1. Introduction
Corn, soybean meal (SBM), and sunflower meal (SFM) are among the most important raw materials in poultry and livestock feed formulations, providing essential energy and protein sources [
1]. However, their chemical profiles vary considerably depending on geographical origin, agronomic conditions, and processing methods.
Corn composition shows notable differences in crude protein (CP), ether extract (EE), gross energy (GE), and mineral content. Comparative studies across countries have reported CP values ranging from 7.12% in Brazil to 7.60% in China, EE from 3.30% (USA) to 3.87% (Brazil), and GE values between 3836 and 3995 kcal/kg, with the highest values found in Brazilian corn [
2]. Such differences are influenced by variety, soil fertility, climate, and environmental stress factors [
2].
Similarly, the nutritional value of soybean meal differs according to its origin. A large-scale near-infrared spectroscopy (NIRS) analysis of 77,478 SBM samples collected from 2018 to 2021 showed that Argentine SBM generally had the lowest CP, EE, and fiber contents, whereas Brazilian SBM had the highest CP, EE, fiber, and digestible lysine values. U.S. SBM, in contrast, showed the highest metabolizable energy (ME, MEn) and amino acid digestibility [
3]. Meta-analysis results support these findings, indicating significant differences between Brazilian, Argentine, U.S., and Indian SBM in terms of CP, fiber, sugars, iron, and essential amino acids such as lysine, methionine, threonine, and cysteine [
3].
Sunflower meal composition is also highly variable, particularly depending on the proportion of hulls remaining after processing. Standard SFM typically contains 30–34% CP, 20–25% crude fiber, and 8–10% lignin, which limits its value for poultry and swine due to its high fiber content [
4]. However, dehulled sunflower meal (HiSFM) can reach ~46% CP and reduced fiber content (8–14%), demonstrating the impact of both raw material origin (seed hull content) and processing technology on its chemical profile [
5].
The objective of this study is to present the average values of the chemical characteristics, key nutrients, and antinutritional factors of three major feed ingredients—corn, soybean meal, and sunflower meal—sourced from different regions of the world over the three years 2019, 2020, and 2021. Mycotoxin analyses were conducted on the maize batches to complement the study. Specifically, ELISA assays were performed, with results cross-validated using the HPLC method for quality control. It is hypothesized that the nutritional values of these ingredients vary according to their geographical origin, and that the choice of origin represents a critical nutritional factor in the formulation of compound feeds for poultry and livestock.
2. Materials and Methods
2.1. Experimental Protocol
Table 1 presents the imports of the main raw materials used in the formulation of compound feeds for poultry and livestock at the BENWAY feed manufacturing plant. The data cover the years 2019, 2020, and 2021, highlighting three key raw materials: corn, soybean meal (SBM), and sunflower meal (SFM). For each product, the table provides information on the country of origin, the number of analyzed samples representing the received batches, and the annual imported tonnage. These are essential data for assessing traceability, quality, and associated health risks.
All samples were analyzed in triplicate under identical laboratory conditions. Sampling was carried out upon reception of the batches, prior to storage at the plant.
In 2020, the total imported volumes of raw materials intended for animal feed—particularly corn, soybean meal, and sunflower meal—experienced a marked decline. This reduction is largely attributed to global logistical disruptions linked to the COVID-19 pandemic, which severely impacted international supply chains [
6].
2.2. Chemical Analyses Performed on the Samples
The following parameters were determined as part of the chemical analysis of the samples: moisture content, water activity, crude protein content, crude fat content, crude fiber content, ash content (mineral matter), starch content (for corn), protein dispersibility index (PDI), digestible protein content in KOH, reactive lysine content, ratio of reactive lysine to total lysine, PCI index (to assess soybean meal batches), urease activity (mg N/g/min), and protein solubility in potassium hydroxide (KOH).
Additional nutritional parameters were predicted using the calibration equations from the NIR device database (EVONIK calibration): poultry metabolizable energy, digestible lysine, digestible methionine + cystine, and digestible threonine.
Two analytical methods were employed for the analysis of mycotoxins: ELISA (Enzyme-Linked Immunosorbent Assay) as the primary screening method using Romer Labs kits, and HPLC (High-Performance Liquid Chromatography) for confirmation and verification purposes. An ELISA was applied for the initial quantification of mycotoxin levels due to its efficiency and suitability for large sample screening. A subset of samples was subsequently analyzed using HPLC to verify ELISA results and ensure accuracy and reliability through cross-validation.
2.3. Sampling
For each received batch, a representative sample was analyzed. The sampling procedure was as follows:
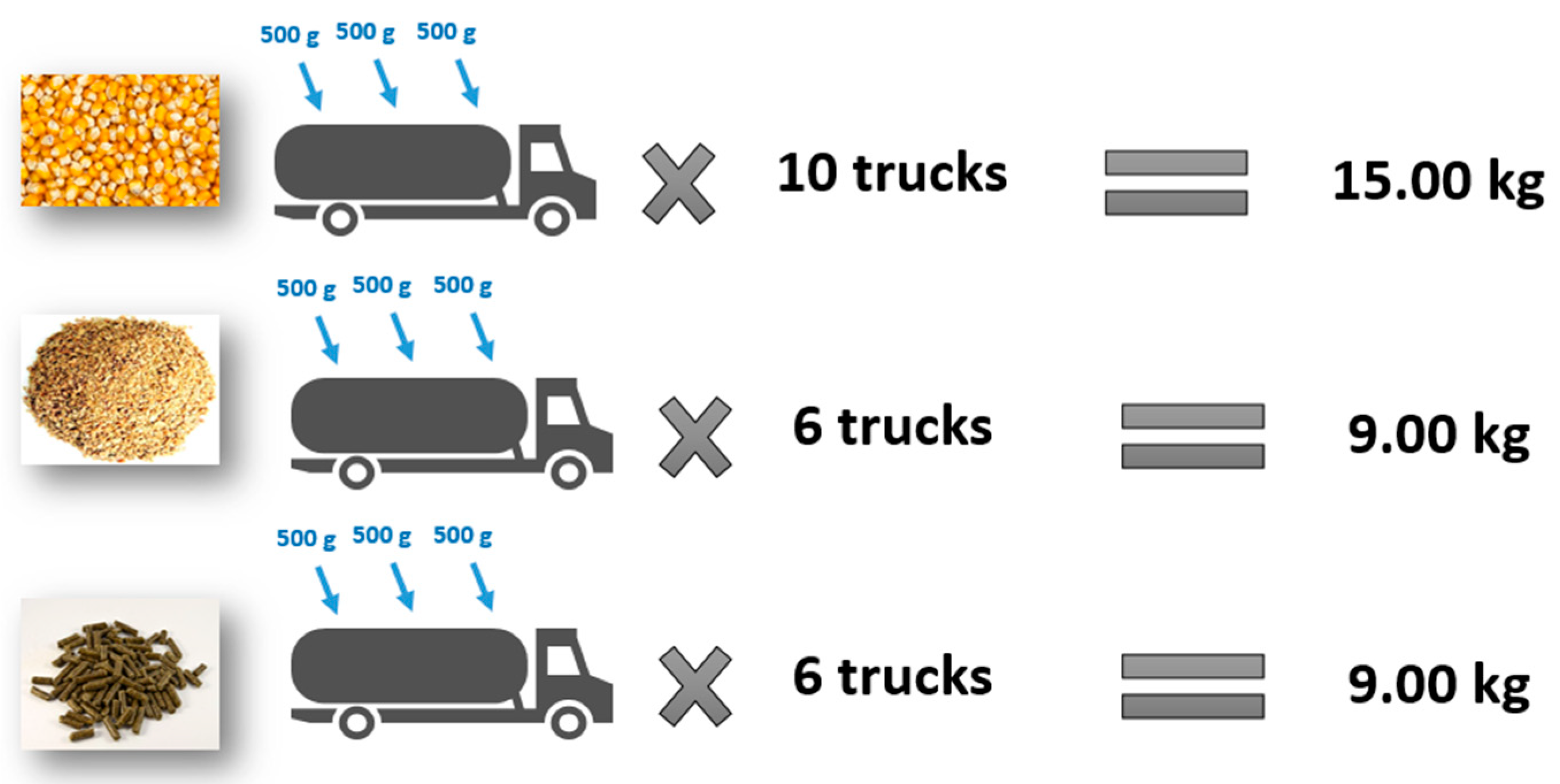
Three sampling points were obtained from each truck (with a capacity of 14 metric tons).
Ten trucks per batch were randomly selected for corn sampling.
Six trucks per batch were randomly selected for soybean meal and sunflower meal sampling.
Three samples were obtained from each truck: the front, middle, and rear, using a deep sampling probe.
2.4. Sample Preparation
For each batch, around twenty trucks deliver the raw materials, with arrivals usually spread over 3 to 5 days. The trucks are randomly selected according to the required number of samples, the designated sampling points, and the standardized sampling method. The collected samples are then stored in sealed containers to preserve their nutritional value and moisture content until all samples are obtained. Subsequently, the samples are pooled and divided using a sample divider to obtain one representative sample per batch for each raw material.
For each batch, sampling was carried out using three probe collections of 500 g each, resulting in a total of 1.5 kg per truck. For corn, this protocol was applied to 10 trucks, yielding a composite sample of 15 kg per corn batch. For soybean meal and sunflower meal, the total sampled quantity per batch was 9 kg for each raw material (
Figure 1).
The selection of trucks was performed randomly, in accordance with the sampling protocol established for the three raw materials studied, and applied to all batches.
The individual sub-samples were homogenized to obtain a single composite sample per batch and per raw material. These composite samples were then processed using a sample divider (
Figure 2) to produce equal fractions, allowing for the collection of a final representative sample intended for analysis.
The principle of a sample divider is to reduce the size of a bulk sample into several representative subsamples, ensuring that each subsample retains the chemical and physical properties of the original material.
Before analysis, all samples underwent homogeneous grinding. This grinding step is essential both for conducting reference chemical analyses, including mycotoxin quantification and for nutritional predictions using near-infrared spectroscopy (NIR), particularly for poultry metabolizable energy (MEp) values and digestible amino acid contents.
2.5. Chemical Analysis Procedures Conducted at the Plant Laboratory
Table 2 shows the analytical methods performed and equipment.
2.6. Statistical Analysis
Due to the violation of normality assumptions required for parametric tests (such as ANOVA or Student’s t-test), non-parametric statistical methods were employed to ensure the validity and robustness of the comparative analyses. The Kruskal–Wallis H test was used to assess differences between more than two independent groups, while the Mann–Whitney U test was applied for comparisons between two groups. This approach was selected to accommodate non-normal data distributions and unequal sample sizes, particularly noting the small sample size in group 3 (number of batches analyzed n = 9), which could otherwise compromise the statistical power of parametric tests. Although some variables (e.g., starch content) showed normal distributions, the Kruskal–Wallis test was uniformly applied across all variables to maintain methodological consistency. A significance level of p < 0.05 was considered statistically significant. All analyses were performed using appropriate statistical software SPSS V26.
Since the Shapiro–Wilk and Kolmogorov–Smirnov normality tests indicated that all mycotoxin concentration data deviated significantly from a normal distribution (p < 0.05 for almost all origins and compounds), the assumptions required for a parametric one-way ANOVA were not met. Consequently, non-parametric tests were applied: the Kruskal–Wallis test was used to compare the three origins (Argentina, Brazil, and Ukraine) for each year, and the Mann–Whitney U test was employed for pairwise post hoc comparisons
3. Results
3.1. Chemical Composition of Corn Imported from Argentina, Brazil, and Ukraine to Morocco in 2019–2021 by a Compound Feed Manufacturing Plant for Poultry
3.1.1. Moisture Content (H2O%) and Water Activity (Aw) in Corn
Moisture content values ranged from 12.625% to 14.583%, while water activity (Aw) varied between 0.677 and 0.747. The highest moisture content was observed in Brazilian corn from 2021 (14.583%), which also showed one of the highest water activity values (0.721). (The detailed chemical composition of corn from the three origins over the three years is presented in
Appendix A,
Table A1,
Table A2,
Table A3,
Table A4,
Table A5,
Table A6 and
Table A7).
Water activity (Aw), which is closely linked to the availability of free water for microorganisms, remained around 0.68–0.69.
3.1.2. Crude Protein (CP%) and Digestible Amino Acids in Corn
Crude protein (CP) content in corn samples ranged from 7.013% (Brazil, 2021) to 7.483% (Brazil, 2019). These values comply with established standards for corn grain [
7,
8]. Digestible lysine values were relatively consistent across origins and years, varying from 0.218% to 0.224%, while digestible methionine + cysteine ranged from 0.299% to 0.313%. Digestible threonine content showed more noticeable variation, with Brazilian corn in 2019 presenting the highest value (0.253%). Post hoc Mann–Whitney analysis indicated that Brazilian maize exhibited a significantly higher digestible threonine content compared with other origin
3.1.3. Starch Content (%) and Metabolizable Energy (ME) in Corn
Starch is the main energy source in corn. Its content was high and stable:
Corn AR (Argentina): 64.03%
Corn Ukrainien (Ukraine): 64.07%
Corn Brésilien (Brazil): 63.80%
Metabolizable energy (ME) values ranged from 3318.000 kcal/kg (Argentina, 2020) to 3352.200 kcal/kg (Brazil, 2019), while starch content varied between 63.800% (Argentina, 2019; Brazil, 2019) and 64.207% (Argentina, 2021).
3.1.4. Crude Fat (CF%), Crude Fiber (CFb%), and Ash (MM%) in Corn
Crude fat content in the analyzed corn samples ranged from 3.929% (Argentina, 2020) to 4.167% (Ukraine, 2019), while crude fiber varied between 1.850% (Argentina, 2020) and 2.133% (Ukraine, 2019). Ash content showed a narrower range, from 1.090% (Ukraine, 2020) to 1.267% (Brazil, 2021). The relatively higher fat levels in Ukrainian corn from 2019 may have contributed to its metabolizable energy value, whereas variations in fiber content could partially explain differences in energy availability among samples. The ash values indicate minor differences in mineral content across origins and years, although slight variations were observed between origins and years, statistical analysis indicated that these differences were not significant for the three origins studied
3.1.5. Nutritional Variability of Corn by Origin
Moisture Content
Argentine corn showed the highest variability in moisture (CV = 6.35%), followed by Brazilian corn (4.47%), while Ukrainian corn exhibited the lowest variability (2.82%). This suggests that Argentine corn batches were less homogeneous in terms of moisture, likely reflecting differences in harvesting, storage, or transport conditions.
Water Activity (Aw)
The greatest variability was observed in Argentine corn (CV = 7.16%), indicating substantial heterogeneity in preservation conditions. Brazilian corn showed moderate variability (2.82%), whereas Ukrainian corn was highly homogeneous (0.26%), suggesting greater stability in this parameter.
Starch Content
Coefficients of variation were low across all origins (1.95% for Argentina, 0.91% for Brazil, and 1.36% for Ukraine), indicating that starch levels were relatively consistent among batches regardless of origin. This was expected, as starch is a major and relatively stable component of corn.
Variability was mainly observed in moisture and particularly in water activity, especially in Argentine corn, which may affect storage quality and increase the risk of fungal growth and mycotoxin development. In contrast, major nutritional components such as starch remained relatively stable across batches and origins.
3.2. Kruskal–Wallis Test for Comparing the Corn Origins (2019–2021)
Kruskal–Wallis tests (
Table 3) were conducted for all measured variables to assess differences between maize samples from three different origins over the years 2019, 2020, and 2021. Variables such as moisture (%), crude protein (%), crude fat (%), crude fiber (%), ash (%), metabolizable energy (kcal/kg), digestible lysine (%), and starch (%) showed no significant effect of origin (
p > 0.05), indicating comparable values across Argentina, Brazil, and Ukraine.
For water activity (Aw), the Kruskal–Wallis test revealed a significant overall difference between origins (H = 19.539; p = 0.000). Post hoc pairwise comparisons using Mann–Whitney tests with Bonferroni correction indicated that this difference was driven exclusively by the comparison between Argentinian and Brazilian maize (p < 0.017), while the other pairs did not show significant differences. Argentinian maize had an average water activity of 0.680, compared to 0.716 for Brazilian maize, making it the most at risk for mold development and potential mycotoxin contamination. Argentine corn presented the lowest average water activity value (0.68), indicating better preservation of its nutritional quality during storage. Overall, the origin factor appears to have a limited impact on corn composition over the three years studied, with water activity being the most sensitive parameter.
Similarly, Digestible Threonine (%) showed a significant difference between origins (H = 15.118; p = 0.001). Post hoc Mann–Whitney tests demonstrated that two pairs of origins had significant differences after Bonferroni correction, contributing to the overall Kruskal–Wallis result. Brazilian maize showed a significantly higher digestible threonine content, with an average value of 0.253%, which is the highest among all the maize origins studied
For digestible methionine + cysteine (%), a trend toward significance was observed (H = 5.684; p = 0.058), suggesting that some origins may have slightly different values, but this difference is not statistically significant at the 5% level.
In summary, only water activity (Aw) and digestible threonine (%) showed significant differences between maize origins, Digestible methionine + cysteine (%) showed a trend toward variation, and all other variables were comparable across Argentina, Brazil, and Ukraine.
3.3. Chemical Composition of Soybean Meal Imported from the USA and Argentina to Morocco in 2019–2021 by a Poultry Compound Feed Manufacturing Plant
3.3.1. Analysis Results of Soybean Meal Imported from the USA
The imported soybean meal from the USA showed a moisture content of 11.22% and water activity (Aw) of 0.658. Crude protein was 46.91%, with 2.35% crude fat, 3.66% crude fiber, and 6.24% ash. Starch content was low at 0.71%, while metabolizable energy (MEp) was 2369.73 kcal/kg. Digestible amino acids were 2.56% lysine, 1.10% methionine + cysteine, and 1.51% threonine. Protein quality indicators were high, with PDI at 11.89%, urease activity of 0.045 mg N/g/min, and protein solubility in KOH at 76.91%, indicating good nutritional quality for poultry feed (the detailed chemical composition of soybean meal imported from the USA is presented in
Appendix A,
Table A8,
Table A9 and
Table A10).
3.3.2. Results of the Analysis of Soybean Meal Imported from Argentina
The soybean meal imported from Argentina exhibited a moisture content of 10.85% and water activity (Aw) of 0.647. Crude protein was high at 46.61%, with 2.08% crude fat, 3.67% crude fiber, and 6.55% ash. Starch content was low (0.72%), and metabolizable energy (MEp) was 2365.83 kcal/kg. Digestible amino acids included 2.55% lysine, 1.11% methionine + cysteine, and 1.53% threonine. Protein quality indicators were strong, with a PDI of 11.91%, urease activity of 0.042 mg N/g/min, and protein solubility in KOH of 80.38%. Notably, the Argentine soybean meal showed larger standard deviations for most parameters compared with the USA soybean meal, which may affect the consistency and stability of the Argentine product. (The detailed chemical composition of soybean meal imported from Argentina is presented in
Appendix A,
Table A11 and
Table A12).
3.3.3. Comparison of the Quality of Soybean Meal Imported from the United States and Argentina (2019–2021)
Crude Protein and Stability of Protein Content
CP USA: 47.26% (2019), 46.43% (2020), 46.82% (2021)
CP Argentina: 46.76% (2019), 46.46% (2021), with a minimum observed at 43.60%
Digestible Amino Acid Profile
American soybean meal shows excellent digestibility for essential amino acids, key indicators in monogastric nutrition:
Digestible Lysine USA: 2.58% (2019), 2.54% (2021)
Digestible Methionine + Cystine: 1.11% (2019), 1.08% (2021)
Digestible Threonine: 1.52–1.50%
Thermal Processing Control
Processing quality is critical to preserving protein digestibility and neutralizing antinutritional factors. American soybean meal shows exemplary thermal processing indicators: IDP: controlled (11.01–12.76%), versus 10.74–12.23% for Argentine meal, urease activity: very low (0.05 mg N/g/min in 2019), similar to Argentina (0.04), KOH protein solubility: 75–78% in the USA, versus 77.85–82.90% in Argentina higher values sometimes indicating overcooking risk [
9]. The optimum PDI range for monogastric feeds is generally between 15 and 25%, guaranteeing good protein digestibility while ensuring the inactivation of anti-nutritional compounds [
10].
Reactive lysine/total lysine (USA 2021): 89.37%, indicating minimal thermal degradation of amino acids. These values show optimal heat treatment for American soybean meal, neither undercooked nor overcooked, promoting protein bioavailability.
3.3.4. Statistical Comparison Between U.S. and Argentine Soybean Meal (Mann–Whitney U Test)
This analysis evaluates whether there are statistically significant differences between two origins of soybean meal across various nutritional and quality parameters. The Mann–Whitney U test (non-parametric test for two independent samples) was applied. A significance threshold of p < 0.05 was used.
The moisture content in American soybean meal is significantly higher than that of Argentine soybean meal (
p < 0.05) (
Table 4).
Among the parameters analyzed, only moisture and water activity showed statistically significant differences between the two soybean meal origins, with the U.S. samples generally showing slightly higher values. All other parameters—including protein content, digestible amino acids, urease activity, and protein solubility—did not differ significantly between the two sources.
3.3.5. Comparative Variability of American and Argentine Soybean Meals
The American soybean meal exhibited a coefficient of variation in moisture content of 8.65% in 2019 compared to 4.35% in 2021. Similarly, the Argentine soybean meal showed a high coefficient of variation in 2019 (9.45%) versus 2.23% in 2021. Crude protein content did not display considerable variability between batches, with the American soybean meal presenting a CV of 2.15% in 2019 and the Argentine soybean meal 2.73% in the same year. In contrast, higher variability between batches was observed in secondary parameters such as crude fat (27.13% for American soybean meal in 2019 and 30.78% for Argentine soybean meal in 2019) and starch (26.46% for American soybean meal in 2019 and 10.50% for Argentine soybean meal in 2019). The protein dispersibility index (PDI) also exhibited marked variability between batches, particularly in Argentine soybean meal (15.82% in 2019) compared to American soybean meal (12.05% in 2019).
3.4. Chemical Composition of Sunflower Meal Mainly Imported from Ukraine to Morocco in 2019–2021 by a Poultry Feed Manufacturing Plant
Sunflower meal constitutes a partial alternative to soybean meal in poultry feed formulations, notably due to its availability and more competitive cost. However, its high fiber content and less balanced amino acid profile compared to soybean limit its inclusion in fast-growing poultry diets.
Ukrainian sunflower meal is the primary source used in the poultry industry in Morocco, which explains why the factory does not import sunflower meal from any other origin. An analysis of its chemical composition over the years 2019, 2020, and 2021 shows a notable evolution in its nutritional characteristics. The crude protein (CP) content steadily increased from 35.97% in 2019 to 36.99% in 2021, reflecting an improvement in its nutritional value. Similarly, the metabolizable energy (MEp) rose from 1553.61 kcal/kg to 1600.17 kcal/kg, which is beneficial for poultry feed efficiency. Crude fiber content decreased from 17.97% to 17.03%, enhancing digestibility. Meanwhile, ash content showed slight fluctuations, reaching 6.79% in 2021. Regarding digestible amino acids, digestible methionine + cysteine showed a slight increase from 1.17% to 1.23%, while digestible lysine and digestible threonine remained relatively stable across the three years. (The detailed chemical composition of sunflower meal imported from Ukraine is presented in
Appendix A,
Table A13,
Table A14 and
Table A15).
A Kruskal–Wallis H test was conducted to determine whether there were statistically significant differences in the chemical composition of sunflower meal across three years (df = 2). The results are shown in
Table 5.
The results indicate that the year has a statistically significant effect on several key nutritional parameters of sunflower meal, including the following:
No significant year-to-year variation was found for water activity, crude fiber, digestible lysine (borderline), and digestible threonine.
4. Mycotoxin Profiling of Maize Batches Analyzed in This Study
The assessment of mycotoxin contamination in imported corn revealed substantial variations among origins. For deoxynivalenol (DON), Brazilian corn exhibited the highest mean concentration (1354.44 ppb; range: 200–3980 ppb), followed by Argentine corn (752.81 ppb; 270–3300 ppb) and Ukrainian corn (456.00 ppb; 410–560 ppb). Fumonisins (B1 + B2) were most elevated in Argentine corn (1165.26 ppb; 210–6040 ppb), with Brazilian corn showing slightly lower levels (1019.52 ppb; 280–2120 ppb) and Ukrainian corn presenting the lowest and constant value (200 ppb). Ochratoxin A (OTA) concentrations were highest in Brazilian corn (3.02 ppb; 1.9–7.15 ppb), while Argentine and Ukrainian corn showed lower means of 2.26 ppb (1–6.22 ppb) and 1.90 ppb (constant), respectively. T-2 toxin levels were highest in Argentine corn (13.91 ppb; 10–71.34 ppb) compared to Brazilian (10.00 ppb) and Ukrainian corn (10.05 ppb). Zearalenone contamination was markedly higher in Argentine corn (36.99 ppb; 0–199.25 ppb), followed by Brazilian (21.92 ppb; 2.62–61.75 ppb) and Ukrainian corn (4.90 ppb; 3.10–6.86 ppb). In contrast, aflatoxin B1 concentrations were low and uniform across all origins (0.20 ppb).
Effect of Corn Origin on Mycotoxin Profile
An overview of the contamination by the six mycotoxins across the different corn origins is presented in detail in
Appendix A,
Table A19.
The mycotoxin contamination profile of maize varied according to its origin (Argentina, Brazil, and Ukraine), as demonstrated by both descriptive statistics and the Kruskal–Wallis H test.
Fumonisins B1 + B2 showed significant differences among origins (H = 6.145, p = 0.046), with Argentine corn exhibiting the highest mean level (1165.26 ppb), followed closely by Brazilian corn (1019.52 ppb). Ukrainian corn showed minimal contamination, with a consistent value of 200 ppb across all samples.
Ochratoxin A also varied significantly (H = 8.230, p = 0.016), with Brazilian corn displaying the highest average (3.02 ppb) and Ukrainian corn the lowest (1.90 ppb).
A significant difference was also observed for T-2 toxin (H = 6.624, p = 0.036), where Argentine corn had a notably high mean (13.91 ppb), in contrast to the low and consistent levels in Brazilian and Ukrainian corn (around 10 ppb).
Similarly, zearalenone levels significantly differed across origins (H = 6.772, p = 0.034), with Argentine corn again showing the highest mean value (36.99 ppb), followed by Brazilian (21.92 ppb), and Ukrainian corn presenting the lowest average (4.90 ppb).
In contrast, deoxynivalenol (DON) levels did not significantly differ by origin (H = 1.683, p = 0.431), despite Brazilian corn showing the highest mean (1354.44 ppb) and Ukrainian corn the lowest (456.00 ppb).
Aflatoxin B1 levels were identical across all samples (0.2 ppb), with no statistical difference (H = 0.000, p = 1.000), indicating uniform and minimal contamination.
These results confirm that maize origin significantly affects contamination levels for several mycotoxins, particularly fumonisins, ochratoxin A, T-2 toxin, and zearalenone, while DON and aflatoxin B1 remained statistically similar between sources. Ukrainian maize consistently showed the lowest mycotoxin burden, making it the least contaminated among the three origins analyzed (
Table 6).
5. Discussion
5.1. Chemical Profile of Corn Imported from Argentina, Brazil, and Ukraine to Morocco in 2019–2021 by a Compound Feed Manufacturing Plant for Poultry
5.1.1. Analysis of Moisture Content (H2O%) and Water Activity (Aw) in Corn
The highest moisture content was observed in Brazilian corn from 2021 (14.583%), which also showed one of the highest water activity values (0.721). Such elevated values may increase the risk of fungal growth and mycotoxin production during storage, particularly when exceeding the generally recommended thresholds for safe grain conservation [
11].
Water activity (Aw), which is closely linked to the availability of free water for microorganisms, remained around 0.68–0.69. While these values are acceptable, they are close to the critical threshold of 0.70, beyond which mold growth—such as
Aspergillus spp.—becomes possible [
12]. Regular monitoring of mycotoxin contamination is therefore recommended.
5.1.2. Analysis of Crude Protein (CP%) and Digestible Amino Acids in Corn
Crude protein (CP) content in corn samples ranged from 7.013% to 7.483%.
These values are typical of corn but relatively low compared to poultry nutritional requirements. This confirms the necessity of systematically supplementing formulas with protein-rich sources (such as soybean meal and synthetic amino acids) [
13].
5.1.3. Analysis of Starch Content (%) and Metabolizable Energy (ME) in Corn
The values fall within the optimal range (60–70% of dry matter), ensuring good digestibility and an appropriate energy supply for poultry. The moderate standard deviation (~1–1.5) reflects consistent quality.
In general, higher starch levels tended to correspond to higher ME values, although exceptions were observed, suggesting that factors such as protein, fat, and fiber composition may also influence ME. Brazilian corn from 2019 showed the highest ME value despite having one of the lowest starch contents, indicating that other nutritional components likely contributed to its energy value.
The mean (2019, 2020, and 2021) metabolizable energy (ME) values followed the same trend: corn from Argentina: 3331.06 kcal, corn from Brazil: 3352.20 kcal, and corn from Ukraine: 3343.67 kcal.
The values of EMp values align with [
14] recommendations and are suitable for formulating grower and finisher diets for broiler chickens.
5.1.4. Analysis of Crude Fat (CF%), Crude Fiber (CFb%), and Ash (MM%) in Corn
The relatively higher fat levels in Ukrainian corn from 2019 may have contributed to its metabolizable energy value, whereas variations in fiber content could partially explain differences in energy availability among samples. The ash values indicate minor differences in mineral content across origins and years.
5.2. Chemical Composition of Soybean Meal Imported from the USA and Argentina to Morocco in 2019–2021
A general positive trend was observed in American soybean meal, which exhibited higher average levels of crude protein, crude fat, metabolizable energy for poultry, and digestible lysine compared to Argentine soybean meal. However, statistical analysis revealed that only moisture content and water activity showed significant differences between the two origins.
Numerous studies have highlighted the superior nutritional quality of American soybean meal, particularly regarding amino acid digestibility and compositional stability [
14,
15]. Thermal processing standards, which are well controlled in the United States, allow for protein dispersibility index (PDI) and urease activity levels compatible with good nutritional value and minimal protein denaturation [
16]. Moreover, the ratios of digestible lysine to total lysine—indicators of heat treatment quality and preservation of essential amino acids—are generally more favorable for American soybean meals [
17]. In comparison, Argentine soybean meal exhibits greater variability, notably in thermal processing and amino acid digestibility [
18], which can affect zootechnical performance in practical settings [
19]. Differences in industrial methods, storage, and logistics partly explain this variability [
20].
5.3. Chemical Composition of Sunflower Meal Mainly Imported from Ukraine to Morocco in 2019–2021
These findings confirm that Ukrainian sunflower meal, as used in Morocco, provides consistent and high-quality nutritional value suitable for the poultry industry. These values are typical of dehulled sunflower meals, whose protein content generally ranges from 33 to 38% [
21]. In comparison, soybean meals contain CP levels of 46–48%. Therefore, sunflower meal remains a complementary protein coproduct but not a full substitute for soybean meal.
The moisture levels measured in sunflower meal batches imported from Ukraine between 2019 and 2021 ranged from 9.42% (2019) to 9.93% (2020), with an average of 9.84% in 2021 (
Appendix A Table A17,
Table A18 and
Table A19). These values are below the critical threshold of 12%, considered the limit beyond which the risk of mold growth and mycotoxin development increases [
22]. Controlled moisture therefore ensures good product stability during storage.
The level of crude fiber (17.561%) is typical of sunflower meal, even when dehulled, and represents a usage limitation, especially in young poultry and broilers due to its negative impact on overall digestibility and feed conversion ratio [
23]; conversely, this fiber richness can be beneficial in diets for layers or breeders, promoting digestive health and transit.
Metabolizable energy values ranged from 1553 to 1600 kcal/kg. This low-energy density is well documented [
24], limiting its use in diets requiring high caloric density (rapid growth, starter phase). Complementation with dense energy sources (oil, corn) is thus necessary.
5.4. Analysis of Mycotoxin Contamination
5.4.1. Analysis of Mycotoxin Contamination in Argentine Corn Imported
The analysis of Argentine corn samples reveals variable contamination levels due to several mycotoxins. Fumonisins B1 + B2 and deoxynivalenol (DON) recorded the highest mean values, at 1165 ppb and 752 ppb, respectively, with maximum levels reaching 6040 ppb for fumonisins and 3300 ppb for DON. When compared to European Union (EU) regulatory limits for animal feed [
25], the average fumonisin level in Argentine corn exceeds the guidance value for poultry feed ingredients, which is set at 20,000 ppb for maize products, but the observed maximum values could pose concerns. For DON, the EU recommends a maximum of 5000 ppb for maize products intended for animal feed, meaning some Argentine corn samples may exceed these limits.
Zearalenone showed an average of 36.99 ppb, remaining below the EU guidance value of 250 ppb for maize feed materials. T-2 toxin levels averaged 13.9 ppb, which is also within the indicative levels set by EFSA [
26], although no official EU guidance value exists specifically for T-2 in corn. Ochratoxin A and aflatoxin B1 were detected at very low concentrations; notably, aflatoxin B1 was consistently at 0.2 ppb, well below the strict EU maximum limit of 20 ppb for maize feed materials. (The detailed mycotoxin contamination data of Argentine corn are provided in
Appendix A,
Table A16).
These findings confirm that while Argentine corn generally complies with European standards for most mycotoxins, particular attention is needed regarding fumonisins and DON due to their high variability and occasional exceedance of EU limits. Systematic monitoring remains essential to guarantee feed safety, especially considering animal health risks associated with chronic exposure [
27].
5.4.2. Analysis of Mycotoxin Contamination in Brazilian Corn Imported
The analysis of Brazilian corn samples shows that deoxynivalenol (DON) and fumonisins B1 + B2 are the most prevalent mycotoxins, with mean concentrations of 1354.44 ppb and 1019.52 ppb, respectively. The maximum values reached 3980 ppb for DON and 2120 ppb for fumonisins. When compared to European Union guidance values [
25], these levels remain below the recommended maximum limits for maize products intended for animal feed.
Ochratoxin A, T-2 toxin, zearalenone, and aflatoxin B1 were present at much lower levels. Notably, aflatoxin B1 was detected at a constant value of 0.20 ppb, which is well below the EU maximum limit of 20 ppb for feed materials. Zearalenone averaged 21.92 ppb with a maximum of 61.75 ppb, remaining under the EU threshold of 250 ppb.
Despite these being acceptable levels relative to EU standards, the high coefficient of variation for DON (1.04) and zearalenone (0.88) reflects considerable variability between samples. This variability indicates potential contamination risk in certain batches, which is consistent with findings reported by [
27], who observed that South American maize, including Brazilian corn, is particularly susceptible to fusarium mycotoxins such as DON and fumonisins due to climate conditions favoring fungal growth. (The detailed mycotoxin contamination data of Brazilian corn are provided in
Appendix A,
Table A17).
In conclusion, while Brazilian corn appears compliant with European limits overall, continuous monitoring is essential to prevent feed safety issues, especially regarding DON and fumonisin variability. A mycotoxin management strategy adapted to tropical-origin cereals is recommended for feed manufacturers.
5.4.3. Analysis of Mycotoxin Contamination in Ukrainian Corn Imported
Analyses performed on five corn samples imported from Ukraine revealed the presence of several important mycotoxins affecting animal nutrition and feed safety: deoxynivalenol (DON), fumonisins (B1 + B2), ochratoxin A, T-2 toxin, zearalenone, and aflatoxin B1.
Deoxynivalenol (DON): The mean concentration was 456 ppb (µg/kg), ranging from 410 to 560 ppb. According to European regulations (European Commission (EC) Regulation No 1881/2006) [
25], DON is known to have immunosuppressive effects and can impair growth performance in animals [
28].
Fumonisins B1 + B2: The concentrations were stable at 200 ppb and thus well below safety thresholds, which is reassuring. Fumonisins mainly cause liver and kidney toxicity [
29].
Ochratoxin A: It was present at approximately 1.9 ppb (µg/kg), but there are no strict European limits for ochratoxin A, in animal feed,
T-2 Toxin: The mean level was 10.05 ppb, close to the maximum measured value. T-2 toxin is a highly toxic trichothecene with immunosuppressive effects. No specific European regulatory limits exist for T-2 toxin in feed, but levels below 100 ppb are generally considered of low concern [
30].
Zearalenone: The mean concentration was 4.9 ppb, with a maximum of 6.86 ppb. The European recommended maximum level for zearalenone in corn is 100 ppb [
26], well above the detected values, indicating low contamination. This mycotoxin has estrogenic effects.
Aflatoxin B1: The concentration was very low at 0.2 ppb. The European maximum limit for aflatoxin B1 in animal feed is 20 ppb, confirming minimal contamination. Aflatoxin B1 is known to be the most carcinogenic mycotoxin [
31]. (The detailed mycotoxin contamination data of Ukrainian corn are provided in
Appendix A,
Table A18).
5.5. Interpretation of the Relationship Between Water Activity and Mycotoxin Contamination
Water activity (Aw) plays a critical role in the growth of molds and subsequent production of mycotoxins in stored grains such as maize. In this study, variations in Aw values among the maize samples from different origins were observed, with Brazilian corn showing the highest average Aw (0.716), followed by Argentine corn (0.680), and Ukrainian corn with the lowest Aw (0.662).
These differences in water activity appear to be closely associated with the mycotoxin contamination profile. Indeed, Brazilian corn, which had the highest Aw, also exhibited the highest average levels of deoxynivalenol (DON), ochratoxin A, and relatively high fumonisin concentrations. In contrast, Ukrainian corn, which showed the lowest Aw, consistently presented the lowest levels of all major mycotoxins, including DON, fumonisins, and zearalenone.
This pattern supports the well-established understanding that higher Aw levels promote fungal growth and mycotoxin biosynthesis, especially under conditions of poor storage, high humidity, or inadequate drying [
32,
33]. While Aw alone does not entirely determine contamination levels—since environmental conditions, crop management, and post-harvest handling also play important roles [
34]—it remains a key predictive factor. Maintaining Aw below critical thresholds (typically < 0.70) is essential for reducing the risk of mold development and mycotoxin accumulation in maize during storage and transport [
32,
33,
34,
35].
6. Conclusions
This study provides a valuable scientific contribution by delivering a comprehensive chemical and mycotoxin profile of key imported feed ingredients—corn from Argentina, Brazil, and Ukraine; soybean meal from the USA and Argentina; and sunflower meal from Ukraine—used in animal feed production in Morocco. By analyzing and comparing the nutritional quality and contamination risks of these commodities, the research offers critical insights into feed safety, raw material sourcing, and quality control practices. The findings support evidence-based decision making for the Moroccan livestock industry and contribute to the global understanding of feed ingredient variability across different origins.
For corn, moisture content ranged from 12.63% to 14.58%, with water activity (Aw) values between 0.677 and 0.747. While nutritional values such as crude protein, fat, and starch were relatively stable and suitable for poultry feeding, Brazilian corn showed the highest average A_w (0.716), nearing the critical threshold for mold development and correlating with higher mycotoxin levels. In contrast, Ukrainian corn exhibited the lowest A_w (0.662) and consistently lower mycotoxin contamination, highlighting the role of low water activity in ensuring storage safety.
To mitigate mycotoxin-related risks, especially in poultry, the study recommends improving raw material monitoring, using antifungal preservatives or binders, and managing high-moisture batches more carefully.
For soybean meal, the American origin outperformed the Argentine in terms of crude protein, crude fat, metabolizable energy, and digestible lysine. However, statistical differences were only significant for moisture and A_w. These results align with previous studies showing better consistency and processing quality in U.S. SBM.
Sunflower meal from Ukraine showed a stable and improving nutritional profile over three years, with increased crude protein (35.97–36.99%) and metabolizable energy, and decreased fiber. Despite lower protein and amino acid content compared to SBM, SFM remains a valuable complementary protein source, particularly for layers and breeders due to its high fiber content. ME values for SFM were lower (1553–1600 kcal/kg), as expected, but consistent across years.
Regular analysis of the chemical composition of these raw materials is therefore essential to ensure feed safety, allow precise diet formulation, and optimize zootechnical performance. Moreover, new alternative sources are beginning to be explored to diversify nutrient inputs. In this regard, a recent study [
36] reports that non-industrial Moroccan hemp seed ecotypes can be included at low levels in laying hen diets without adverse effects on performance, partially replacing corn, soybean, or other oilseeds, thus opening new perspectives in feed formulation.
These findings provide valuable insights for poultry and livestock feed formulators, offering a general overview of the nutritional quality and potential risks associated with different ingredient origins. Special attention should be paid to water activity, which strongly influences the development of mycotoxins. In particular, Argentine corn may pose a risk for fumonisin and T-2 toxin contamination, while Brazilian corn may require close monitoring for DON and ochratoxin A. It is therefore essential to systematically test each incoming batch to ensure feed safety and performance.
This study successfully highlighted the nutritional advantages and limitations associated with each origin for the three raw materials, providing a scientific basis for making optimal sourcing decisions. It also lays the groundwork for future field-based research aimed at evaluating the impact of ingredient origin on animal performance. Such studies would enable a critical analysis of additional variables linking nutrients to animals, including digestibility, nutrient absorption, and overall zootechnical outcomes.