviews: A new edition of the Cochrane Handbook for Systematic Reviews of Interventions. Cochrane Database Syst. Rev. 2019, 10, Ed000142. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Leite, M.; Charamba, C.D.F.; Lima, R.; Meireles, S.; Duarte, R.; Andrade, A.K.M. Bond strength of universal adhesive applied to dry and wet dentin. Braz. J. Oral Sci. 2020, 19, e201662. [Google Scholar] [CrossRef]
- Nonato, R.; Moreira, P.; Silva, D.; Ferreira, M.; Reis, A.; Cardenas, A.; Loguércio, A.; Siqueira, F. Long-term Evaluation of Bonding Performance of Universal Adhesives based on Different Dentinal Moisture Levels. J. Adhes. Dent. 2022, 24, b3559027. [Google Scholar] [CrossRef]
- Saeed, N.A.; Tichý, A.; Yusuke, K.; Hosaka, K.; Tagami, J.; Nakajima, M. Effect of Surface Moisture on Bur-cut Dentin on Bonding of HEMA-free and HEMA-containing Universal Adhesives with or without Methacrylamide Monomer. J. Adhes. Dent. 2021, 23, 327. [Google Scholar] [CrossRef] [PubMed]
- Ekambaram, M.; Yiu, C.; Matinlinna, J.; King, N.; Tay, F. Adjunctive application of chlorhexidine and ethanol-wet bonding on durability of bonds to sound and caries-affected dentine. J. Dent. 2014, 42, 709–719. [Google Scholar] [CrossRef] [PubMed]
- Elbanna , A.A.; El-Toukhy, R.I.; Abbas, M.; Zaghloul, N.M. Effect of Dimethyl Sulfoxide Primer on Microtensile Bond Strength and Micromorphological Pattern of HEMA-free Universal Adhesive to Dry/Wet Dentin After Thermomechanical Aging. J. Clin. Exp. Dent. 2024, 16, e323. [Google Scholar] [CrossRef]
- Hamouda, M.A.; Kandil, M.M.; Nassif, M. Effect of dentin moisture and bonding strategies on micro-tensile bond strength using two chemically different universal adhesives. Ain Shams Dent. J. 2022, 28, 1–12. [Google Scholar] [CrossRef]
- Faria-e-Silva, A.L.; Fabião, M.M.; Sfalcin, R.A. Bond strength of one-step adhesives under different substrate moisture. Int. J. Dent. 2009, 20, 227–233. [Google Scholar] [CrossRef]
- Popescu, M.; Perieanu, V.S.; Burlibașa, M.; Vorovenci, A.; Malița, M.A.; Petri, D.-C.; Ștețiu, A.A.; Costea, R.C.; Costea, R.M.; Burlibașa, A.; et al. Comparative Cost-Effectiveness of Resin 3D Printing Protocols in Dental Prosthodontics: A Systematic Review. Prosthesis 2025, 7, 78. [Google Scholar] [CrossRef]
- Marcov, E.; Burlibasa, M.; Marcov, N.; Costea, R.; Voinescu, I.; Popescu, M.; Costea, R.-C.; Bodnar, D. In vitro Comparison of the Efficiency of Celluloid and Metallic Matrices in Proximal Restorations with a Bulk Polymer-based Biomaterial. Mater. Plast. 2024, 61, 1–14. [Google Scholar] [CrossRef]
- Zhao, X.-Y. Bonding Strength and Interface Effects of Different Dentin Bonding Surfaces on Alcohol-water-based Adhesives. Ph.D. Thesis, University of Luxembourg, Luxembourg, 2004. [Google Scholar]
- Albaladejo, A.; Osorio, R.; Toledano, M.; Ferrari, M. Hybrid layers of etch-and-rinse versus self-etching adhesive systems. Med. Oral Patol. Oral Cir. Bucal 2010, 15, e112–e118. [Google Scholar] [CrossRef] [PubMed]
- Takahashi, R.; Nikaido, T.; Tagami, J.; Hickel, R.; Kunzelmann, K.-H. Contemporary adhesives: Marginal adaptation and microtensile bond strength of class II composite restorations. Am. J. Dent. 2012, 25, 181–188. [Google Scholar]
- Lőrincz, H.; Bardocz-Veres, Z.; Strnad, G.; Kerekes-Máthé, B. In Vitro Study of Mechanical Properties of Teeth Restored with Bulk-fill and Universal Composites Using Different Dentin Adhesives. J. Interdiscip. Med. 2022, 7, 38–43. [Google Scholar] [CrossRef]
- Elkaffas, A.A.; Hamama, H.H.H.; Mahmoud, S.H. Do universal adhesives promote bonding to dentin? A systematic review and meta-analysis. Restor. Dent. Endod. 2018, 43, e29. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Hardan, L.; Bourgi, R.; Kharouf, N.; Davide, M.; Zarow, M.; Jakubowicz, N.; Haikel, Y.; Cuevas Suárez, C. Bond Strength of Universal Adhesives to Dentin: A Systematic Review and Meta-Analysis. Polymers 2021, 13, 814. [Google Scholar] [CrossRef] [PubMed]
- Forville, H.; Wendlinger, M.; Cochinski, G.D.; Favoreto, M.W.; Wambier, L.M.; Chibinski, A.C.; Loguercio, A.D.; Reis, A. Re-evaluating the role of moist dentin of adhesive systems used in the etch-and-rinse bonding strategy. A systematic review and meta-analysis. J. Dent. 2025, 152, 105495. [Google Scholar] [CrossRef] [PubMed]
- Tsujimoto, A.; Barkmeier, W.W.; Takamizawa, T.; Watanabe, H.; Johnson, W.W.; Latta, M.A.; Miyazaki, M. Relationship between mechanical properties and bond durability of short fiber-reinforced resin composite with universal adhesive. Eur. J. Oral Sci. 2016, 124, 480–489. [Google Scholar] [CrossRef] [PubMed]
- Takamizawa, T.; Barkmeier, W.W.; Tsujimoto, A.; Berry, T.P.; Watanabe, H.; Erickson, R.L.; Latta, M.A.; Miyazaki, M. Influence of different etching modes on bond strength and fatigue strength to dentin using universal adhesive systems. Dent. Mater. Off. Publ. Acad. Dent. Mater. 2016, 32, e9–e21. [Google Scholar] [CrossRef] [PubMed]
- Li, X.-R.; Deng, Q.-S.; Liu, P.-L.; He, S.-H.; Gao, Y.; Wei, Z.-Y.; Zhang, C.-R.; Wang, F.; Dou, X.-Q.; Dawes, H.; et al. Three-dimensional printed biomimetic multilayer scaffolds coordinated with sleep-related small extracellular vesicles: A strategy for extracellular matrix homeostasis and macrophage polarization to enhance osteochondral regeneration. View 2024, 5, 20230069. [Google Scholar] [CrossRef]
- Shen, L.; Ye, X.; Wu, S.; Yu, J.; Chen, H.; Zhang, S.; Wang, C.; Jiang, S.; Mao, J.; He, S.; et al. Evaluation of the inner retinal layers following traditional and modified ILM peeling surgery using automatic CMDS quantification algorithm. View 2023, 4, 20220061. [Google Scholar] [CrossRef]

| PICO Element | Definition |
|---|---|
| Population (P) | Extracted human dentin from permanent teeth (primarily third molars) prepared for composite bonding. |
| Intervention (I) | Wet-bonding techniques, including water-wet-, ethanol-wet-, and acetone-wet-bonding protocols. |
| Comparison (C) | Conventional dry-bonding techniques (air-dried dentin without intentional rewetting). |
| Outcome (O) | Primary: Microtensile bond strength (μTBS, in MPa). Secondary: Adhesive interface quality (e.g., nanoleakage, hybrid layer integrity, and long-term durability). |
| Study | Study Design; Main Measurement | Sample Size | Bonding Technique | Measurement Methods |
|---|---|---|---|---|
| Leite et al., 2020 [45] | In vitro experimental; microtensile bond strength (TBS) | human third molars | Wet vs. dry bonding (Scotchbond Universal, Adper Single Bond 2) | Microtensile bond strength at 24 h, 1 year; t-test; Weibull analysis. |
| Saeed et al., 2021 [47] | In vitro experimental; microtensile bond strength | human third molars | Wet vs. dry; multiple adhesives (2-hydroxyethyl methacrylate (HEMA) containing/HEMA- free) | Microtensile bond strength at 24 h, after thermocycling; three-way analysis of variance. |
| Elbanna et al., 2024 [49] | In vitro experimental; microtensile bond strength | mandibular first molars (microtensile bond strength); 16 maxillary premolars (morphology) | Wet/dry; dimethyl sulfoxide (DMSO) pretreatment; Gluma Bond Universal | Microtensile bond strength at 24 h, after thermomechanical aging; three-way analysis of variance; environmental scanning electron microscopy (ESEM). |
| Nonato et al., 2022 [46] | In vitro experimental; microtensile bond strength | 378 human molars | Dry, wet, oversaturated; three adhesives; etch-and-rinse (ER)/self-etch (SE) | Microtensile bond strength, SNU at 24 h, 1 year; four-way analysis of variance; scanning electron microscopy (SEM), Fourier-transform infrared spectroscopy (FTIR). |
| Choi et al., 2017 [39] | In vitro experimental; microtensile bond strength | 72 human third molars | Etch-and-rinse/self-etch; 0, 5, 10 s air-drying; three adhesives | Microtensile bond strength at 24 h, two-way analysis of variance, confocal laser scanning microscopy (CLSM). |
| Hamouda et al., 2022 [50] | In vitro experimental; microtensile bond strength | 60 human third molars | Etch-and-rinse/self-etch; dry, wet, moist; two adhesives | Microtensile bond strength at 24 h, three-way analysis of variance, scanning electron microscopy. |
| Caceres et al., 2020 [22] | Experimental ex vivo; microtensile bond strength | 48 premolars | Water vs. ethanol wet bonding; Single Bond 2 | Microtensile bond strength (immediate), t-test. |
| Zhao et al., 2023 [12] | In vitro experimental; microtensile bond strength | 60 third molars | Water/ethanol/acetone wet bonding; Singlebond Universal | Microtensile bond strength at 24 h, after thermocycling and collagenase aging; two-way analysis of variance. |
| Ekambaram et al., 2014 [48] | In vitro experimental; microtensile bond strength | 48 molars (caries affected) | Ethanol/water wet bonding with or without chlorhexidine; hydrophobic adhesive | Microtensile bond strength at 24 h, 12 months; three-way analysis of variance. |
| Study | Year | Clearly Stated Aim | Inclusion of Consecutive Specimens | Prospective Data Collection | Endpoints Appropriate | Unbiased Assessment | Adequate Follow-Up | <5% Loss to Follow-Up | Sample Size Calculation | Total (/16) |
|---|---|---|---|---|---|---|---|---|---|---|
| Elbanna et al. [49] | 2024 | 2 | 2 | 2 | 2 | 1 | 2 | 2 | 2 | 15 |
| Caceres et al. [22] | 2020 | 2 | 2 | 2 | 2 | 1 | 2 | 2 | 2 | 15 |
| Choi et al. [39] | 2017 | 2 | 1 | 1 | 2 | 1 | 2 | 2 | 0 | 11 |
| Ekambaram et al. [48] | 2014 | 2 | 1 | 1 | 2 | 1 | 2 | 2 | 0 | 11 |
| Leite et al. [45] | 2020 | 2 | 1 | 2 | 2 | 2 | 2 | 2 | 2 | 15 |
| Hamouda et al. [50] | 2022 | 2 | 2 | 2 | 2 | 1 | 2 | 2 | 1 | 14 |
| Saeed et al. [47] | 2021 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 16 |
| Zhao et al. [12] | 2023 | 2 | 2 | 2 | 2 | 1 | 2 | 2 | 1 | 14 |
| Nonato et al. [46] | 2022 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 2 | 16 |
| Outcome | No. of Studies | Study Design | Risk of Bias | Inconsistency | Indirectness | Imprecision | Publication Bias | Certainty of Evidence |
|---|---|---|---|---|---|---|---|---|
| Microtensile Bond Strength (μTBS) | 9 | In vitro experimental | Not serious | Serious | Very serious | Serious | Unclear | Very low |
| Nanoleakage | 6 | In vitro experimental | Not serious | Serious | Very serious | Serious | Unclear | Very low |
| Hybrid Layer Degradation | 4 | In vitro experimental | Not serious | Serious | Very serious | Serious | Unclear | Very low |
| Long-term Durability | 7 | In vitro experimental | Not serious | Serious | Very serious | Serious | Unclear | Very low |
| Adhesive | Condition | μTBS (MPa ± SD) | Interpretation |
|---|---|---|---|
| Prime&Bond Universal (P&BU) | Dry | 47.6 ± 7.1 | Favored dry |
| Prime&Bond Universal (P&BU) | Moist | 36.2 ± 6.4 | Lower |
| Scotchbond Universal (SBU) | Dry | 33.9 ± 6.1 | Lower |
| Scotchbond Universal (SBU) | Moist | 41.8 ± 6.7 | Favored moist |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Popescu, M.; Malița, M.; Vorovenci, A.; Ștețiu, A.A.; Perieanu, V.Ș.; Costea, R.C.; David, M.; Costea, R.M.; Ștețiu, M.A.; Drăguș, A.C.; et al. Wet vs. Dry Dentin Bonding: A Systematic Review and Meta-Analysis of Adhesive Performance and Hybrid Layer Integrity. Oral 2025, 5, 63. https://doi.org/10.3390/oral5030063
Popescu M, Malița M, Vorovenci A, Ștețiu AA, Perieanu VȘ, Costea RC, David M, Costea RM, Ștețiu MA, Drăguș AC, et al. Wet vs. Dry Dentin Bonding: A Systematic Review and Meta-Analysis of Adhesive Performance and Hybrid Layer Integrity. Oral. 2025; 5(3):63. https://doi.org/10.3390/oral5030063
Chicago/Turabian StylePopescu, Mircea, Mădălina Malița, Andrei Vorovenci, Andreea Angela Ștețiu, Viorel Ștefan Perieanu, Radu Cătălin Costea, Mihai David, Raluca Mariana Costea, Maria Antonia Ștețiu, Andi Ciprian Drăguș, and et al. 2025. "Wet vs. Dry Dentin Bonding: A Systematic Review and Meta-Analysis of Adhesive Performance and Hybrid Layer Integrity" Oral 5, no. 3: 63. https://doi.org/10.3390/oral5030063
APA StylePopescu, M., Malița, M., Vorovenci, A., Ștețiu, A. A., Perieanu, V. Ș., Costea, R. C., David, M., Costea, R. M., Ștețiu, M. A., Drăguș, A. C., Șerbănescu, C. M., Burlibașa, A., Eftene, O., & Burlibașa, M. (2025). Wet vs. Dry Dentin Bonding: A Systematic Review and Meta-Analysis of Adhesive Performance and Hybrid Layer Integrity. Oral, 5(3), 63. https://doi.org/10.3390/oral5030063





