Dolphin Welfare Assessment under Professional Care: ‘Willingness to Participate’, an Indicator Significantly Associated with Six Potential ‘Alerting Factors’
Abstract
1. Introduction
2. Methods
2.1. Involved Dolphinaria
2.2. Information Used on the QBA
2.3. Statistical Analysis and Sample Sizes
3. Results
3.1. Effects of Different Training Conditions and Individual Features on Behavioural Parameters
3.1.1. Dolphins’ Speed of Approach in Presence of the Trainer
3.1.2. Dolphins’ Level of Excitement
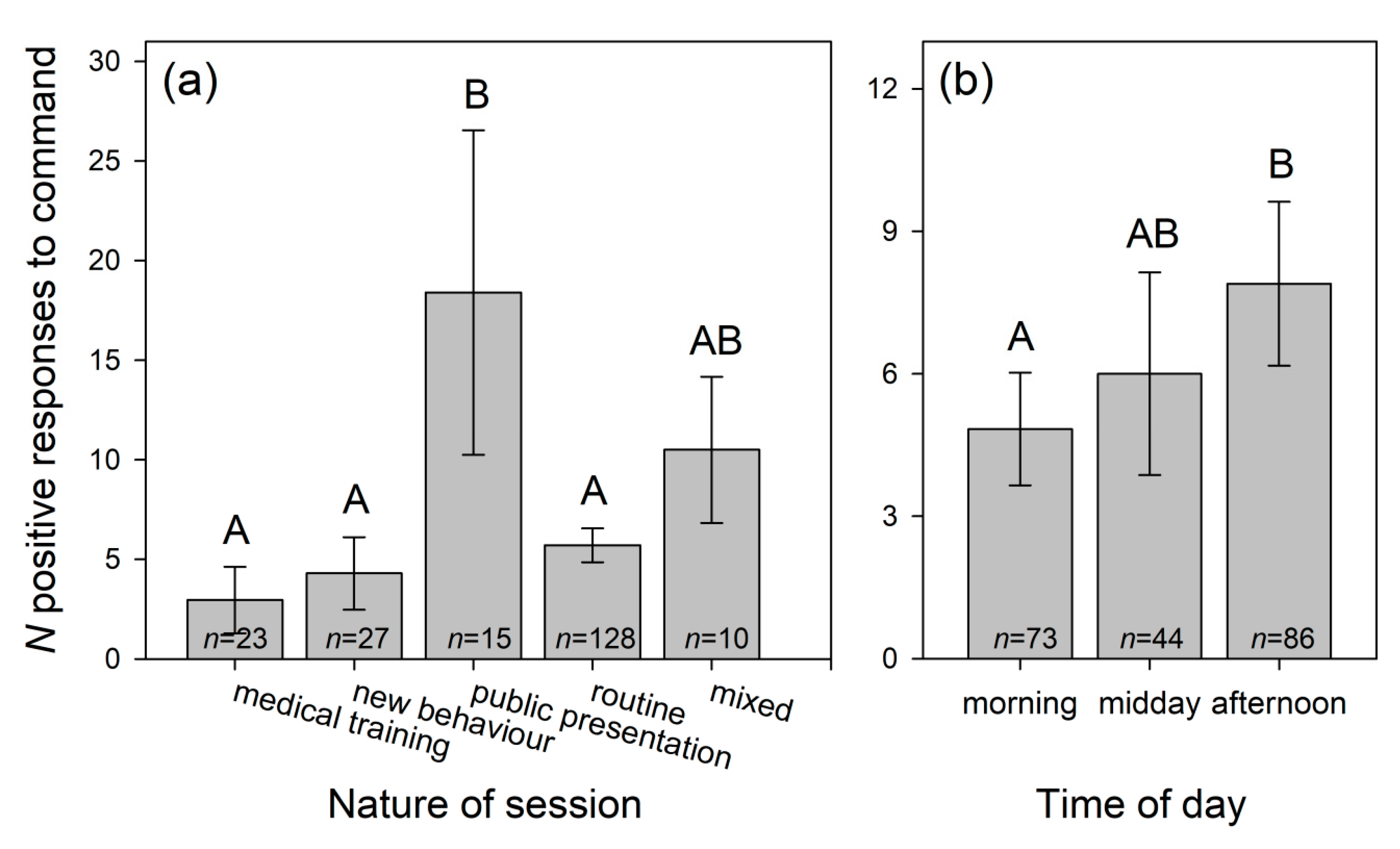
3.1.3. Dolphins’ Positive Responses to Trainers’ Signals
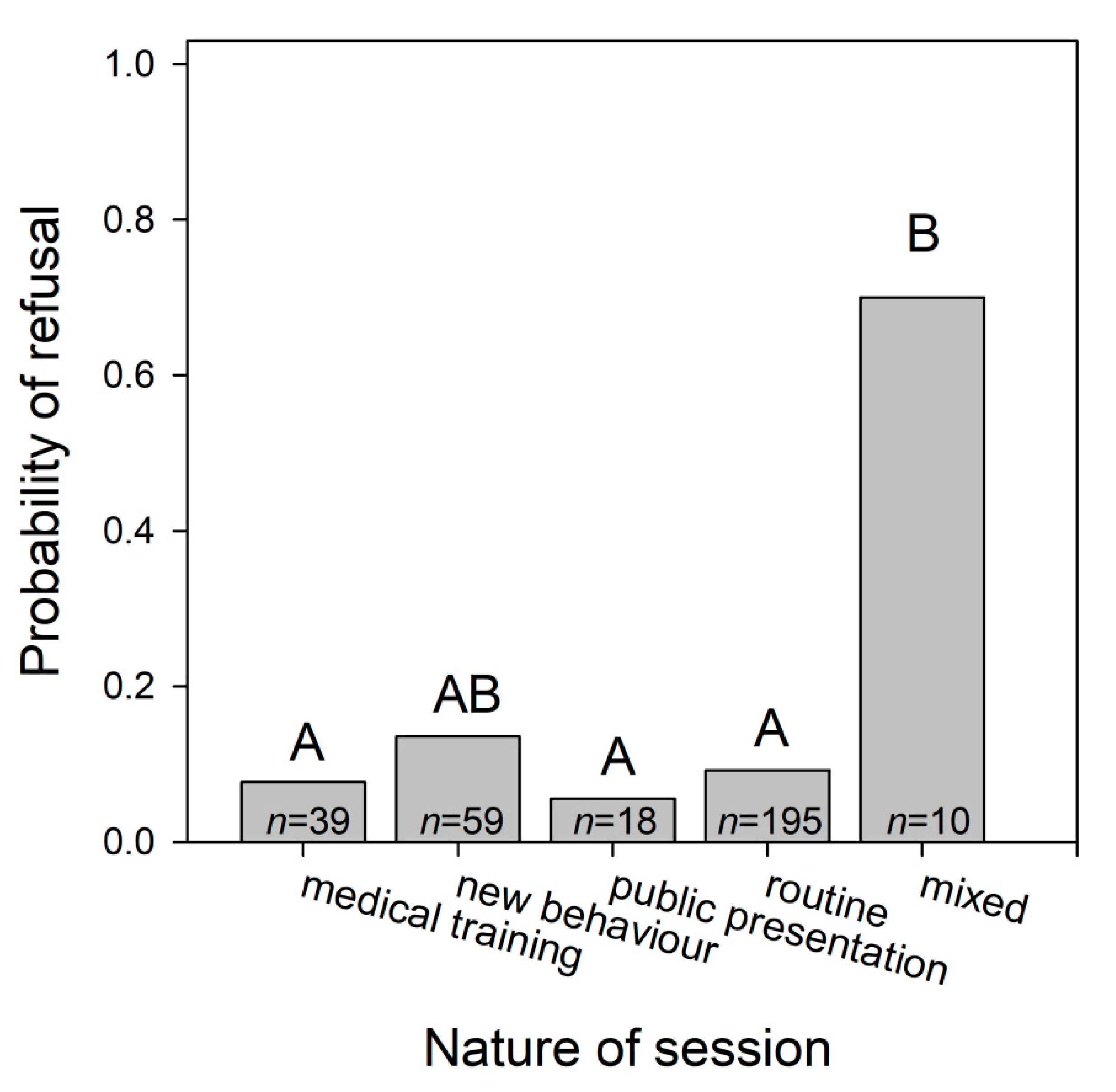
3.1.4. Dolphins’ Refusal to Perform Particular Behaviours
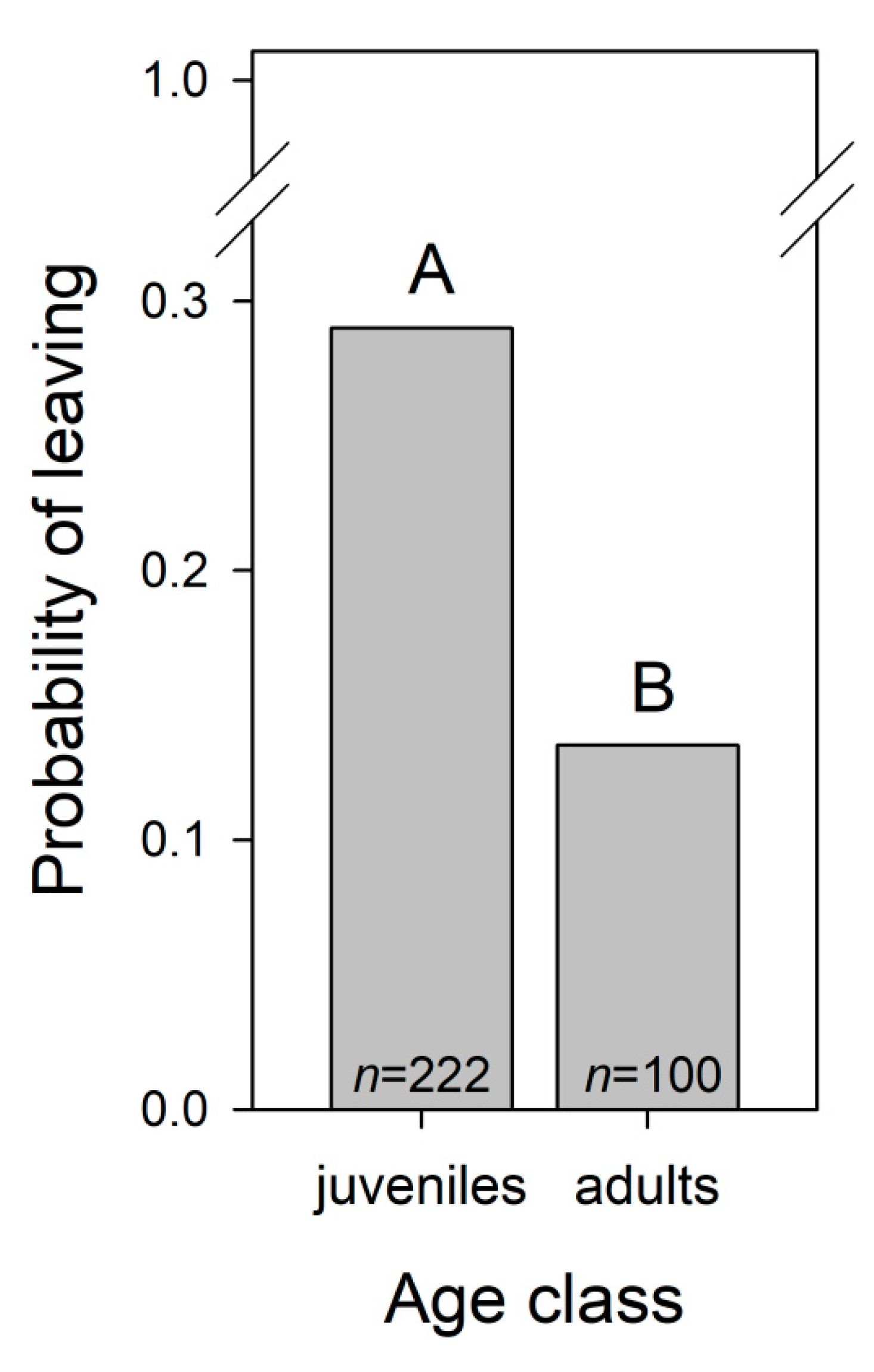
3.1.5. Dolphins’ Leave Trainer during Trainer-Dolphin Sessions
3.1.6. Dolphins’ Eye Contact with Trainers
3.1.7. Dolphins’ Approach Speed Once the Trainer Entered the Pool
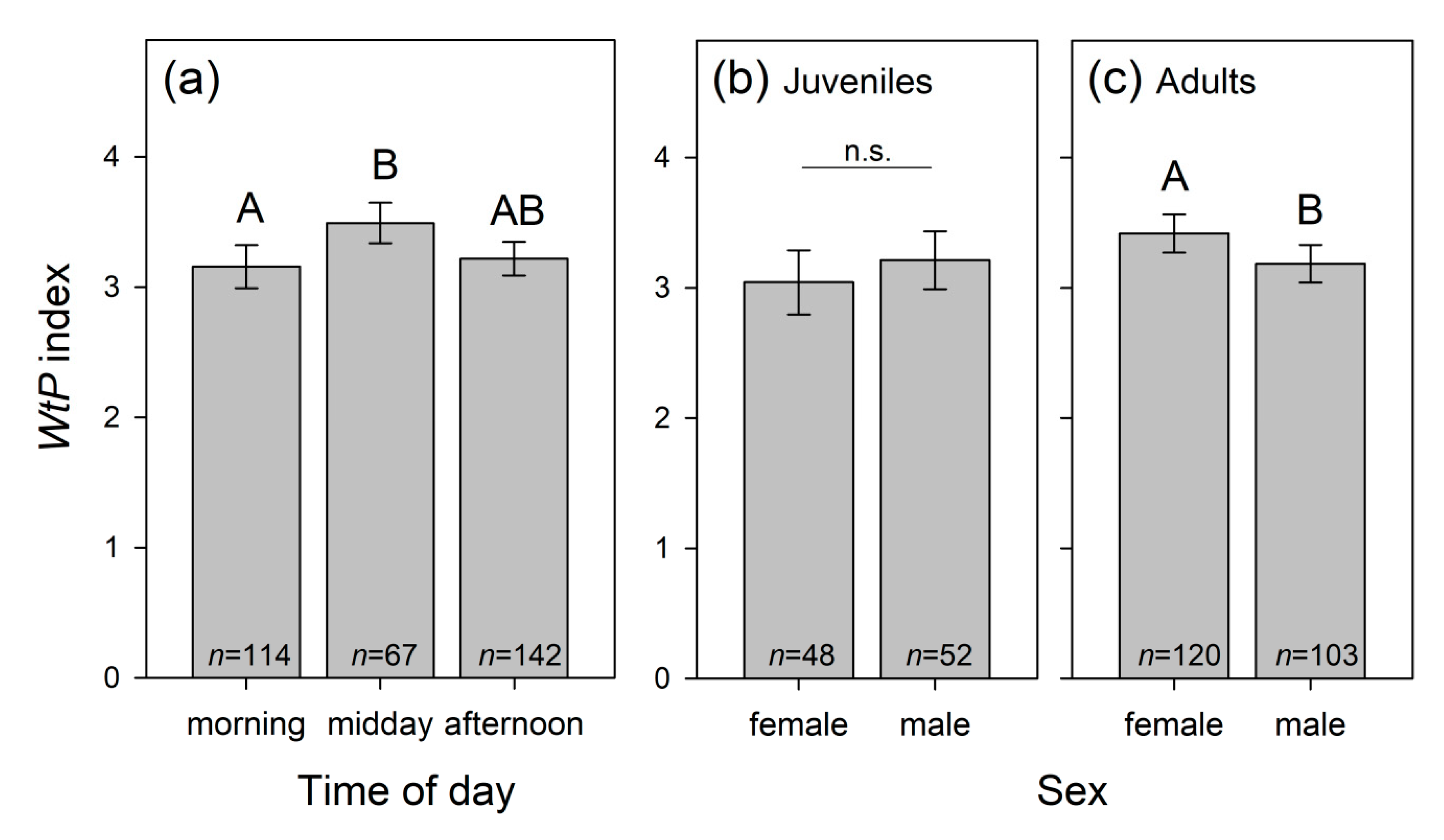
3.1.8. Dolphins’ Willingness to Participate (WtP) during Training
3.2. Associations between WtP and Other Behavioural Parameters
4. Discussion
4.1. Factors Explaining the Variation of the Different Behavioural Parameters Reflecting Dolphin Welfare
4.2. “WtP” and Potential Alerting Factors to assess Dolphin Welfare
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hosey, G.R. A preliminary model of human–animal relationships in the zoo. Appl. Anim. Behav. Sci. 2008, 109, 105–127. [Google Scholar] [CrossRef]
- Carlstead, K. A comparative approach to the study of keeper–animal relationships in the zoo. Zoo Biol. 2009, 28, 589–608. [Google Scholar] [CrossRef]
- Highfill, L.E.; Kuczaj, S.A., II. Do bottlenose dolphins (Tursiops truncatus) have distinct and stable personalities? Aquat. Mamm. 2007, 33, 380–389. [Google Scholar] [CrossRef]
- Hosey, H. Hediger Revisited: How do zoo animals see us? J. Appl. Anim. Welf. Sci. 2013, 16, 338–359. [Google Scholar] [CrossRef] [PubMed]
- Hemsworth, P.H. Human–animal interactions in livestock production. Appl. Anim. Behav. Sci. 2003, 81, 185–198. [Google Scholar] [CrossRef]
- Waiblinger, S.; Boivin, X.; Pederson, V.; Tosi, M.; Janczak, A.M.; Visser, E.K.; Jones, R.B. Assessing the human–animal relationship in farmed species: A critical review. Appl. Anim. Behav. Sci. 2006, 101, 185–242. [Google Scholar] [CrossRef]
- Hosey, G.R.; Melfi, V. Human–animal bonds between zoo professionals and the animals in their care. Zoo Biol. 2012, 31, 13–26. [Google Scholar] [CrossRef]
- Boivin, X.; Lensink, J.; Tallet, C.; Veissier, I. Stockmanship and farm animal welfare. Anim. Welf. 2003, 12, 479–492. [Google Scholar]
- Brando, S. Advances in husbandry training in marine mammal care programs. Int. J. Comp. Psychol. 2010, 23, 777–791. [Google Scholar]
- Smith, J.J. Human-animal relationships in zoo-housed orangutans (P. abelii) and gorillas (G. g. gorilla): The effects of familiarity. Am. J. Primatol. 2014, 76, 942–955. [Google Scholar] [CrossRef]
- Cole, J.; Fraser, D. Zoo animal welfare: The human dimension. J. Appl. Anim. Welf. Sci. 2008, 21, 49–58. [Google Scholar] [CrossRef] [PubMed]
- Carlstead, K.; Brown, J.L. Relationships between patterns of fecal corticoid excretion and behavior, reproduction and environmental factors in captive black (Diceros bicornis) and white (Ceratotherium simum) rhinoceros. Zoo Biol. 2005, 24, 215–232. [Google Scholar] [CrossRef]
- Wielebnowski, N.C.; Fletchall, N.; Carlstead, K.; Busso, J.M.; Brown, J.L. Non-invasive assessment of adrenal activity associated with husbandry and behavioural factors in the North American clouded leopard population. Zoo Biol. 2002, 21, 77–98. [Google Scholar] [CrossRef]
- Claxton, A.M. The potential of the human–animal relationship as an environmental enrichment for the welfare of zoo-housed animals. Appl. Anim. Behav. Sci. 2011, 133, 1–10. [Google Scholar] [CrossRef]
- Hosey, G.R. Zoo animals and their human audiences: What is the visitor effect? Anim. Welf. 2000, 9, 343–357. [Google Scholar]
- Melfi, V.A.; Thomas, S. Can training zoo- housed primates compromise their conservation? A case study using Abyssinian colobus monkeys (Colobus guereza). Anthrozoos. 2005, 18, 304–317. [Google Scholar] [CrossRef]
- Webster, J. Animal Welfare: Limping Towards Eden: A Practical Approach to Redressing the Problem of Our Dominion Over the Animals; Blackwell Publishing Ltd.: Oxford, UK, 2005. [Google Scholar]
- Farm Animal Welfare Council (FAWC). FAWC updates the five freedoms. Vet. Rec. 1992, 17, 357. [Google Scholar]
- Welfare Quality® Assessment Protocol for Cattle (Fattening Cattle, Dairy Cows, Veal Calves); Welfare Quality® Consortium: Lelystad, The Netherlands, 2009.
- Welfare Quality® Assessment Protocol for Pigs. Welfare; Quality®Consortium: Lelystad, The Netherlands, 2009.
- Welfare Quality® Assessment Protocol for Poultry; Welfare Quality® Consortium: Lelystad, The Netherlands, 2009.
- Breuer, K.; Hemsworth, P.H.; Coleman, G.J. The effect of positive or negative handling on the behavioural and physiological responses of nonlactating heifers. Appl. Anim. Behav. Sci. 2003, 84, 3–22. [Google Scholar] [CrossRef]
- Campbell, M. Avian reactions towards human approaches in different urban greenery structures in Nanaimo. Urban For. Urban Green. 2016, 19, 47–55. [Google Scholar] [CrossRef]
- Smith, A.V.; Wilson, C.; McComb, K.; Proops, L. Domestic horses (Equus caballus) prefer to approach humans displaying a submissive body posture rather than a dominant body posture. Anim. Cogn. 2018, 21, 307–312. [Google Scholar] [CrossRef]
- Wemelsfelder, F.; Lawrence, A.B. Qualitative assessment of animal behaviour as an on-farm welfare monitoring tool. Acta Agr Scand a-An. 2001, 51, 21–25. [Google Scholar]
- Brscic, M.; Wemelsfelder, F.; Tessitore, E.; Gottardo, F.; Cozzi, G.; Van Reenen, C.G. Welfare assessment: Correlations and integration between a Qualitative Behavioural Assessment and a clinical/health protocol applied in veal calves farms. Ital. J. Anim. Sci. 2010, 8, 601–603. [Google Scholar] [CrossRef]
- Wemelsfelder, F.; Hunter, E.A.; Mendl, M.T.; Lawrence, A.B. Assessing the ‘whole animal’: A Free-Choice-Profiling approach. Anim. Behav. 2001, 62, 209–220. [Google Scholar] [CrossRef]
- Wemelsfelder, F. How animals communicate quality of life: The qualitative assessment of animal behaviour. Anim. Welf. 2007, 16, 25–31. [Google Scholar]
- Patel, F.; Whitehouse-Tedd, K.; Ward, S.J. Redefining human-animal relationships: An evaluation of methods to allow their empirical measurement in zoos. Anim. Welf. 2019, 28, 247–259. [Google Scholar] [CrossRef]
- Rutherford, K.M.D.; Donald, R.D.; Lawrence, A.B.; Wemelsfelder, F. Qualitative Behavioural Assessment of emotionality in pigs. Appl. Anim. Behav. Sci. 2012, 139, 218–224. [Google Scholar] [CrossRef] [PubMed]
- Wemelsfelder, F.; Mullan, S. Applying ethological and health indicators to practical animal welfare assessment. Anim. Welf.: Focusing on the Future. 2014, 33, 111–120. [Google Scholar] [CrossRef]
- Fleming, P.A.; Clarke, T.; Wickham, S.L.; Stockman, C.A.; Barnes, A.L.; Collins, T.; Miller, D.W. The contribution of qualitative behavioural assessment to appraisal of livestock welfare. Anim. Prod. Sci. 2016, 56, 1569–1578. [Google Scholar] [CrossRef]
- Ellingsen, K.; Coleman, G.J.; Lund, V.; Mejdell, C.M. Using qualitative behaviour assessment to explore the link between stockperson behaviour and dairy calf behaviour. Appl. Anim. Behav. Sci. 2014, 153, 10–17. [Google Scholar] [CrossRef]
- Patel, F.; Wemelsfelder, F.; Ward, S. Using qualitative behaviour assessment to investigate human-animal relationships in zoo-housed giraffes (Giraffa camelopardalis). Animals 2019, 9, 381. [Google Scholar] [CrossRef] [PubMed]
- Minero, M.; Dalla Costa, E.; Dai, F.; Canali, E.; Barbieri, S.; Zanella, A.; Pascuzzo, R.; Wemelsfelder, F. Using qualitative behaviour assessment (QBA) to explore the emotional state of horses and its association with human-animal relationship. Appl. Anim. Behav. Sci. 2018, 204, 53–59. [Google Scholar] [CrossRef]
- Carrasco, L.; Colell, M.; Calvo, M.; Abello, M.T.; Velasco, M.; Posada, S. Benefits of training/playing therapy in a group of captive lowland gorillas (Gorilla gorilla gorilla). Anim. Welf. 2009, 18, 9–19. [Google Scholar]
- Chelluri, G.I.; Ross, S.R.; Wagner, K.E. Behavioral correlates and welfare implications of informal interactions between caretakers and zoo-housed chimpanzees and gorillas. Appl. Anim. Behav. Sci. 2013, 147, 306–315. [Google Scholar] [CrossRef]
- Carlstead, K.; Mellen, J.; Kleiman, D.G. Black rhinoceros (Diceros bicornis) in U.S. zoos: I. Individual behavior profiles and their relationship to breeding success. Zoo Biol. 1999, 18, 14–34. [Google Scholar] [CrossRef]
- Carlstead, K.; Fraser, J.; Bennett, C.; Kleiman, D.G. Black rhinoceros (Diceros bicornis) in U.S. zoos: II. Behavior, breeding success and mortality in relation to housing facilities. Zoo Biol. 1999, 18, 35–52. [Google Scholar] [CrossRef]
- Carlstead, K.; Shepherdson, D.; Sheppard, C.; Mellen, J.; Bennet, C. Constructing Behavioural Profiles for Zoo Animals: Incorporating Behavioural Information into Captive Population Management; American Zoo and Aquarium Association’s Behaviour Husbandry Advisory Group, Oregon Zoo: Silver Spring, MD, USA, 2000. [Google Scholar]
- Swaisgood, R.R.; Shepherdson, D. Scientific approaches to enrichment and stereotypies in zoo animals: What’s been done and where should we go next? Zoo Biol. 2005, 24, 499–518. [Google Scholar] [CrossRef]
- Powell, D.M.; Hong, L.; Carlstead, K.; Kleiman, D.; Zhang, H.; Zhang, G.; Zhang, Z.; Yu, J.; Zhang, J.G.; Lu, Y.; et al. Relationships between temperament, husbandry, management, and socio-sexual behavior in capive male and female giant pandas (Ailuropoda melanoleuca). Acta Zool. Sinica. 2008, 1, 169–175. [Google Scholar]
- Mellor, D.J.; Hunt, S.; Gusset, M. Caring for Wildlife: The World Zoo and Aquarium Animal Welfare Strategy; WAZA Executive Office: Gland, Switzerland, 2015. [Google Scholar]
- Brando, S. Animal Learning and Training Implications for Animal Welfare. Vet. Clin. N. Am. Ex. Anim. Prac. 2012, 15, 387–398. [Google Scholar] [CrossRef]
- Ramirez, K. Marine Mammal Training: The history of training animals for medical behaviours and keys to their success. Vet. Clin. N. Am.: Exotic Anim. Practice. 2012, 15, 413–423. [Google Scholar]
- Laule, G.E.; Bloomsmith, M.A.; Schapiro, S.J. The use of positive reinforcement training techniques to enhance the care, management, and welfare of primates in the laboratory. J. Appl. Anim. Welf. Sci. 2003, 6, 163–173. [Google Scholar] [CrossRef]
- Pomerantz, O.; Terkel, J. Effects of positive reinforcement training techniques on the psychological welfare of zoo-housed chimpanzees (Pan troglodytes). Am. J. Primatol. 2009, 71, 687–695. [Google Scholar] [CrossRef]
- Ward, S.J.; Melfi, V. Keeper-animal interactions: Differences between the behaviour of zoo animals affect stockmanship. PLoS ONE 2015, 10, e0140237. [Google Scholar] [CrossRef]
- Clegg, I.L.; Borger-Turner, J.L.; Eskelinen, H.C. C-Well: The development of a welfare assessment index for captive bottlenose dolphins (Tursiops truncatus). Anim. Welf. 2015, 24, 267–282. [Google Scholar] [CrossRef]
- Clegg, I.L.K.; Van Elk, C.E.; Delfour, F. Applying welfare science to bottlenose dolphins (Tursiops truncatus). Anim. Welf. 2017, 26, 165–176. [Google Scholar] [CrossRef]
- Clegg, I.L.K.; Delfour, F. Can we assess marine mammal welfare in captivity and in the wild? Considering the example of bottlenose dolphins. Aquat. Mamm. 2018, 44, 181–200. [Google Scholar] [CrossRef]
- Tallo-Parra, O.; Delfour, F.; von Fersen, L.; Garcia-Parraga, D.; Manteca, X.; Monreal-Pawlowsky, T.; Pilenga, C.; Ternes, K.; Clegg, I.L.K.; Garcia Hartmann, M.; et al. Dolphin-WET (Welfare Evaluation Tool): A new conceptual framework for welfare evaluation of bottlenose dolphins (Tursiops truncatus) under human care. In Proceedings of the E.A.Z.A. Annual Conf. Symposium, Valencia, Spain, 17–21 September 2019. [Google Scholar]
- Clegg, I.L.K.; Rödel, H.G.; Mercera, B.; van der Heul, S.; Schrijvers, T.; de Laender, P.; Gojceta, R.; Zimmitti, M.; Verhoeven, E.; Burger, J.; et al. Dolphins’ willingness to participate (WtP) in positive reinforcement training as a potential welfare indicator, where WtP predicts early changes in health status. Front. Psychol. 2019, 10, 2112. [Google Scholar] [CrossRef]
- Dalla Costa, E.; Dai, F.; Lebelt, D.; Scholz, P.; Barbieri, S.; Canali, E.; Zanella, A.J.; Minero, M. Welfare assessment of horses: The AWIN approach. Anim. Welf. 2016, 25, 481–488. [Google Scholar] [CrossRef]
- Yon, L.; Williams, E.; Harvey, N.D.; Asher, L. Development of a behavioural welfare assessment tool for routine use with captive elephants. PLoS ONE 2019, 14, 1–20. [Google Scholar] [CrossRef]
- Clegg, I.L.K.; Rödel, H.G.; Boivin, X.; Delfour, F. Looking forward to interacting with their caretakers: Dolphins’ anticipatory behaviour indicates motivation to participate in specific events. Appl. Anim. Behav. Sci. 2018, 202, 85–93. [Google Scholar] [CrossRef]
- Welsh, T.; Ward, S.J. Implications of human-animal interactions on mother-calf interactions in a Bottlenose Dolphin (Tursiops truncatus) dyad. J. Zoo Aquar. Res. 2019, 7, 162–169. [Google Scholar]
- Tomonaga, M.; Uwano, Y.; Ogura, S.; Saito, T. Bottlenose dolphins’ (Tursiops truncatus) theory of mind as demonstrated by responses to their trainers’ attentional states. Intern. J. Comp. Psychol. 2010, 23, 386–400. [Google Scholar]
- Tomonaga, M.; Uwano, Y.; Ogura, S.; Chin, H.; Dozaki, M.; Saito, T. Which person is my trainer? Spontaneous visual discrimination of human individuals by bottlenose dolphins (Tursiops truncatus). SpringerPlus 2015, 4, 352. [Google Scholar] [CrossRef] [PubMed]
- Pack, A.A.; Herman, L.M. Bottlenosed dolphins (Tursiops truncatus) comprehend the referent of both static and dynamic human gazing and pointing in an object-choice task. J. Compar. Psychol. 2004, 118, 160–171. [Google Scholar] [CrossRef]
- Xitco, M.J.; Gory, J.D.; Kuczaj, S.A. Dolphin pointing is linked to the attentional behavior of a receiver. Anim. Cogn. 2004, 7, 231–238. [Google Scholar] [CrossRef] [PubMed]
- R Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- Bates, D.; Maechler, M.; Bolker, B.; Walker, S. Fitting linear mixed-effects models using lme4. J. Stat. Softw. 2015, 67, 1–48. [Google Scholar] [CrossRef]
- Christensen, R.H.B. Ordinal—Regression Models for Ordinal Data. R Package Version 2019. Available online: http://www.cran.r-project.org/package=ordinal/ (accessed on 12 April 2020).
- Faraway, J.J. Extending the Linear Model with R. Generalized Linear, Mixed Effects and Nonparametric Regression Models; Chapman & Hall: Boca Raton, CA, USA, 2006. [Google Scholar]
- Browne, W.J.; Subramanian, S.V.; Jones, K.; Goldstein, H. Variance partitioning in multilevel logistic models that exhibit over dispersion. J. Roy. Stat. Soc. Ser. A 2005, 168, 599–613. [Google Scholar] [CrossRef]
- Serrapica, M.; Boivin, X.; Coulon, M.; Braghieri, A.; Napolitano, F. Positive perception of human stroking by lambs: Qualitative behaviour assessment confirms previous interpretation of quantitative data. Appl. Anim. Behav. Sci. 2017, 187, 31–37. [Google Scholar] [CrossRef]
- Whitham, J.C.; Wielebnowski, N. Animal-based welfare monitoring: Using keeper ratings as an assessment tool. Zoo Biol. 2009, 28, 545–560. [Google Scholar] [CrossRef]
- Servais, V.; Delfour, F. Comment étudier la communication entre dauphins captifs et soigneurs? In Proceedings of the International Conference III 2011 “Cross-Understanding: From Intraspecific to Interspecific”, Landaul, France, 25–26 November 2011; pp. 141–156. [Google Scholar]
- Birgersson, S.; de la Pommeraye, S.B.; Delfour, F. Dolphin Personality Study Based on Ethology and Social Network Theory; LAP Lambert, Academic Publishing: Riga, Lettonia, 2014. [Google Scholar]
- Estep, D.Q.; Hetts, S. Interactions, relationships, and bonds: The conceptual basis for scientist–animal relations. In The Inevitable Bond: Examining Scientist-Animal Interactions; Davis, H., Balfour, D., Eds.; Cambridge University Press: Cambridge, UK, 1992; pp. 6–26. [Google Scholar]
- Tsai, Y.J.J.; Mann, J. Dispersal, philopatry, and the role of fission-fusion dynamics in bottlenose dolphins. Mar. Mam. Sci. 2013, 29, 261–279. [Google Scholar] [CrossRef]
- Aplin, L.M.; Farine, D.R.; Mann, R.P.; Sheldon, B.C. Individual level personality influences social foraging and collective behaviour in wild birds. Proc. Royal Soc. Biol. Sci. 2014, 281, 1789. [Google Scholar] [CrossRef]
- Dall, S.R.X.; Houston, A.I.; McNamara, J.M. The behavioural ecology of personality: Consistent individual differences from an adaptive perspective. Ecol. Lett. 2004, 7, 734–739. [Google Scholar] [CrossRef]
- Gosling, S.D. Personality in non-human animals. Soc. Personal. Psychol. Compass 2008, 2, 985–1001. [Google Scholar] [CrossRef]
- Sih, A.; Bell, A.M.; Johnson, J.C.; Ziemba, R.E. Behavioural syndromes: An integrative overview. Q. Rev. Biol. 2004, 79, 241–277. [Google Scholar] [CrossRef] [PubMed]
- Wolf, M.; Weissing, F.J. Animal personalities: Consequences for ecology and evolution. Trends Ecol. Evol. 2012, 27, 452–461. [Google Scholar] [CrossRef] [PubMed]
- Serres, A.; Delfour, F. Environmental changes and anthropogenic factors modulate social play in captive bottlenose dolphins (Tursiops truncatus). Zoo Biol. 2017, 36, 99–111. [Google Scholar] [CrossRef]
- Birke, L.; Hockenhull, J.; Creighton, E.; Pinno, L.; Mee, J.; Mills, D. Horses’ responses to variation in human approach. Appl. Anim. Behav. Sci. 2011, 134, 56–63. [Google Scholar] [CrossRef]
- Herron, M.E.; Kirby-Madden, T.M.; Lord, L.K. Effects of environmental enrichment on the behavior of shelter dogs. J. Am. Vet. Medic. Assoc. 2014, 244, 687–692. [Google Scholar] [CrossRef] [PubMed]
- Henriksson, J.; Sauveroche, M.; Roth, L.S. Effects of size and personality on social learning and human-directed behaviour in horses (Equus caballus). Anim. Cogn. 2019, 22, 1001–1011. [Google Scholar] [CrossRef]
- Pack, A.A.; Herman, L.M. Dolphin social cognition and joint attention: Our current understanding. Aquat. Mamm. 2006, 32, 443–460. [Google Scholar] [CrossRef]
- Highfill, L.E.; Yeater, D.; Kuczaj II, S.A. Catch! Dolphin (Tursiops truncatus) ball tossing to humans is affected by human perspective. Aquat. Mamm. 2016, 42, 253–258. [Google Scholar] [CrossRef]
- Herzing, D.L.; Delfour, F.; Pack, A.A. Responses of human-habituated wild atlantic spotted dolphins to play behaviors using a two-way human/dolphin interface. Int. J. Comp. Psychol. 2012, 25, 137–165. [Google Scholar]
- Harvey, A.M.; Beausoleil, N.J.; Ramp, D.; Mellor, D.J. A Ten-Stage protocol for assessing the welfare of individual non-captive wild animals: Free-roaming horses (Equus ferus caballus) as an example. Animals 2020, 10, 148. [Google Scholar] [CrossRef] [PubMed]
- Walker, R.T.; Miller, L.J.; Kuczaj, S.; Solangi, M. Seasonal, diel, and age differences in activity budgets of a group of bottlenose dolphins (Tursiops truncatus) under professional care. Int. J. Comp. Psychol. 2017, 30, 1–12. [Google Scholar]


| Facility | Females | Males | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| N Dolphins | Range of Age | N Dolphins | Range of Age | N Sessions Observed | |||||||
| N total | Juv. | Ad. | Juv. | Ad. | Juv. | Ad. | Juv. | Ad. | Total | Per Dolphin | |
| Parc Astérix | 7 | 2 | 3 | 2–4 | 20–46 | 0 | 2 | - | 35–37 | 70 | 10 |
| Mundomar | 7 | 0 | 5 | - | 12–45 | 1 | 1 | 4 | 33 | 68 | 10 |
| Oceanogràfic Valencia | 16 | 1 | 8 | 2 | 13-40 | 2 | 5 | 6–10 | 14–35 | 52 | 1-10 |
| Zoomarine Italia | 10 | 2 | 2 | 3–8 | 18 | 2 | 4 | 5–10 | 21–36 | 110 | 10–12 |
| Barcelona | 3 | 0 | 0 | - | - | 1 | 2 | 8 | 17–22 | 23 | 7–8 |
| Total | 43 | 5 | 18 | - | - | 6 | 14 | 323 | - | ||
| Behavioural Parameters | N | Predictors | χ2 | df | P |
|---|---|---|---|---|---|
| (a) Speed of approach | 322 | Sex S | 1.552 | 1 | 0.213 |
| Age class A | 0.385 | 1 | 0.535 | ||
| Trainers’ experience TE | 0.219 | 1 | 0.640 | ||
| Ind./group session IG | 2.148 | 1 | 0.143 | ||
| Nature of session NS | 2.284 | 4 | 0.684 | ||
| Time of day TD | 3.418 | 2 | 0.181 | ||
| A × IG | 4.504 | 1 | 0.034 | ||
| (b) Excitement level | 320 | Sex S | 1.585 | 1 | 0.208 |
| Age class A | 9.615 | 1 | 0.002 | ||
| Trainers’ experience TE | 1.486 | 1 | 0.223 | ||
| Ind./group session IG | 0.135 | 1 | 0.713 | ||
| Nature of session NS | 5.754 | 4 | 0.218 | ||
| Time of day TD | 4.576 | 2 | 0.101 | ||
| TE × IG | 6.532 | 1 | 0.010 | ||
| (c) Positive responses to commands | 203 | Sex S | 0.428 | 1 | 0.513 |
| Age class A | 3.387 | 1 | 0.066 | ||
| Trainers’ experience TE | 0.190 | 1 | 0.663 | ||
| Ind./group session IG | 1.182 | 1 | 0.277 | ||
| Nature of session NS | 82.615 | 4 | <0.001 | ||
| Time of day TD | 7.530 | 2 | 0.023 | ||
| (d) Refusal to perform a behaviour | 321 | Sex S | 0.293 | 1 | 0.588 |
| Age class A | 2.311 | 1 | 0.128 | ||
| Trainers’ experience TE | 2.858 | 1 | 0.091 | ||
| Ind./group session IG | 1.493 | 1 | 0.221 | ||
| Nature of session NS | 10.382 | 4 | 0.034 | ||
| Time of day TD | 0.179 | 2 | 0.915 | ||
| (e) Leaving during session | 322 | Sex S | 0.795 | 1 | 0.372 |
| Age class A | 14.231 | 1 | <0.001 | ||
| Trainers’ experience TE | 0.162 | 1 | 0.687 | ||
| Ind./group session IG | 2.218 | 1 | 0.136 | ||
| Nature of session NS | 0.898 | 4 | 0.925 | ||
| Time of day TD | 1.523 | 2 | 0.467 | ||
| (f) Eye contact with trainer | 323 | Sex S | 0.436 | 1 | 0.509 |
| Age class A | 0.310 | 1 | 0.578 | ||
| Trainers’ experience TE | 0.205 | 1 | 0.651 | ||
| Ind./group session IG | 0.493 | 1 | 0.483 | ||
| Nature of session NS | 3.528 | 4 | 0.474 | ||
| Time of day TD | 3.348 | 2 | 0.188 | ||
| (g) Approach speed when trainer enters pool | 99 | Sex S | 0.093 | 1 | 0.761 |
| Age class A | 0.446 | 1 | 0.504 | ||
| Trainers’ experience TE | 1.913 | 1 | 0.167 | ||
| Ind./group session IG | 0.001 | 1 | 0.998 | ||
| Nature of session NS | 0.001 | 4 | 0.999 | ||
| Time of day TD | 1.063 | 2 | 0.587 | ||
| (h) Willingness to participate | 323 | Sex S | 0.001 | 1 | 0.999 |
| Age class A | 0.001 | 1 | 0.999 | ||
| Trainers’ experience TE | 0.053 | 1 | 0.818 | ||
| Ind./group session IG | 3.706 | 1 | 0.054 | ||
| Nature of session NS | 4.151 | 4 | 0.386 | ||
| Time of day TD | 8.647 | 2 | 0.013 | ||
| S × A | 4.058 | 1 | 0.044 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Delfour, F.; Monreal-Pawlowsky, T.; Vaicekauskaite, R.; Pilenga, C.; Garcia-Parraga, D.; Rödel, H.G.; García Caro, N.; Perlado Campos, E.; Mercera, B. Dolphin Welfare Assessment under Professional Care: ‘Willingness to Participate’, an Indicator Significantly Associated with Six Potential ‘Alerting Factors’. J. Zool. Bot. Gard. 2020, 1, 42-60. https://doi.org/10.3390/jzbg1010004
Delfour F, Monreal-Pawlowsky T, Vaicekauskaite R, Pilenga C, Garcia-Parraga D, Rödel HG, García Caro N, Perlado Campos E, Mercera B. Dolphin Welfare Assessment under Professional Care: ‘Willingness to Participate’, an Indicator Significantly Associated with Six Potential ‘Alerting Factors’. Journal of Zoological and Botanical Gardens. 2020; 1(1):42-60. https://doi.org/10.3390/jzbg1010004
Chicago/Turabian StyleDelfour, Fabienne, Tania Monreal-Pawlowsky, Ruta Vaicekauskaite, Cristina Pilenga, Daniel Garcia-Parraga, Heiko G. Rödel, Nuria García Caro, Enrique Perlado Campos, and Birgitta Mercera. 2020. "Dolphin Welfare Assessment under Professional Care: ‘Willingness to Participate’, an Indicator Significantly Associated with Six Potential ‘Alerting Factors’" Journal of Zoological and Botanical Gardens 1, no. 1: 42-60. https://doi.org/10.3390/jzbg1010004
APA StyleDelfour, F., Monreal-Pawlowsky, T., Vaicekauskaite, R., Pilenga, C., Garcia-Parraga, D., Rödel, H. G., García Caro, N., Perlado Campos, E., & Mercera, B. (2020). Dolphin Welfare Assessment under Professional Care: ‘Willingness to Participate’, an Indicator Significantly Associated with Six Potential ‘Alerting Factors’. Journal of Zoological and Botanical Gardens, 1(1), 42-60. https://doi.org/10.3390/jzbg1010004





