KSHV/HHV8-Associated Lymphoproliferative Disorders: Lessons Learnt from People Living with HIV
Abstract
:1. Introduction
2. Pathological and Clinical Features
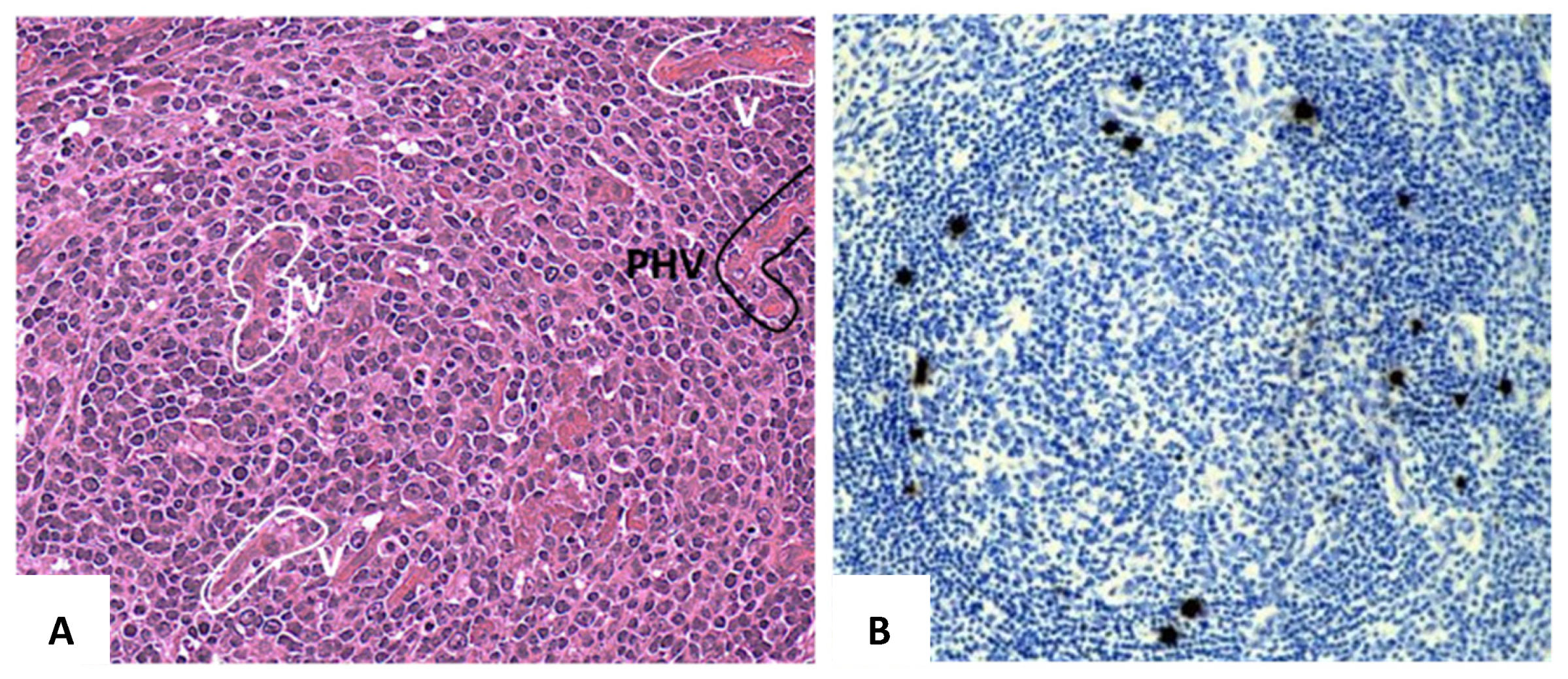
2.1. KSHV/HHV8-Associated Multicentric Castleman Disease (KSHV/HHV8-MCD)
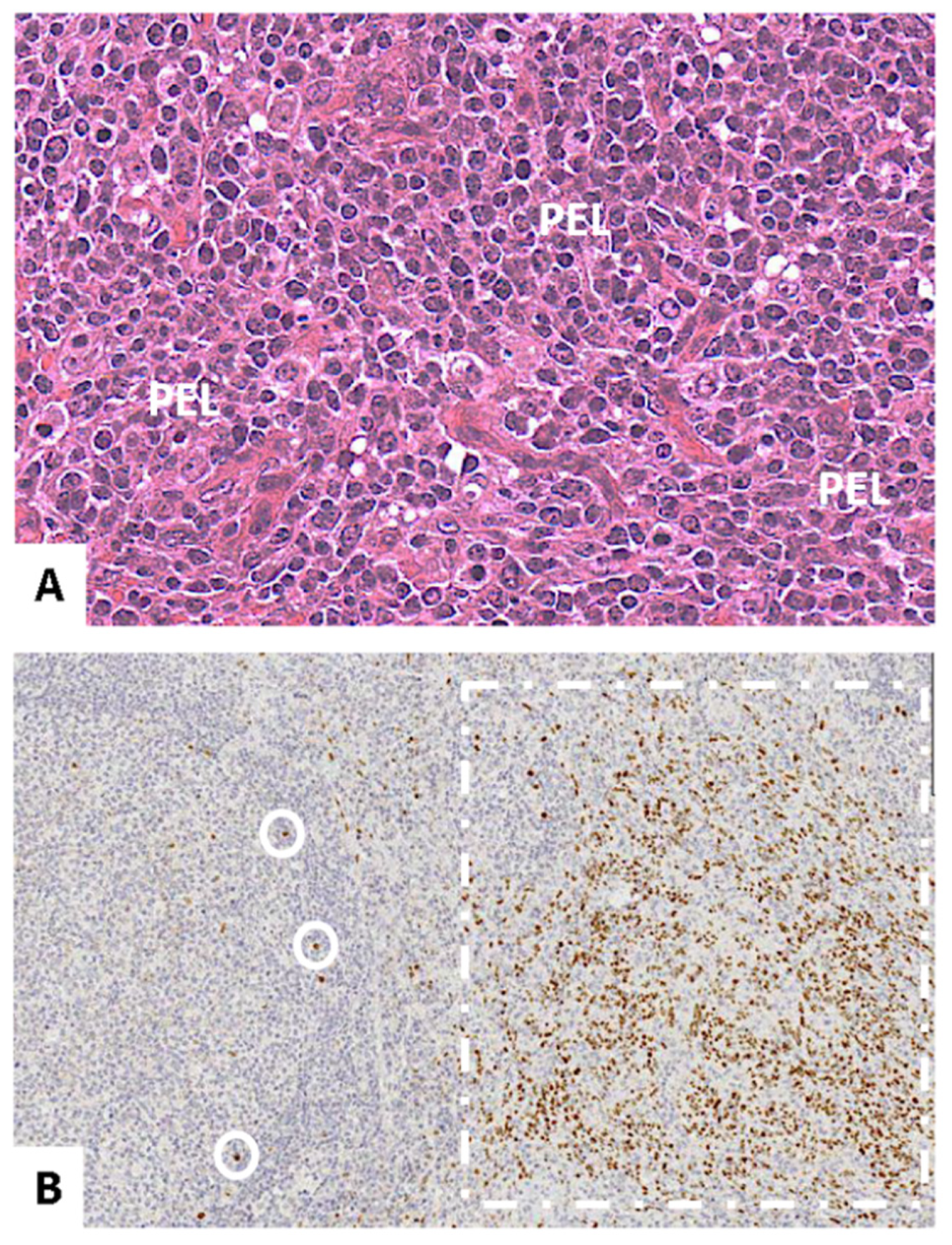
2.2. Primary Effusion Lymphoma (PEL), Classic, and Solid Variants
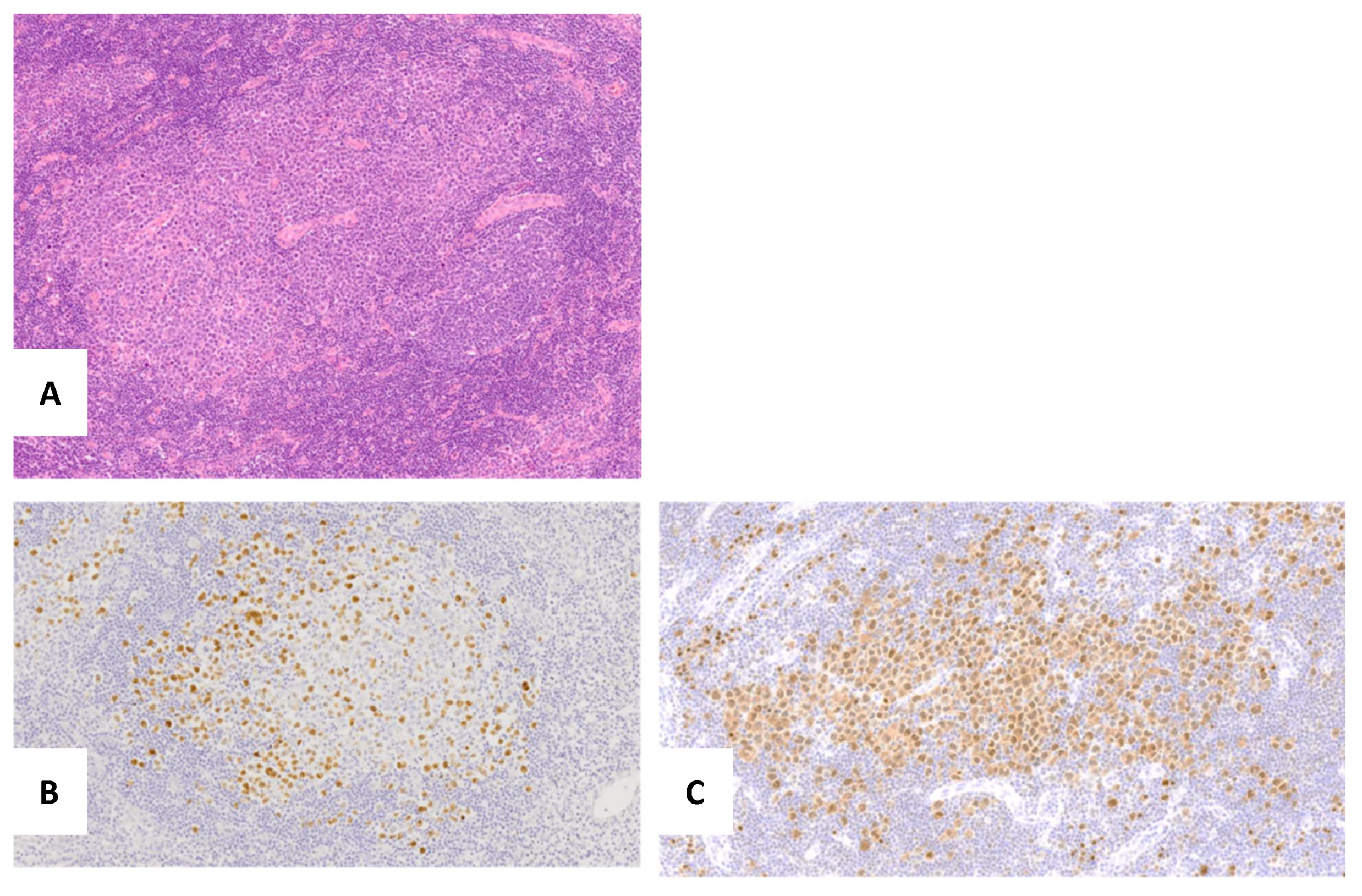
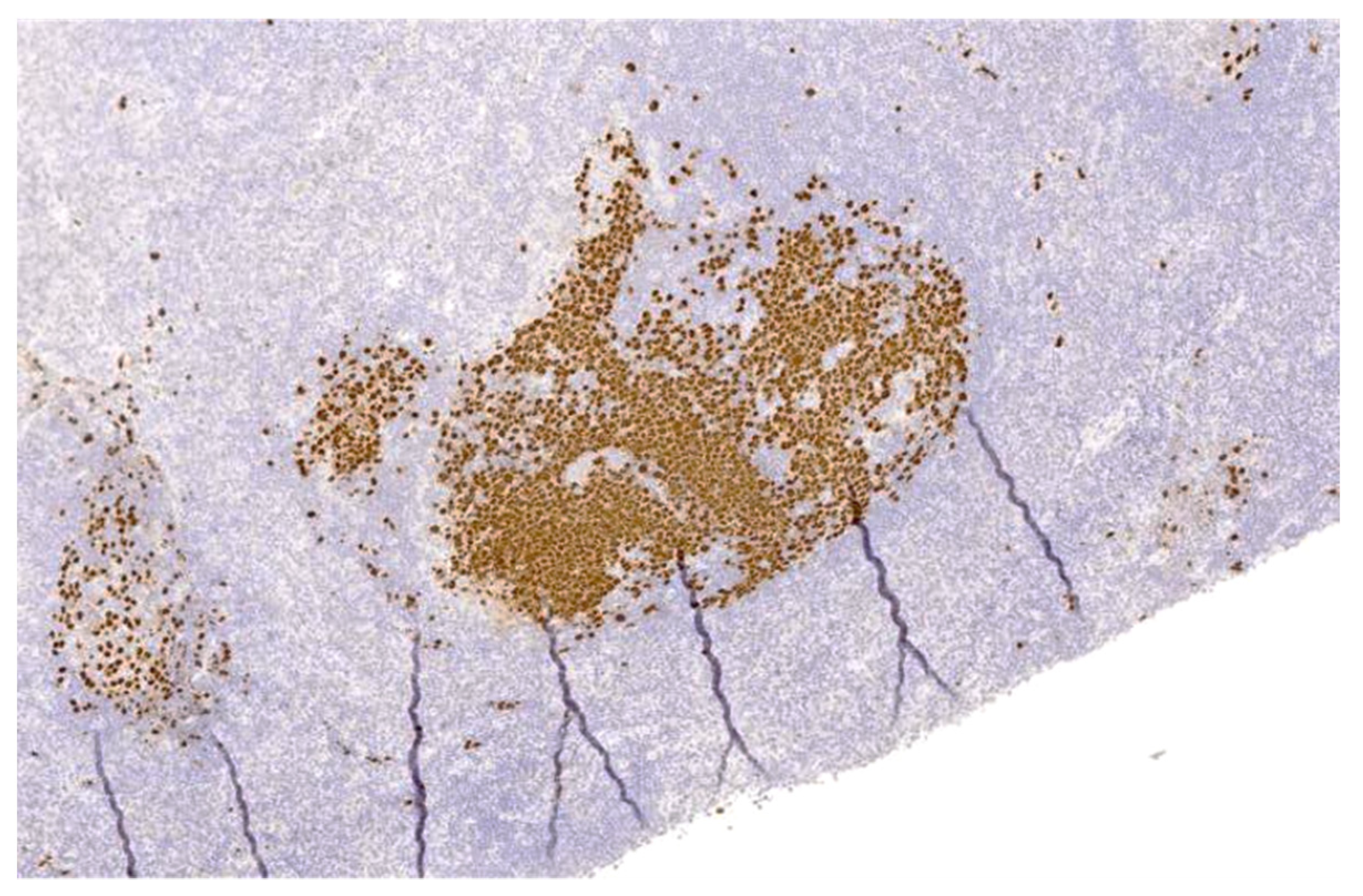
2.3. KSHV/HHV8-Positive Germinotropic Lymphoproliferative Disorder (GLPD)
3. Treatment and Outcome
3.1. KSHV/HHV8-MCD
3.2. PEL
3.3. KSHV/HHV8-Associated DLBCL
3.4. GLPD
4. Conclusions
Funding
Conflicts of Interest
References
- Beral, V.; Peterman, T.A.; Berkelman, R.L.; Jaffe, H.W. Kaposi’s sarcoma among persons with AIDS: A sexually transmitted infection? Lancet 1990, 335, 123–128. [Google Scholar] [CrossRef] [Green Version]
- Chang, Y.; Cesarman, E.; Pessin, M.S.; Lee, F.; Culpepper, J.; Knowles, D.M.; Moore, P.S. Identification of herpesvirus-like DNA sequences in AIDS-associated Kaposi’s sarcoma. Science 1994, 266, 1865–1869. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Judde, J.G.; Lacoste, V.; Briere, J.; Kassa-Kelembho, E.; Clyti, E.; Couppié, P.; Buchrieser, C.; Tulliez, M.; Morvan, J.; Gessain, A. Monoclonality or oligoclonality of human herpesvirus 8 terminal repeat sequences in Kaposi’s sarcoma and other diseases. J. Natl. Cancer Inst. 2000, 92, 729–736. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Whitby, D.; Boshoff, C.; Hatzioannou, T.; Weiss, R.A.; Schulz, T.F.; Howard, M.R.; Brink, N.S.; Tedder, R.S.; Tenant-Flowers, M.; Copas, A. Detection of Kaposi’s sarcoma-associated herpesvirus (KSHV) in peripheral blood of HIV-infected individuals predicts progression to Kaposi’s sarcoma. Lancet 1995, 364, 799–802. [Google Scholar] [CrossRef]
- Soulier, J.; Grollet, L.; Oksenhendler, E.; Cacoub, P.; Cazals-Hatem, D.; Babinet, P.; d’Agay, M.F.; Clauvel, J.P.; Raphael, M.; Degos, L. Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in multicentric Castleman’s disease. Blood 1995, 86, 1276–1280. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cesarman, E.; Chang, Y.; Moore, P.S.; Said, J.W.; Knowles, D.M. Kaposi’s sarcoma-associated herpesvirus-like DNA sequences in AIDS-related body-cavity-based lymphomas. N. Engl. J. Med. 1995, 332, 1186–1191. [Google Scholar] [CrossRef] [PubMed]
- Nador, R.G.; Cesarman, E.; Chadburn, A.; Dawson, D.B.; Ansari, M.Q.; Sald, J.; Knowles, D.M. Primary effusion lymphoma: A distinct clinicopathologic entity associated with the Kaposi’s sarcoma-associated herpes virus. Blood 1996, 88, 645–656. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Du, M.-Q.; Diss, T.C.; Liu, H.; Ye, H.; Hamoudi, R.A.; Cabeçadas, J.; Dong, H.Y.; Harris, N.L.; Chan, J.K.C.; Rees, J.W.; et al. KSHV- and EBV-associated germinotropic lymphoproliferative disorder. Blood 2002, 100, 3415–3418. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carbone, A.; Vaccher, E.; Gloghini, A.; Pantanowitz, L.; Abayomi, A.; de Paoli, P.; Franceschi, S. Diagnosis and management of lymphomas and other cancers in HIV-infected patients. Nat. Rev. Clin. Oncol. 2014, 11, 223–238. [Google Scholar] [CrossRef]
- Carbone, A.; Vaccher, E.; Gloghini, A. Hematological cancers in individuals infected by HIV. Blood 2021. [Google Scholar] [CrossRef]
- Kessler, E. Multicentric giant lymph node hyperplasia. A report of seven cases. Cancer 1985, 56, 2446–2451. [Google Scholar] [CrossRef]
- Van Rhee, F.; Oksenhendler, E.; Srkalovic, G.; Voorhees, P.; Lim, M.; Dispenzieri, A.; Ide, M.; Parente, S.; Schey, S.; Streetly, M.; et al. International evidence-based consensus diagnostic and treatment guidelines for unicentric Castleman disease. Blood Adv. 2020, 4, 6039–6050. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-W.; Pittaluga, S.; Jaffe, E.S. Multicentric Castleman disease: Where are we now? Semin. Diagn. Pathol. 2016, 33, 294–306. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vega, F.; Miranda, R.N.; Medeiros, L.J. KSHV/HHV8-positive large B-cell lymphomas and associated diseases: A heterogeneous group of lymphoproliferative processes with significant clinicopathological overlap. Mod. Pathol. 2019, 33, 18–28. [Google Scholar] [CrossRef] [PubMed]
- Gérard, L.; Bérezné, A.; Galicier, L.; Meignin, V.; Obadia, M.; de Castro, N.; Jacomet, C.; Verdon, R.; Madelaine-Chambrin, I.; Boulanger, E. Prospective Study of Rituximab in Chemotherapy-Dependent Human Immunodeficiency Virus Associated Multicentric Castleman’s Disease: ANRS 117 CastlemaB Trial. J. Clin. Oncol. 2007, 25, 3350–3356. [Google Scholar] [CrossRef] [PubMed]
- Uldrick, T.S.; Polizzotto, M.; Yarchoan, R. Recent advances in Kaposi sarcoma herpesvirus-associated multicentric Castleman disease. Curr. Opin. Oncol. 2012, 24, 495–505. [Google Scholar] [CrossRef] [PubMed]
- Bower, M.; Pria, A.D.; Coyle, C.; Nelson, M.; Naresh, K. Diagnostic Criteria Schemes for Multicentric Castleman Disease in 75 Cases. JAIDS J. Acquir. Immune Defic. Syndr. 2014, 65, e80–e82. [Google Scholar] [CrossRef]
- Knowles, D.M.; Inghirami, G.; Ubriaco, A.; Dalla-Favera, R. Molecular genetic analysis of three AIDS-associated neo-plasms of uncertain lineage demonstrates their B-cell derivation and the possible pathogenetic role of the Epstein-Barr virus. Blood 1989, 73, 792–799. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Simonelli, C.; Spina, M.; Cinelli, R.; Talamini, R.; Tedeschi, R.; Gloghini, A.; Vaccher, E.; Carbone, A.; Tirelli, U. Clinical features and outcome of primary effusion lymphoma in HIV-infected patients: A single-institution study. J. Clin. Oncol. 2003, 21, 3948–3954. [Google Scholar] [CrossRef]
- Riva, G.; Luppi, M.; Barozzi, P.; Forghieri, F.; Potenza, L. How I treat HHV8/KSHV-related diseases in posttransplant patients. Blood 2012, 120, 4150–4159. [Google Scholar] [CrossRef]
- Said, J.W.; Tasaka, T.; Takeuchi, S.; Asou, H.; de Vos, S.; Cesarman, E.; Knowles, D.M.; Koeffler, H.P. Primary effusion lymphoma in women: Report of two cases of Kaposi’s sarcoma herpes virus-associated effusion-based lymphoma in human immunodeficiency virus-negative women. Blood 1996, 88, 3124–3128. [Google Scholar] [CrossRef]
- Ascoli, V.; Scalzo, C.C.; Danese, C.; Vacca, K.; Pistilli, A.; Coco, F.L. Human herpes virus-8 associated primary effusion lymphoma of the pleural cavity in HIV-negative elderly men. Eur. Respir. J. 1999, 14, 1231–1234. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chadburn, A.; Hyjek, E.; Mathew, S.; Cesarman, E.; Said, J.; Knowles, D.M. KSHV-Positive solid lymphomas represent an extra-cavitary variant of primary effusion lymphoma. Am. J. Surg. Pathol. 2004, 28, 1401–1416. [Google Scholar] [CrossRef]
- Carbone, A.; Gloghini, A.; Vaccher, E.; Marchetti, G.; Gaidano, G.; Tirelli, U. KSHV/HHV-8 associated lymph node based lymphomas in HIV seronegative subjects. Report of two cases with anaplastic large cell morphology and plasmablastic immunophenotype. J. Clin. Pathol. 2005, 58, 1039–1045. [Google Scholar] [CrossRef]
- Aguilar, C.; Laberiano, C.; Beltran, B.; Diaz, C.; Taype-Rondan, A.; Castillo, J.J. Clinicopathologic characteristics and survival of patients with primary effusion lymphoma. Leuk. Lymphoma 2020, 61, 2093–2102. [Google Scholar] [CrossRef] [PubMed]
- Gérard, L.; Michot, J.M.; Burcheri, S.; Fieschi, C.; Longuet, P.; Delcey, V.; Meignin, V.; Agbalika, F.; Chevret, S.; Oksenhendler, E. Rituximab decreases the risk of lymphoma in patients with HIV-associated multicentric Castleman disease. Blood 2012, 119, 2228–2233. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oksenhendler, E.; Boulanger, E.; Galicier, L.; Du, M.Q.; Dupin, N.; Diss, T.C.; Hamoudi, R.; Daniel, M.T.; Agbalika, F.; Boshoff, C.; et al. High incidence of Kaposi sarcoma-associated herpesvirus-related non-Hodgkin lym-phoma in patients with HIV infection and multicentric Castleman disease. Blood 2002, 99, 2331–2336. [Google Scholar] [CrossRef]
- Pria, A.D.; Pinato, D.; Roe, J.; Naresh, K.; Nelson, M.; Bower, M. Relapse of HHV8-positive multicentric Castleman disease following rituximab-based therapy in HIV-positive patients. Blood 2017, 129, 2143–2147. [Google Scholar] [CrossRef] [Green Version]
- Zanelli, M.; Zizzo, M.; Bisagni, A.; Froio, E.; de Marco, L.; Valli, R.; Filosa, A.; Luminari, S.; Martino, G.; Massaro, F.; et al. Germinotropic lymphoproliferative disorder: A systematic review. Ann. Hematol. 2020, 99, 2243–2253. [Google Scholar] [CrossRef]
- Bower, M.; Powles, T.; Williams, S.; Davis, T.N.; Atkins, M.; Montoto, S.; Orkin, C.; Webb, A.; Fisher, M.; Nelson, M.; et al. Brief Communication: Rituximab in HIV-Associated Multicentric Castleman Disease. Ann. Intern. Med. 2007, 147, 836–839. [Google Scholar] [CrossRef] [PubMed]
- Hoffmann, C.; Schmid, H.; Müller, M.; Teutsch, C.; van Lunzen, J.; Esser, S.; Wolf, T.; Wyen, C.; Sabranski, M.; Horst, H.-A.; et al. Improved outcome with rituximab in patients with HIV-associated multicentric Castleman disease. Blood 2011, 118, 3499–3503. [Google Scholar] [CrossRef] [PubMed]
- Uldrick, T.S.; Polizzotto, M.; Aleman, K.; O’Mahony, D.; Wyvill, K.M.; Wang, V.; Marshall, V.; Pittaluga, S.; Steinberg, S.M.; Tosato, G.; et al. High-dose zidovudine plus valganciclovir for Kaposi sarcoma herpesvirus-associated multicentric Castleman disease: A pilot study of virus-activated cytotoxic therapy. Blood 2011, 117, 6977–6986. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Casper, C.; Nichols, W.G.; Huang, M.L.; Corey, L.; Wald, A. Remission of HHV-8 and HIV-associated multi-centric Castleman disease with ganciclovir treatment. Blood 2004, 103, 1632–1634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Oksenhendler, E.; Duarte, M.; Soulier, J.; Cacoub, P.; Welker, Y.; Cadranel, J.; Cazals-Hatem, D.; Autran, B.; Clauvel, J.P.; Raphael, M. Multicentric Castleman’s disease in HIV infection: A clinical and pathological study of 20 patients. AIDS 1996, 10, 61–67. [Google Scholar] [CrossRef] [PubMed]
- Berezne, A.; Agbalika, F.; Oksenhendler, E. Failure of cidofovir in HIV-associated multicentric Castleman disease. Blood 2004, 103, 4368–4369. [Google Scholar] [CrossRef] [Green Version]
- Senanayake, S.; Kelly, J.; Lloyd, A.; Waliuzzaman, Z.; Goldstein, D.; Rawlinson, W. Multicentric Castleman’s disease treated with antivirals and immunosuppressants. J. Med. Virol. 2003, 71, 399–403. [Google Scholar] [CrossRef]
- Ramaswami, R.; Lurain, K.; Polizzotto, M.N.; Ekwede, I.; Waldon, K.; Steinberg, S.M.; Mangusan, R.; Widell, A.; Rupert, A.; George, J.; et al. Characteristics and outcomes of KSHV-associated multicentric Castleman disease with or without other KSHV diseases. Blood Adv. 2021, 5, 1660–1670. [Google Scholar] [CrossRef]
- Ramaswami, R.; Lurain, K.; Peer, C.J.; Serquiña, A.; Wang, V.Y.; Widell, A.; Goncalves, P.; Steinberg, S.M.; Marshall, V.; George, J.; et al. Tocilizumab in patients with symptomatic Kaposi sarcoma herpesvirus–associated multicentric Castleman disease. Blood 2020, 135, 2316–2319. [Google Scholar] [CrossRef]
- Writing Group; Bower, M.; Palfreeman, A.; Alfa-Wali, M.; Bunker, C.; Burns, F.; Churchill, D.; Collins, S.; Cwynarski, K.; Edwards, S.; et al. British HIV Association guidelines for HIV-associated malignancies 2014. HIV Med. 2014, 15, 1–92. [Google Scholar] [CrossRef]
- Guillet, S.; Gérard, L.; Meignin, V.; Agbalika, F.; Cuccini, W.; Denis, B.; Katlama, C.; Galicier, L.; Oksenhendler, E. Classic and extracavitary primary effusion lymphoma in 51 HIV-infected patients from a single institution. Am. J. Hematol. 2016, 91, 233–237. [Google Scholar] [CrossRef] [Green Version]
- Lurain, K.; Polizzotto, M.N.; Aleman, K.; Bhutani, M.; Wyvill, K.M.; Gonçalves, P.H.; Ramaswami, R.; Marshall, V.A.; Miley, W.; Steinberg, S.M.; et al. Viral, immunologic, and clinical features of primary effusion lymphoma. Blood 2019, 133, 1753–1761. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Olszewski, A.J.; Fallah, J.; Castillo, J.J. Human immunodeficiency virus-associated lymphomas in the antiretroviral therapy era: Analysis of the National Cancer Data Base. Cancer 2016, 122, 2689–2697. [Google Scholar] [CrossRef] [PubMed]
- El-Fattah, M.A. Clinical characteristics and survival outcome of primary effusion lymphoma: A review of 105 patients. Hematol. Oncol. 2017, 35, 878–883. [Google Scholar] [CrossRef] [PubMed]
- Shimada, K.; Hayakawa, F.; Kiyoi, H. Biology and management of primary effusion lymphoma. Blood 2018, 132, 1879–1888. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Castillo, J.J.; Winer, E.S.; Stachurski, D.; Perez, K.; Jabbour, M.; Milani, C.; Colvin, G.; Butera, J.N. Prognostic Factors in Chemotherapy-Treated Patients with HIV-Associated Plasmablastic Lymphoma. Oncology 2010, 15, 293–299. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ronaghy, A.; Wang, H.-Y.; Thorson, J.A.; Medeiros, L.J.; Xie, Y.; Zhang, X.; Sheikh-Fayyaz, S. PD-L1 and Notch1 expression in KSHV/HHV-8 and EBV associated germinotropic lymphoproliferative disorder: Case report and review of the literature. Pathology 2017, 49, 430–435. [Google Scholar] [CrossRef]
| CD20 | IRF4/MUM1 | CD138 | Other Positive Cell Markers | |
|---|---|---|---|---|
| Classic PEL | Negative | Positive | Positive | CD30, CD31, CD71, EMA |
| Solid PEL | Negative | Positive | Positive | CD30, EMA |
| KSHV/HHV8-MCD-DLBCL | Positive (may be lost) | Positive | Positive | Lambda light chain, CD45 |
| KSHV/HHV8-MCD | Negative | Positive | Negative | CD38, IgM, Lambda light chain |
| GLPD | Negative | Positive | Positive (may be lost) | Monotypic light chain |
| EBV Infection (Frequency) | KSHV/HHV8 Infection | Genetic Features | |
|---|---|---|---|
| Classic PEL | Positive (80–100%) | Positive (100%) | Complex karyotype, no recurrent translocation, Tp53 and RAS rarely mutated |
| Solid PEL | Positive (80–100%) | Positive (100%) | Occasional p53 positive cells |
| KSHV/HHV8-MCD-DLBCL | Negative | Positive (100%) | Myc rearrangement, TP53 point mutation |
| KSHV/HHV8-MCD | Negative | Positive (100%) | |
| GLPD | Positive (100%) | Positive (100%) |
| ANRS Criteria | NCI Criteria |
|---|---|
| Fever | Fatigue CTAE grade > 1 |
| C-reactive protein > 20 mg/L in the absence of any other aetiology | Fever or night sweats |
| Peripheral lymphadenopathy | Weight loss |
| Enlarged spleen | Respiratory symptoms |
| Oedema | Gastrointestinal symptoms |
| Pleural effusion | Neurological symptoms |
| Ascites | Oedema or effusion |
| Cough | Xerostomia |
| Nasal obstruction | Rash |
| Xerostomia | |
| Rash | |
| Central neurologic symptoms | Anaemia (Hb < 12 g/dL) |
| Jaundice | Thrombocytopenia (<100 × 109/L) |
| Autoimmune haemolytic anaemia | Hypoalbuminemia (<35 g/L) |
| Serum CRP > 3 mg/L |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bower, M.; Carbone, A. KSHV/HHV8-Associated Lymphoproliferative Disorders: Lessons Learnt from People Living with HIV. Hemato 2021, 2, 703-712. https://doi.org/10.3390/hemato2040047
Bower M, Carbone A. KSHV/HHV8-Associated Lymphoproliferative Disorders: Lessons Learnt from People Living with HIV. Hemato. 2021; 2(4):703-712. https://doi.org/10.3390/hemato2040047
Chicago/Turabian StyleBower, Mark, and Antonino Carbone. 2021. "KSHV/HHV8-Associated Lymphoproliferative Disorders: Lessons Learnt from People Living with HIV" Hemato 2, no. 4: 703-712. https://doi.org/10.3390/hemato2040047
APA StyleBower, M., & Carbone, A. (2021). KSHV/HHV8-Associated Lymphoproliferative Disorders: Lessons Learnt from People Living with HIV. Hemato, 2(4), 703-712. https://doi.org/10.3390/hemato2040047







