Clinical Evaluation of AMNIODERM+® Wound Dressing Containing Non-Viable Human Amniotic Membrane: Retrospective-Perspective Clinical Trial
Abstract
1. Introduction
2. Materials and Methods
2.1. New Medical Device AMNIODERM+®
2.2. Mechanism of Action
- Protection from external environment.
- Prevention of liquid evaporation.
- Prevention of microorganisms from entering the wound.
2.3. Method of Conducting the Study and Its Evaluation
3. Results
3.1. Primary Endpoint
3.2. Secondary Endpoints
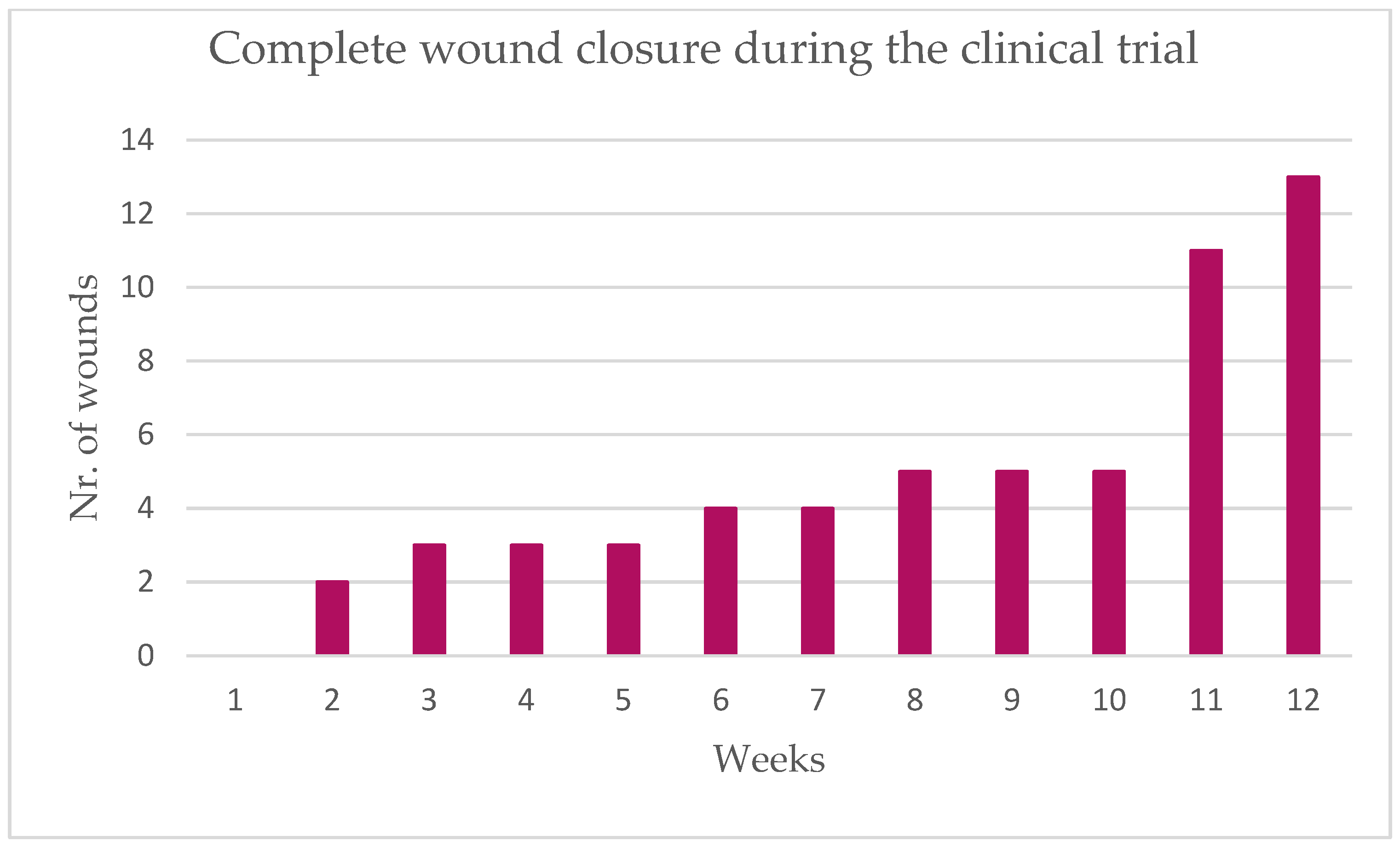
- Wound closure after 12 weeks was observed in 13 patients, which equals 65.0% (95% CI: 40.8–84.6%). Compared to the null hypothesis that wound closure would be observed in 50% of patients, there was no significant difference (p = 263; one-sample exact binomial test; Table 3).
- Time to complete wound closure was 11.4 weeks (95% CI: 8.3 months—upper confidence limit not reached).
- The proportion of patients with complete wound closure each week is described in Table 3.
- Subject satisfaction and user assessment at the final visit is described in Table 4.
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Baena-Díez, J.M.; Peñafiel, J.; Subirana, I.; Ramos, R.; Elosua, R.; Marín-Ibañez, A.; Grau, M. Risk of Cause-Specific Death in Individuals With Diabetes: A Competing Risks Analysis. Diabetes Care 2016, 39, 1987–1995. [Google Scholar] [CrossRef] [PubMed]
- World Health Organization. Global Health Risks: Mortality and Burden of Disease Attributable to Selected Major Risks; World Health Organization: Geneva, Switzerland, 2009; Available online: https://books.google.cz/books?hl=cs&lr=&id=Ycbr2e2WPdcC&oi=fnd&pg=PR5&ots=ahG2VyYdas&sig=1h6IOwVB9ZsV67QfEWDiL_Q3Ffs&redir_esc=y#v=onepage&q&f=false (accessed on 29 August 2022).
- Grennan, D. Diabetic Foot Ulcers. JAMA 2019, 321, 114. [Google Scholar] [CrossRef]
- Cho, N.H.; Shaw, J.E.; Karuranga, S.; Huang, Y.; da Rocha Fernandes, J.D.; Ohlrogge, A.W.; Malanda, B.I.D.F. IDF diabetes atlas: Global estimates of diabetes prevalence for 2017 and projections for 2045. Diabetes Res. Clin. Pract. 2018, 138, 271–281. [Google Scholar] [CrossRef] [PubMed]
- Moulik, P.K.; Mtonga, R.; Gill, G.V. Amputation and mortality in new-onset diabetic foot ulcers stratified by etiology. Diabetes Care 2003, 26, 491–494. [Google Scholar] [CrossRef] [PubMed]
- Lipsky, B.A.; Berendt, A.R.; Cornia, P.B.; Pile, J.C.; Peters, E.J.; Armstrong, D.G.; Deery, H.G.; Embil, J.M.; Joseph, W.S.; Karchmer, A.W. Infectious Diseases Society of America clinical practice guideline for the diagnosis and treatment of diabetic foot infections. Clin. Infect. Dis. 2012, 54, e132–e173. [Google Scholar] [CrossRef]
- Everett, E.; Mathioudakis, N. Update on management of diabetic foot ulcers. Ann. N. Y. Acad. Sci. 2018, 1411, 153–165. [Google Scholar] [CrossRef]
- Mulder, G.; Tenenhaus, M.; D’Souza, G.F. Reduction of Diabetic Foot Ulcer Healing Times through Use of Advanced Treatment Modalities. Int. J. Low. Extrem. Wounds 2014, 13, 335–346. [Google Scholar] [CrossRef]
- Schmiedova, I.; Ozanova, Z.; Stastna, E.; Kiselakova, L.; Lipovy, B.; Forostyak, S. Case Report: Freeze-Dried Human Amniotic Membrane Allograft for the Treatment of Chronic Wounds: Results of a Multicentre Observational Study. Front. Bioeng. Biotechnol. 2021, 9, 649446. [Google Scholar] [CrossRef]
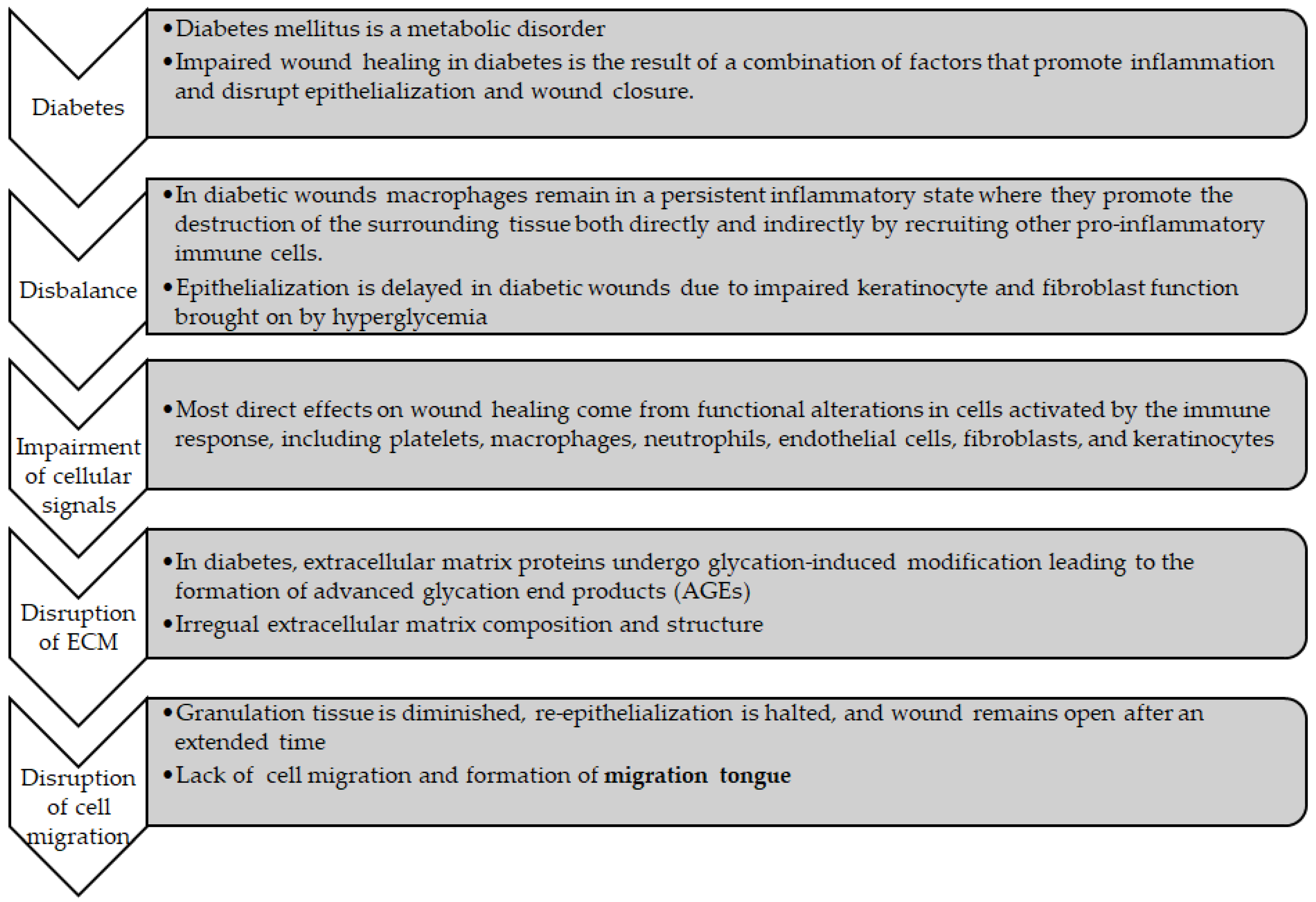
- Burgess, J.L.; Wyant, W.A.; Abdo Abujamra, B.; Kirsner, R.S.; Jozic, I. Diabetic Wound-Healing Science. Medicina 2021, 57, 1072. [Google Scholar] [CrossRef]
- O’Toole, E.A. Extracellular matrix and keratinocyte migration. Clin. Exp. Dermatol. 2001, 26, 525–530. [Google Scholar] [CrossRef]
- Michopoulou, A.; Rousselle, P. How do epidermal matrix metalloproteinases support re-epithelialization during skin healing? Eur. J. Dermatol. 2015, 25, 33–42. [Google Scholar] [CrossRef] [PubMed]
- Solarte, D.V.A.; Güiza-Argüello, V.R.; Arango-Rodríguez, M.L.; Sossa, C.L.; Becerra-Bayona, S.M. Decellularized Tissues for Wound Healing: Towards Closing the Gap Between Scaffold Design and Effective Extracellular Matrix Remodeling. Front. Bioeng. Biotechnol. 2022, 10, 821852. [Google Scholar] [CrossRef] [PubMed]
- Eming, S.A.; Koch, M.; Krieger, A.; Brachvogel, B.; Kreft, S.; Bruckner-Tuderman, L.; Fox, J.W. Differential Proteomic Analysis Distinguishes Tissue Repair Biomarker Signatures in Wound Exudates Obtained from Normal Healing and Chronic Wounds. J. Proteome Res. 2010, 9, 4758–4766. [Google Scholar] [CrossRef]
- Goldin, A. Advanced Glycation End Products: Sparking the Development of Diabetic Vascular Injury. Circulation 2006, 114, 597–605. [Google Scholar] [CrossRef]
- Leaper, D.J.; Schultz, G.; Carville, K.; Fletcher, J.; Swanson, T.; Drake, R. Extending the TIME concept: What have we learned in the past 10 years?(*). Int. Wound J. 2012, 9 (Suppl. S2), 1–19. [Google Scholar] [CrossRef]
- Mohammed, Y.A.; Farouk, H.K.; Gbreel, M.I.; Ali, A.M.; Salah, A.A.; Nourelden, A.Z.; Gawad, M.M.A.E. Human amniotic membrane products for patients with diabetic foot ulcers. Do they help? A systematic review and meta-analysis. J. Foot Ankle Res. 2022, 15, 71. [Google Scholar] [CrossRef]
- DiDomenico, L.A.; Orgill, D.P.; Galiano, R.D.; Serena, T.E.; Carter, M.J.; Kaufman, J.P.; Young, N.J.; Zelen, C.M. Aseptically Processed Placental Membrane Improves Healing of Diabetic Foot Ulcerations: Prospective, Randomized Clinical Trial. Plast. Reconstr. Surg. Glob. Open 2016, 4, e1095. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Jones, R.R.; Mi, S.; Foster, J.; Alcock, S.G.; Hamley, I.W.; Connon, C.J. The mechanical properties of amniotic membrane influence its effect as a biomaterial for ocular surface repair. Soft Matter 2012, 8, 8379. [Google Scholar] [CrossRef]
- Buerzle, W.; Mazza, E. On the deformation behavior of human amnion. J. Biomech. 2013, 46, 1777–1783. [Google Scholar] [CrossRef]
- Grémare, A.; Jean-Gilles, S.; Musqui, P.; Magnan, L.; Torres, Y.; Fenelon, M.; Brun, S.; Fricain, J.C.; L’heureux, N. Cartography of the mechanical properties of the human amniotic membrane. J. Mech. Behav. Biomed. Mater. 2019, 99, 18–26. [Google Scholar] [CrossRef]
- Ilic, D.; Vicovac, L.; Nikolic, M.; Lazic Ilic, E. Human amniotic membrane grafts in therapy of chronic non-healing wounds. Br. Med. Bull. 2016, 117, 59–67. [Google Scholar] [CrossRef] [PubMed]
- Enoch, S.; Leaper, D.J. Basic science of wound healing. Surgery 2008, 26, 31–37. [Google Scholar] [CrossRef]
- Frykberg, R.G.; Banks, J. Challenges in the Treatment of Chronic Wounds. Adv. Wound Care 2015, 4, 560–582. [Google Scholar] [CrossRef]
- Castellanos, G.; Bernabé-García, Á.; Moraleda, J.M.; Nicolás, F.J. Amniotic membrane application for the healing of chronic wounds and ulcers. Placenta 2017, 59, 146–153. [Google Scholar] [CrossRef] [PubMed]

| AMNIODERM+® Composition | ||
|---|---|---|
| The Component | Description of the Component | The Purpose of the Component |
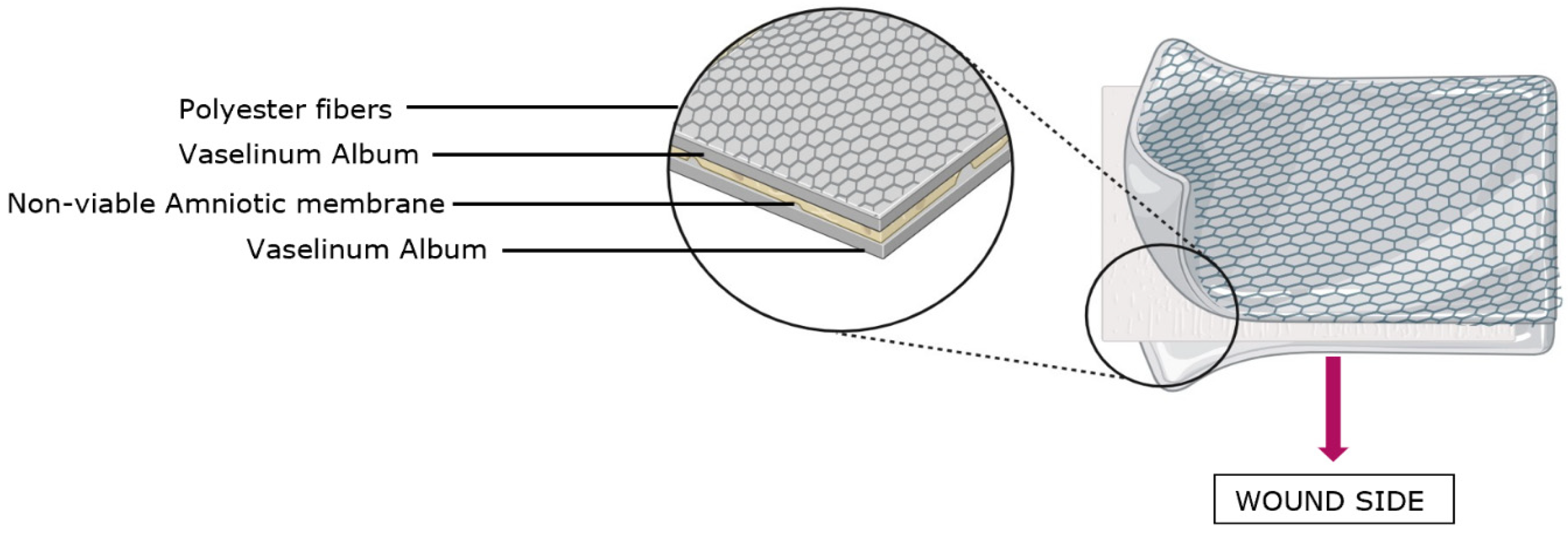
| Polyester fabric layer | The outer structural layer is a highly porous knitted fabric manufactured from 100% polyester fibers. This fabric is flexible, enabling the entire dressing to conform to the wound, but also provides support to allow the entire wound dressing to be repositioned and removed, as desired. The polyester fibers do not grow into the newly formed tissue, which allows for extended dressing application times. | As this component is flexible, it serves the purpose of enabling the entire dressing to conform to the wound, but also provides support to allow the entire wound dressing to be repositioned and removed, as desired. |
| White petroleum jelly (Vaselinum album) layer | This non-structural layer comprises white petroleum jelly (Vaselinum album), which acts as a protective film over the wound. This hydrophobic layer provides the main action and microbial protection, and minimizes moisture loss, enabling a favorable moist wound-healing environment. The white petrolatum provides an occlusive layer that reduces the risk of wound dehydration, thus helping to prevent scab formation and promoting faster healing. | The purpose of this hydrophobic layer is to provide the main action, microbial protection, and minimize moisture loss, enabling a favorable moist wound-healing environment. |
| Non-viable amniotic tissue (HAM) layer | This layer is made from deactivated non-viable amniotic tissue and provides an ancillary action assuring that the wound dressing conforms to the surface of the wound, including the wound edges, supporting physical protection of the device to the wound bed. The amniotic tissue layer also provides physical support for the middle layer, which comprises white petroleum jelly. It is important to note that while the non-viable HAM layer in this context does not contribute to the AMNIODERM+® wound dressing with its traditional biological functions due to its non-viable state, it still adds an exceptional mechanical dimension to the wound dressing. The combination of the non-viable HAM scaffold coated by Vaselinum album serves as a supportive platform that complements the body’s natural healing mechanisms. | The purpose of this component is to add an exceptional mechanical dimension to the wound dressing. The combination of the non-viable HAM scaffold coated by Vaselinum album serves as a supportive platform that complements the body’s natural healing mechanisms. |
| Objectives | Primary Endpoint | The primary endpoint is a percentage reduction in the wound area after 12 weeks. |
| Secondary Endpoint | • A proportion of subjects with a complete wound closure (based on investigator’s decision) after 12 weeks. • Time to complete wound closure. Closed wound is defined as follows: the skin remains intact, and the tissue that lies underneath is not exposed. • The proportion of subjects with complete wound closure each week. • The percentage reduction in the wound area each week. • The Numeric Rating Scale (NRS-11) during 12 weeks. • Foot Health Status Questionnaire (FHSQ) during 12 weeks. • Subject satisfaction and user assessment at the final visit. | |
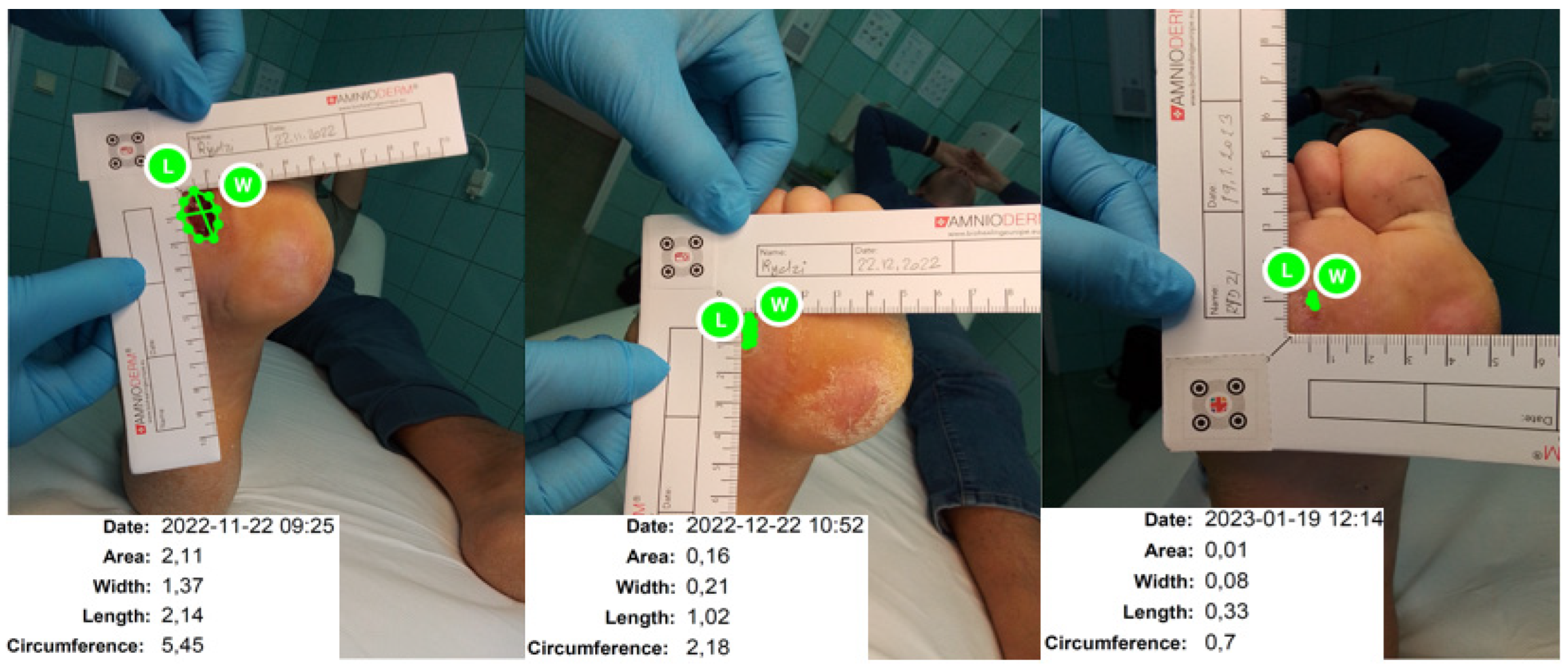
| Methodology | Process of data collection All assessments must take place at the investigational site. The subjects who fully recovered before the planned Final Visit (wound closure = defined as the skin remains intact and the tissue that lies underneath is not exposed, based on investigator´s decision) will be terminated. Their data from the final visit will be processed according to the last carried forward principle. Assessment of change Documentation of the wound area and circumference from a photograph of the wound with an adjacently placed ruler using the Imito® application. Assessment using Leg Ulcer Measurement Tool [17] | |
| Number of patients | Planned: | 20 |
| Analyzed: | 20 | |
| Diagnosis and main criteria for inclusion | Subjects with a diabetic neuropathic or neuroischemic wounds, anywhere on the leg, ranging in size between 2 cm2 and 16 cm2, that has not healed at least 20% after the 6 weeks of the (SoC) with the known therapeutic history ≥6 weeks and ≤104 weeks prior to clinical trial inclusion. | |
| Test product, dose, and mode of administration | The wound must be cleaned from infection and necrotic tissue before the subject’s enrolment. During the initial period, the revision of the wound will be conducted once a week. However, the frequency of visits can be adjusted if the earlier change in the dressing is needed. A change of new AMNIODERM+® wound dressing is expected to be done once a week at each visit, but the change of the dressing is not obligatory and depends on the wound condition. The fixation of AMNIODERM+® will be conducted using a secondary dressing with (e.g., Mepitel One (Mölnlycke Health Care s.r.o., Karvina, Czech Republic), Adaptic (Systagenix Wound Management Limited., Berkshire, England) or Silflex (Advancis Medical, Nottingham, United Kingdom)). The primary cover can be attached to the wound area with a soft patch such as COSMOPOR. | |
| Duration of treatment | The planned maximal length of participation of one subject is 12 weeks = 84 days. The expected overall duration of the trial (FSI to LSO) is 6 months. The subject may cancel his/her participation without giving the reason at any time. | |
| Reference therapy | SoC during 6 weeks prior to AMNIODERM+® wound treatment. | |
| Criteria for evaluation | Efficacy | Percentage reduction after 12 weeks of treatment is at least 20%. |
| Safety | Serious adverse events, adverse events, and device deficiency were collected in eCRF. | |
| Statistical methods | Primary hypothesis was tested using a two-sided one-sample Wilcoxon test. The reference value of a reduction in the wound area was 20%. The proportion of subjects with a complete wound closure after 12 weeks was analyzed using a one-sample binomial test. The reference value was 50%. Standard descriptive statistics were used in the analysis. Categorical parameters were described using absolute and relative frequencies. Relative frequencies were calculated from the non-missing data. Continuous parameters were described using the mean with standard deviation (SD) and 95% confidence interval (CI) for mean and with median supplemented by range. Time to wound closure was computed using Kaplan–Meier analysis (patients without complete wound closure after week 12 were censored at 12 weeks). | |
| N | % | 95% CI for Proportion | p Value 1 | |
|---|---|---|---|---|
| Number of patients | 20 | 100 | ||
| Complete wound closure after 12 weeks | 13 | 65.0 | 40.8; 84.6 | 0.263 |
| Complete wound closure each week | ||||
| Week 1 | 0 | 0 | 0.0; 16.8 | |
| Week 2 | 2 | 10.0 | 1.2; 31.7 | |
| Week 3 | 3 | 15.0 | 3.2; 37.9 | |
| Week 4 | 3 | 15.0 | 3.2; 37.9 | |
| Week 5 | 3 | 15.0 | 3.2; 37.9 | |
| Week 6 | 4 | 20.0 | 5.7; 43.7 | |
| Week 7 | 4 | 20.0 | 5.7; 43.7 | |
| Week 8 | 5 | 25.0 | 8.7; 49.1 | |
| Week 9 | 5 | 25.0 | 8.7; 49.1 | |
| Week 10 | 5 | 25.0 | 8.7; 49.1 | |
| Week 11 | 11 | 55.0 | 31.5; 76.9 | |
| Week 12 | 13 | 65.0 | 40.8; 84.6 |
| N | % | |
|---|---|---|
| Number of patients | 20 | 100 |
| The application process of AMNIODERM+® was no big deal for me | ||
| I totally agree | 14 | 70.0 |
| I agree | 6 | 30.0 |
| Neither agree or disagree | 0 | 0 |
| I disagree | 0 | 0 |
| I totally disagree | 0 | 0 |
| I did not notice higher discomfort while wearing the AMNIODEM+® | ||
| I totally agree | 14 | 70.0 |
| I agree | 6 | 30.0 |
| Neither agree or disagree | 0 | 0 |
| I disagree | 0 | 0 |
| I totally disagree | 0 | 0 |
| Please evaluate your total satisfaction with AMNIODERM+® management | ||
| Very satisfied | 17 | 85.0 |
| Satisfied | 3 | 15.0 |
| Unsatisfied | 0 | 0 |
| Very unsatisfied | 0 | 0 |
| Please evaluate the manipulation with AMNIODERM+® | ||
| Excellent | 14 | 70.0 |
| Very good | 6 | 30.0 |
| Good | 0 | 0 |
| Acceptable | 0 | 0 |
| Poor | 0 | 0 |
| Insufficient | 0 | 0 |
| Please assess the improvement of the wound condition with AMNIODERM+® | ||
| Excellent | 20 | 100 |
| Very good | 0 | 0 |
| Good | 0 | 0 |
| Acceptable | 0 | 0 |
| Poor | 0 | 0 |
| Insufficient | 0 | 0 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Schmiedova, I.; Slama, P.; Dembickaja, A.; Kozova, B.; Hyneckova, V.; Gogolkova, S.; Stastna, E.; Zahradnicek, M.; Savic, S.; Davani, A.; et al. Clinical Evaluation of AMNIODERM+® Wound Dressing Containing Non-Viable Human Amniotic Membrane: Retrospective-Perspective Clinical Trial. BioTech 2024, 13, 36. https://doi.org/10.3390/biotech13030036
Schmiedova I, Slama P, Dembickaja A, Kozova B, Hyneckova V, Gogolkova S, Stastna E, Zahradnicek M, Savic S, Davani A, et al. Clinical Evaluation of AMNIODERM+® Wound Dressing Containing Non-Viable Human Amniotic Membrane: Retrospective-Perspective Clinical Trial. BioTech. 2024; 13(3):36. https://doi.org/10.3390/biotech13030036
Chicago/Turabian StyleSchmiedova, Iveta, Petr Slama, Alena Dembickaja, Beata Kozova, Vendula Hyneckova, Sona Gogolkova, Elen Stastna, Michal Zahradnicek, Stefan Savic, Arash Davani, and et al. 2024. "Clinical Evaluation of AMNIODERM+® Wound Dressing Containing Non-Viable Human Amniotic Membrane: Retrospective-Perspective Clinical Trial" BioTech 13, no. 3: 36. https://doi.org/10.3390/biotech13030036
APA StyleSchmiedova, I., Slama, P., Dembickaja, A., Kozova, B., Hyneckova, V., Gogolkova, S., Stastna, E., Zahradnicek, M., Savic, S., Davani, A., Hulo, E., & Martinka, E. (2024). Clinical Evaluation of AMNIODERM+® Wound Dressing Containing Non-Viable Human Amniotic Membrane: Retrospective-Perspective Clinical Trial. BioTech, 13(3), 36. https://doi.org/10.3390/biotech13030036






