Association Between Diabetes Mellitus and Head and Neck Cancer: An Umbrella Review of Systematic Reviews and Meta-Analyses
Abstract
1. Introduction
2. Materials and Methods
2.1. Literature Search
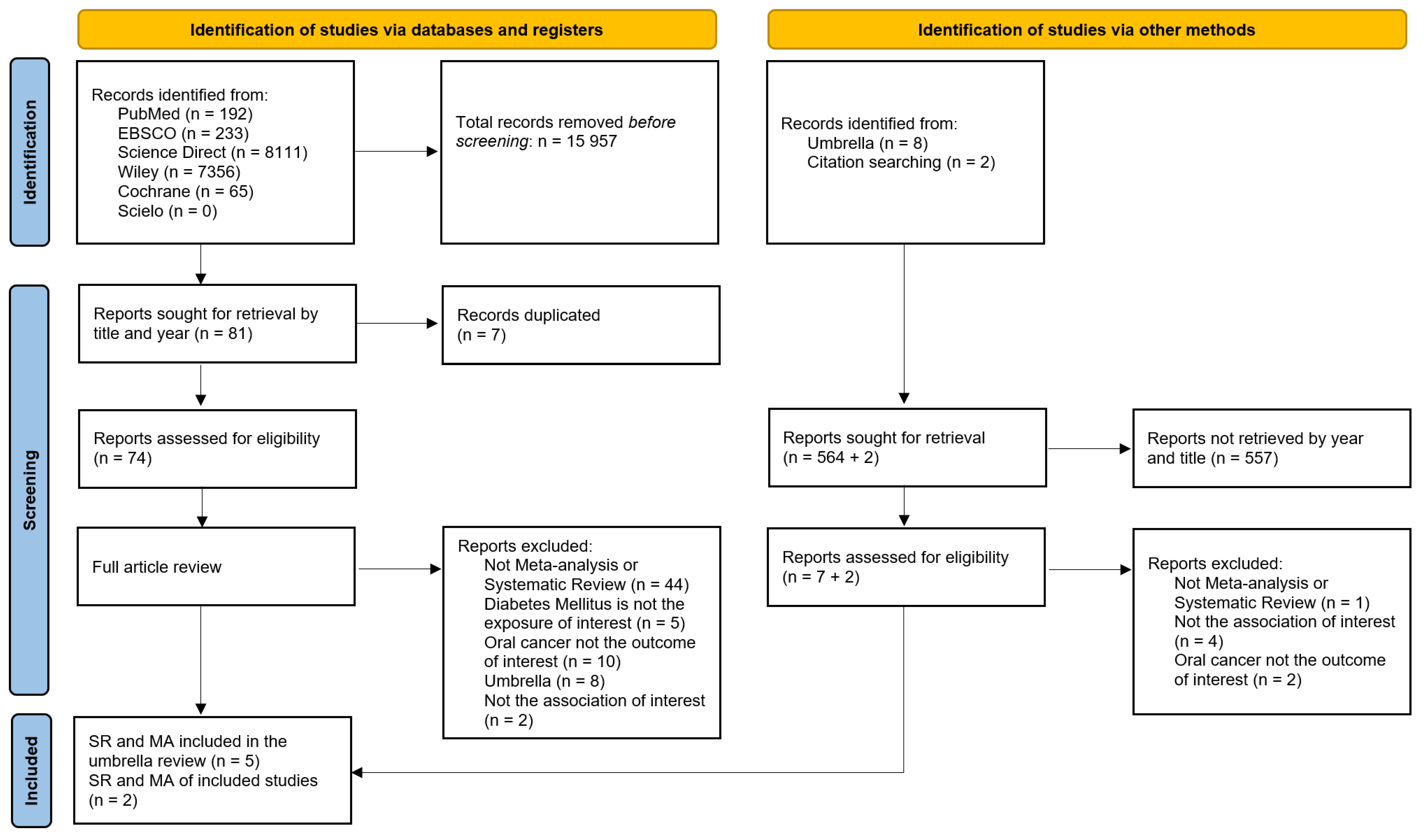
2.2. Selection of Studies and Eligibility
2.3. Data Extraction
2.4. Risk of Bias
2.5. Strategy for Data Synthesis
3. Results
3.1. Study Characteristics
3.2. Quantitative Evaluation
| Authors (Year) | Population | Outcome | Sample Size (Patients) | Guidelines | PROSPERO Code | Search Strategy (Date) | Eligibility Criteria Restrictions |
|---|---|---|---|---|---|---|---|
| [14] Gong et al. (2015) | Patients with diabetes mellitus type 2 | Oral cancer and precancerous lesions | 17 studies total; 13 for oral cancer with 4,8 million participants and 6465 cases with oral cancer | MOOSE | ND | MEDLINE and EMBASE through 31 May 2014 | (a) original data from case–control or cohort studies; (b) exposure was type 2 DM; (c) outcome was oral cancers or precancerous lesions; (d) studies should report either adjusted OR, RR, hazard ratio (HR), or standardized incidence/mortality ratios (SIR/SMR) with their 95% Cis (or data to calculate them). |
| [18] Sona et al. (2018) | Type 1 Diabetes mellitus patients | Various types of cancer including head and neck | 15 observational studies with two case–control studies and 13 cohort studies from 11 articles | ND | ND | PubMed and EMBASE in April 2017 | (a) case–control studies and prospective or retrospective cohort studies; (b) association between type 1 diabetes and the risk of cancer; (c) reporting measures of outcomes with adjusted odds ratios (Ors) or relative risks (RRs) and 95% confidence intervals (Cis); (d) data identical in more than one study or duplicated, the more comprehensive or first published study was included in the analysis. |
| [19] Ohkuma et al. (2018) | Diabetes of type 1 or type 2 | Cancer, overall and by site | 108 studies total, 19 million participants | PRISMA | ND | PubMed on 23 December 2016 | (a) observational cohort studies if they had provided RR; (b) exclusion if they had not adjusted at least for age or did not provide information about the variability around the point estimate or if they only had data for one sex; (c) in duplicate reports from the same study, the one with the longest follow-up or the highest number of cases was included. |
| [20] Ramos-Garcia et al. (2020) | Diabetes of type 1 or type 2 | Oral cancer or oral potentially malignant disorders | 52 studies, 559,927 participants | PRISMA | CRD42020162848 | PubMed; Embase, Web of Science; Scopus; until November 2019 | (a) original studies, without language, publication date, sex, or age restrictions; (b) oral cancer in type 1 or type 2 DM; (c) cohort, case–control and cross-sectional studies; (d) exclusion of animal research or in vitro studies; (e) results from the same study where included the most recent or with more data. |
| [21] Yan et al. (2021) | Diabetes mellitus type 2 | Head and neck cancer subtypes | 27 studies, 1.4 million T2DM cases and 23,045 HNC cases | PRISMA | ND | PubMed, Web of Science and Embase, on 31 July 2020 | (a) cohort or case–control study; (b) exposure of T2DM; (c) primary outcome was incidence of HNC, oral cancer, pharyngeal cancer, or laryngeal cancer; (d) reported the risk estimates OR, standard incidence ratio (SIR), RR or hazard ratio (HR), and 95% confidence intervals; (e) when multiple articles come from the same, we only include the longest duration of follow-up. |
| [22] Dos Santos et al. (2022) | Patients with systemic conditions | Oral squamous cell carcinoma | 95 studies total, 14,442,487 participants | PRISMA—PECOS | CRD42021242702 | PubMed; Scopus; Web of Science; Embase; Google Scholar; OpenGrey; ProQuest Dissertation & Theses Global; on 27 February 2021 and updated on 19 February 2022 | (a) English articles; (b) with systemic conditions; (c) systemic conditions associated with increased risk to develop OSCC; (d) observational studies. |
| [23] Gormley et al. (2022) | Metabolic disorders | Head and neck cancer | 36 studies total; 15 between diabetes type 2 and HNC | PRISMA | CRD42021250520 | January 1966 to November 2021, including Cochrane Library, OVID SP versions of Medline and EMBASE; Cochrane Library, EthOS, Google Scholar, Open Grey and ClinicalTrials.gov (gray literature) | (a) participants over 18 years old, of either sex, from any ethnic background; (b) with T2D, obesity, dyslipidemia or hypertension; (c) outcome HNSCC which may be human papilloma virus (HPV) positive or negative and high-risk types HPV16, 18, 31 and 33 only will be included; (d) All studies published from January 1966 in the English language; (e) observational with head and neck squamous cell carcinoma; (f) with OR or risk ratio, or data which will allow to calculate |
| Authors (Year) | Diagnostic Criteria for Inclusion | Time Range of the Studies Included | Risk of Bias | Meta-Analysis | Risk Factors | Funding |
|---|---|---|---|---|---|---|
| [14] Gong et al. (2015) | ND | 1994 to 2013 | Funnel plots and the further Begg’s adjusted rank correlation and Egger’ regression asymmetry tests | Yes | Cigarette smoking, alcohol consumption, betel-quid chewing and some types of viral infections | ND |
| [18] Sona et al. (2018) | ND | 1965 to 2014 | funnel plots and the further Begg’s adjusted rank correlation and Egger’ regression asymmetry tests | Yes | Age; sex and age at onset of diabetes; alcohol; tobacco consumption; history of hepatitis; liver cirrhosis; BMI; history of cancer in first degree relatives; calendar year at follow-up; duration of diabetes and its status; period of diagnosis; socioeconomic status; region | ND |
| [19] Ohkuma et al. (2018) | ND | 1982 to 2016 | Funnel plots and Egger’s and Begg’s tests | Yes | ND | ND |
| [20] Ramos-Garcia et al. (2020) | ND | 1983 to 2016 | Joanna Briggs Institute, University of Adelaide, Australia; QUIPS | Yes | ND | ND |
| [21] Yan et al. (2021) | ND | 1994 to 2018 | Egger’s linear regression test and Begg’s test; | Yes | Smoking, alcohol use, or BMI/obesity. | ND |
| [22] Dos Santos et al. (2022) | ND | 1961 to 2022 | Joanna Briggs Institute; In the Diabetes Mellitus study the risk of Bias is LOW | Yes | Having systemic conditions | ND |
| [23] Gormley et al. (2022) | Symptoms such as polyuria or polydipsia, plus: A random blood plasma glucose concentration ≥ 11.1 mmol/L; A fasting plasma glucose concentration ≥ 7.0 mmol/L (whole blood ≥ 6.1 mmol/L); Two-hour plasma glucose concentration ≥ 11.1 mmol/L, two hours after 75 g anhydrous glucose in an oral glucose tolerance test; Glycated hemoglobin (HbA1c) 6.5% or more (48 mmol/mol and above). | 1992 to 2021 | ROBINS-E | Yes | Smoking, alcohol and presence of human papillomavirus | ND |
| Authors (Year) | Association Between Diabetes and the Outcome: | Nº of Studies | Nº of Cases/Population | Countries in Each Revision |
|---|---|---|---|---|
| [14] Gong et al. (2015) | Oral cancer or precancerous lesions | 17 studies total; 13 for oral cancer and 4 for precancerous lesions | More than 4.8 million patients and 6465 cases of oral cancer; 1407 cases with oral precancerous lesions (1137 with leukoplakia, 100 with erythroplakia and 170 with submucous fibrosis) | USA, Hungary, India, Poland, Germany, Italy, Japan, Switzerland, Denmark, England, Taiwan |
| [18] Sona et al. (2018) | All types of cancer, including head and neck | 15 total, 2 with buccal cancer | 31,893 cancer patients among a total of 1,915,179 participants | Sweden (n = 3), Australia (n = 2), UK (n = 2), Denmark (n = 2), Italy (n = 2), Finland (n = 1), Scotland (n = 1), US (n = 1) and Taiwan (n = 1) |
| [19] Ohkuma et al. (2018) | Lip, oral cavity, pharynx, head and neck, nasopharynx and oral cancer; and sex differences | 108 total; 3 with lip, oral cavity and pharynx; 3 with head and neck; 2 with nasopharynx; 2 with oral cancer | 108 total; 3 with lip, oral cavity and pharynx; 3 with head and neck; 2 with nasopharynx; 2 with oral cancer | USA, Japan, Italy, Denmark, Australia, Finland, Scotland, Sweden, Germany, Austria, Korea, China, Taiwan, Israel, UK, New Zealand, Singapore, Spain, Netherlands, Poland, France, Mauritius, Fiji, Nauru |
| [20] Ramos-Garcia et al. (2020) | Oral cancer in general or oral potentially malignant disorders | 52; 28 studies only evaluated cancer; 23 reported data about prevalence and risk of oral cavity and oropharynx cancer | 559,927 | Asia (n = 21), Europe (n = 20), North America (n = 6), South America (n = 4) and Global Multicontinent (n = 1) |
| [21] Yan et al. (2021) | Oral cavity; pharynx; larynx; oral; head and neck general; specific squamous cell carcinoma; Nasopharyngeal | 27 total; all with DM2 | 1.4 million T2DM cases and 23,045 HNC cases in 15 studies; 28,451 HNC cases in the remaining 12 studies | Europe (n = 11), East Asia (n = 8), North America (n = 6), and Brazil (n = 1) |
| [22] Dos Santos et al. (2022) | Oral squamous cell carcinoma | 86 for qualitative synthesis; 9 for quantitative synthesis; 1 study with the diabetes mellitus condition | 1,442,487 patients with systemic conditions; 89,089 with diabetes mellitus; Tseng: n(DM) = 89,530 and n(without DM) = 89,530 | Japan, United States, Denmark, Taiwan and South Korea. |
| [23] Gormley et al. (2022) | Head and neck squamous cell carcinoma | 36 total, 15 with diabetes mellitus type 2 | 39,002 total; 17,582 cases with diabetes mellitus | Sweden, UK, USA, Netherland, Other European Countries, Denmark, Israel, Taiwan, Scotland, Japan, Finland, Korea, China, Hawaii |
| Authors (Year) | Association Between Diabetes Mellitus and the Outcome | Random Effects | Largest Study of Our Outcome | Random p-Value | Egger’s p-Value | I2; P-Value | Evidence of Reclassification (I-IV) | AMSTAR |
|---|---|---|---|---|---|---|---|---|
| [14] Gong et al. (2015) | oral cancer | SRR = 1.15 (95% CI: 1.02–1.29) | ORLS1 = 1.04 (0.63–1.63); ORLS2 = 0.95 (0.43–1.84) | p = 0.277 | Egger’s p = 0.176; Begg’s p = 0.392 | p = 0.277, I2 = 15.4% | The quality scores ranged from 5 to 9, with the median score 7. Most of the included studies (13/17) were of high quality (NOS score ≥ 7). | Newcastle–Ottawa quality assessment Scale (NOS) |
| [18] Sona et al. (2018) | buccal cancer | OR or RR = 1.79 (95% CI: 0.96–3.36) | ND | ND | p = 0.218 | I2 = 0.0% | One of the studies was qualified as Low and another one High | Newcastle–Ottawa Scale (NOS) |
| [19] Ohkuma et al. (2018) | lip, oral cavity, pharynx cancer | RRR = 0.94 (95% CI: 0.68–1.32); | Oral OR = 1.13 (1.00–1.28) | ND | Egger’s p = 0.13; Begg’s p = 0.16 | ND | Lower score (<7 points) = 1.02 (0.97–1.07) Higher score (≥7 points) = 1.07 (1.04–1.10) | Newcastle–Ottawa Quality Assessment Scale (NOS) |
| head and neck cancer | RRR = 1.01 (95% CI: 0.59–1.72); | ND | ND | |||||
| nasopharynx cancer | RRR = 1.04 (95% CI: 0.92–1.18); | ND | ND | |||||
| oral cancer | RRR = 1.13 (95% CI: 1.00–1.28) | p = 0.009 | ND | |||||
| [20] Ramos-Garcia et al. (2020) | oral cancer | OR = 1.32 (95% CI: 1.12–1.56) I2 = 75.5% | Mixed OR = 1.21 (0.95–1.53) | p < 0.001 | p = 0.492 | I2 = 75.5%; p < 0.001 | “low” quality of evidence for the association between oropharyngeal cancer development and diabetes; and “very low” quality of evidence for the rest of analyzed outcomes. The most influential domain was “inconsistency,” mainly due to the high heterogeneity found. | GRADE |
| oral squamous cell carcinoma | OR = 1.41 (95% CI: 1.1–1.81) I2 = 83.2% | p < 0.001 | ||||||
| oropharyngeal cancer | OR = 1.17 (95% CI: 0.82–1.65) I2 = 32.2% | p = 0.229 | ||||||
| mixed | OR = 1.21 (95% CI: 0.95;1.53) I2 = 41.7% | p = 0.127 | ||||||
| [21] Yan et al. (2021) | head and neck cancer | RR = 1.04 (95% CI: 0.88–1.23); p = 0.635; | head and neck squamous cell carcinoma: OR = 0.92 (0.88–0.96) | p < 0.10 | Egger’s p = 0.437; Begg’s p = 0.951 | p < 0.001; I2 = 83.2% | ND | Newcastle–Ottawa Quality Assessment Scale (NOS) |
| oral cancer | RR = 1.28, (95% CI, 1.04–1.58); | p < 0.05 | ND | |||||
| oropharyngeal cancer | RR = 1.18, (95% CI, 1.02–1.37) | p < 0.05 | ND | |||||
| [22] Dos Santos et al. (2022) | head and neck cancer | HR = 1.47 (95% CI: 1.30–1.66) * | ND | p < 0.05 | ND | p < 0.01, I2 = 95% | 87%, low risk of bias | Joanna Briggs Institute |
| oral cancer | HR = 1.73 (95% CI: 1.46–2.05) * | p < 0.05 | ||||||
| oropharyngeal cancer | HR = 1.53 (95% CI: 1.01–2.31) * | p < 0.05 | ||||||
| nasopharyngeal carcinoma | OR = 1.40 (95% CI: 1.03–1.90) * | p < 0.05 | ||||||
| [23] Gormley et al. (2022) | overall cancer | RRoverall = 1.13, (95% CI: 0.95–1.34); RRfixed effects = 1.22, (95% CI: 1.16- 1.29) | OR = 1.21 (1.13–1.30) | p < 0.001 | ND | p < 0.0001, I2 = 80.0% | One study was rated as ‘Very high’ risk of bias (3.7%), 21 studies were rated as ‘High’ (77.8%), one study as ‘Some concerns’ (3.7%) and the remaining studies as ‘Low’ (14.8%) risk of bias | ROBINS-E tool |
| oral cancer | RR = 1.13, (95% CI: 0.97–1.31) | OR = 1.10 (0.98–1.24) | ND | |||||
| oropharyngeal cancer | RR = 1.16, (95% CI: 0.73–1.83) | OR = 0.95 (0.84–1.08) | ND |
3.3. Risk of Bias
3.4. Strategy for Data Synthesis
| [14] Gong et al. (2015) | [18] Sona et al. (2018) | [19] Ohkuma et al. (2018) | [20] Ramos-Garcia et al. (2020) | [21] Yan et al. (2021) | [22] Dos Santos et al. (2022) | [23] Gormley et al. (2022) | |
|---|---|---|---|---|---|---|---|
| Is the review question clearly and explicitly stated? | Met | Met | Met | Met | Met | Met | Met |
| Were the inclusion criteria appropriate for the review question? | Met | Met | Met | Met | Met | Met | Met |
| Was the search strategy appropriate? | Met | Met | Met | Met | Met | Met | Met |
| Were the sources and resources used to search for studies adequate? | Met | Met | Met | Met | Met | Met | Met |
| Were the criteria for appraising studies appropriate? | Met | Met | Met | Met | Met | Met | Met |
| Was critical appraisal conducted by two or more reviewers independently? | Met | Met | Unclear | Met | Met | Met | Met |
| Were the methods used to combine studies appropriate? | Met | Met | Met | Met | Met | Met | Met |
| Was the likelihood of publication bias assessed? | Met | Met | Met | Met | Met | Met | Met |
| Were recommendations for policy and/or practice supported by the reported data? | Met | Unclear | Met | Met | Unclear | Met | Met |
| Were the specific directives for new research appropriate? | Met | Not met | Met | Met | Unclear | Not Met | Not met |
| Include? | Yes | Yes | Yes | Yes | Yes | Yes | Yes |
| Low, Moderate or High quality? | High | High | High | High | High | High | High |
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| AMSTAR | Assessing the Methodological Quality of Systematic Reviews |
| BMI | Body Mass Index |
| DM2 | Diabetes Mellitus 2 |
| DNA | Deoxyribonucleic Acid |
| GDM | Gestational Diabetes Mellitus |
| GRADE | Grading of Recommendations Assessment Development and Evaluation GROOVE—Graphical Representation of Overlap for OVErviews |
| HR | Hazard Ratio |
| IGF | Insulin-like Growth Factors |
| IL-6 | Interleukin-6 |
| JBI | Joanna Briggs Institute |
| MA | Meta Analysis |
| MOOSE | Meta-analyses Of Observational Studies in Epidemiology NOS—Newcastle–Ottawa Quality Assessment Scale |
| OR | Odds Ratio |
| PECO | Population, Exposure, Comparison, Outcome |
| PRISMA | Preferred Reporting Items for Systematic reviews and Meta-Analyses RR—Relative Risk |
| RRR | Relative Risk Ratio |
| SR | Systematic Review |
References
- Feller, G.; Khammissa, R.A.G.; Nemutandani, M.S.; Feller, L. Biological consequences of cancer radiotherapy in the context of oral squamous cell carcinoma. Head Face Med. 2021, 17, 35. [Google Scholar] [CrossRef] [PubMed]
- Mouth Cancer Foundation. Symptoms. Available online: https://www.mouthcancerfoundation.org/symptoms/ (accessed on 23 February 2024).
- Liu, C.; Wang, M.; Zhang, H.; Li, C.; Zhang, T.; Liu, H.; Zhu, S.; Chen, J. Tumor microenvironment and immunotherapy of oral cancer. Eur. J. Med. Res. 2022, 27, 198. [Google Scholar] [CrossRef] [PubMed]
- National Health System. Mouth Cancer—Treatment. 2017. Available online: https://www.nhs.uk/conditions/mouth-cancer/treatment/ (accessed on 23 February 2024).
- Omura, K. Current status of oral cancer treatment strategies: Surgical treatments for oral squamous cell carcinoma. Int. J. Clin. Oncol. 2014, 19, 423–430. [Google Scholar] [CrossRef] [PubMed]
- Kawakita, D.; Matsuo, K. Alcohol and head and neck cancer. Cancer Metastasis Rev. 2017, 36, 425–434. [Google Scholar] [CrossRef] [PubMed]
- Alterio, D.; Marvaso, G.; Ferrari, A.; Volpe, S.; Orecchia, R.; Jereczek-Fossa, B.A. Modern radiotherapy for head and neck cancer. Semin. Oncol. 2019, 46, 233–245. [Google Scholar] [CrossRef] [PubMed]
- American Diabetes Association. Diagnosis and Classification of Diabetes Mellitus. Diabetes Care 2010, 34 (Suppl. S1), S62–S69. [Google Scholar] [CrossRef]
- Leahy, J.L. Pathogenesis of Type 2 Diabetes Mellitus. Arch. Med. Res. 2005, 36, 197–209. [Google Scholar] [CrossRef] [PubMed]
- Tomic, D.; Shaw, J.E.; Magliano, D.J. The Burden and Risks of Emerging Complications of Diabetes Mellitus. Nat. Rev. Endocrinol. 2022, 18, 525–539. [Google Scholar] [CrossRef] [PubMed]
- Morss, A.S.; Edelman, E.R. Glucose Modulates Basement Membrane Fibroblast Growth Factor-2 via Alterations in Endothelial Cell Permeability. J. Biol. Chem. 2007, 282, 14635–14644. [Google Scholar] [CrossRef] [PubMed]
- Richardson, L.C.; Pollack, L.A. Therapy insight: Influence of type 2 diabetes on the development, treatment and outcomes of cancer. Nat. Clin. Pract. Oncol. 2005, 2, 48–53. [Google Scholar] [CrossRef] [PubMed]
- Noto, H.; Osame, K.; Sasazuki, T.; Noda, M. Substantially increased risk of cancer in patients with diabetes mellitus. J. Diabetes Its Complicat. 2010, 24, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Gong, Y.; Wei, B.; Yu, L.; Pan, W. Type 2 diabetes mellitus and risk of oral cancer and precancerous lesions: A meta-analysis of observational studies. Oral Oncol. 2015, 51, 332–340. [Google Scholar] [CrossRef] [PubMed]
- Page, M.J.; Moher, D.; Bossuyt, P.M.; Boutron, I.; Hoffmann, T.C.; Mulrow, C.D.; Shamseer, L.; Tetzlaff, J.M.; Akl, E.A.; Brennan, S.E. PRISMA 2020 explanation and elaboration: Updated guidance and exemplars for reporting systematic reviews. BMJ 2021, 372, n160. [Google Scholar] [CrossRef] [PubMed]
- Aromataris, E.; Fernandez, R.; Godfrey, C.M.; Holly, C.; Khalil, H.; Tungpunkom, P. Summarizing systematic reviews: Methodological development, conduct and reporting of an umbrella review approach. Int. J. Evid.-Based Healthc. 2015, 13, 132–140. [Google Scholar] [CrossRef] [PubMed]
- Aromataris, E.; Munn, Z. JBI Reviewer’s Manual; JBI: Singapore, 2020. [Google Scholar] [CrossRef]
- Sona, M.F.; Myung, S.K.; Park, K.; Jargalsaikhan, G. Type 1 diabetes mellitus and risk of cancer: A meta-analysis of observational studies. Jpn. J. Clin. Oncol. 2018, 48, 426–433. [Google Scholar] [CrossRef] [PubMed]
- Ohkuma, T.; Peters, S.A.E.; Woodward, M. Sex differences in the association between diabetes and cancer: A systematic review and meta-analysis of 121 cohorts including 20 million individuals and one million events. Diabetologia 2018, 61, 2140–2154. [Google Scholar] [CrossRef] [PubMed]
- Ramos-Garcia, P.; del Mar Roca-Rodriguez, M.; Aguilar-Diosdado, M.; Gonzalez-Moles, M.A. Diabetes mellitus and oral cancer/oral potentially malignant disorders: A systematic review and meta-analysis. Oral Dis. 2020, 27, 404–421. [Google Scholar] [CrossRef] [PubMed]
- Yan, P.; Wang, Y.; Yu, X.; Liu, Y.; Zhang, Z.J. Type 2 diabetes mellitus and risk of head and neck cancer subtypes: A systematic review and meta-analysis of observational studies. Acta Diabetol. 2021, 58, 549–565. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos, E.S.; Pérez-de-Oliveira, M.E.; Normando, A.G.C.; Gueiros, L.A.M.; Rogatto, S.R.; Vargas, P.A.; da Silva Guerra, E.N.; Leme, A.F.P.; Santos-Silva, A.R. Systemic conditions associated with increased risk to develop oral squamous cell carcinoma: Systematic review and meta-analysis. Head Neck 2022, 44, 2925–2937. [Google Scholar] [CrossRef] [PubMed]
- Gormley, A.; Richards, C.; Spiga, F.; Gray, E.; Hooper, J.; Main, B.; Vincent, E.E.; Richmond, R.; Higgins, J.; Gormley, M. Metabolic disorders and the risk of head and neck cancer: A protocol for a systematic review and meta-analysis. BMJ Open 2022, 12, e058392. [Google Scholar] [CrossRef] [PubMed]
- Shih, Y.H.; Wang, T.H.; Shieh, T.M.; Tseng, Y.H. Oral Submucous Fibrosis: A Review on Etiopathogenesis, Diagnosis, and Therapy. Int. J. Mol. Sci. 2019, 20, 2940–2962. [Google Scholar] [CrossRef] [PubMed]
- Barclay, A.W.; Petocz, P.; McMillan-Price, J.; Flood, V.M.; Prvan, T.; Mitchell, P.; Brand-Miller, J.C. Glycemic index, glycemic load, and chronic disease risk—A meta-analysis of observational studies. Am. J. Clin. Nutr. 2008, 87, 627–637. [Google Scholar] [CrossRef] [PubMed]
- Denley, A.; Carroll, J.M.; Brierley, G.V.; Cosgrove, L.; Wallace, J.; Forbes, B.; Roberts, C.T. Differential Activation of Insulin Receptor Substrates 1 and 2 by Insulin-Like Growth Factor-Activated Insulin Receptors. Mol. Cell. Biol. 2007, 27, 3569–3577. [Google Scholar] [CrossRef] [PubMed]
- Wolf, I.; Sadetzki, S.; Catane, R.; Karasik, A.; Kaufman, B. Diabetes mellitus and breast cancer. Lancet Oncol. 2005, 6, 103–111. [Google Scholar] [CrossRef] [PubMed]
- Giovannucci, E. Insulin, insulin-like growth factors and colon cancer: A review of the evidence. J. Nutr. 2001, 131 (Suppl. S11), 3109S–3120S. [Google Scholar] [CrossRef] [PubMed]
- Giovannucci, E.; Harlan, D.M.; Archer, M.C.; Bergenstal, R.M.; Gapstur, S.M.; Habel, L.A.; Pollak, M.; Regensteiner, J.G.; Yee, D. Diabetes and Cancer: A consensus report. Diabetes Care 2010, 33, 1674–1685. [Google Scholar] [CrossRef] [PubMed]
- Yu, H.; Pardoll, D.; Jove, R. STATs in cancer inflammation and immunity: A leading role for STAT3. Nat. Rev. Cancer 2009, 9, 798–809. [Google Scholar] [CrossRef] [PubMed]
- Calle, E.; Kaaks, R. Overweight, obesity and cancer: Epidemiological evidence and proposed mechanisms. Nat. Rev. Cancer 2004, 4, 579–591. [Google Scholar] [CrossRef] [PubMed]
- American Cancer Society. Signs and Symptoms of Oral Cavity and Oropharyngeal Cancer. Available online: https://www.cancer.org/cancer/types/oral-cavity-and-oropharyngeal-cancer/causes-risks-prevention/risk-factors.html (accessed on 20 March 2024).
- Okumura, M.; Yamamoto, M.; Sakuma, H.; Kojima, T.; Maruyama, T.; Jamali, M.; Cooper, D.R.; Yasuda, K. Leptin and high glucose stimulate cell proliferation in MCF-7 human breast cancer cells: Reciprocal involvement of PKC-α and PPAR expression. Biochim. Biophys. Acta (BBA) Mol. Cell Res. 2002, 1592, 107–116. [Google Scholar] [CrossRef]
- Barone, B.B. Long-term All-Cause Mortality in Cancer Patients with Preexisting Diabetes Mellitus. JAMA 2008, 300, 2754–2764. [Google Scholar] [CrossRef] [PubMed]

| Component | Explanation |
|---|---|
| Population | Any kind of patients |
| Exposure | Having diabetes |
| Comparison | Not having diabetes |
| Outcome | Probability of having head and neck cancer |
| Database | Query |
|---|---|
| PubMED | ((“diabetes”[All Fields] OR “diabetes mellitus”[MeSH Terms] OR (“diabetes”[All Fields] AND “mellitus”[All Fields]) OR “diabetes mellitus”[All Fields] OR “diabetes”[All Fields] OR “diabetes insipidus”[MeSH Terms] OR (“diabetes”[All Fields] AND “insipidus”[All Fields]) OR “diabetes insipidus”[All Fields] OR “diabetic”[All Fields] OR “diabetics”[All Fields] OR “diabets”[All Fields]) AND (“cancer s”[All Fields] OR “cancerated”[All Fields] OR “canceration”[All Fields] OR “cancerization”[All Fields] OR “cancerized”[All Fields] OR “cancerous”[All Fields] OR “neoplasms”[MeSH Terms] OR “neoplasms”[All Fields] OR “cancer”[All Fields] OR “cancers”[All Fields] OR (“carcinoma”[MeSH Terms] OR “carcinoma”[All Fields] OR “carcinomas”[All Fields] OR “carcinoma s”[All Fields]) OR (“neoplasms”[MeSH Terms] OR “neoplasms”[All Fields] OR “neoplasia”[All Fields] OR “neoplasias”[All Fields]) OR (“cysts”[MeSH Terms] OR “cysts”[All Fields] OR “cyst”[All Fields] OR “neurofibroma”[MeSH Terms] OR “neurofibroma”[All Fields] OR “neurofibromas”[All Fields] OR “tumor s”[All Fields] OR “tumoral”[All Fields] OR “tumorous”[All Fields] OR “tumour”[All Fields] OR “neoplasms”[MeSH Terms] OR “neoplasms”[All Fields] OR “tumor”[All Fields] OR “tumour s”[All Fields] OR “tumoural”[All Fields] OR “tumourous”[All Fields] OR “tumours”[All Fields] OR “tumors”[All Fields]) OR (“neoplasm s”[All Fields] OR “neoplasms”[MeSH Terms] OR “neoplasms”[All Fields] OR “neoplasm”[All Fields]) OR (“maligna”[All Fields] OR “malignas”[All Fields]) OR (“adenocarcinoma”[MeSH Terms] OR “adenocarcinoma”[All Fields] OR “adenocarcinomas”[All Fields] OR “adenocarcinoma s”[All Fields])) AND (“meta analysis”[Publication Type] OR “meta analysis as topic”[MeSH Terms] OR “meta analysis”[All Fields] OR (“systematic review”[Publication Type] OR “systematic reviews as topic”[MeSH Terms] OR “systematic review”[All Fields])) AND (“mouth”[MeSH Terms] OR “mouth”[All Fields] OR “oral”[All Fields] OR (“buccal”[All Fields] OR “buccally”[All Fields]) OR (“head”[MeSH Terms] OR “head”[All Fields]) OR (“neck”[MeSH Terms] OR “neck”[All Fields]))) AND ((meta-analysis[Filter] OR systematicreview[Filter]) AND (fft[Filter]) AND (humans[Filter]) AND (english[Filter]) AND (2000:2024[pdat])) |
| EBSCO | “((diabetes) AND (cancer OR carcinoma OR neoplasm OR tumour OR neoplasm OR malignant) AND (meta-analysis OR systematic review) AND (oral OR buccal OR head OR neck))” 2000-2024 |
| Science Direct | “((diabetes) AND (cancer OR carcinoma OR neoplasm) AND (meta-analysis OR systematic review) AND (oral OR head))” 2000–2024 |
| Wiley | “((diabetes) AND (cancer OR carcinoma OR neoplasm OR tumour OR neoplasm OR malignant) AND (meta-analysis OR systematic review) AND (oral OR buccal OR head OR neck)) 2000–2024 and open access only |
| Cochrane | “((diabetes) AND (cancer OR carcinoma OR neoplasm OR tumor OR neoplasm OR malignant) AND (meta-analysis OR systematic review) AND (oral OR buccal OR head OR neck))” 2000–2024 |
| Inclusion Criteria | Exclusion Criteria |
|---|---|
| Meta-analyses and systematic reviews | Types of cancer other than head and neck as an outcome |
| From the beginning of 2000 to the end of January 2024 | Associations other than diabetes mellitus with oral cancer |
| English only | Studies with no original data or effect statistics reported. Results that cannot be interpreted/converted into categorical |
| Open access (in the case of Science Direct) | |
| Human participants, regardless of age and gender | Human studies only: no in vivo, animal, or in vitro cell line studies |
| Study subjects must have type 1 or type 2 diabetes | Associations other than diabetes mellitus with oral cancer |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Formosinho, F.; Arcanjo, A.; Manso, M.C. Association Between Diabetes Mellitus and Head and Neck Cancer: An Umbrella Review of Systematic Reviews and Meta-Analyses. Oral 2025, 5, 52. https://doi.org/10.3390/oral5030052
Formosinho F, Arcanjo A, Manso MC. Association Between Diabetes Mellitus and Head and Neck Cancer: An Umbrella Review of Systematic Reviews and Meta-Analyses. Oral. 2025; 5(3):52. https://doi.org/10.3390/oral5030052
Chicago/Turabian StyleFormosinho, Filipa, Alexandra Arcanjo, and Maria Conceição Manso. 2025. "Association Between Diabetes Mellitus and Head and Neck Cancer: An Umbrella Review of Systematic Reviews and Meta-Analyses" Oral 5, no. 3: 52. https://doi.org/10.3390/oral5030052
APA StyleFormosinho, F., Arcanjo, A., & Manso, M. C. (2025). Association Between Diabetes Mellitus and Head and Neck Cancer: An Umbrella Review of Systematic Reviews and Meta-Analyses. Oral, 5(3), 52. https://doi.org/10.3390/oral5030052






