1. Introduction
Behaviour is considered a relevant and sensitive signal that informs us of individuals’ specific preferences, requirements, and internal states that correspond to their welfare and subjective wellbeing [
1,
2,
3,
4]. It is well established that postnatal environmental factors during development play a significant role in determining behavioural responses [
5]. Amongst these factors is the early social rearing environment, which is often manipulated within zoological institutions for conservation purposes [
6]. As a result, the postnatal rearing conditions for captive young tend to vary. Parent-rearing (i.e., by the natural parents of the offspring), hand-rearing (i.e., by a human caregiver), peer-rearing (i.e., reared in the presence of same age peers), foster-rearing, and cross foster-rearing are common practice, and it is important to understand their unique impact on ontogenetic behaviour. Foster-rearing and cross foster-foster rearing refers to “rearing non-maternal young by intraspecific or interspecific surrogate parents”, respectively [
7].
The literature investigating the behavioural impact of the early social rearing environment has mostly documented the developmental effects of hand-rearing. Hand-reared birds have shown altered behavioural patterns including increased abnormal and stereotypic behaviours (parrots, Psittaciformes [
8]), increased aggression (parrots [
9]), reduced vigilance (cranes, Gruidae [
10,
11,
12], and reduced neophobia (parrots [
13]; starlings, Sturnidae [
14,
15]).
Altered patterns of associations are also evident, with hand-reared lovebirds (
Agapornis sp.) showing an inability to successfully integrate with conspecifics [
16] and hand-reared ravens (
Corvus corax) possessing larger quantities of less meaningful relationships [
17]. Birds reared in the presence of same-age peers (i.e., peer-reared) have also shown behavioural differences when compared to parent-reared conspecifics (e.g., in starlings,
Sturnus vulgaris [
18]; canaries,
Serinus canaria domestica [
19]; and zebra finches,
Taeniopygia guttata [
20]).
The behavioural alterations incurred through hand-rearing and peer-rearing practices suggests that secure, maternal bonds promote the development of relevant and adaptive behaviours [
21]. As such, foster-rearing and cross foster-rearing, which may provide a suitable alternative biologically relevant social environments, presents themselves as useful husbandry practices in zoological institutions [
22]. Cross-fostering has been used extensively in captivity with many species, to let experienced parents care for abandoned young or to give inexperienced parents a chance to learn how to raise young [
23]. This has benefited conservation efforts in rodents [
24], marsupials [
25], and birds [
26] to increase the productivity of breeding individuals in managed programmes.
Although its use is extensive, few reports have investigated the developmental impacts of foster rearing to the same extent as the hand-rearing and peer-rearing literature. Studies that use foster-rearing to assist with conservation efforts typically utilise survival analyses as a measure of success (e.g., [
7,
27,
28,
29]). Although survival prerequisites the appropriateness of husbandry manipulations, it does not necessarily equate to the development of adaptive behaviours or positive welfare. Altered patterns of behaviour and sociality induced by disruptions of the early rearing environment are likely to go unnoticed by survival analyses (Beck 2002 as cited in [
12]). A research bias toward primates and megafauna within this literature also hinders our understanding of the welfare needs across taxa [
3].
Where behavioural analyses have been conducted on birds, cross-fostering has shown to result in misprinting (i.e., chicks show similarities and preferences to their foster species). Misimprinting in blue tits (
Cyanistes caeruleus) and great tits (
Parus major) is well documented for several behaviours, including mate choice [
30], aggression [
31], social dominance [
32], alarm calls [
33], bird song [
34], and paternal behaviour [
35]. Similar findings for communication and mate preference are demonstrated research on male zebra finches cross-fostered to Bengalese finches (
Lonchura striata var.
domestica) [
36,
37,
38]. Consequently, where cross fostering is used as a husbandry tool, monitoring of the behavioural development throughout the juvenile stages into adulthood should be performed to evaluate the efficacy of this procedure.
Indications that the early rearing environment influences the development of social networks is evident in sandhill cranes (
Antigone canadensis) who show association preferences for their foster species rather than conspecifics [
23]. Foster-reared Hawaiian goose (nene) goslings (
Branta sandvicensis) were less able to integrate and adjust into new environments compared to parent-reared conspecifics [
39]. Foster-reared goslings displayed longer latencies to associate with flock members and performed behaviours indicative of an inferior antipredator strategy (e.g., reduced vigilance and predator avoidance) [
39]. However, these foster-reared goslings only had visual and auditory contact with their foster parents due to a mesh wire barrier. Parent-reared goslings had more social opportunities and were housed in larger enclosures. Hence, full maternal care was precluded and environmental conditions were not consistent between rearing practices.
When interactions with parents and exploration opportunities have been provided, no differences in the activity patterns and associations can be seen, as highlighted by research comparing the development of captive killdeer (
Charadrius vociferous) cross-fostered to wild spotted sandpipers (
Actitis macularia) and wild parent-reared killdeer [
40] (
Charadrius vociferous); Nonetheless, the use of fostering and cross-fostering alike has expected and unexpected fitness outcomes [
41]. As a result, authors have advised against, and limited, the use of fostering as a husbandry technique for reintroduction programmes; due to the potential for unsuitable coping strategies and mate choice both in the wild and in captivity (e.g., [
23,
40,
42]). However, the use of foster rearing techniques can be widespread for particular species across and within zoos. To fully understand the long term of effects of such strategies, consistent methods and repeatable data collection techniques need to be applied to the young animals involved and provide a contemporary investigation of behavioural development.
Captive flamingos offer excellent opportunities for strong empirical behavioural research due to their large sample sizes, diverse behavioural repertoire, ability to identify ringed individuals, and their global captive presence permitting research replication [
43]. As such, this study investigates how disruption of the early rearing environment affects the behavioural development of Chilean flamingo (
Phoenicopterus chilensis) chick’s cross-fostered to Andean flamingos (
Phoenicoparrus andinus) at WWT Slimbridge Wetland Centre. This unique opportunity to collect data on an understudied area of zoo management became apparent when a sudden and unexpected breeding event in the group of Andean flamingos occurred. Behavioural differences between Andean and Chilean flamingos are few and the two species potentially have a close evolutionary relationship [
44]. Food selectivity varies between the two, and Chilean flamingo chicks show looser social bonds out of the breeding season [
45,
46,
47,
48]. Differences between these two close taxonomic relatives are most prominent in their visual characteristics as adults (
Figure 1).
There is potential that these behavioural and physiological differences may manifest into altered behavioural repertoires and social preferences in cross-fostered Chilean flamingo chicks. We aimed to use well established welfare measures of captive flamingo activity and enclosure usage (e.g., [
48]), as well as social network analyses (e.g., [
50,
51]) to compare temporal patterns of behaviour and sociality of cross-fostered Chilean flamingo chicks with that of parent-reared Chilean flamingo chicks housed in the same enclosure. Chilean flamingos are a commonly housed zoo flamingo, whereas the Andean flamingo is not (being held, as of 2021 at two institutions). Although husbandry enclosure factors were similar for these two study flocks, limited information on captive Andean flamingo breeding behaviour means that no prior assumptions on the behavioural development of cross fostered chicks were made. Our findings have the potential to inform flamingo husbandry regimes, with an aim of helping produce self-sustaining populations across many zoological institutions. Our results also provide a baseline of flamingo behavioural development that are useful for others wishing to evaluate breeding behaviour in captive flocks.
3. Results
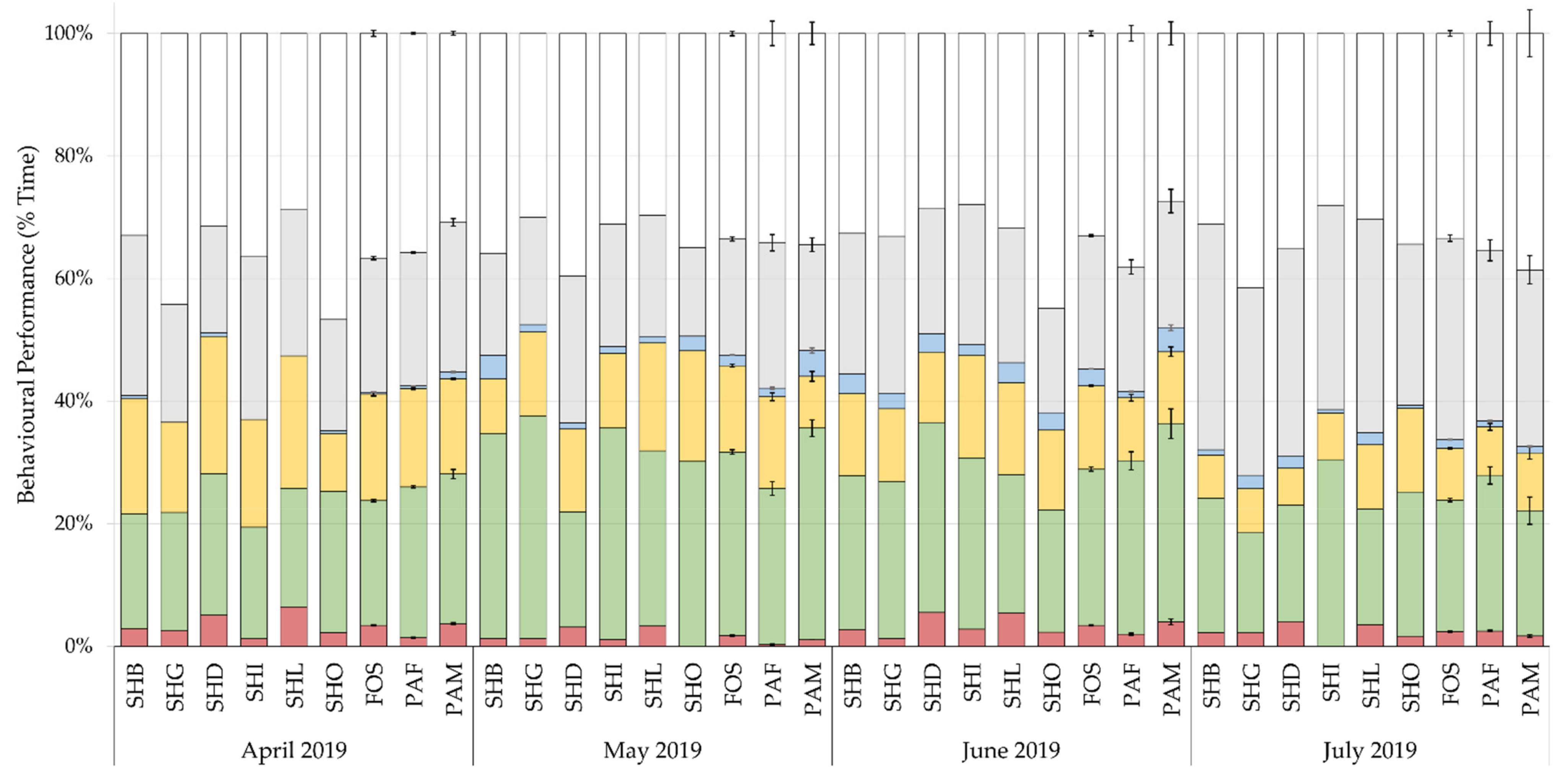
3.1. Activity
Differences in the development of chick behaviour by month are shown in
Figure 3. Chicks spent most of their time resting, 35%, preening, 24%, and feeding, 25%. The effect of rearing background, sex, climate, and visitors with the random slope, explained 1.8 to 12.4% of the variance in key state behaviours, with rearing background accounting for 0.3% to 4.1% of the variance. Model fit values are displayed in
Appendix A.
Key state behaviour model comparison results are displayed in
Table 2. FC models for each key state behaviour performed significantly better than null models,
X2 s (10, N = 8059) ≥ 22.01,
p’s ≤ 0.015. RO models performed significantly better than null models when explaining feeding and movement behaviour,
X2 s (1, N = 8059) ≥ 4.37,
p’s ≤ 0.037. FC models performed significantly better than FC-R models when explaining feeding behaviour,
X2 (2, N = 8059) = 6.21,
p = 0.045. TO models performed significantly better than the null model when explaining preening and movement behaviour,
X2 s(1, N = 8059) ≥ 11.24,
p’s ≤ 0.001.
All fixed factor outputs for behaviour are displayed in
Appendix B. Foster-reared chicks were 0.829 times less likely to feed than parent-reared chicks, OR = 0.829 (95% CI: 0.703, 0.978), β = −0.187, SE = 0.084,
p = 0.026. Female chicks were 0.405 (95% CI: 0.246, 0.666) times less likely to perform other behaviours than male chicks, β = −0.904, SE = 0.254,
p < 0.001.
A unit increase in time increased the odds of preening, β = 0.206, SE = 0.039, p < 0.001, OR = 1.228 (95% CI: 1.138, 1.326), and resting behaviour, β = 0.147, SE = 0.037, p < 0.001, OR = 1.158 (95% CI: 1.078, 1.244) and decreased the odds of feeding, β = −0.164, SE = 0.038, p < 0.001, OR = 0.849 (95% CI: 0.788, 0.914), movement, β = −0.320, SE = 0.052, p < 0.001, OR = 0.726 (95% CI: 0.656, 0.804), and vigilance, β = 0.264, SE = 0.103, p = 0.103, OR = 0.768 (95% CI: 0.628, 0.939).
An increase in total park visitors reduced feeding, β = −0.256, SE = 0.037, p < 0.001, OR = 0.774 (95% CI: 0.720, 0.832) and increased resting β = 0.223, SE = 0.032, p < 0.001, OR = 1.250 (95% CI: 1.173, 1.333). An increase in enclosure visitors increased feeding, β = 0.124, SE = 0.034, p < 0.001, OR = 1.132 (95% CI: 1.060, 1.209), preening, β = 0.071, SE = 0.034, p = 0.036, OR = 1.074 (95% CI: 1.005, 1.148), vigilance, β = 0.341, SE = 0.090, p < 0.001, OR = 1.406 (95% CI: 1.179, 1.677), and other behaviours, β = 0.232, SE = 0.111, p = 0.036, OR = 1.262 (95% CI: 1.015, 1.568), and decreased resting, β = −0.245, SE = 0.033, p < 0.001, OR = 0.783 (95% CI: 0.733, 0.835).
Feeding increased when temperature, β = 0.229, SE = 0.040, p < 0.001, OR = 1.257 (95% CI: 1.163, 1.358), and daily rainfall increased, β = 0.186, SE = 0.034, p < 0.001, OR = 1.204 (95% CI: 1.127, 1.288). Feeding decreased when humidity increased, β = −0.226, SE = 0.047, p < 0.001, OR = 0.798 (95% CI: 0.727, 0.876). Preening increased when daily rainfall increased, β = 0.137, SE = 0.034, p < 0.001, OR = 1.147 (95% CI: 1.073, 1.226). Resting decreased when temperature, β = −0.267, SE = 0.037, p < 0.001, OR = 0.766 (95% CI: 0.712, 0.824), and rainfall increased, β = −0.206, SE = 0.034, p < 0.001, OR = 0.814 (95% CI: 0.761, 0.870). Resting increased when humidity increased, β = 0.188, SE = 0.045, p < 0.001, OR = 1.207 (95% CI: 1.106, 1.318). Movement decreased as sunshine increased, β = −0.115, SE = 0.051, p = 0.024, OR = 0.892 (95% CI: 0.807, 0.985). Vigilance increased as temperature, β = 0.419, SE = 0.106, p < 0.001, OR = 1.520 (95% CI: 1.234, 1.873), and humidity increased, β = 0.583, SE = 0.137, p < 0.001, OR = 1.792 (95% CI: 1.371, 2.342). Vigilance decreased when rainfall increased, β = −0.579, SE = 0.140, p < 0.001, OR = 0.560 (95% CI: 0.426, 0.737).
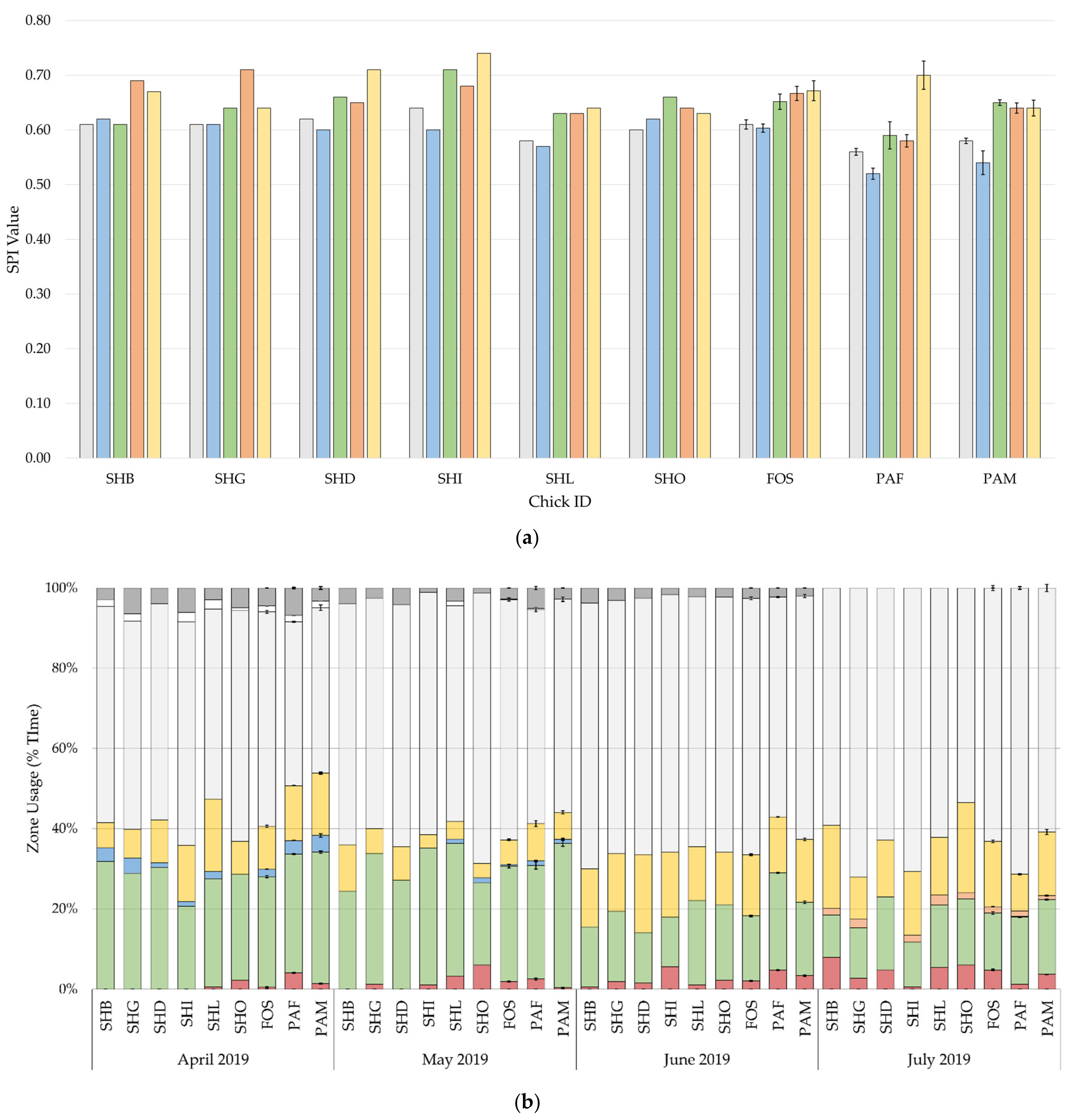
3.2. Enclosure Usage
Differences in the development of SPIs and zone usage by month are shown in
Figure 4. SPI values were high and showed an increasing trend over time, indicating discriminative use of enclosure zones. Foster-reared and parent-reared chicks spent most of their time in the nesting area, 60% and 55%, respectively. The effect rearing background, sex, climate, and visitors with the random slope, explained 9.2% to 20.7% of the variance in usage of the nest, shelter, and mid-water areas, with rearing background accounting for 1.9% to 10.5% of the variance (
Appendix C).
Zone usage model comparisons are displayed in
Table 3. FC models performed significantly better than null models,
X2s (10, N = 8059) ≥ 106.65,
p’s ≤ 0.001. RO models performed significantly better than null models when explaining usage of the nesting area,
X2 (1, N = 8059) ≥ 9.50,
p’s = 0.002. FC models did not perform significantly better than FC-R models when explaining occupancy in the nesting area,
X2 (2, N = 8059) ≤ 3.63,
p’s ≥ 0.0163. TO models performed significantly better than the null model when explaining usage of shelter and mid-water areas,
X2 (1, N = 8059) ≥ 8.03,
p’s ≤ 0.005.
The influence of predictors on zone usage, indicated through binomial GLMMs, are displayed in
Appendix D. Foster-reared chicks were 1.281 (95% CI: 1.099, 1.494) times more likely to occupy the nesting area than parent-reared chicks, β = 0.248, SE = 0.078,
p = 0.002. Female chicks were 1.729 (95% CI: 1.137, 2.629) times more likely to occupy the shelter zone than male chicks, β = 0.547, SE = 0.214,
p = 0.011, and 0.822 (95% CI: 0.684, 0.989) times less likely to occupy the middle water zone than males, β = −0.195, SE = 0.094,
p = 0.037. The effect of sex on occupancy in the shelter and mid-water zones were not dependent on rearing background, β = −0.551, SE = 0.313,
p = 0.078; β = 0.117, SE = 0.138,
p = 0.394.
A unit increase in time increased occupancy in the nesting zone, β = 0.392, SE = 0.036, p < 0.001, OR = 1.480 (95% CI: 1.380, 1.587), and decreased occupancy in the shelter, β = −1.064, SE = 0.109, p < 0.001, OR = 0.345 (95% CI: 0.279, 0.428), and mid-water zones, β = −0.098, SE = 0.050, p = 0.049, OR = 0.907 (95% CI: 0.823, 1.000).
An increase in enclosure visitors reduced occupancy of the nesting zone, β = −0.063, SE = 0.032, p = 0.046, OR = 0.939 (95% CI: 0.882, 0.999), and the mid-water zone, β = −0.246, SE = 0.048, p < 0.001, OR = 0.782 (95% CI: 0.712, 0.860). An increase in total park visitors decreased occupancy in the nest zone, β = −0.397, SE = 0.035, p < 0.001, OR = 0.672 (95% CI: 0.628, 0.720).
Occupancy in the nesting zone increased with humidity, β = 0.439, SE = 0.044, p < 0.001, OR = 1.551 (95% CI: 1.422, 1.693), and sunshine, β = 0.677, SE = 0.037, p < 0.001, OR = 1.968 (95% CI: 1.830, 2.117), and decreased as temperature, β = −0.494, SE = 0.038, p < 0.001, OR = 0.610 (95% CI: 0.567, 0.657), and rainfall increased, β = −0.203, SE = 0.032, p < 0.001, OR = 0.816 (95% CI: 0.766, 0.869). Occupancy in the shelter zone increased with temperature, β = 0.673, SE = 0.103, p < 0.001, OR = 1.960 (95% CI: 1.603, 2.398), and decreased as humidity, β = −0.293, SE = 0.145, p = 0.043, OR = 0.746 (95% CI: 0.561, 0.991), and sunshine increased, β = −0.472, SE = 0.111, p < 0.001, OR = 0.624 (95% CI: 0.502, 0.775). Occupancy of the mid-water zone increased with temperature, β = 0.518, SE = 0.053, p < 0.001, OR = 1.679 (95% CI: 1.513, 1.862), and daily rainfall, β = 0.378, SE = 0.038, p < 0.001, OR = 1.460 (95% CI: 1.354, 1.574), and decreased as sunshine increased, β = −0.263, SE = 0.054, p < 0.001, OR = 0.769 (95% CI: 0.692, 0.855).
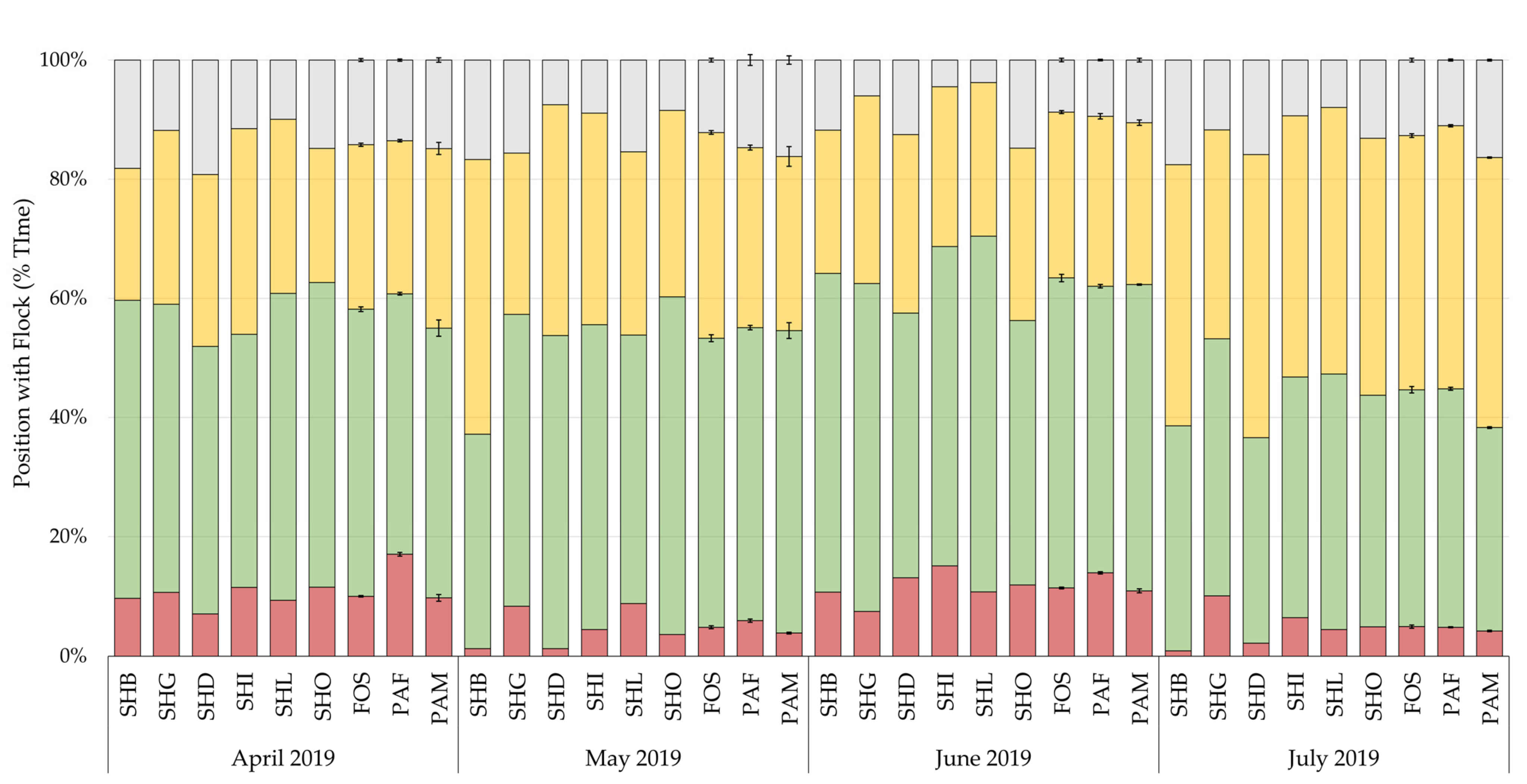
3.3. Flock Position
Differences in the position of chicks within the flock by month are shown in
Figure 5. Chilean flamingo spent on average 92% of their time associating with other flamingos, spending most of their time in peripheral, 45%, and outer, 33%, positions to the flock. The effect of rearing background, sex, climate, and visitors with the random slope, explained 2.8% to 8.6% of the variance in flock position, with rearing background accounting for 0.6% to 2.1% of the variance (
Appendix E).
Flock position model comparisons are displayed in
Table 4. FC models for flock positions performed better than null models,
X2 s (10, N = 8059) ≥ 106.65,
p’s ≤ 0.001. RO models did not perform better than null models,
X2 s (1, N = 8059) ≤ 3.46,
p’s ≥ 0.063. FC models did not perform better than FC-R models,
X2 s (2, N = 8059) ≤ 4.60,
p’s ≥ 0.0100. TO models performed better than null models for each flock position,
X2 s (1, N = 8059) ≥ 4.69,
p’s ≤ 0.030.
The influence of predictors on chicks’ position within the flock, indicated through binomial GLMMs, are displayed in
Appendix F. Rearing background did not influence flock position. Female chicks were 1.505 (95% CI: 1.121, 2.021) times more likely than male chicks to be alone, β = 0.409, SE = 0.150,
p = 0.007.
A unit increase in time decreased occupancy in alone positions, β = −0.204, SE = 0.060, p < 0.001, OR = 0.816 (95% CI: 0.725, 0.917), and peripheral positions, β = −0.266, SE = 0.033, p < 0.001, OR = 1.766 (95% CI: 0.718, 0.818), and increased occupancy in outer positions, β = 0.410, SE = 0.036, p < 0.001, OR = 1.507 (95% CI: 1.404, 1.617).
An increase in total park visitors increased occupancy in alone positions, β = 0.118, SE = 0.054, p = 0.029, OR = 1.125 (95% CI: 1.012, 1.252). An increase in enclosure visitors increased occupancy in alone positions, β = 0.278, SE = 0.048, p < 0.001, OR = 1.320 (95% CI: 1.202, 1.450), and decreased occupancy in peripheral positions, β = −0.156, SE = 0.030, p < 0.001, OR = 0.856 (95% CI: 0.807, 0.907).
An increase in temperature increased occupancy in alone, β = 0.290, SE = 0.061, p < 0.001, OR = 1.337 (95% CI: 1.186, 1.506), and peripheral positions, β = 0.211, SE = 0.035, p < 0.001, OR = 1.235 (95% CI: 1.153, 1.322), and reduced occupancy in outer, β = −0.281, SE = 0.037, p < 0.001, OR = 0.755 (95% CI: 0.702, 0.813), and central flock positions, β = −0.125, SE = 0.052, p = 0.016, OR = 0.882 (95% CI: 0.797, 0.977). An increase in humidity increased occupancy in alone positions, β = 0.564, SE = 0.076, p < 0.001, OR = 1.757 (95% CI: 1.514, 2.040), and reduced occupancy in peripheral positions, β = −0.289, SE = 0.042, p < 0.001, OR = 0.749 (95% CI: 0.690, 0.813). An increase in sunshine increased occupancy in outer, β = 0.201, SE = 0.036, p < 0.001, OR = 1.223 (95% CI: 1.140, 1.311), and central positions to the flock, β = 0.263, SE = 0.052, p < 0.001, OR = 1.300 (95% CI: 1.175, 1.438), and reduced occupancy in alone, β = −0.235, SE = 0.060, p < 0.001, OR = 0.791 (95% CI: 0.703, 0.890), and peripheral positions, β = −0.201, SE = 0.034, p < 0.001, OR = 0.800 (95% CI: 0.749, 0.855). An increase in rainfall reduced occupancy in alone positions, β = −0.352, SE = 0.060, p < 0.001, OR = 0.703 (95% CI: 0.626, 0.790) and increased occupancy in peripheral positions, β = 0.124, SE = 0.031, p < 0.001, OR = 1.132 (95% CI: 1.066, 1.202).
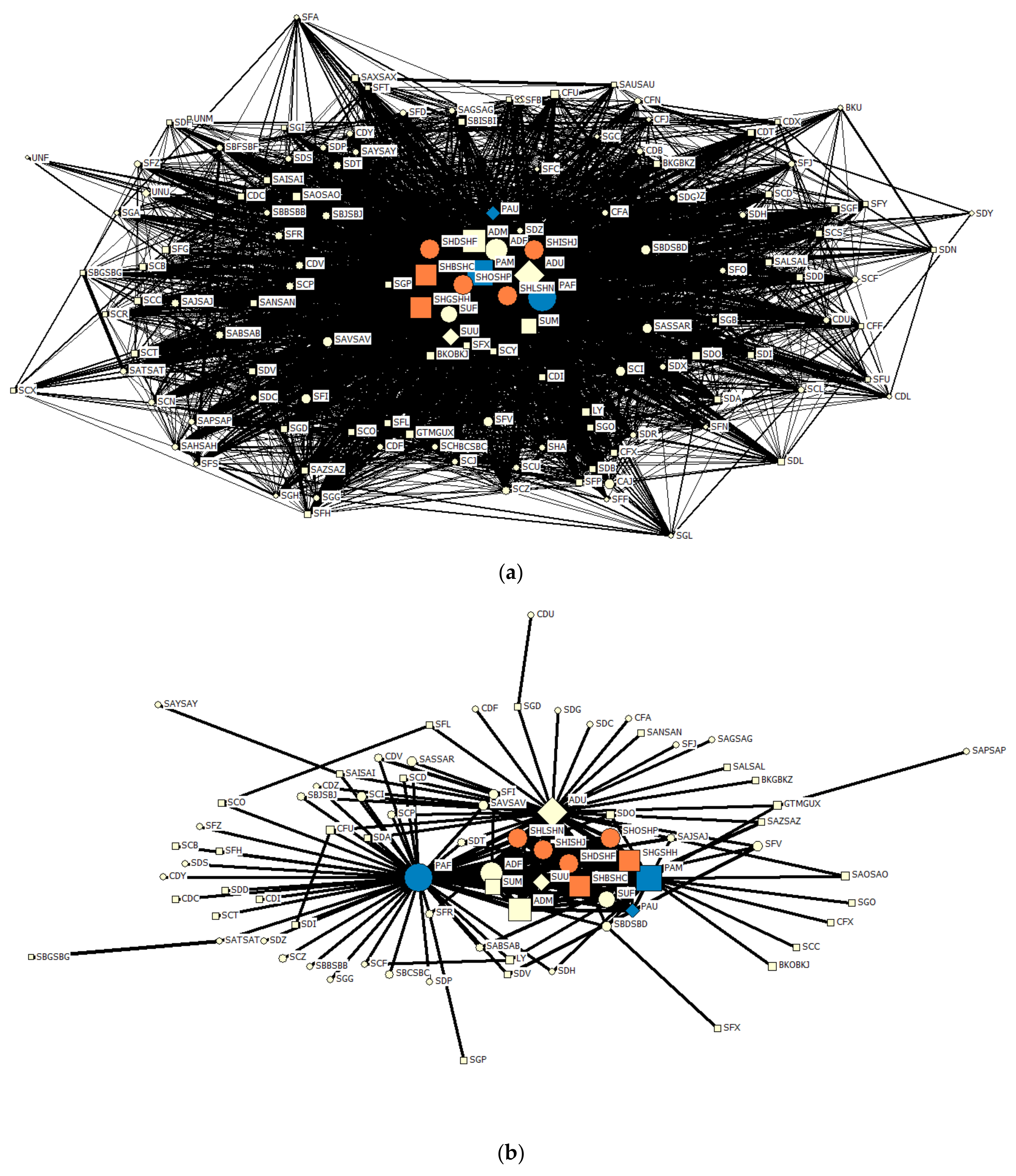
3.4. Social Preferences
Networks of associations of parent-reared and foster-reared chicks are displayed in
Figure 6. The overall mean association rate of both foster-reared (Mean Assoc. = 0.08, SD = 0.08) and parent-reared chicks (Mean Assoc. = 0.10, SD = 0.07) indicated that their associations within this network were weak; but large group sizes may distort this (Rose & Croft, 2018). Estimation of social differentiation using a likelihood method revealed that this network was well differentiated (CV = 1.139, SE = 0.072) as defined by Whitehead (2009) and the power to detect the true social network was moderate (P = 0.494, SE = 0.006). Modularity analysis indicated that the network could not be divided into separate communities (Modularity = 0.079 for 13 clusters). Furthermore, a cophenetic correlation coefficient of 0.778 did not justify the use of cluster analysis.
Permutation tests revealed that foster-reared chicks held less significant dyads than expected (obs = 78, exp = 133.2) and parent-reared chicks held more significant dyads than expected (obs = 155, exp = 125.25). See
Appendix G for significant avoided and preferred associations. The observed CV was significantly greater than the expected CV for both foster-reared and parent-reared chicks, indicating that Chilean flamingo chicks were preferentially associating or disassociating with other flamingos, CV
observed = 1.908, CV
expected = 1.886,
p = 0.001; CV
observed = 1.644, CV
expected = 1.526,
p < 0.001.
A Mantel test revealed that patterns of associations were significantly different between rearing classes, t = −4.632, p < 0.001. MRQAP tests also revealed that rearing background predicted the association matrix whilst controlling for sex, age, and network measures, r = −0.215, p < 0.001. Sex and age were not significant predictors of the association matrix, r = −0.0347, p = 0.066; r = −0.036, p = 0.262. Amongst the network measures, only eigenvector centrality was a significant predictor of the association matrix, r = −0.048, p = 0.009.
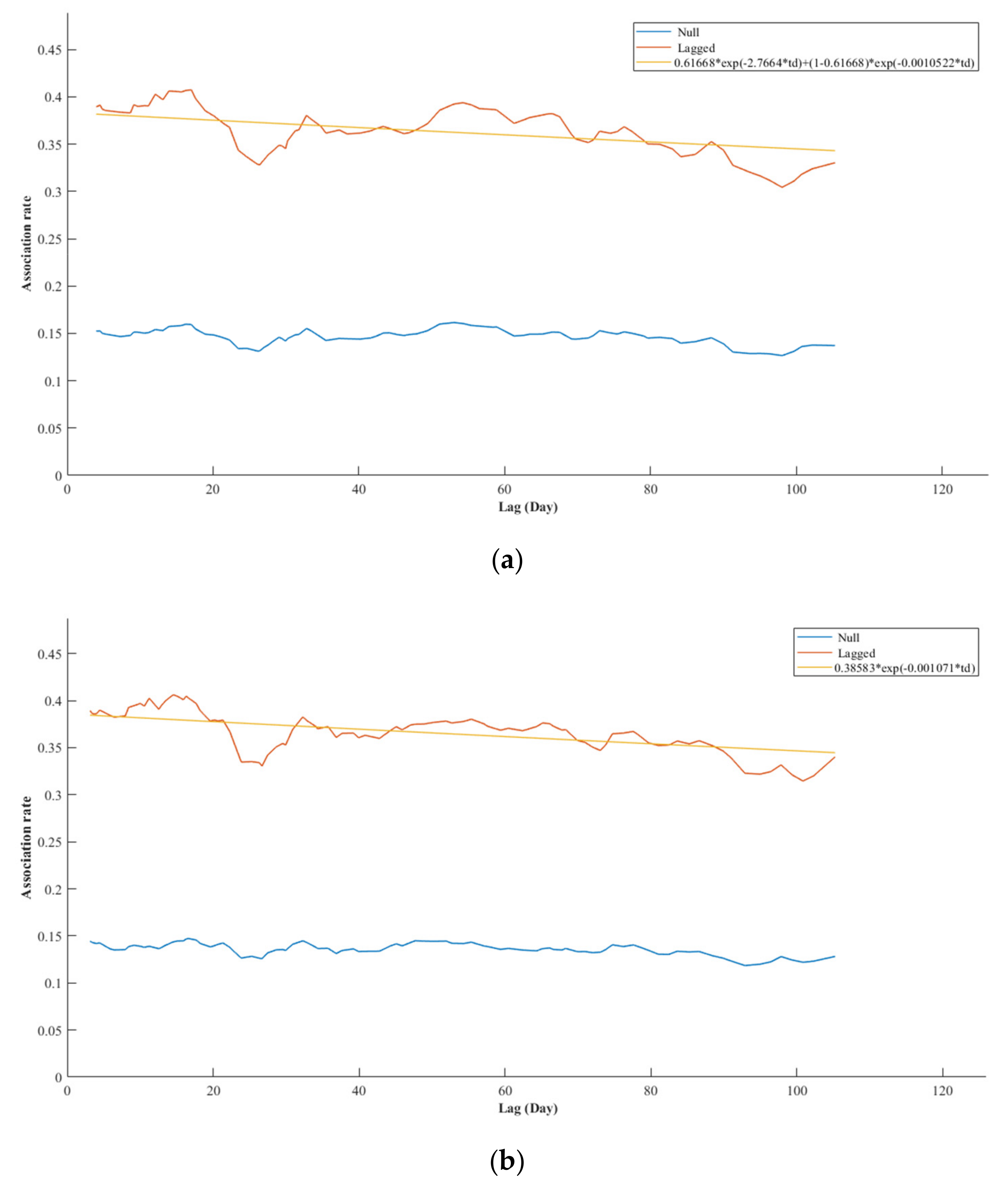
Differences between the plotted lagged and null association rates indicated that foster-reared and parent-reared chicks were both showing preferred associations over time (
Figure 7) and that the strength of a typical dyadic association is similar between the two groups of chicks. The best fitting model (i.e., model with lowest QAIC value) explaining the lagged association rate of foster-reared chicks to all other flamingos indicated two levels of casual acquaintances. The best fitting model explaining the lagged association rate of parent-reared chicks to all other flamingos indicated rapid dispersal and casual acquaintances. Other models also showed support (see
Appendix H).
Results from Mantel tests are displayed in
Table 5. The overall associations matrix from the first month of sampling consistently and accurately correlated with all other monthly association matrices, suggesting that the association patterns of chicks were consistent over the sampling period. Behavioural association matrices also accurately correlated with one another. However, the partial correlation coefficient reduced when looking at the associations when chicks were alert or performing other behaviours, indicating that associations varied across some behaviours. Similarly, association matrices observed in different enclosure zones accurately correlated but the strength of correlation was not consistent, indicating that associations varied across zones. Associations observed in public viewing areas did not correlate with associations elsewhere in the enclosure. Associations across central, outer, and peripheral locations relative to the flock strongly and accurately correlated; indicating that chicks moved around and within the flock with a consistent set of associates. As expected, the association matrix of when chicks were alone did not correlate with the association’s chicks held when they were closer to the flock. Patterns of association across visitor and climatic variables strongly and accurately correlated with little variation.
4. Discussion
Our results show no major differences between the juvenile Chilean flamingos reared as chicks by Andean flamingo foster-surrogates when compared to juvenile Chilean flamingos reared by their own parents. Such zoo husbandry interventions can be successful and enhance the welfare of individuals involved (e.g., in this case enabling a full breeding cycle to experience by Andean flamingos) if the ecology and behaviour of all species involved is carefully considered, and the intervention and output carefully monitored. Comparison of full conditional (FC) against null models indicated that parent-reared and fostered chick behaviour, enclosure usage, and flock positioning were non-random. Rearing background explained a small proportion of variance in behaviour. Comparisons of rearing background models (RO) against null models suggested that the predictive use of rearing background was limited to feeding, movement, and occupancy in the nesting zone. Comparison of FC models to FC models without rearing background (FC-R) revealed that the proportion of variance in movement and nest occupancy explained by rearing background did not significantly improve model fit. Despite this, the fixed effect of rearing background on nesting occupancy within the FC model was significant. Although the variance in nest occupancy explained by rearing background was small, this variance was unique and not encapsulated by other predictors in the model. Conversely, rearing background did not influence movement; the initial variance explained by rearing background could be better explained by other predictors in the model. For feeding behaviour, rearing background significantly improved model fit and significantly influenced time spent feeding.
The Chilean flamingo chicks formed a highly variable and well differentiated network of associations, indicative of non-random, discriminative patterns of association (
Figure 6a). The filtered network shows that all chicks held several preferred associations (
Figure 6b). Although foster-reared chicks showed fewer significant preferred associations, differences the null and lagged association rates (
Figure 7) show that these bonds were equally strong and stable as the bonds that parent-reared chicks formed. Both groups of chicks formed long-lasting preferential bonds and demonstrated several short-lived relationships.
Chicks showed an impressive ability to maintain their pattern of associations across the sampling period, behavioural states, enclosure zones, positions within the flock, varying climates, and varying numbers of visitors. Chosen associates significantly differed between foster-reared and parent-reared chicks. Rearing background predicted the association patterns, but the sex or age of a bird had no influence. Foster-reared chicks associated with other foster-reared chicks, but also formed relationships with other flamingos. Eigenvector centrality was also a predictor of the association matrix, as birds with more associates are more likely to be connected to the well connected.
Patterns of behaviour (
Figure 3), enclosure usage (
Figure 4), and positioning within the flock (
Figure 5) were also non-random. Chicks spent most of their time feeding, resting, and preening, occupying the nesting area, and occupying peripheral and outer locations to the flock. Foster-reared and parent-reared chicks’ behavioural states, use of the enclosure, and positions within their flock over the sampling period showed complex relationships with temporal, identity, and environmental factors. However, early rearing experience did not explain a significant amount of variation in most behaviours. The effect of rearing background on Chilean flamingo chick behaviour was limited to feeding and use of the nesting zone. Independent of their sex, foster-reared chicks had greater odds of occupying the nesting zone than parent-reared chicks and were also less likely to be observed feeding.
Taken together, our results show that altering the early social rearing environment of captive Chilean flamingo chicks, through foster-rearing intervention with Andean flamingos, is associated with few behavioural and social differences relative to parent-reared conspecifics within the same flock. Our findings conflict with previous studies where cross-fostered birds display numerous behavioural similarities to their foster species [
30,
31,
34] and develop altered social preferences [
23,
39]. Instead, both foster-reared and parent-reared Chilean flamingo chicks were able to express patterns of activity and association similar to their captive and wild counterparts [
47,
50,
51,
71].
Cross-fostering studies are typically used to assess the relative influence of genetics and the environment [
72]. Experimental procedures utilise two species that are closely related enough that fostering is successful, but also dissimilar in observable aspects of behaviour so that misimprinting is easily recognisable (e.g., vocalisations [
34]). The resulting behavioural differences between cross-fostered and parent-reared conspecifics are therefore the result of pre-existing behavioural differences between the two species as a whole. It follows, then, that the numerous similarities in various aspects of the Chilean flamingo chick’s behaviour can be largely attributed to the ecological similarities between Andean and Chilean flamingos.
Previous social network analyses on the Chilean and Andean flocks involved in our study indicate that both show similar non-random patterns of assortment [
47]. Individuals formed strong preferential bonds which were maintained in and outside of the breeding season [
47]. The social dynamics of the foster flock were therefore a good representative of the biological flock. The early social rearing environment equipped foster-chicks with relevant experiences that facilitated their ability to form long-lasting bonds with other flamingos whilst avoiding others.
Both parent-reared and foster-reared chicks showed a preference for the nesting area (
Figure 4b). Over time, chicks were less likely to be found alone or on the periphery of the flock and were more likely to be associating with five or more older flamingos. Foster-reared chicks also occupied the nest area more than parent-reared chicks. Captive greater flamingos have also shown biased use of the nesting area when studied during the breeding season, as in this study [
73]. Flamingos naturally group together and are more likely to congregate in the nesting zone during breeding season [
49,
74]. By assembling together into the popular nesting area, chicks can settle into the flock and benefit from the welfare advantages of flocking (e.g., foraging efficiency, predator detection and avoidance, access to mates [
75,
76,
77].
Wild Andean and Chilean flamingos inhabit intersecting geographical regions in South America [
51,
52], utilising similar wetland resources. This ecology is represented in their enclosure design at this animal collection. The two enclosures at WWT Slimbridge (South American Pen and the Andean Flamingo Pen) incorporated biologically relevant features to both species providing, as best possible, opportunities for normal time-activity patterns to be performed. Pre-existing preferences developed during early rearing mean that when relocating, birds should prefer to settle in habitats similar to their natal foster-rearing site [
78,
79]. When later life environments are different to the natal rearing environment, cross-fostered young show negative altered behaviour patterns that reduce fitness [
39]. Dispersing birds are under strong selection pressure to quickly settle into a habitat, and so make discriminative choices based on their natal experience [
78,
80]. Once settled, individuals are then predicted to increase activity in the breeding area [
81]. For instance, after dispersal, cross-fostered pied flycatchers show preferences to their foster species habitat and choose to breed there later in life [
82].
Andean and Chilean flamingos spend most of their time preening, resting, and feeding [
48,
70]; analogous to chicks behavioural patterns (
Figure 3). Less time spent feeding by foster-reared chicks may initially seem concerning since reduced feeding is linked to deferred maturity and higher mortality in birds [
83]. Wild juvenile Chilean flamingo chicks feed less than adults due to aggressive displacement [
84]. However, the abundance of food appropriately dispersed across the Chilean flamingo enclosure and freedom of movement would minimise aggression between birds [
85]. This is seen in other wild flamingo flocks; for instance, the superabundant supplies of food at Kamfers Dam in South Africa reduced the effects of any lost feeding time by allowing lesser flamingos to spread out and avoid aggressive interactions [
75]. Foster-reared Chilean flamingo chicks, motivated by hunger, could freely move to non-competitive zones to feed. Instead, greater foraging efficiency requires individuals to spend less time feeding [
84,
86]. Generalist feeding birds cross-fostered to specialist feeding birds have previously shown foraging patterns that align with their foster species and that are more efficient than parent-reared controls [
35,
87].
It is not well-known if feeding behaviours of the generalist feeding Chilean flamingo are shared by the specialist feeding Andean flamingo 49]. Andean and Chilean flamingos show different food preferences which enable them to coexist together in the wild [
45,
46]. Differences in bill structure [
88] and an understanding that flamingos display distinct feeding behaviours that are relevant to their ecological niche (Rooth, 1976 as cited in 49]) suggest it is likely that feeding behaviour varies between Andean and Chilean flamingos. Understanding species-specific feeding behaviour of the understudied Andean flamingo would help clarify if distinct foraging niches exist and if these can be culturally transmitted into non-biological offspring.
Whilst we present these results to show the efficacy of a husbandry intervention, such information can benefit ex situ conservation efforts by encouraging increased reproductive output in populations that may need assistance for breeding. Conservation efforts may require animals being moved between groups to ensure genetic management of the population [
89]. For captive flamingos, this is almost a certainty, since zoos can no longer remove flamingos from the wild and their current captive populations are not self-sustaining 49]. Foster-rearing presents a way to address this reproductive dilemma. Introducing reproductively viable individuals into foreign populations increases genetic diversity and reduces inbreeding [
22]. Providing inexperienced individuals with paternal experience also increases future rearing success in flamingos and removing eggs from breeding pairs encourages further egg production 49]. The individuals that can benefit from foster-rearing are vast and understanding the effects are imperative to its success. The evidence-based approach is key here; assessing the ecology of the selected species for cross fostering and monitoring development (i.e., growth and behavioural) in young animals to ensure any impacts on adult characteristics and activity patterns are not detrimental or long-lasting.
Cases against the use of foster-rearing are valid, e.g., [
23,
40,
42]. Although our small sample size restricts generalisability, we suggest that foster-rearing husbandry practices need to be implemented on a case-by-case basis to suit individual welfare needs. Rather than generalising knowledge across species, future fostering interventions should be informed by comprehensive habitat, behavioural, and social assessments of the specific animals involved; analogous to reintroduction programmes [
26,
90,
91,
92]. Previous experiences place specific demands on individuals that must be factored into husbandry provision in captivity [
89]. Further research assessing the reproductive behaviours of the foster-reared chicks, once they reach sexual maturity, would help reveal the propagative viability of these captive foster-reared flamingos. It would also be beneficial to study patterns of sociality over longer sampling periods since captive Chilean flamingos show looser association patterns outside of the breeding season than Andean flamingos [
47].