Functions and Therapeutic Use of Heat Shock Proteins in Hepatocellular Carcinoma
Abstract
1. Introduction
2. Chaperones in Hepatocellular Carcinoma
2.1. HSP110 Family
2.2. HSP90 Family
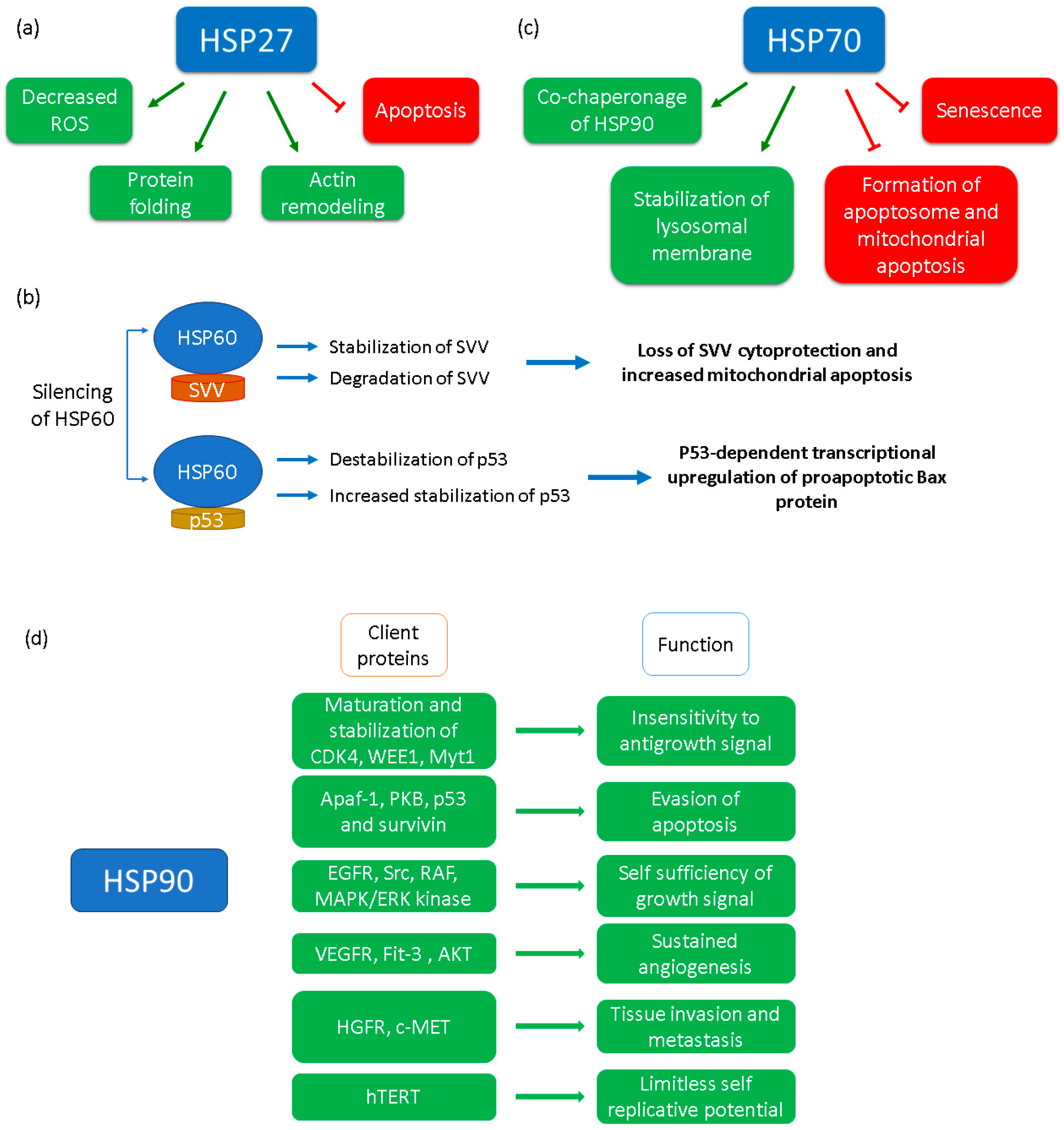
2.3. HSP70 Family
2.4. HSP60
2.5. HSP27
2.6. HSP20
2.7. Clusterin
3. Role of Chaperones in HCC Treatment and Therapeutics
3.1. HSP90 as a Therapeutic Target
3.2. HSP70 as a Therapeutic Target
3.3. HSP60 as a Therapeutic Target
3.4. HSP27 as a Therapeutic Target
3.5. Clusterin as a Therapeutic Target
| Chemicals/Inhibitors | Tested on | Functions | References |
|---|---|---|---|
| HSP27 Inhibitors | |||
| KRIBB11 | Mouse model | Inhibit tumor growth | [124,135] |
| KRIBB3 | Inhibit growth and proliferation of HCC cells | [124] | |
| 15-deoxy-Delta (12,14)-prostaglandin J (2) | Antitumor effects in HCC | [125,136] | |
| Quercetin Celastrol Triptolide | HCC cell lines Phase 2 | Inhibit HSP27 expression. | [126,127,137,138,139] |
| OGX-427 | Suppress metastasis. | [95] | |
| RP101 | Phase II trial | Bind with HSP27 and inhibits its interaction with other proteins | [122,123,140] |
| 1,3,5-trihydroxy-13,13- dimethyl-2H-pyran [7,6-b] xanthone (TDP) | Directly binds HSP60 and stimulates it to form aggregates followed by its ubiquitin-mediated proteolysis | [124,125,130,141] | |
| HSP90 inhibitors | |||
| 17AAG | Phase I/II/III clinical trial | Tumor growth arrest and reduce microvessels. | [142,143] |
| Ganetespib | Phase I/II/III clinical trials | Suppress HCC cell proliferation. | [105,144,145] |
| AUY 922 | Phase I/II Clinical Trial | Inhibit expression of HSP90 | [146] |
| SNX 2112 | HCC cell lines | Induce apoptosis. | [107,147,148] |
| PU-H71 | Phase I clinical trial | Decrease cancer growth by inactive UPR. | [106,149] |
| Geldanamycin (GA) | Phase I clinical trial | Prevent cell growth by binding to the ATP-binding site of HSP90. | [150,151,152,153] |
| Carboranylphenoxyacetanilide | Multiple cancer cell lines | Directly interacts with HSP60 to inhibit its function | [154,155] |
| Gold(III) porphyrin | Multiple cancer cell lines | Directly interacts with HSP60 to inhibit its function | [156,157] |
| 17-DMAG | Phase I clinical trial | Analogue of GA, Prevent cell growth by binding to the ATP-binding site of HSP90. | [158] |
| HSP70 inhibitors | |||
| VER155008 | Inhibit expression of HSP70 and cause cell death. | [159,160] | |
| 2-Phenylethynesulfonamide | Binds C terminal PBD of HSP70, resulting in aggregation of misfolded protein and finally apoptosis | [110,161] | |
| MKT-077 | Phase I clinical trial | disrupts ATPase domain of HSP70 | [111] |
| Peptide vaccine | Mouse model | ||
| HSP60 inhibitors | |||
| Mizoribine | HCC cell lines | inhibit the folding capacity of HSP60. | [128,162] |
| Myrtucommulone A | HCC cell lines | anti-bacterial, anti-inflammatory, anti-tumor property | [163,164,165] |
| Epolactaene | inhibits HSP60 by alkylating cys442 | [115,166] | |
| Stephacidin B, Avarainvillamide | in vitro anticancer activities | [119,167] | |
| KIRA6 | Multiple cancer cell lines | interacts with HSP60 and decreases its ATPase and folding ability. | [121] |
| KHS101 | Inhibits HSP60-dependent substrate refolding activity | [168,169] | |
| Clusterin inhibitor | |||
| OGX-011 | HCC cell lines | Suppress metastasis. | [95,170] |
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| HSP | Heat shock protein |
| HCC | Hepatocellular carcinoma |
| ATP | Adenosine triphosphate |
| PIKK | Phosphatidylinositol-3 kinase-related kinases |
| Bcl2 | B-cell lymphoma 2 |
| Bclaf | Bcl2 associated transcription factor1 |
| mTOR | Mammalian target of rapamycin |
| PI3K | Phosphoinositide 3-kinase |
| 17-AAG | 17-N-Allylamino-17-demethoxygeldanamycin |
| NBD | Nucleotide-binding domain |
| SBD | Substrate binding domain |
| NTD | N-terminal domain |
| CTD | C-terminal domain |
| TNF | Tumor necrosis factor |
| TRAP1 | TNF receptor-associated protein 1 |
| NEF | Nucleotide exchange factor |
| EMT | Epithelial to mesenchymal transition |
| ECM | Extracellular matrix |
| WASF2 | Wiskott-Aldrich syndrome family member 2 |
| HIF1 | Hypoxia-inducible factor 1 |
| MDR | Multiple drug resistance |
| ATF | Activating transcription factor |
| CHOP | C/EBP homologous protein |
| CypD | Cyclophilin D |
| FHIP | Fragile histidine triad protein |
| VEGF | Vascular endothelial growth factors |
| MMPs | Metalloproteinases |
| TCTP | Translationally controlled tumor proteins |
| MAPK | Mitogen-activated protein kinase |
| ERK | Extracellular signal-regulated kinase |
| PDIA3 | Protein disulfide isomerase A3 |
| GRP78 | Glucose-regulated protein 78 |
| CRT | Calreticulin |
References
- Jafri, W.; Kamran, M. Hepatocellular Carcinoma in Asia: A Challenging Situation. Euroasian J. Hepatogastroenterol. 2019, 9, 27–33. [Google Scholar] [CrossRef] [PubMed]
- Omata, M.; Cheng, A.-L.; Kokudo, N.; Kudo, M.; Lee, J.M.; Jia, J.; Tateishi, R.; Han, K.H.; Chawla, Y.K.; Shiina, S.; et al. Asia-Pacific clinical practice guidelines on the management of hepatocellular carcinoma: A 2017 update. Hepatol. Int. 2017, 11, 317–370. [Google Scholar] [CrossRef]
- Camberg, J.L.; Doyle, S.M.; Johnston, D.M.; Wickner, S. Molecular Chaperones. In Brenner’s Encyclopedia of Genetics, 2nd ed.; Maloy, S., Hughes, K., Eds.; Academic Press: San Diego, CA, USA, 2013; pp. 456–460. [Google Scholar] [CrossRef]
- Deuerling, E.; Gamerdinger, M.; Kreft, S.G. Chaperone Interactions at the Ribosome. Cold Spring Harb. Perspect. Biol. 2019, 11, a033977. [Google Scholar] [CrossRef]
- Hartl, F.U.; Bracher, A.; Hayer-Hartl, M. Molecular chaperones in protein folding and proteostasis. Nature 2011, 475, 324–332. [Google Scholar] [CrossRef]
- Saibil, H. Chaperone machines for protein folding, unfolding and disaggregation. Nat. Rev. Mol. Cell Biol. 2013, 14, 630–642. [Google Scholar] [CrossRef] [PubMed]
- Pearl, L.H. Review: The HSP90 molecular chaperone-an enigmatic ATPase. Biopolymers 2016, 105, 594–607. [Google Scholar] [CrossRef]
- Wankhede, N.L.; Kale, M.B.; Upaganlawar, A.B.; Taksande, B.G.; Umekar, M.J.; Behl, T.; Abdellatif, A.A.H.; Bhaskaran, P.M.; Dachani, S.R.; Sehgal, A. Involvement of molecular chaperone in protein-misfolding brain diseases. Biomed. Pharmacother. 2022, 147, 112647. [Google Scholar] [CrossRef] [PubMed]
- Chu, Y.; Kordower, J.H. Age-associated increases of α-synuclein in monkeys and humans are associated with nigrostriatal dopamine depletion: Is this the target for Parkinson’s disease? Neurobiol. Dis. 2007, 25, 134–149. [Google Scholar] [CrossRef]
- Kuzuhara, S.; Mori, H.; Izumiyama, N.; Yoshimura, M.; Ihara, Y. Lewy bodies are ubiquitinated. Acta Neuropathol. 1988, 75, 345–353. [Google Scholar] [CrossRef]
- Hamley, I.W. The Amyloid Beta Peptide: A Chemist’s Perspective. Role in Alzheimer’s and Fibrillization. Chem. Rev. 2012, 112, 5147–5192. [Google Scholar] [CrossRef]
- Hoshino, T.; Murao, N.; Namba, T.; Takehara, M.; Adachi, H.; Katsuno, M.; Sobue, G.; Matsushima, T.; Suzuki, T.; Mizushima, T. Suppression of Alzheimer’s Disease-Related Phenotypes by Expression of Heat Shock Protein 70 in Mice. J. Neurosci. 2011, 31, 5225–5234. [Google Scholar] [CrossRef] [PubMed]
- Takano, M.; Yamashita, T.; Nagano, K.; Otani, M.; Maekura, K.; Kamada, H.; Tsunoda, S.; Tsutsumi, Y.; Tomiyama, T.; Mori, H.; et al. Proteomic analysis of the hippocampus in Alzheimer’s disease model mice by using two-dimensional fluorescence difference in gel electrophoresis. Neurosci. Lett. 2013, 534, 85–89. [Google Scholar] [CrossRef] [PubMed]
- Liu, J.; Shinobu, L.A.; Ward, C.M.; Young, D.; Cleveland, D.W. Elevation of the Hsp70 chaperone does not effect toxicity in mouse models of familial amyotrophic lateral sclerosis. J. Neurochem. 2005, 93, 875–882. [Google Scholar] [CrossRef] [PubMed]
- Rosen, D.R.; Siddique, T.; Patterson, D.; Figlewicz, D.A.; Sapp, P.; Hentati, A.; Donaldson, D.; Goto, J.; O’Regan, J.P.; Deng, H.X. Mutations in Cu/Zn superoxide dismutase gene are associated with familial amyotrophic lateral sclerosis. Nature 1993, 362, 59–62. [Google Scholar] [CrossRef] [PubMed]
- Hansson, G.K. Inflammation, Atherosclerosis, and Coronary Artery Disease. N. Engl. J. Med. 2005, 352, 1685–1695. [Google Scholar] [CrossRef] [PubMed]
- Jiang, Y.; Lv, H.; Liao, M.; Xu, X.; Huang, S.; Tan, H.; Peng, T.; Zhang, Y.; Li, H. GRP78 counteracts cell death and protein aggregation caused by mutant huntingtin proteins. Neurosci. Lett. 2012, 516, 182–187. [Google Scholar] [CrossRef]
- Orr, H.T.; Zoghbi, H.Y. Trinucleotide Repeat Disorders. Annu. Rev. Neurosci. 2007, 30, 575–621. [Google Scholar] [CrossRef]
- Vos, M.J.; Zijlstra, M.P.; Kanon, B.; van Waarde-Verhagen, M.A.; Brunt, E.R.; Oosterveld-Hut, H.M.; Carra, S.; Sibon, O.C.; Kampinga, H.H. HSPB7 is the most potent polyQ aggregation suppressor within the HSPB family of molecular chaperones. Hum. Mol. Genet. 2010, 19, 4677–4693. [Google Scholar] [CrossRef]
- Ciocca, D.R.; Calderwood, S.K. Heat shock proteins in cancer: Diagnostic, prognostic, predictive, and treatment implications. Cell Stress Chaperones 2005, 10, 86–103. [Google Scholar] [CrossRef]
- Workman, P.; Burrows, F.; Neckers, L.; Rosen, N. Drugging the cancer chaperone HSP90: Combinatorial therapeutic exploitation of oncogene addiction and tumor stress. Ann. N. Y. Acad. Sci. 2007, 1113, 202–216. [Google Scholar] [CrossRef] [PubMed]
- Kubelka, J.; Hofrichter, J.; Eaton, W.A. The protein folding “speed limit”. Curr. Opin. Struct. Biol. 2004, 14, 76–88. [Google Scholar] [CrossRef]
- Sučec, I.; Bersch, B.; Schanda, P. How do Chaperones Bind (Partly) Unfolded Client Proteins? Front. Mol. Biosci. 2021, 8, 762005. [Google Scholar] [CrossRef]
- Mayer, M.P. Hsp70 chaperone dynamics and molecular mechanism. Trends Biochem. Sci. 2013, 38, 507–514. [Google Scholar] [CrossRef]
- Jakob, U.; Gaestel, M.; Engel, K.; Buchner, J. Small heat shock proteins are molecular chaperones. J. Biol. Chem. 1993, 268, 1517–1520. [Google Scholar] [CrossRef]
- Kosmaoglou, M.; Schwarz, N.; Bett, J.S.; Cheetham, M.E. Molecular chaperones and photoreceptor function. Prog. Retin. Eye Res. 2008, 27, 434–449. [Google Scholar] [CrossRef] [PubMed]
- Oh, H.J.; Easton, D.; Murawski, M.; Kaneko, Y.; Subjeck, J.R. The Chaperoning Activity of hsp110: Identification of Functional Domains by Use of Targeted Deletions. J. Biol. Chem. 1999, 274, 15712–15718. [Google Scholar] [CrossRef] [PubMed]
- Kampinga, H.H.; Hageman, J.; Vos, M.J.; Kubota, H.; Tanguay, R.M.; Bruford, E.A.; Cheetham, M.E.; Chen, B.; Hightower, L.E. Guidelines for the nomenclature of the human heat shock proteins. Cell Stress Chaperones 2009, 14, 105–111. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Sarbeng, E.B.; Vorvis, C.; Kumar, D.P.; Zhou, L.; Liu, Q. Unique Peptide Substrate Binding Properties of 110-kDa Heat-shock Protein (Hsp110) Determine Its Distinct Chaperone Activity. J. Biol. Chem. 2012, 287, 5661–5672. [Google Scholar] [CrossRef] [PubMed]
- Kamran, M.Z.; Patil, P.; Gude, R.P. Role of STAT3 in Cancer Metastasis and Translational Advances. BioMed Res. Int. 2013, 2013, e421821. [Google Scholar] [CrossRef] [PubMed]
- Saini, J.; Sharma, P.K. Clinical, Prognostic and Therapeutic Significance of Heat Shock Proteins in Cancer. Curr. Drug Targets 2018, 19, 1478–1490. [Google Scholar] [CrossRef] [PubMed]
- Manjili, M.H.; Park, J.; Facciponte, J.G.; Subjeck, J.R. HSP110 induces “danger signals” upon interaction with antigen presenting cells and mouse mammary carcinoma. Immunobiology 2005, 210, 295–303. [Google Scholar] [CrossRef] [PubMed]
- Duennwald, M.L.; Echeverria, A.; Shorter, J. Small heat shock proteins potentiate amyloid dissolution by protein disaggregases from yeast and humans. PLoS Biol. 2012, 10, e1001346. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Shen, F.; Yin, Y.X.; Yang, Y.Y.; Xiang, D.J.; Chen, Q. Increased expression of heat shock protein 27 correlates with peritoneal metastasis in epithelial ovarian cancer. Reprod. Sci. 2012, 19, 748–753. [Google Scholar] [CrossRef]
- Picard, D. Heat-shock protein 90, a chaperone for folding and regulation. Cell. Mol. Life Sci. 2002, 59, 1640–1648. [Google Scholar] [CrossRef] [PubMed]
- Chen, B.; Zhong, D.; Monteiro, A. Comparative genomics and evolution of the HSP90 family of genes across all kingdoms of organisms. BMC Genom. 2006, 7, 156. [Google Scholar] [CrossRef]
- Harris, S.F.; Shiau, A.K.; Agard, D.A. The crystal structure of the carboxy-terminal dimerization domain of htpG, the Escherichia coli Hsp90, reveals a potential substrate binding site. Structure 2004, 12, 1087–1097. [Google Scholar] [CrossRef]
- Minami, Y.; Kimura, Y.; Kawasaki, H.; Suzuki, K.; Yahara, I. The carboxy-terminal region of mammalian HSP90 is required for its dimerization and function in vivo. Mol. Cell. Biol. 1994, 14, 1459–1464. [Google Scholar]
- Prodromou, C.; Roe, S.M.; O’Brien, R.; Ladbury, J.E.; Piper, P.W.; Pearl, L.H. Identification and structural characterization of the ATP/ADP-binding site in the Hsp90 molecular chaperone. Cell 1997, 90, 65–75. [Google Scholar] [CrossRef] [PubMed]
- Zuehlke, A.; Johnson, J.L. Hsp90 and co-chaperones twist the functions of diverse client proteins. Biopolymers 2010, 93, 211–217. [Google Scholar] [CrossRef]
- Schueller, G.; Kettenbach, J.; Sedivy, R.; Bergmeister, H.; Stift, A.; Fried, J.; Gnant, M.; Lammer, J. Expression of heat shock proteins in human hepatocellular carcinoma after radiofrequency ablation in an animal model. Oncol. Rep. 2004, 12, 495–499. [Google Scholar] [CrossRef] [PubMed]
- Nouri-Vaskeh, M.; Alizadeh, L.; Hajiasgharzadeh, K.; Mokhtarzadeh, A.; Halimi, M.; Baradaran, B. The role of HSP90 molecular chaperones in hepatocellular carcinoma. J. Cell Physiol. 2020, 235, 9110–9120. [Google Scholar] [CrossRef] [PubMed]
- Tian, L.-Y.; Smit, D.J.; Jücker, M. The Role of PI3K/AKT/mTOR Signaling in Hepatocellular Carcinoma Metabolism. Int. J. Mol. Sci. 2023, 24, 2652. [Google Scholar] [CrossRef] [PubMed]
- Yu, S.; Wang, X.; Dou, N.; Zhou, J.; Gao, Y.; Li, Y. B-cell lymphoma-2-associated transcription factor 1 is overexpressed and contributes to sorafenib resistance in hepatocellular carcinoma. Hepatol. Res. 2019, 49, 1329–1340. [Google Scholar] [CrossRef] [PubMed]
- Gao, M.; Geng, X.-P.; Xiang, H.-P. HSP90 and SIRT3 expression in hepatocellular carcinoma and their effect on invasive capability of human hepatocellular carcinoma cells. Asian Pac. J. Trop. Med. 2015, 8, 305–308. [Google Scholar] [CrossRef] [PubMed]
- Cheng, W.; Ainiwaer, A.; Xiao, L.; Cao, Q.; Wu, G.; Yang, Y.; Mao, R.; Bao, Y. Role of the novel HSP90 inhibitor AUY922 in hepatocellular carcinoma: Potential for therapy. Mol. Med. Rep. 2015, 12, 2451–2456. [Google Scholar] [CrossRef] [PubMed]
- Sun, E.J.; Wankell, M.; Palamuthusingam, P.; McFarlane, C.; Hebbard, L. Targeting the PI3K/Akt/mTOR Pathway in Hepatocellular Carcinoma. Biomedicines 2021, 9, 1639. [Google Scholar] [CrossRef]
- Akt Pathway|Thermo Fisher Scientific-IN. Available online: https://www.thermofisher.com/in/en/home/life-science/antibodies/antibodies-learning-center/antibodies-resource-library/cell-signaling-pathways/akt-signaling-pathway.html (accessed on 25 January 2024).
- Li, Z.-N.; Luo, Y. HSP90 inhibitors and cancer: Prospects for use in targeted therapies (Review). Oncol. Rep. 2023, 49, 6. [Google Scholar] [CrossRef]
- Balchin, D.; Hayer-Hartl, M.; Hartl, F.U. In vivo aspects of protein folding and quality control. Science 2016, 353, aac4354. [Google Scholar] [CrossRef]
- Mayer, M.P.; Bukau, B. Hsp70 chaperones: Cellular functions and molecular mechanism. Cell. Mol. Life Sci. 2005, 62, 670–684. [Google Scholar] [CrossRef]
- Mogk, A.; Kummer, E.; Bukau, B. Cooperation of Hsp70 and Hsp100 chaperone machines in protein disaggregation. Front. Mol. Biosci. 2015, 2, 22. [Google Scholar] [CrossRef]
- Li, H.; Li, Y.; Liu, D.; Sun, H.; Su, D.; Yang, F.; Liu, J. Extracellular HSP70/HSP70-PCs Promote Epithelial-Mesenchymal Transition of Hepatocarcinoma Cells. PLoS ONE 2013, 8, e84759. [Google Scholar] [CrossRef] [PubMed]
- Sossey-Alaoui, K.; Li, X.; Ranalli, T.A.; Cowell, J.K. WAVE3-mediated cell migration and lamellipodia formation are regulated downstream of phosphatidylinositol 3-kinase. J. Biol. Chem. 2005, 280, 21748–21755. [Google Scholar] [CrossRef] [PubMed]
- Teng, Y.; Ren, M.Q.; Cheney, R.; Sharma, S.; Cowell, J.K. Inactivation of the WASF3 gene in prostate cancer cells leads to suppression of tumorigenicity and metastases. Br. J. Cancer 2010, 103, 1066–1075. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.-H.; Lin, K.-H.; Yu, J.-S.; Wu, T.J.; Lee, W.C.; Chao, C.C.; Pan, T.L.; Yeh, C.T. Targeting HSP60 by subcutaneous injections of jetPEI/HSP60-shRNA destabilizes cytoplasmic survivin and inhibits hepatocellular carcinoma growth. Mol. Carcinog. 2018, 57, 1087–1101. [Google Scholar] [CrossRef] [PubMed]
- Kim, T.-K.; Na, H.J.; Lee, W.R.; Jeoung, M.H.; Lee, S. Heat shock protein 70-1A is a novel angiogenic regulator. Biochem. Biophys. Res. Commun. 2016, 469, 222–228. [Google Scholar] [CrossRef] [PubMed]
- Wang, C.; Zhang, Y.; Guo, K.; Wang, N.; Jin, H.; Liu, Y.; Qin, W. Heat shock proteins in hepatocellular carcinoma: Molecular mechanism and therapeutic potential. Int. J. Cancer 2016, 138, 1824–1834. [Google Scholar] [CrossRef]
- Yaglom, J.A.; Wang, Y.; Li, A.; Li, Z.; Monti, S.; Alexandrov, I.; Lu, X.; Sherman, M.Y. Cancer cell responses to Hsp70 inhibitor JG-98: Comparison with Hsp90 inhibitors and finding synergistic drug combinations. Sci. Rep. 2018, 8, 3010. [Google Scholar] [CrossRef]
- Kumar, S.; Stokes, J.; Singh, U.P.; Scissum Gunn, K.; Acharya, A.; Manne, U.; Mishra, M. Targeting Hsp70: A possible therapy for cancer. Cancer Lett. 2016, 374, 156–166. [Google Scholar] [CrossRef]
- Henderson, B.; Fares, M.A.; Lund, P.A. Chaperonin 60: A paradoxical, evolutionarily conserved protein family with multiple moonlighting functions. Biol. Rev. 2013, 88, 955–987. [Google Scholar] [CrossRef]
- Jindal, S.; Dudani, A.K.; Singh, B.; Harley, C.B.; Gupta, R.S. Primary Structure of a Human Mitochondrial Protein Homologous to the Bacterial and Plant Chaperonins and to the 65-Kilodalton Mycobacterial Antigen. Mol. Cell. Biol. 1989, 9, 2279–2283. [Google Scholar]
- Ostermann, J.; Horwich, A.L.; Neupert, W.; Hartl, F.-U. Protein folding in mitochondria requires complex formation with hsp60 and ATP hydrolysis. Nature 1989, 341, 125–130. [Google Scholar] [CrossRef] [PubMed]
- Kalderon, B.; Kogan, G.; Bubis, E.; Pines, O. Cytosolic Hsp60 Can Modulate Proteasome Activity in Yeast. J. Biol. Chem. 2015, 290, 3542–3551. [Google Scholar] [CrossRef] [PubMed]
- Soltys, B.J.; Gupta, R.S. Cell Surface Localization of the 60 Kda Heat Shock Chaperonin Protein (hsp60) in Mammalian Cells. Cell Biol. Int. 1997, 21, 315–320. [Google Scholar] [CrossRef] [PubMed]
- Lim, S.O.; Park, S.-J.; Kim, W.; Park, S.G.; Kim, H.J.; Kim, Y.I.; Sohn, T.S.; Noh, J.H.; Jung, G. Proteome Analysis of Hepatocellular Carcinoma. Biochem. Biophys. Res. Commun. 2002, 291, 1031–1037. [Google Scholar] [CrossRef] [PubMed]
- Hoter, A.; Rizk, S.; Naim, H.Y. Heat Shock Protein 60 in Hepatocellular Carcinoma: Insights and Perspectives. Front. Mol. Biosci. 2020, 7, 60. [Google Scholar] [CrossRef]
- Chen, Y.; Li, X.; Shao, S. The Clinical Value of HSP60 in Digestive System Cancers: A Systematic Review and Meta-Analysis. Clin. Lab. 2019, 65, 1937. [Google Scholar] [CrossRef]
- Zhang, J.; Zhou, X.; Chang, H.; Huang, X.; Guo, X.; Du, X.; Tian, S.; Wang, L.; Lyv, Y.; Yuan, P.; et al. Hsp60 exerts a tumor suppressor function by inducing cell differentiation and inhibiting invasion in hepatocellular carcinoma. Oncotarget 2016, 7, 68976–68989. [Google Scholar] [CrossRef]
- Huang, Y.-H.; Wang, F.-S.; Wang, P.-W.; Lin, H.-Y.; Luo, S.-D.; Yang, Y.-L. Heat Shock Protein 60 Restricts Release of Mitochondrial dsRNA to Suppress Hepatic Inflammation and Ameliorate Non-Alcoholic Fatty Liver Disease in Mice. Int. J. Mol. Sci. 2022, 23, 577. [Google Scholar] [CrossRef]
- Deocaris, C.C.; Widodo, N.; Shrestha, B.G.; Kaur, K.; Ohtaka, M.; Yamasaki, K.; Kaul, S.C.; Wadhwa, R. Mortalin sensitizes human cancer cells to MKT-077-induced senescence. Cancer Lett. 2007, 252, 259–269. [Google Scholar] [CrossRef]
- Ghosh, J.C.; Siegelin, M.D.; Dohi, T.; Altieri, D.C. Heat Shock Protein 60 Regulation of the Mitochondrial Permeability Transition Pore in Tumor Cells. Cancer Res. 2010, 70, 8988–8993. [Google Scholar] [CrossRef]
- Rodríguez, M.E.; Cogno, I.S.; Sanabria, L.S.M.; Morán, Y.S.; Rivarola, V.A. Heat shock proteins in the context of photodynamic therapy: Autophagy, apoptosis and immunogenic cell death. Photochem. Photobiol. Sci. 2016, 15, 1090–1102. [Google Scholar] [CrossRef]
- Tsai, Y.-P.; Yang, M.-H.; Huang, C.-H.; Chang, S.Y.; Chen, P.M.; Liu, C.J.; Teng, S.C.; Wu, K.J. Interaction between HSP60 and β-catenin promotes metastasis. Carcinogenesis 2009, 30, 1049–1057. [Google Scholar] [CrossRef]
- Mehlen, P.; Mehlen, A.; Godet, J.; Arrigo, A.P. hsp27 as a switch between differentiation and apoptosis in murine embryonic stem cells. J. Biol. Chem. 1997, 272, 31657–31665. [Google Scholar] [CrossRef] [PubMed]
- Vidyasagar, A.; Wilson, N.A.; Djamali, A. Heat shock protein 27 (HSP27): Biomarker of disease and therapeutic target. Fibrogenesis Tissue Repair 2012, 5, 7. [Google Scholar] [CrossRef] [PubMed]
- Baylot, V.; Katsogiannou, M.; Andrieu, C.; Taieb, D.; Acunzo, J.; Giusiano, S.; Fazli, L.; Gleave, M.; Garrido, C.; Rocchi, P. Targeting TCTP as a New Therapeutic Strategy in Castration-resistant Prostate Cancer. Mol. Ther. 2012, 20, 2244–2256. [Google Scholar] [CrossRef] [PubMed]
- Voss, O.H.; Batra, S.; Kolattukudy, S.J.; Gonzalez-Mejia, M.E.; Smith, J.B.; Doseff, A.I. Binding of Caspase-3 Prodomain to Heat Shock Protein 27 Regulates Monocyte Apoptosis by Inhibiting Caspase-3 Proteolytic Activation. J. Biol. Chem. 2007, 282, 25088–25099. [Google Scholar] [CrossRef] [PubMed]
- Katsogiannou, M.; Andrieu, C.; Baylot, V.; Baudot, A.; Dusetti, N.J.; Gayet, O.; Finetti, P.; Garrido, C.; Birnbaum, D.; Bertucci, F.; et al. The functional landscape of Hsp27 reveals new cellular processes such as DNA repair and alternative splicing and proposes novel anticancer targets. Mol. Cell. Proteom. 2014, 13, 3585–3601. [Google Scholar] [CrossRef]
- Arrigo, A.P. sHsp as novel regulators of programmed cell death and tumorigenicity. Pathol. Biol. 2000, 48, 280–288. [Google Scholar]
- Eto, D.; Hisaka, T.; Horiuchi, H.; Uchida, S.; Ishikawa, H.; Kawashima, Y.; Kinugasa, T.; Nakashima, O.; Yano, H.; Okuda, K.; et al. Expression of HSP27 in Hepatocellular Carcinoma. Anticancer Res. 2016, 36, 3775–3779. [Google Scholar] [PubMed]
- Pavan, S.; Musiani, D.; Torchiaro, E.; Migliardi, G.; Gai, M.; Di Cunto, F.; Erriquez, J.; Olivero, M.; Di Renzo, M.F. HSP27 is required for invasion and metastasis triggered by hepatocyte growth factor. Int. J. Cancer 2014, 134, 1289–1299. [Google Scholar] [CrossRef]
- Cordonnier, T.; Bishop, J.L.; Shiota, M.; Nip, K.M.; Thaper, D.; Vahid, S.; Heroux, D.; Gleave, M.; Zoubeidi, A. Hsp27 regulates EGF/β-catenin mediated epithelial to mesenchymal transition in prostate cancer. Int. J. Cancer 2015, 136, E496–E507. [Google Scholar] [CrossRef]
- Matsushima-Nishiwaki, R.; Adachi, S.; Yoshioka, T.; Yasuda, E.; Yamagishi, Y.; Matsuura, J.; Muko, M.; Iwamura, R.; Noda, T.; Toyoda, H. Suppression by heat shock protein 20 of hepatocellular carcinoma cell proliferation via inhibition of the mitogen-activated protein kinases and AKT pathways. J. Cell. Biochem. 2011, 112, 3430–3439. [Google Scholar] [CrossRef]
- Matsushima-Nishiwaki, R.; Toyoda, H.; Nagasawa, T.; Yasuda, E.; Chiba, N.; Okuda, S.; Maeda, A.; Kaneoka, Y.; Kumada, T.; Kozawa, O. Phosphorylated Heat Shock Protein 20 (HSPB6) Regulates Transforming Growth Factor-α-Induced Migration and Invasion of Hepatocellular Carcinoma Cells. PLoS ONE 2016, 11, e0151907. [Google Scholar] [CrossRef]
- Koltai, T. Clusterin: A key player in cancer chemoresistance and its inhibition. Onco Targets Ther. 2014, 7, 447–456. [Google Scholar] [CrossRef]
- Nafee, A.M.; Pasha, H.F.; El Aal, S.M.A.; Mostafa, N.A. Clinical significance of serum clusterin as a biomarker for evaluating diagnosis and metastasis potential of viral-related hepatocellular carcinoma. Clin. Biochem. 2012, 45, 1070–1074. [Google Scholar] [CrossRef]
- Kang, Y.K.; Hong, S.W.; Lee, H.; Kim, W.H. Overexpression of clusterin in human hepatocellular carcinoma. Hum. Pathol. 2004, 35, 1340–1346. [Google Scholar] [CrossRef]
- De Maio, A. Heat shock proteins: Facts, thoughts, and dreams. Shock 1999, 11, 1–12. [Google Scholar] [CrossRef]
- Fan, G.-C. Role of heat shock proteins in stem cell behavior. Prog. Mol. Biol. Transl. Sci. 2012, 111, 305–322. [Google Scholar] [PubMed]
- Jolly, C.; Morimoto, R.I. Role of the heat shock response and molecular chaperones in oncogenesis and cell death. J. Natl. Cancer Inst. 2000, 92, 1564–1572. [Google Scholar] [CrossRef] [PubMed]
- Jee, H. Size dependent classification of heat shock proteins: A mini-review. J. Exerc. Rehabil. 2016, 12, 255–259. [Google Scholar] [CrossRef] [PubMed]
- Karademir, B.; Sari-Kaplan, G. Heat Shock Protein (HSP). In Encyclopedia of Signaling Molecules; Choi, S., Ed.; Springer: New York, NY, USA, 2016; pp. 1–10. [Google Scholar] [CrossRef]
- Yang, S.; Xiao, H.; Cao, L. Recent advances in heat shock proteins in cancer diagnosis, prognosis, metabolism and treatment. Biomed. Pharmacother. 2021, 142, 112074. [Google Scholar] [CrossRef] [PubMed]
- Lelj-Garolla, B.; Kumano, M.; Beraldi, E.; Nappi, L.; Rocchi, P.; Ionescu, D.N.; Fazli, L.; Zoubeidi, A.; Gleave, M.E. Hsp27 Inhibition with OGX-427 Sensitizes Non–Small Cell Lung Cancer Cells to Erlotinib and Chemotherapy. Mol. Cancer Ther. 2015, 14, 1107–1116. [Google Scholar] [CrossRef] [PubMed]
- Shevtsov, M.; Multhoff, G. Heat Shock Protein-Peptide and HSP-Based Immunotherapies for the Treatment of Cancer. Front. Immunol. 2016, 7, 171. [Google Scholar] [CrossRef] [PubMed]
- Nylandsted, J.; Brand, K.; Jäättelä, M. Heat shock protein 70 is required for the survival of cancer cells. Ann. N. Y. Acad. Sci. 2000, 926, 122–125. [Google Scholar] [CrossRef]
- Kondo, R.; Ishino, K.; Wada, R.; Takata, H.; Peng, W.X.; Kudo, M.; Kure, S.; Kaneya, Y.; Taniai, N.; Yoshida, H.; et al. Downregulation of protein disulfide-isomerase A3 expression inhibits cell proliferation and induces apoptosis through STAT3 signaling in hepatocellular carcinoma. Int. J. Oncol. 2019, 54, 1409–1421. [Google Scholar] [CrossRef]
- Feng, R.; Ye, J.; Zhou, C.; Qi, L.; Fu, Z.; Yan, B.; Liang, Z.; Li, R.; Zhai, W. Calreticulin down-regulation inhibits the cell growth, invasion and cell cycle progression of human hepatocellular carcinoma cells. Diagn. Pathol. 2015, 10, 149. [Google Scholar] [CrossRef]
- Lee, A.S. Glucose-regulated proteins in cancer: Molecular mechanisms and therapeutic potential. Nat. Rev. Cancer 2014, 14, 263–276. [Google Scholar] [CrossRef]
- Chen, W.-T.; Zhu, G.; Pfaffenbach, K.; Kanel, G.; Stiles, B.; Lee, A.S. GRP78 as a regulator of liver steatosis and cancer progression mediated by loss of the tumor suppressor PTEN. Oncogene 2014, 33, 4997–5005. [Google Scholar] [CrossRef]
- Cho, W.C.S. Contribution of oncoproteomics to cancer biomarker discovery. Mol. Cancer 2007, 6, 25. [Google Scholar] [CrossRef]
- Liu, H.-Q.; Sun, L.-X.; Yu, L.; Sun, L.C.; Yang, Z.H.; Shu, X.; Ran, Y.L. HSP90, as a functional target antigen of a mAb 11C9, promotes stemness and tumor progression in hepatocellular carcinoma. Stem Cell Res. Ther. 2023, 14, 273. [Google Scholar] [CrossRef]
- Jego, G.; Hazoumé, A.; Seigneuric, R.; Garrido, C. Targeting heat shock proteins in cancer. Cancer Lett. 2013, 332, 275–285. [Google Scholar] [CrossRef] [PubMed]
- Goyal, L.; Wadlow, R.C.; Blaszkowsky, L.S.; Wolpin, B.M.; Abrams, T.A.; McCleary, N.J.; Sheehan, S.; Sundaram, E.; Karol, M.D.; Chen, J.; et al. A phase I and pharmacokinetic study of ganetespib (STA-9090) in advanced hepatocellular carcinoma. Investig. New Drugs 2015, 33, 128–137. [Google Scholar] [CrossRef] [PubMed]
- Breinig, M.; Caldas-Lopes, E.; Goeppert, B.; Malz, M.; Rieker, R.; Bergmann, F.; Schirmacher, P.; Mayer, M.; Chiosis, G.; Kern, M.A. Targeting heat shock protein 90 with non-quinone inhibitors: A novel chemotherapeutic approach in human hepatocellular carcinoma. Hepatology 2009, 50, 102–112. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Wang, Y.; Chen, Z.; Fu, L.; Wang, S.; Zhang, X.; Zhang, P.; Lu, X.; Jie, H.; Li, M.; et al. Hsp90 Inhibitor SNX-2112 Enhances TRAIL-Induced Apoptosis of Human Cervical Cancer Cells via the ROS-Mediated JNK-p53-Autophagy-DR5 Pathway. Oxid. Med. Cell. Longev. 2019, 2019, 9675450. [Google Scholar] [CrossRef] [PubMed]
- Leu, J.I.-J.; Pimkina, J.; Frank, A.; Murphy, M.E.; George, D.L. A Small Molecule Inhibitor of Inducible Heat Shock Protein 70. Mol. Cell 2009, 36, 15–27. [Google Scholar] [CrossRef] [PubMed]
- Wadhwa, R.; Sugihara, T.; Yoshida, A.; Nomura, H.; Reddel, R.R.; Simpson, R.; Maruta, H.; Kaul, S.C. Selective Toxicity of MKT-077 to Cancer Cells Is Mediated by Its Binding to the hsp70 Family Protein mot-2 and Reactivation of p53 Function. Cancer Res. 2000, 60, 6818–6821. [Google Scholar]
- Gyrd-Hansen, M.; Nylandsted, J.; Jäättelä, M. Heat shock protein 70 promotes cancer cell viability by safeguarding lysosomal integrity. Cell Cycle 2004, 3, 1484–1485. [Google Scholar] [CrossRef]
- Britten, C.D.; Rowinsky, E.K.; Baker, S.D.; Weiss, G.R.; Smith, L.; Stephenson, J.; Rothenberg, M.; Smetzer, L.; Cramer, J.; Collins, W.; et al. A phase I and pharmacokinetic study of the mitochondrial-specific rhodacyanine dye analog MKT 077. Clin. Cancer Res. 2000, 6, 42–49. [Google Scholar]
- Ge, C.; Xing, Y.; Wang, Q.; Xiao, W.; Lu, Y.; Hu, X.; Gao, Z.; Xu, M.; Ma, Y.; Cao, R.; et al. Improved efficacy of therapeutic vaccination with dendritic cells pulsed with tumor cell lysate against hepatocellular carcinoma by introduction of 2 tandem repeats of microbial HSP70 peptide epitope 407-426 and OK-432. Int. Immunopharmacol. 2011, 11, 2200–2207. [Google Scholar] [CrossRef]
- Nakamura, H.; Minegishi, H. HSP60 as a Drug Target. Curr. Pharm. Des. 2013, 19, 441–451. [Google Scholar] [CrossRef]
- Mizuno, K.; Tsujino, M.; Takada, M.; Hayashi, M.; Atsumi, K. Studies on bredinin. I. Isolation, characterization and biological properties. J. Antibiot. 1974, 27, 775–782. [Google Scholar] [CrossRef]
- Nagumo, Y.; Kakeya, H.; Shoji, M.; Hayashi, Y.; Dohmae, N.; Osada, H. Epolactaene binds human Hsp60 Cys442 resulting in the inhibition of chaperone activity. Biochem. J. 2005, 387, 835–840. [Google Scholar] [CrossRef] [PubMed]
- Wiechmann, K.; Müller, H.; König, S.; Wielsch, N.; Svatoš, A.; Jauch, J.; Werz, O. Mitochondrial Chaperonin HSP60 Is the Apoptosis-Related Target for Myrtucommulone. Cell Chem. Biol. 2017, 24, 614–623.e6. [Google Scholar] [CrossRef]
- Nobili, S.; Mini, E.; Landini, I.; Gabbiani, C.; Casini, A.; Messori, L. Gold compounds as anticancer agents: Chemistry, cellular pharmacology, and preclinical studies: Gold compounds as anticancer agents. Med. Res. Rev. 2010, 30, 550–580. [Google Scholar] [CrossRef]
- Izgi, K.; Iskender, B.; Jauch, J.; Sezen, S.; Cakir, M.; Charpentier, M.; Canatan, H.; Sakalar, C. Myrtucommulone-A Induces both Extrinsic and Intrinsic Apoptotic Pathways in Cancer Cells. J. Biochem. Mol. Toxicol. 2015, 29, 432–439. [Google Scholar] [CrossRef]
- Qian-Cutrone, J.; Huang, S.; Shu, Y.Z.; Vyas, D.; Fairchild, C.; Menendez, A.; Krampitz, K.; Dalterio, R.; Klohr, S.E.; Gao, Q. Stephacidin A and B: Two Structurally Novel, Selective Inhibitors of the Testosterone-Dependent Prostate LNCaP Cells. J. Am. Chem. Soc. 2002, 124, 14556–14557. [Google Scholar] [CrossRef]
- Wulff, J.E.; Siegrist, R.; Myers, A.G. The Natural Product Avrainvillamide Binds to the Oncoprotein Nucleophosmin. J. Am. Chem. Soc. 2007, 129, 14444–14451. [Google Scholar] [CrossRef]
- Rufo, N.; Korovesis, D.; Van Eygen, S.; Derua, R.; Garg, A.D.; Finotello, F.; Vara-Perez, M.; Rožanc, J.; Dewaele, M.; de Witte, P.A.; et al. Stress-induced inflammation evoked by immunogenic cell death is blunted by the IRE1α kinase inhibitor KIRA6 through HSP60 targeting. Cell Death Differ. 2022, 29, 230–245. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, J.-C.; Tuukkanen, A.; Schroeder, M.; Fahrig, T.; Fahrig, R. RP101 (brivudine) binds to heat shock protein HSP27 (HSPB1) and enhances survival in animals and pancreatic cancer patients. J. Cancer Res. Clin. Oncol. 2011, 137, 1349–1361. [Google Scholar] [CrossRef] [PubMed]
- Heinrich, J.C.; Donakonda, S.; Haupt, V.J.; Lennig, P.; Zhang, Y.; Schroeder, M. New HSP27 inhibitors efficiently suppress drug resistance development in cancer cells. Oncotarget 2016, 7, 68156–68169. [Google Scholar] [CrossRef]
- Shin, K.D.; Yoon, Y.J.; Kang, Y.-R.; Son, K.H.; Kim, H.M.; Kwon, B.M.; Han, D.C. KRIBB3, a novel microtubule inhibitor, induces mitotic arrest and apoptosis in human cancer cells. Biochem. Pharmacol. 2008, 75, 383–394. [Google Scholar] [CrossRef] [PubMed]
- Hwang, J.; Lee, H.-I.; Chang, Y.-S.; Lee, S.J.; Kim, K.P.; Park, S.I. 15-deoxy-Delta12,14-prostaglandin J2-induced down-regulation of endothelial nitric oxide synthase in association with HSP70 induction. Biochem. Biophys. Res. Commun. 2007, 357, 206–211. [Google Scholar] [CrossRef] [PubMed]
- Murakami, A.; Ashida, H.; Terao, J. Multitargeted cancer prevention by quercetin. Cancer Lett. 2008, 269, 315–325. [Google Scholar] [CrossRef]
- Du, S.; Song, X.; Li, Y.; Cao, Y.; Chu, F.; Durojaye, O.A.; Su, Z.; Shi, X.; Wang, J.; Cheng, J.; et al. Celastrol inhibits ezrin-mediated migration of hepatocellular carcinoma cells. Sci. Rep. 2020, 10, 11273. [Google Scholar] [CrossRef] [PubMed]
- Valvezan, A.J.; McNamara, M.C.; Miller, S.K.; Torrence, M.E.; Asara, J.M.; Henske, E.P.; Manning, B.D. IMPDH inhibitors for antitumor therapy in tuberous sclerosis complex. JCI Insight 2020, 5, e135071. [Google Scholar] [CrossRef]
- Zhang, Y.; Tao, X.; Jin, G.; Jin, H.; Wang, N.; Hu, F.; Luo, Q.; Shu, H.; Zhao, F.; Yao, M.; et al. A Targetable Molecular Chaperone Hsp27 Confers Aggressiveness in Hepatocellular Carcinoma. Theranostics 2016, 6, 558–570. [Google Scholar] [CrossRef]
- Fu, W.-M.; Wang, W.-M.; Wang, H.; Zhu, X.; Liang, Y.; Kung, H.F.; Zhang, J.F. 1,3,5-Trihydroxy-13,13-dimethyl-2H-pyran [7,6-b] xanthone directly targets heat shock protein 27 in hepatocellular carcinoma. Cell Biol. Int. 2014, 38, 272–276. [Google Scholar] [CrossRef]
- Xiu, P.; Xu, Z.; Liu, F.; Li, Z.; Li, T.; Zou, F.; Sun, X.; Li, J. Downregulating sCLU Enhances the Sensitivity of Hepatocellular Carcinoma Cells to Gemcitabine by Activating the Intrinsic Apoptosis Pathway. Dig. Dis. Sci. 2014, 59, 1798–1809. [Google Scholar] [CrossRef]
- Zheng, W.; Sai, W.; Yao, M.; Gu, H.; Yao, Y.; Qian, Q.; Yao, D. Silencing clusterin gene transcription on effects of multidrug resistance reversing of human hepatoma HepG2/ADM cells. Tumor. Biol. 2015, 36, 3995–4003. [Google Scholar] [CrossRef]
- Peng, M.; Deng, J.; Zhou, S.; Tao, T.; Su, Q.; Yang, X.; Yang, X. The role of Clusterin in cancer metastasis. Cancer Manag. Res. 2019, 11, 2405–2414. [Google Scholar] [CrossRef]
- Ammar, H.; Closset, J.L. Clusterin activates survival through the phosphatidylinositol 3-kinase/Akt pathway. J. Biol. Chem. 2008, 283, 12851–12861. [Google Scholar] [CrossRef]
- Yoon, Y.J.; Kim, J.A.; Shin, K.D.; Shin, D.S.; Han, Y.M.; Lee, Y.J.; Lee, J.S.; Kwon, B.M.; Han, D.C. KRIBB11 inhibits HSP70 synthesis through inhibition of heat shock factor 1 function by impairing the recruitment of positive transcription elongation factor b to the hsp70 promoter. J. Biol. Chem. 2011, 286, 1737–1747. [Google Scholar] [CrossRef]
- Song, N.-Y.; Kim, D.-H.; Kim, E.-H.; Na, H.-K.; Surh, Y.-J. 15-Deoxy-delta 12, 14-prostaglandin J2 induces upregulation of multidrug resistance-associated protein 1 via Nrf2 activation in human breast cancer cells. Ann. N. Y. Acad. Sci. 2009, 1171, 210–216. [Google Scholar] [CrossRef]
- Noel, P.; Von Hoff, D.D.; Saluja, A.K.; Velagapudi, M.; Borazanci, E.; Han, H. Triptolide and Its Derivatives as Cancer Therapies. Trends Pharmacol. Sci. 2019, 40, 327–341. [Google Scholar] [CrossRef]
- Chin, Y.; Gumilar, K.E.; Li, X.-G.; Tjokroprawiro, B.A.; Lu, C.H.; Lu, J.; Zhou, M.; Sobol, R.W.; Tan, M. Targeting HSF1 for cancer treatment: Mechanisms and inhibitor development. Theranostics 2023, 13, 2281–2300. [Google Scholar] [CrossRef]
- Lee, C.-H.; Hong, H.-M.; Chang, Y.-Y.; Chang, W.-W. Inhibition of heat shock protein (Hsp) 27 potentiates the suppressive effect of Hsp90 inhibitors in targeting breast cancer stem-like cells. Biochimie 2012, 94, 1382–1389. [Google Scholar] [CrossRef]
- Lampros, M.; Vlachos, N.; Voulgaris, S.; Alexiou, G.A. The Role of Hsp27 in Chemotherapy Resistance. Biomedicines 2022, 10, 897. [Google Scholar] [CrossRef]
- Yun, C.W.; Kim, H.J.; Lim, J.H.; Lee, S.H. Heat Shock Proteins: Agents of Cancer Development and Therapeutic Targets in Anti-Cancer Therapy. Cells 2019, 9, 60. [Google Scholar] [CrossRef] [PubMed]
- Talaei, S.; Mellatyar, H.; Asadi, A.; Akbarzadeh, A.; Sheervalilou, R.; Zarghami, N. Spotlight on 17-AAG as an Hsp90 inhibitor for molecular targeted cancer treatment. Chem. Biol. Drug Des. 2019, 93, 760–786. [Google Scholar] [CrossRef] [PubMed]
- Georgakis, G.V.; Li, Y.; Younes, A. The heat shock protein 90 inhibitor 17-AAG induces cell cycle arrest and apoptosis in mantle cell lymphoma cell lines by depleting cyclin D1, Akt, Bid and activating caspase 9. Br. J. Haematol. 2006, 135, 68–71. [Google Scholar] [CrossRef] [PubMed]
- Chettiar, S.T.; Malek, R.; Annadanam, A.; Nugent, K.M.; Kato, Y.; Wang, H.; Cades, J.A.; Taparra, K.; Belcaid, Z.; Ballew, M.; et al. Ganetespib radiosensitization for liver cancer therapy. Cancer Biol. Ther. 2016, 17, 457–466. [Google Scholar] [CrossRef] [PubMed]
- Youssef, M.E.; Cavalu, S.; Hasan, A.M.; Yahya, G.; Abd-Eldayem, M.A.; Saber, S. Role of Ganetespib, an HSP90 Inhibitor, in Cancer Therapy: From Molecular Mechanisms to Clinical Practice. Int. J. Mol. Sci. 2023, 24, 5014. [Google Scholar] [CrossRef] [PubMed]
- Colunga Biancatelli, R.M.L.; Solopov, P.; Gregory, B.; Catravas, J.D. The HSP90 Inhibitor, AUY-922, Protects and Repairs Human Lung Microvascular Endothelial Cells from Hydrochloric Acid-Induced Endothelial Barrier Dysfunction. Cells 2021, 10, 1489. [Google Scholar] [CrossRef]
- Wang, X.; Wang, S.; Liu, Y.; Ding, W.; Zheng, K.; Xiang, Y.; Liu, K.; Wang, D.; Zeng, Y.; Xia, M.; et al. The Hsp90 inhibitor SNX-2112 induces apoptosis of human hepatocellular carcinoma cells: The role of ER stress. Biochem. Biophys. Res. Commun. 2014, 446, 160–166. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Qin, L.; Deng, L.; Zhu, X.; Li, Y.; Wu, X.; Zheng, Y. SNX-2112 Induces Apoptosis and Inhibits Proliferation, Invasion, and Migration of Non-Small Cell Lung Cancer by Downregulating Epithelial-Mesenchymal Transition via the Wnt/β-Catenin Signaling Pathway. J. Cancer 2021, 12, 5825–5837. [Google Scholar] [CrossRef] [PubMed]
- Gallerne, C.; Prola, A.; Lemaire, C. Hsp90 inhibition by PU-H71 induces apoptosis through endoplasmic reticulum stress and mitochondrial pathway in cancer cells and overcomes the resistance conferred by Bcl-2. Biochim. Biophys. Acta (BBA)-Mol. Cell Res. 2013, 1833, 1356–1366. [Google Scholar] [CrossRef] [PubMed]
- Miyata, Y. Hsp90 inhibitor geldanamycin and its derivatives as novel cancer chemotherapeutic agents. Curr. Pharm. Des. 2005, 11, 1131–1138. [Google Scholar] [CrossRef] [PubMed]
- Wurnig, S.; Vogt, M.; Hogenkamp, J.; Dienstbier, N.; Borkhardt, A.; Bhatia, S.; Hansen, F.K. Development of the first geldanamycin-based HSP90 degraders. Front. Chem. 2023, 11, 1219883. [Google Scholar] [CrossRef] [PubMed]
- Hadden, M.K.; Lubbers, D.J.; Blagg, B.S.J. Geldanamycin, radicicol, and chimeric inhibitors of the Hsp90 N-terminal ATP binding site. Curr. Top. Med. Chem. 2006, 6, 1173–1182. [Google Scholar] [CrossRef]
- Johnson, V.A.; Singh, E.K.; Nazarova, L.A.; Alexander, L.D.; McAlpine, S.R. Macrocyclic Inhibitors of Hsp90. Curr. Top. Med. Chem. 2010, 10, 1380–1402. [Google Scholar] [CrossRef]
- Ban, H.S.; Shimizu, K.; Minegishi, H.; Nakamura, H. Identification of HSP60 as a primary target of o-carboranylphenoxyacetanilide, an HIF-1alpha inhibitor. J. Am. Chem. Soc. 2010, 132, 11870–11871. [Google Scholar] [CrossRef]
- Cappello, F.; Marino Gammazza, A.; Palumbo Piccionello, A.; Campanella, C.; Pace, A.; Conway de Macario, E.; Macario, A.J. Hsp60 chaperonopathies and chaperonotherapy: Targets and agents. Expert Opin. Ther. Targets 2014, 18, 185–208. [Google Scholar] [CrossRef]
- Hu, D.; Liu, Y.; Lai, Y.-T.; Tong, K.C.; Fung, Y.M.; Lok, C.N.; Che, C.M. Anticancer Gold(III) Porphyrins Target Mitochondrial Chaperone Hsp60. Angew. Chem. Int. Ed Engl. 2016, 55, 1387–1391. [Google Scholar] [CrossRef]
- Tong, K.-C.; Hu, D.; Wan, P.-K.; Lok, C.-N.; Che, C.-M. Anticancer Gold(III) Compounds With Porphyrin or N-heterocyclic Carbene Ligands. Front. Chem. 2020, 8, 587207. [Google Scholar] [CrossRef] [PubMed]
- Kummar, S.; Gutierrez, M.E.; Gardner, E.R.; Chen, X.; Figg, W.D.; Zajac-Kaye, M.; Chen, M.; Steinberg, S.M.; Muir, C.A.; Yancey, M.A.; et al. Phase I trial of 17-dimethylaminoethylamino-17-demethoxygeldanamycin (17-DMAG), a heat shock protein inhibitor, administered twice weekly in patients with advanced malignancies. Eur. J. Cancer 2010, 46, 340–347. [Google Scholar] [CrossRef] [PubMed]
- Xu, F.; Lin, D.; Jiang, W.; Meng, L.; Xu, Y.; Wang, C.; Wang, X.; He, H.; Xu, D.; Zhu, Y.; et al. HSP70 inhibitor VER155008 suppresses pheochromocytoma cell and xenograft growth by inhibition of PI3K/AKT/mTOR and MEK/ERK pathways. Int. J. Clin. Exp. Pathol. 2019, 12, 2585–2594. [Google Scholar] [PubMed]
- Huang, L.; Wang, Y.; Bai, J.; Yang, Y.; Wang, F.; Feng, Y.; Zhang, R.; Li, F.; Zhang, P.; Lv, N.; et al. Blockade of HSP70 by VER-155008 synergistically enhances bortezomib-induced cytotoxicity in multiple myeloma. Cell Stress Chaperones 2020, 25, 357–367. [Google Scholar] [CrossRef] [PubMed]
- Leu, J.I.-J.; Pimkina, J.; Pandey, P.; Murphy, M.E.; George, D.L. HSP70 inhibition by the small-molecule 2-phenylethynesulfonamide impairs protein clearance pathways in tumor cells. Mol. Cancer Res. 2011, 9, 936–947. [Google Scholar] [CrossRef] [PubMed]
- Barazi, H.O.; Zhou, L.; Templeton, N.S.; Krutzsch, H.C.; Roberts, D.D. Identification of Heat Shock Protein 60 as a Molecular Mediator of α3β1 Integrin Activation. Cancer Res. 2002, 62, 1541–1548. [Google Scholar]
- Tretiakova, I.; Blaesius, D.; Maxia, L.; Wesselborg, S.; Schulze-Osthoff, K.; Cinatl, J., Jr.; Michaelis, M.; Werz, O. Myrtucommulone from Myrtus communis induces apoptosis in cancer cells via the mitochondrial pathway involving caspase-9. Apoptosis 2008, 13, 119–131. [Google Scholar] [CrossRef]
- Mir, M.A.; Elbehairi, S.E.; Memish, L.A.; Saif, F.; Bashir, N.; Shati, A.A.; Alfaifi, M.Y.; Alamri, A.M.; Alkahtani, S.A.; Ahmad, I.; et al. Myrtus Communis Leaf Extract—A Source of Secondary Metabolites Exhibiting Anticancer and Antimycobacterial Activities. Res. Sq. 2021; in review. [Google Scholar] [CrossRef]
- Ogur, R. Studies with Myrtus communis L.: Anticancer properties. J. Intercult. Ethnopharmacol. 2014, 3, 135–137. [Google Scholar] [CrossRef] [PubMed]
- Kakeya, H.; Takahashi, I.; Okada, G.; Isono, K.; Osada, H. Epolactaene, a Novel Neuritogenic Compound in Human Neuroblastoma Cells, Produced by a Marine Fungus. J. Antibiot. 1995, 48, 733–735. [Google Scholar] [CrossRef] [PubMed]
- Baran, P.S.; Hafensteiner, B.D.; Ambhaikar, N.B.; Guerrero, C.A.; Gallagher, J.D. Enantioselective total synthesis of avrainvillamide and the stephacidins. J. Am. Chem. Soc. 2006, 128, 8678–8693. [Google Scholar] [CrossRef] [PubMed]
- Polson, E.S.; Kuchler, V.B.; Abbosh, C.; Ross, E.M.; Mathew, R.K.; Beard, H.A.; da Silva, B.; Holding, A.N.; Ballereau, S.; Chuntharpursat-Bon, E.; et al. KHS101 disrupts energy metabolism in human glioblastoma cells and reduces tumor growth in mice. Sci. Transl. Med. 2018, 10, eaar2718. [Google Scholar] [CrossRef]
- Tang, Y.; Zhou, Y.; Fan, S.; Wen, Q. The multiple roles and therapeutic potential of HSP60 in cancer. Biochem. Pharmacol. 2022, 201, 115096. [Google Scholar] [CrossRef]
- Wang, C.; Jin, G.; Jin, H.; Wang, N.; Luo, Q.; Zhang, Y.; Gao, D.; Jiang, K.; Gu, D.; Shen, Q.; et al. Clusterin facilitates metastasis by EIF3I/Akt/MMP13 signaling in hepatocellular carcinoma. Oncotarget 2015, 6, 2903–2916. [Google Scholar] [CrossRef]
- Karampa, A.D.; Goussia, A.C.; Glantzounis, G.K.; Mastoridou, E.M.; Anastasopoulos, N.-A.T.; Charchanti, A.V. The Role of Macroautophagy and Chaperone-Mediated Autophagy in the Pathogenesis and Management of Hepatocellular Carcinoma. Cancers 2022, 14, 760. [Google Scholar] [CrossRef]


| S. N° | HSP Family | Members of the HSP Family | Function | References |
|---|---|---|---|---|
| 1 | Small HSP | HSP10 and HSP27 | Molecular chaperon, HSP10 acts as a cofactor for HSP60. | [89,90] |
| 2 | HSP40/DNAJ | HSP40 | Molecular chaperon, HSP40 acts as a cofactor for HSP70. | [91] |
| 3 | HSP60 | HSP60 | Chaperonin | [92] |
| 4 | HSP70 | HSP70, HSC70, GRP75, GRP78 | Molecular chaperone | [93] |
| 5 | HSP90 | HSP90A, HSP90B, GRP94, TRAP1 | Molecular chaperone | [93] |
| 6 | Large HSPs | HSP110, GRP170 | Molecular chaperone, Holdase | [93] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Paul, R.; Shreya, S.; Pandey, S.; Shriya, S.; Abou Hammoud, A.; Grosset, C.F.; Prakash Jain, B. Functions and Therapeutic Use of Heat Shock Proteins in Hepatocellular Carcinoma. Livers 2024, 4, 142-163. https://doi.org/10.3390/livers4010011
Paul R, Shreya S, Pandey S, Shriya S, Abou Hammoud A, Grosset CF, Prakash Jain B. Functions and Therapeutic Use of Heat Shock Proteins in Hepatocellular Carcinoma. Livers. 2024; 4(1):142-163. https://doi.org/10.3390/livers4010011
Chicago/Turabian StylePaul, Ramakrushna, Smriti Shreya, Shweta Pandey, Srishti Shriya, Aya Abou Hammoud, Christophe F. Grosset, and Buddhi Prakash Jain. 2024. "Functions and Therapeutic Use of Heat Shock Proteins in Hepatocellular Carcinoma" Livers 4, no. 1: 142-163. https://doi.org/10.3390/livers4010011
APA StylePaul, R., Shreya, S., Pandey, S., Shriya, S., Abou Hammoud, A., Grosset, C. F., & Prakash Jain, B. (2024). Functions and Therapeutic Use of Heat Shock Proteins in Hepatocellular Carcinoma. Livers, 4(1), 142-163. https://doi.org/10.3390/livers4010011








