6-Amino-4-Aryl-3-Carbamoyl-5-Cyano-1,4-Dihydropyridine-2-Thiolates: Synthesis, Reactions and Docking Studies †
Abstract
:1. Introduction
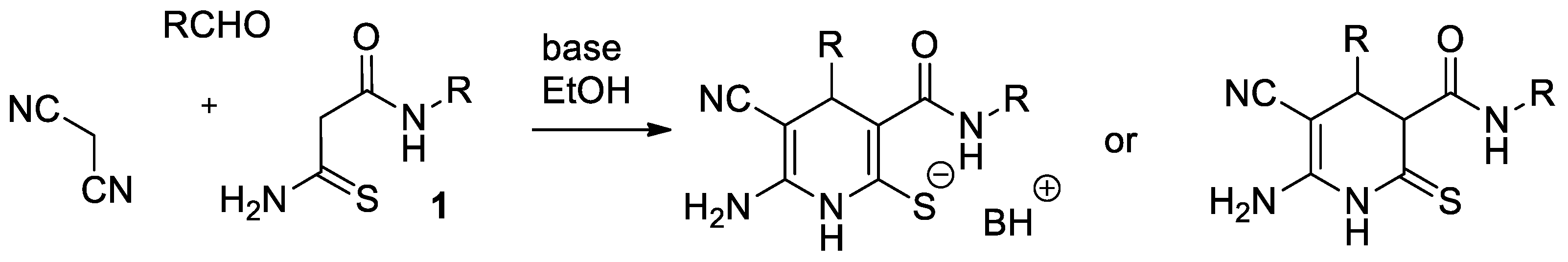
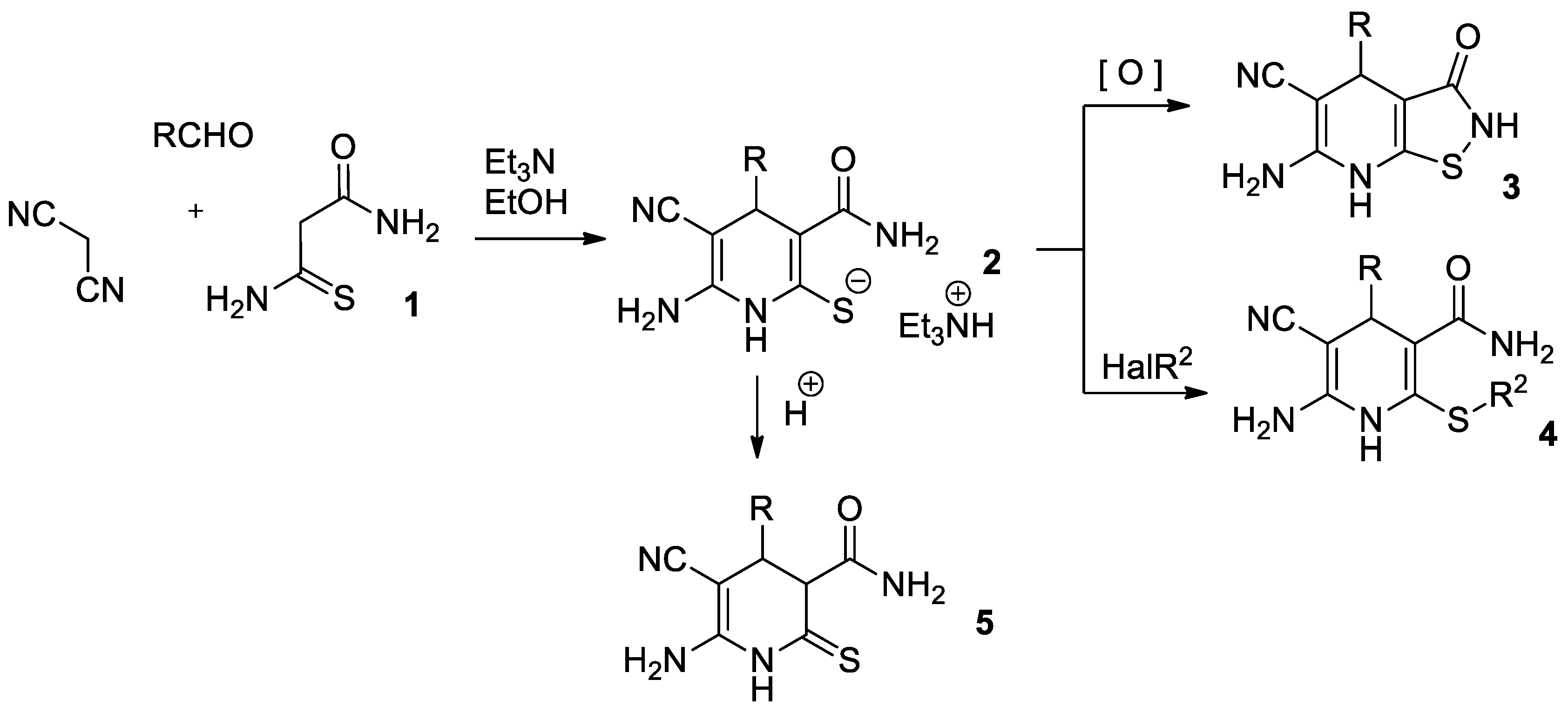
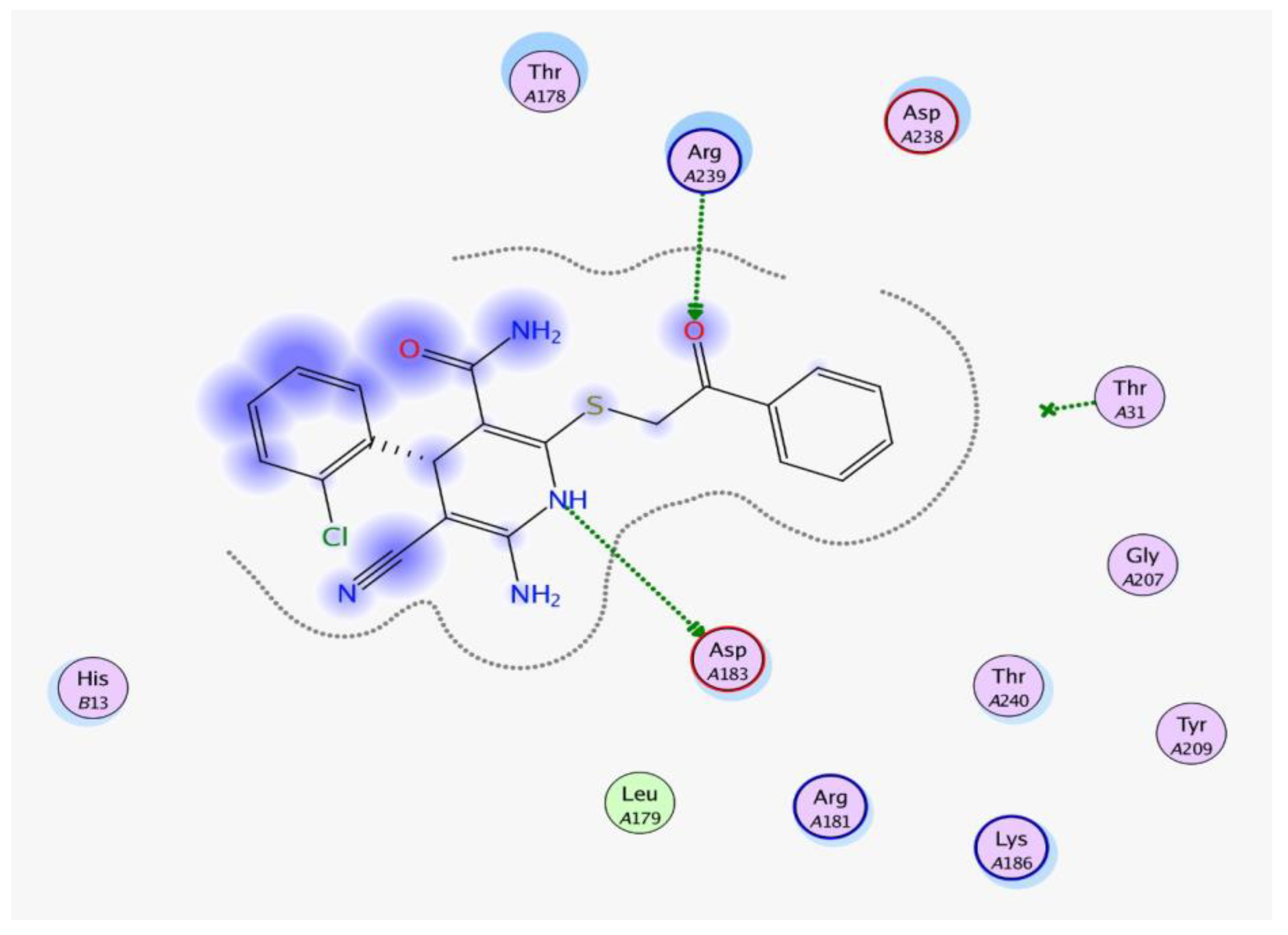
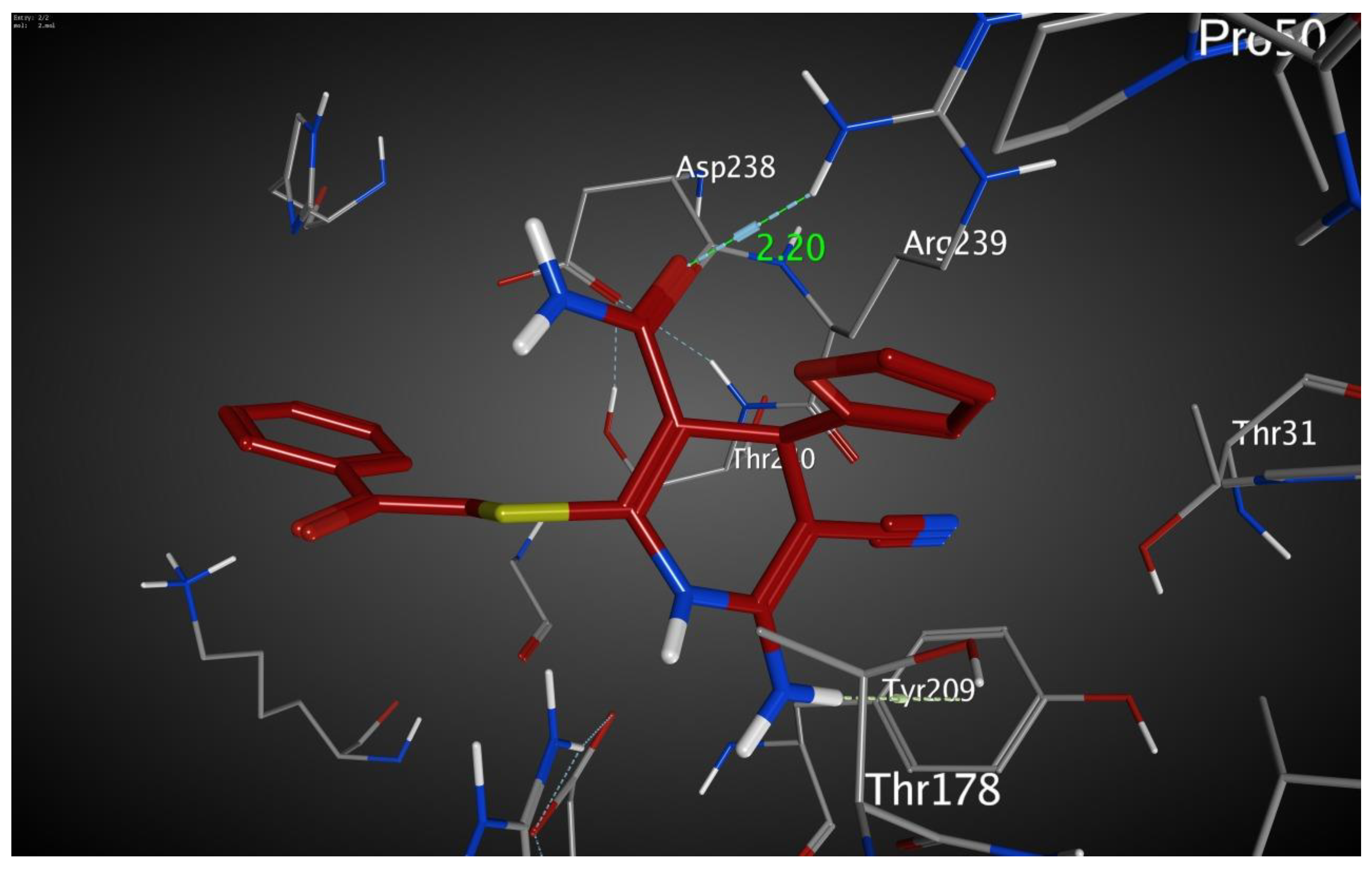
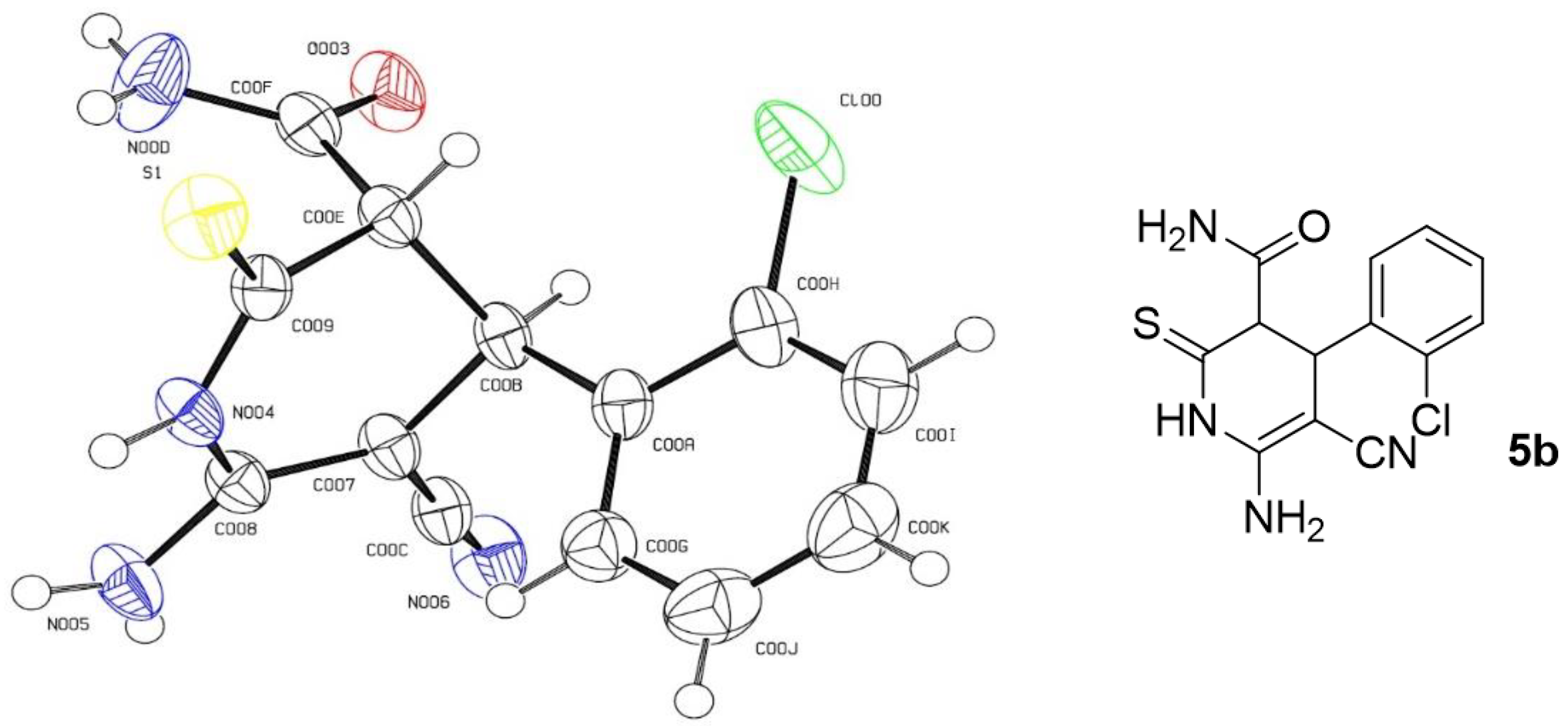
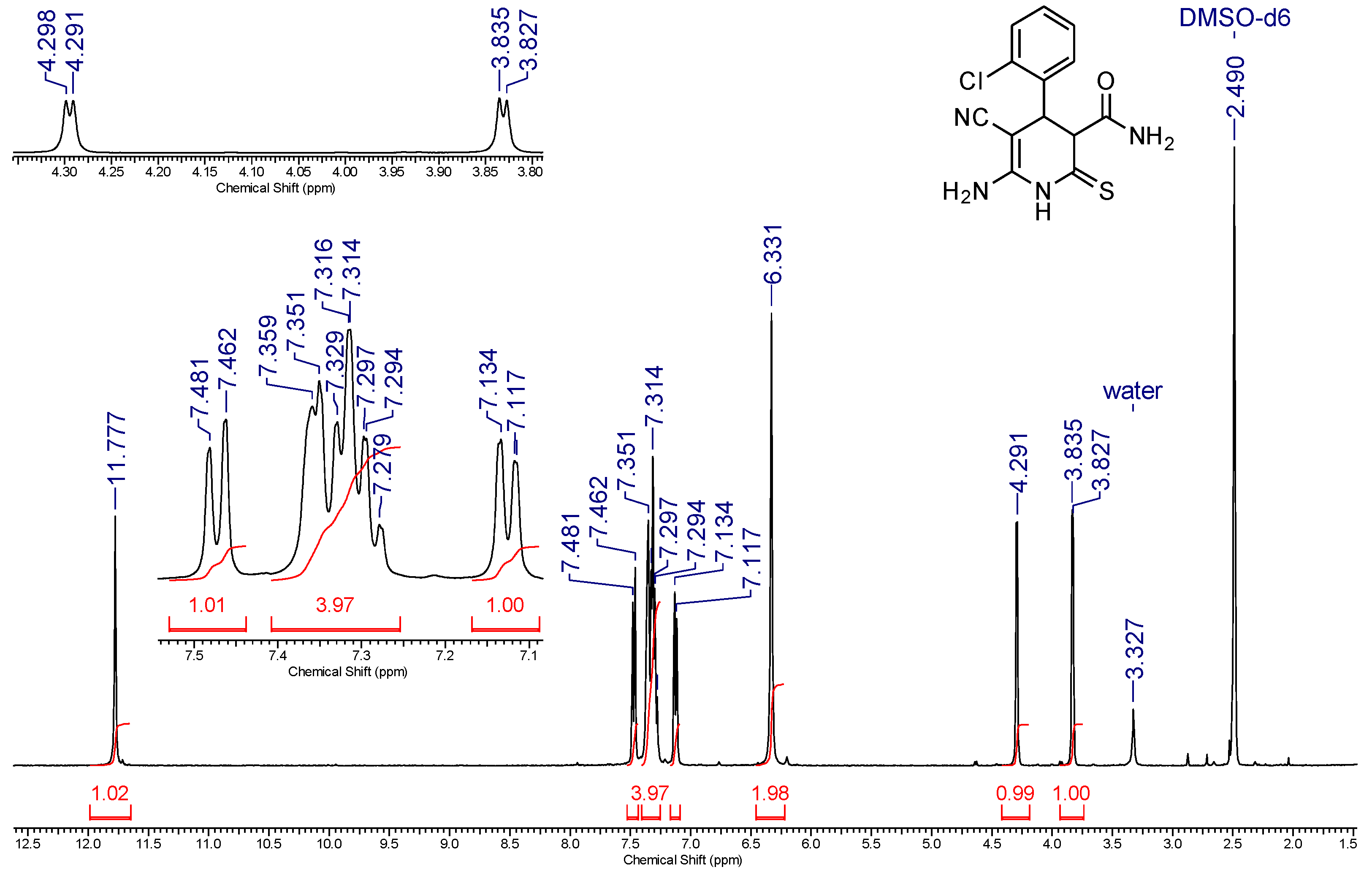
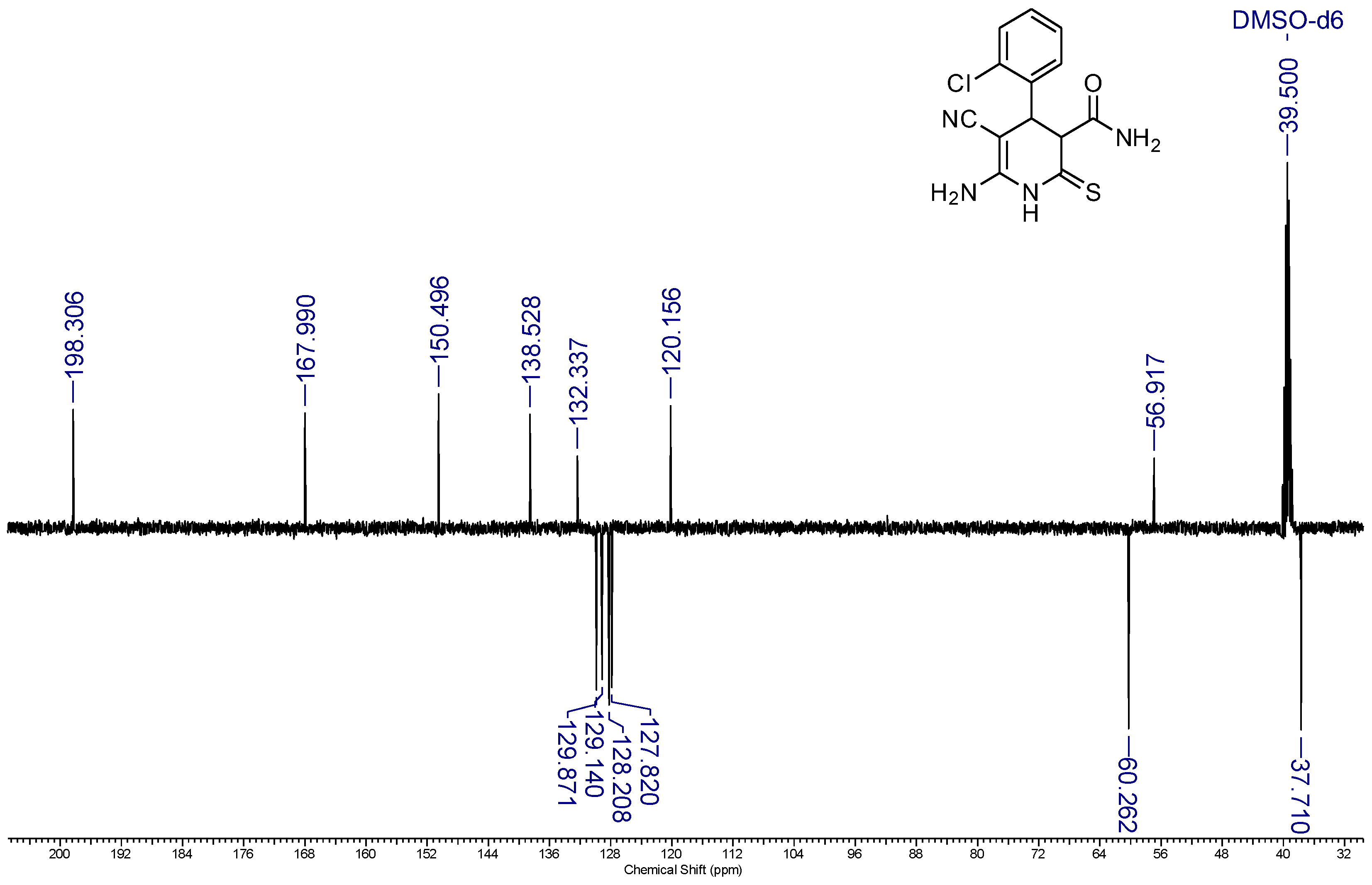
2. Results and Discussion
3. Experimental
3.1. Preparation of Triethylammonium 6-Amino-4-aryl-3-carbamoyl-5-cyano-1,4-dihydropyridine-2-thiolates 2. General Procedure
3.2. Preparation of Compounds 5. General Procedure
Author Contributions
Funding
Conflicts of Interest
References
- Safak, C.; Simsek, R. Fused 1,4-dihydropyridines as potential calcium modulatory compounds. Mini Rev. Med. Chem. 2006, 6, 747–755. [Google Scholar] [CrossRef] [PubMed]
- Hilgeroth, A. Dimeric 4-Aryl-1,4-dihydropyridines: Development of a third class of nonpeptidic HIV-1 protease inhibitors. Mini Rev. Med. Chem. 2002, 2, 235–245. [Google Scholar] [CrossRef] [PubMed]
- Triggle, D.J. 1, 4-Dihydropyridines as calcium channel ligands and privileged structures. Cell. Mol. Neurobiol. 2003, 23, 293–303. [Google Scholar] [CrossRef] [PubMed]
- Triggle, D.J.; Langs, D.A.; Janis, R.A. Ca2+ channel ligands: Structure-function relationships of the 1,4-dihydropyridines. Med. Res. Rev. 1989, 9, 123–180. [Google Scholar] [CrossRef] [PubMed]
- Wan, J.P.; Liu, Y. Recent advances in new multicomponent synthesis of structurally diversified 1,4-dihydropyridines. RSC Adv. 2012, 2, 9763–9777. [Google Scholar] [CrossRef]
- Sharma, V.K.; Singh, S.K. Synthesis, utility and medicinal importance of 1,2-& 1,4-dihydropyridines. RSC Adv. 2017, 7, 2682–2732. [Google Scholar] [CrossRef]
- Triggle, D.J. The 1,4-dihydropyridine nucleus: A pharmacophoric template part 1. Actions at ion channels. Mini Rev. Med. Chem. 2003, 3, 215–223. [Google Scholar] [CrossRef] [PubMed]
- Krauze, A.; Popelis, J.; Duburs, G. Efficient regioselective one-pot synthesis of partially hydrogenated thiazolo [3, 2-a] pyridines. Tetrahedron 1998, 54, 9161–9168. [Google Scholar] [CrossRef]
- Krauze, A.; Liepinsh, E.; Vilums, M.; Duburs, G. A new regioselective synthesis of 2-methoxymethyl-3-oxo-2,3-dihydro-7H-thiazolo [3,2-a]pyridine-8-carboxylic acid methylamide. Chem. Heterocycl. Compd. 2009, 45, 250–252. [Google Scholar] [CrossRef]
- Krauze, A.; Viļums, M.; Sīle, L.; Duburs, G. Alternative products in one-pot reaction of benzylidenemalononitrile, N-methyl-2-thiocarbamoylacetamide, and ω-bromoacetophenone. Chem. Heterocycl. Compd. 2007, 43, 653–657. [Google Scholar] [CrossRef]
- Krauze, A.; Vilums, M.; Sīle, L.; Duburs, G. Alternative Products in One-Pot Reaction of Benzylidene-Malononitrile, Thiocarbamoylacetamide And Alkyl Halides. Heterocycl. Commun. 2009, 15, 239–244. [Google Scholar] [CrossRef]
- Dotsenko, V.V.; Krivokolysko, S.G.; Litvinov, V.P. The Mannich reaction in the synthesis of N, S-containing heterocycles 12. First example of aminomethylation involving 2-thioxonicotinamide derivative: Synthesis of 3,5,7,11-tetraazatricyclo [7.3.1.02,7]tridec-2-ene-9-carboxamides. Russ. Chem. Bull. 2012, 61, 136–140. [Google Scholar] [CrossRef]



Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Jassim, N.T.; Dotsenko, V.V.; Aksenov, N.A. 6-Amino-4-Aryl-3-Carbamoyl-5-Cyano-1,4-Dihydropyridine-2-Thiolates: Synthesis, Reactions and Docking Studies. Chem. Proc. 2021, 3, 13. https://doi.org/10.3390/ecsoc-24-08394
Jassim NT, Dotsenko VV, Aksenov NA. 6-Amino-4-Aryl-3-Carbamoyl-5-Cyano-1,4-Dihydropyridine-2-Thiolates: Synthesis, Reactions and Docking Studies. Chemistry Proceedings. 2021; 3(1):13. https://doi.org/10.3390/ecsoc-24-08394
Chicago/Turabian StyleJassim, Nawras T., Victor V. Dotsenko, and Nicolai A. Aksenov. 2021. "6-Amino-4-Aryl-3-Carbamoyl-5-Cyano-1,4-Dihydropyridine-2-Thiolates: Synthesis, Reactions and Docking Studies" Chemistry Proceedings 3, no. 1: 13. https://doi.org/10.3390/ecsoc-24-08394
APA StyleJassim, N. T., Dotsenko, V. V., & Aksenov, N. A. (2021). 6-Amino-4-Aryl-3-Carbamoyl-5-Cyano-1,4-Dihydropyridine-2-Thiolates: Synthesis, Reactions and Docking Studies. Chemistry Proceedings, 3(1), 13. https://doi.org/10.3390/ecsoc-24-08394





