Mathematical Models to Compare the Pharmacokinetics of Methadone, Buprenorphine, Tramadol, and Tapentadol †
Abstract
:1. Introduction
2. Methodology
2.1. Oral Administration
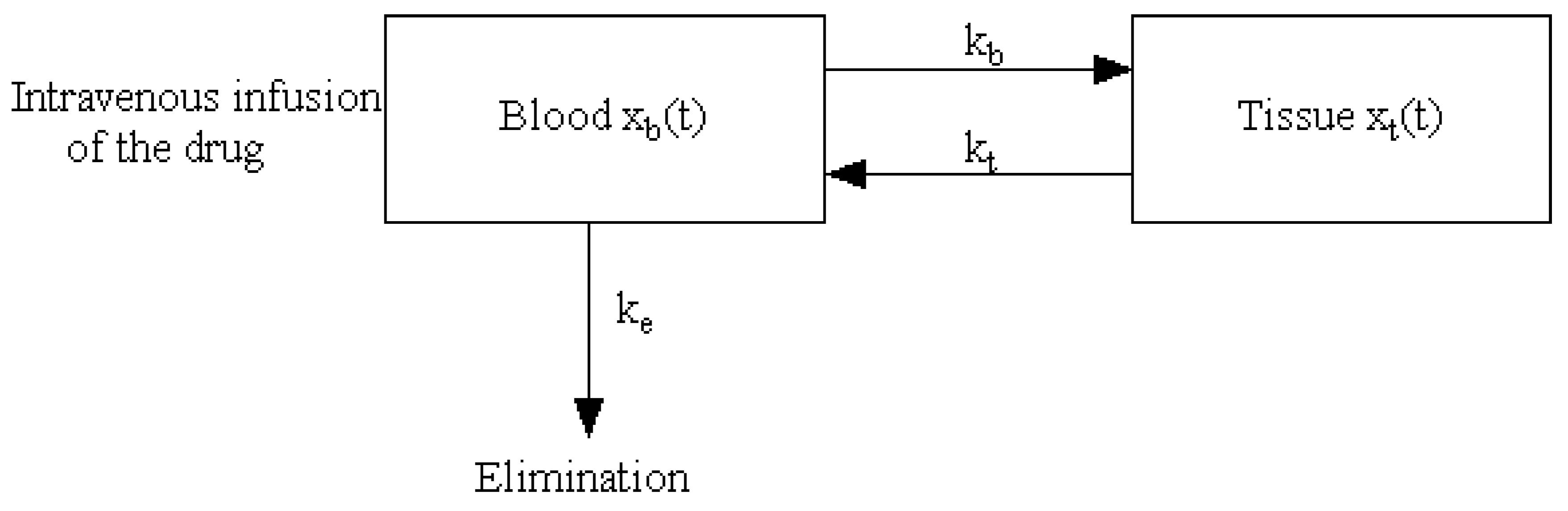
2.2. Intravenous Administration
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Cohen, M.; Quintner, J.; van Rysewyk, S. Reconsidering the International Association for the Study of Pain definition of pain. Pain Rep. 2018, 3, e634. [Google Scholar] [CrossRef] [PubMed]
- Negus, S.S.; Vanderah, T.W.; Brandt, M.R.; Bilsky, E.J.; Becerra, L.; Borsook, D. Preclinical Assessment of Candidate Analgesic Drugs: Recent Advances and Future Challenges. J. Pharmacol. Exp. Ther. 2006, 319, 507–514. [Google Scholar] [CrossRef] [PubMed]
- Cooper, T.E.; Fisher, E.; Gray, A.L.; Krane, E.; Sethna, N.; van Tilburg, M.A.L.; Zernikow, B.; Wiffen, P.J. Opioids for chronic non-cancer pain in children and adolescents. Cochrane Database Syst. Rev. 2017, 7. [Google Scholar] [CrossRef]
- Currie, G.M. Pharmacology, Part 2: Introduction to Pharmacokinetics. J. Nucl. Med. Technol. 2018, 46, 221–230. [Google Scholar] [CrossRef]
- Doogue, M.P.; Polasek, T.M. The ABCD of clinical pharmacokinetics. Ther. Adv. Drug Saf. 2013, 4, 5–7. [Google Scholar] [CrossRef] [PubMed]
- Davis, M.P. Twelve reasons for considering buprenorphine as a frontline analgesic in the management of pain. J. Support. Oncol. 2012, 10, 209–219. [Google Scholar] [CrossRef] [PubMed]
- Pasternak, G.W.; Pan, Y.-X. Mu Opioids and Their Receptors: Evolution of a Concept. Pharmacol. Rev. 2013, 65, 1257–1317. [Google Scholar] [CrossRef] [PubMed]
- Hain, R.; Goldman, A.; Rapoport, A.; Meiring, M. Oxford Textbook of Palliative Care for Children; Oxford University Press: Oxford, UK, 2021. [Google Scholar]
- Nicholson, B. Benefits of Extended-Release Opioid Analgesic Formulations in the Treatment of Chronic Pain. Pain Pract. 2009, 9, 71–81. [Google Scholar] [CrossRef]
- Subedi, M.; Bajaj, S.; Kumar, M.S.; Yc, M. An overview of tramadol and its usage in pain management and future perspective. Biomed. Pharmacother. 2019, 111, 443–451. [Google Scholar] [CrossRef]
- Flecknell, P. Chapter 4—Analgesia and Post-Operative Care. In Laboratory Animal Anaesthesia, 4th ed.; Flecknell, P., Ed.; Academic Press: Boston, MA, USA, 2016; pp. 141–192. [Google Scholar] [CrossRef]
- Arifin, D.Y.; Lee, L.Y.; Wang, C.-H. Mathematical modeling and simulation of drug release from microspheres: Implications to drug delivery systems. Adv. Drug Deliv. Rev. 2006, 58, 1274–1325. [Google Scholar] [CrossRef] [PubMed]
- Siepmann, J.; Siepmann, F. Mathematical modeling of drug delivery. Int. J. Pharm. 2008, 364, 328–343. [Google Scholar] [CrossRef] [PubMed]
- Alqahtani, M.S.; Kazi, M.; Alsenaidy, M.A.; Ahmad, M.Z. Advances in Oral Drug Delivery. Front. Pharmacol. 2021, 12. Available online: https://www.frontiersin.org/articles/10.3389/fphar.2021.618411 (accessed on 24 July 2023). [CrossRef] [PubMed]
- Khanday, M.A.; Rafiq, A.; Nazir, K. Mathematical models for drug diffusion through the compartments of blood and tissue medium. Alex. J. Med. 2017, 53, 245–249. [Google Scholar] [CrossRef]
- Peterfreund, R.A. Intravenous administration of medications during an anesthetic: A deceptively simple process. J. Clin. Monit. Comput. 2019, 33, 547–548. [Google Scholar] [CrossRef] [PubMed]
- Huestis, M.A.; Cone, E.J.; Pirnay, S.O.; Umbricht, A.; Preston, K.L. Intravenous buprenorphine and norbuprenorphine pharmacokinetics in humans. Drug Alcohol. Depend. 2013, 131, 258–262. [Google Scholar] [CrossRef] [PubMed]
- Minkowitz, H.; Leiman, D.; Lu, L.; Reines, S.; Ryan, M.; Harnett, M.; Singla, N. IV Tramadol—A New Treatment Option for Management of Post-Operative Pain in the US: An Open-Label, Single-Arm, Safety Trial Including Various Types of Surgery. J. Pain Res. 2020, 13, 1155–1162. [Google Scholar] [CrossRef] [PubMed]
- Therapeutic Guidelines. Tapentadol for analgesia. Aust. Prescr. 2013, 36, 105–107. [Google Scholar] [CrossRef]
- Dormand, J.R.; Prince, P.J. A family of embedded Runge-Kutta formulae. J. Comput. Appl. Math. 1980, 6, 19–26. [Google Scholar] [CrossRef]
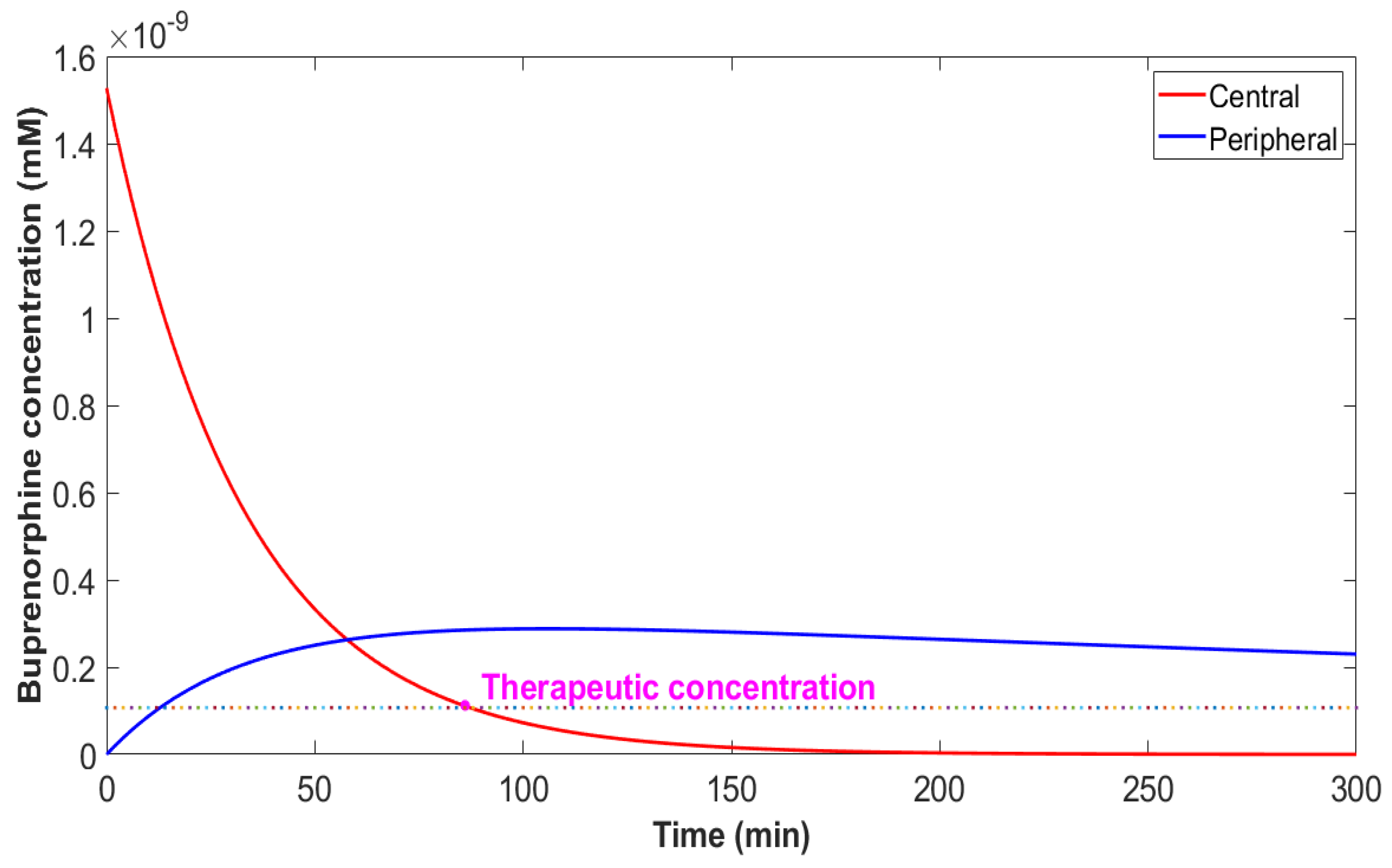


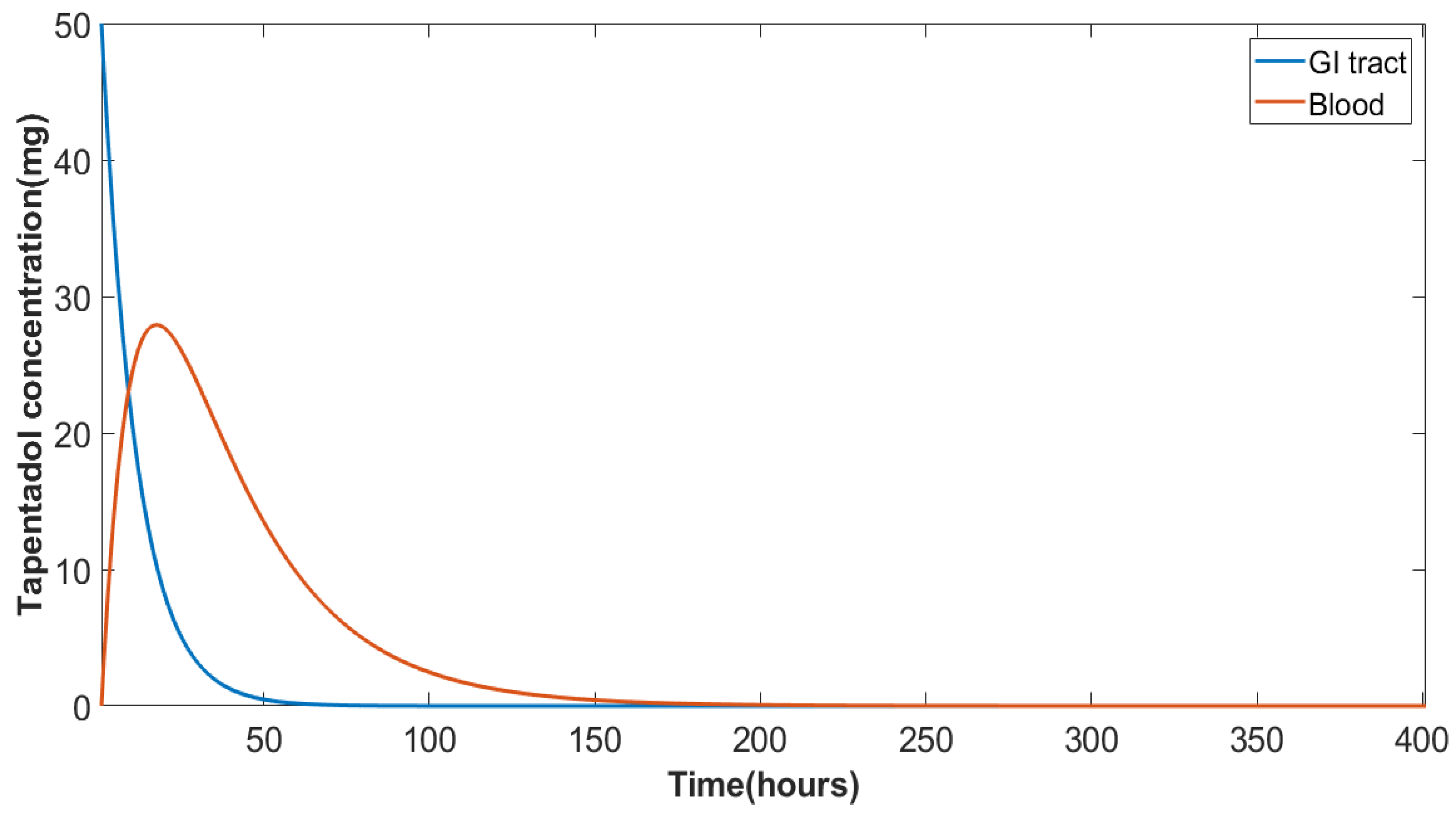
| Sl. No. | Drug | Therapeutic Concentration (mM) | T_therapeutic (minutes) | T_clearance (minutes) |
|---|---|---|---|---|
| 1 | Buprenorphine | 86 | 3578 | |
| 2 | Methadone | 230 | 4590 | |
| 3 | Tramadol | 250 | 4990 | |
| 4 | Tapentadol | 409 | 764 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shenoy, P.; D’Souza, J.; Rao, M.; Chokkadi, S.; Salins, N. Mathematical Models to Compare the Pharmacokinetics of Methadone, Buprenorphine, Tramadol, and Tapentadol. Eng. Proc. 2023, 59, 55. https://doi.org/10.3390/engproc2023059055
Shenoy P, D’Souza J, Rao M, Chokkadi S, Salins N. Mathematical Models to Compare the Pharmacokinetics of Methadone, Buprenorphine, Tramadol, and Tapentadol. Engineering Proceedings. 2023; 59(1):55. https://doi.org/10.3390/engproc2023059055
Chicago/Turabian StyleShenoy, Prathvi, Joslin D’Souza, Mahadev Rao, Shreesha Chokkadi, and Naveen Salins. 2023. "Mathematical Models to Compare the Pharmacokinetics of Methadone, Buprenorphine, Tramadol, and Tapentadol" Engineering Proceedings 59, no. 1: 55. https://doi.org/10.3390/engproc2023059055
APA StyleShenoy, P., D’Souza, J., Rao, M., Chokkadi, S., & Salins, N. (2023). Mathematical Models to Compare the Pharmacokinetics of Methadone, Buprenorphine, Tramadol, and Tapentadol. Engineering Proceedings, 59(1), 55. https://doi.org/10.3390/engproc2023059055






