Clostridium perfringens-Induced Necrotic Diseases: An Overview
Abstract
1. Overview of C. perfringens Produced Toxins and Enzymes
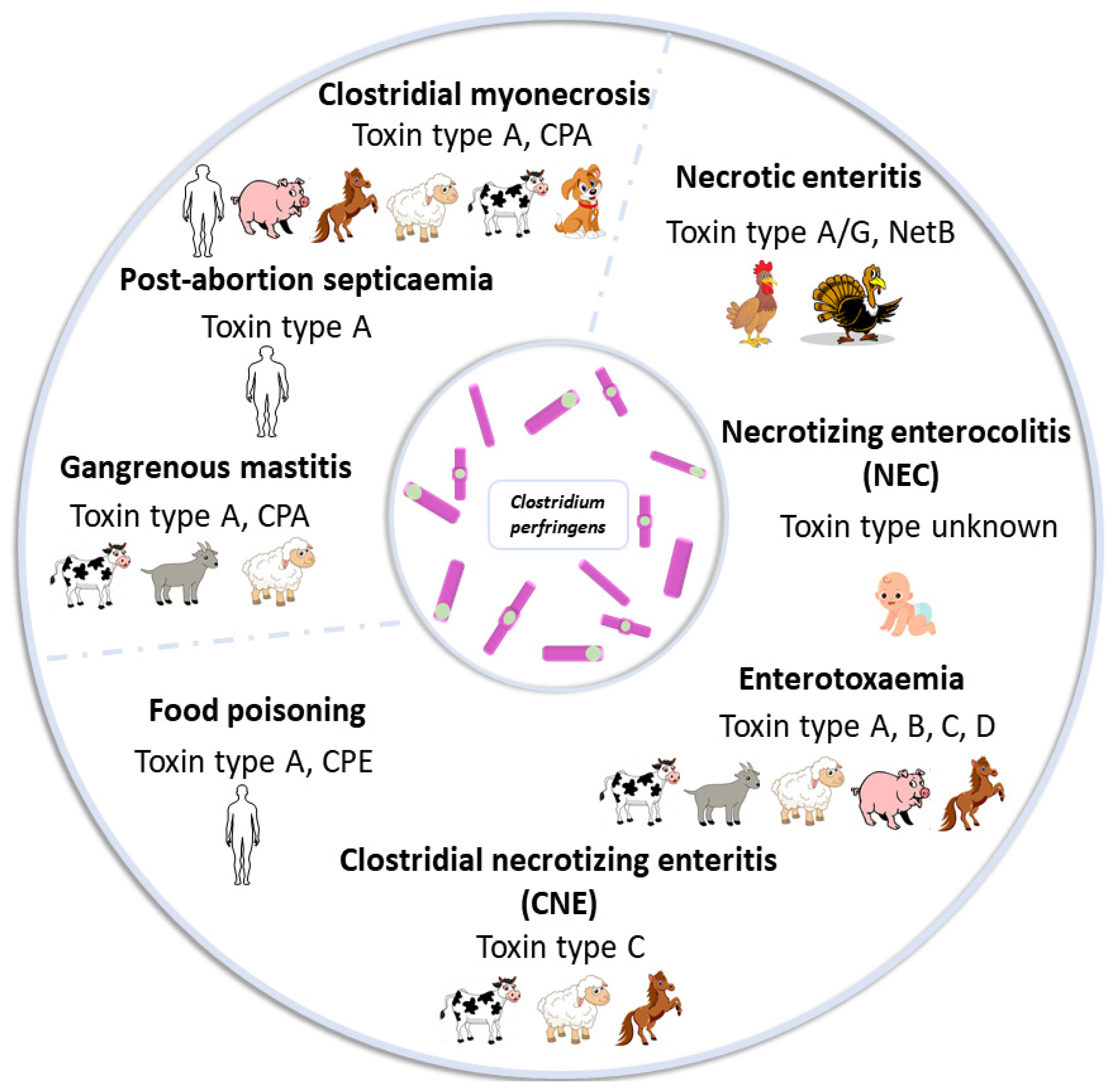
2. Overview of C. perfringens-Induced Diseases
2.1. C. perfringens-Induced Necrotic Diseases in Wounded Soft Tissue
2.2. C. perfringens-Induced Enteritis with Tissue Necrosis
3. C. perfringens-Induced Enteritis, Immunity, and Microbiome
3.1. The Interaction of C. perfringens and Immunity in Myonecrosis
3.2. The Interaction of C. perfringens, Immunity, Microbiota, and Its Metabolites in Enteritis
3.3. Ischemia in C. perfringens-Induced Diseases
4. Prevention and Treatment of C. perfringens-Induced Necrotic Diseases
4.1. Preventing C. perfringens-Induced Diseases with Vaccines
4.2. Treating C. perfringens-Induced Diseases
4.3. Potential New Interventions against C. perfringens-Induced Necrotic Diseases
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Data Availability Statement
Conflicts of Interest
References
- CDC. Prevent Illness from C. perfringens; CDC: Atlanta, GA, USA, 2021.
- Rood, J.I.; Keyburn, A.L.; Moore, R.J. NetB and necrotic enteritis: The hole movable story. Avian Pathol. 2016, 45, 295–301. [Google Scholar] [CrossRef]
- Navarro, M.A.; McClane, B.A.; Uzal, F.A. Mechanisms of Action and Cell Death Associated with Clostridium perfringens Toxins. Toxins 2018, 10, 212. [Google Scholar] [CrossRef]
- Rood, J.I.; Adams, V.; Lacey, J.; Lyras, D.; McClane, B.A.; Melville, S.B.; Moore, R.J.; Popoff, M.R.; Sarker, M.R.; Songer, J.G.; et al. Expansion of the Clostridium perfringens toxin-based typing scheme. Anaerobe 2018, 53, 5–10. [Google Scholar] [CrossRef]
- Fernandes da Costa, S.P.; Mot, D.; Bokori-Brown, M.; Savva, C.G.; Basak, A.K.; Van Immerseel, F.; Titball, R.W. Protection against avian necrotic enteritis after immunisation with NetB genetic or formaldehyde toxoids. Vaccine 2013, 31, 4003–4008. [Google Scholar] [CrossRef]
- Fernandez-Miyakawa, M.E.; Marcellino, R.; Uzal, F.A. Clostridium perfringens type A toxin production in 3 commonly used culture media. J. Vet. Diagn. Investig. 2007, 19, 184–186. [Google Scholar] [CrossRef]
- Duncan, C.L.; Strong, D.H. Improved medium for sporulation of Clostridium perfringens. Appl. Microbiol. 1968, 16, 82–89. [Google Scholar] [CrossRef]
- Willardsen, R.R.; Busta, F.F.; Allen, C.E. Growth of Clostridium perfringens in Three Different Beef Media and Fluid Thioglycollate Medium at Static and Constantly Rising Temperatures (1). J. Food Prot. 1979, 42, 144–148. [Google Scholar] [CrossRef]
- Wang, H.; Latorre, J.D.; Bansal, M.; Abraha, M.; Al-Rubaye, B.; Tellez-Isaias, G.; Hargis, B.; Sun, X. Microbial metabolite deoxycholic acid controls Clostridium perfringens-induced chicken necrotic enteritis through attenuating inflammatory cyclooxygenase signaling. Sci. Rep. 2019, 9, 14541. [Google Scholar] [CrossRef]
- Hauschild, A.H.; Hilsheimer, R.; Griffith, D.W. Enumeration of fecal Clostridium perfringens spores in egg yolk-free tryptose-sulfite-cycloserine agar. Appl. Microbiol. 1974, 27, 527–530. [Google Scholar] [CrossRef]
- Goldstein, M.R.; Kruth, S.A.; Bersenas, A.M.; Holowaychuk, M.K.; Weese, J.S. Detection and characterization of Clostridium perfringens in the feces of healthy and diarrheic dogs. Can. J. Vet. Res. 2012, 76, 161–165. [Google Scholar]
- Rhodehamel, E.J.; Harmon, S.M. BAM Chapter 16: Clostridium perfringens. In Bacteriological Analytical Manual, 8th ed.; Revision, A., Ed.; FDA: Silver Spring, MD, USA, 2001. [Google Scholar]
- BioNumbers. Comparison of Experimentally Obtained Generation Times of Ent+ and Ent− Grown in Autoclaved Beef with Those Predicted by the Gompertz Model (Shortest Known Cellular Doubling Time). Available online: https://bionumbers.hms.harvard.edu/bionumber.aspx?id=105474&ver=1 (accessed on 16 February 2022).
- Titball, R.W.; Naylor, C.E.; Basak, A.K. The Clostridium perfringens alpha-toxin. Anaerobe 1999, 5, 51–64. [Google Scholar] [CrossRef]
- Uzal, F.A.; Freedman, J.C.; Shrestha, A.; Theoret, J.R.; Garcia, J.; Awad, M.M.; Adams, V.; Moore, R.J.; Rood, J.I.; McClane, B.A. Towards an understanding of the role of Clostridium perfringens toxins in human and animal disease. Future Microbiol. 2014, 9, 361–377. [Google Scholar] [CrossRef]
- Marius, N.; Manoilescu, I.; Velnic, A.-A.; Ioan, B. Gas Gangrene: Case Presentation and Literature Data. J. Chir. 2017, 139–142. [Google Scholar] [CrossRef]
- Hickey, M.J.; Kwan, R.Y.; Awad, M.M.; Kennedy, C.L.; Young, L.F.; Hall, P.; Cordner, L.M.; Lyras, D.; Emmins, J.J.; Rood, J.I. Molecular and cellular basis of microvascular perfusion deficits induced by Clostridium perfringens and Clostridium septicum. PLoS Pathog. 2008, 4, e1000045. [Google Scholar] [CrossRef]
- Sayeed, S.; Li, J.; McClane, B.A. Characterization of virulence plasmid diversity among Clostridium perfringens type B isolates. Infect. Immun. 2010, 78, 495–504. [Google Scholar] [CrossRef]
- Gurjar, A.; Li, J.; McClane, B.A. Characterization of toxin plasmids in Clostridium perfringens type C isolates. Infect. Immun. 2010, 78, 4860–4869. [Google Scholar] [CrossRef]
- Oda, M.; Terao, Y.; Sakurai, J.; Nagahama, M. Membrane-Binding Mechanism of Clostridium perfringens Alpha-Toxin. Toxins 2015, 7, 5268–5275. [Google Scholar] [CrossRef]
- Sayeed, S.; Uzal, F.A.; Fisher, D.J.; Saputo, J.; Vidal, J.E.; Chen, Y.; Gupta, P.; Rood, J.I.; McClane, B.A. Beta toxin is essential for the intestinal virulence of Clostridium perfringens type C disease isolate CN3685 in a rabbit ileal loop model. Mol. Microbiol. 2008, 67, 15–30. [Google Scholar] [CrossRef]
- Uzal, F.A.; Saputo, J.; Sayeed, S.; Vidal, J.E.; Fisher, D.J.; Poon, R.; Adams, V.; Fernandez-Miyakawa, M.E.; Rood, J.I.; McClane, B.A. Development and application of new mouse models to study the pathogenesis of Clostridium perfringens type C Enterotoxemias. Infect. Immun. 2009, 77, 5291–5299. [Google Scholar] [CrossRef]
- Freedman, J.C.; McClane, B.A.; Uzal, F.A. New insights into Clostridium perfringens epsilon toxin activation and action on the brain during enterotoxemia. Anaerobe 2016, 41, 27–31. [Google Scholar] [CrossRef]
- Harkness, J.M.; Li, J.; McClane, B.A. Identification of a lambda toxin-negative Clostridium perfringens strain that processes and activates epsilon prototoxin intracellularly. Anaerobe 2012, 18, 546–552. [Google Scholar] [CrossRef]
- Minami, J.; Katayama, S.; Matsushita, O.; Matsushita, C.; Okabe, A. Lambda-toxin of Clostridium perfringens activates the precursor of epsilon-toxin by releasing its N- and C-terminal peptides. Microbiol. Immunol. 1997, 41, 527–535. [Google Scholar] [CrossRef]
- Popoff, M.R. Epsilon toxin: A fascinating pore-forming toxin. FEBS J. 2011, 278, 4602–4615. [Google Scholar] [CrossRef]
- Filho, E.J.; Carvalho, A.U.; Assis, R.A.; Lobato, F.F.; Rachid, M.A.; Carvalho, A.A.; Ferreira, P.M.; Nascimento, R.A.; Fernandes, A.A.; Vidal, J.E.; et al. Clinicopathologic features of experimental Clostridium perfringens type D enterotoxemia in cattle. Vet. Pathol. 2009, 46, 1213–1220. [Google Scholar] [CrossRef] [PubMed]
- Li, J.; Miyamoto, K.; McClane, B.A. Comparison of virulence plasmids among Clostridium perfringens type E isolates. Infect. Immun. 2007, 75, 1811–1819. [Google Scholar] [CrossRef]
- Nagahama, M.; Umezaki, M.; Oda, M.; Kobayashi, K.; Tone, S.; Suda, T.; Ishidoh, K.; Sakurai, J. Clostridium perfringens iota-toxin b induces rapid cell necrosis. Infect. Immun. 2011, 79, 4353–4360. [Google Scholar] [CrossRef] [PubMed]
- Czeczulin, J.R.; Hanna, P.C.; McClane, B.A. Cloning, nucleotide sequencing, and expression of the Clostridium perfringens enterotoxin gene in Escherichia coli. Infect. Immun. 1993, 61, 3429–3439. [Google Scholar] [CrossRef]
- McClane, B.A.; Robertson, S.L.; Li, J. Clostridium perfringens. In Food Microbiology; Wiley: Hoboken, NJ, USA, 2012; pp. 465–489. [Google Scholar]
- Abdel-Glil, M. In Silico Genome Analysis and Molecular Typing of Clostridium perfringens. Ph.D. Thesis, Freie Universität, Berlin, Germany, 2020. [Google Scholar]
- Xiao, Y. Clostridium perfringens Sporulation, Germination and Outgrowth in Food: A Functional Genomics Approach; Wageningen University and Research: Wageningen, The Netherlands, 2014. [Google Scholar]
- Miyamoto, K.; Yumine, N.; Mimura, K.; Nagahama, M.; Li, J.; McClane, B.A.; Akimoto, S. Identification of novel Clostridium perfringens type E strains that carry an iota toxin plasmid with a functional enterotoxin gene. PLoS ONE 2011, 6, e20376. [Google Scholar] [CrossRef]
- Billington, S.J.; Wieckowski, E.U.; Sarker, M.R.; Bueschel, D.; Songer, J.G.; McClane, B.A. Clostridium perfringens type E animal enteritis isolates with highly conserved, silent enterotoxin gene sequences. Infect. Immun. 1998, 66, 4531–4536. [Google Scholar] [CrossRef]
- Sarker, M.R.; Carman, R.J.; McClane, B.A. Inactivation of the gene (cpe) encoding Clostridium perfringens enterotoxin eliminates the ability of two cpe-positive C. perfringens type A human gastrointestinal disease isolates to affect rabbit ileal loops. Mol. Microbiol. 1999, 33, 946–958. [Google Scholar]
- Bannam, T.L.; Yan, X.X.; Harrison, P.F.; Seemann, T.; Keyburn, A.L.; Stubenrauch, C.; Weeramantri, L.H.; Cheung, J.K.; McClane, B.A.; Boyce, J.D.; et al. Necrotic enteritis-derived Clostridium perfringens strain with three closely related independently conjugative toxin and antibiotic resistance plasmids. mBio 2011, 2, e00190-11. [Google Scholar] [CrossRef] [PubMed]
- Lepp, D.; Roxas, B.; Parreira, V.R.; Marri, P.R.; Rosey, E.L.; Gong, J.; Songer, J.G.; Vedantam, G.; Prescott, J.F. Identification of novel pathogenicity loci in Clostridium perfringens strains that cause avian necrotic enteritis. PLoS ONE 2010, 5, e10795. [Google Scholar] [CrossRef]
- Keyburn, A.L.; Yan, X.X.; Bannam, T.L.; Van Immerseel, F.; Rood, J.I.; Moore, R.J. Association between avian necrotic enteritis and Clostridium perfringens strains expressing NetB toxin. Vet. Res. 2010, 41, 21. [Google Scholar] [CrossRef]
- Buboltz, J.B.; Murphy-Lavoie, H.M. Gas Gangrene. 3 February 2022. Available online: https://www.ncbi.nlm.nih.gov/books/NBK537030/ (accessed on 18 February 2022).
- Giri, B.; Kole, L. Combating the Insidious Enemy: Epidemiology, Pathophysiology, and Treatment of Clostridial Gas Gangrene. In Biological Toxins and Bioterrorism; Gopalakrishnakone, P., Balali-Mood, M., Llewellyn, L., Singh, B.R., Eds.; Springer: Dordrecht, The Netherlands, 2015; pp. 425–448. [Google Scholar]
- Giri, B.; Kole, L. Combating the Perilous Consequence of Clostridial Gas Gangrene: An Overview. In Toxinology: Biological Toxins and Bioterrorism; Gopalakrishnakone, P., Ed.; Springer: Dordrecht, The Netherlands, 2013; pp. 1–21. [Google Scholar]
- Goossens, E.; Valgaeren, B.R.; Pardon, B.; Haesebrouck, F.; Ducatelle, R.; Deprez, P.R.; Van Immerseel, F. Rethinking the role of alpha toxin in Clostridium perfringens-associated enteric diseases: A review on bovine necro-haemorrhagic enteritis. Vet. Res. 2017, 48, 9. [Google Scholar] [CrossRef] [PubMed]
- McDonel, J.L. Clostridium perfringens toxins (type A, B, C, D, E). Pharmacol. Ther. 1980, 10, 617–655. [Google Scholar] [CrossRef]
- Bansal, M.; Alenezi, T.; Fu, Y.; Almansour, A.; Wang, H.; Gupta, A.; Liyanage, R.; Graham, D.B.; Hargis, B.M.; Sun, X. Specific Secondary Bile Acids Control Chicken Necrotic Enteritis. Pathogens 2021, 10, 1041. [Google Scholar] [CrossRef]
- Nagpal, R.; Ogata, K.; Tsuji, H.; Matsuda, K.; Takahashi, T.; Nomoto, K.; Suzuki, Y.; Kawashima, K.; Nagata, S.; Yamashiro, Y. Sensitive quantification of Clostridium perfringens in human feces by quantitative real-time PCR targeting alpha-toxin and enterotoxin genes. BMC Microbiol. 2015, 15, 219. [Google Scholar] [CrossRef]
- Bryant, A.E.; Chen, R.Y.; Nagata, Y.; Wang, Y.; Lee, C.H.; Finegold, S.; Guth, P.H.; Stevens, D.L. Clostridial gas gangrene. II. Phospholipase C-induced activation of platelet gpIIbIIIa mediates vascular occlusion and myonecrosis in Clostridium perfringens gas gangrene. J. Infect. Dis. 2000, 182, 808–815. [Google Scholar]
- Timbermont, L.; Haesebrouck, F.; Ducatelle, R.; Van Immerseel, F. Necrotic enteritis in broilers: An updated review on the pathogenesis. Avian Pathol. 2011, 40, 341–347. [Google Scholar] [CrossRef]
- Stevens, D.L.; Troyer, B.E.; Merrick, D.T.; Mitten, J.E.; Olson, R.D. Lethal effects and cardiovascular effects of purified alpha- and theta-toxins from Clostridium perfringens. J. Infect. Dis. 1988, 157, 272–279. [Google Scholar] [CrossRef]
- Kiu, R.; Hall, L.J. An update on the human and animal enteric pathogen Clostridium perfringens. Emerg. Microbes Infect. 2018, 7, 141. [Google Scholar] [CrossRef] [PubMed]
- Bunting, M.; Lorant, D.E.; Bryant, A.E.; Zimmerman, G.A.; McIntyre, T.M.; Stevens, D.L.; Prescott, S.M. Alpha toxin from Clostridium perfringens induces proinflammatory changes in endothelial cells. J. Clin. Investig. 1997, 100, 565–574. [Google Scholar] [CrossRef] [PubMed]
- Qureshi, S. Clostridial Gas Gangrene. Medscape. 2019. Available online: https://emedicine.medscape.com/article/214992-overview#a5 (accessed on 18 February 2022).
- Uzal, F.A.; McClane, B.A.; Cheung, J.K.; Theoret, J.; Garcia, J.P.; Moore, R.J.; Rood, J.I. Animal models to study the pathogenesis of human and animal Clostridium perfringens infections. Vet. Microbiol. 2015, 179, 23–33. [Google Scholar] [CrossRef] [PubMed]
- Azar, F.M.; Canale, S.T.; Beaty, J.H. Campbell’s Operative Orthopaedics; Elsevier: Philadelphia, PA, USA, 2021; p. 4536. [Google Scholar]
- Bryant, A.E.; Stevens, D.L. Chapter 11—The Pathogenesis of Gas Gangrene. In The Clostridia; Rood, J.I., McClane, B.A., Songer, J.G., Titball, R.W., Eds.; Academic Press: San Diego, CA, USA, 1997; pp. 185–196. [Google Scholar]
- Buboltz, J.B.; Murphy-Lavoie, H.M. Gas Gangrene. In StatPearls; StatPearls Publishing LLC.: Treasure Island, FL, USA, 2022. [Google Scholar]
- Yao, P.; Annamaraju, P. Clostridium perfringens. 2021. Available online: https://www.ncbi.nlm.nih.gov/books/NBK559049/ (accessed on 18 February 2022).
- Stevens, D.L.; Maier, K.A.; Laine, B.M.; Mitten, J.E. Comparison of Clindamycin, Rifampin, Tetracycline, Metronidazole, and Penicillin for Efficacy in Prevention of Experimental Gas Gangrene Due to Clostridium perfringens. J. Infect. Dis. 1987, 155, 220–228. [Google Scholar] [CrossRef]
- Gazioglu, A.; Karagülle, B.; Yüksel, H.; Nuri Açık, M.; Keçeci, H.; Dörtbudak, M.B.; Çetinkaya, B. Sudden death due to gas gangrene caused by Clostridium septicum in goats. BMC Vet. Res. 2018, 14, 406. [Google Scholar] [CrossRef]
- Salari Sedigh, H.; Rajabioun, M.; Razmyar, J.; Kazemi Mehrjerdi, H. An unusual necrotic myositis by Clostridium perfringens in a German Shepherd dog: A clinical report, bacteriological and molecular identification. Vet. Res. Forum Int. Q. J. 2015, 6, 349–353. [Google Scholar]
- Silva, R.; Uzal, F.; Oliveira, C., Jr.; Lobato, F. Clostridial Diseases of Animals; John Wiley & Sons, Ltd.: Hoboken, NJ, USA, 2016. [Google Scholar]
- Junior, C.A.O.; Silva, R.O.S.; Lobato, F.C.F.; Navarro, M.A.; Uzal, F.A. Gas gangrene in mammals: A review. J. Vet. Diagn. Investig. 2020, 32, 175–183. [Google Scholar] [CrossRef]
- Peek, S.F.; Semrad, S.D.; Perkins, G.A. Clostridial myonecrosis in horses (37 cases 1985–2000). Equine Vet. J. 2003, 35, 86–92. [Google Scholar] [CrossRef]
- Reef, V.B. Clostridium perfringens cellulitis and immune-mediated hemolytic anemia in a horse. J. Am. Vet. Med. Assoc. 1983, 182, 251–254. [Google Scholar]
- Robinson, A.; Manser, P.A. Mastitis in a heifer caused by Clostridium welchii, type “A”. Vet. Rec. 1977, 101, 37. [Google Scholar] [CrossRef]
- CDC. Foodborne Germs and Illnesses; CDC: Atlanta, GA, USA, 2020.
- Shrestha, A.; Uzal, F.A.; McClane, B.A. Enterotoxic Clostridia: Clostridium perfringens Enteric Diseases. Microbiol. Spectr. 2018, 6. [Google Scholar] [CrossRef] [PubMed]
- Uçar, A.; Yilmaz, M.V.; Cakiroglu, F.P. Food Safety—Problems and Solutions. In Significance, Prevention and Control of Food Related Diseases; Makun, H., Ed.; IntechOpen: London, UK, 2016. [Google Scholar]
- Clostridium Perfringens. 2022. Available online: http://www.bccdc.ca/health-info/diseases-conditions/clostridium-perfringens (accessed on 18 February 2022).
- MyHealth.Alberta.ca. Foodborne Illness: Clostridium perfringens. 2021. Available online: https://myhealth.alberta.ca/Health/pages/conditions.aspx?hwid=te6324 (accessed on 18 February 2022).
- Chakrabarti, G.; Zhou, X.; McClane, B.A. Death Pathways Activated in CaCo-2 Cells by Clostridium perfringens. Enterotoxin 2003, 71, 4260–4270. [Google Scholar] [CrossRef] [PubMed]
- Chakrabarti, G.; McClane, B.A. The importance of calcium influx, calpain and calmodulin for the activation of CaCo-2 cell death pathways by Clostridium perfringens enterotoxin. Cell Microbiol. 2005, 7, 129–146. [Google Scholar] [CrossRef] [PubMed]
- Contributors, W. Clostridium perfringens. 2022. Available online: https://en.wikipedia.org/w/index.php?title=Clostridium_perfringens&oldid=1071364430 (accessed on 18 February 2022).
- Pathogens, M.C.F. Most Common Foodborne Pathogens. 2019. Available online: https://www.eatright.org/homefoodsafety/safety-tips/food-poisoning/most-common-foodborne-pathogens (accessed on 18 February 2022).
- Ma, M.; Li, J.; McClane, B.A. Genotypic and phenotypic characterization of Clostridium perfringens isolates from Darmbrand cases in post-World War II Germany. Infect. Immun. 2012, 80, 4354–4363. [Google Scholar] [CrossRef]
- Larry, M.; Bush, M.T.V.-P. Clostridial Necrotizing Enteritis. 2021. Available online: https://www.merckmanuals.com/professional/infectious-diseases/anaerobic-bacteria/clostridial-necrotizing-enteritis (accessed on 18 February 2022).
- Gui, L.; Subramony, C.; Fratkin, J.; Hughson, M.D. Fatal Enteritis Necroticans (Pigbel) in a Diabetic Adult. Mod. Pathol. 2002, 15, 66–70. [Google Scholar] [CrossRef]
- Murrell, T.G.; Walker, P.D. The pigbel story of Papua New Guinea. Trans. R. Soc. Trop. Med. Hygen. 1991, 85, 119–122. [Google Scholar] [CrossRef]
- Shulhan, J.; Dicken, B.; Hartling, L.; Larsen, B.M. Current Knowledge of Necrotizing Enterocolitis in Preterm Infants and the Impact of Different Types of Enteral Nutrition Products. Adv. Nutr. 2017, 8, 80–91. [Google Scholar] [CrossRef]
- Gregory, K.E.; Deforge, C.E.; Natale, K.M.; Phillips, M.; Van Marter, L.J. Necrotizing enterocolitis in the premature infant: Neonatal nursing assessment, disease pathogenesis, and clinical presentation. Adv. Neonatal Care Off. J. Natl. Assoc. Neonatal Nurses 2011, 11, 155–166. [Google Scholar] [CrossRef]
- Heida, F.H.; van Zoonen, A.; Hulscher, J.B.F.; Te Kiefte, B.J.C.; Wessels, R.; Kooi, E.M.W.; Bos, A.F.; Harmsen, H.J.M.; de Goffau, M.C. A Necrotizing Enterocolitis-Associated Gut Microbiota is Present in the Meconium: Results of a Prospective Study. Clin. Infect. Dis. 2016, 62, 863–870. [Google Scholar] [CrossRef]
- Neu, J.; Walker, W.A. Necrotizing enterocolitis. N. Engl. J. Med. 2011, 364, 255–264. [Google Scholar] [CrossRef]
- Bode, L. Human Milk Oligosaccharides in the Prevention of Necrotizing Enterocolitis: A Journey from in vitro and in vivo Models to Mother-Infant Cohort Studies. Front. Pediatr. 2018, 6, 385. [Google Scholar] [CrossRef] [PubMed]
- Kaldhusdal, M.; Benestad, S.L.; Lovland, A. Epidemiologic aspects of necrotic enteritis in broiler chickens—Disease occurrence and production performance. Avian Pathol. 2016, 45, 271–274. [Google Scholar] [CrossRef]
- Hargis, B.M. Overview of Necrotic Enteritis in Poultry. 2016. Available online: https://www.merckvetmanual.com/poultry/necrotic-enteritis/overview-of-necrotic-enteritis-in-poultry (accessed on 20 February 2022).
- Cooper, K.K.; Songer, J.G.; Uzal, F.A. Diagnosing clostridial enteric disease in poultry. J. Vet.-Diagn. Investig. 2013, 25, 314–327. [Google Scholar] [CrossRef] [PubMed]
- Gu, C.; Lillehoj, H.S.; Sun, Z.; Lee, Y.; Zhao, H.; Xianyu, Z.; Yan, X.; Wang, Y.; Lin, S.; Liu, L.; et al. Characterization of Virulent netB(+)/tpeL(+) Clostridium perfringens Strains from Necrotic Enteritis-Affected Broiler Chicken Farms. Avian Dis. 2019, 63, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Lee, K.-W.; Lillehoj, H.S. Role of Clostridium perfringens Necrotic Enteritis B-like Toxin in Disease Pathogenesis. Vaccines 2022, 10, 61. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.B.; Rodgers, N.; Choct, M. Optimized necrotic enteritis model producing clinical and subclinical infection of Clostridium perfringens in broiler chickens. Avian Dis. 2010, 54, 1058–1065. [Google Scholar] [CrossRef] [PubMed]
- Shojadoost, B.; Vince, A.R.; Prescott, J.F. The successful experimental induction of necrotic enteritis in chickens by Clostridium perfringens: A critical review. Vet. Res. 2012, 43, 74. [Google Scholar] [CrossRef]
- Nauerby, B.; Pedersen, K.; Madsen, M. Analysis by pulsed-field gel electrophoresis of the genetic diversity among Clostridium perfringens isolates from chickens. Vet. Microbiol. 2003, 94, 257–266. [Google Scholar] [CrossRef]
- Gholamiandekhordi, A.R.; Ducatelle, R.; Heyndrickx, M.; Haesebrouck, F.; Van Immerseel, F. Molecular and phenotypical characterization of Clostridium perfringens isolates from poultry flocks with different disease status. Vet. Microbiol. 2006, 113, 143–152. [Google Scholar] [CrossRef]
- Wilkie, D.C.; Van Kessel, A.G.; Dumonceaux, T.J.; Drew, M.D. The effect of hen-egg antibodies on Clostridium perfringens colonization in the gastrointestinal tract of broiler chickens. Prev. Vet. Med. 2006, 74, 279–292. [Google Scholar] [CrossRef]
- Keyburn, A.L.; Sheedy, S.A.; Ford, M.E.; Williamson, M.M.; Awad, M.M.; Rood, J.I.; Moore, R.J. Alpha-toxin of Clostridium perfringens is not an essential virulence factor in necrotic enteritis in chickens. Infect. Immun. 2006, 74, 6496–6500. [Google Scholar] [CrossRef]
- Keyburn, A.L.; Boyce, J.D.; Vaz, P.; Bannam, T.L.; Ford, M.E.; Parker, D.; Di Rubbo, A.; Rood, J.I.; Moore, R.J. NetB, a new toxin that is associated with avian necrotic enteritis caused by Clostridium perfringens. PLoS Pathog. 2008, 4, e26. [Google Scholar] [CrossRef] [PubMed]
- Ballard, J.; Sokolov, Y.; Yuan, W.L.; Kagan, B.L.; Tweten, R.K. Activation and mechanism of Clostridium septicum alpha toxin. Mol. Microbiol. 1993, 10, 627–634. [Google Scholar] [CrossRef] [PubMed]
- Chalmers, G.; Bruce, H.L.; Hunter, D.B.; Parreira, V.R.; Kulkarni, R.R.; Jiang, Y.F.; Prescott, J.F.; Boerlin, P. Multilocus sequence typing analysis of Clostridium perfringens isolates from necrotic enteritis outbreaks in broiler chicken populations. J. Clin. Microbiol. 2008, 46, 3957–3964. [Google Scholar] [CrossRef] [PubMed]
- Johansson, A.; Aspán, A.; Kaldhusdal, M.; Engström, B.E. Genetic diversity and prevalence of netB in Clostridium perfringens isolated from a broiler flock affected by mild necrotic enteritis. Vet. Microbiol. 2010, 144, 87–92. [Google Scholar] [CrossRef] [PubMed]
- Hibberd, M.C.; Neumann, A.P.; Rehberger, T.G.; Siragusa, G.R. Multilocus sequence typing subtypes of poultry Clostridium perfringens isolates demonstrate disease niche partitioning. J. Clin. Microbiol. 2011, 49, 1556–1567. [Google Scholar] [CrossRef] [PubMed]
- Abildgaard, L.; Sondergaard, T.E.; Engberg, R.M.; Schramm, A.; Højberg, O. In vitro production of necrotic enteritis toxin B, NetB, by netB-positive and netB-negative Clostridium perfringens originating from healthy and diseased broiler chickens. Vet. Microbiol. 2010, 144, 231–235. [Google Scholar] [CrossRef]
- Goossens, E.; Verherstraeten, S.; Timbermont, L.; Valgaeren, B.R.; Pardon, B.; Haesebrouck, F.; Ducatelle, R.; Deprez, P.R.; Van Immerseel, F. Clostridium perfringens strains from bovine enterotoxemia cases are not superior in in vitro production of alpha toxin, perfringolysin O and proteolytic enzymes. BMC Vet. Res. 2014, 10, 32. [Google Scholar] [CrossRef]
- Henry, R.; Stämpfli, O.J.O.-E. Enterotoxemias in Animals (Enterotoxemias in Animals). 2021. Available online: https://www.merckvetmanual.com/generalized-conditions/clostridial-diseases/enterotoxemias-in-animals (accessed on 20 February 2022).
- Uzal, F.A.; Songer, J.G. Diagnosis of Clostridium perfringens Intestinal Infections in Sheep and Goats. J. Vet.-Diagn. Investig. 2008, 20, 253–265. [Google Scholar] [CrossRef]
- Simpson, K.M.; Callan, R.J.; van Metre, D.C. Clostridial Abomasitis and Enteritis in Ruminants. Vet. Clin. N. Am. Food Anim. Pract. 2018, 34, 155–184. [Google Scholar] [CrossRef]
- Hines, M.E., II. Enterotoxemia in Sheep and Goats. 2013. Available online: https://vet.uga.edu/enterotoxemia-in-sheep-and-goats/ (accessed on 20 February 2022).
- O’Brien, D.K.; Therit, B.H.; Woodman, M.E.; Melville, S.B. The role of neutrophils and monocytic cells in controlling the initiation of Clostridium perfringens gas gangrene. FEMS Immunol. Med. Microbiol. 2007, 50, 86–93. [Google Scholar] [CrossRef]
- Stevens, D.L.; Tweten, R.K.; Awad, M.M.; Rood, J.I.; Bryant, A.E. Clostridial gas gangrene: Evidence that alpha and theta toxins differentially modulate the immune response and induce acute tissue necrosis. J. Infect. Dis. 1997, 176, 189–195. [Google Scholar] [CrossRef]
- Low, L.Y.; Harrison, P.F.; Gould, J.; Powell, D.R.; Choo, J.M.; Forster, S.C.; Chapman, R.; Gearing, L.J.; Cheung, J.K.; Hertzog, P.; et al. Concurrent Host-Pathogen Transcriptional Responses in a Clostridium perfringens Murine Myonecrosis Infection. mBio 2018, 9, e00473-18. [Google Scholar] [CrossRef]
- Doeing, D.C.; Borowicz, J.L.; Crockett, E.T. Gender dimorphism in differential peripheral blood leukocyte counts in mice using cardiac, tail, foot, and saphenous vein puncture methods. BMC Clin. Pathol. 2003, 3, 3. [Google Scholar] [CrossRef]
- Mestas, J.; Hughes, C.C. Of mice and not men: Differences between mouse and human immunology. J. Immunol. 2004, 172, 2731–2738. [Google Scholar] [CrossRef]
- Thomas, G.D.; Hamers, A.A.J.; Nakao, C.; Marcovecchio, P.; Taylor, A.M.; McSkimming, C.; Nguyen, A.T.; McNamara, C.A.; Hedrick, C.C. Human Blood Monocyte Subsets: A New Gating Strategy Defined Using Cell Surface Markers Identified by Mass Cytometry. Arterioscler. Thromb. Vasc. Biol. 2017, 37, 1548–1558. [Google Scholar] [CrossRef]
- Van Furth, R.; Sluiter, W. Distribution of blood monocytes between a marginating and a circulating pool. J. Exp. Med. 1986, 163, 474–479. [Google Scholar] [CrossRef]
- Van Furth, R.; Cohn, Z.A. The origin and kinetics of mononuclear phagocytes. J. Exp. Med. 1968, 128, 415–435. [Google Scholar] [CrossRef]
- Kurihara, T.; Warr, G.; Loy, J.; Bravo, R. Defects in macrophage recruitment and host defense in mice lacking the CCR2 chemokine receptor. J. Exp. Med. 1997, 186, 1757–1762. [Google Scholar] [CrossRef]
- Shi, C.; Pamer, E.G. Monocyte recruitment during infection and inflammation. Nat. Rev. Immunol. 2011, 11, 762–774. [Google Scholar] [CrossRef]
- Ginhoux, F.; Jung, S. Monocytes and macrophages: Developmental pathways and tissue homeostasis. Nat. Rev. Immunol. 2014, 14, 392–404. [Google Scholar] [CrossRef]
- Takehara, M.; Takagishi, T.; Seike, S.; Ohtani, K.; Kobayashi, K.; Miyamoto, K.; Shimizu, T.; Nagahama, M. Clostridium perfringens α-Toxin Impairs Innate Immunity via Inhibition of Neutrophil Differentiation. Sci. Rep. 2016, 6, 28192. [Google Scholar] [CrossRef]
- Bryant, A.E.; Bayer, C.R.; Aldape, M.J.; Wallace, R.J.; Titball, R.W.; Stevens, D.L. Clostridium perfringens phospholipase C-induced platelet/leukocyte interactions impede neutrophil diapedesis. J. Med. Microbiol. 2006, 55 Pt 5, 495–504. [Google Scholar] [CrossRef]
- Manz, M.G.; Boettcher, S. Emergency granulopoiesis. Nat. Rev. Immunol. 2014, 14, 302–314. [Google Scholar] [CrossRef]
- Lieschke, G.J.; Grail, D.; Hodgson, G.; Metcalf, D.; Stanley, E.; Cheers, C.; Fowler, K.J.; Basu, S.; Zhan, Y.F.; Dunn, A.R. Mice lacking granulocyte colony-stimulating factor have chronic neutropenia, granulocyte and macrophage progenitor cell deficiency, and impaired neutrophil mobilization. Blood 1994, 84, 1737–1746. [Google Scholar] [CrossRef]
- Ley, K.; Smith, E.; Stark, M.A. IL-17A-producing neutrophil-regulatory Tn lymphocytes. Immunol. Res. 2006, 34, 229–242. [Google Scholar] [CrossRef]
- Chen, K.; Kolls, J.K. Interluekin-17A (IL17A). Gene 2017, 614, 8–14. [Google Scholar] [CrossRef]
- Lehman, H.K.; Segal, B.H. The role of neutrophils in host defense and disease. J. Allergy Clin. Immunol. 2020, 145, 1535–1544. [Google Scholar] [CrossRef]
- Papayannopoulos, V. Neutrophil extracellular traps in immunity and disease. Nat. Rev. Immunol. 2018, 18, 134–147. [Google Scholar] [CrossRef]
- Williams, R.B. Intercurrent coccidiosis and necrotic enteritis of chickens: Rational, integrated disease management by maintenance of gut integrity. Avian Pathol. 2005, 34, 159–180. [Google Scholar] [CrossRef]
- McReynolds, J.L.; Byrd, J.A.; Anderson, R.C.; Moore, R.W.; Edrington, T.S.; Genovese, K.J.; Poole, T.L.; Kubena, L.F.; Nisbet, D.J. Evaluation of immunosuppressants and dietary mechanisms in an experimental disease model for necrotic enteritis. Poult. Sci. 2004, 83, 1948–1952. [Google Scholar] [CrossRef]
- Vincent, D.; Klinke, M.; Eschenburg, G.; Trochimiuk, M.; Appl, B.; Tiemann, B.; Bergholz, R.; Reinshagen, K.; Boettcher, M. NEC is likely a NETs dependent process and markers of NETosis are predictive of NEC in mice and humans. Sci. Rep. 2018, 8, 12612. [Google Scholar] [CrossRef]
- Fasina, Y.O.; Lillehoj, H.S. Characterization of intestinal immune response to Clostridium perfringens infection in broiler chickens. Poult. Sci. 2019, 98, 188–198. [Google Scholar] [CrossRef]
- Daneshmand, A.; Kermanshahi, H.; Mohammed, J.; Sekhavati, M.H.; Javadmanesh, A.; Ahmadian, M.; Alizadeh, M.; Razmyar, J.; Kulkarni, R.R. Intestinal changes and immune responses during Clostridium perfringens-induced necrotic enteritis in broiler chickens. Poult. Sci. 2022, 101, 101652. [Google Scholar] [CrossRef]
- Su, S.; Dwyer, D.M.; Miska, K.B.; Fetterer, R.H.; Jenkins, M.C.; Wong, E.A. Expression of host defense peptides in the intestine of Eimeria-challenged chickens. Poult. Sci. 2017, 96, 2421–2427. [Google Scholar] [CrossRef]
- Zaytsoff, S.J.M.; Lyons, S.M.; Garner, A.M.; Uwiera, R.R.E.; Zandberg, W.F.; Abbott, D.W.; Inglis, G.D. Host responses to Clostridium perfringens challenge in a chicken model of chronic stress. Gut Pathog. 2020, 12, 24. [Google Scholar] [CrossRef]
- Rooks, M.G.; Garrett, W.S. Gut microbiota, metabolites and host immunity. Nat. Rev. Immunol. 2016, 16, 341–352. [Google Scholar] [CrossRef]
- Humphrey, S.; Chaloner, G.; Kemmett, K.; Davidson, N.; Williams, N.; Kipar, A.; Humphrey, T.; Wigley, P. Campylobacter jejuni is not merely a commensal in commercial broiler chickens and affects bird welfare. mBio 2014, 5, e01364-14. [Google Scholar] [CrossRef]
- Belkaid, Y.; Naik, S. Compartmentalized and systemic control of tissue immunity by commensals. Nat. Immunol. 2013, 14, 646–653. [Google Scholar] [CrossRef]
- Shan, M.; Gentile, M.; Yeiser, J.R.; Walland, A.C.; Bornstein, V.U.; Chen, K.; He, B.; Cassis, L.; Bigas, A.; Cols, M.; et al. Mucus enhances gut homeostasis and oral tolerance by delivering immunoregulatory signals. Science 2013, 342, 447–453. [Google Scholar] [CrossRef]
- Macpherson, A.J.; Uhr, T. Induction of protective IgA by intestinal dendritic cells carrying commensal bacteria. Science 2004, 303, 1662–1665. [Google Scholar] [CrossRef]
- Pabst, O.; Mowat, A.M. Oral tolerance to food protein. Mucosal. Immunol. 2012, 5, 232–239. [Google Scholar] [CrossRef]
- Davis, C.P. Normal Flora. In Medical Microbiology; Baron, S., Ed.; The University of Texas Medical Branch at Galveston: Galveston, TX, USA, 1996. [Google Scholar]
- Bereswill, S.; Fischer, A.; Plickert, R.; Haag, L.M.; Otto, B.; Kuhl, A.A.; Dasti, J.I.; Zautner, A.E.; Munoz, M.; Loddenkemper, C.; et al. Novel murine infection models provide deep insights into the “menage a trois” of Campylobacter jejuni, microbiota and host innate immunity. PLoS ONE 2011, 6, e20953. [Google Scholar] [CrossRef]
- Warner, B.B.; Tarr, P.I. Necrotizing enterocolitis and preterm infant gut bacteria. Semin. Fetal Neonatal Med. 2016, 21, 394–399. [Google Scholar] [CrossRef]
- Cotten, C.M.; Taylor, S.; Stoll, B.; Goldberg, R.N.; Hansen, N.I.; Sanchez, P.J.; Ambalavanan, N.; Benjamin, D.K., Jr.; Network, N.N.R. Prolonged duration of initial empirical antibiotic treatment is associated with increased rates of necrotizing enterocolitis and death for extremely low birth weight infants. Pediatrics 2009, 123, 58–66. [Google Scholar] [CrossRef]
- Warner, B.B.; Deych, E.; Zhou, Y.; Hall-Moore, C.; Weinstock, G.M.; Sodergren, E.; Shaikh, N.; Hoffmann, J.A.; Linneman, L.A.; Hamvas, A.; et al. Gut bacteria dysbiosis and necrotising enterocolitis in very low birthweight infants: A prospective case-control study. Lancet 2016, 387, 1928–1936. [Google Scholar] [CrossRef]
- Mai, V.; Young, C.M.; Ukhanova, M.; Wang, X.; Sun, Y.; Casella, G.; Theriaque, D.; Li, N.; Sharma, R.; Hudak, M.; et al. Fecal microbiota in premature infants prior to necrotizing enterocolitis. PLoS ONE 2011, 6, e20647. [Google Scholar] [CrossRef]
- Morrow, A.L.; Lagomarcino, A.J.; Schibler, K.R.; Taft, D.H.; Yu, Z.; Wang, B.; Altaye, M.; Wagner, M.; Gevers, D.; Ward, D.V.; et al. Early microbial and metabolomic signatures predict later onset of necrotizing enterocolitis in preterm infants. Microbiome 2013, 1, 13. [Google Scholar] [CrossRef]
- McMurtry, V.E.; Gupta, R.W.; Tran, L.; Blanchard, E.E.; Penn, D.; Taylor, C.M.; Ferris, M.J. Bacterial diversity and Clostridia abundance decrease with increasing severity of necrotizing enterocolitis. Microbiome 2015, 3, 11. [Google Scholar] [CrossRef]
- Yang, Q.; Liu, J.; Wang, X.; Robinson, K.; Whitmore, M.A.; Stewart, S.N.; Zhao, J.; Zhang, G. Identification of an Intestinal Microbiota Signature Associated With the Severity of Necrotic Enteritis. Front. Microbiol. 2021, 12, 703693. [Google Scholar] [CrossRef]
- Kim, J.E.; Lillehoj, H.S.; Hong, Y.H.; Kim, G.B.; Lee, S.H.; Lillehoj, E.P.; Bravo, D.M. Dietary Capsicum and Curcuma longa oleoresins increase intestinal microbiome and necrotic enteritis in three commercial broiler breeds. Res. Vet. Sci. 2015, 102, 150–158. [Google Scholar] [CrossRef]
- Latorre, J.D.; Adhikari, B.; Park, S.H.; Teague, K.D.; Graham, L.E.; Mahaffey, B.D.; Baxter, M.F.A.; Hernandez-Velasco, X.; Kwon, Y.M.; Ricke, S.C.; et al. Evaluation of the Epithelial Barrier Function and Ileal Microbiome in an Established Necrotic Enteritis Challenge Model in Broiler Chickens. Front. Vet. Sci. 2018, 5, 199. [Google Scholar] [CrossRef]
- Lacey, J.A.; Stanley, D.; Keyburn, A.L.; Ford, M.; Chen, H.; Johanesen, P.; Lyras, D.; Moore, R.J. Clostridium perfringens-mediated necrotic enteritis is not influenced by the pre-existing microbiota but is promoted by large changes in the post-challenge microbiota. Vet. Microbiol. 2018, 227, 119–126. [Google Scholar] [CrossRef]
- Bortoluzzi, C.; Vieira, B.S.; Hofacre, C.; Applegate, T.J. Effect of different challenge models to induce necrotic enteritis on the growth performance and intestinal microbiota of broiler chickens. Poult. Sci. 2019, 98, 2800–2812. [Google Scholar] [CrossRef]
- Bansal, M.; Fu, Y.; Alrubaye, B.; Abraha, M.; Almansour, A.; Gupta, A.; Liyanage, R.; Wang, H.; Hargis, B.; Sun, X. A secondary bile acid from microbiota metabolism attenuates ileitis and bile acid reduction in subclinical necrotic enteritis in chickens. J. Anim. Sci. Biotechnol. 2020, 11, 37. [Google Scholar] [CrossRef]
- Eltzschig, H.K.; Eckle, T. Ischemia and reperfusion--from mechanism to translation. Nat. Med. 2011, 17, 1391–1401. [Google Scholar] [CrossRef]
- Yellon, D.M.; Hausenloy, D.J. Myocardial reperfusion injury. N. Engl. J. Med. 2007, 357, 1121–1135. [Google Scholar] [CrossRef]
- Bryant, A.E.; Chen, R.Y.; Nagata, Y.; Wang, Y.; Lee, C.H.; Finegold, S.; Guth, P.H.; Stevens, D.L. Clostridial gas gangrene. I. Cellular and molecular mechanisms of microvascular dysfunction induced by exotoxins of Clostridium perfringens. J. Infect. Dis. 2000, 182, 799–807. [Google Scholar]
- Schropfer, E.; Rauthe, S.; Meyer, T. Diagnosis and misdiagnosis of necrotizing soft tissue infections: Three case reports. Cases J. 2008, 1, 252. [Google Scholar] [CrossRef]
- Jame, R.; Vilímová, V.; Lakatoš, B.; Varečka, Ľ. The hydrogen production by anaerobic bacteria grown on glucose and glycerol. Acta Chim. Slovaca 2011, 4, 145–157. [Google Scholar]
- Bhatia, M. Hydrogen sulfide as a vasodilator. IUBMB Life 2005, 57, 603–606. [Google Scholar] [CrossRef] [PubMed]
- Fuhrmann, D.C.; Brune, B. Mitochondrial composition and function under the control of hypoxia. Redox Biol. 2017, 12, 208–215. [Google Scholar] [CrossRef]
- Bedard, K.; Krause, K.H. The NOX family of ROS-generating NADPH oxidases: Physiology and pathophysiology. Physiol. Rev. 2007, 87, 245–313. [Google Scholar] [CrossRef]
- Kimura, M.; Kataoka, M.; Kuwabara, Y.; Sato, A.; Kato, T.; Narita, K.; Okahira, T.; Fujii, Y. Beneficial effects of verapamil on intestinal ischemia and reperfusion injury: Pretreatment versus postischemic treatment. Eur. Surg. Res. 1998, 30, 191–197. [Google Scholar] [CrossRef]
- Erdogan, O.; Yildiz, S.; Basaran, A.; Demirbas, A.; Yesilkaya, A. Effect of intraportal verapamil infusion on hepatic ischemia-reperfusion injury. Pol. J. Pharm. 2001, 53, 137–141. [Google Scholar]
- World Health Organization. Vaccines and Immunization: What is Vaccination? 2021. Available online: https://www.who.int/news-room/questions-and-answers/item/vaccines-and-immunization-what-is-vaccination (accessed on 20 February 2022).
- Ardehali, M.; Darakhshan, H. Production and standardization of polyvalent Clostridium perfringens vaccine in Iran. Dev. Biol. Stand. 1976, 32, 31–34. [Google Scholar] [PubMed]
- Baxter, D. Active and passive immunity, vaccine types, excipients and licensing. Occup. Med. 2007, 57, 552–556. [Google Scholar] [CrossRef] [PubMed]
- Keyburn, A.L.; Portela, R.W.; Sproat, K.; Ford, M.E.; Bannam, T.L.; Yan, X.; Rood, J.I.; Moore, R.J. Vaccination with recombinant NetB toxin partially protects broiler chickens from necrotic enteritis. Vet. Res. 2013, 44, 54. [Google Scholar] [CrossRef]
- Lovland, A.; Kaldhusdal, M.; Redhead, K.; Skjerve, E.; Lillehaug, A. Maternal vaccination against subclinical necrotic enteritis in broilers. Avian Pathol. 2004, 33, 83–92. [Google Scholar] [CrossRef]
- Fisher, D.J.; Fernandez-Miyakawa, M.E.; Sayeed, S.; Poon, R.; Adams, V.; Rood, J.I.; Uzal, F.A.; McClane, B.A. Dissecting the contributions of Clostridium perfringens type C toxins to lethality in the mouse intravenous injection model. Infect. Immun. 2006, 74, 5200–5210. [Google Scholar] [CrossRef]
- Siqueira, F.F.; Silva, R.O.S.; do Carmo, A.O.; de Oliveira-Mendes, B.B.R.; Horta, C.C.R.; Lobato, F.C.F.; Kalapothakis, E. Immunization with a nontoxic naturally occurring Clostridium perfringens alpha toxin induces neutralizing antibodies in rabbits. Anaerobe 2018, 49, 48–52. [Google Scholar] [CrossRef] [PubMed]
- Dwivedi, P.; Alam, S.I.; Kumar, O.; Kumar, R.B. Lipoproteins from Clostridium perfringens and their protective efficacy in mouse model. Infect. Genet. Evol. 2015, 34, 434–443. [Google Scholar] [CrossRef]
- Hoang, T.H.; Hong, H.A.; Clark, G.C.; Titball, R.W.; Cutting, S.M. Recombinant Bacillus subtilis expressing the Clostridium perfringens alpha toxoid is a candidate orally delivered vaccine against necrotic enteritis. Infect. Immun. 2008, 76, 5257–5265. [Google Scholar] [CrossRef]
- Alimolaei, M.; Golchin, M.; Abshenas, J.; Ezatkhah, M.; Bafti, M.S. A Recombinant Probiotic, Lactobacillus casei, Expressing the Clostridium perfringens alpha-toxoid, as an Orally Vaccine Candidate Against Gas Gangrene and Necrotic Enteritis. Probiotics Antimicrob. Proteins 2018, 10, 251–257. [Google Scholar] [CrossRef]
- Alimolaei, M.; Golchin, M.; Daneshvar, H. Oral immunization of mice against Clostridium perfringens epsilon toxin with a Lactobacillus casei vector vaccine expressing epsilon toxoid. Infect. Genet. Evol. 2016, 40, 282–287. [Google Scholar] [CrossRef]
- Shreya, D.; Uppalapati, S.R.; Kingston, J.J.; Sripathy, M.H.; Batra, H.V. Immunization with recombinant bivalent chimera r-Cpae confers protection against alpha toxin and enterotoxin of Clostridium perfringens type A in murine model. Mol. Immunol. 2015, 65, 51–57. [Google Scholar] [CrossRef] [PubMed]
- Alam, S.I.; Dwivedi, P. Putative function of hypothetical proteins expressed by Clostridium perfringens type A strains and their protective efficacy in mouse model. Infect. Genet. Evol. 2016, 44, 147–156. [Google Scholar] [CrossRef]
- Hakkarainen, T.W.; Kopari, N.M.; Pham, T.N.; Evans, H.L. Necrotizing soft tissue infections: Review and current concepts in treatment, systems of care, and outcomes. Curr. Probl. Surg. 2014, 51, 344–362. [Google Scholar] [CrossRef] [PubMed]
- Yang, Z.; Hu, J.; Qu, Y.; Sun, F.; Leng, X.; Li, H.; Zhan, S. Interventions for treating gas gangrene. Cochrane Database Syst. Rev. 2015, 2015, CD010577. [Google Scholar] [CrossRef]
- Devaney, B.; Frawley, G.; Frawley, L.; Pilcher, D.V. Necrotising soft tissue infections: The effect of hyperbaric oxygen on mortality. Anaesth. Intensiv. Care 2015, 43, 685–692. [Google Scholar] [CrossRef]
- Bao, P.; Kodra, A.; Tomic-Canic, M.; Golinko, M.S.; Ehrlich, H.P.; Brem, H. The role of vascular endothelial growth factor in wound healing. J. Surg. Res. 2009, 153, 347–358. [Google Scholar] [CrossRef] [PubMed]
- Pierro, A. The surgical management of necrotising enterocolitis. Early Hum. Dev. 2005, 81, 79–85. [Google Scholar] [CrossRef] [PubMed]
- Lu, R.-M.; Hwang, Y.-C.; Liu, I.J.; Lee, C.-C.; Tsai, H.-Z.; Li, H.-J.; Wu, H.-C. Development of therapeutic antibodies for the treatment of diseases. J. Biomed. Sci. 2020, 27, 1. [Google Scholar] [CrossRef] [PubMed]
- Garcia, J.P.; Beingesser, J.; Bohorov, O.; Bohorova, N.; Goodman, C.; Kim, D.; Pauly, M.; Velasco, J.; Whaley, K.; Zeitlin, L.; et al. Prevention and treatment of Clostridium perfringens epsilon toxin intoxication in mice with a neutralizing monoclonal antibody (c4D7) produced in Nicotiana benthamiana. Toxicon Off. J. Int. Soc. Toxinol. 2014, 88, 93–98. [Google Scholar] [CrossRef] [PubMed]
- Linden, J.R.; Telesford, K.; Shetty, S.; Winokour, P.; Haigh, S.; Cahir-McFarland, E.; Antognetti, G.; Datta, A.; Wang, T.; Meier, W.; et al. A Novel Panel of Rabbit Monoclonal Antibodies and Their Diverse Applications Including Inhibition of Clostridium perfringens Epsilon Toxin Oligomerization. Antibodies 2018, 7, 37. [Google Scholar] [CrossRef]
- Bryant, A.E. Biology and pathogenesis of thrombosis and procoagulant activity in invasive infections caused by group A streptococci and Clostridium perfringens. Clin. Microbiol. Rev. 2003, 16, 451–462. [Google Scholar] [CrossRef]
- Lee, J.W.; Ko, J.; Ju, C.; Eltzschig, H.K. Hypoxia signaling in human diseases and therapeutic targets. Exp. Mol. Med. 2019, 51, 1–13. [Google Scholar] [CrossRef]
- Shrestha, A.; Gohari, I.M.; McClane, B.A.; Ballard, J.D. RIP1, RIP3, and MLKL Contribute to Cell Death Caused by Clostridium perfringens Enterotoxin. mBio 2019, 10, e02985-19. [Google Scholar] [CrossRef]
- Khalique, A.; Zeng, D.; Shoaib, M.; Wang, H.; Qing, X.; Rajput, D.S.; Pan, K.; Ni, X. Probiotics mitigating subclinical necrotic enteritis (SNE) as potential alternatives to antibiotics in poultry. AMB Express 2020, 10, 50. [Google Scholar] [CrossRef]
- Caly, D.L.; D’Inca, R.; Auclair, E.; Drider, D. Alternatives to Antibiotics to Prevent Necrotic Enteritis in Broiler Chickens: A Microbiologist’s Perspective. Front. Microbiol. 2015, 6, 1336. [Google Scholar] [CrossRef]
- Gong, L.; Wang, B.; Zhou, Y.; Tang, L.; Zeng, Z.; Zhang, H.; Li, W. Protective Effects of Lactobacillus plantarum 16 and Paenibacillus polymyxa 10 Against Clostridium perfringens Infection in Broilers. Front. Immunol. 2021, 11, 3934. [Google Scholar] [CrossRef] [PubMed]
- Zhou, M.; Zeng, D.; Ni, X.; Tu, T.; Yin, Z.; Pan, K.; Jing, B. Effects of Bacillus licheniformis on the growth performance and expression of lipid metabolism-related genes in broiler chickens challenged with Clostridium perfringens-induced necrotic enteritis. Lipids Health Dis. 2016, 15, 48. [Google Scholar] [CrossRef]
- Agus, A.; Clément, K.; Sokol, H. Gut microbiota-derived metabolites as central regulators in metabolic disorders. Gut 2021, 70, 1174–1182. [Google Scholar] [CrossRef] [PubMed]
- Diaz Carrasco, J.M.; Redondo, L.M.; Redondo, E.A.; Dominguez, J.E.; Chacana, A.P.; Fernandez Miyakawa, M.E. Use of Plant Extracts as an Effective Manner to Control Clostridium perfringens Induced Necrotic Enteritis in Poultry. BioMed Res. Int. 2016, 2016, 3278359. [Google Scholar] [CrossRef] [PubMed]
- Song, B.; Li, P.; Yan, S.; Liu, Y.; Gao, M.; Lv, H.; Lv, Z.; Guo, Y. Effects of Dietary Astragalus Polysaccharide Supplementation on the Th17/Treg Balance and the Gut Microbiota of Broiler Chickens Challenged With Necrotic Enteritis. Front. Immunol. 2022, 13, 781934. [Google Scholar] [CrossRef]
| Toxins | Toxin Types | Encoded Genes | Gene Location | Protein Size, kDa |
|---|---|---|---|---|
| Alpha (CPA or PLC) | A, B, C, D, E, F, G | cpa or plc | Chromosome | 42.5 |
| Beta (CPB) | B, C | cpb | Plasmid | 35 |
| Epsilon (ETX) | B, D | etx | Plasmid | 34 |
| Iota (ITX) | E | iab and iap | Plasmid | Ia: 47.5, Ib: 71.5 |
| Enterotoxin (CPE) | C, D, E, F | cpe | Chromosome or Plasmid | 35.5 |
| Enteritis B-like toxin (NetB) | G | netB | Plasmid | 28 |
| Prevention |
| ∗Vaccine |
| 1. Bacterin vaccines |
| 2. Toxoid vaccines |
| 3. Recombinant proteins vaccines |
| ∗Potential new interventions |
| 1. Monoclonal antibodies (mAb) |
| 2. Blood thinners (anticoagulants) |
| 3. Hypoxia relief |
| 4. Cell death inhibitors |
| 5. Probiotics, gut microbiota and its metabolites |
| Treatment |
| C. perfringens-induced myonecrosis and severe enteritis |
| 1. A combination of intravenous high doses broad-spectrum antibiotics |
| 2. Wound debridement of all bacteria carriers |
| 3. Surgical removal of dead or infected tissues |
| 4. Hyperbaric oxygen therapy |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fu, Y.; Alenezi, T.; Sun, X. Clostridium perfringens-Induced Necrotic Diseases: An Overview. Immuno 2022, 2, 387-407. https://doi.org/10.3390/immuno2020024
Fu Y, Alenezi T, Sun X. Clostridium perfringens-Induced Necrotic Diseases: An Overview. Immuno. 2022; 2(2):387-407. https://doi.org/10.3390/immuno2020024
Chicago/Turabian StyleFu, Ying, Tahrir Alenezi, and Xiaolun Sun. 2022. "Clostridium perfringens-Induced Necrotic Diseases: An Overview" Immuno 2, no. 2: 387-407. https://doi.org/10.3390/immuno2020024
APA StyleFu, Y., Alenezi, T., & Sun, X. (2022). Clostridium perfringens-Induced Necrotic Diseases: An Overview. Immuno, 2(2), 387-407. https://doi.org/10.3390/immuno2020024






