Investigating Vernal Pool Fairy Shrimp Exposure to Organophosphate Pesticides: Implications for Population-Level Risk Assessment
Abstract
:1. Introduction
2. Methods
2.1. Overview of Life History Relevant to Model Development
2.2. Utilization of Pop-GUIDE for Model Development
2.2.1. Phase 1: Model Objectives
2.2.2. Phase 2: Data Collection
2.2.3. Phase 3: Decision Steps
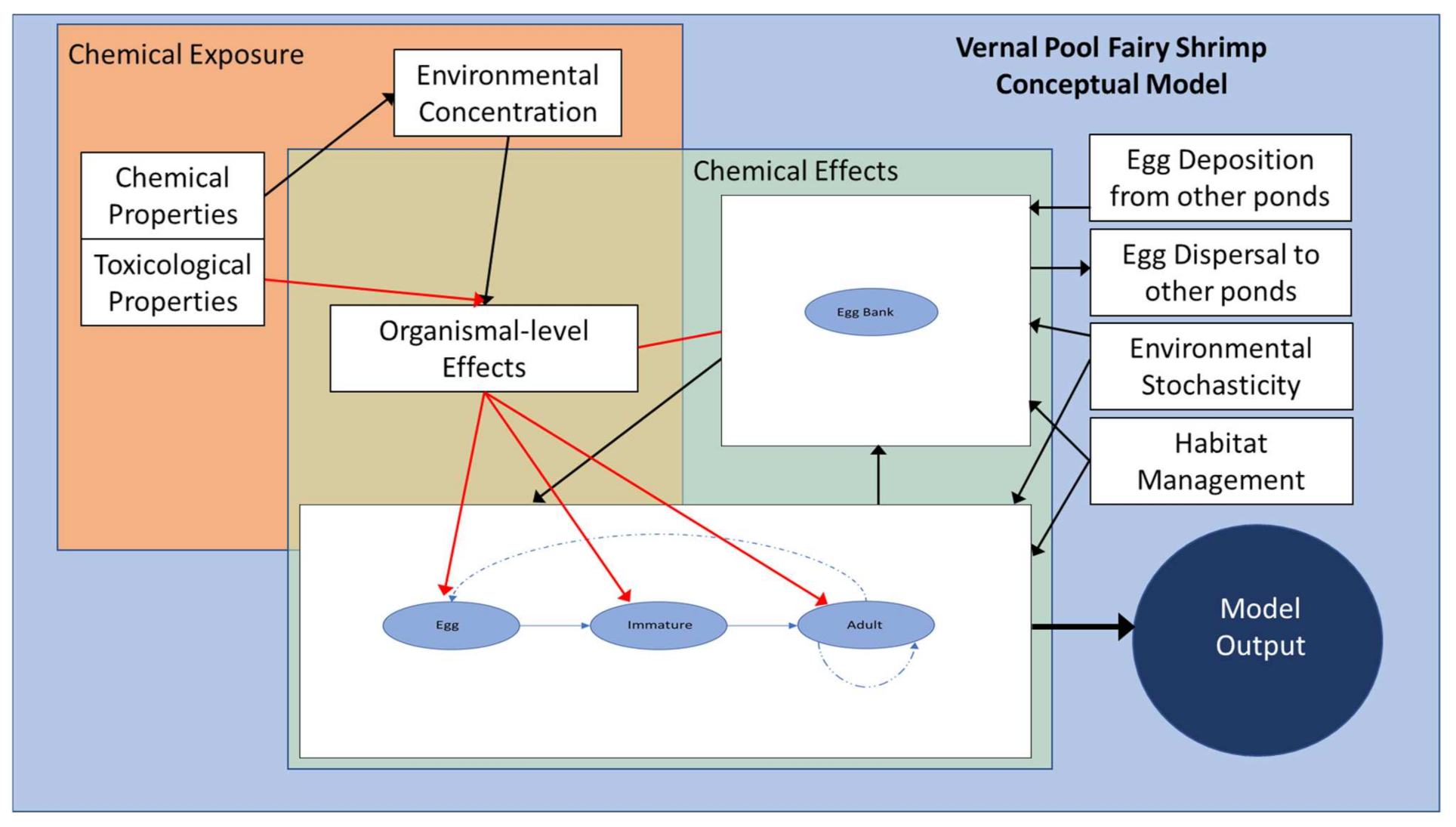
2.2.4. Phase 4: Conceptual Model
2.2.5. Phase 5: Evaluation and Implementation
3. Results
3.1. Utilization of Pop-GUIDE for Model Development
3.1.1. Phase 1: Model Objectives
3.1.2. Phase 2: Data Collection
3.1.3. Phase 3: Decision Steps
3.1.4. Phase 4: Conceptual Model
3.1.5. Phase 5: Evaluation and implementation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Eng, L.L.; Belk, D.; Eriksen, C.H. California Anostraca: Distribution, habitat, and status. J. Crustac. Biol. 1990, 10, 247–277. [Google Scholar] [CrossRef]
- Silveira, J.G. Avian uses of vernal pools and implications for conservation practice. In Ecology, Conservation, and Management of Vernal Pool Ecosystems; Witham, C.W., Bauder, E.T., Belk, D., Ferren, W.R., Jr., Ornduff, R., Eds.; California Native Plant Society: Sacramento, CA, USA, 1998; pp. 92–106. [Google Scholar]
- Rogers, D.C. Anostracan (Crustacea: Branchiopoda) zoogeography II. Relating distribution to geochemical substrate properties in the USA. Zootaxa 2014, 3856, 1–49. [Google Scholar] [CrossRef] [PubMed]
- Raimondo, S.; Sharpe, L.; Oliver, L.; McCaffrey, K.R.; Purucker, S.T.; Sinnathamby, S.; Minucci, J. A unified approach for protecting listed species and ecosystem services in geographically isolated wetlands using community-level protection goals. Sci. Total Environ. 2019, 663, 465–478. [Google Scholar] [CrossRef]
- Federal Register. Endangered and threatened plants; determination of endangered status for the conservancy fairy shrimp, longhorn fairy shrimp, and the vernal pool tadpole shrimp; and threatened status for the vernal pool fairy shrimp. Fed. Regist. 1994, 59, 48136–48153. [Google Scholar]
- Holland, R.F. Great Valley Vernal Pool Distribution, Rephotorevised 2005. 2009. Consultant Report. Prepared for Placer Land Trust. Available online: www.placerlandtrust.org (accessed on 1 October 2021).
- Johnson, C.S. Evaluation of Pesticides in Vernal Pools of the Central Valley, California; Final Report, Investigation ID #: 200210103.1; Congressional Districts—CA 2, 18; U.S. Department of the Interior Fish and Wildlife Service: Sacramento, CA, USA, 2006.
- Sinnathamby, S.; Minucci, J.M.; Denton, D.L.; Raimondo, S.M.; Oliver, L.; Yuan, Y.f.; Young, D.F.; Hook, J.; Pitchford, A.M.; Waits, E.; et al. A sensitivity analysis of pesticide concentrations in California Central Valley vernal pools. Environ. Pollut. 2020, 257, 113486. [Google Scholar] [CrossRef]
- Seiber, J.N.; Wilson, B.W.; McChesney, M.M. Air and fog deposition residues of four organophosphorus insecticides used on dormant orchards in the San Joaquin Valley, California. Environ. Sci. Technol. 1993, 27, 2236–2243. [Google Scholar] [CrossRef]
- Domagalski, J.L.; Dubrovsky, N.M.; Kratzer, C.R. Pesticides in the san Joaquin River, California: Inputs from dormant sprayed orchards. J. Environ. Qual. 1997, 26, 454–465. [Google Scholar] [CrossRef]
- Bailey, H.C.; Deanovic, L.; Reyes, E.; Kimball, T.; Larson, K.; Cortright, K.; Connor, V.; Hinton, D.E. Diazinon and chlorpyrifos in urban waterways in northern California. Environ. Toxicol. Chem. 2000, 19, 82–87. [Google Scholar] [CrossRef]
- Battaglin, W.A.; Rice, K.C.; Focazio, M.J.; Salmons, S.; Barry, R.X. The occurrence of glyphosate, atrazine, and other pesticides in vernal pools and adjacent streams in Washington, DC, Maryland, Iowa, and Wyoming, 2005–2006. Environ. Monit. Assess. 2009, 155, 281–307. [Google Scholar] [CrossRef]
- Trumble, J.T.; Butler, C.T. Climate change will exacerbate California’s insect pest problems. Calif. Agric. 2009, 62, 73–78. [Google Scholar] [CrossRef]
- Delcour, I.; Spanoghe, P.; Uyttendaele, M. Literature review: Impact of climate change on pesticide use. Food Res. Int. 2015, 68, 7–15. [Google Scholar] [CrossRef]
- Schomburg, C.J.; Glotfelty, D.E.; Seiber, J.N. Pesticide occurrence and distribution in fog collected near Monterey, California. Environ. Sci. Technol. 1991, 25, 155–160. [Google Scholar] [CrossRef]
- McConnell, L.L.; LeNoir, J.S.; Datta, S.; Seiber, J.N. Wet deposition of current use pesticides in the sierra Nevada mountain range, California USA. Environ. Toxicol. Chem. 1998, 17, 1908–1916. [Google Scholar] [CrossRef]
- LeNoir, J.S.; McConnell, L.L.; Fellers, G.M.; Cahill, T.M.; Seiber, J.N. Summertime transport of current-use pesticides from California’s Central Valley to the sierra Nevada mountain range. Environ. Toxicol. Chem. 1999, 18, 2715–2722. [Google Scholar] [CrossRef]
- CDPR (California Department of Pesticide Regulation). Summary of Pesticide Use Report Data: Indexed by Chemical; State of California, Environmental Protection Agency, Department of Pesticide Regulation: Sacramento, CA, USA, 2006.
- Raimondo, S.; Jackson, C.R.; Barron, M.G. Web-Based Interspecies Correlation Estimation (Web-ICE) for Acute Toxicity: 2015, User Manual; EPA/600/R-15/192, Version 3.3; USA Environmental Protection Agency, Office of Research and Development, Gulf Ecology Division: Gulf Breeze, FL, USA, 2015.
- Spromberg, J.A.; Meador, J.P. Relating chronic toxicity response to population-level effects: A comparison of population-level parameters for three salmon species as a function of low-level toxicity. Ecol. Model. 2006, 199, 240–252. [Google Scholar] [CrossRef] [Green Version]
- Miller, D.H.; Tietge, J.E.; McMaster, M.E.; Munkittrick, K.R.; Xia, X.; Ankley, G.T. Assessment of status of white sucker (Catostomus commersoni) populations exposed to bleached kraft pulp mill effluent. Environ. Toxicol. Chem. 2013, 32, 1592–1603. [Google Scholar] [CrossRef]
- Schmolke, A.; Brain, R.; Thorbek, P.; Perkins, D.; Forbes, V. Population modeling for pesticide risk assessment of threatened species—A case study of a terrestrial plant, Boltonia decurrens. Environ. Toxicol. Chem. 2017, 36, 480–491. [Google Scholar] [CrossRef]
- Schmolke, A.; Roy, C.; Brain, R.; Forbes, V. Adapting population models for application in pesticide risk assessment: A case study with Mead’s milkweed. Environ. Toxicol. Chem. 2018, 37, 2235–2245. [Google Scholar] [CrossRef]
- Thursby, G.; Sappington, K.; Etterson, M. Coupling toxicokinetic–toxicodynamic and population models for assessing aquatic ecological risks to time-varying pesticide exposures. Environ. Toxicol. Chem. 2018, 37, 2633–2644. [Google Scholar] [CrossRef]
- Forbes, V.E.; Railsback, S.; Accolla, C.; Birnir, B.; Bruins, R.J.; Ducrot, V.; Galic, N.; Garber, K.; Harvey, B.C.; Jager, H.I.; et al. Predicting impacts of chemicals from organisms to ecosystem service delivery: A case study of endocrine disruptor effects on trout. Sci. Total Environ. 2019, 649, 949–959. [Google Scholar] [CrossRef] [Green Version]
- EFSA. Scientific Opinion on good modelling practice in the context of mechanistic effect models for risk assessment of plant protection products. Eur. Food Saf. Authority. EFSA J. 2014, 12, 3589. [Google Scholar]
- Raimondo, S.; Etterson, M.; Pollesch, N.; Garber, K.; Kanarek, A.; Lehmann, W.; Awkerman, J.A. A framework for linking population model development with ecological risk assessment objectives. Integr. Environ. Assess. Man. 2018, 14, 369–380. [Google Scholar] [CrossRef] [PubMed]
- Raimondo, S.; Schmolke, A.; Pollesch, N.; Accolla, C.; Galic, N.; Moore, A.; Vaugeois, M.; Rueda-Cediel, P.; Kanarek, A.; Awkerman, J.; et al. Pop-GUIDE: Population modeling Guidance, Use, Interpretation, and Development for Ecological Risk Assessment. Integr. Environ. Assess. Man. 2021, 17, 767–784. [Google Scholar] [CrossRef] [PubMed]
- Thorne, R.F. Are California’s vernal pools unique? In Vernal Pools and Intermittent Streams; Jain, S., Moyle, P., Eds.; University of California Institute of Ecology: Berkeley, CA, USA, 1984; Volume 28, pp. 1–8. [Google Scholar]
- Rogers, D.C. Branchiopoda (Anostraca, Notostraca, Laevicaudata, Spinicaudata, Cyclestherida). In Encyclopedia of Inland Waters; Likens, G.F., Ed.; Elsevier: Amsterdam, The Netherlands, 2009; Volume 2, pp. 242–249. [Google Scholar]
- Rogers, D.C. Larger hatching fractions in avian dispersed anostracan eggs (Branchiopoda). J. Crustacean Biol. 2014, 34, 135–143. [Google Scholar] [CrossRef] [Green Version]
- Rogers, D.C. A conceptual model for Anostracan biogeography. J. Crustacean Biol. 2015, 35, 686–699. [Google Scholar] [CrossRef] [Green Version]
- Levins, R. The strategy of model building in population biology. Am. Sci. 1966, 54, 421–431. [Google Scholar]
- Gallagher, S.P. Seasonal occurrence and habitat characteristics of some vernal pool Branchiopoda in northern California, USA. J. Crustacean Biol. 1996, 16, 322–329. [Google Scholar] [CrossRef]
- Rogers, D.C. Hatching response to temperature along a latitudinal gradient by the fairy shrimp Branchinecta lindahli (Crustacea; Branchiopoda; Anostraca) in culture conditions. J. Limnol. 2015, 74, 85–94. [Google Scholar] [CrossRef] [Green Version]
- Lavens, P.; Tackaert, W.; Sorgeloos, P. International study on Artemia. XLI. Influence of culture conditions and specific diapause deactivation methods on the hatchability of Artemia cysts produced in a standard culture system. Mar. Ecol. Program Ser. 1986, 31, 197–203. [Google Scholar] [CrossRef]
- Lavens, P.; Sorgeloos, P. The Cryptobiotic State of Artemia Cysts, Its Diapause Deactivation and Hatching: A Review. Available online: file:///C:/Users/MDPI/Downloads/133988.pdf (accessed on 10 March 2022).
- Brendonck, L. Diapause, quiescence, hatching requirements: What we can learn from large freshwater branchiopods (Crustacea: Anostraca, Notostraca, Conchostraca). Hydrobiologia 1996, 320, 85–97. [Google Scholar] [CrossRef]
- Mura, G.; Zarattini, P. Influence of parental rearing conditions on cyst production and hatching of Chirocephalus ruffoi, an endemic fairy shrimp from Italy. Crustaceana 1999, 72, 449–465. [Google Scholar]
- Zarattini, P.; Rossi, V.; Mantovani, B.; Mura, G. A preliminary study in the use of RAPD markers in detecting genetic differences in hatching patterns of Chirocephalus diaphanus Prévost, 1803 (Crustacea, Anostraca). Hydrobiologia 2002, 486, 315–323. [Google Scholar] [CrossRef]
- Patton, S. The Life History Patterns and the Distribution of Two Anostraca, Linderiella occidentalis and Branchinecta sp. Master’s Thesis, California State University, Chico, CA, USA, 1984. [Google Scholar]
- Gharibi, M.R.; Nematollahi, M.A.; Agh, N.; Atashbar, B. Life cycle of the fairy shrimp, Phallocryptus spinosa Milne Edwards, 1840 (Crustacea: Anostraca) at different temperatures. J. Agr. Sci. Technol. 2016, 18, 171–177. [Google Scholar]
- Thursby, G.B. Matrix Population Modeling for Estimating Effects from Time-Varying Aquatic Exposures: Technical Documentation; EPA/600/R-16/062; US Environmental Protection Agency: Washington, DC, USA, 2016.
- Thursby, G.B. Americamysis Bahia Stochastic Matrix Population Model for Laboratory Populations: Technical Documentation; EPA/600/R-09/121; US Environmental Protection Agency: Washington, DC, USA, 2009.
- Lussier, S.M.; Kuhn, A.; Chammas, M.J.; Sewall, J. Techniques for the laboratory culture of mysidopsis species (crustacea: Mysidacea). Environ. Toxicol. Chem. 1988, 7, 969–977. [Google Scholar] [CrossRef]
- The National Wildlife Federation. Vernal Pool Fairy Shrimp. Available online: https://www.nwf.org/Educational-Resources/Wildlife-Guide/Invertebrates/Vernal-Pool-Fairy-Shrimp (accessed on 1 March 2021).
- Rogers, D.C. Aquatic Macroinvertebrate Occurrences and Population Trends in Constructed and Natural Vernal Pools in Folsom, California. In Ecology, Conservation, and Management of Vernal Pool Ecosystems—Proceedings from a 1996 Conference; Witham, C.W., Bauder, E.T., Belk, D., Ferren, W.R., Jr., Ornduff, R., Eds.; California Native Plant Society: Sacramento, CA, USA, 1998; pp. 224–235. [Google Scholar]
- Eriksen, C.H.; Belk, D. The Fairy Shrimps of California’s Pools, Puddles, and Playas; Mad River Press: Eureka, CA, USA, 1999. [Google Scholar]
- IUCN. The IUCN Red List of Threatened Species. 2021, Version 2021-1. Available online: https://www.iucnredlist.org (accessed on 17 June 2021).
- Montrone, A.; Saito, L.; Weisberg, P.J.; Gosejohan, M.; Merriam, K.; Mejia, J.F. Climate change impacts on vernal pool hydrology and vegetation in northern California. J. Hydrol. 2019, 574, 1003–1013. [Google Scholar] [CrossRef]
- Young, D.F. Pesticide in Water Calculator User Manual for Versions 1.50 and 1.52; U.S. Environmental Protection Agency, Office of Pesticide Programs: Washington, DC, USA, 2016.
- Palumbo, A.J.; TenBrook, P.L.; Fojut, T.L.; Faria, I.R.; Tjeerdema, R.S. Aquatic Life Water Quality Criteria Derived via the UC Davis Method: I. Organophosphate Insecticides. In Aquatic Life Water Quality Criteria for Selected Pesticides, Reviews of Environmental Contamination and Toxicology; Tjeerdema, R.S., Ed.; Springer: New York, NY, USA, 2012; Volume 216, pp. 1–49. [Google Scholar] [CrossRef]
- USA Environmental Protection Agency (USEPA). ECOTOX User Guide: ECOTOXicology Knowledgebase System. 2021, Version 5.4. Available online: https://cfpub.epa.gov/ecotox/ (accessed on 30 July 2021).
- CDPR (California Department of Pest Regulation). California Department of Pesticide Regulation, Environmental Monitoring and Pest Management Branch. Pesticide Use Reporting Data 1999–2016. Data File. 2018. California Department of Pesticide Regulation, Sacramento, CA. Available online: https://www.cdpr.ca.gov/docs/pur/purmain.htm (accessed on 25 September 2021).
- Gruber, S.J.; Munn, M.D. Organophosphate and carbamate insecticides in agricultural waters and cholinesterase (ChE) inhibition in common carp (Cyprinus carpio). Arch. Environ. Contam. Toxicol. 1988, 35, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Brady, J.A.; Wallender, W.W.; Werner, I.; Mostafazadeh Fard, B.; Zalom, F.G.; Oliver, M.N.; Wilson, B.W.; Mata, M.M.; Henderdon, J.D.; Deanovic, L.A.; et al. Pesticide runoff from orchard floors in Davis, California, USA: A comparative analysis of diazinon and esfenvalerate. Agric. Ecosyst. Environ. 2006, 115, 56–68. [Google Scholar] [CrossRef]
- Willming, M.M.; Lilavois, C.R.; Barron, M.G.; Raimondo, S. Acute toxicity prediction to threatened and endangered species using Interspecies Correlation Estimation (ICE) models. Environ. Sci. Technol. 2016, 50, 10700–10707. [Google Scholar] [CrossRef]
- Caswell, H. Matrix Population Models; Sinauer Associates: Sunderland, MA, USA, 2001. [Google Scholar]
- Ellner, S.P. Lectures on Theoretical Ecology. Department of Ecology and Evolutionary Biology. 2009. Cornell University. Available online: http://www.eeb.cornell.edu/Ellner/lte/TheoreticalEcology2009.pdf (accessed on 5 March 2021).
- Goodman, L.A. The analysis of population growth when the birth and death rates depend on several factors. Biometrics 1969, 25, 659–681. [Google Scholar] [CrossRef]
- Rogers, D.C. Anostraca Catalogus. Raffles Bull. Zool. 2013, 61, 525–546. [Google Scholar]
- Central Valley Regional Water Quality Control Board. Central Valley Region Climate Change Work Plan. 2017; 64p. Available online: https://www.waterboards.ca.gov/centralvalley (accessed on 16 June 2021).
- Brooks, R.T. Potential impacts of global climate change on the hydrology and ecology of ephemeral freshwater systems of the forests of the northeastern United States. Clim. Chang. 2009, 95, 469–483. [Google Scholar] [CrossRef]
- Shin, H.R.; Kneitel, J.M. Warming interacts with inundation timing to influence the species composition of California vernal pool communities. Hydrobiologia 2019, 843, 93–105. [Google Scholar] [CrossRef]
- Pyke, C.R. Assessing climate change impacts on vernal pool ecosystems and endemic branchiopods. Ecosystems 2005, 8, 95–105. [Google Scholar] [CrossRef]
- Choudhury, P.P.; Saha, S. Dynamics of pesticides under changing climatic scenario. Environ. Monit. Assess. 2020, 192 (Suppl. 1), 814. [Google Scholar] [CrossRef]



| Number * | Question |
|---|---|
| 1. | Ideally, how will the population model be used in the ERA, e.g., as a direct assessment tool in species- or location-specific ERA (e.g., endangered species, superfund) or as part of a weight of evidence for broader ecological protections? If the model will be used as a direct assessment tool, its tradeoffs should match that of the ERA category. |
| 2. | What assessment endpoint(s) are most relevant to the ERA objective and the intended model use (e.g., population growth, abundance, quasi-extinction probability, etc.)? |
| 3. | Are there temporal considerations that are important to the realism of the ERA, e.g., seasonal chemical application or persistence in the environment? |
| 4. | What uncertainties are acceptable for the ERA? |
| 5. | What are the project resources (timeline, budget, etc.)? |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Miller, D.H.; Etterson, M.; Oliver, L.; Paulukonis, E.; Pollesch, N.; Purucker, S.T.; Rogers, D.C.; Sinnathamby, S.; Raimondo, S. Investigating Vernal Pool Fairy Shrimp Exposure to Organophosphate Pesticides: Implications for Population-Level Risk Assessment. Ecologies 2022, 3, 308-322. https://doi.org/10.3390/ecologies3030024
Miller DH, Etterson M, Oliver L, Paulukonis E, Pollesch N, Purucker ST, Rogers DC, Sinnathamby S, Raimondo S. Investigating Vernal Pool Fairy Shrimp Exposure to Organophosphate Pesticides: Implications for Population-Level Risk Assessment. Ecologies. 2022; 3(3):308-322. https://doi.org/10.3390/ecologies3030024
Chicago/Turabian StyleMiller, David H., Matthew Etterson, Leah Oliver, Elizabeth Paulukonis, Nathan Pollesch, S. Thomas Purucker, D. Christopher Rogers, Sumathy Sinnathamby, and Sandy Raimondo. 2022. "Investigating Vernal Pool Fairy Shrimp Exposure to Organophosphate Pesticides: Implications for Population-Level Risk Assessment" Ecologies 3, no. 3: 308-322. https://doi.org/10.3390/ecologies3030024






