Imaging of Vascular Graft/Endograft Infection with Radiolabeled White Blood Cell Scan and [18F]FDG PET/CT
Abstract
1. Introduction
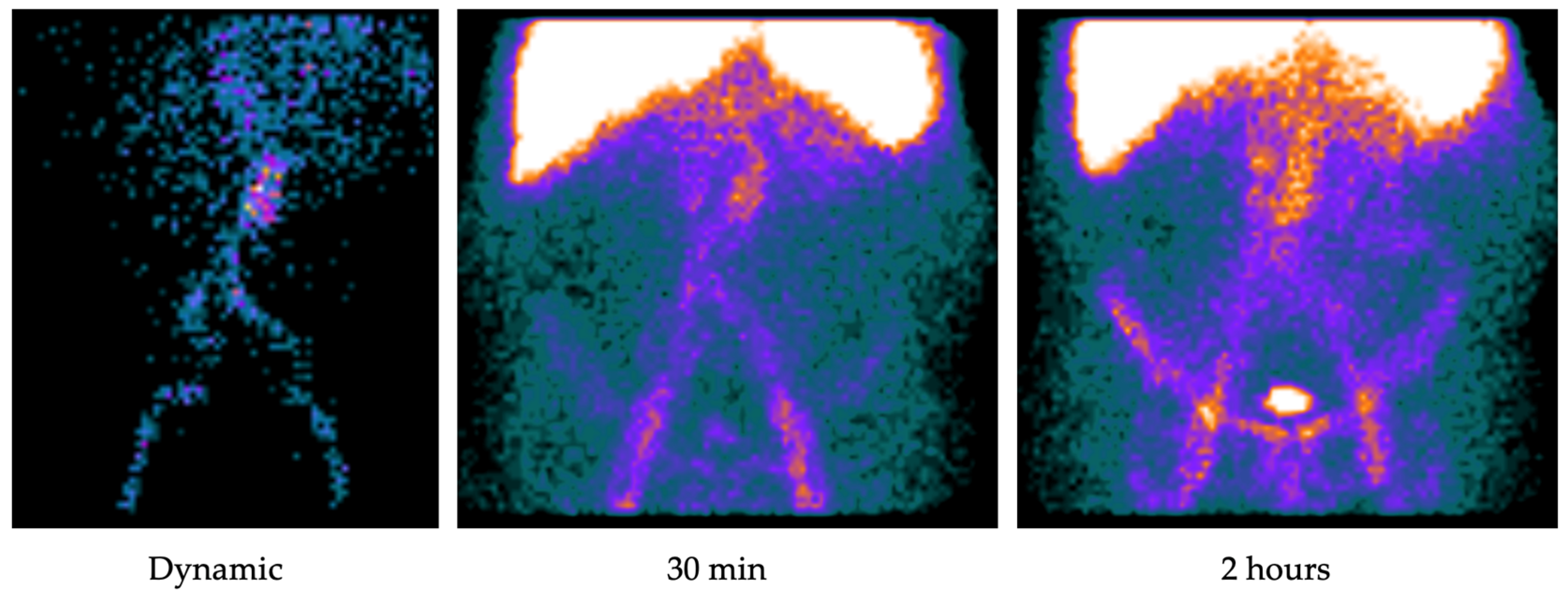
2. Radiolabeled White Blood Cell Scan
2.1. Image Analysis
2.2. Literature Review
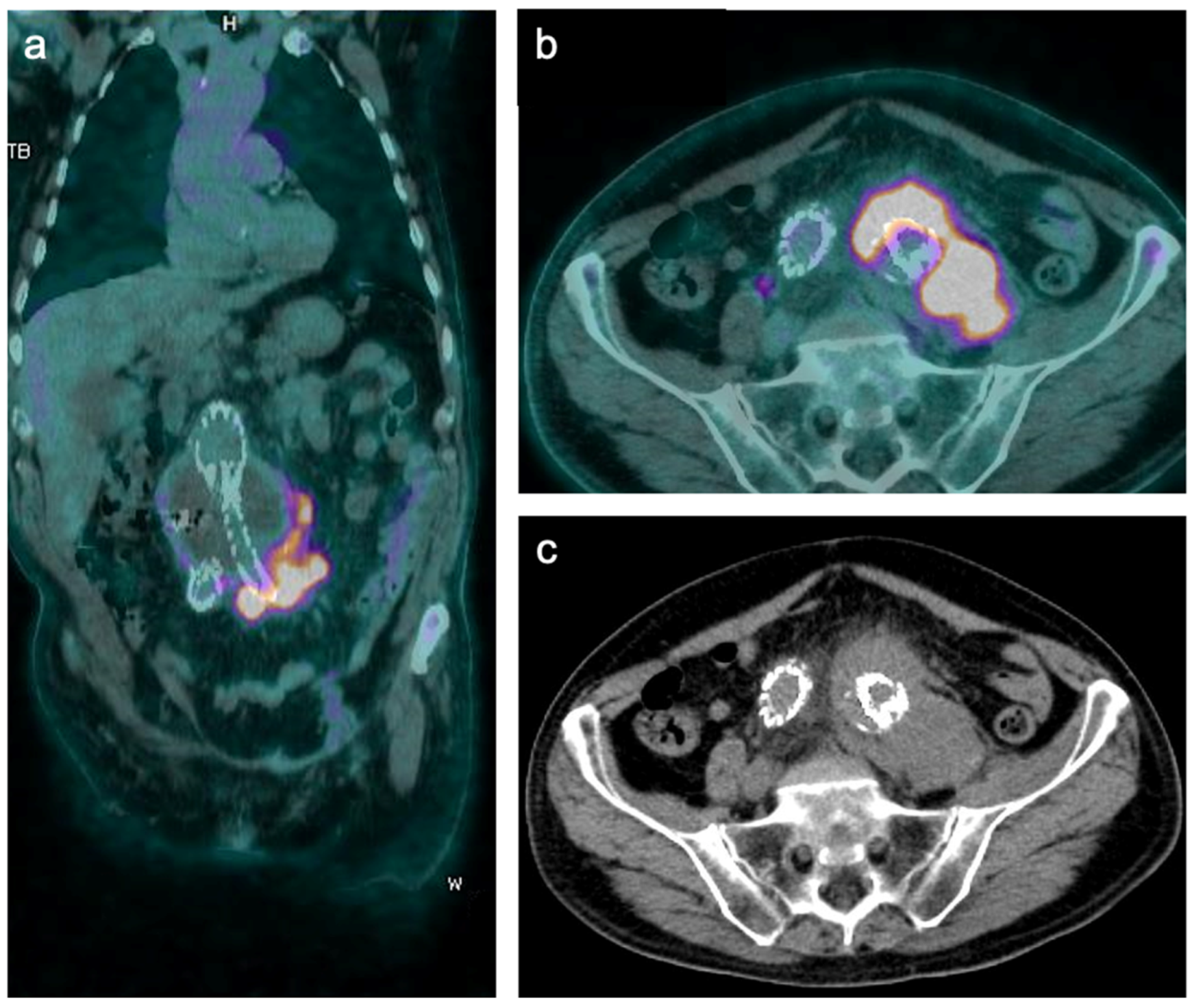
3. [18F]FDG PET/CT
3.1. Image Analysis
3.2. Literature Review
- Grade I: normal background activity;
- Grade II: mildly increased but diffuse FDG uptake along the graft (mild uptake: less than twice the blood pool activity in the ascending aorta; strong uptake: more than twice the blood pool activity in the ascending aorta);
- Grade III: focal but only mild FDG uptake or strong diffuse FDG uptake along the graft;
- Grade IV: focal and intense FDG uptake (±diffuse FDG uptake along the graft);
- Grade V: focal and intense FDG uptake plus fluid collections/abscess formation.
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| PTFE | Polytetrafluoroethylene |
| VGEI | Vascular graft/endograft infection |
| MAGIC | Management of Aortic Graft Infection Collaboration |
| WBCs | White blood cells |
| [18F]FDG PET/CT | Fluorine-18 fluorodeoxyglucose positron emission tomography/computed tomography |
| AGI | Aortic graft infection |
| ESVS | European Society for Vascular Surgery |
| VASGRA | Vascular Graft Infection Cohort study |
| US | Ultrasonography |
| CTA | Computed tomography angiography |
| MRA | Magnetic resonance angiography |
| EANM | European Association of Nuclear Medicine |
| HMPAO | Hexamethylpropyleneamine |
| p.i. | Post-injection |
| EVAR | Endovascular aneurysm repair |
| SUVmax | Maximum standardized uptake value |
| SUVmean | Mean standardized uptake value |
| TBR | Target-to-background ratio |
| EVA | Endovascular aneurysm sealing |
| VGS | Visual grading scale |
References
- Ambler, G.K.; Twine, C.P. Graft type for femoro-popliteal bypass surgery. Cochrane Database Syst. Rev. 2018, 2018, CD001487. [Google Scholar] [CrossRef] [PubMed]
- Hasse, B.; Husmann, L.; Zinkernagel, A.; Weber, R.; Lachat, M.; Mayer, D. Vascular graft infections. Swiss Med. Wkly. 2013, 143, w13754. [Google Scholar] [CrossRef][Green Version]
- Li, H.L.; Chan, Y.C.; Cheng, S.W. Current Evidence on Management of Aortic Stent-graft Infection: A Systematic Review and Meta-Analysis. Ann. Vasc. Surg. 2018, 51, 306–313. [Google Scholar] [CrossRef] [PubMed]
- Legout, L.; D’Elia, P.V.; Sarraz-Bournet, B.; Haulon, S.; Meybeck, A.; Senneville, E.; Leroy, O. Diagnosis and management of prosthetic vascular graft infections. Med. Mal. Infect. 2012, 42, 102–109. [Google Scholar] [CrossRef] [PubMed]
- Gharamti, A.; Kanafani, Z.A. Vascular Graft Infections: An update. Infect. Dis. Clin. N. Am. 2018, 32, 789–809. [Google Scholar] [CrossRef]
- Saleem, B.R.; Meerwaldt, R.; Tielliu, I.F.J.; Verhoeven, E.L.G.; Van Den Dungen, J.J.A.M.; Zeebregts, C.J. Conservative treatment of vascular prosthetic graft infection is associated with high mortality. Am. J. Surg. 2010, 200, 47–52. [Google Scholar] [CrossRef]
- Lyons, O.T.A.; Baguneid, M.; Barwick, T.D.; Bell, R.E.; Foster, N.; Homer-Vanniasinkam, S.; Hopkins, S.; Hussain, A.; Katsanos, K.; Modarai, B.; et al. Diagnosis of Aortic Graft Infection: A Case Definition by the Management of Aortic Graft Infection Collaboration (MAGIC). Eur. J. Vasc. Endovasc. Surg. 2016, 52, 758–763. [Google Scholar] [CrossRef]
- Chakfé, N.; Diener, H.; Lejay, A.; Assadian, O.; Berard, X.; Caillon, J.; Fourneau, I.; Glaudemans, A.W.J.M.; Koncar, I.; Lindholt, J.; et al. Editor’s Choice—European Society for Vascular Surgery (ESVS) 2020 Clinical Practice Guidelines on the Management of Vascular Graft and Endograft Infections. Eur. J. Vasc. Endovasc. Surg. 2020, 59, 339–384. [Google Scholar] [CrossRef]
- Anagnostopoulos, A.; Mayer, F.; Ledergerber, B.; Bergadà-Pijuan, J.; Husmann, L.; Mestres, C.A.; Rancic, Z.; Hasse, B.; VASGRA Cohort Study. Editor’s Choice—Validation of the Management of Aortic Graft Infection Collaboration (MAGIC) Criteria for the Diagnosis of Vascular Graft/Endograft Infection: Results from the Prospective Vascular Graft Cohort Study. Eur. J. Vasc. Endovasc. Surg. 2021, 62, 251–257. [Google Scholar] [CrossRef]
- Lauri, C.; Signore, A.; Glaudemans, A.W.J.M.; Treglia, G.; Gheysens, O.; Slart, R.H.J.A.; Iezzi, R.; Prakken, N.H.J.; Debus, E.S.; Honig, S.; et al. Evidence-based guideline of the European Association of Nuclear Medicine (EANM) on imaging infection in vascular grafts. Eur. J. Nucl. Med. Mol. Imaging 2022, 49, 3430–3451. [Google Scholar] [CrossRef]
- Reinders Folmer, E.I.; Von Meijenfeldt, G.C.I.; Van der Laan, M.J.; Glaudemans, A.W.J.M.; Slart, R.H.J.A.; Saleem, B.R.; Zeebregts, C.J. Diagnostic Imaging in Vascular Graft Infection: A Systematic Review and Meta-Analysis. Eur. J. Vasc. Endovasc. Surg. 2018, 56, 719–729. [Google Scholar] [CrossRef] [PubMed]
- Signore, A.; Jamar, F.; Israel, O.; Buscombe, J.; Martin-Comin, J.; Lazzeri, E. Clinical indications, image acquisition and data interpretation for white blood cells and anti-granulocyte monoclonal antibody scintigraphy: An EANM procedural guideline. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 1816–1831. [Google Scholar] [CrossRef] [PubMed]
- Erba, P.A.; Leo, G.; Sollini, M.; Tascini, C.; Boni, R.; Berchiolli, R.N.; Menichetti, F.; Ferrari, M.; Lazzeri, E.; Mariani, G. Radiolabelled leucocyte scintigraphy versus conventional radiological imaging for the management of late, low-grade vascular prosthesis infections. Eur. J. Nucl. Med. Mol. Imaging 2014, 41, 357–368. [Google Scholar] [CrossRef]
- Khaja, M.S.; Sildiroglu, O.; Hagspiel, K.; Rehm, P.K.; Cherry, K.J.; Turba, U.C. Prosthetic vascular graft infection imaging. J. Clin. Imaging. 2013, 37, 239–244. [Google Scholar] [CrossRef]
- Puges, M.; Bérard, X.; Ruiz, J.B.; Debordeaux, F.; Desclaux, A.; Stecken, L.; Pereyre Sc Hocquelet, A.; Bordenave, J.; Pinaquy, J.B.; Cazanave, C. Retrospective Study Comparing WBC scan and 18F-FDG PET/CT in Patients with Suspected Prosthetic Vascular Graft Infection. Eur. J. Vasc. Endovasc. Surg. 2019, 57, 876–884. [Google Scholar] [CrossRef] [PubMed]
- de la Rubia-Marcos, M.; García-Alonso, P.; Mena-Melgar, C.; Tagliatori-Nogueira, B.; Herrero-Muñoz, A.; Sandoval-Moreno, C.; Paniagua-Correa, C.; Castillejos-Rodríguez, L.; Ortega-Valle, A.; Balsa-Bretón, M.A. 99mTC-white blood cell scintigraphy with SPECT/CT in the diagnosis of vascular graft infection. Rev. Española Med. Nucl. Imagen Mol. (Engl. Ed.) 2020, 39, 347–352. [Google Scholar] [CrossRef]
- Vasquez, L.; Ong, Q.H.; Zaman, Z.; Allen, B.; Khashram, M. Clinical utility of technetium-99m-labelled leukocyte scan in diagnosis of vascular infections. J. Med. Imaging Radiat. Oncol. 2022, 67, 344–348. [Google Scholar] [CrossRef]
- Lauri, C.; Campagna, G.; Aloisi, F.; Posa, A.; Iezzi, R.; Sirignano, P.; Taurino, M.; Signore, A. How to combine CTA, 99mTc-WBC SPECT/CT, and [18F]FDG PET/CT in patients with suspected abdominal vascular endograft infections? Eur. J. Nucl. Med. Mol. Imaging 2023. [Google Scholar] [CrossRef]
- Folmer, E.I.; von Meijenfeldt, G.C.; van der Laan, M.J.; Glaudemans, A.W.; Slart, R.H.; Zeebregts, C.J.; Saleem, B.R. A systematic review and meta-analysis of 18F-fluoro-D-deoxyglucose positron emission tomography interpretation methods in vascular graft and endograft infection. J. Vasc. Surg. 2020, 72, 2174–2185.e2. [Google Scholar] [CrossRef]
- Liberatore, M.; Misuraca, M.; Calandri, E.; Rizzo, L.; Speziale, F.; Iurilli, A.P.; Anagnostou, C. White blood cell scintigraphy in the diagnosis of infection of endovascular prostheses within the first month after implantation. Med. Sci. Monit. 2006, 12, 5–9. [Google Scholar]
- Spacek, M.; Belohlavek, O.; Votrubova, J.; Sebesta, P. Diagnostics of “ non-acute ” vascular prosthesis infection using 18 F-FDG PET/CT: Our experience with 96 prostheses. Eur. J. Nucl. Med. Mol. Imaging 2009, 36, 850–858. [Google Scholar] [CrossRef] [PubMed]
- Keidar, Z.; Engel, A.; Hoffman, A.; Israel, O.; Nitecki, S. Prosthetic vascular graft infection: The role of 18F-FDG PET/CT. J. Nucl. Med. 2007, 48, 1230–1236. [Google Scholar] [CrossRef] [PubMed]
- Keidar, Z.; Nitecki, S. FDG-PET in Prosthetic Graft Infections. Semin Nucl. Med. 2013, 43, 396–402. [Google Scholar] [CrossRef]
- Saleem, B.R.; Pol, R.A.; Slart, R.H.J.A.; Reijnen, M.M.P.J.; Zeebregts, C.J. 18F-Fluorodeoxyglucose Positron Emission Tomography/CT Scanning in Diagnosing Vascular Prosthetic Graft Infection. Biomed. Res. Int. 2014, 2014, 471971. [Google Scholar] [CrossRef]
- Bruggink, J.L.M.; Glaudemans, A.W.J.M.; Saleem, B.R.; Meerwaldt, R.; Alkefaji, H.; Prins, T.R.; Slart, R.H.; Zeebregts, C.J. Accuracy of FDG-PET/CT in the diagnostic work-up of vascular prosthetic graft infection. Eur. J. Vasc. Endovasc. Surg. 2010, 40, 348–354. [Google Scholar] [CrossRef] [PubMed]
- Lauri, C.; Iezzi, R.; Rossi, M.; Tinelli, G.; Sica, S.; Signore, A.; Posa, A.; Tanzilli, A.; Panzera, C.; Taurino, M.; et al. Imaging Modalities for the Diagnosis of Vascular Graft Infections: A Consensus Paper amongst Different Specialists. J. Clin. Med. 2020, 9, 1510. [Google Scholar] [CrossRef]
- Mitra, A.; Pencharz, D.; Davis, M.; Wagner, T. Determining the Diagnostic Value of 18F-Fluorodeoxyglucose Positron Emission/Computed Tomography in Detecting Prosthetic Aortic Graft Infection. Ann. Vasc. Surg. 2018, 53, 78–85. [Google Scholar] [CrossRef]
- Chrapko, B.E.; Chrapko, M.; Nocuń, A.; Zubilewicz, T.; Stefaniak, B.; Mitura, J.; Wolski, A.; Terelecki, P. Patterns of vascular graft infection in 18F-FDG PET/CT. Nucl. Med. Rev. 2020, 23, 63–70. [Google Scholar] [CrossRef]
- Tokuda, Y.; Oshima, H.; Araki, Y.; Narita, Y.; Mutsuga, M.; Kato, K.; Usui, A. Detection of thoracic aortic prosthetic graft infection with 18F-fluorodeoxyglucose positron emission tomography/computed tomography. Eur. J. Cardio-Thoracic Surg. 2013, 43, 1183–1187. [Google Scholar] [CrossRef]
- Lauri, C.; Signore, A.; Campagna, G.; Aloisi, F.; Taurino, M.; Sirignano, P. [18F]FDG Uptake in Non-Infected Endovascular Grafts: A Retrospective Study. Diagnostics 2023, 13, 409. [Google Scholar] [CrossRef]
- Keidar, Z.; Pirmisashvili, N.; Leiderman, M.; Nitecki, S.; Israel, O. 18F-FDG uptake in noninfected prosthetic vascular grafts: Incidence, patterns, and changes over time. J. Nucl. Med. 2014, 55, 392–395. [Google Scholar] [CrossRef] [PubMed]
- Groot Jebbink, E.; van Den Ham, L.H.; van Woudenberg, B.B.J.; Slart, R.H.J.A.; Zeebregts, C.J.; Rijnders, T.J.M.; Lardenoije, J.H.P.; Reijnen, M.M.P.J. Physiological Appearance of Hybrid FDG–Positron Emission Tomography/Computed Tomography Imaging Following Uncomplicated Endovascular Aneurysm Sealing Using the Nellix Endoprosthesis. J. Endovasc. Ther. 2020, 27, 509–515. [Google Scholar] [CrossRef] [PubMed]
- Marie, P.Y.; Plissonnier, D.; Bravetti, S.; Coscas, R.; Rouer, M.; Haulon, S.; Mandry, D.; Alsac, J.M.; Malikov, S.; Settembre, N. Low baseline and subsequent higher aortic abdominal aneurysm FDG uptake are associated with poor sac shrinkage post endovascular repair. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 549–557. [Google Scholar] [CrossRef]
- Saleem, B.R.; Berger, P.; Vaartjes, I.; De Keizer, B.; Vonken, E.J.P.A.; Slart, R.H.J.A.; de Borst, G.J.; Zeebregts, C.J. Modest utility of quantitative measures in 18F-fluorodeoxyglucose positron emission tomography scanning for the diagnosis of aortic prosthetic graft infection. J. Vasc. Surg. 2015, 61, 965–971. [Google Scholar] [CrossRef] [PubMed]
- Rahimi, M.; Adlouni, M.; Ahmed, A.I.; Alnabelsi, T.; Chinnadurai, P.; Al-Mallah, M.H. Diagnostic Accuracy of FDG PET for the Identification of Vascular Graft Infection. Ann. Vasc. Surg. 2022, 87, 422–429. [Google Scholar] [CrossRef] [PubMed]
- Sah, B.R.; Husmann, L.; Mayer, D.; Scherrer, A.; Rancic, Z.; Puippe, G.; Weber, R.; Hasse, B.; VASGRA Cohort. Diagnostic performance of 18F-FDG-PET/CT in vascular graft infections. Eur. J. Vasc. Endovasc. Surg. 2015, 49, 455–464. [Google Scholar] [CrossRef]
- Husmann, L.; Huellner, M.W.; Ledergerber, B.; Anagnostopoulos, A.; Stolzmann, P.; Sah, B.R.; Burger, I.A.; Rancic, Z.; Hasse, B.; Vasgra Cohort. Comparing diagnostic accuracy of 18 F-FDG-PET/CT, contrast enhanced CT and combined imaging in patients with suspected vascular graft infections. Eur. J. Nucl. Med. Mol. Imaging 2019, 46, 1359–1368. [Google Scholar] [CrossRef]
- Berger, P.; Vaartjes, I.; Scholtens, A.; Moll, F.L.; De Borst, G.J.; De Keizer, B.; Bots, M.L. Differential FDG-PET Uptake Patterns in Uninfected and Infected Central Prosthetic Vascular Grafts. Eur. J. Vasc. Endovasc. Surg. 2015, 50, 376–383. [Google Scholar] [CrossRef]
- Fukuchi, K.; Ishida, Y.; Higashi, M.; Tsunekawa, T.; Ogino, H.; Minatoya, K.; Naito, H. Detection of aortic graft infection by fluorodeoxyglucose positron emission tomography: Comparison with computed tomographic findings. J. Vasc. Surg. 2005, 42, 919–925. [Google Scholar] [CrossRef]
- Einspieler, I.; Mergen, V.; Wendorff, H.; Haller, B.; Eiber, M.; Schwaiger, M.; Nekolla, S.G.; Mustafa, M. Diagnostic performance of quantitative and qualitative parameters for the diagnosis of aortic graft infection using [18F]-FDG PET/CT. J. Nucl. Cardiol. 2021, 28, 2220–2228. [Google Scholar] [CrossRef]
- Zogala, D.; Rucka, D.; Ptacnik, V.; Cerny, V.; Trnka, J.; Varejka, P.; Heller, S.; Lambert, L. How to recognize stent graft infection after endovascular aortic repair: The utility of 18F-FDG PET/CT in an infrequent but serious clinical setting. Ann. Nucl. Med. 2019, 33, 594–605. [Google Scholar] [CrossRef]
- Dong, W.; Li, Y.; Zhu, J.; Xia, J.; He, L.; Yun, M.; Jiao, J.; Zhu, G.; Hacker, M.; Wei, Y. Detection of aortic prosthetic graft infection with 18F-FDG PET/CT imaging, concordance with consensus MAGIC graft infection criteria. J. Nucl. Cardiol. 2021, 28, 1005–1016. [Google Scholar] [CrossRef]
- Husmann, L.; Eberhard, N.; Huellner, M.W.; Ledergerber, B.; Mueller, A.; Gruenig, H.; Messerli, M.; Mestres, C.A.; Rancic, Z.; Zimmermann, A.; et al. Impact of unknown incidental findings in PET/CT examinations of patients with proven or suspected vascular graft or endograft infections. Sci. Rep. 2021, 11, 13747. [Google Scholar] [CrossRef]
- Kagna, O.; Kurash, M.; Ghanem-Zoubi, N.; Keidar, Z.; Israel, O. Does antibiotic treatment affect the diagnostic accuracy of18F-FDG PET/CT studies in patients with uspected infectious processes? J. Nucl. Med. 2017, 58, 1827–1830. [Google Scholar] [CrossRef]
- Husmann, L.; Sah, B.R.; Scherrer, A.; Burger, I.A.; Stolzmann, P.; Weber, R.; Rancic, Z.; Mayer, D.; Hasse, B.; VASGRA Cohort. 18F-FDG PET/CT for therapy control in vascular graft infections: A first feasibility study. J. Nucl. Med. 2015, 56, 1024–1029. [Google Scholar] [CrossRef] [PubMed]
- Husmann, L.; Ledergerber, B.; Anagnostopoulos, A.; Stolzmann, P.; Sah, B.R.; Burger, I.A.; Pop, R.; Weber, A.; Mayer, D.; Rancic, Z.; et al. The role of FDG PET/CT in therapy control of aortic graft infection. Eur. J. Nucl. Med. Mol. Imaging 2018, 45, 1987–1997. [Google Scholar] [CrossRef] [PubMed]
- Saleem, B.R.; Beukinga, R.J.; Boellaard, R.; Glaudemans, A.W.J.M.; Reijnen, M.M.P.J.; Zeebregts, C.J.; Slart, R.H. Textural features of 18F-fluorodeoxyglucose positron emission tomography scanning in diagnosing aortic prosthetic graft infection. Eur. J. Nucl. Med. Mol. Imaging 2017, 44, 886–894. [Google Scholar] [CrossRef] [PubMed]


| Clinical | Radiological | Laboratory | |
|---|---|---|---|
| Major criteria | Pus (confirmed by microscopy) around graft of aneurysm sac at surgery Open wound with exposed graft or communicating sinus Fistula development Graft insertion in an infected site | Peri-graft fluid on CT > 3 months after insertion Peri-graft gas on CT scan > 7 weeks after insertion Increase in peri-graft volume demonstrated on serial imaging | Organism recovered from an explanted graft Organism recovered from an intra-operative specimen Organism recovered from a radiologically guided aspirate of peri-graft fluid |
| Minor criteria | Localized clinical features of graft infection at site (e.g., erythema, warmth, swelling, purulent discharge, pain) Febrile > 38 °C with AGI as the most likely cause | Other signs (e.g., suspicious peri-graft gas/fluid/soft tissue inflammation, aneurysm expansion, pseudoaneurysm formation, focal bowel wall thickening, discitis/osteomyelitis); suspicious metabolic activity on [18F]FDG PET/CT; radiolabeled leukocyte uptake | Blood culture(s) positive and no apparent source except graft infection Abnormally elevated inflammatory markers with graft infection as most likely cause (e.g., ESR, CRP, white cell count) |
| Patients | Methodology | Reference Standard | Imaging Protocol | Grafts | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Accuracy (%) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Erba et al. [13], 2013 | 55 (47 confirmed) | Retrospective | Microbiological, clinical, and paraclinical criteria | [99mTc]Tc-HMPAO Images obtained 30 min, 2 h or 4–6 h (delayed), and 20–24 h (late) p.i. | Peripheral and aortic grafts | 100 | 100 | 100 | 100 | 100 |
| Khaja et al. [14], 2013 | 20 | Retrospective | Results of tissue cultures from open surgical or percutaneous procedures and/or blood cultures | [111In]In-oxine Images obtained 20 h p.i. | Peripheral and aortic grafts | 75 | 100 | 100 | 50 | 80 |
| Puges et al. [15], 2019 | 39 (15 confirmed) | Retrospective | Microbiological, clinical, and paraclinical criteria | [99mTc]Tc-HMPAO Images obtained after 4–6 h (early) and 20–24 h (delayed) p.i. | Peripheral and aortic grafts | 89.5 | 90.9 | 70.8 | 97.2 | 90.6 |
| De la Rubia-Marcos et al. [16], 2020 | 30 (10 confirmed) | Retrospective | Microbiological, clinical, and paraclinical criteria | [99mTc]Tc-HMPAO Images obtained 30 min and 2 h p.i. | Peripheral and aortic grafts | 100 | 95 | 91 | 100 | |
| Vasquez et al. [17], 2022 | 43 (32 confirmed) | Retrospective | MAGIC criteria | [99mTc]Tc-HMPAO Images obtained after a minimum of 3 h p.i. | Aortic grafts and | 85.7 | 75 | |||
| peripheral grafts | 84.6 | 100 | ||||||||
| Lauri et al. [18], 2023 | 26 (11 confirmed) | Prospective | Microbiological/histological findings | [99mTc]Tc-HMPAO Images obtained 30 min, 2 h, and 20 h p.i. | Aortic (abdominal) | 81.8 | 100 | 88.2 | 100 | 92.3 |
| Advantages | Drawbacks | |
|---|---|---|
| WBC scan | Established acquisition protocols and interpretation criteria High accuracy both in early and late post-operative periods Accurately differentiates a VGEI from a sterile post-surgical inflammatory reaction Detection of alternative sites of infection Better inter-observer agreement than [18F]FDG PET/CT, as shown by Puges et al. [15] | Expected lower accuracy in aortic grafts due to hepato-biliary secretion of [99mTc]Tc-HMPAO and physiological bone marrow uptake Limited image resolution Long acquisition and labeling time Manipulation of potentially infected blood Requires highly trained personnel for in vitro labeling Limited availability High radiation dose High cost |
| [18F]FDG PET/CT | High sensitivity and ability to rule out infection High image resolution Accurately diagnoses a VGEI in the late post-operative period Detection of alternative sites of infections Short acquisition time Available in most centers | No established interpretation criteria Moderate specificity Lower accuracy inflammatory reaction in the early post-operative period than in the late post-operative period |
| Patients | Methodology | Reference Standard | Grafts | Interpretation Tool | Sensitivity (%) | Specificity (%) | PPV (%) | NPV (%) | Accuracy (%) | |
|---|---|---|---|---|---|---|---|---|---|---|
| Keidar et al. [22], 2007 | 39, with a total of 69 implanted grafts (15 confirmed) | Prospective | Histopathological/microbiological findings and clinical/imaging follow-up | Peripheral and aortic grafts | Intense focal pattern | 93 | 91 | 88 | 96 | |
| Spacek et al. [21], 2009 | 76, with a total of 96 implanted grafts (55 confirmed) | Prospective | Histopathology findings and clinical follow-up | Peripheral and aortic grafts | Intense focal pattern | 78.2 | 92.7 | 93.5 | 76.0 | 84.4 |
| Focal or inhomogenous pattern | 98.2 | 75.6 | 84.4 | 96.9 | 88.5 | |||||
| Inhomogenous pattern + suspicious CT findings | 72.7 | 85.77 | 88.9 | 66.7 | 77.8 | |||||
| Bruggink et al. [25] | 25 (15 confirmed) | Retrospective | Microbiological findings | Peripheral and aortic grafts | Overall (based on VGS I–IV ≥ III (establshed by Fukuchi et al. [39]), SUVmax, and TBR) | 93 | 70 | 82 | 88 | |
| Tokuda et al. [29], 2013 | 9 (4 confirmed) | Retrospective | Microbiological findings and clinical follow-up | Thoracic aortic grafts | SUVmax ≥ 8 | 100 | 80 | |||
| Saleem et al. [34], 2015 | 37 (21 confirmed) | Prospective | Microbiological findings | Peripheral and aortic grafts (35 patients with aortoiliac grafts; 2 patients with axillobifemoral grafts) | VGS 0-IV ≥ III (established by Fukuchi et al. [39]) | 86 | 63 | 75 | 77 | |
| Focal pattern | 90 | 25 | 61 | 67 | ||||||
| SUVmax ≥ 8 | 40 | 88 | 80 | 54 | ||||||
| TBR ≥ 6 | 40 | 81 | 73 | 52 | ||||||
| Sah et al. [36], 2015 | 34 (27 confirmed) | Prospective | Microbiological findings | Peripheral and aortic grafts | VGS I-V ≥ III | 100 | 86 | 96 | 100 | 97 |
| Focal pattern | 96 | 86 | 96 | 86 | 94 | |||||
| SUVmax ≥ 3.8 | 100 | 86 | ||||||||
| Mitra et al. [27], 2018 | 21 (13 confirmed) | Retrospective | Microbiological findings | Peripheral and aortic grafts | Overall (based on VGS I-IV modified from Sah et al. [36]) | 92 | 63 | 80 | 83 | |
| SUVmax ≥ 6.3 | 92 | 88 | 92 | 88 | ||||||
| Puges et al. [15], 2018 | 39 (15 confirmed) | Retrospective | Microbiological, clinical, and paraclinical criteria | Peripheral and aortic grafts | VGS I-V ≥ III (Sah et al. [36]) | 85 | 68.4 | 41.5 | 94.5 | 71.9 |
| Husmann et al. [37], 2019 | 23 (13 confirmed) | Prospective | Microbiological/ hisopathological findings and clinical follow-up | Aortic grafts | SUVmax ≥ 4.9 | 100 | 50 | 100 | 72.2 | 78.3 |
| Einspieler et al. [40], 2019 | 50 (28 confirmed) | Retrospective | MAGIC criteria irrespective of the results of the PET/CT | Aortic grafts | VGS I-V ≥ III (Sah et al. [36]) | 100 | 85.3 | 84.8 | 100 | 91.9 |
| SUVmax ≥ 4.96 | 89.3 | 100 | 100 | 91.2 | 95.2 | |||||
| GBRBP ≥ 2.99 | 89.3 | 94.1 | 92.6 | 91.4 | 90.3 | |||||
| GBRNIAW ≥ 6.21 | 82.1 | 97.1 | 95.8 | 86.8 | 93.5 | |||||
| GBRT ≥ 3.24 | 85.7 | 100 | 100 | 89.7 | 91.9 | |||||
| Zogala et al. [41], 2019 | 17 (9 confirmed) | Retrospective | Operative microbiological and clinical findings | EVAR | SUVmax ≥ 5.6 | 90 | 100 | |||
| TBRhep ≥ 2.2 TBRBP ≥ 3.5 | ||||||||||
| Overall (based on focal pattern and VGS 0-IV ≥ III modified from Saleem et al. [34]) | 89 | 100 | 100 | 89 | ||||||
| Dong et al. [42], 2020 | 35 (25 confirmed) | Prospective | MAGIC criteria irrespective of the results of the PET/CT | Aortic grafts | VGS I–V ≥ III (Sah et al. [36]) | 96 | 70 | 88.6 | ||
| Focal pattern | 84 | 90 | 85.7 | |||||||
| SUVmax ≥ 7.3 | 88 | 80 | 85.7 | |||||||
| TBRmax ≥ 4.2 | 92 | 80 | 88.6 | |||||||
| Overall (VGS ≥ III plus ≥ 1 other parameter) | 96 | 80 | 91.4 | |||||||
| Rahimi et al. [35], 2022 | 28 (15 confirmed) | Prospective | Clinical, laboratory, and radiologic findings | Peripheral and aortic grafts | SUVmax ≥ 4.5 | 93 | 92 | |||
| SUVmean ≥ 3.7 | 100 | 92 | ||||||||
| TBR ≥ 1.6 | 93 | 92 | ||||||||
| Lauri et al. [18], 2023 | 26 (11 confirmed) | Prospective | Microbiological findings | Aortic (abdominal) | VGS I–V ≥ III (Sah et al. [36]) | 100 | 40 | 55 | 100 | 65.4 |
| VGS I–V ≥ IV (Lauri’s scale [18]) | 100 | 73.3 | 73.3 | 100 | 84.6 | |||||
| SUVmax ≥ 4.52 | 90.3 | 53.3 | ||||||||
| SUVmean | 100 | 73.3 | ||||||||
| TBR | 90.9 | 66.7 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manta, R.; Lauri, C.; Taurino, M.; Signore, A. Imaging of Vascular Graft/Endograft Infection with Radiolabeled White Blood Cell Scan and [18F]FDG PET/CT. Hemato 2023, 4, 285-300. https://doi.org/10.3390/hemato4040023
Manta R, Lauri C, Taurino M, Signore A. Imaging of Vascular Graft/Endograft Infection with Radiolabeled White Blood Cell Scan and [18F]FDG PET/CT. Hemato. 2023; 4(4):285-300. https://doi.org/10.3390/hemato4040023
Chicago/Turabian StyleManta, Ringo, Chiara Lauri, Maurizio Taurino, and Alberto Signore. 2023. "Imaging of Vascular Graft/Endograft Infection with Radiolabeled White Blood Cell Scan and [18F]FDG PET/CT" Hemato 4, no. 4: 285-300. https://doi.org/10.3390/hemato4040023
APA StyleManta, R., Lauri, C., Taurino, M., & Signore, A. (2023). Imaging of Vascular Graft/Endograft Infection with Radiolabeled White Blood Cell Scan and [18F]FDG PET/CT. Hemato, 4(4), 285-300. https://doi.org/10.3390/hemato4040023








