Pemafibrate Improves Alanine Aminotransferase Levels Independently of Its Lipid-Lowering Effect
Abstract
:1. Introduction
2. Methods
2.1. Liver Function Evaluation
2.2. Statistical Analysis
3. Results
3.1. Baseline
3.2. Pre- and Post Treatment
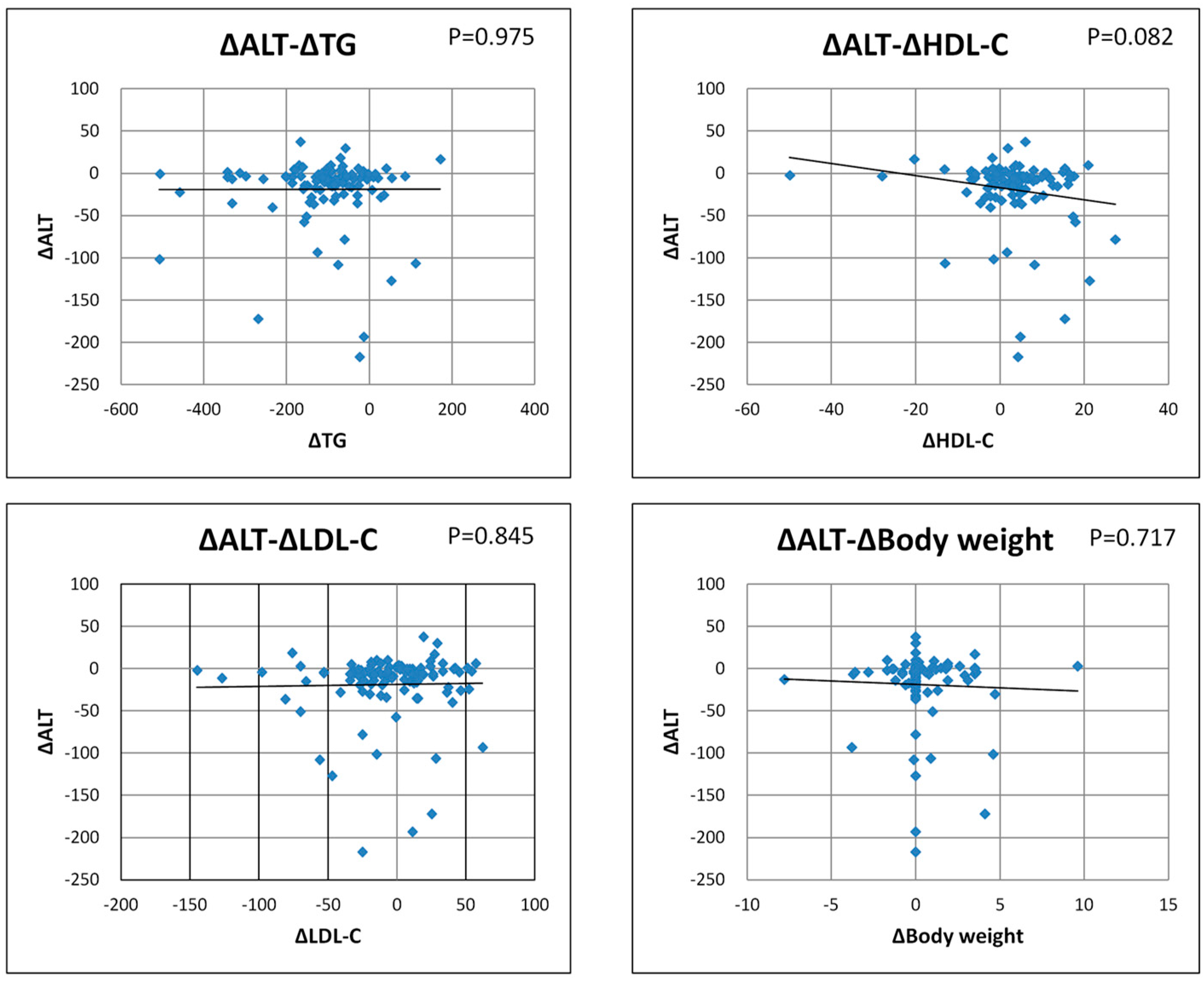
3.3. ALT Correlation
4. Discussion
5. Conclusions
6. Limitation
- This was a single-facility, retrospective observational study.
- The pemafibrate administration period was not unified.
- The control group was not set.
- No histopathological evaluation of the liver was performed after administration.
- There are quite a lot of concomitant medications, and pemafibrate alone could not be evaluated. In particular, SGLT2 inhibitors and concomitant drugs such as statins and ezetimibe may greatly affect the effects of NAFLD.
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kim, M.K.; Han, K.; Kim, H.-S.; Park, Y.-M.; Kwon, H.-S.; Yoon, K.-H.; Lee, S.-H. Cholesterol variability and the risk of mortality, myocardial infarction, andstroke: A nationwide population-based study. Eur. Heart J. 2017, 38, 3560–3566. [Google Scholar] [CrossRef] [PubMed]
- Watanabe, S.; Hashimoto, E.; Ikejima, K.; Uto, H.; Ono, M.; Sumida, Y.; Seike, M.; Takei, Y.; Takehara, T.; Tokushige, K.; et al. Evidence-based clinical practice guidelines for nonalcoholic fatty liver disease/nonalcoholic steatohepatitis. J. Gastroenterol. 2015, 50, 364–377. [Google Scholar] [CrossRef] [PubMed]
- Eguchi, Y.; Hyogo, H.; Ono, M.; Mizuta, T.; Ono, N.; Fujimoto, K.; Chayama, K.; Saibara, T. Prevalence and associated metabolic factors of nonalcoholic fatty liver disease in the general population from 2009 to 2010 in Japan: A multicenter large retrospective study. J. Gastroenterol. 2012, 47, 586–595. [Google Scholar] [CrossRef] [PubMed]
- Sumida, Y.; Yoneda, M. Current and future pharmacological therapies for NAFLD/NASH. J. Gastroenterol. 2018, 53, 362–376. [Google Scholar] [CrossRef]
- Tenenbaum, A.; Motro, M.; Fisman, E.Z. Dual and pan-peroxisome proliferator-activated receptors (PPAR) co-agonism: The bezafibrate lessons. Cardiovasc. Diabetol. 2005, 4, 14. [Google Scholar] [CrossRef]
- Fruchart, J.C. Selective peroxisome proliferator-activated receptor α modulators (SPPARMα): The next generation of peroxisome proliferator-activated receptor α-agonists. Cardiovasc. Diabetol. 2013, 12, 82. [Google Scholar] [CrossRef]
- Honda, Y.; Kessoku, T.; Ogawa, Y.; Tomeno, W.; Imajo, K.; Fujita, K.; Yoneda, M.; Takizawa, T.; Saito, S.; Nagashima, Y.; et al. Pemafibrate, a novel selective peroxisome proliferator-activated receptor α modulator, improves the pathogenesis in a rodent model of nonalcoholic steatohepatitis. Sci. Rep. 2017, 7, 42477. [Google Scholar] [CrossRef]
- Ishibashi, S.; Yamashita, S.; Arai, H.; Araki, E.; Yokote, K.; Suganami, H.; Fruchart, J.C.; Kodama, T. Effects of K-877, a novel selective PPARα modulator (SPPARMα), in dyslipidaemic patients: A randomized, double-blind, active- and placebo-controlled, phase 2 trial. Atherosclerosis 2016, 249, 36–43. [Google Scholar] [CrossRef]
- Seko, Y.; Sumida, Y.; Tanaka, S.; Mori, K.; Taketani, H.; Ishiba, H.; Hara, T.; Okajima, A.; Yamaguchi, K.; Moriguchi, M.; et al. Serum alanine aminotransferase predicts the histological course of non-alcoholic steatohepatitis in Japanese patients. Hepatol. Res. 2015, 45, E53–E61. [Google Scholar] [CrossRef]
- Kozumi, K.; Kodama, T.; Murai, H.; Sakane, S.; Govaere, O.; Cockell, S.; Motooka, D.; Kakita, N.; Yamada, Y.; Kondo, Y.; et al. Transcriptomics Identify Thrombospondin-2 as a Biomarker for NASH and Advanced Liver Fibrosis. Hepatology 2021, 74, 2452–2466. [Google Scholar] [CrossRef]
- Sterling, R.K.; Lissen, E.; Clumeck, N.; Sola, R.; Correa, M.C.; Montaner, J.; Sulkowski, M.S.; Torriani, F.J.; Dieterich, D.T.; Thomas, D.L.; et al. Development of a simple noninvasive index to predict significant fibrosis in patients with HIV/HCV coinfection. Hepatology 2006, 43, 1317–1325. [Google Scholar] [CrossRef] [PubMed]
- Iso, H.; Imano, H.; Yamagishi, K.; Ohira, T.; Cui, R.; Noda, H.; Sato, S.; Kiyama, M.; Okada, T.; Hitsumoto, S.; et al. Fasting and non-fasting triglycerides and risk of ischemic cardiovascular disease in Japanese men and women: The Circulatory Risk in Communities Study (CIRCS). Atherosclerosis 2014, 237, 361–368. [Google Scholar] [CrossRef] [PubMed]
- Sone, H.; Tanaka, S.; Tanaka, S.; Iimuro, S.; Oida, K.; Yamasaki, Y.; Oikawa, S.; Ishibashi, S.; Katayama, S.; Ohashi, Y.; et al. Serum level of triglycerides is a potent risk factor comparable to LDL cholesterol for coronary heart disease in Japanese patients with type 2 diabetes: Subanalysis of the Japan Diabetes Complications Study (JDCS). J. Clin. Endocrinol. Metab. 2011, 96, 3448–3456. [Google Scholar] [CrossRef]
- Angulo, P.; Kleiner, D.E.; Dam-Larsen, S.; Adams, L.A.; Bjornsson, E.S.; Charatcharoenwitthaya, P.; Mills, P.R.; Keach, J.C.; Lafferty, H.D.; Stahler, A.; et al. Liver fibrosis, but No Other Histologic Features, Is Associated with Long-term Outcomes of Patients with Nonalcoholic Fatty Liver Disease. Gastroenterology. 2015, 149, 389–397. [Google Scholar] [CrossRef]
- Ishibashi, S.; Arai, H.; Yokote, K.; Araki, E.; Suganami, H.; Yamashita, S. Efficacy and safety of pemafibrate (K-877), a selective peroxisome proliferator-activated receptor α modulator, in patients with dyslipidemia: Results from a 24-week, randomized, double-blind, active-controlled, phase 3 trial. J. Clin. Lipidol. 2018, 12, 173–184. [Google Scholar] [CrossRef]
- Seko, Y.; Yamaguchi, K.; Umemura, A.; Yano, K.; Takahashi, A.; Okishio, S.; Kataoka, S.; Okuda, K.; Moriguchi, M.; Okanoue, T.; et al. Effect of pemafibrate on fatty acid levels and liver enzymes in non-alcoholic fatty liver disease patients with dyslipidemia: A single-arm, pilot study. Hepatol. Res. 2020, 50, 1328–1336. [Google Scholar] [CrossRef] [PubMed]
- Shinozaki, S.; Tahara, T.; Lefor, A.K.; Ogura, M. Pemafibrate decreases markers of hepatic inflammation in patients with non-alcoholic fatty liver disease. Clin. Exp. Hepatol. 2020, 6, 270–274. [Google Scholar] [CrossRef]
- Hatanaka, T.; Kakizaki, S.; Saito, N.; Nakano, Y.; Nakano, S.; Hazama, Y.; Yoshida, S.; Hachisu, Y.; Tanaka, Y.; Kashiwabara, K.; et al. Impact of Pemafibrate in Patients with Hypertriglyceridemia and Metabolic Dysfunction-associated Fatty Liver Disease Pathologically Diagnosed with Non-alcoholic Steatohepatitis: A Retrospective, Single-arm Study. Intern. Med. 2021, 60, 2167–2174. [Google Scholar] [CrossRef]
- Nakajima, A.; Eguchi, Y.; Yoneda, M.; Imajo, K.; Tamaki, N.; Suganami, H.; Nojima, T.; Tanigawa, R.; Iizuka, M.; Iida, Y.; et al. Randomised clinical trial: Pemafibrate, a novel selective peroxisome proliferator-activated receptor α modulator (SPPARM α), versus placebo in patients with non-alcoholic fatty liver disease. Aliment. Pharmacol. Ther. 2021, 54, 1263–1277. [Google Scholar] [CrossRef]
- Chianale, J.; Vollrath, V.; Wielandt, A.M.; Amigo, L.; Rigotti, A.; Nervi, F.; Gonzalez, S.; Andrade, L.; Pizarro, M.; Accatino, L. Fibrates induce mdr2 gene expression and biliary phospholipid secretion in the mouse. Biochem. J. 1996, 314, 781–786. [Google Scholar] [CrossRef]
- Smit, J.; Schinkel, A.; Elferink, R.; Groen, A.; Wagenaar, E.; van Deemter, L.; Mol, C.; Ottenhoff, R.; van der Lugt, N.; van Roon, M.; et al. Homozygous disruption of the murine mdr2 Pglycoprotein gene leads to a complete absence of phospholipid from bile and to liver disease. Cell 1993, 75, 451–462. [Google Scholar] [CrossRef]
- Joshita, S.; Umemura, T.; Yamashita, Y.; Sugiura, A.; Yamazaki, T.; Fujimori, N.; Matsumoto, A.; Tanaka, E. Biochemical and plasma lipid responses to pemafibrate in patients with primary biliary cholangitis. Hepatol. Res. 2019, 49, 1236–1243. [Google Scholar] [CrossRef] [PubMed]
- Yamaguchi, M.; Asano, T.; Arisaka, T.; Mashima, H.; Irisawa, A.; Tamano, M. Effects of pemafibrate on primary biliary cholangitis with dyslipidemia. Hepatol. Res. 2022, 52, 522–531. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Chu, E.S.; Zhang, J.; Li, X.; Liang, Q.; Chen, J.; Chen, M.; Teoh, N.; Farrell, G.; Sung, J.J.; et al. Peroxisome proliferator-activated receptor α inhibits hepatocarcinogenesis through mediating NF-κB signaling pathway. Oncotarget 2014, 5, 8330–8340. [Google Scholar] [CrossRef] [PubMed]
- Stienstra, R.; Mandard, S.; Patsouris, D.; Maass, C.; Kersten, S.; Müller, M. Peroxisome proliferator-activated receptor α protects against obesity-induced hepatic inflammation. Endocrinology 2007, 148, 2753–2763. [Google Scholar] [CrossRef]
- Lefebvre, P.; Chinetti, G.; Fruchart, J.C.; Staels, B. Sorting out the roles of PPAR α in energy metabolism and vascular homeostasis. J. Clin. Investig. 2006, 116, 571–580. [Google Scholar] [CrossRef]
- Sasaki, Y.; Asahiyama, M.; Tanaka, T.; Yamamoto, S.; Murakami, K.; Kamiya, W.; Matsumura, Y.; Osawa, T.; Anai, M.; Fruchart, J.-C.; et al. Pemafibrate, a selective PPARα modulator, prevents non-alcoholic steatohepatitis development without reducing the hepatic triglyceride content. Sci. Rep. 2020, 10, 7818. [Google Scholar] [CrossRef]
- Konishi, H.; Miyauchi, K.; Onishi, A.; Suzuki, S.; Fuchimoto, D.; Shitara, J.; Endo, H.; Wada, H.; Doi, S.; Naito, R.; et al. Effect of pemafibrate (K-877), a novel selective peroxisome proliferator-activated receptor αmodular (SPPARMα), in atherosclerosis model, using low-density lipoprotein receptor knockout swine with balloon injury. PLoS ONE 2020, 15, e0241195. [Google Scholar] [CrossRef]
| Age (years) | 62.2 ± 13.7 (Mean ± SD) |
| Males/Females | 77/35 |
| BMI (kg/m2) | 25.4 ± 4.2 (Mean ± SD) |
| Follow-up period (days) | 224.1 ± 83.6 (Mean ± SD) |
| Liver disease | 90 (80.4%) |
| NAFLD | 71 (63.4%) |
| Hypertension | 54 (48.2%) |
| Diabetes mellitus | 43 (38.4%) |
| Statin | 51 (45.5%) |
| Ezetimibe | 5 (4.5%) |
| EPA·DHA | 0 (0%) |
| SGLT2 inhibitor | 31 (27.7%) |
| DPP4 inhibitor | 28 (25.0%) |
| Metformin | 28 (25.0%) |
| Angiotensin II receptor blockers | 16 (14.3%) |
| UDCA | 30 (26.8%) |
| Vitamin E | 23 (20.5%) |
| Variables | Before | After | p Value |
|---|---|---|---|
| Weight (kg) | 68.3 ± 14.2 | 68.9 ± 14.1 | <0.05 |
| BMI (kg/m2) | 25.4 ± 4.2 | 25.7 ± 4.1 | <0.05 |
| AST (IU/L) | 36.1 ± 32.1 | 27.8 ± 17.0 | 0.005 |
| ALT (IU/L) | 43.7 ± 43.8 | 24.0 ± 13.8 | <0.001 |
| γ-GPT (IU/L) | 93.8 ± 210.2 | 44.3 ± 129.3 | <0.001 |
| Platelet (104/μL) | 24.6 ± 7.2 | 26.9 ± 8.6 | <0.001 |
| M2BPGi | 0.9 ± 0.7 | 0.7 ± 0.5 | <0.05 |
| Triglyceride (mg/dL) | 234.6 ± 126.3 | 126.7 ± 68.4 | <0.001 |
| HDL-C (mg/dL) | 52.6 ± 12.9 | 55.7 ± 11.6 | 0.001 |
| LDL-C (mg/dL) | 118.4 ± 35.2 | 111.9 ± 28.0 | 0.06 |
| Cre (mg/dL) | 0.8 ± 0.2 | 0.8 ± 0.2 | 0.57 |
| eGFR (mL/min/1.73 m2) | 73.0 ± 15.2 | 73.1 ± 15.5 | 0.70 |
| Glucose (mg/dL) | 138.7 ± 42.1 | 130.4 ± 38.1 | 0.15 |
| HbA1c (%) | 6.5 ± 1.0 | 6.4 ± 1.0 | 0.87 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Watanabe, A.; Horigome, R.; Nakatsuka, Y.; Terai, S. Pemafibrate Improves Alanine Aminotransferase Levels Independently of Its Lipid-Lowering Effect. Livers 2023, 3, 562-568. https://doi.org/10.3390/livers3040038
Watanabe A, Horigome R, Nakatsuka Y, Terai S. Pemafibrate Improves Alanine Aminotransferase Levels Independently of Its Lipid-Lowering Effect. Livers. 2023; 3(4):562-568. https://doi.org/10.3390/livers3040038
Chicago/Turabian StyleWatanabe, Azuma, Ryoko Horigome, Yumiko Nakatsuka, and Shuji Terai. 2023. "Pemafibrate Improves Alanine Aminotransferase Levels Independently of Its Lipid-Lowering Effect" Livers 3, no. 4: 562-568. https://doi.org/10.3390/livers3040038
APA StyleWatanabe, A., Horigome, R., Nakatsuka, Y., & Terai, S. (2023). Pemafibrate Improves Alanine Aminotransferase Levels Independently of Its Lipid-Lowering Effect. Livers, 3(4), 562-568. https://doi.org/10.3390/livers3040038






