Immune Modulatory Effects of Ketogenic Diet in Different Disease Conditions
Abstract
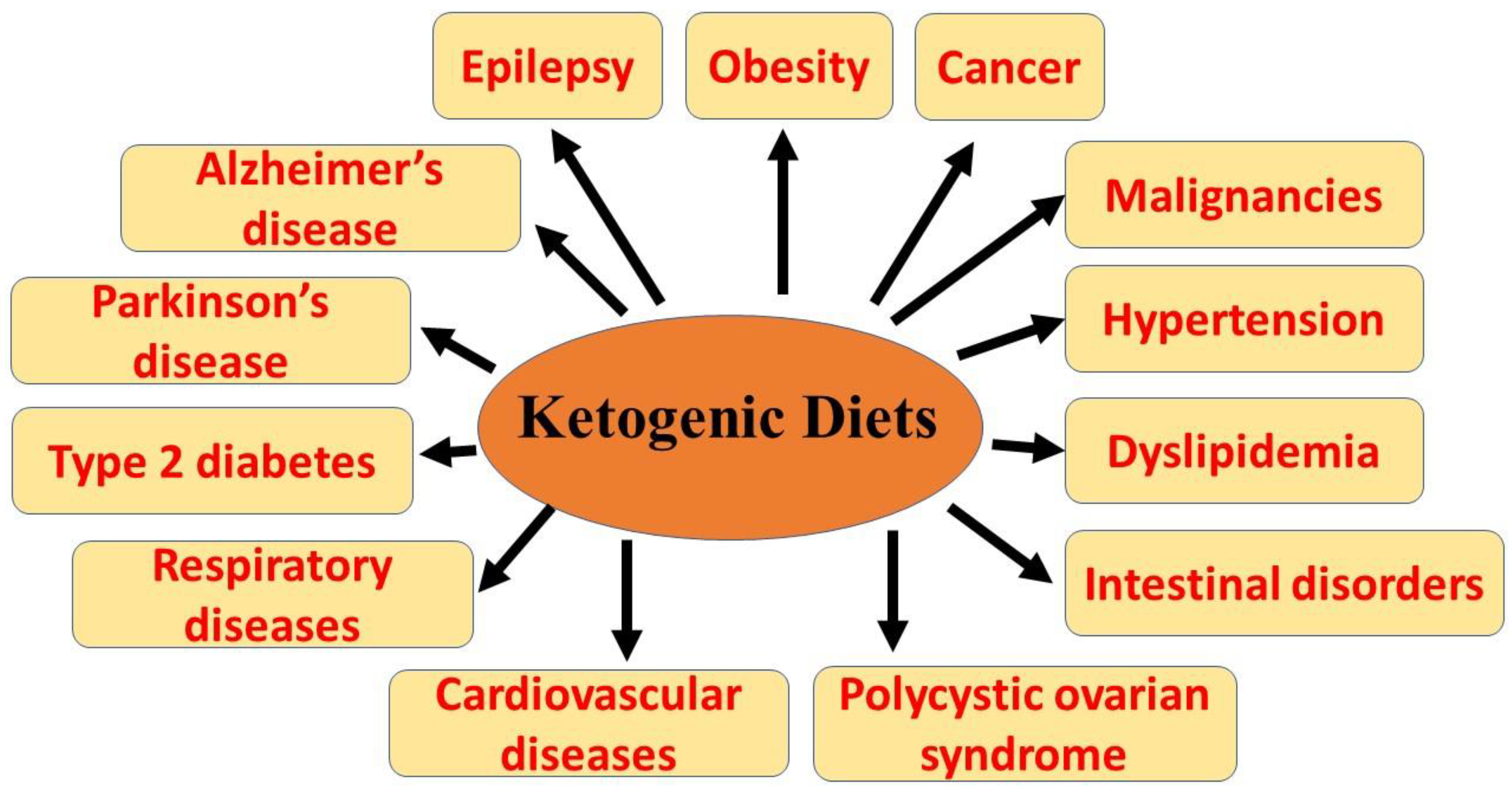
:1. Introduction
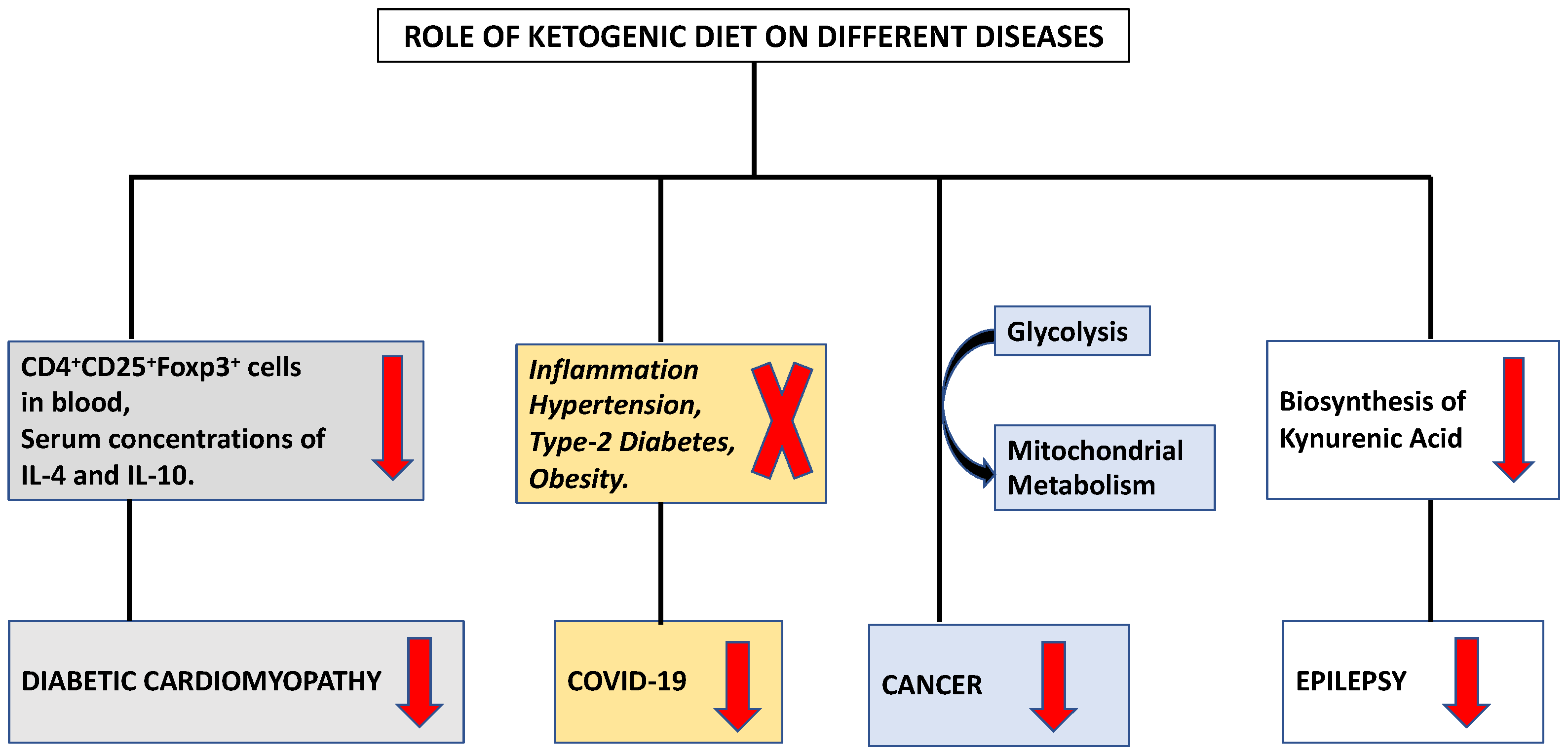
2. Immune Modulation by Ketogenic Diet in Viral Infection
3. Ketogenic Diet Mediated Immune Regulation in Cancer
4. Ketogenic Diet Mediated Immune Regulation in Cardiovascular Diseases
5. Ketogenic Diet Mediated Immune Modulation in Central Nervous System Diseases
6. Keto Diet Complications:
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Zhu, H.; Bi, D.; Zhang, Y.; Kong, C.; Du, J.; Wu, X.; Wei, Q.; Qin, H. Ketogenic diet for human diseases: The underlying mechanisms and potential for clinical implementations. Signal Transduct. Target. Ther. 2022, 7, 1–21. [Google Scholar] [CrossRef] [PubMed]
- Joanne Carroll, M.R.D.K. The Ketogenic Diet: A Practical Guide for Caregivers. J. Am. Diet. Assoc. 1998, 98, 316–321. [Google Scholar] [CrossRef] [PubMed]
- Pardo, A.C. Ketogenic Diet: A Role in Immunity? Pediatr. Neurol. Briefs 2020, 34, 5. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dowis, K.; Banga, S. The Potential Health Benefits of the Ketogenic Diet: A Narrative Review. Nutrients 2021, 13, 1654. [Google Scholar] [CrossRef] [PubMed]
- Kakodkar, P.; Arnold, A.; Ali, A.; Kaka, N. Ketogenic Diet: Biochemistry, Weight Loss and Clinical Applications. Nutr. Food Sci. Int. J. 2020, 10, 1–9. [Google Scholar] [CrossRef]
- Ludwig, D.S. The Ketogenic Diet: Evidence for Optimism but High-Quality Research Needed. J. Nutr. 2019, 150, 1354–1359. [Google Scholar] [CrossRef] [Green Version]
- Westman, E.C.; Mavropoulos, J.; Yancy, W.S.; Volek, J.S. A review of low-carbohydrate ketogenic diets. Curr. Atheroscler. Rep. 2003, 5, 476–483. [Google Scholar] [CrossRef]
- Ciaffi, J.; Mitselman, D.; Mancarella, L.; Brusi, V.; Lisi, L.; Ruscitti, P.; Cipriani, P.; Meliconi, R.; Giacomelli, R.; Borghi, C.; et al. The Effect of Ketogenic Diet on Inflammatory Arthritis and Cardiovascular Health in Rheumatic Conditions: A Mini Review. Front. Med. 2021, 8. [Google Scholar] [CrossRef]
- Castaldo, G.; Pagano, I.; Grimaldi, M.; Marino, C.; Molettieri, P.; Santoro, A.; Stillitano, I.; Romano, R.; Montoro, P.; D’Ursi, A.M.; et al. Effect of Very-Low-Calorie Ketogenic Diet on Psoriasis Patients: A Nuclear Magnetic Resonance-Based Metabolomic Study. J. Proteome Res. 2020, 20, 1509–1521. [Google Scholar] [CrossRef]
- Kong, C.; Yan, X.; Liu, Y.; Huang, L.; Zhu, Y.; He, J.; Gao, R.; Kalady, M.F.; Goel, A.; Qin, H.; et al. Ketogenic diet alleviates colitis by reduction of colonic group 3 innate lymphoid cells through altering gut microbiome. Signal Transduct. Target. Ther. 2021, 6, 1–12. [Google Scholar] [CrossRef]
- Charlot, A.; Boumiza, R.; Roux, M.; Zoll, J. Obésité, inflammation et COVID-19: Intérêt préventif de l’alimentation cétogène? Biol. Aujourd’hui 2021, 215, 63–72. [Google Scholar] [CrossRef] [PubMed]
- Paoli, A.; Gorini, S.; Caprio, M. The dark side of the spoon—glucose, ketones and COVID-19: A possible role for ketogenic diet? J. Transl. Med. 2020, 18, 441. [Google Scholar] [CrossRef] [PubMed]
- Gangitano, E.; Tozzi, R.; Gandini, O.; Watanabe, M.; Basciani, S.; Mariani, S.; Lenzi, A.; Gnessi, L.; Lubrano, C. Ketogenic Diet as a Preventive and Supportive Care for COVID-19 Patients. Nutrients 2021, 13, 1004. [Google Scholar] [CrossRef] [PubMed]
- Soliman, S.; Faris, M.E.; Ratemi, Z.; Halwani, R. Switching Host Metabolism as an Approach to Dampen SARS-CoV-2 Infection. Ann. Nutr. Metab. 2020, 76, 297–303. [Google Scholar] [CrossRef] [PubMed]
- Sukkar, S.G.; Cogorno, L.; Pisciotta, L.; Pasta, A.; Vena, A.; Gradaschi, R.; Dentone, C.; Guiddo, E.; Martino, E.; Beltramini, S.; et al. Clinical efficacy of eucaloric ketogenic nutrition in the COVID-19 cytokine storm: A retrospective analysis of mortality and intensive care unit admission. Nutrition 2021, 89, 111236. [Google Scholar] [CrossRef] [PubMed]
- Karagiannis, F.; Peukert, K.; Surace, L.; Michla, M.; Nikolka, F.; Fox, M.; Weiss, P.; Feuerborn, C.; Maier, P.; Schulz, S.; et al. Impaired ketogenesis ties metabolism to T cell dysfunction in COVID-19. Nature 2022, 609, 801–807. [Google Scholar] [CrossRef]
- Ryu, S.; Shchukina, I.; Youm, Y.-H.; Qing, H.; Hilliard, B.; Dlugos, T.; Zhang, X.; Yasumoto, Y.; Booth, C.J.; Fernández-Hernando, C.; et al. Ketogenic diet restrains aging-induced exacerbation of coronavirus infection in mice. Elife 2021, 10. [Google Scholar] [CrossRef]
- Goldberg, E.L.; Molony, R.D.; Kudo, E.; Sidorov, S.; Kong, Y.; Dixit, V.D.; Iwasaki, A. Ketogenic diet activates protective γδ T cell responses against influenza virus infection. Sci. Immunol. 2019, 4. [Google Scholar] [CrossRef]
- Bradshaw, P.C.; Seeds, W.A.; Miller, A.C.; Mahajan, V.R.; Curtis, W.M. COVID-19: Proposing a Ketone-Based Metabolic Therapy as a Treatment to Blunt the Cytokine Storm. Oxidative Med. Cell. Longev. 2020, 2020, 6401341. [Google Scholar] [CrossRef]
- Hirschberger, S.; Gellert, L.; Effinger, D.; Muenchhoff, M.; Herrmann, M.; Briegel, J.-M.; Zwißler, B.; Kreth, S. Ketone Bodies Improve Human CD8+ Cytotoxic T-Cell Immune Response During COVID-19 Infection. Front. Med. 2022, 9. [Google Scholar] [CrossRef]
- Klement, R.J. Fasting, Fats, and Physics: Combining Ketogenic and Radiation Therapy against Cancer. Complement. Med. Res. 2017, 25, 102–113. [Google Scholar] [CrossRef]
- Rous, P. The influence of diet on transplanted and spontaneous mouse tumors. J. Exp. Med. 1914, 20, 433–451. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tisdale, M.; Brennan, R.; Fearon, K. Reduction of weight loss and tumour size in a cachexia model by a high fat diet. Br. J. Cancer 1987, 56, 39–43. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klement, R.J.; Champ, C.E.; Otto, C.; Kämmerer, U. Anti-Tumor Effects of Ketogenic Diets in Mice: A Meta-Analysis. PLOS ONE 2016, 11, e0155050. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferrere, G.; Alou, M.T.; Liu, P.; Goubet, A.-G.; Fidelle, M.; Kepp, O.; Durand, S.; Iebba, V.; Fluckiger, A.; Daillère, R.; et al. Ketogenic diet and ketone bodies enhance the anticancer effects of PD-1 blockade. J. Clin. Investig. 2021, 6. [Google Scholar] [CrossRef] [PubMed]
- Hao, G.-W.; Chen, Y.-S.; He, D.-M.; Wang, H.-Y.; Wu, G.-H.; Zhang, B. Growth of Human Colon Cancer Cells in Nude Mice is Delayed by Ketogenic Diet With or Without Omega-3 Fatty Acids and Medium-chain Triglycerides. Asian Pac. J. Cancer Prev. 2015, 16, 2061–2068. [Google Scholar] [CrossRef] [Green Version]
- Nebeling, L.C.; Miraldi, F.; Shurin, S.B.; Lerner, E. Effects of a ketogenic diet on tumor metabolism and nutritional status in pediatric oncology patients: Two case reports. J. Am. Coll. Nutr. 1995, 14, 202–208. [Google Scholar] [CrossRef]
- Seyfried, T.N.; Shelton, L.M. Cancer as a metabolic disease. Nutr. Metab. 2010, 7, 7. [Google Scholar] [CrossRef] [Green Version]
- Abdelwahab, M.G.; Fenton, K.E.; Preul, M.C.; Rho, J.M.; Lynch, A.; Stafford, P.; Scheck, A.C. The Ketogenic Diet Is an Effective Adjuvant to Radiation Therapy for the Treatment of Malignant Glioma. PLoS ONE 2012, 7, e36197. [Google Scholar] [CrossRef]
- Sharpe, A.; Pauken, K. The diverse functions of the PD1 inhibitory pathway. Nat. Rev. Immunol. 2018, 18, 153–167. [Google Scholar] [CrossRef]
- Dai, X.; Bu, X.; Gao, Y.; Guo, J.; Hu, J.; Jiang, C.; Zhang, Z.; Xu, K.; Duan, J.; He, S.; et al. Energy status dictates PD-L1 protein abundance and anti-tumor immunity to enable checkpoint blockade. Mol. Cell 2021, 81, 2317–2331.e6. [Google Scholar] [CrossRef]
- Mauldin, I.S.; Wages, N.A.; Stowman, A.M.; Wang, E.; Smolkin, M.E.; Olson, W.C.; Deacon, D.H.; Smith, K.T.; Galeassi, N.V.; Chianese-Bullock, K.A.; et al. Intratumoral interferon-gamma increases chemokine production but fails to increase T cell infiltration of human melanoma metastases. Cancer Immunol. Immunother. 2016, 65, 1189–1199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sun, W.; Yang, J.; Liu, B.; Liu, Q.; Wang, T.; Wang, Q.; Liu, M.; Li, L.; Wang, Z.; Li, S.; et al. Ketogenic diet inhibits tumor growth by enhancing immune response, attenuating immunosuppression, inhibiting angiogenesis and EMT in CT26 colon tumor allografts mouse model. J. Funct. Foods 2022, 92, 105067. [Google Scholar] [CrossRef]
- Meade, T. Hypercoagulability and ischaemic heart disease. Blood Rev. 1987, 1, 2–8. [Google Scholar] [CrossRef] [PubMed]
- Lussier, D.M.; Woolf, E.C.; Johnson, J.L.; Brooks, K.S.; Blattman, J.N.; Scheck, A.C. Enhanced immunity in a mouse model of malignant glioma is mediated by a therapeutic ketogenic diet. BMC Cancer 2016, 16, 1–10. [Google Scholar] [CrossRef] [Green Version]
- Sun, H.Y.; Sebaratnam, D.F. Prurigo pigmentosa following a ketogenic diet: A case report. Eur. J. Clin. Nutr. 2021, 76, 624–625. [Google Scholar] [CrossRef]
- Talib, W.H.; Mahmod, A.I.; Kamal, A.; Rashid, H.M.; Alashqar, A.; Khater, S.; Jamal, D.; Waly, M. Ketogenic Diet in Cancer Prevention and Therapy: Molecular Targets and Therapeutic Opportunities. Curr. Issues Mol. Biol. 2021, 43, 558–589. [Google Scholar] [CrossRef]
- Meynet, O.; Ricci, J.-E. Caloric restriction and cancer: Molecular mechanisms and clinical implications. Trends Mol. Med. 2014, 20, 419–427. [Google Scholar] [CrossRef]
- Hursting, S.D.; Ford, N.A.; Dunlap, S.M.; Hursting, M.J.; Lashinger, L.M. Calorie Restriction and Cancer Prevention: Established and Emerging Mechanisms. In Obesity, Inflammation and Cancer; Springer: New York, NY, USA, 2013; pp. 363–379. [Google Scholar] [CrossRef]
- Mulrooney, T.J.; Marsh, J.; Urits, I.; Seyfried, T.; Mukherjee, P. Influence of Caloric Restriction on Constitutive Expression of NF-κB in an Experimental Mouse Astrocytoma. PLoS ONE 2011, 6, e18085. [Google Scholar] [CrossRef]
- Zhang, N.; Liu, C.; Zhang, R.; Jin, L.; Yin, X.; Zheng, X.; Siebert, H.-C.; Li, Y.; Wang, Z.; Loers, G.; et al. Amelioration of clinical course and demyelination in the cuprizone mouse model in relation to ketogenic diet. Food Funct. 2020, 11, 5647–5663. [Google Scholar] [CrossRef]
- Murray, P.J.; Wynn, T.A. Protective and pathogenic functions of macrophage subsets. Nat. Rev. Immunol. 2011, 11, 723–737. [Google Scholar] [CrossRef] [Green Version]
- Choi, J.-W.; Hua, T.N. Impact of Lifestyle Behaviors on Cancer Risk and Prevention. J. Lifestyle Med. 2021, 11, 1–7. [Google Scholar] [CrossRef] [PubMed]
- Zou, Y.; Fineberg, S.; Pearlman, A.; Feinman, R.D.; Fine, E.J. The effect of a ketogenic diet and synergy with rapamycin in a mouse model of breast cancer. PLOS ONE 2020, 15, e0233662. [Google Scholar] [CrossRef] [PubMed]
- Rosenzweig, R.; Kumar, V.; Gupta, S.; Bermeo-Blanco, O.; Stratton, M.S.; Gumina, R.J.; Bansal, S.S. Estrogen Receptor-β Agonists Modulate T-Lymphocyte Activation and Ameliorate Left Ventricular Remodeling During Chronic Heart Failure. Circ. Hear. Fail. 2022, 15. [Google Scholar] [CrossRef] [PubMed]
- Rosenzweig, R.; Gupta, S.; Kumar, V.; Gumina, R.J.; Bansal, S.S. Estrogenic bias in T-Lymphocyte biology: Implications for cardiovascular disease. Pharmacol. Res. 2021, 170, 105606. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Prabhu, S.D.; Bansal, S.S. CD4+ T-lymphocytes exhibit biphasic kinetics post-myocardial infarction. Front. Cardiovasc. Med. 2022, 9. [Google Scholar] [CrossRef] [PubMed]
- Kumar, V.; Rosenzweig, R.; Asalla, S.; Nehra, S.; Prabhu, S.D.; Bansal, S.S. TNFR1 Contributes to Activation-Induced Cell Death of Pathological CD4+ T Lymphocytes During Ischemic Heart Failure. JACC: Basic Transl. Sci. 2022, 7, 1038–1049. [Google Scholar] [CrossRef]
- Visioli, F.; Mucignat-Caretta, C.; Anile, F.; Panaite, S.-A. Traditional and Medical Applications of Fasting. Nutrients 2022, 14, 433. [Google Scholar] [CrossRef]
- Kirkpatrick, C.F.; Bolick, J.P.; Kris-Etherton, P.M.; Sikand, G.; Aspry, K.E.; Soffer, D.E.; Willard, K.-E.; Maki, K.C. Review of current evidence and clinical recommendations on the effects of low-carbohydrate and very-low-carbohydrate (including ketogenic) diets for the management of body weight and other cardiometabolic risk factors: A scientific statement from the National Lipid Association Nutrition and Lifestyle Task Force. J. Clin. Lipidol. 2019, 13, 689–711.e1. [Google Scholar] [CrossRef]
- Guarente, L. Calorie restriction and sirtuins revisited. Genes Dev. 2013, 27, 2072–2085. [Google Scholar] [CrossRef] [Green Version]
- Imai, S.-I.; Guarente, L. It takes two to tango: NAD+ and sirtuins in aging/longevity control. npj Aging Mech. Dis. 2016, 2, 16017. [Google Scholar] [CrossRef] [Green Version]
- De Toledo, F.W.; Grundler, F.; Sirtori, C.R.; Ruscica, M. Unravelling the health effects of fasting: A long road from obesity treatment to healthy life span increase and improved cognition. Ann. Med. 2020, 52, 147–161. [Google Scholar] [CrossRef] [PubMed]
- Osataphan, S.; Macchi, C.; Singhal, G.; Chimene-Weiss, J.; Sales, V.; Kozuka, C.; Dreyfuss, J.M.; Pan, H.; Tangcharoenpaisan, Y.; Morningstar, J.; et al. SGLT2 inhibition reprograms systemic metabolism via FGF21-dependent and -independent mechanisms. J. Clin. Investig. 2019, 4, e123130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cantó, C.; Auwerx, J. Calorie Restriction: Is AMPK a Key Sensor and Effector? Physiology 2011, 26, 214–224. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mercken, E.M.; Crosby, S.D.; Lamming, D.W.; JeBailey, L.; Krzysik-Walker, S.; Villareal, D.T.; Capri, M.; Franceschi, C.; Zhang, Y.; Becker, K.; et al. Calorie restriction in humans inhibits the PI 3 K / AKT pathway and induces a younger transcription profile. Aging Cell 2013, 12, 645–651. [Google Scholar] [CrossRef] [Green Version]
- Glatzel, D.K.; Koeberle, A.; Pein, H.; Loeser, K.; Stark, A.; Keksel, N.; Werz, O.; Mueller, R.; Bischoff, I.; Fürst, R. Acetyl-CoA carboxylase 1 regulates endothelial cell migration by shifting the phospholipid composition. J. Lipid Res. 2018, 59, 298–311. [Google Scholar] [CrossRef] [Green Version]
- Niu, C.; Chen, Z.; Kim, K.T.; Sun, J.; Xue, M.; Chen, G.; Li, S.; Shen, Y.; Zhu, Z.; Wang, X.; et al. Metformin alleviates hyperglycemia-induced endothelial impairment by downregulating autophagy via the Hedgehog pathway. Autophagy 2019, 15, 843–870. [Google Scholar] [CrossRef]
- Chen, L.; Duan, Y.; Wei, H.; Ning, H.; Bi, C.; Zhao, Y.; Qin, Y.; Li, Y. Acetyl-CoA carboxylase (ACC) as a therapeutic target for metabolic syndrome and recent developments in ACC1/2 inhibitors. Expert Opin. Investig. Drugs 2019, 28, 917–930. [Google Scholar] [CrossRef]
- Wang, X.; Zhou, Y.; Tang, D.; Zhu, Z.; Li, Y.; Huang, T.; Müller, R.; Yu, W.; Li, P. ACC1 (Acetyl Coenzyme A Carboxylase 1) Is a Potential Immune Modulatory Target of Cerebral Ischemic Stroke. Stroke 2019, 50, 1869–1878. [Google Scholar] [CrossRef]
- Norlander, A.E.; Saleh, M.A.; Kamat, N.V.; Ko, B.; Gnecco, J.; Zhu, L.; Dale, B.L.; Iwakura, Y.; Hoover, R.S.; McDonough, A.A.; et al. Interleukin-17A Regulates Renal Sodium Transporters and Renal Injury in Angiotensin II–Induced Hypertension. Hypertension 2016, 68, 167–174. [Google Scholar] [CrossRef] [Green Version]
- Ye, J.; Ji, Q.; Liu, J.; Liu, L.; Huang, Y.; Shi, Y.; Shi, L.; Wang, M.; Liu, M.; Feng, Y.; et al. Interleukin 22 Promotes Blood Pressure Elevation and Endothelial Dysfunction in Angiotensin II–Treated Mice. J. Am. Hear. Assoc. 2017, 6. [Google Scholar] [CrossRef] [Green Version]
- Caillon, A.; Mian, M.O.R.; Fraulob-Aquino, J.C.; Huo, K.-G.; Barhoumi, T.; Ouerd, S.; Sinnaeve, P.R.; Paradis, P.; Schiffrin, E.L. γδ T Cells Mediate Angiotensin II-Induced Hypertension and Vascular Injury. Circulation 2017, 135, 2155–2162. [Google Scholar] [CrossRef] [PubMed]
- Kiran, S.; Rakib, A.; Kodidela, S.; Kumar, S.; Singh, U.P. High-Fat Diet-Induced Dysregulation of Immune Cells Correlates with Macrophage Phenotypes and Chronic Inflammation in Adipose Tissue. Cells 2022, 11, 1327. [Google Scholar] [CrossRef] [PubMed]
- Mathis, K.W.; Wallace, K.; Flynn, E.R.; Maric-Bilkan, C.; Lamarca, B.; Ryan, M.J. Preventing Autoimmunity Protects Against the Development of Hypertension and Renal Injury. Hypertension 2014, 64, 792–800. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Taylor, E.B.; Barati, M.T.; Powell, D.W.; Turbeville, H.R.; Ryan, M.J. Plasma Cell Depletion Attenuates Hypertension in an Experimental Model of Autoimmune Disease. Hypertension 2018, 71, 719–728. [Google Scholar] [CrossRef]
- Chan, C.T.; Sobey, C.G.; Lieu, M.; Ferens, D.; Kett, M.M.; Diep, H.; Kim, H.A.; Krishnan, S.M.; Lewis, C.; Salimova, E.; et al. Obligatory Role for B Cells in the Development of Angiotensin II–Dependent Hypertension. Hypertension 2015, 66, 1023–1033. [Google Scholar] [CrossRef]
- Sundgren, N.C.; Vongpatanasin, W.; Boggan, B.-M.D.; Tanigaki, K.; Yuhanna, I.S.; Chambliss, K.L.; Mineo, C.; Shaul, P.W. IgG Receptor FcγRIIB Plays a Key Role in Obesity-Induced Hypertension. Hypertension 2015, 65, 456–462. [Google Scholar] [CrossRef]
- Guo, Y.; Zhang, C.; Shang, F.-F.; Luo, M.; You, Y.; Zhai, Q.; Xia, Y.; Suxin, L. Ketogenic Diet Ameliorates Cardiac Dysfunction via Balancing Mitochondrial Dynamics and Inhibiting Apoptosis in Type 2 Diabetic Mice. Aging Dis. 2020, 11, 229–240. [Google Scholar] [CrossRef] [Green Version]
- Yu, Y.; Wang, F.; Wang, J.; Zhang, D.; Zhao, X. Ketogenic diet attenuates aging-associated myocardial remodeling and dysfunction in mice. Exp. Gerontol. 2020, 140, 111058. [Google Scholar] [CrossRef]
- Ma, Y.; Zou, H.; Zhu, X.-X.; Pang, J.; Xu, Q.; Jin, Q.-Y.; Ding, Y.-H.; Zhou, B.; Huang, D.-S. Transforming growth factor β: A potential biomarker and therapeutic target of ventricular remodeling. Oncotarget 2017, 8, 53780–53790. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.; Tang, K.; Ma, J.; Zhou, L.; Liu, J.; Zeng, L.; Zhu, L.; Xu, P.; Chen, J.; Wei, K.; et al. Ketogenesis-generated β-hydroxybutyrate is an epigenetic regulator of CD8+ T-cell memory development. Nature 2019, 22, 18–25. [Google Scholar] [CrossRef]
- Mey, J.T.; Erickson, M.L.; Axelrod, C.L.; King, W.T.; A Flask, C.; McCullough, A.J.; Kirwan, J.P. β-Hydroxybutyrate is reduced in humans with obesity-related NAFLD and displays a dose-dependent effect on skeletal muscle mitochondrial respiration in vitro. Am. J. Physiol. Metab. 2020, 319, E187–E195. [Google Scholar] [CrossRef] [PubMed]
- Wu, M.; Wang, S.; Xie, Y.; Chen, Z.; Guo, Q.; Yuan, W.; Guan, C.; Xu, C.; Huang, Y.; Wang, J.; et al. Interleukin-33 alleviates diabetic cardiomyopathy through regulation of endoplasmic reticulum stress and autophagy via insulin-like growth factor-binding protein 3. J. Cell. Physiol. 2020, 236, 4403–4419. [Google Scholar] [CrossRef] [PubMed]
- Tao, J.; Chen, H.; Wang, Y.-J.; Qiu, J.-X.; Meng, Q.-Q.; Zou, R.-J.; Li, L.; Huang, J.-G.; Zhao, Z.-K.; Huang, Y.-L.; et al. Ketogenic Diet Suppressed T-Regulatory Cells and Promoted Cardiac Fibrosis via Reducing Mitochondria-Associated Membranes and Inhibiting Mitochondrial Function. Oxidative Med. Cell. Longev. 2021, 2021, 5512322. [Google Scholar] [CrossRef]
- Franchi, L.; Eigenbrod, T.; Muñoz-Planillo, R.; Nuñez, G. The inflammasome: A caspase-1-activation platform that regulates immune responses and disease pathogenesis. Nat. Immunol. 2009, 10, 241–247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Duewell, P.; Kono, H.; Rayner, K.J.; Sirois, C.M.; Vladimer, G.; Bauernfeind, F.G.; Abela, G.S.; Franchi, L.; Nuñez, G.; Schnurr, M.; et al. NLRP3 inflammasomes are required for atherogenesis and activated by cholesterol crystals. Nature 2010, 464, 1357–1361. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Youm, Y.-H.; Nguyen, K.Y.; Grant, R.W.; Goldberg, E.L.; Bodogai, M.; Kim, D.; D’Agostino, D.; Planavsky, N.; Lupfer, C.; Kanneganti, T.-D.; et al. The ketone metabolite β-hydroxybutyrate blocks NLRP3 inflammasome–mediated inflammatory disease. Nat. Med. 2015, 21, 263–269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Klement, R.J.; Meyer, D.; Kanzler, S.; Sweeney, R.A. Ketogenic diets consumed during radio-chemotherapy have beneficial effects on quality of life and metabolic health in patients with rectal cancer. Eur. J. Nutr. 2021, 61, 69–84. [Google Scholar] [CrossRef]
- Sukkar, S.G.; Bassetti, M. Induction of ketosis as a potential therapeutic option to limit hyperglycemia and prevent cytokine storm in COVID-19. Nutrition 2020, 79-80, 110967. [Google Scholar] [CrossRef]
- Crujeiras, A.B.; Izquierdo, A.G.; Primo, D.; Milagro, F.I.; Sajoux, I.; Jácome, A.; Fernandez-Quintela, A.; Portillo, M.P.; Martínez, J.; Martinez-Olmos, M.A.; et al. Epigenetic landscape in blood leukocytes following ketosis and weight loss induced by a very low calorie ketogenic diet (VLCKD) in patients with obesity. Clin. Nutr. 2021, 40, 3959–3972. [Google Scholar] [CrossRef]
- Schreck, K.C.; Lwin, M.; Strowd, R.E.; Henry-Barron, B.J.; Blakeley, J.O.; Cervenka, M.C. Effect of ketogenic diets on leukocyte counts in patients with epilepsy. Nutr. Neurosci. 2017, 22, 522–527. [Google Scholar] [CrossRef]
- Wilhelm, C.; Surendar, J.; Karagiannis, F. Enemy or ally? Fasting as an essential regulator of immune responses. Trends Immunol. 2021, 42, 389–400. [Google Scholar] [CrossRef] [PubMed]
- Beghi, E.; Giussani, G.; Nichols, E.; Abd-Allah, F.; Abdela, J.; Abdelalim, A.; Abraha, H.N.; Adib, M.G.; Agrawal, S.; Alahdab, F.; et al. Global, regional, and national burden of epilepsy, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Neurol. 2019, 18, 357–375. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Leeman, B.; Cole, A. Advancements in the Treatment of Epilepsy. Annu. Rev. Med. 2008, 59, 503–523. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Scher, M.S.; Kidder, B.M.; Bangert, B.A. Pediatric Epilepsy Evaluations From the Prenatal Perspective. J. Child Neurol. 2007, 22, 396–401. [Google Scholar] [CrossRef] [PubMed]
- Razaz, N.; Tomson, T.; Wikström, A.-K.; Cnattingius, S. Association Between Pregnancy and Perinatal Outcomes Among Women With Epilepsy. JAMA Neurol. 2017, 74, 983–991. [Google Scholar] [CrossRef] [PubMed]
- Perucca, P.; Bahlo, M.; Berkovic, S.F. The Genetics of Epilepsy. Annu. Rev. Genom. Hum. Genet. 2020, 21, 205–230. [Google Scholar] [CrossRef] [PubMed]
- Leu, C.; Balestrini, S.; Maher, B.; Hernández-Hernández, L.; Gormley, P.; Hämäläinen, E.; Heggeli, K.; Schoeler, N.; Novy, J.; Willis, J.; et al. Genome-wide Polygenic Burden of Rare Deleterious Variants in Sudden Unexpected Death in Epilepsy. eBioMedicine 2015, 2, 1063–1070. [Google Scholar] [CrossRef] [Green Version]
- Guerrini, R.; Balestrini, S.; Wirrell, E.C.; Walker, M.C. Monogenic Epilepsies. Neurology 2021, 97, 817–831. [Google Scholar] [CrossRef]
- Wang, J.; Lin, Z.-J.; Liu, L.; Xu, H.-Q.; Shi, Y.-W.; Yi, Y.-H.; He, N.; Liao, W.-P. Epilepsy-associated genes. Seizure 2016, 44, 11–20. [Google Scholar] [CrossRef] [Green Version]
- Koh, S.; Dupuis, N.; Auvin, S. Ketogenic diet and Neuroinflammation. Epilepsy Res. 2020, 167, 106454. [Google Scholar] [CrossRef]
- Panhans, C.M.; Gresham, G.; Amaral, L.J.; Hu, J. Exploring the Feasibility and Effects of a Ketogenic Diet in Patients With CNS Malignancies: A Retrospective Case Series. Front. Neurosci. 2020, 14, 390. [Google Scholar] [CrossRef] [PubMed]
- Lorenzo, P.M.; Sajoux, I.; Izquierdo, A.G.; Gomez-Arbelaez, D.; Zulet, M.A.; Abete, I.; Castro, A.I.; Baltar, J.; Portillo, M.P.; Tinahones, F.J.; et al. Immunomodulatory effect of a very-low-calorie ketogenic diet compared with bariatric surgery and a low-calorie diet in patients with excessive body weight. Clin. Nutr. 2022, 41, 1566–1577. [Google Scholar] [CrossRef] [PubMed]
- Okawa, T.; Nagai, M.; Hase, K. Dietary Intervention Impacts Immune Cell Functions and Dynamics by Inducing Metabolic Rewiring. Front. Immunol. 2021, 11. [Google Scholar] [CrossRef] [PubMed]
- Brunner, B.; Ari, C.; D’Agostino, D.P.; Kovács, Z. Adenosine Receptors Modulate the Exogenous Ketogenic Supplement-Evoked Alleviating Effect on Lipopolysaccharide-Generated Increase in Absence Epileptic Activity in WAG/Rij Rats. Nutrients 2021, 13, 4082. [Google Scholar] [CrossRef] [PubMed]
- Chen, F.; He, X.; Luan, G.; Li, T. Role of DNA Methylation and Adenosine in Ketogenic Diet for Pharmacoresistant Epilepsy: Focus on Epileptogenesis and Associated Comorbidities. Front. Neurol. 2019, 10. [Google Scholar] [CrossRef] [PubMed]
- Kovacs, Z.; Brunner, B.; D’Agostino, D.P.; Ari, C. Inhibition of adenosine A1 receptors abolished the nutritional ketosis-evoked delay in the onset of isoflurane-induced anesthesia in Wistar Albino Glaxo Rijswijk rats. BMC Anesthesiol. 2020, 20, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Masino, S.A.; Rho, J.M. Mechanisms of Ketogenic Diet Action. In Jasper’s Basic Mechanisms of the Epilepsies; National Center for Biotechnology Information (US): Bethesda, MD, USA, 2012. [Google Scholar]
- Watanabe, M.; Tozzi, R.; Risi, R.; Tuccinardi, D.; Mariani, S.; Basciani, S.; Spera, G.; Lubrano, C.; Gnessi, L. Beneficial effects of the ketogenic diet on nonalcoholic fatty liver disease: A comprehensive review of the literature. Obes. Rev. 2020, 21, e13024. [Google Scholar] [CrossRef] [Green Version]
- Żarnowska, I.; Wróbel-Dudzińska, D.; Tulidowicz-Bielak, M.; Kocki, T.; Mitosek-Szewczyk, K.; Gasior, M.; Turski, W.A. Changes in tryptophan and kynurenine pathway metabolites in the blood of children treated with ketogenic diet for refractory epilepsy. Seizure 2019, 69, 265–272. [Google Scholar] [CrossRef]
- Hirschberger, S.; Strauß, G.; Effinger, D.; Marstaller, X.; Ferstl, A.; Müller, M.B.; Wu, T.; Hübner, M.; Rahmel, T.; Mascolo, H.; et al. Very-low-carbohydrate diet enhances human T-cell immunity through immunometabolic reprogramming. EMBO Mol. Med. 2021, 13, e14323. [Google Scholar] [CrossRef]
- Dressler, A.; Benninger, F.; Trimmel-Schwahofer, P.; Gröppel, G.; Porsche, B.; Abraham, K.; Mühlebner, A.; Samueli, S.; Male, C.; Feucht, M. Efficacy and tolerability of the ketogenic diet versus high-dose adrenocorticotropic hormone for infantile spasms: A single-center parallel-cohort randomized controlled trial. Epilepsia 2019, 60, 441–451. [Google Scholar] [CrossRef]
- Olmi, S.; Petkoski, S.; Guye, M.; Bartolomei, F.; Jirsa, V. Controlling seizure propagation in large-scale brain networks. PLoS Comput. Biol. 2019, 15, e1006805. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ruskin, D.N.; Murphy, M.I.; Slade, S.L.; Masino, S.A. Ketogenic diet improves behaviors in a maternal immune activation model of autism spectrum disorder. PLOS ONE 2017, 12, e0171643. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Asadi-Pooya, A.A.; Katibeh, P. Lennox-Gastaut Syndrome. In Comprehensive Pharmacology; Elsevier: Amsterdam, The Netherlands, 2021; pp. 105–122. [Google Scholar] [CrossRef]
- Gokten, E.S.; Duman, N. West syndrome and autism: A case report. Eur. Res. J. 2015, 1, 154–156. [Google Scholar] [CrossRef] [Green Version]
- De Alba, G.O.-G.; Valdez, J.M.; Crespo, F.V. West Syndrome Evolving into the Lennox-Gastaut Syndrome. Clin. Electroencephalogr. 1984, 15, 61–68. [Google Scholar] [CrossRef]
- Calvo, A.; Buompadre, M.C.; Gallo, A.; Gutiérrez, R.; Valenzuela, G.R.; Caraballo, R. Electroclinical pattern in the transition from West to Lennox-Gastaut syndrome. Epilepsy Res. 2020, 167, 106446. [Google Scholar] [CrossRef]
- Amador, J.B.; García-Segovia, R.; Boyano, M.G.; Miño-León, G. West Syndrome in Children With Congenital Zika Virus Infection. Pediatr. Infect. Dis. J. 2021, 40, 1108–1110. [Google Scholar] [CrossRef]
- Pavone, P.; Polizzi, A.; Marino, S.D.; Corsello, G.; Falsaperla, R.; Marino, S.; Ruggieri, M. West syndrome: A comprehensive review. Neurol. Sci. 2020, 41, 3547–3562. [Google Scholar] [CrossRef]
- Kramer, U.; Chi, C.-S.; Lin, K.-L.; Specchio, N.; Sahin, M.; Olson, H.; Nabbout, R.; Kluger, G.; Lin, J.-J.; Van Baalen, A. Febrile infection-related epilepsy syndrome (FIRES): Pathogenesis, treatment, and outcome. Epilepsia 2011, 52, 1956–1965. [Google Scholar] [CrossRef]
- Peng, P.; Peng, J.; Yin, F.; Deng, X.; Chen, C.; He, F.; Wang, X.; Guang, S.; Mao, L. Ketogenic Diet as a Treatment for Super-Refractory Status Epilepticus in Febrile Infection-Related Epilepsy Syndrome. Front. Neurol. 2019, 10, 423. [Google Scholar] [CrossRef] [Green Version]
- Obara, K.; Ono, T. Ketogenic Diet for a Young Adult Patient With Chronic-Phase Febrile Infection-Related Epilepsy Syndrome. Cureus 2022, 14, e22099. [Google Scholar] [CrossRef] [PubMed]
- Li, W.-J.; Xue, C.-L.; Zhang, Y.; Wu, L.-H.; Chen, D.-M.; Chen, F.; Xu, J.; Li, Z.; Miao, H.-J. Ketogenic diet (KD) therapy in the acute phase of febrile infection-related epilepsy syndrome (FIRES): A case report. Transl. Pediatr. 2021, 10, 2392–2397. [Google Scholar] [CrossRef] [PubMed]
- Mehta, V.; Ferrie, C.D.; Cross, H.; Vadlamani, G. Corticosteroids including ACTH for childhood epilepsy other than epileptic spasms. Cochrane Database Syst. Rev. 2015, 2015, CD005222. [Google Scholar] [CrossRef]
- A Kelley, S.; Kossoff, E.H. Doose syndrome (myoclonic-astatic epilepsy): 40 years of progress. Dev. Med. Child Neurol. 2010, 52, 988–993. [Google Scholar] [CrossRef] [PubMed]
- Storoni, M.; Plant, G.T. The Therapeutic Potential of the Ketogenic Diet in Treating Progressive Multiple Sclerosis. Mult. Scler. Int. 2015, 2015, 681289. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kim, D.Y.; Hao, J.; Liu, R.; Turner, G.; Shi, F.-D.; Rho, J.M. Inflammation-Mediated Memory Dysfunction and Effects of a Ketogenic Diet in a Murine Model of Multiple Sclerosis. PLOS ONE 2012, 7, e35476. [Google Scholar] [CrossRef] [Green Version]
- Valburg, C.; Sonti, A.; Stern, J.N.; Najjar, S.; Harel, A. Dietary factors in experimental autoimmune encephalomyelitis and multiple sclerosis: A comprehensive review. Mult. Scler. J. 2020, 27, 494–502. [Google Scholar] [CrossRef]
- Bock, M.; Karber, M.; Kuhn, H. Ketogenic diets attenuate cyclooxygenase and lipoxygenase gene expression in multiple sclerosis. eBioMedicine 2018, 36, 293–303. [Google Scholar] [CrossRef]
- Düking, T.; Spieth, L.; Berghoff, S.A.; Piepkorn, L.; Schmidke, A.M.; Mitkovski, M.; Kannaiyan, N.; Hosang, L.; Scholz, P.; Shaib, A.H.; et al. Ketogenic Diet Uncovers Differential Metabolic Plasticity of Brain Cells. Science 2022, 8, eabo7639. Available online: https://www.science.org (accessed on 28 November 2022). [CrossRef]
- Lin, W.-S.; Lin, S.-J.; Liao, P.-Y.; Suresh, D.; Hsu, T.-R.; Wang, P.-Y. Role of Ketogenic Diets in Multiple Sclerosis and Related Animal Models: An Updated Review. Adv. Nutr. Int. Rev. J. 2022, 13, 2002–2014. [Google Scholar] [CrossRef]
- Piacente, F.; Bottero, M.; Benzi, A.; Vigo, T.; Uccelli, A.; Bruzzone, S.; Ferrara, G. Neuroprotective Potential of Dendritic Cells and Sirtuins in Multiple Sclerosis. Int. J. Mol. Sci. 2022, 23, 4352. [Google Scholar] [CrossRef]
- Göschl, L.; Preglej, T.; Hamminger, P.; Bonelli, M.; Andersen, L.; Boucheron, N.; Gülich, A.F.; Müller, L.; Saferding, V.; Mufazalov, I.A.; et al. A T cell-specific deletion of HDAC1 protects against experimental autoimmune encephalomyelitis. J. Autoimmun. 2017, 86, 51–61. [Google Scholar] [CrossRef] [PubMed]
- Martin, A.; Tegla, C.A.; Cudrici, C.D.; Kruszewski, A.M.; Azimzadeh, P.; Boodhoo, D.; Mekala, A.P.; Rus, V.; Rus, H. Role of SIRT1 in autoimmune demyelination and neurodegeneration. Immunol. Res. 2014, 61, 187–197. [Google Scholar] [CrossRef] [PubMed]
- Liu, C.; Zhang, N.; Zhang, R.; Jin, L.; Petridis, A.K.; Loers, G.; Zheng, X.; Wang, Z.; Siebert, H.-C. Cuprizone-Induced Demyelination in Mouse Hippocampus Is Alleviated by Ketogenic Diet. J. Agric. Food Chem. 2020, 68. [Google Scholar] [CrossRef] [PubMed]
- Wouters, E.; Grajchen, E.; Jorissen, W.; Dierckx, T.; Wetzels, S.; Loix, M.; Tulleners, M.P.; Staels, B.; Stinissen, P.; Haidar, M.; et al. Altered PPARγ Expression Promotes Myelin-Induced Foam Cell Formation in Macrophages in Multiple Sclerosis. Int. J. Mol. Sci. 2020, 21, 9329. [Google Scholar] [CrossRef] [PubMed]
- Zhang, K.; Guo, Y.; Ge, Z.; Zhang, Z.; Da, Y.; Li, W.; Zhang, Z.; Xue, Z.; Li, Y.; Ren, Y.; et al. Adiponectin Suppresses T Helper 17 Cell Differentiation and Limits Autoimmune CNS Inflammation via the SIRT1/PPARγ/RORγt Pathway. Mol. Neurobiol. 2016, 54, 4908–4920. [Google Scholar] [CrossRef]
- Ai, R.-S.; Xing, K.; Deng, X.; Han, J.-J.; Hao, D.-X.; Qi, W.-H.; Han, B.; Yang, Y.-N.; Li, X.; Zhang, Y. Baicalin Promotes CNS Remyelination via PPARγ Signal Pathway. Neurol.-Neuroimmunol. Neuroinflammation 2022, 9, e1142. [Google Scholar] [CrossRef]
- Kanakasabai, S.; Pestereva, E.; Chearwae, W.; Gupta, S.K.; Ansari, S.; Bright, J.J. PPARγ Agonists Promote Oligodendrocyte Differentiation of Neural Stem Cells by Modulating Stemness and Differentiation Genes. PLoS ONE 2012, 7, e50500. [Google Scholar] [CrossRef]
- Brenton, J.N.; Banwell, B.; Bergqvist, A.C.; Lehner-Gulotta, D.; Gampper, L.; Leytham, E.; Coleman, R.; Goldman, M.D. Pilot study of a ketogenic diet in relapsing-remitting MS. Neurol.-Neuroimmunol. Neuroinflammation 2019, 6, e565. [Google Scholar] [CrossRef] [Green Version]
- Bahr, L.S.; Bock, M.; Liebscher, D.; Bellmann-Strobl, J.; Franz, L.; Prüß, A.; Schumann, D.; Piper, S.K.; Kessler, C.S.; Steckhan, N.; et al. Ketogenic diet and fasting diet as Nutritional Approaches in Multiple Sclerosis (NAMS): Protocol of a randomized controlled study. Trials 2020, 21, 1–9. [Google Scholar] [CrossRef] [Green Version]
- Alizadeh, A.; Dyck, S.M.; Karimi-Abdolrezaee, S. Traumatic spinal cord injury: An overview of pathophysiology, models and acute injury mechanisms. Front. Neurol. 2019, 10, 282. [Google Scholar] [CrossRef] [Green Version]
- Streijger, F.; Plunet, W.T.; Lee, J.H.T.; Liu, J.; Lam, C.K.; Park, S.; Hilton, B.; Fransen, B.L.; Matheson, K.A.J.; Assinck, P.; et al. Ketogenic Diet Improves Forelimb Motor Function after Spinal Cord Injury in Rodents. PLoS ONE 2013, 8, e78765. [Google Scholar] [CrossRef] [PubMed]
- Seira, O.; Kolehmainen, K.; Liu, J.; Streijger, F.; Haegert, A.; Lebihan, S.; Boushel, R.; Tetzlaff, W. Ketogenesis controls mitochondrial gene expression and rescues mitochondrial bioenergetics after cervical spinal cord injury in rats. Sci. Rep. 2021, 11, 1–19. [Google Scholar] [CrossRef] [PubMed]
- Liu, T.; Zhang, L.; Joo, D.; Sun, S.-C. NF-κB signaling in inflammation. Signal Transduct. Target. Ther. 2017, 2, 17023. [Google Scholar] [CrossRef] [Green Version]
- Kracht, M.; Müller-Ladner, U.; Schmitz, M.L. Mutual regulation of metabolic processes and proinflammatory NF-κB signaling. J. Allergy Clin. Immunol. 2020, 146, 694–705. [Google Scholar] [CrossRef] [PubMed]
- Roberti, A.; Chaffey, L.E.; Greaves, D.R. NF-κB Signaling and Inflammation—Drug Repurposing to Treat Inflammatory Disorders? Biology 2022, 11, 372. [Google Scholar] [CrossRef]
- Lawrence, T. The Nuclear Factor NF-kappa B Pathway in Inflammation. Cold Spring Harb. Perspect. Biol. 2009, 1, a001651. [Google Scholar] [CrossRef] [Green Version]
- Poewe, W.; Seppi, K.; Tanner, C.M.; Halliday, G.M.; Brundin, P.; Volkmann, J.; Schrag, A.E.; Lang, A.E. Parkinson disease. Nat. Rev. Dis. Primers 2017, 3, 17013. [Google Scholar] [CrossRef]
- Bloem, B.R.; Okun, M.S.; Klein, C. Parkinson’s disease. Lancet 2021, 397, 2284–2303. [Google Scholar] [CrossRef]
- Jankovic, J. Parkinson’s disease: Clinical features and diagnosis. J. Neurol. Neurosurg. Psychiatry 2008, 79, 368–376. [Google Scholar] [CrossRef] [Green Version]
- Mullin, S.; Stokholm, M.G.; Hughes, D.; Mehta, A.; Parbo, P.; Hinz, R.; Pavese, N.; Brooks, D.J.; Schapira, A.H. Brain Microglial Activation Increased in Glucocerebrosidase ( GBA ) Mutation Carriers without Parkinson’s disease. Mov. Disord. 2020, 36, 774–779. [Google Scholar] [CrossRef]
- Ferreira, S.A.; Romero-Ramos, M. Microglia Response During Parkinson’s Disease: Alpha-Synuclein Intervention. Front. Cell. Neurosci. 2018, 12, 247. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Armstrong, M.J.; Okun, M.S. Diagnosis and Treatment of Parkinson Disease. JAMA 2020, 323, 548–560. [Google Scholar] [CrossRef] [PubMed]
- Sveinbjornsdottir, S. The clinical symptoms of Parkinson’s disease. J. Neurochem. 2016, 139, 318–324. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, X.; Cheng, B. Neuroprotective and Anti-inflammatory Activities of Ketogenic Diet on MPTP-induced Neurotoxicity. J. Mol. Neurosci. 2010, 42, 145–153. [Google Scholar] [CrossRef] [PubMed]
- George, S.; Rey, N.L.; Tyson, T.; Esquibel, C.; Meyerdirk, L.; Schulz, E.; Pierce, S.; Burmeister, A.R.; Madaj, Z.; Steiner, J.A.; et al. Microglia affect α-synuclein cell-to-cell transfer in a mouse model of Parkinson’s disease. Mol. Neurodegener. 2019, 14, 1–22. [Google Scholar] [CrossRef]
- Ingelfinger, F.; Gerdes, L.A.; Kavaka, V.; Krishnarajah, S.; Friebel, E.; Galli, E.; Zwicky, P.; Furrer, R.; Peukert, C.; Dutertre, C.-A.; et al. Twin study reveals non-heritable immune perturbations in multiple sclerosis. Nature 2022, 603, 152–158. [Google Scholar] [CrossRef]
- Badanjak, K.; Fixemer, S.; Smajić, S.; Skupin, A.; Grünewald, A. The Contribution of Microglia to Neuroinflammation in Parkinson’s Disease. Int. J. Mol. Sci. 2021, 22, 4676. [Google Scholar] [CrossRef]
- Ma, Z.; Zhang, D.; Li, J. A dedicated collaboration platform for integrated project delivery. Autom. Constr. 2018, 86, 199–209. [Google Scholar] [CrossRef]
- Olson, C.A.; Vuong, H.E.; Yano, J.M.; Liang, Q.Y.; Nusbaum, D.J.; Hsiao, E.Y. The Gut Microbiota Mediates the Anti-Seizure Effects of the Ketogenic Diet. Cell 2018, 173, 1728–1741.e13. [Google Scholar] [CrossRef] [Green Version]
- Sampson, T.R.; Challis, C.; Jain, N.; Moiseyenko, A.; Ladinsky, M.S.; Shastri, G.G.; Thron, T.; Needham, B.D.; Horvath, I.; Debelius, J.W.; et al. A gut bacterial amyloid promotes α-synuclein aggregation and motor impairment in mice. eLife 2020, 9, e53111. [Google Scholar] [CrossRef]
- Kam, T.-I.; Hinkle, J.T.; Dawson, T.M.; Dawson, V.L. Microglia and astrocyte dysfunction in parkinson’s disease. Neurobiol. Dis. 2020, 144, 105028. [Google Scholar] [CrossRef] [PubMed]
- Batch, J.T.; Lamsal, S.P.; Adkins, M.; Sultan, S.; Ramirez, M.N. Advantages and Disadvantages of the Ketogenic Diet: A Review Article. Cureus 2020, 12, e9639. [Google Scholar] [CrossRef] [PubMed]
- Crosby, L.; Davis, B.; Joshi, S.; Jardine, M.; Paul, J.; Neola, M.; Barnard, N.D. Ketogenic Diets and Chronic Disease: Weighing the Benefits Against the Risks. Front. Nutr. 2021, 8. [Google Scholar] [CrossRef] [PubMed]
- Newmaster, K.; Zhu, Z.; Bolt, E.; Chang, R.J.; Day, C.; Mhanna, A.; Paudel, S.; Farooq, O.; Swaminathan, A.; Acharya, P.; et al. A Review of the Multi-Systemic Complications of a Ketogenic Diet in Children and Infants with Epilepsy. Children 2022, 9, 1372. [Google Scholar] [CrossRef]
- Valencia, I.; Pfeifer, H.; Thiele, E.A. General Anesthesia and the Ketogenic Diet: Clinical Experience in Nine Patients. Epilepsia 2002, 43, 525–529. [Google Scholar] [CrossRef]
- Breu, M.; Häfele, C.; Glatter, S.; Trimmel-Schwahofer, P.; Golej, J.; Male, C.; Feucht, M.; Dressler, A. Ketogenic Diet in the Treatment of Super-Refractory Status Epilepticus at a Pediatric Intensive Care Unit: A Single-Center Experience. Front. Neurol. 2021, 12. [Google Scholar] [CrossRef]
- Sondhi, V.; Agarwala, A.; Pandey, R.M.; Chakrabarty, B.; Jauhari, P.; Lodha, R.; Toteja, G.S.; Sharma, S.; Paul, V.K.; Kossoff, E.; et al. Efficacy of Ketogenic Diet, Modified Atkins Diet, and Low Glycemic Index Therapy Diet Among Children With Drug-Resistant Epilepsy. JAMA Pediatr. 2020, 174, 944. [Google Scholar] [CrossRef]
- Alten, E.; Chaturvedi, A.; Cullimore, M.; Fallon, A.; Habben, L.; Hughes, I.; O’Malley, N.; Rahimi, H.; Renodin-Mead, D.; Schmidt, B.; et al. No longer a historical ailment: Two cases of childhood scurvy with recommendations for bone health providers. Osteoporos. Int. 2020, 31, 1001–1005. [Google Scholar] [CrossRef]
- Willmott, N.S.; Bryan, R.A.E. Scurvy in child with epilepsy on a ketogenic diet with oral complications. Eur. Arch. Paediatr. Dent. 2008, 9, 148–152. [Google Scholar] [CrossRef]
- Kidwell, R.; Tobias, J.D. Anesthetic care of a child with receiving a ketogenic diet. Compr. Pharmacol. 2021, 9, 85–93. [Google Scholar] [CrossRef]
- Grinspan, Z.M.; Shellhaas, R.A.; Coryell, J.; Sullivan, J.E.; Wirrell, E.C.; Mytinger, J.R.; Gaillard, W.D.; Kossoff, E.H.; Valencia, I.; Knupp, K.G.; et al. Comparative Effectiveness of Levetiracetam vs Phenobarbital for Infantile Epilepsy. JAMA Pediatr. 2018, 172, 352–360. [Google Scholar] [CrossRef] [PubMed]
- Erickson, J.C.; Jabbari, B.; DiFazio, M.P. Basal ganglia injury as a complication of the ketogenic diet. Mov. Disord. 2003, 18, 448–451. [Google Scholar] [CrossRef] [PubMed]
- Desai, A.A.; Thompson, L.M.; Abdelmoity, A.T.; Kayyali, H.; Peter, S.D.S. Management of Symptomatic Cholelithiasis While on Ketogenic Diet: A Case Report. Pediatr. Neurol. 2014, 51, 439–440. [Google Scholar] [CrossRef] [PubMed]
- Kapetanakis, M.; Liuba, P.; Odermarsky, M.; Lundgren, J.; Hallböök, T. Effects of ketogenic diet on vascular function. Eur. J. Paediatr. Neurol. 2014, 18, 489–494. [Google Scholar] [CrossRef] [PubMed]
| Type of Ketogenic Diet (KD) | Mechanism of Action | Conclusions/Effects | References |
|---|---|---|---|
| Medium-Chain Triglycerides (MCT) | Reduced Leukocyte Count | Anti-Tumor Effect | [79] |
| Eucaloric Ketogenic Diet (EKD) | M1 Recruitment of Neutrophil and Platelets in Thrombo-Inflammation | Prevention of Cytokine Storm in COVID-19. | [80] |
| Very Low Calorie Ketogenic Diet (VLCKD) | Altered Leukocyte Methylation | Obesity Prevention | [81] |
| KD | Reduced WBC & Neutrophil Count | In Epilepsy, baseline immunosuppression does not worsen with KD. | [82] |
| KD | Enhanced activity of Th1 cells | Promoted cellular immune function in a CT26 colon tumor allografts mouse model. | [33] |
| KD | Suppression of Th1 & Th17 | Protection against autoinflammation (Central Nervous System). | [83] |
| KD | Suppression of macrophages & neutrophils | Attenuation of autoinflammation (Muckle-Wells syndrome/ Gout) | [83] |
| KD | Activation of protective γδ T cells and decreases myeloid cell subset | Ketogenesis-induced protection from mCoV-A59-driven inflammatory damage in aging. | [17] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Srivastava, S.; Pawar, V.A.; Tyagi, A.; Sharma, K.P.; Kumar, V.; Shukla, S.K. Immune Modulatory Effects of Ketogenic Diet in Different Disease Conditions. Immuno 2023, 3, 1-15. https://doi.org/10.3390/immuno3010001
Srivastava S, Pawar VA, Tyagi A, Sharma KP, Kumar V, Shukla SK. Immune Modulatory Effects of Ketogenic Diet in Different Disease Conditions. Immuno. 2023; 3(1):1-15. https://doi.org/10.3390/immuno3010001
Chicago/Turabian StyleSrivastava, Shivani, Vishakha Anand Pawar, Anuradha Tyagi, Kanti Prakash Sharma, Vinay Kumar, and Surendra Kumar Shukla. 2023. "Immune Modulatory Effects of Ketogenic Diet in Different Disease Conditions" Immuno 3, no. 1: 1-15. https://doi.org/10.3390/immuno3010001
APA StyleSrivastava, S., Pawar, V. A., Tyagi, A., Sharma, K. P., Kumar, V., & Shukla, S. K. (2023). Immune Modulatory Effects of Ketogenic Diet in Different Disease Conditions. Immuno, 3(1), 1-15. https://doi.org/10.3390/immuno3010001







