A Two-Species Finite Volume Scalar Model for Modeling the Diffusion of Poly(lactic-co-glycolic acid) into a Coronary Arterial Wall from a Single Half-Embedded Drug Eluting Stent Strut
Abstract
1. Introduction
- A theoretical methodology for computational modeling of the diffusion of PLGA into a coronary arterial wall from a single half-embedded drug eluting stent strut.
- A computational drug diffusion model that considers the pharmaco-kinetic reactions in the arterial wall as equilibrium reversible binding reaction source terms for the free and bound-drug.
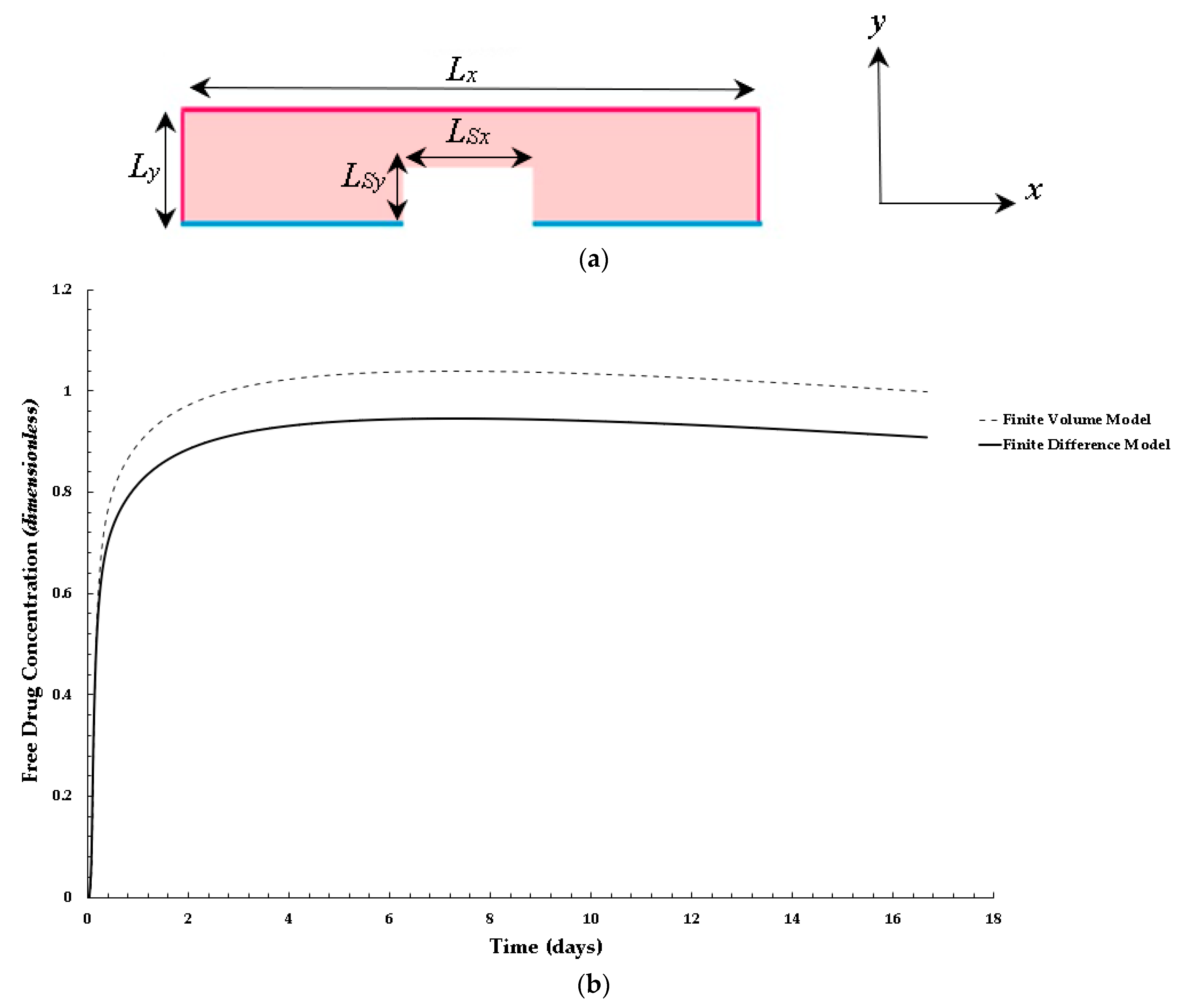
- Validation of the reported computational model via simulation-based results from a finite difference model developed from methods reported in previous works.
- A computational drug diffusion model that provides an understanding of the relationship between drug physicochemical properties and the local transport environment which is crucial to the success of new stent designs.
- The model reported in this work is the second reported model in literature that successfully uses an ANSYS FLUENT user-defined scalar (UDS) model to model the diffusion of the free and bound drug in the arterial wall with reversible binding source terms. Additionally, this is the first reported model to use a UDS model to incorporate the polymer layer in the computational domain.
2. Material and Methods
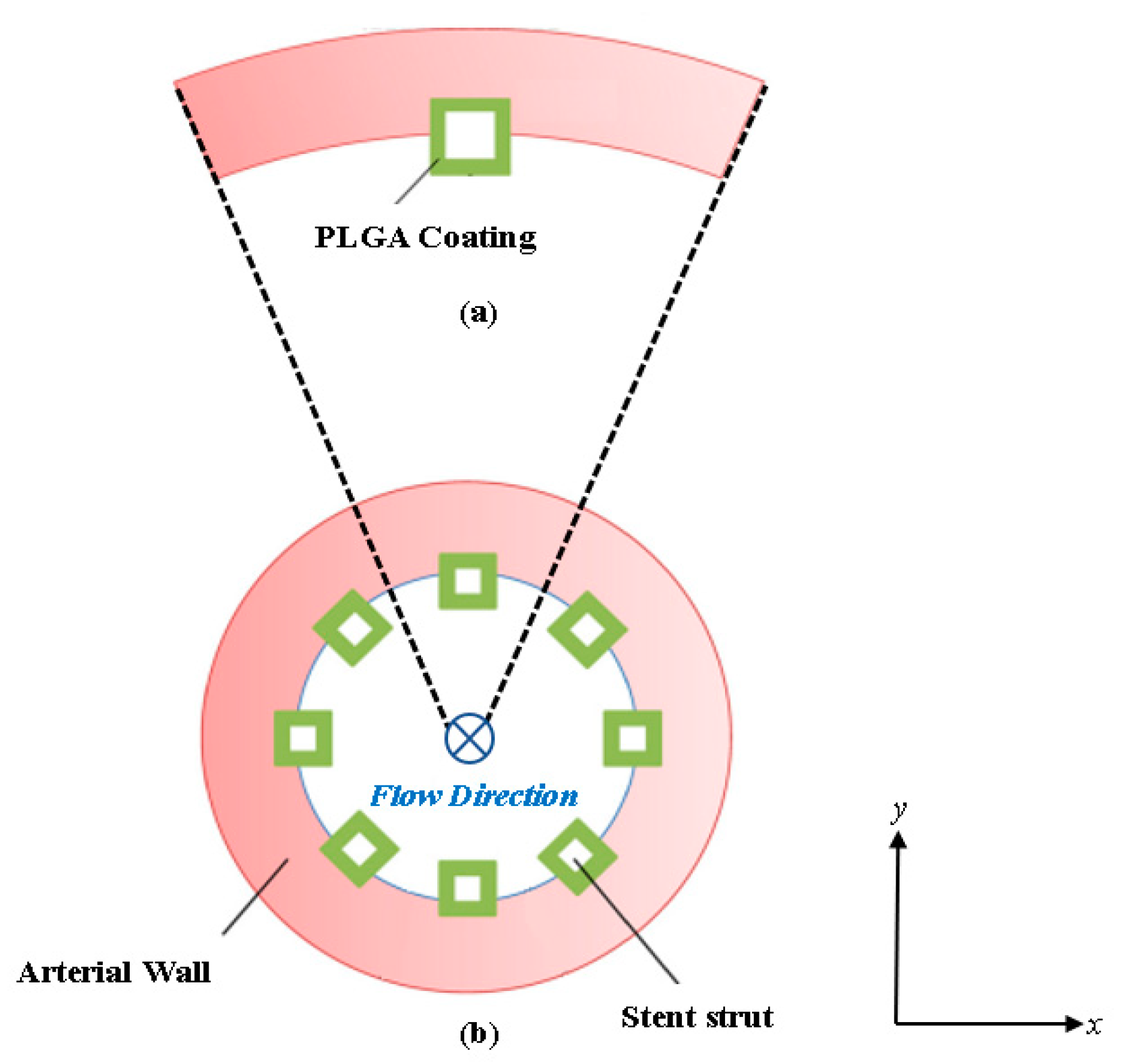
2.1. Model Development
2.2. Boundary Conditions and Meshing
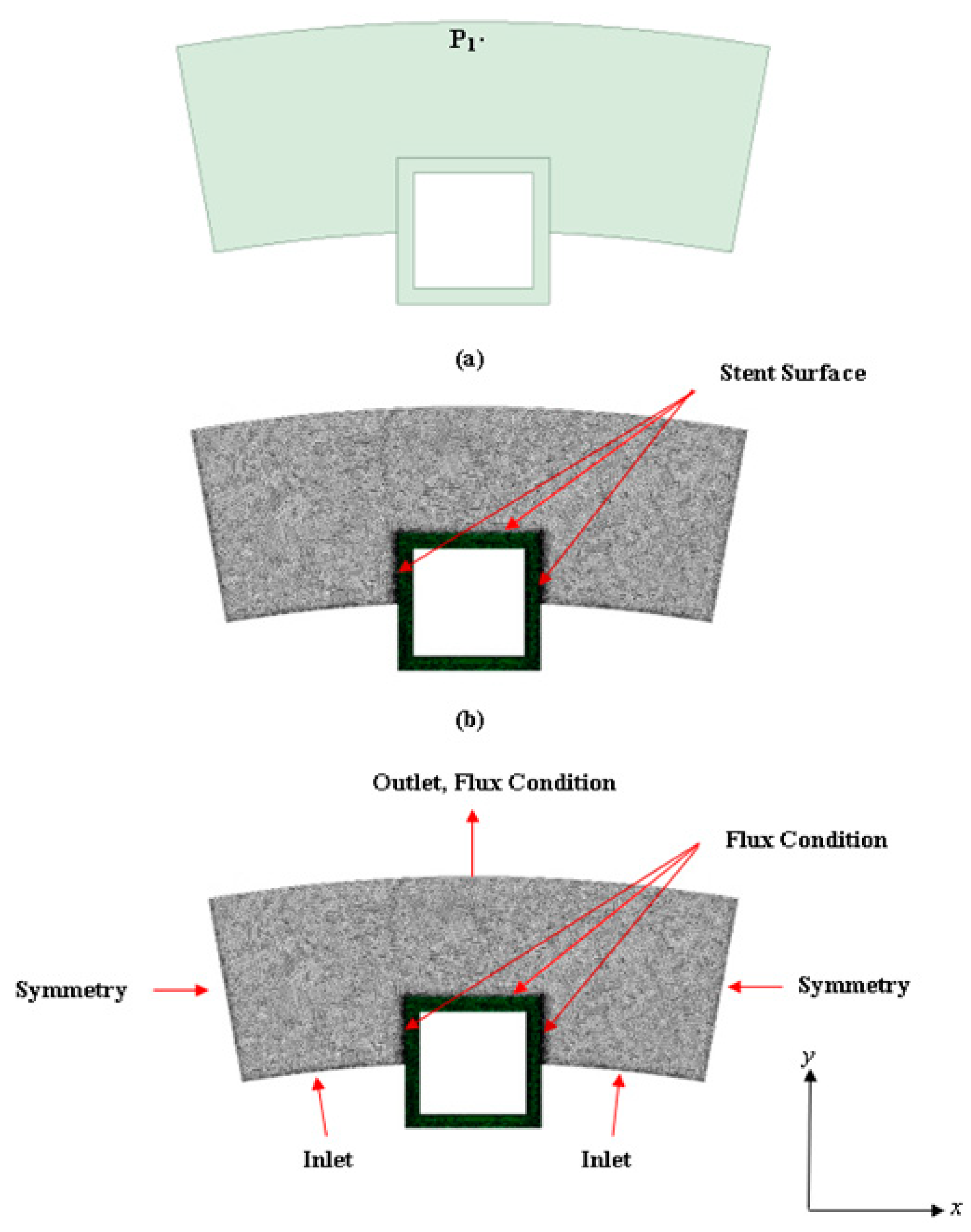
2.3. Plasma Flow
2.4. Drug Transport in the PLGA Coating and Arterial Domains
- Drug Binding:
- Free Drug in the PLGA Coating Domain:
- Free Drug in the Arterial Domain:
- Bound Drug in the Arterial Domain:
- Drug Transport Boundary Conditions:
- User Defined Scalar and Numerical Modelling
2.5. Non-Dimensional Pre-Analyses
2.6. Grid Independence Analysis, Modelling Parameters, and Validation and Verification
3. Results
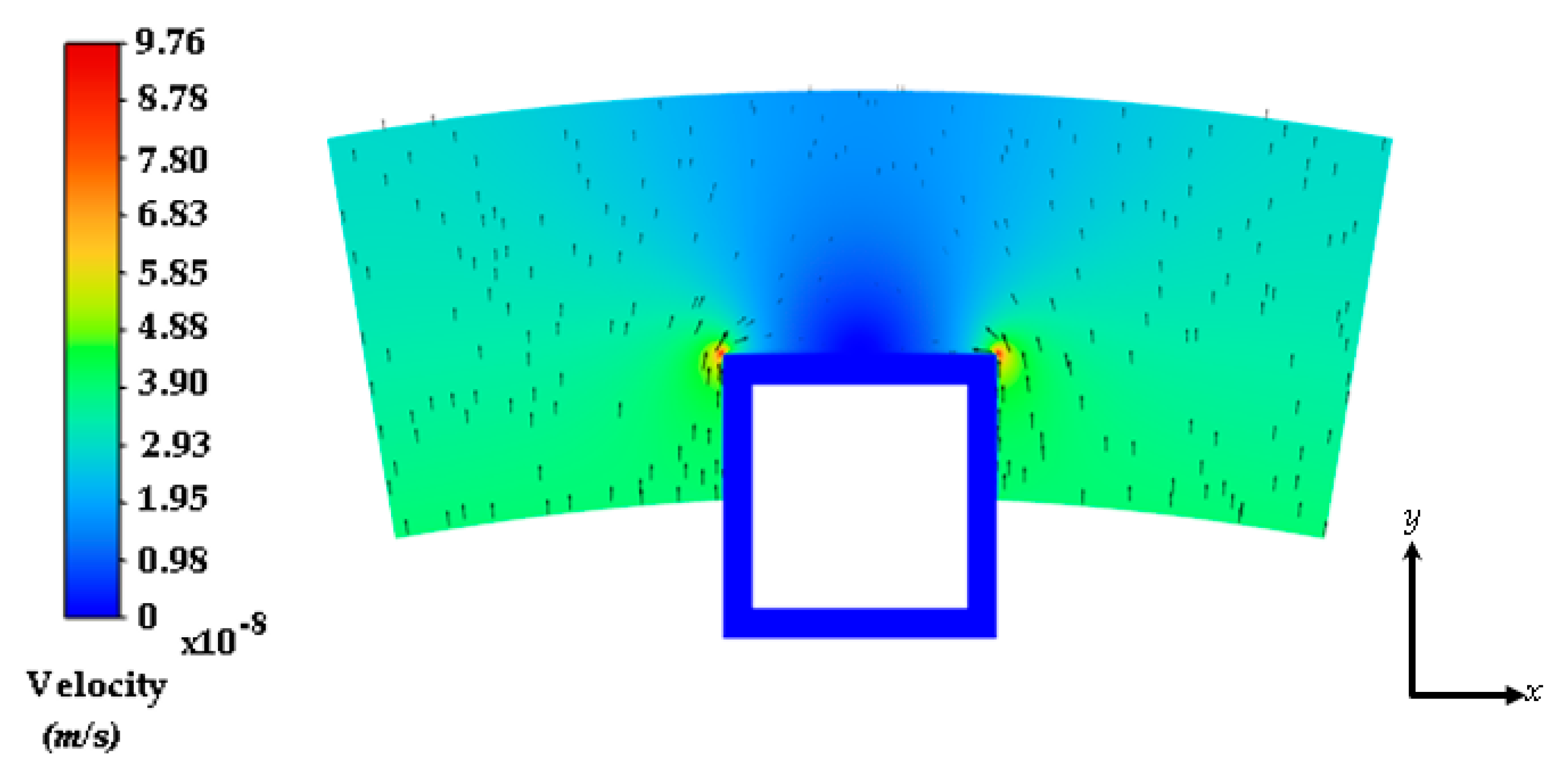
3.1. Interstitial Flow into the Arterial Wall
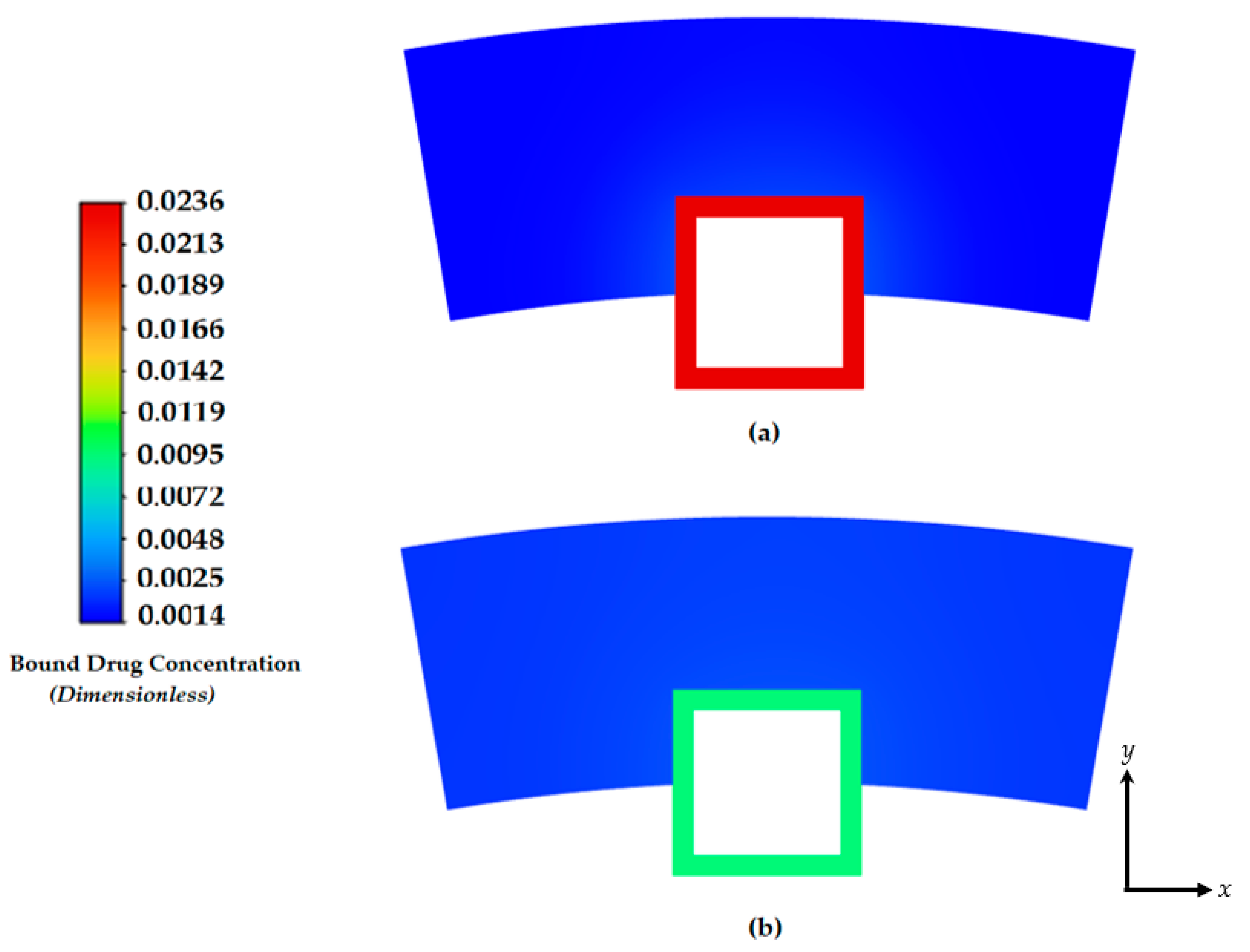
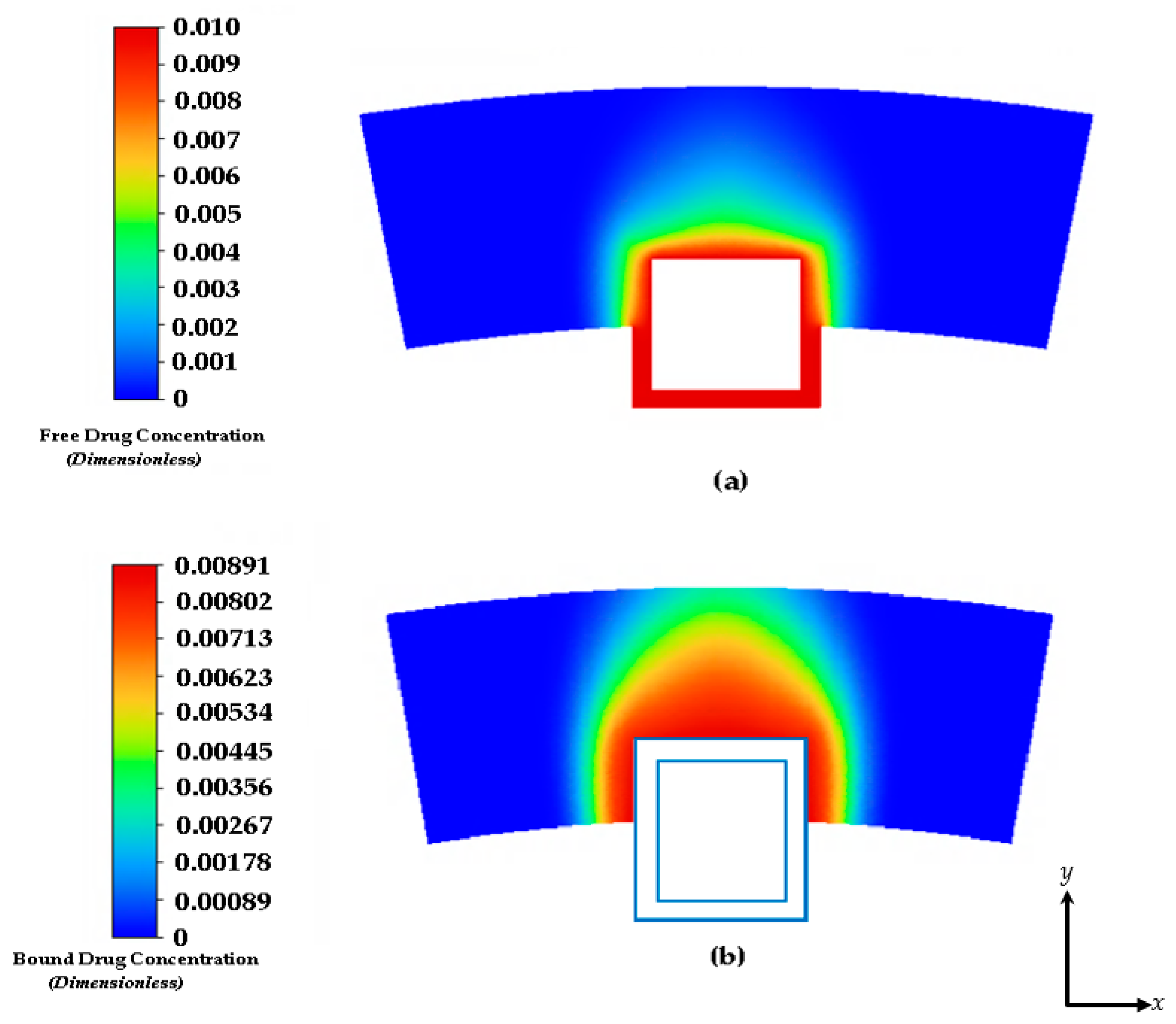
3.2. Free and Bound Drug Concentration Profiles with Erosion and Interstitial Flow
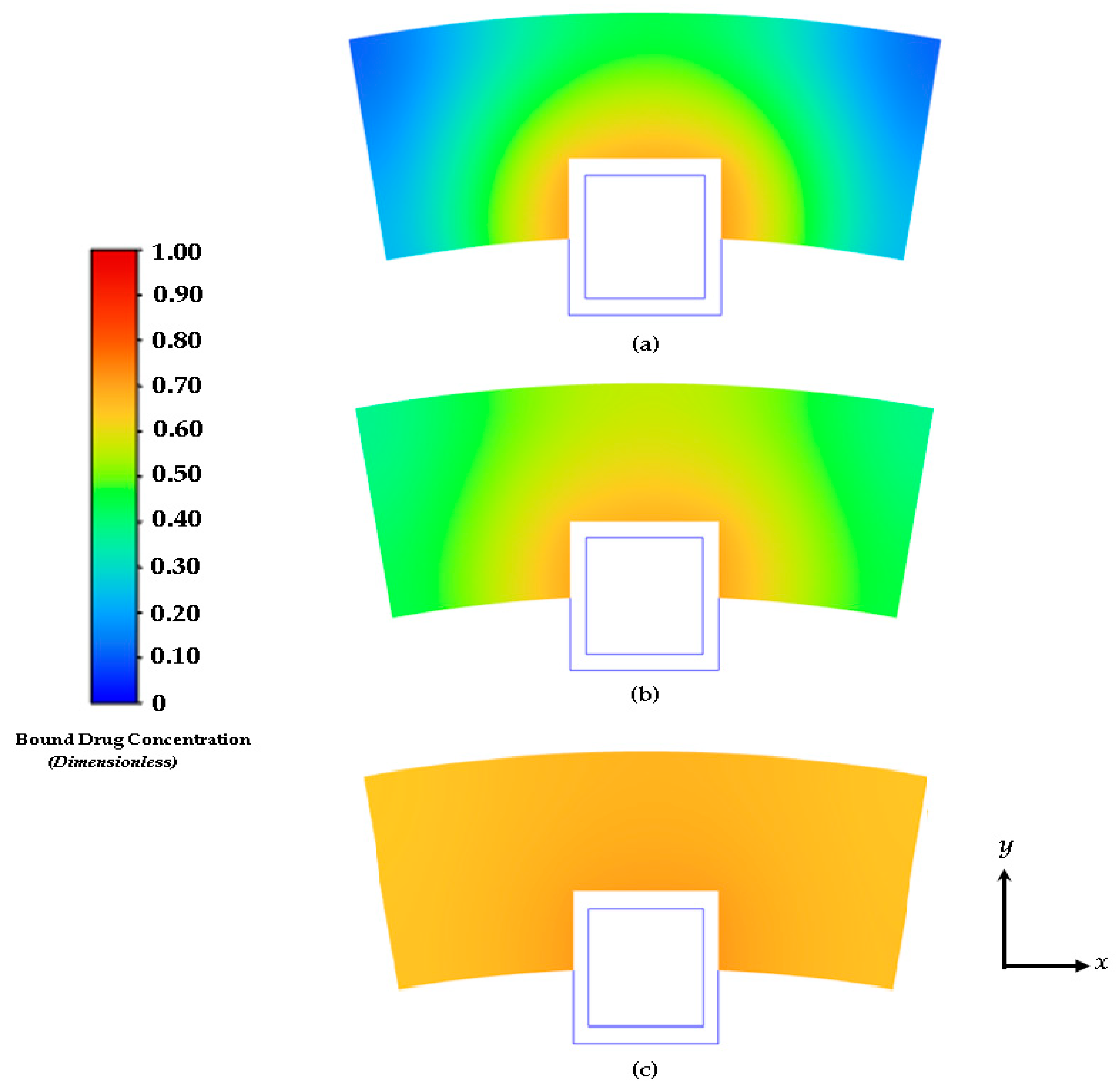
3.3. Free and Bound Drug Concentration Profiles with Erosion and Convection
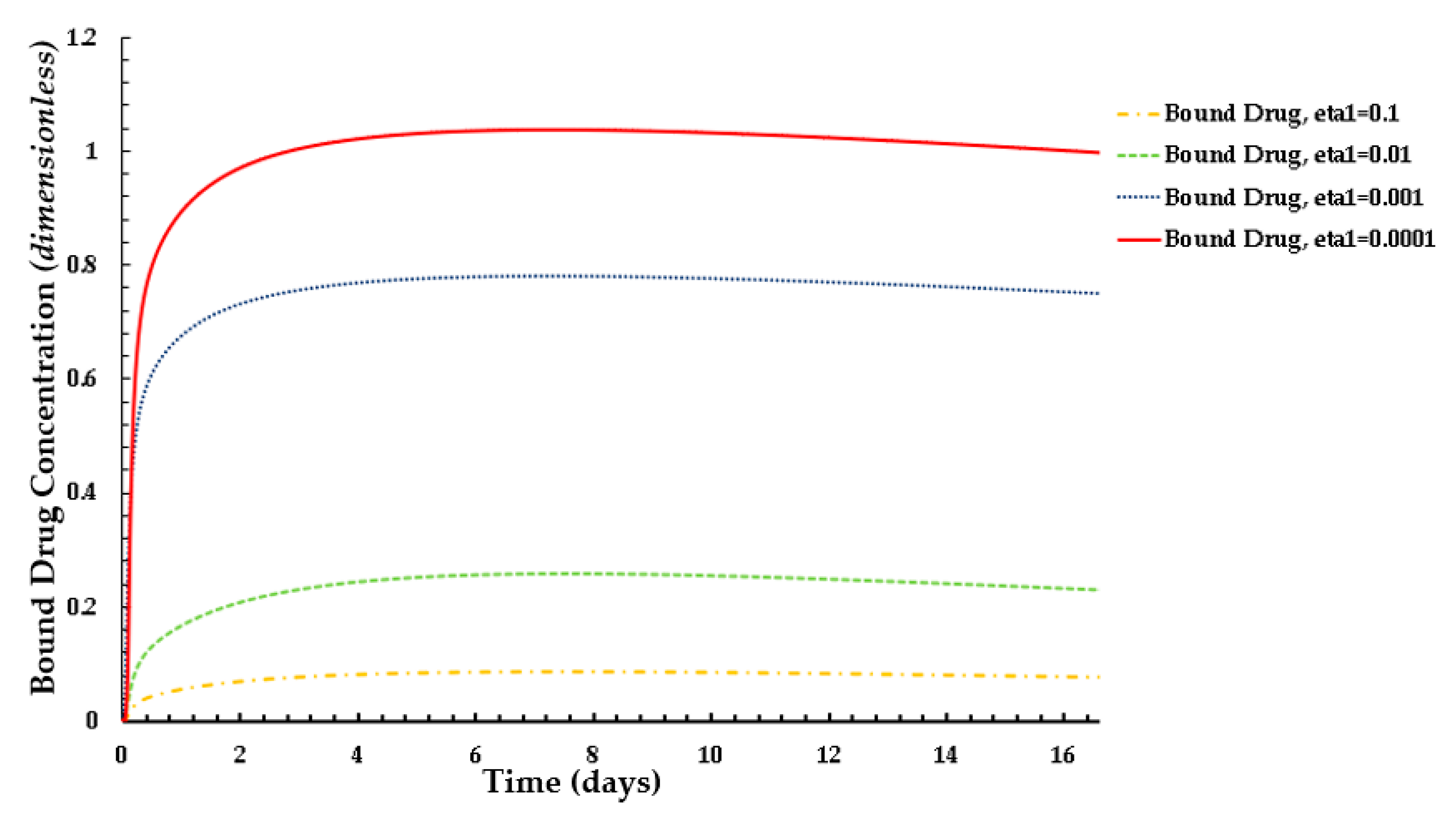
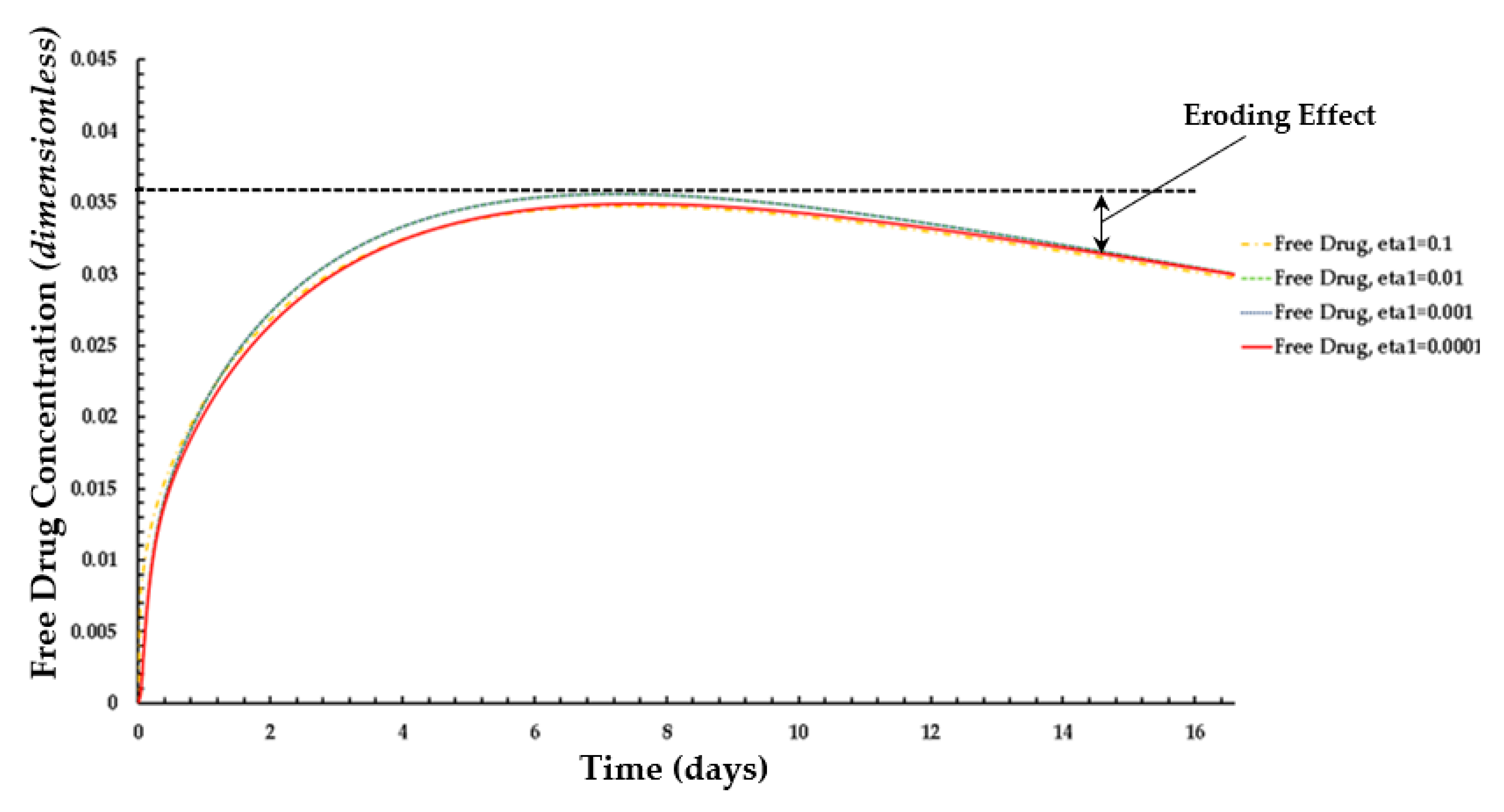
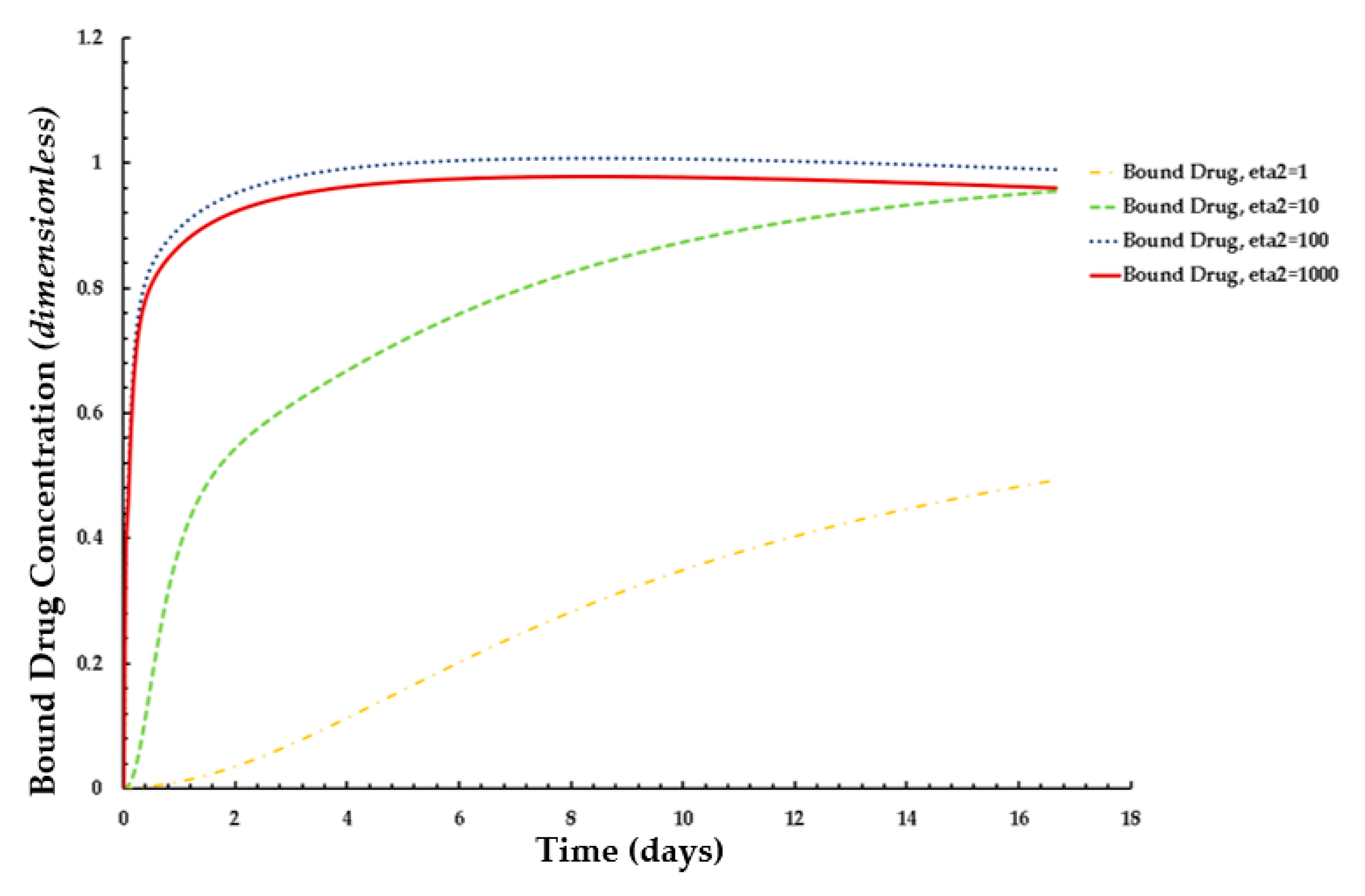
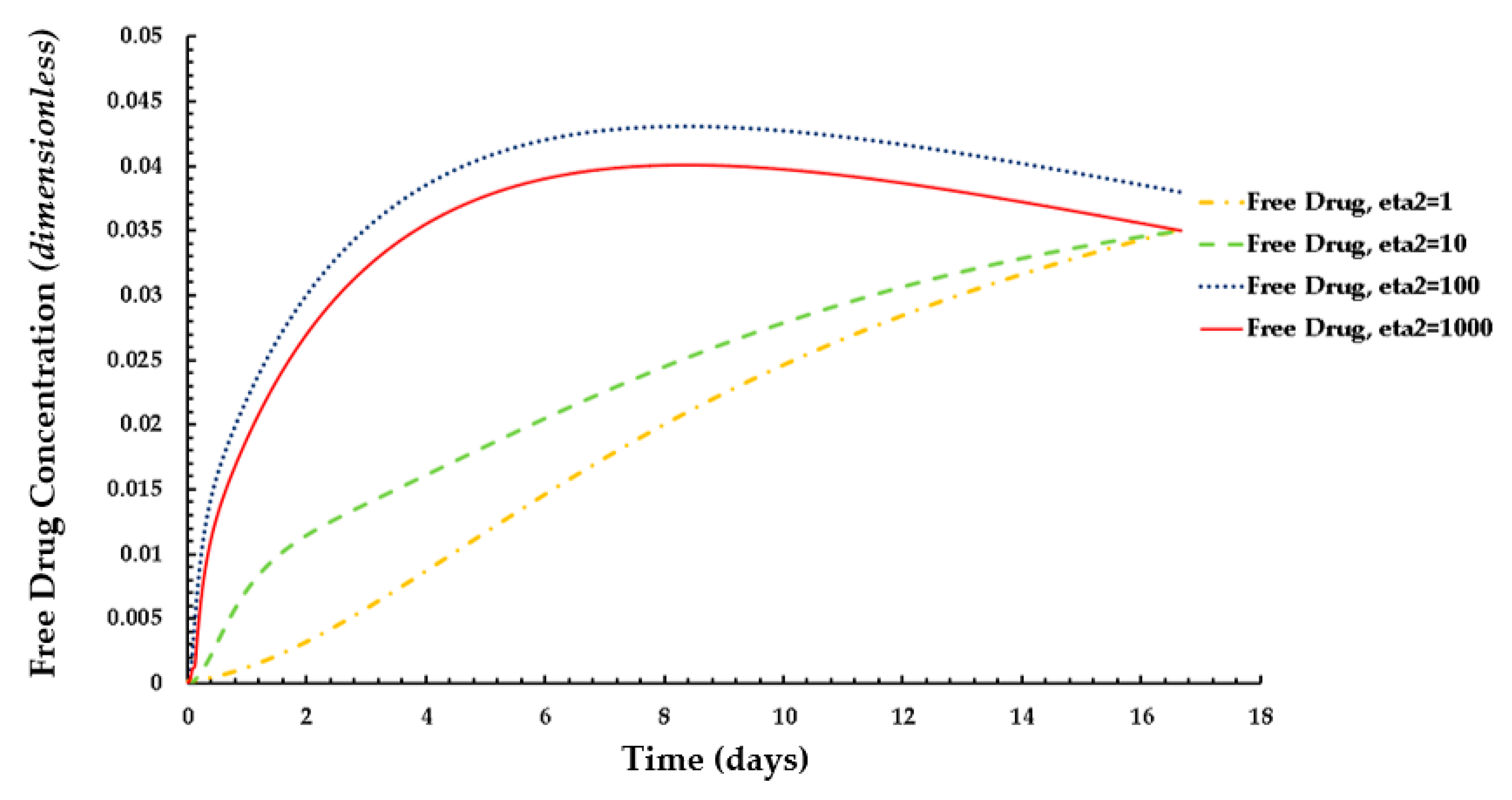
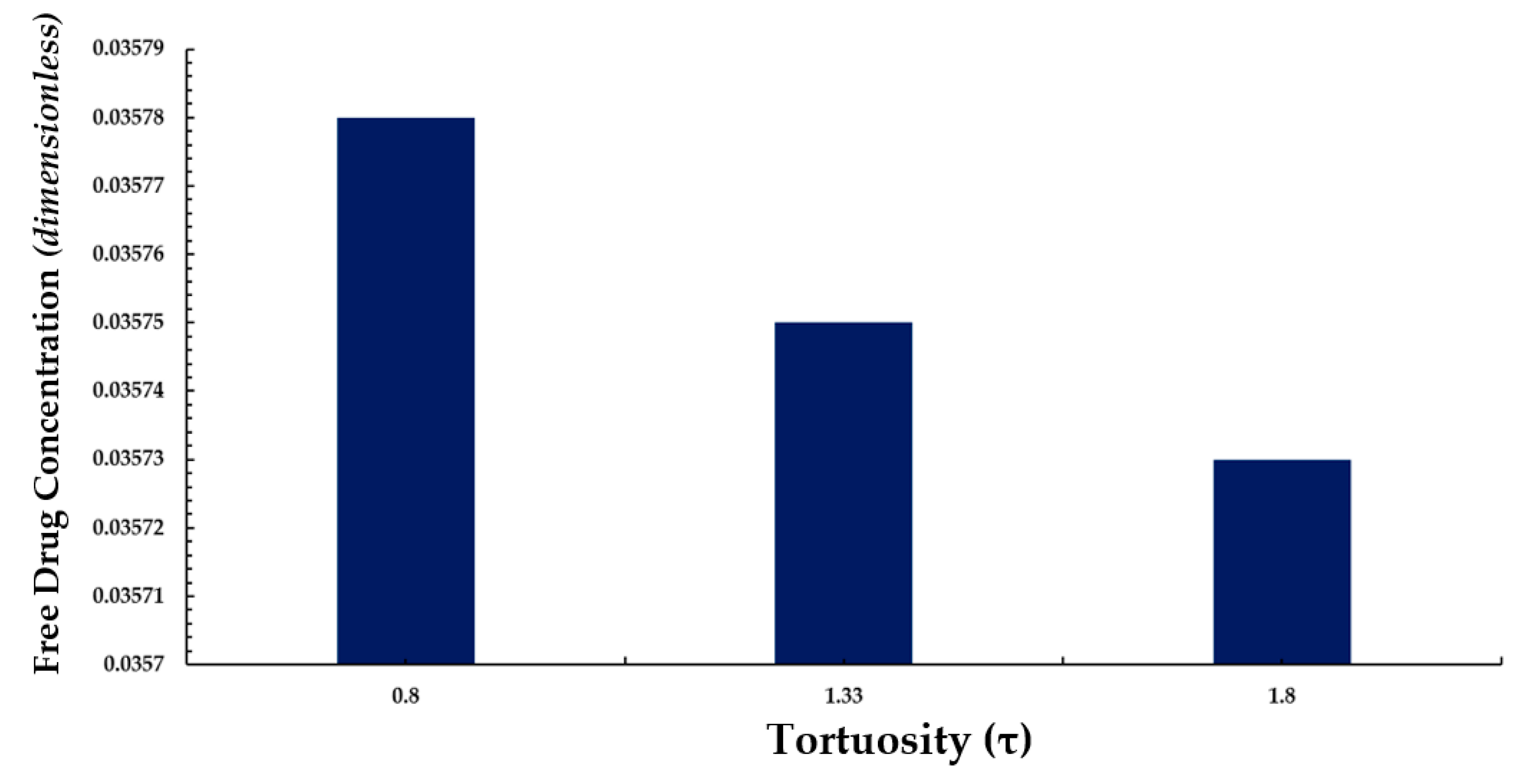
3.4. Average Weighted Concentration Results for Varying Tortuosity
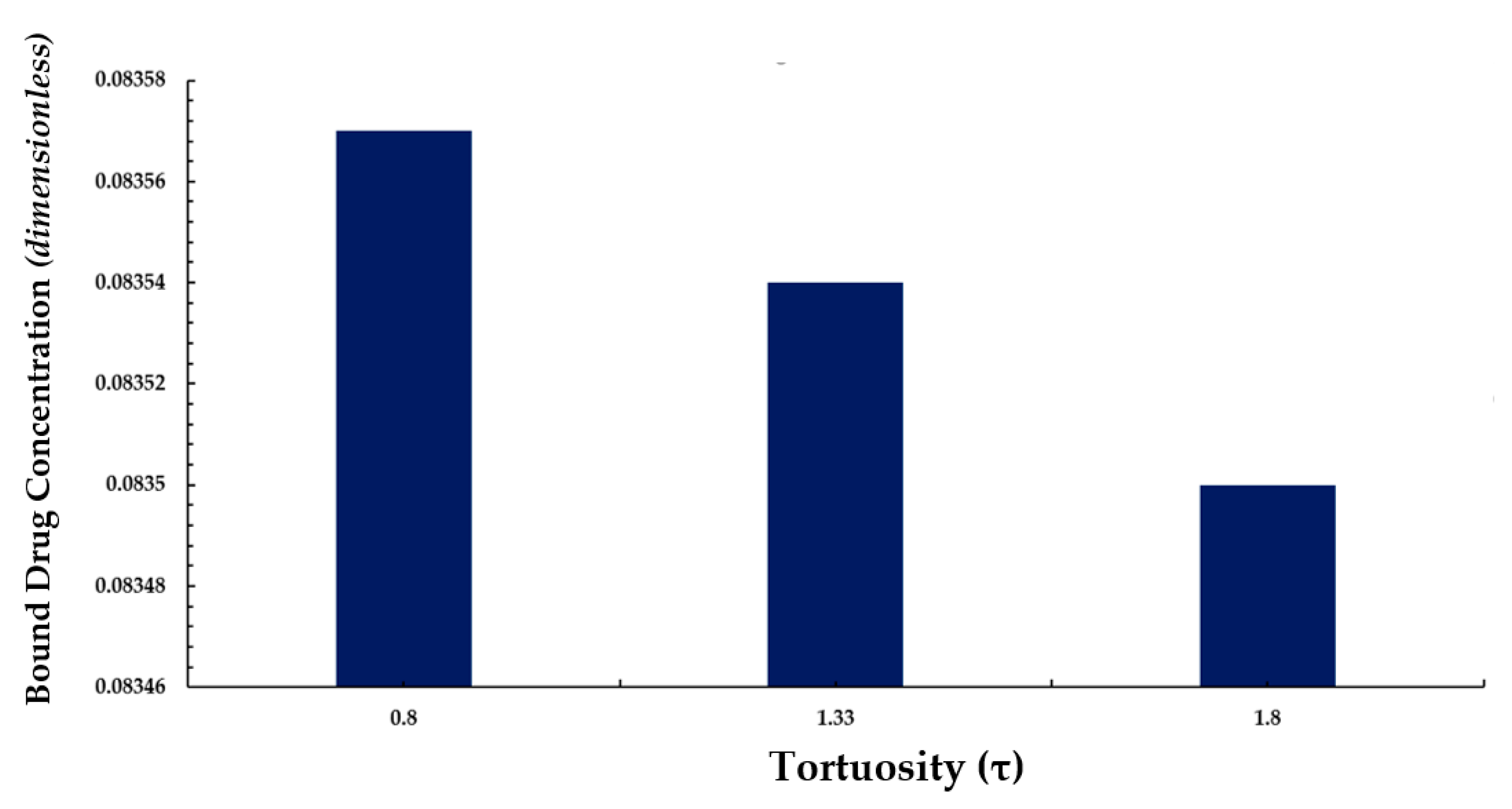
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
Abbreviations
| Cf | Free drug |
| Cb | Bound drug |
| Cp | Perivascular drug concentration |
| Cw | Wall drug concentration |
| C0 | Initial drug concentration |
| D | Outer diameter of the artery |
| DC | Coefficient of the coating diffusion |
| Da | Dimensionless DamKöhler number in the tissue |
| Dfree | Coefficient of free diffusivity |
| Deff | Coefficient of effective diffusivity |
| DT | True diffusivity of the free drug |
| Jwp | PLGA flux parameter |
| Lx | Arterial domain length |
| Ly | Arterial domain wall thickness |
| Lsx | Stent length |
| Lsy | Stent thickness |
| ka | Tissue binding capacity |
| kd | Dissociation rate constant |
| PeC | Dimensionless Peclet number in the coating |
| PeT | Dimensionless Peclet number in the tissue |
| Rd | Equilibrium dissociation constant |
| Rwp | Mass transfer resistance |
| S0 | Available binding sites |
| T | Time |
| Vy | Transmural filtration velocity |
| x | x-coordinate |
| y | y-coordinate |
| δ | Strut dimension |
| ε | Porosity of the arterial wall |
| ε1 and ε2 | Dimensionless scaling parameters |
| τ | Tortuosity of the arterial wall |
| τ1, τ2, and τ2 | Characteristic time scales |
| PLGA | Poly(lactic-co-glycolic acid) |
| UDS | User defined scalar |
Appendix A
- Free-drug in the PLGA Coating Domain:
- Free-drug in the Arterial Domain:
- Bound-drug in the Arterial Domain:
- Scaled free-drug concentration time derivative:
- Scaled bound-drug concentration time derivative:
- Scaled free-drug concentration first-order x-direction derivative:
- Scaled free-drug concentration first-order y-direction derivative:
- Scaled free-drug concentration second-order x-direction derivative:
- Scaled free-drug concentration second-order y-direction derivative:
References
- Edwards, M.; Hewlin, R.L., Jr.; Smith, M. A 2-D Transient Computational Multi-Physics Model for Analyzing Magnetic and Non-Magnetic (Red Blood Cells and E. Coli Bacteria) Particle Dynamics in a Travelling Wave Ferro-Magnetic Microfluidic Device. ASME J. Eng. Sci. Med. Ther. Diagn. 2023, 1–47. [Google Scholar] [CrossRef]
- Hewlin, R.L., Jr.; Edwards, M. Continuous Flow Separation of Red Blood Cells and Platelets in a Y-Microfluidic Channel Device with Saw-Tooth Profile Elec-trodes via Low Voltage Dielectrophoresis. Curr. Issues Mol. Biol. 2023, 45, 3048–3067. [Google Scholar] [CrossRef] [PubMed]
- Hewlin, R.L., Jr.; Ciero, A.; Kizito, J.P. Development of a Two-Way Coupled Eulerian-Lagrangian Computational Magnetic Nanoparticle Targeting Model for Pulsatile Flow in a Patient-Specific Diseased Left Carotid Bifurcation Artery. Cardiovasc. Eng. Technol. 2019, 10, 299–313. [Google Scholar] [CrossRef] [PubMed]
- Stanley, N.; Ciero, A.; Timms, W.; Hewlin, R.L., Jr. A 3-D Printed Optically Clear Rigid Diseased Carotid Bifurcation Arterial Mock Vessel Model for Particle Image Velocimetry Analysis in Pulsatile Flow. ASME Open J. Eng. ASME 2023, 2, 021010. [Google Scholar] [CrossRef]
- Hewlin, R.L., Jr.; Tindall, J.M. Computational Assessment of Magnetic Nanoparticle Targeting Efficiency in a Simplified Circle of Willis Arterial Model. Int. J. Mol. Sci. 2023, 24, 2545. [Google Scholar] [CrossRef] [PubMed]
- Hewlin, R.L., Jr.; Kizito, J.P. Comparison of Carotid Bifurcation Hemodynamics in Patient-Specific Geometries at Rest and During Exercise. In Proceedings of the Fluids Engineering Division Summer Meeting. American Society of Mechanical Engineers, Volume 1A, Symposia: Advances in Fluids Engineering Education; Advances in Numerical Modeling for Turbomachinery Flow Optimization; Applications in CFD; Bio-Inspired Fluid Mechanics; CFD Verification and Validation; Development and Applications of Immersed Boundary Methods; DNS, LES, and Hybrid RANS/LES Methods, Incline Village, NV, USA, 7–11 July 2013; p. V01AT04A001. [Google Scholar] [CrossRef]
- Stanley, N.; Ciero, A.; Timms, W.; Hewlin, R.L., Jr. Development of 3-D Printed Optically Clear Rigid Anatomical Vessels for Particle Image Velocimetry Analysis in Cardiovascular Flow. In Proceedings of the ASME International Mechanical Engineering Congress and Exposition. American Society of Mechanical Engineers, Volume 7: Fluids Engineering, Salt Lake City, UT, USA, 11–14 November 2019; p. V007T08A004. [Google Scholar] [CrossRef]
- Hewlin, R.L., Jr.; Kizito, J.P. Development of an Experimental and Digital Cardiovascular Arterial Model for Transient Hemodynamic and Postural Change Studies: “A Preliminary Framework Analysis”. Cardiovasc. Eng. Tech. 2018, 9, 1–31. [Google Scholar] [CrossRef]
- Daemen, J.; Serruys, P.W. Drug-Eluting Stent Update 2007 Part I: A Survey of Current and Future Generation Drug-Eluting Stents: Meaningful Advances or More of the Same. Circulation 2007, 116, 316–328. [Google Scholar] [CrossRef]
- Beshchasna, N.; Saqib, M.; Kraskiewicz, H.; Wasyluk, Ł.; Kuzmin, O.; Duta, O.C.; Ficai, D.; Ghizdavet, Z.; Marin, A.; Ficai, A.; et al. Recent Advances in Manufacturing Innovative Stents. Pharnaceutics 2020, 12, 349. [Google Scholar] [CrossRef]
- Conway, C. Clinical Evidence vs. the Testing Paradigm. Cardiovasc. Eng. Technol. 2018, 9, 752–760. [Google Scholar] [CrossRef]
- Beckman, J.A.; White, C.J. Paclitaxel-Coated Balloons and Eluting Stents: Is There a Mortality Risk in Patients with Peripheral Artery Disease. Circulation 2019, 140, 1342–1351. [Google Scholar] [CrossRef]
- Capell, W.H.; Bonaca, M.P.; Nehler, M.R.; Kittelson, J.M.; Anand, S.S.; Berkowitz, S.D.; Debus, E.S.; Fanelli, F.; Haskell, L.; Patel, M.R.; et al. Rationale and Desitn for the Vascular Outcomes Study of ASA Along with Rivaroxaban in Endovascular or Surgical Limb Revascularization for Peripheral Artery Disease. Am. Heart J. 2018, 199, 83–91. [Google Scholar] [CrossRef]
- US Food and Drug Administration. Update: Treatment of Peripheral Arterial Disease with Paclitaxel-Coated Ballons and Paclitaxel-Eluting Stents Potentiall Associated with Increased Mortality: Letter to Health Care Providers. Available online: https://www.fda.gov/medical-devices/letters-health-care-providers/update-treatment-peripheral-arterial-disease-paclitaxel-coated-balloons-and-paclitaxel-eluting (accessed on 5 May 2022).
- Levin, A.; Jonas, M.; Hwang, C.W.; Edelman, E. Local and Systemic Drug Competition in Drug-Eluting Stent Tissue Deposition Properties. J. Control. Release 2005, 109, 226–243. [Google Scholar] [CrossRef] [PubMed]
- Granada, J.F.; Stenoien, M.; Buszman, P.P.; Tellez, A.; Langanki, D.; Kaluza, G.L.; Leon, M.B.; Gray, W.; Jaff, M.R.; Schwartz, R.S. Mechanisms of Tissue Uptake and Retention of Paclitaxel-Coated Balloons: Impact on Neointimal Proliferation and Healing. Open Heart 2014, 1, e000117. [Google Scholar] [CrossRef]
- Lovich, M.A.; Philbrook, M.; Sawyer, S.; Weselcouch, E.; Edelman, E.R. Arterial Heparin Deposition: Role of Diffusion, Convection, and Extravascular Space. Am. J. Physiol. 1998, 275, H2236–H2242. [Google Scholar] [CrossRef]
- Moses, J.W.; Stone, G.W.; Nikolsky, E.; Mintz, G.S.; Dangas, G.; Grube, E. Drug-eluting Stents in the Treatment of Intermediate Lesions: Pooled Analysis from four Randomized Trials. J. Am. Coll. Cardiol. 2006, 47, 2164–2171. [Google Scholar] [CrossRef]
- Serruys, P.W.; Sianos, G.; Abizaid, A.; Aoki, J.; den Heijer, P.; Bonnier, H. The Effect of Variable Dose and Release Kinetics on Neointimal Hyperplasia using a Novel Paclitaxel-eluting Stent Platform: The Paclitaxel In-Stent Controlled Elution Study (Pices). J. Am. Coll. Cardiol. 2005, 46, 253–260. [Google Scholar] [CrossRef] [PubMed]
- Lovich, M.A.; Edelman, E.R. Computational Simulations of Local Vascular Heparin Deposition and Distribution. Am. J. Physiol. 1996, 271, H2014–H2024. [Google Scholar] [CrossRef]
- Sakharov, D.V.; Kalachev, L.V.; Rijken, D.C. Numerical Simulation of Local Pharmcokinetics of a Drug after Intervascular Delivery with an Eluting Stent. J. Drug Target. 2002, 10, 507–513. [Google Scholar] [CrossRef] [PubMed]
- Hwang, C.; Wu, D.; Edelman, E. Physiological Transport Forces Govern Drug Distribution for Stent-Based Delivery. Circulation 2001, 104, 600–605. [Google Scholar] [CrossRef]
- Migiliavacca, F.; Gervaso, F.; Prosi, M.; Zunino, P.; Minisini, S.; Formaggia, L. Expansion and Drug Elution Model of a Coronary Stent. Comput. Methods Biomech. Biomed. Eng. 2007, 10, 63–73. [Google Scholar] [CrossRef]
- Borghi, A.; Foa, E.; Balossino, R.; Migliavacca, F.; Dubini, G. Modelling Drug Elutiogn from Stents: Effects of Reversible Binding in the Vascular Wall and Degradable Polymeric Matrix. Comput. Methods Biomech. Biomed. Eng. 2008, 11, 367–377. [Google Scholar] [CrossRef]
- Horner, M.; Joshi, S.; Dhruva, V.; Sett, S.; Stewart, S.F.C. A Two-Species Drug Delivery Model is Required to Predict Deposition from Drug-Eluting Stents. Cardiovasc. Eng. Technol. 2010, 1, 225–234. [Google Scholar] [CrossRef]
- Tzafriri, A.R.; Levin, A.D.; Edelman, E.R. Diffusion-limited Binding Explains Binary Dose Response for Local Arterial and Tumor Drug Delivery. Cell Prolif. 2009, 42, 348–363. [Google Scholar] [CrossRef]
- Higuchi, T. Theoretical Analysis of Rate of Release of Solid Drugs Dispersed in Solid Matrices. J. Pharm. Sci. 1963, 52, 1145–1149. [Google Scholar] [CrossRef] [PubMed]
- Mandal, A.P.; Mandal, P.K. An Unsteady Analysis of Arterial Drug Transport from Half-Embedded Drug Eluting Stent. Appl. Math. Comput. 2015, 266, 968–981. [Google Scholar] [CrossRef]
- Mandal, A.P.; Mandal, P.K. Computational Modelling of Three-Phase Stent-Based Delivery. J. Explor. Res. Pharm. 2017, 2, 31–40. [Google Scholar] [CrossRef][Green Version]
- Saltzman, W.M. Drug Delivery: Engineering Principles for Drug Therapy; Oxford University Press: Oxford, UK, 2001. [Google Scholar]
- Sarifuddin; Mandal, P.K. Effect of Diffusivity on the Transport of Drug Eluted from Drug Eluting Stent. Int. J. Appl. Comput. Math. 2016, 2, 291–301. [Google Scholar] [CrossRef]
- O’Connel, B.M.; Walsh, M.T. Demonstrating the Influence of Compression on Artery Wall Mass Transport. Ann. Biomed. Eng. 2010, 38, 1354–1366. [Google Scholar] [CrossRef]
- Edwards, M.; Kizito, J.P.; Hewlin, R.L., Jr. A Time-Dependent Two Species Explicit Finite Difference Computational Model for Analyzing Diffusion in a Drug Eluting Stented Coronary Artery Wall: A Phase I Study. In Proceedings of the ASME International Mechanical Engineering Congress and Exposition. American Society of Mechanical Engineers, Volume 4: Biomedical and Biotechnology; Design, Systems, and Complexity, Columbus, OH, USA, 30 October–3 November 2022; p. V004T05A009. [Google Scholar] [CrossRef]
- Zhu, X.; Pack, D.W.; Braatz, R.D. Modelling Intravascular Delivery from Drug-Eluting Stents with Biodegradable Coating: Investigation of Anisotropic Vascular Drug Diffusivity and Arterial Drug Distribution. Comput. Methods Biomech. Biomed. Eng. 2014, 17, 187–198. [Google Scholar] [CrossRef]
- Saha, R.; Mandal, P.K. Modelling Time-dependent Release Kinetics in Stent-based Delivery. J. Explor. Res. Pharmacol. 2018, 3, 61–70. [Google Scholar] [CrossRef][Green Version]
- Zhu, X.; Braatz, R.D. Modelling and Analysis of Drug-Eluting Stents With Biodegradable PLGS Coating: Consequences on Instravascular Drug Delivery. J. Biomech. Eng. 2015, 136, 111004. [Google Scholar] [CrossRef]
- Balakrishnan, B.; Tzafriri, A.R.; Arifin, D.Y.; Edelman, E.R. Intravascular Drug Release Kinetics Dictate Arterial Drug Deposition, Rentention and Distribution. J. Control. Release 2007, 123, 100–108. [Google Scholar] [CrossRef] [PubMed]
- Vairo, G.; Cioffi, M.; Cottone, R.; Dubini, G.; Migliavacca, F. Drug Release From Coronary Eluting Stents: A Multidomain Approach. J. Biomech. Eng. 2010, 43, 1580–1589. [Google Scholar] [CrossRef]
- Hewlin, R.L., Jr. Transient Cardiovascular Hemodynamics in a Patient-Specific Arterial System. Ph.D. Thesis, North Carolina Agricultural and Technical State University, Greensboro, NC, USA, 2015. [Google Scholar]
- Whale, M.; Grodzinsky, A.; Johnson, M. The Effect of Aging and Pressure on the Specific Hydraulic Conductivity of the Aortic Wall. Biorheology 1996, 33, 17–44. [Google Scholar] [CrossRef]
- Kolachalama, V.; Tzafriri, A.; Arifin, D.; Edelman, E. Luminal Flow Patterns Dictate Arterial Drug Deposition in Stent-Based Delivery. J. Control. Release 2009, 133, 24–30. [Google Scholar] [CrossRef]
- Baldwin, A.L.; Wilson, I.; Gardus-Pizlo, R.; Wilensky, R.; March, K. Effect of Atherosclerosis on Transmural Convection and Arterial Ultrastructure: Implications for Loacl Vascualr Drug Delivery. Aterioscler. Thromb. Vasc. Biol. 1997, 17, 3365–3375. [Google Scholar] [CrossRef]
- Wang, D.; Tarbell, J. Modelling Interstitial Flow in an Artery Wall Allows Estimation of Wall Shear Stress on Smooth Muscle Cells. ASME J. Biomech. Eng. 1995, 117, 358–363. [Google Scholar] [CrossRef]
- Santin, M.; Colombo, P.; Brushci, G. Interfacial Biology of In-Stent Restenosis. Expert Rev. Med. Dev. 2005, 2, 429–443. [Google Scholar] [CrossRef]
- Balakrishnan, B.; Tzafriri, A.R.; Seifert, P.; Groothuis, A.; Rogers, C.; Edelman, E.R. Strut Position, Blood Flow, and Drug Deposition-Implications for Single and Overlapping Drug-Eluting Stents. Circulation 2005, 111, 2958–2965. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.T.; Venkatraman, S.S.; Boey, F.Y.C.; Loo, J.S.C.; Tan, L.P. Controlled Release of Sirolimus from a Multilayered PLGA Stent Matrix. Biomaterials 2006, 27, 5588–5595. [Google Scholar] [CrossRef] [PubMed]
- Castellot, J.J.; Wong, K.; Herman, B.; Hoover, R.L.; Albertini, D.F.; Wright, T.C.; Caleb, B.L.; Karnovsky, M.J. Binding and Internalization of Heparin by Vascular Smooth Muscle Cells. J. Cell. Physiol. 1985, 124, 13–20. [Google Scholar] [CrossRef] [PubMed]
- Deux, J.-F.; Meddahi-Pelle, A.; Le Blanche, A.F.; Feldman, L.J.; Colliec-Jouault, S.; Brée, F.; Boudghène, F.; Michel, J.-B.; Letourneur, D. Low Molecular Weight Fucoidan Prevents Neointimal Hyperplasia in Rabbit Iliac Artery In-Stent Restenosis Model. Arter. Thromb. Vasc. Biol. 2002, 22, 1604–1609. [Google Scholar] [CrossRef] [PubMed]
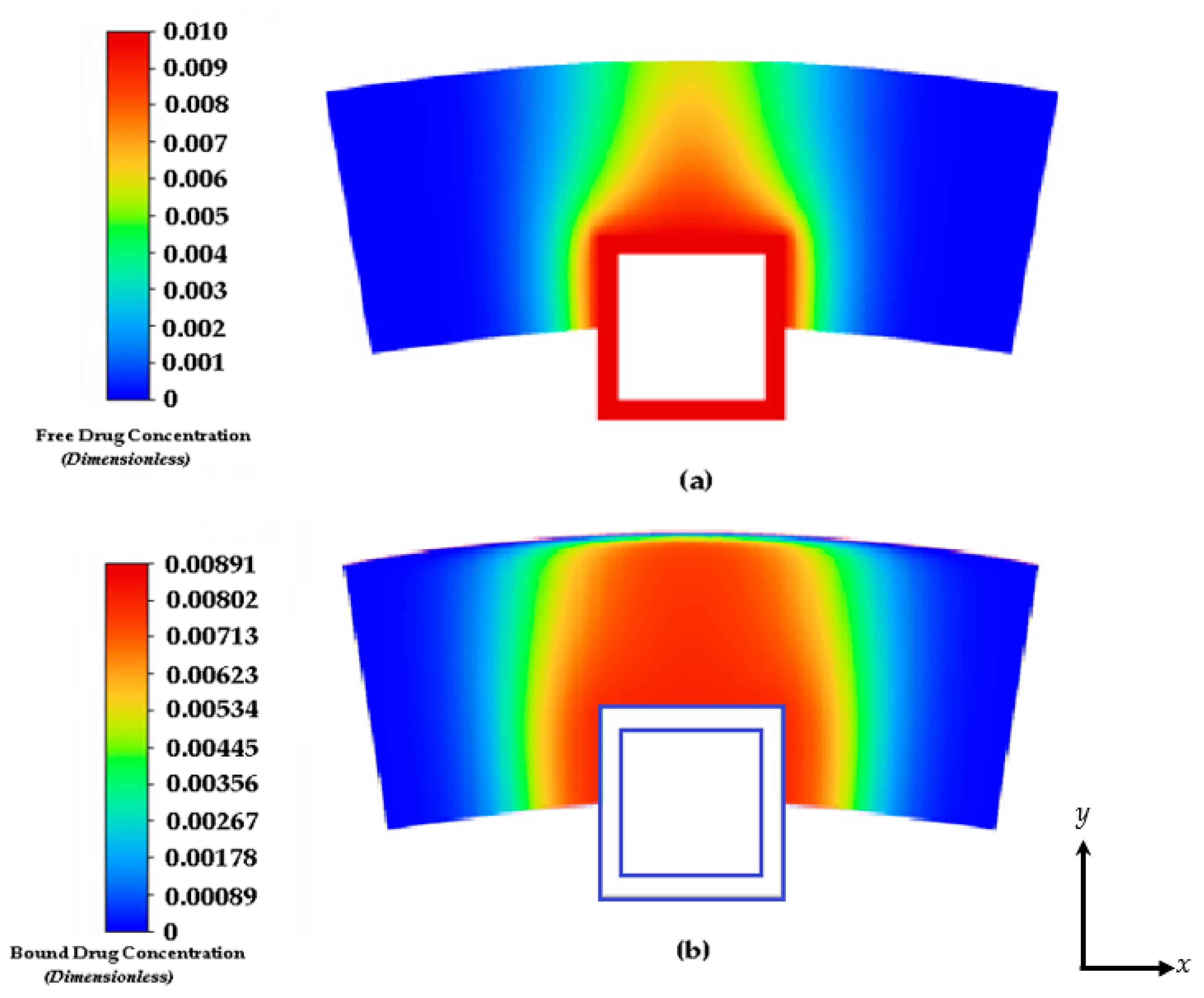
| Element Number | Average Weighted Concentration | Average Velocity |
|---|---|---|
| 74,212 | 1.121327 | 13.72 × 10−6 |
| 82,458 | 1.057641 | 12.68 × 10−6 |
| 91,621 | 1.034241 | 10.23 × 10−6 |
| 101,802 | 0.983541 | 9.83 × 10−6 |
| 113,114 | 0.977732 | 9.77 × 10−6 |
| 372,125 | 0.977654 | 9.76 × 10−6 |
| Description | Parameter | Value |
|---|---|---|
| Outer diameter of the artery, mm | D | 3 |
| Artery wall thickness, µm | Ly | 200 |
| Strut dimension, m | δ | 0.00014 |
| Transmural filtration velocity, m/s | Vy | 4 × 10−8 |
| Porosity of the arterial wall | ε | 0.787 |
| Tortuosity of the arterial wall | τ | 1.333 |
| Coating drug diffusivity, m2/s | Dc | 1.0 × 10−12 |
| Coefficient of free diffusivity, m2/s | Dfree | 3.65 × 10−12 |
| Coefficient of effective diffusivity, m2/s | Deff | 2.15 × 10−12 |
| True diffusivity of the free drug, m2/s | DT | 24 × 10−12 |
| Initial drug concentration in the coating, mol/m3 | C0 | 0.01 |
| Tissue binding capacity, mol/m3 | ka | 10 |
| Dissociation rate constant | kd | 0.01 |
| Equilibrium dissociation constant, mol/m3 | Rd | 0.001 |
| Dimensionless Peclet number in the coating | PeC | 100 |
| Dimensionless Peclet number in the tissue | PeT | 2 |
| Dimensionless DamKöhler number in the tissue | Da | 40 |
| Dimensionless scaling parameter 1 | ε1 | 0.001 |
| Dimensionless scaling parameter 2 | ε2 | 100 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hewlin, R.L., Jr.; Edwards, M.; Kizito, J.P. A Two-Species Finite Volume Scalar Model for Modeling the Diffusion of Poly(lactic-co-glycolic acid) into a Coronary Arterial Wall from a Single Half-Embedded Drug Eluting Stent Strut. Biophysica 2023, 3, 385-408. https://doi.org/10.3390/biophysica3020026
Hewlin RL Jr., Edwards M, Kizito JP. A Two-Species Finite Volume Scalar Model for Modeling the Diffusion of Poly(lactic-co-glycolic acid) into a Coronary Arterial Wall from a Single Half-Embedded Drug Eluting Stent Strut. Biophysica. 2023; 3(2):385-408. https://doi.org/10.3390/biophysica3020026
Chicago/Turabian StyleHewlin, Rodward L., Jr., Maegan Edwards, and John P. Kizito. 2023. "A Two-Species Finite Volume Scalar Model for Modeling the Diffusion of Poly(lactic-co-glycolic acid) into a Coronary Arterial Wall from a Single Half-Embedded Drug Eluting Stent Strut" Biophysica 3, no. 2: 385-408. https://doi.org/10.3390/biophysica3020026
APA StyleHewlin, R. L., Jr., Edwards, M., & Kizito, J. P. (2023). A Two-Species Finite Volume Scalar Model for Modeling the Diffusion of Poly(lactic-co-glycolic acid) into a Coronary Arterial Wall from a Single Half-Embedded Drug Eluting Stent Strut. Biophysica, 3(2), 385-408. https://doi.org/10.3390/biophysica3020026






