Nutritional Composition of the Atlantic Seaweeds Ulva rigida, Codium tomentosum, Palmaria palmata and Porphyra purpurea †
Abstract
:1. Introduction
2. Material and Methods
2.1. Sample Preparation
2.2. Proximate Composition
2.2.1. Inorganic Material
2.2.2. Protein Content
2.2.3. Lipids
2.2.4. Fiber and Hydrocarbons
2.3. Organic Acids
2.4. Mineral Content
2.5. Fatty Acid Profile
3. Results and Discussion
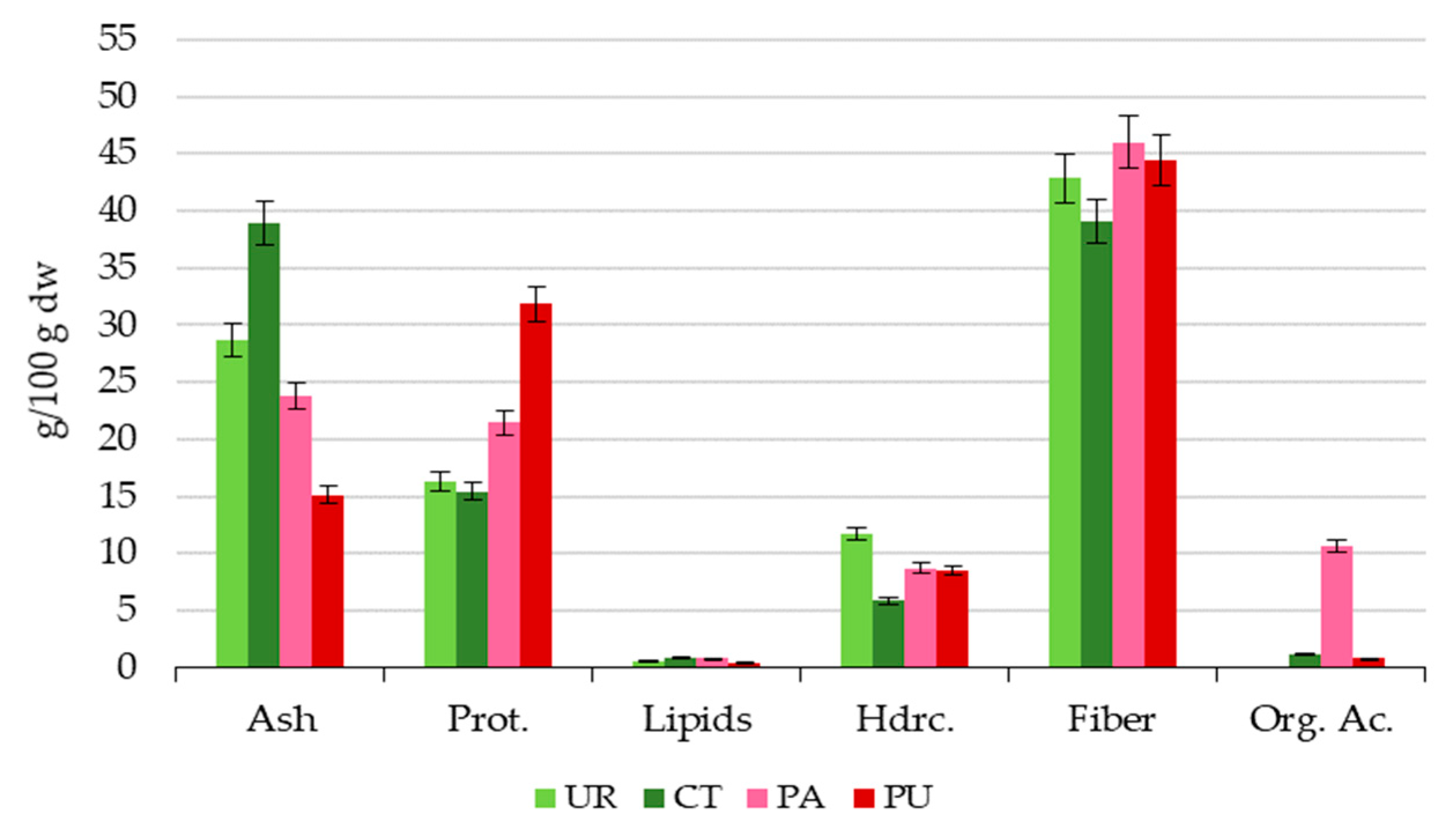
3.1. Proximate Composition
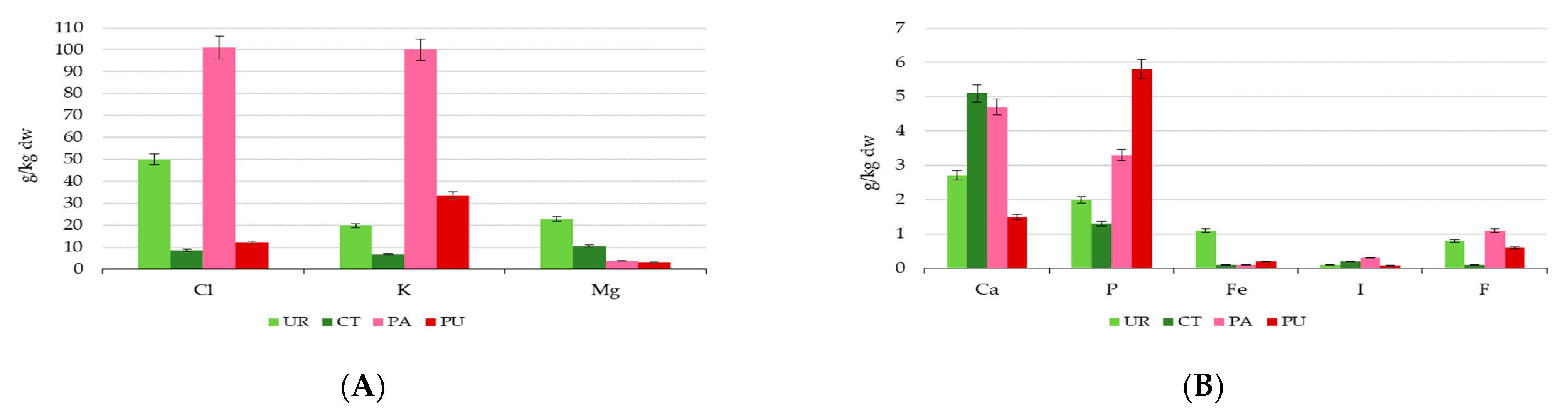
3.2. Mineral Composition
3.3. Fatty Acid Profile
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Martínez-Hernández, G.B.; Castillejo, N.; del M Carrión-Monteagudo, M.; Artés, F.; Artés-Hernández, F. Nutritional and bioactive compounds of commercialized algae powders used as food supplements. Food Sci. Technol. Int. 2018, 24, 172–182. [Google Scholar] [CrossRef] [PubMed]
- Li, B.; Xu, H.; Wang, X.; Wan, Y.; Jiang, N.; Qi, H.; Liu, X. Antioxidant and antihyperlipidemic activities of high sulfate content purified polysaccharide from Ulva pertusa. Int. J. Biol. Macromol. 2020, 146, 756–762. [Google Scholar] [CrossRef] [PubMed]
- Wells, M.L.; Potin, P.; Craigie, J.S.; Raven, J.A.; Merchant, S.S.; Helliwell, K.E.; Smith, A.G.; Camire, M.E.; Brawley, S.H. Algae as nutritional and functional food sources: Revisiting our understanding. J. Appl. Phycol. 2017, 29, 949–982. [Google Scholar] [CrossRef] [PubMed]
- Pliego-Cortés, H.; Wijesekara, I.; Lang, M.; Bourgougnon, N.; Bedoux, G. Current knowledge and challenges in extraction, characterization and bioactivity of seaweed protein and seaweed-derived proteins. In Advances in Botanical Research; Elsevier Ltd.: Amsterdam, The Netherlands, 2020; Volume 95, pp. 289–326. ISBN 9780081027103. [Google Scholar]
- Paiva, L.; Lima, E.; Patarra, R.F.; Neto, A.I.; Baptista, J. Edible Azorean macroalgae as source of rich nutrients with impact on human health. Food Chem. 2014, 164, 128–135. [Google Scholar] [CrossRef] [PubMed]
- Paiva, L.; Lima, E.; Neto, A.I.; Marcone, M.; Baptista, J. Health-promoting ingredients from four selected Azorean macroalgae. Food Res. Int. 2016, 89, 432–438. [Google Scholar] [CrossRef] [PubMed]
- Mæhre, H.K.; Malde, M.K.; Eilertsen, K.E.; Elvevoll, E.O. Characterization of protein, lipid and mineral contents in common Norwegian seaweeds and evaluation of their potential as food and feed. J. Sci. Food Agric. 2014, 94, 3281–3290. [Google Scholar] [CrossRef] [PubMed]
- Mouritsen, O.G.; Dawczynski, C.; Duelund, L.; Jahreis, G.; Vetter, W.; Schröder, M. On the human consumption of the red seaweed dulse (Palmaria palmata (L.) Weber & Mohr). J. Appl. Phycol. 2013, 25, 1777–1791. [Google Scholar] [CrossRef]
- Association of Official Chemistry (AOAC). Official Methods of Analysis of the Association of Official Analytical Chemists, 16th ed.; AOAC International: Arlington, TX, USA, 1995; Volume 1, ISBN 0935584544. [Google Scholar]
- Bradstreet, R.B. Kjeldahl Method for Organic Nitrogen. Anal. Chem. 1954, 26, 185–187. [Google Scholar] [CrossRef]
- Peinado, I.; Girón, J.; Koutsidis, G.; Ames, J.M. Chemical composition, antioxidant activity and sensory evaluation of five different species of brown edible seaweeds. Food Res. Int. 2014, 66, 36–44. [Google Scholar] [CrossRef] [Green Version]
- Jayakody, M.M.; Vanniarachchy, M.P.G.; Wijesekara, W.L.I. Development and characterization of a seaweed snack using Ulva fasciata. J. Food Sci. Technol. 2021, 58, 1617–1622. [Google Scholar] [CrossRef] [PubMed]
- Taboada, M.C.; Millán, R.; Miguez, M.I. Nutritional value of the marine algae wakame (Undaria pinnatifida) and nori (Porphyra purpurea) as food supplements. J. Appl. Phycol. 2013, 25, 1271–1276. [Google Scholar] [CrossRef]
- Rubio, C.; Napoleone, G.; Luis-González, G.; Gutiérrez, A.J.; González-Weller, D.; Hardisson, A.; Revert, C. Metals in edible seaweed. Chemosphere 2017, 173, 572–579. [Google Scholar] [CrossRef] [PubMed]
- Darias-Rosales, J.; Rubio, C.; Gutiérrez, Á.J.; Paz, S.; Hardisson, A. Risk assessment of iodine intake from the consumption of red seaweeds (Palmaria palmata and Chondrus crispus). Environ. Sci. Pollut. Res. 2020, 27, 45737–45741. [Google Scholar] [CrossRef] [PubMed]
- Taboada, C.; Millan, R.; Miguez, I. Evaluation of marine algae Undaria pinnatifida and Porphyra purpurea as a food supplement: Composition, nutritional value and effect of intake on intestinal, hepatic and renal enzyme activities in rats. J. Sci. Food Agric. 2013, 93, 1863–1868. [Google Scholar] [CrossRef] [PubMed]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Echave, J.; Lourenço-Lopes, C.; Carreira-Casais, A.; Chamorro, F.; Fraga-Corral, M.; Otero, P.; Garcia-Perez, P.; Baamonde, S.; Fernández-Saa, F.; Cao, H.; et al. Nutritional Composition of the Atlantic Seaweeds Ulva rigida, Codium tomentosum, Palmaria palmata and Porphyra purpurea. Chem. Proc. 2021, 5, 67. https://doi.org/10.3390/CSAC2021-10681
Echave J, Lourenço-Lopes C, Carreira-Casais A, Chamorro F, Fraga-Corral M, Otero P, Garcia-Perez P, Baamonde S, Fernández-Saa F, Cao H, et al. Nutritional Composition of the Atlantic Seaweeds Ulva rigida, Codium tomentosum, Palmaria palmata and Porphyra purpurea. Chemistry Proceedings. 2021; 5(1):67. https://doi.org/10.3390/CSAC2021-10681
Chicago/Turabian StyleEchave, Javier, Catarina Lourenço-Lopes, Anxo Carreira-Casais, Franklin Chamorro, Maria Fraga-Corral, Paz Otero, Pascual Garcia-Perez, Sergio Baamonde, Fermín Fernández-Saa, Hui Cao, and et al. 2021. "Nutritional Composition of the Atlantic Seaweeds Ulva rigida, Codium tomentosum, Palmaria palmata and Porphyra purpurea" Chemistry Proceedings 5, no. 1: 67. https://doi.org/10.3390/CSAC2021-10681
APA StyleEchave, J., Lourenço-Lopes, C., Carreira-Casais, A., Chamorro, F., Fraga-Corral, M., Otero, P., Garcia-Perez, P., Baamonde, S., Fernández-Saa, F., Cao, H., Xiao, J., Prieto, M. A., & Simal-Gandara, J. (2021). Nutritional Composition of the Atlantic Seaweeds Ulva rigida, Codium tomentosum, Palmaria palmata and Porphyra purpurea. Chemistry Proceedings, 5(1), 67. https://doi.org/10.3390/CSAC2021-10681












