Type 2 Diabetes and Impaired Physical Function: A Growing Problem
Abstract
:1. Physical Function and Frailty
2. The Phenotype of Frailty in Type 2 Diabetes
3. Wider Recognition of Impaired Physical Function form Health Experts
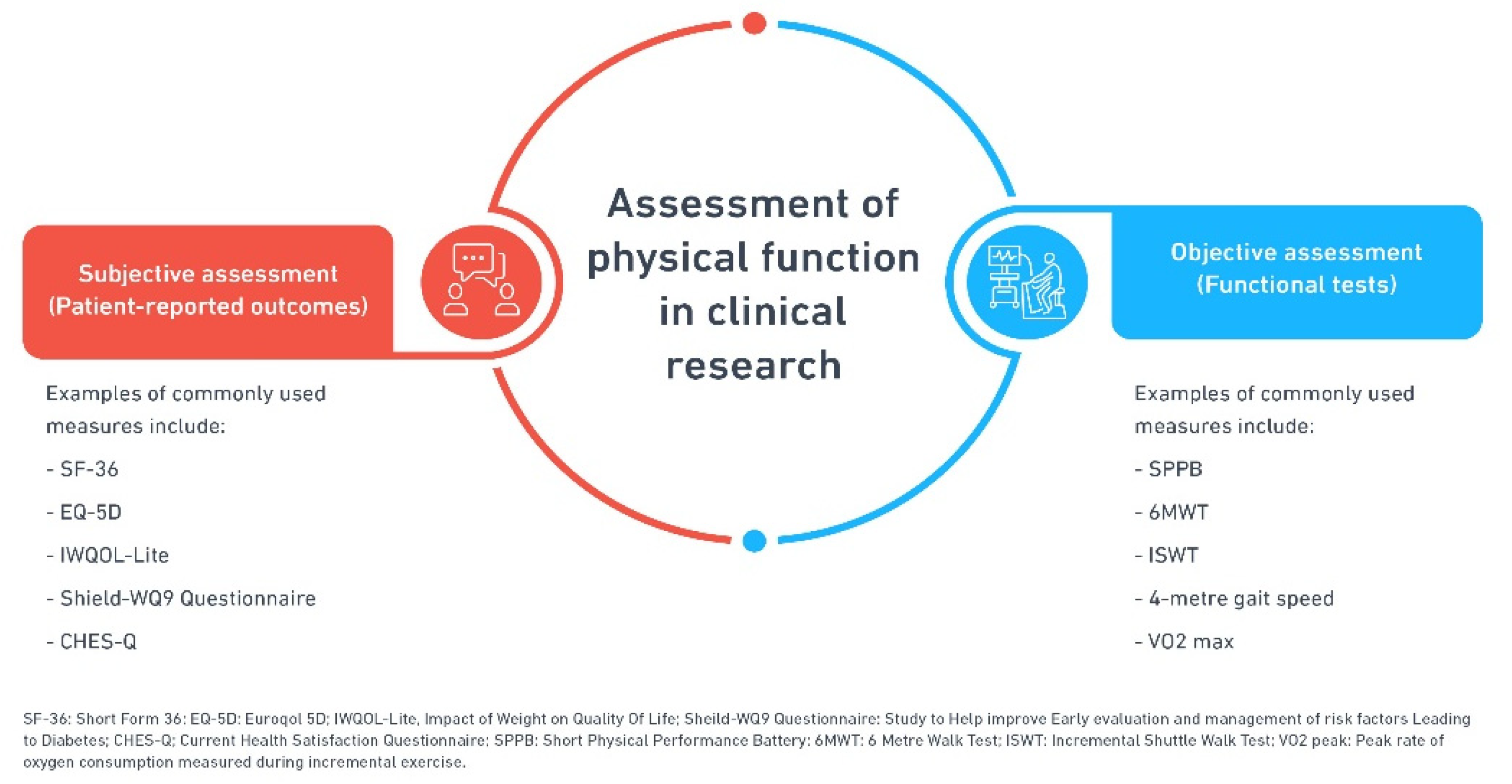
4. Assessment of Physical Function
4.1. Assessment of Physical Function in Clinical Practice
4.2. Assessment of Physical Function in Clinical Trials
5. The Importance of Physical Function in People with T2D
5.1. High Prevalence of Impaired Physical Function across All Age Groups
5.2. Physical Function Is a Dynamic Process
5.3. Adverse Health Outcomes Associated with Decline in Physical Function
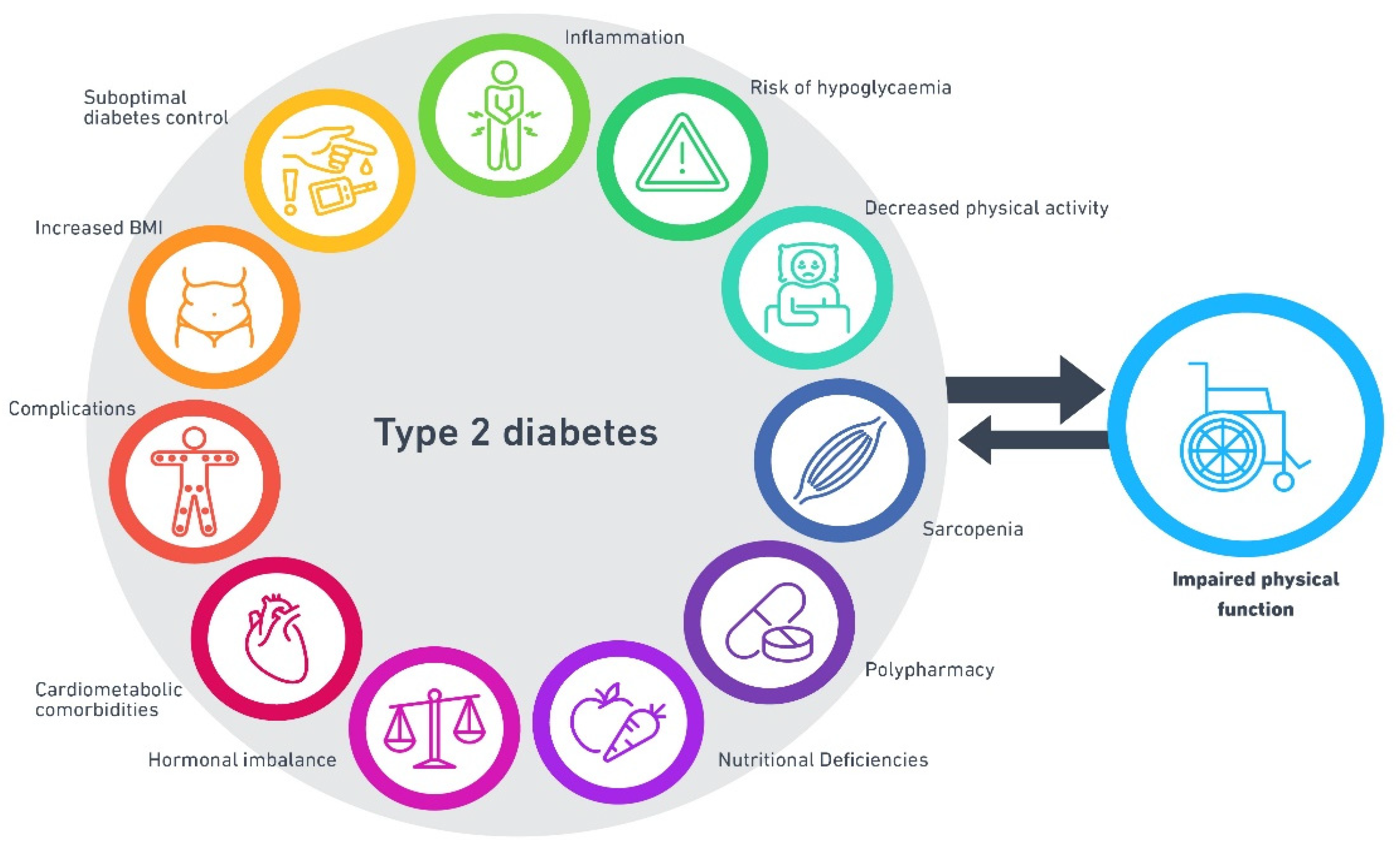
6. Mechanisms Underlying Reduced Physical Function in T2D
6.1. Multimorbidity in T2D
6.2. Sarcopenia and T2D
7. Interventions to Improve Physical Function
7.1. Role of Exercise
7.2. Controlling the Risk Factors
7.3. Physical Function and Novel Glucose Lowering Agents
7.4. Interplay between Exercise and Novel GLTs
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Painter, P.; Stewart, A.L.; Carey, S. Physical Functioning: Definitions, Measurement, and Expectations. Adv. Ren. Replace. Ther. 1999, 6, 110–123. [Google Scholar] [CrossRef]
- Garber, C.E.; Greaney, M.L.; Riebe, D.; Nigg, C.R.; Burbank, P.A.; Clark, P.G. Physical and mental health-related correlates of physical function in community dwelling older adults: A cross sectional study. BMC Geriatr. 2010, 10, 6–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bergman, H.; Ferrucci, L.; Guralnik, J.; Hogan, D.; Hummel, S.; Karunananthan, S.; Wolfson, C. Frailty: An Emerging Research and Clinical Paradigm—Issues and Controversies. J. Gerontol. Ser. A 2007, 62, 731–737. [Google Scholar] [CrossRef] [PubMed]
- Fried, L.P.; Tangen, C.M.; Walston, J.; Newman, A.B.; Hirsch, C.; Gottdiener, J.; Seeman, T.; Tracy, R.; Kop, W.J.; Burke, G.; et al. Frailty in Older adults: Evidence for a phenotype. J. Gerontol. A Biol. Sci. Med. Sci. 2001, 56, M146–M156. [Google Scholar] [CrossRef]
- Fried, L.P.; Ferrucci, L.; Darer, J.; Williamson, J.D.; Anderson, G. Untangling the Concepts of Disability, Frailty, and Comorbidity: Implications for Improved Targeting and Care. J. Gerontol. Ser. A Biol. Sci. Med. Sci. 2004, 59, M255–M263. [Google Scholar] [CrossRef] [Green Version]
- Chalé-Rush, A.; Guralnik, J.M.; Walkup, M.P.; Miller, M.E.; Rejeski, W.J.; Katula, J.A.; King, A.C.; Glynn, N.W.; Manini, T.M.; Blair, S.N.; et al. Relationship Between Physical Functioning and Physical Activity in the Lifestyle Interventions and Independence for Elders Pilot. J. Am. Geriatr. Soc. 2010, 58, 1918–1924. [Google Scholar] [CrossRef] [Green Version]
- Magliano, D.J.; Sacre, J.W.; Harding, J.L.; Gregg, E.W.; Zimmet, P.Z.; Shaw, J.E. Young-onset type 2 diabetes mellitus—Implications for morbidity and mortality. Nat. Rev. Endocrinol. 2020, 16, 321–331. [Google Scholar] [CrossRef]
- Volpato, S.; Maraldi, C.; Fellin, R. Type 2 diabetes and risk for functional decline and disability in older persons. Curr. Diabetes Rev. 2010, 6, 134–143. [Google Scholar] [CrossRef]
- Huang, E.S.; Gorawara-Bhat, R.; Chin, M.H. Self-Reported Goals of Older Patients with Type 2 Diabetes Mellitus. J. Am. Geriatr. Soc. 2005, 53, 306–311. [Google Scholar] [CrossRef] [Green Version]
- NICE. New QOF Indicators Added to Menu by NICE. 2018. Available online: https://diabetestimes.co.uk/new-qof-indicators-added-to-menu-by-nice/ (accessed on 21 June 2021).
- Group PCSaNC. 2019/20 General Medical Services (GMS) contract Quality and Outcomes Framework (QOF) Guidance for GMS contract 2019/20 in Englan. BMA 2019. Available online: https://www.england.nhs.uk/wp-content/uploads/2019/05/gms-contract-qof-guidance-april-2019.pdf (accessed on 22 June 2021).
- Dunning, T.; Sinclair, A.; Colagiuri, S. New IDF Guideline for managing type 2 diabetes in older people. Diabetes Res. Clin. Pract. 2014, 103, 538–540. [Google Scholar] [CrossRef]
- Sinclair, A.J.; Abdelhafiz, A.; Dunning, T.; Izquierdo, M.; Manas, L.R.; Bourdel-Marchasson, I.; Morley, J.E.; Munshi, M.; Woo, J.; Vellas, B. An international position statement on the management of frailty in diabetes mellitus: Summary of recommendations 2017. J. Frailty Aging 2018, 7, 1–11. [Google Scholar] [CrossRef]
- Sinclair, A.; Gallaher, A. Managing Frailty and Associated Comorbidities in Older Adults with Diabetes: Position Statement on behalf of the Association of British Clinical Diabetologists (ABCD). Available online: https://abcd.care/sites/abcd.care/files/site_uploads/Resources/Position-Papers/ABCD-Position-Paper-Frailty.pdf. (accessed on 22 June 2021).
- Strain, W.D.; Down, S.; Brown, P.; Puttanna, A.; Sinclair, A. Diabetes and Frailty: An Expert Consensus Statement on the Management of Older Adults with Type 2 Diabetes. Diabetes Ther. 2021, 12, 1227–1247. [Google Scholar] [CrossRef]
- Castro-Rodríguez, M.; Carnicero, J.A.; Garcia-Garcia, F.J.; Walter, S.; Morley, J.E.; Rodríguez-Artalejo, F.; Sinclair, A.J.; Rodríguez-Mañas, L. Frailty as a Major Factor in the Increased Risk of Death and Disability in Older People with Diabetes. J. Am. Med. Dir. Assoc. 2016, 17, 949–955. [Google Scholar] [CrossRef]
- Sinclair, A.; Dunning, T.; Rodríguez-Mañas, L. Diabetes in older people: New insights and remaining challenges. Lancet Diabetes Endocrinol. 2015, 3, 275–285. [Google Scholar] [CrossRef]
- Strain, W.D.; Hope, S.; Green, A.; Kar, P.; Valabhji, J.; Sinclair, A.J. Type 2 diabetes mellitus in older people: A brief statement of key principles of modern day management including the assessment of frailty. A national collaborative stakeholder initiative. Diabet. Med. 2018, 35, 838–845. [Google Scholar] [CrossRef] [Green Version]
- NHS Electronic Frailty Index. Available online: https://www.england.nhs.uk/ourwork/clinical-policy/older-people/frailty/efi/. (accessed on 22 June 2021).
- Sinclair, A. (September 2019) Key Learning Points: Diabetes in Older People with Frailty. Available online: https://www.guidelinesinpractice.co.uk/diabetes/key-learning-points-diabetes-in-older-people-with-frailty/454910.article (accessed on 22 June 2021).
- Treacy, D.; Hassett, L. The Short Physical Performance Battery. J. Physiother. 2018, 64, 61. [Google Scholar] [CrossRef] [PubMed]
- Maggio, M.; Ceda, G.P.; Ticinesi, A.; De Vita, F.; Gelmini, G.; Costantino, C.; Meschi, T.; Kressig, R.W.; Cesari, M.; Fabi, M.; et al. Instrumental and Non-Instrumental Evaluation of 4-Meter Walking Speed in Older Individuals. PLoS ONE 2016, 11, e0153583. [Google Scholar] [CrossRef] [PubMed]
- Barry, E.; Galvin, R.; Keogh, C.; Horgan, F.; Fahey, T. Is the Timed Up and Go test a useful predictor of risk of falls in community dwelling older adults: A systematic review and meta- analysis. BMC Geriatr. 2014, 14, 14. [Google Scholar] [CrossRef] [PubMed]
- Nicolini-Panisson, R.D.; Donadio, M. Timed “Up & Go” test in children and adolescents. Rev. Paul. Pediatr. 2013, 31, 377–383. [Google Scholar] [CrossRef]
- Yates, T.; Zaccardi, F.; Dhalwani, N.; Davies, M.; Bakrania, K.; Celis-Morales, C.A.; Gill, J.M.R.; Franks, P.; Khunti, K. Association of walking pace and handgrip strength with all-cause, cardiovascular, and cancer mortality: A UK Biobank observational study. Eur. Heart J. 2017, 38, 3232–3240. [Google Scholar] [CrossRef]
- Timmins, I.R.; Zaccardi, F.; Nelson, C.P.; Franks, P.W.; Yates, T.; Dudbridge, F. Genome-wide association study of self-reported walking pace suggests beneficial effects of brisk walking on health and survival. Commun. Biol. 2020, 3, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Argyridou, S.; Zaccardi, F.; Davies, M.; Khunti, K.; Yates, T. Walking pace improves all-cause and cardiovascular mortality risk prediction: A UK Biobank prognostic study. Eur. J. Prev. Cardiol. 2020, 27, 1036–1044. [Google Scholar] [CrossRef] [PubMed]
- Zaccardi, F.; Davies, M.; Khunti, K.; Yates, T. Comparative Relevance of Physical Fitness and Adiposity on Life Expectancy: A UK Biobank Observational Study. Mayo Clin. Proc. 2019, 94, 985–994. [Google Scholar] [CrossRef] [PubMed]
- Bravata, D.M.; Smith-Spangler, C.; Sundaram, V.; Gienger, A.L.; Lin, N.; Lewis, R.; Stave, C.D.; Olkin, I.; Sirard, J.R. Using Pedometers to Increase Physical Activity and Improve Health: A systematic review. J. Am. Med. Assoc. 2007, 298, 2296–2304. [Google Scholar] [CrossRef]
- Van Kan, G.A.; Rolland, Y.; Bergman, H.; Morley, J.E.; Kritchevsky, S.B.; Vellas, B. The I.A.N.A. task force on frailty assessment of older people in clinical practice. J. Nutr. Health Aging 2008, 12, 29–37. [Google Scholar] [CrossRef]
- Morley, J.E.; Vellas, B.; Van Kan, G.A.; Anker, S.D.; Bauer, J.M.; Bernabei, R.; Cesari, M.; Chumlea, W.; Doehner, W.; Evans, J.; et al. Frailty Consensus: A Call to Action. J. Am. Med. Dir. Assoc. 2013, 14, 392–397. [Google Scholar] [CrossRef] [Green Version]
- Lins, L.; Carvalho, F. SF-36 total score as a single measure of health-related quality of life: Scoping review. SAGE Open Med. 2016, 4. [Google Scholar] [CrossRef] [Green Version]
- Balestroni, G.; Bertolotti, G. EuroQol-5D (EQ-5D): An instrument for measuring quality of life. Monaldi Arch. Chest Dis. 2012, 78, 155–159. [Google Scholar] [CrossRef]
- Grandy, S.; Fox, K.M.; Bazata, D.D.; for the SHIELD Study Group. Health-related quality of life association with weight change in type 2 diabetes mellitus: Perception vs. reality. Int. J. Clin. Pract. 2013, 67, 455–461. [Google Scholar] [CrossRef]
- Traina, S.B.; Colwell, H.H.; Crosby, R.D.; Mathias, S.D. Pragmatic measurement of health satisfaction in people with type 2 diabetes mellitus using the Current Health Satisfaction Questionnaire. Patient Relat. Outcome Meas. 2015, 6, 103–115. [Google Scholar] [CrossRef] [Green Version]
- Kolotkin, R.L.; Williams, V.S.L.; Ervin, C.M.; Williams, N.; Meincke, H.H.; Qin, S.; Smith, L.V.H.; Fehnel, S. Validation of a new measure of quality of life in obesity trials: Impact of Weight on Quality of Life-Lite Clinical Trials Version. Clin. Obes. 2019, 9, e12310. [Google Scholar] [CrossRef]
- Villareal, D.T.; Chode, S.; Parimi, N.; Sinacore, D.R.; Hilton, T.; Armamento-Villareal, R.; Napoli, N.; Qualls, C.; Shah, K. Weight Loss, Exercise, or Both and Physical Function in Obese Older Adults. N. Engl. J. Med. 2011, 364, 1218–1229. [Google Scholar] [CrossRef] [Green Version]
- Gregg, E.W.; Beckles, G.L.; Williamson, D.F.; Leveille, S.G.; Langlois, J.A.; Engelgau, M.M.; Narayan, K.M. Diabetes and physical disability among older U.S. adults. Diabetes Care 2000, 23, 1272–1277. [Google Scholar] [CrossRef] [Green Version]
- Enright, P.L. The six-minute walk test. Respir Care 2003, 48, 783–785. [Google Scholar]
- Costa, I.P.; Corso, S.D.; Borghi-Silva, A.; Peixoto, F.; Stirbulov, R.; Arena, R.; Cahalin, L.P.; Sampaio, L.M.M. Reliability of the Shuttle Walk Test with Controlled Incremental Velocity in Patients with Difficult-to-Control Asthma. J. Cardiopulm. Rehabil. Prev. 2018, 38, 54–57. [Google Scholar] [CrossRef] [PubMed]
- Betik, A.C.; Hepple, R.T. Determinants ofVO2 maxdecline with aging: An integrated perspective. Appl. Physiol. Nutr. Metab. 2008, 33, 130–140. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Fitchett, M.A. Predictability of VO2 max from submaximal cycle ergometer and bench stepping tests. Br. J. Sports Med. 1985, 19, 85–88. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Laucis, N.C.; Hays, R.D.; Bhattacharyya, T. Scoring the SF-36 in Orthopaedics: A Brief Guide. J. Bone Jt. Surg. 2015, 97, 1628–1634. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Wong, E.; Backholer, K.; Gearon, E.; Harding, J.; Freak-Poli, R.; Stevenson, C.; Peeters, A. Diabetes and risk of physical disability in adults: A systematic review and meta-analysis. Lancet Diabetes Endocrinol. 2013, 1, 106–114. [Google Scholar] [CrossRef] [Green Version]
- Morley, J.E.; Malmstrom, T.K.; Rodríguez-Mañas, L.; Sinclair, A.J. Frailty, Sarcopenia and Diabetes. J. Am. Med. Dir. Assoc. 2014, 15, 853–859. [Google Scholar] [CrossRef] [PubMed]
- Hanlon, P.; Nicholl, B.I.; Jani, B.D.; Lee, D.; McQueenie, R.; Mair, F.S. Frailty and pre-frailty in middle-aged and older adults and its association with multimorbidity and mortality: A prospective analysis of 493 737 UK Biobank participants. Lancet Public Health 2018, 3, e323. [Google Scholar] [CrossRef]
- Hubbard, R.E.; Andrew, M.K.; Fallah, N.; Rockwood, K. Comparison of the prognostic importance of diagnosed diabetes, co-morbidity and frailty in older people. Diabet. Med. 2010, 27, 603–606. [Google Scholar] [CrossRef] [PubMed]
- Ryerson, B.; Tierney, E.F.; Thompson, T.J.; Engelgau, M.M.; Wang, J.; Gregg, E.W.; Geiss, L.S. Excess physical limitations among adults with diabetes in the U.S. population, 1997–1999. Diabetes Care 2003, 26, 206–210. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kalyani, R.R.; Saudek, C.D.; Brancati, F.L.; Selvin, E. Association of Diabetes, Comorbidities, and A1C With Functional Disability in Older Adults: Results from the National Health and Nutrition Examination Survey (NHANES), 1999–2006. Diabetes Care 2010, 33, 1055–1060. [Google Scholar] [CrossRef] [Green Version]
- Godino, J.G.; Appel, L.J.; Gross, A.L.; Schrack, J.A.; Parrinello, C.M.; Kalyani, R.R.; Windham, B.G.; Pankow, J.S.; Kritchevsky, S.B.; Bandeen-Roche, K.; et al. Diabetes, hyperglycemia, and the burden of functional disability among older adults in a community-based study. J. Diabetes 2017, 9, 76–84. [Google Scholar] [CrossRef] [Green Version]
- Rodríguez-Saldaña, J.; Morley, J.E.; Reynoso, M.T.; Medina, C.A.; Salazar, P.; Cruz, E.; Torres, A.L.N. Diabetes Mellitus in a Subgroup of Older Mexicans: Prevalence, Association with Cardiovascular Risk Factors, Functional and Cognitive Impairment, and Mortality. J. Am. Geriatr. Soc. 2002, 50, 111–116. [Google Scholar] [CrossRef]
- Mickute, M.; Henson, J.; Rowlands, A.V.; Sargeant, J.A.; Webb, D.; Hall, A.P.; Edwardson, C.L.; Baldry, E.L.; Brady, E.M.; Khunti, K.; et al. Device-measured physical activity and its association with physical function in adults with type 2 diabetes mellitus. Diabet. Med. 2021, 38. [Google Scholar] [CrossRef]
- Chhetri, J.K.; Zheng, Z.; Xu, X.; Ma, C.; Chan, P. The prevalence and incidence of frailty in Pre-diabetic and diabetic community-dwelling older population: Results from Beijing longitudinal study of aging II (BLSA-II). BMC Geriatr. 2017, 17, 47. [Google Scholar] [CrossRef] [Green Version]
- Gregg, E.W.; Mangione, C.M.; Cauley, J.A.; Thompson, T.J.; Schwartz, A.V.; Ensrud, K.E.; Nevitt, M.C.; for the Study of Osteoporotic Fractures Research Group. Diabetes and Incidence of Functional Disability in Older Women. Diabetes Care 2002, 25, 61–67. [Google Scholar] [CrossRef] [Green Version]
- Sayer, A.A.; Dennison, E.M.; Syddall, H.E.; Gilbody, H.J.; Phillips, D.I.; Cooper, C. Type 2 diabetes, muscle strength, and impaired physical function: The tip of the iceberg? Diabetes Care 2005, 28, 2541–2542. [Google Scholar] [CrossRef] [Green Version]
- Chen, C.Y.; Gan, P.; How, C.H. Approach to frailty in the elderly in primary care and the community. Singap. Med. J. 2018, 59, 240–245. [Google Scholar] [CrossRef] [Green Version]
- Radner, H.; Smolen, J.S.; Aletaha, D. Comorbidity affects all domains of physical function and quality of life in patients with rheumatoid arthritis. Rheumatology 2010, 50, 381–388. [Google Scholar] [CrossRef] [Green Version]
- Chao, C.-T.; COhort of GEriatric Nephrology in NTUH (COGENT) Study Group; Wang, J.; Chien, K.-L. Both pre-frailty and frailty increase healthcare utilization and adverse health outcomes in patients with type 2 diabetes mellitus. Cardiovasc. Diabetol. 2018, 17, 130. [Google Scholar] [CrossRef] [PubMed]
- Wong, E.; Stevenson, C.; Backholer, K.; Woodward, M.; Shaw, J.E.; Peeters, A. Predicting the risk of physical disability in old age using modifiable mid-life risk factors. J. Epidemiol. Community Health 2014, 69, 70–76. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huo, L.; Shaw, J.E.; Wong, E.; Harding, J.L.; Peeters, A.; Magliano, D.J. Burden of diabetes in Australia: Life expectancy and disability-free life expectancy in adults with diabetes. Diabetologia 2016, 59, 1437–1445. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Turnbull, P.J.; Sinclair, A.J. Evaluation of nutritional status and its relationship with functional status in older citizens with diabetes mellitus using the mini nutritional assessment (MNA) tool—A preliminary investigation. J. Nutr. Health Aging 2002, 6, 185–189. [Google Scholar]
- Van Sloten, T.T.; Savelberg, H.H.; Duimel-Peeters, I.G.; Meijer, K.; Henry, R.M.; Stehouwer, C.D.; Schaper, N.C. Peripheral neuropathy, decreased muscle strength and obesity are strongly associated with walking in persons with type 2 diabetes without manifest mobility limitations. Diabetes Res. Clin. Pract. 2011, 91, 32–39. [Google Scholar] [CrossRef]
- Tyrovolas, S.; Koyanagi, A.; Garin, N.; Olaya, B.; Ayuso-Mateos, J.L.; Miret, M.; Chatterji, S.; Tobiasz-Adamczyk, B.; Koskinen, S.; Leonardi, M.; et al. Diabetes mellitus and its association with central obesity and disability among older adults: A global perspective. Exp. Gerontol. 2015, 64, 70–77. [Google Scholar] [CrossRef]
- Atkinson, H.H.; Rosano, C.; Simonsick, E.M.; Williamson, J.D.; Davis, C.; Ambrosius, W.T.; Rapp, S.R.; Cesari, M.; Newman, A.B.; Harris, T.B.; et al. Cognitive Function, Gait Speed Decline, and Comorbidities: The Health, Aging and Body Composition Study. J. Gerontol. Ser. A 2007, 62, 844–850. [Google Scholar] [CrossRef] [Green Version]
- Kirkman, M.S.; Briscoe, V.J.; Clark, N.; Florez, H.; Haas, L.B.; Halter, J.B.; Huang, E.S.; Korytkowski, M.T.; Munshi, M.N.; Odegard, P.S.; et al. Diabetes in Older Adults. Diabetes Care 2012, 35, 2650–2664. [Google Scholar] [CrossRef] [Green Version]
- Cacciatore, F.; Testa, G.; Galizia, G.; Della-Morte, D.; Mazzella, F.; Langellotto, A.; Pirozzi, G.; Ferro, G.; Gargiulo, G.; Ferrara, N.; et al. Clinical frailty and long-term mortality in elderly subjects with diabetes. Acta Diabetol. 2013, 50, 251–260. [Google Scholar] [CrossRef]
- Gadsby, R.; Hope, S.; Hambling, C.A.C. Frailty, older people and type 2 diabetes. J. Diabetes Nurs. 2017, 21, 138–142. [Google Scholar]
- Helmersson, J.; Vessby, B.; Larsson, A.; Basu, S. Association of Type 2 Diabetes with Cyclooxygenase-Mediated Inflammation and Oxidative Stress in an Elderly Population. Circulation 2004, 109, 1729–1734. [Google Scholar] [CrossRef]
- Szewieczek, J.; Bieniek, J.; Wilczyński, K. Fried frailty phenotype assessment components as applied to geriatric inpatients. Clin. Interv. Aging 2016, 11, 453–459. [Google Scholar] [CrossRef] [Green Version]
- Huang, E.S.; Zhang, Q.; Gandra, N.; Chin, M.H.; Meltzer, D.O. The Effect of Comorbid Illness and Functional Status on the Expected Benefits of Intensive Glucose Control in Older Patients with Type 2 Diabetes: A Decision Analysis. Ann. Intern. Med. 2008, 149, 11–19. [Google Scholar] [CrossRef] [Green Version]
- Koye, D.N.; Shaw, J.E.; Magliano, D. Diabetes and disability in older Australians: The Australian Diabetes, Obesity and Lifestyle (AusDiab) study. Diabetes Res. Clin. Pr. 2017, 126, 60–67. [Google Scholar] [CrossRef]
- Maggi, S.; Noale, M.; Gallina, P.; Marzari, C.; Bianchi, D.; Limongi, F.; Crepaldi, G.; For the ILSA Group. Physical disability among older Italians with diabetes. The ILSA Study. Diabetologia 2004, 47, 1957–1962. [Google Scholar] [CrossRef] [Green Version]
- Sinclair, A.J.; Conroy, S.; Bayer, A.J. Impact of Diabetes on Physical Function in Older People. Diabetes Care 2007, 31, 233–235. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cesari, M.; Landi, F.; Vellas, B.; Bernabei, R.; Marzetti, E. Sarcopenia and Physical Frailty: Two Sides of the Same Coin. Front. Aging Neurosci. 2014, 6, 192. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Park, S.W.; Goodpaster, B.H.; Strotmeyer, E.S.; Kuller, L.H.; Broudeau, R.; Kammerer, C.; de Rekeneire, N.; Harris, T.B.; Schwartz, A.V.; Tylavsky, F.A.; et al. Accelerated Loss of Skeletal Muscle Strength in Older Adults with Type 2 Diabetess: The health, aging, and body composition study. Diabetes Care 2007, 30, 1507–1512. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rolland, Y.; Czerwinski, S.; Van Kan, G.A.; Morley, J.E.; Cesari, M.; Onder, G.; Woo, J.; Baumgartner, R.; Pillard, F.; Boirie, Y.; et al. Sarcopenia: Its assessment, etiology, pathogenesis, consequences and future perspectives. J. Nutr. Health Aging 2008, 12, 433–450. [Google Scholar] [CrossRef] [Green Version]
- Landi, F.; Onder, G.; Bernabei, R. Sarcopenia and Diabetes: Two Sides of the Same Coin. J. Am. Med. Dir. Assoc. 2013, 14, 540–541. [Google Scholar] [CrossRef] [PubMed]
- McNeil, C.J.; Doherty, T.J.; Stashuk, D.W.; Rice, C.L. Motor unit number estimates in the tibialis anterior muscle of young, old, and very old men. Muscle Nerve 2005, 31, 461–467. [Google Scholar] [CrossRef] [PubMed]
- Giresi, P.G.; Stevenson, E.J.; Theilhaber, J.; Koncarevic, A.; Parkington, J.; Fielding, R.A.; Kandarian, S.C. Identification of a molecular signature of sarcopenia. Physiol. Genom. 2005, 21, 253–263. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cree-Green, M.; Newcomer, B.R.; Katsanos, C.S.; Sheffield-Moore, M.; Chinkes, D.; Aarsland, A.; Urban, R.; Wolfe, R.R. Intramuscular and Liver Triglycerides Are Increased in the Elderly. J. Clin. Endocrinol. Metab. 2004, 89, 3864–3871. [Google Scholar] [CrossRef]
- Sinclair, A.J.; Abdelhafiz, A.H.; Rodríguez-Mañas, L. Frailty and sarcopenia-newly emerging and high impact complications of diabetes. J. Diabetes Its Complicat. 2017, 31, 1465–1473. [Google Scholar] [CrossRef]
- Sinclair, A.J.; Rodríguez-Mañas, L. Diabetes and Frailty: Two Converging Conditions? Can. J. Diabetes 2016, 40, 77–83. [Google Scholar] [CrossRef] [Green Version]
- Park, S.W.; Goodpaster, B.H.; Lee, J.S.; Kuller, L.H.; Boudreau, R.; de Rekeneire, N.; Harris, T.B.; Kritchevsky, S.; Tylavsky, F.A.; Nevitt, M.; et al. Excessive Loss of Skeletal Muscle Mass in Older Adults with Type 2 Diabetes. Diabetes Care 2009, 32, 1993–1997. [Google Scholar] [CrossRef] [Green Version]
- Kalyani, R.R.; Corriere, M.; Ferrucci, L. Age-related and disease-related muscle loss: The effect of diabetes, obesity, and other diseases. Lancet Diabetes Endocrinol. 2014, 2, 819–829. [Google Scholar] [CrossRef] [Green Version]
- Reusch, J.E.B.; Bridenstine, M.; Regensteiner, J.G. Type 2 diabetes mellitus and exercise impairment. Rev. Endocr. Metab. Disord. 2013, 14, 77–86. [Google Scholar] [CrossRef]
- Kelley, D.E.; He, J.; Menshikova, E.V.; Ritov, V.B. Dysfunction of Mitochondria in Human Skeletal Muscle in Type 2 Diabetes. Diabetes 2002, 51, 2944–2950. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Petersen, K.F.; Befroy, D.; Dufour, S.; Dziura, J.; Ariyan, C.; Rothman, D.L.; DiPietro, L.; Cline, G.W.; Shulman, G.I. Mitochondrial Dysfunction in the Elderly: Possible Role in Insulin Resistance. Science 2003, 300, 1140–1142. [Google Scholar] [CrossRef] [Green Version]
- Lanza, I.R.; Short, D.K.; Short, K.R.; Raghavakaimal, S.; Basu, R.; Joyner, M.J.; McConnell, J.P.; Nair, K.S. Endurance Exercise as a Countermeasure for Aging. Diabetes 2008, 57, 2933–2942. [Google Scholar] [CrossRef] [Green Version]
- Cesari, M.; Vellas, B.; Hsu, F.-C.; Newman, A.B.; Doss, H.; King, A.C.; Manini, T.M.; Church, T.; Gill, T.; Miller, M.E.; et al. A Physical Activity Intervention to Treat the Frailty Syndrome in Older Persons—Results From the LIFE-P Study. J. Gerontol. Ser. A 2015, 70, 216–222. [Google Scholar] [CrossRef]
- De Vries, N.M.; Staal, J.B.; van der Wees, P.J.; Adang, E.M.M.; Akkermans, R.; Rikkert, M.G.M.O.; der Sanden, M.W.G.N.-V. Patient-centred physical therapy is (cost-) effective in increasing physical activity and reducing frailty in older adults with mobility problems: A randomized controlled trial with 6 months follow-up. J. Cachex-Sarcopenia Muscle 2015, 7, 422–435. [Google Scholar] [CrossRef]
- Theou, O.; Stathokostas, L.; Roland, K.P.; Jakobi, J.M.; Patterson, C.; Vandervoort, A.A.; Jones, G.R. The Effectiveness of Exercise Interventions for the Management of Frailty: A Systematic Review. J. Aging Res. 2011, 2011, 1–19. [Google Scholar] [CrossRef] [Green Version]
- Cadore, E.L.; Rodríguez-Mañas, L.; Sinclair, A.; Izquierdo, M. Effects of Different Exercise Interventions on Risk of Falls, Gait Ability, and Balance in Physically Frail Older Adults: A Systematic Review. Rejuvenation Res. 2013, 16, 105–114. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Hovanec, N.; Sawant, A.; Overend, T.J.; Petrella, R.J.; Vandervoort, A.A. Resistance Training and Older Adults with Type 2 Diabetes Mellitus: Strength of the Evidence. J. Aging Res. 2012, 2012, 284635. [Google Scholar] [CrossRef]
- Fielding, R.A. The role of progressive resistance training and nutrition in the preservation of lean body mass in the elderly. J. Am. Coll. Nutr. 1995, 14, 587–594. [Google Scholar] [CrossRef] [PubMed]
- Daniels, R.; Van Rossum, E.; De Witte, L.; Kempen, G.I.J.M.; Heuvel, W.V.D. Interventions to prevent disability in frail community-dwelling elderly: A systematic review. BMC Health Serv. Res. 2008, 8, 278. [Google Scholar] [CrossRef] [Green Version]
- Lee, J.; Kim, D.; Kim, C. Resistance Training for Glycemic Control, Muscular Strength, and Lean Body Mass in Old Type 2 Diabetic Patients: A Meta-Analysis. Diabetes Ther. 2017, 8, 459–473. [Google Scholar] [CrossRef] [Green Version]
- Brandon, L.J.; Gaasch, D.A.; Boyette, L.W.; Lloyd, A.M. Effects of Long-Term Resistive Training on Mobility and Strength in Older Adults with Diabetes. J. Gerontol. Ser. A 2003, 58, M740–M745. [Google Scholar] [CrossRef]
- Ibañez, J.; Izquierdo, M.; Argüelles, I.; Forga, L.; Larrión, J.L.; García-Unciti, M.; Idoate, F.; Gorostiaga, E.M. Twice-Weekly Progressive Resistance Training Decreases Abdominal Fat and Improves Insulin Sensitivity in Older Men with Type 2 Diabetes. Diabetes Care 2005, 28, 662–667. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Mourier, A.; Gautier, J.-F.; De Kerviler, E.; Bigard, A.X.; Villette, J.-M.; Garnier, J.-P.; Duvallet, A.; Guezennec, C.Y.; Cathelineau, G. Mobilization of Visceral Adipose Tissue Related to the Improvement in Insulin Sensitivity in Response to Physical Training in NIDDM: Effects of branched-chain amino acid supplements. Diabetes Care 1997, 20, 385–391. [Google Scholar] [CrossRef]
- Eriksson, J.; Taimela, S.; Parviainen, S.; Peltonen, J.; Kujala, U. Resistance Training in the Treatment of Non-Insulin-Dependent Diabetes Mellitus. Int. J. Sports Med. 1997, 18, 242–246. [Google Scholar] [CrossRef]
- Miller, W.J.; Sherman, W.M.; Ivy, J.L. Effect of strength training on glucose tolerance and post-glucose insulin response. Med. Sci. Sports Exerc. 1984, 16, 539–543. [Google Scholar] [CrossRef]
- Poehlman, E.T.; Dvorak, R.V.; DeNino, W.F.; Brochu, M.; Ades, P.A. Effects of Resistance Training and Endurance Training on Insulin Sensitivity in Nonobese, Young Women: A Controlled Randomized Trial1. J. Clin. Endocrinol. Metab. 2000, 85, 2463–2468. [Google Scholar] [CrossRef] [Green Version]
- Gaster, M.; Vach, W.; Beck-Nielsen, H.; Schrøder, H.D. GLUT4 expression at the plasma membrane is related to fibre volume in human skeletal muscle fibres. APMIS 2002, 110, 611–619. [Google Scholar] [CrossRef] [PubMed]
- Rodriguez-Mañas, L.; Laosa, O.; Vellas, B.; Paolisso, G.; Topinkova, E.; Oliva-Moreno, J.; Bourdel-Marchasson, I.; Izquierdo, M.; Hood, K.; Zeyfang, A.; et al. Effectiveness of a multimodal intervention in functionally impaired older people with type 2 diabetes mellitus. J. Cachex-Sarcopenia Muscle 2019, 10, 721–733. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rejeski, W.J.; Ip, E.H.; Bertoni, A.; Bray, G.A.; Evans, G.; Gregg, E.; Zhang, Q. Lifestyle Change and Mobility in Obese Adults with Type 2 Diabetes. N. Engl. J. Med. 2012, 366, 1209–1217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Buman, M.P.; Hekler, E.B.; Haskell, W.L.; Pruitt, L.; Conway, T.L.; Cain, K.L.; Sallis, J.F.; Saelens, B.E.; Frank, L.D.; King, A.C. Objective Light-Intensity Physical Activity Associations with Rated Health in Older Adults. Am. J. Epidemiol. 2010, 172, 1155–1165. [Google Scholar] [CrossRef] [Green Version]
- Izquierdo, M.; Vivifrail Investigators Group; Rodriguez-Mañas, L.; Sinclair, A.J. What is new in exercise regimes for frail older people—How does the Erasmus Vivifrail Project take us forward? J. Nutr. Health Aging 2016, 20, 736–737. [Google Scholar] [CrossRef] [PubMed]
- Henderson, R.M.; Miller, M.E.; Fielding, R.A.; Gill, T.; Glynn, N.W.; Guralnik, J.M.; King, A.; Newman, A.B.; Manini, T.M.; Marsh, A.P.; et al. Maintenance of Physical Function 1 Year After Exercise Intervention in At-Risk Older Adults: Follow-up from the LIFE Study. J. Gerontol. Ser. A Boil. Sci. Med. Sci. 2018, 73, 688–694. [Google Scholar] [CrossRef]
- Morley, J.E.; Von Haehling, S.; Anker, S.D.; Vellas, B. From sarcopenia to frailty: A road less traveled. J. Cachex- Sarcopenia Muscle 2014, 5, 5–8. [Google Scholar] [CrossRef]
- Sinclair, A.; Morley, J.E.; Rodriguez-Mañas, L.; Paolisso, G.; Bayer, T.; Zeyfang, A.; Bourdel-Marchasson, I.; Vischer, U.; Woo, J.; Chapman, I.; et al. Diabetes Mellitus in Older People: Position Statement on behalf of the International Association of Gerontology and Geriatrics (IAGG), the European Diabetes Working Party for Older People (EDWPOP), and the International Task Force of Experts in Diabetes. J. Am. Med. Dir. Assoc. 2012, 13, 497–502. [Google Scholar] [CrossRef]
- Reaney, M.; Elash, C.A.; Litcher-Kelly, L. Patient Reported Outcomes (PROs) used in recent Phase 3 trials for Type 2 Diabetes: A review of concepts assessed by these PROs and factors to consider when choosing a PRO for future trials. Diabetes Res. Clin. Pract. 2016, 116, 54–67. [Google Scholar] [CrossRef]
- Tentolouris, A.; Vlachakis, P.; Tzeravini, E.; Eleftheriadou, I.; Tentolouris, N. SGLT2 Inhibitors: A Review of Their Antidiabetic and Cardioprotective Effects. Int. J. Environ. Res. Public Health 2019, 16, 2965. [Google Scholar] [CrossRef] [Green Version]
- Lee, Y.-S.; Jun, H.-S. Anti-Inflammatory Effects of GLP-1-Based Therapies beyond Glucose Control. Mediat. Inflamm. 2016, 2016, 3094642. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yates, T.; Henson, J.; Sargeant, J.; King, J.A.; Ahmad, E.; Zaccardi, F.; Davies, M.J. Exercise, Pharmaceutical Therapies and Type 2 Diabetes: Looking beyond Glycemic Control to Whole Body Health and Function. Transl. Med. Exerc. Prescr. 2021, 33–42. [Google Scholar] [CrossRef]
- Boulé, N.G.; Robert, C.; Bell, G.J.; Johnson, S.T.; Bell, R.C.; Lewanczuk, R.Z.; Gabr, R.Q.; Brocks, D.R. Metformin and Exercise in Type 2 Diabetes. Diabetes Care 2011, 34, 1469–1474. [Google Scholar] [CrossRef] [Green Version]
- Braun, B.; Eze, P.; Stephens, B.R.; Hagobian, T.A.; Sharoff, C.G.; Chipkin, S.; Goldstein, B. Impact of metformin on peak aerobic capacity. Appl. Physiol. Nutr. Metab. 2008, 33, 61–67. [Google Scholar] [CrossRef]
- Linden, M.A.; Fletcher, J.A.; Morris, E.M.; Meers, G.M.; Kearney, M.L.; Crissey, J.M.; Laughlin, M.H.; Booth, F.W.; Sowers, J.R.; Ibdah, J.A.; et al. Combining metformin and aerobic exercise training in the treatment of type 2 diabetes and NAFLD in OLETF rats. Am. J. Physiol. Metab. 2014, 306, E300–E310. [Google Scholar] [CrossRef] [Green Version]
- Malin, S.K.; Nightingale, J.; Choi, S.-E.; Chipkin, S.R.; Braun, B. Metformin modifies the exercise training effects on risk factors for cardiovascular disease in impaired glucose tolerant adults. Obesity 2013, 21, 93–100. [Google Scholar] [CrossRef]
- Myette-Côté, E.; Terada, T.; Boulé, N.G. The Effect of Exercise with or Without Metformin on Glucose Profiles in Type 2 Diabetes: A Pilot Study. Can. J. Diabetes 2016, 40, 173–177. [Google Scholar] [CrossRef]
- Sharoff, C.G.; Hagobian, T.A.; Malin, S.; Chipkin, S.; Yu, H.; Hirshman, M.F.; Goodyear, L.J.; Braun, B. Combining short-term metformin treatment and one bout of exercise does not increase insulin action in insulin-resistant individuals. Am. J. Physiol. Metab. 2010, 298, E815–E823. [Google Scholar] [CrossRef] [PubMed]
- Newman, A.A.; Grimm, N.C.; Wilburn, J.R.; Schoenberg, H.M.; Trikha, S.R.; Luckasen, G.J.; Biela, L.M.; Melby, C.L.; Bell, C.; Wilburn, J.R. Influence of Sodium Glucose Cotransporter 2 Inhibition on Physiological Adaptation to Endurance Exercise Training. J. Clin. Endocrinol. Metab. 2019, 104, 1953–1966. [Google Scholar] [CrossRef] [PubMed]
- Jensen, S.; Janus, C.; Lundgren, J.R.; Juhl, C.R.; Blond, M.B.; Stallknecht, B.; Holst, J.J.; Madsbad, S.; Torekov, S.S. 686-P: Exercise Alone and in Combination with Liraglutide Exert Clinically Relevant Improvements in Cardiorespiratory Fitness during 1-Year Weight Loss Maintenance: The S-LITE Randomized Trial. Diabetes 2020, 69, 686. [Google Scholar] [CrossRef]
| Age Groups | Percentage with Some Physical Limitation | |
|---|---|---|
| With Diabetes | Without Diabetes | |
| 18–44 | 46 | 18 |
| 45–64 | 63 | 35 |
| 65–74 | 74 | 53 |
| ≥75 | 85 | 70 |
| Physical Function Category | Percentage (%) | 95% Confidence Interval (95% CI) |
|---|---|---|
| General physical activities | 73.6 | 70.2–76.9 |
| Lower extremity mobility | 52.2 | 48.5–55.9 |
| IADL | 43.6 | 40.1–47.2 |
| ADL | 37.2 | 33.1–41.3 |
| Leisure and social activities | 33.8 | 30.8–36.9 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ahmad, E.; Sargeant, J.A.; Yates, T.; Webb, D.R.; Davies, M.J. Type 2 Diabetes and Impaired Physical Function: A Growing Problem. Diabetology 2022, 3, 30-45. https://doi.org/10.3390/diabetology3010003
Ahmad E, Sargeant JA, Yates T, Webb DR, Davies MJ. Type 2 Diabetes and Impaired Physical Function: A Growing Problem. Diabetology. 2022; 3(1):30-45. https://doi.org/10.3390/diabetology3010003
Chicago/Turabian StyleAhmad, Ehtasham, Jack A. Sargeant, Tom Yates, David R. Webb, and Melanie J. Davies. 2022. "Type 2 Diabetes and Impaired Physical Function: A Growing Problem" Diabetology 3, no. 1: 30-45. https://doi.org/10.3390/diabetology3010003
APA StyleAhmad, E., Sargeant, J. A., Yates, T., Webb, D. R., & Davies, M. J. (2022). Type 2 Diabetes and Impaired Physical Function: A Growing Problem. Diabetology, 3(1), 30-45. https://doi.org/10.3390/diabetology3010003






