Abstract
Peripheral developing odontoma is a rare odontogenic lesion that mostly occurs in children. Their clinical and radiological features generally provide clinical suspicion of a benign lesion but only a histological examination may lead to the final diagnosis. Surgical management is generally easy and resolutive, but it can become more complicated in uncooperative patients and may thus require additional procedures to perform a fit surgery. We report a case of peripheral developing odontoma of the palatal aspect of the anterior maxilla that occurred in an uncooperative child subsequently treated by diode laser surgery, highlighting the benefit of this kind of surgery in pediatric patients. Data from the literature on peripheral developing odontoma have been collected and discussed with a narrative review.
1. Introduction
Odontogenic tumors are a heterogeneous group of benign and malignant lesions characterized by different clinical presentations and histopathological features [1]. In pediatric patients, they account for less than 10% of oral pathologies [2]. Among these tumors, odontomas are described as common, hamartomatous odontogenic lesions, associated with the interference of permanent and deciduous teeth eruption or tooth germ replacement at the site of development [3]. Although they are usually intraosseous, peripheral developments in soft tissues are also described in the literature and referred to as “peripheral odontoma” (PO), “gingival odontoma”, “gingival peripheral odontoma”, “extraosseous odontoma”, and “soft tissue odontoma” [3,4]. Due to its origin, nature, and histopathology, Ide et al. described the lesion as “peripheral developing odontoma” [5,6]. In fact, developing odontomas are classified as a subgroup lacking the complete differentiation pattern of odontomas but appearing more differentiated than ameloblastic fibro-odontoma, and are also rarer and mainly occurring in children [7,8]. As a consequence, differential diagnosis between developing odontomas and ameloblastic fibro-odontoma is hardly challenging [8,9].
In the literature, odontomas are clinically divided into three categories, namely intraosseous, peripheral, or extraosseous and erupted [10]. Central odontoma is the most frequently described, followed by erupted odontoma, while PO is extremely rare [10]. Peripheral or extraosseous manifestations usually appear as gingival or mucosal soft tissue swellings and are histologically similar to their intraosseous counterpart [11]. Although there is no consensus about their origin, it is mostly reported that peripheral odontoma originates specifically from the remnants of the dental lamina (rests of Serres) situated in the gingiva; also, another suggestion is that tooth-like structures arise from soft tissue remnants of the odontogenic epithelium with epithelial–mesenchymal interactions [9]. In the etiology of odontoma, some authors have described the possible association with local trauma in the primary dentition, as well as inflammatory and/or infectious processes, odontoblastic hyperactivity, and genetic alterations associated or otherwise with hereditary conditions (e.g., Gardner syndrome, Hermanns syndrome) [12].
The classic clinical appearance includes gingiva or mucosal swelling, slow growing and asymptomatic, and without bone involvement based on radiographs [13,14,15].
The surgical excision (with periosteum debridement) is generally resolutive for PO, while recurrences have been reported as mainly related to incomplete removal of the lesion due to the impossibility or difficulty to eliminate local irritative agents during surgery. Moreover, conventional surgical treatment with a scalpel is frequently characterized by intraoperative bleeding (also frequently related to gingival inflammation and poor dental hygiene), post-operative bleeding (related to frequent impossibility of suture), post-operative edema, and discomfort. PO management by laser has been reported as an alternative and resolutive surgical method. Generally, lasers such as diode, KTP, Neodymium, and YAG have proven surgical advantages for oral surgery procedures because they are associated with rarer intra-operative complications, low to absent intra-operative bleeding, faster mucosal healing, reduced post-operative pain and edema, and higher patient acceptance [16].
We report the case of a peripheral developing odontoma involving the palatal mucosa in a child, removed by diode laser. In this study, we emphasize the benefits of laser excision as a viable surgical alternative in PO management, particularly in patients with special needs, reviewing the last three decades of international literature on such a topic, as regards the diagnosis, treatment, and epidemiology data.
2. Case Presentation
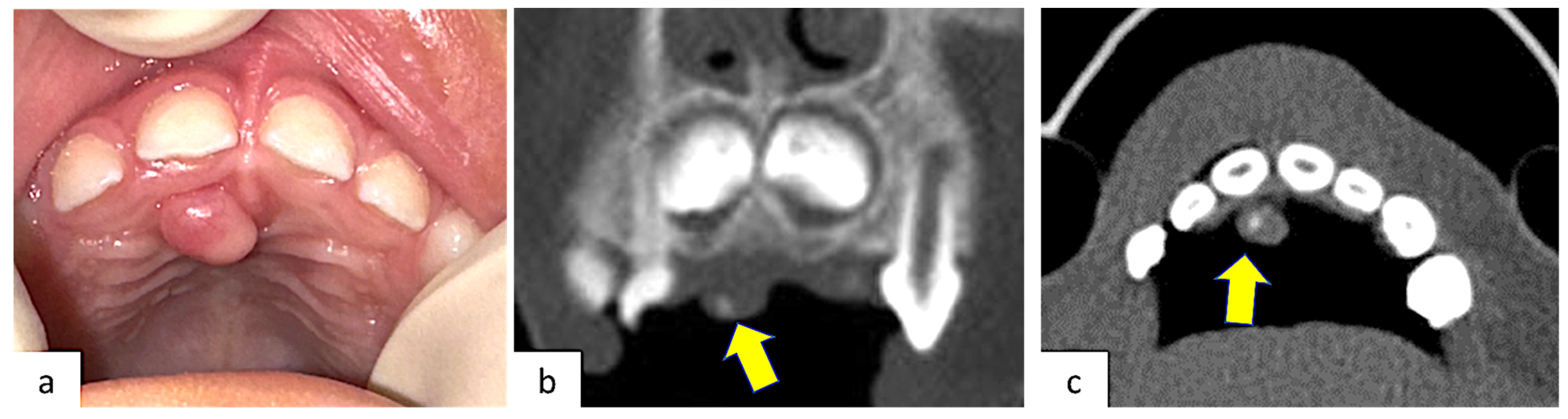
A 4-year-old healthy male patient was referred to the Complex Unit of Odontostomatology of the University Hospital “Policlinico of Bari”, Italy, for a palatal swelling in the upper retro-incisive region. Their parents reported no relevant medical history. The lesion occurred two months earlier with a slow and progressive growth that caused discomfort and difficulties in speech and feeding, but without pain or bleeding. At the intraoral examination, a solitary nodular swelling on the palate, measuring approximately 0.5 × 0.5 cm in size and covered by normally colored mucosa, was observable (Figure 1a). A pre-operative maxillary CT, which required general anesthesia, was performed, showing an intra-lesional calcification. Bone involvement was absent (Figure 1b,c).
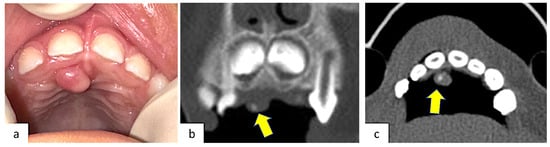
Figure 1.
(a) Clinical presentation of POF occurring in the retro-incisal palatal mucosa, covered by normally colored mucosa without ulceration; (b) coronal CT scan; (c) axial CT scan. Both CT scans showed a small intra-lesion calcification.
3. Results
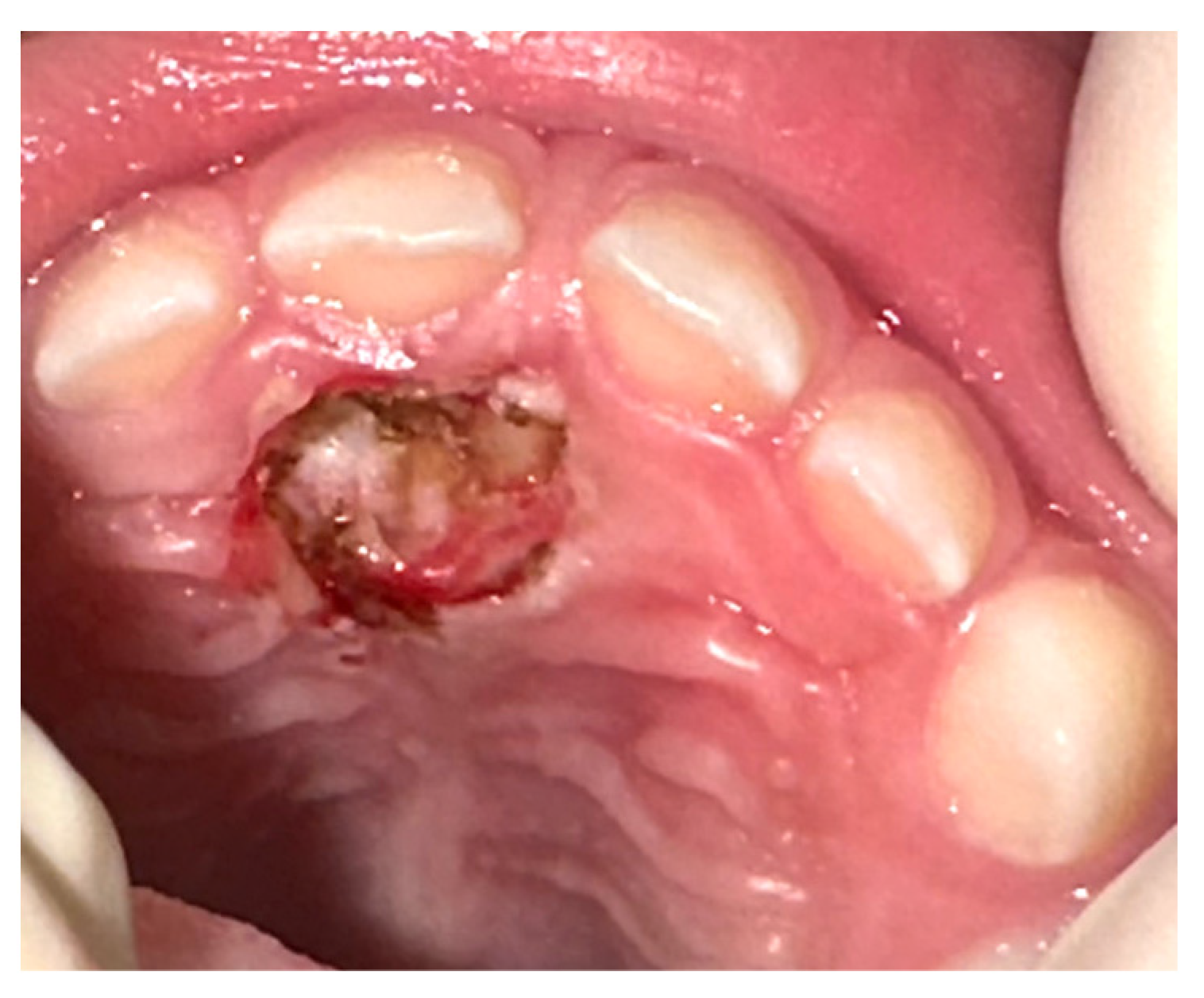
On the basis of the clinical and radiological features, and with the parents’ agreement, it was decided that a diode laser surgical excision, under conscious sedation, should be performed. After a minimal local anesthesia, the excision was performed by diode laser (Lasotronix-Piaseczno, Poland-, 910 nm, 2 W, CW, 300 micron fibre), the marginal gingiva of the 5.1 was preserved, and the lesion was removed with extensive lateral and deep margins, without intra-operative bleeding. A suture was not necessary (Figure 2).
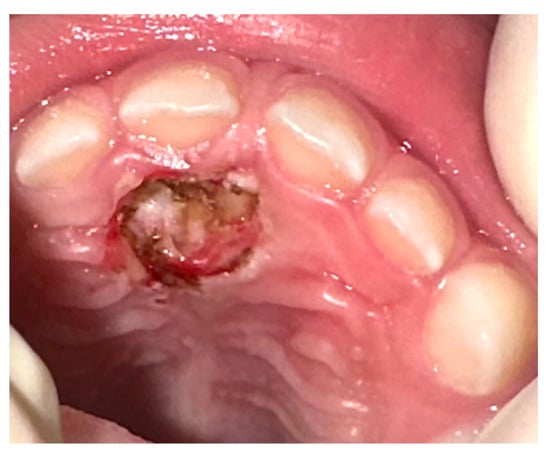
Figure 2.
Immediate post-operative appearance after diode laser removal. Surgical excision showed no bleeding or need for suture.
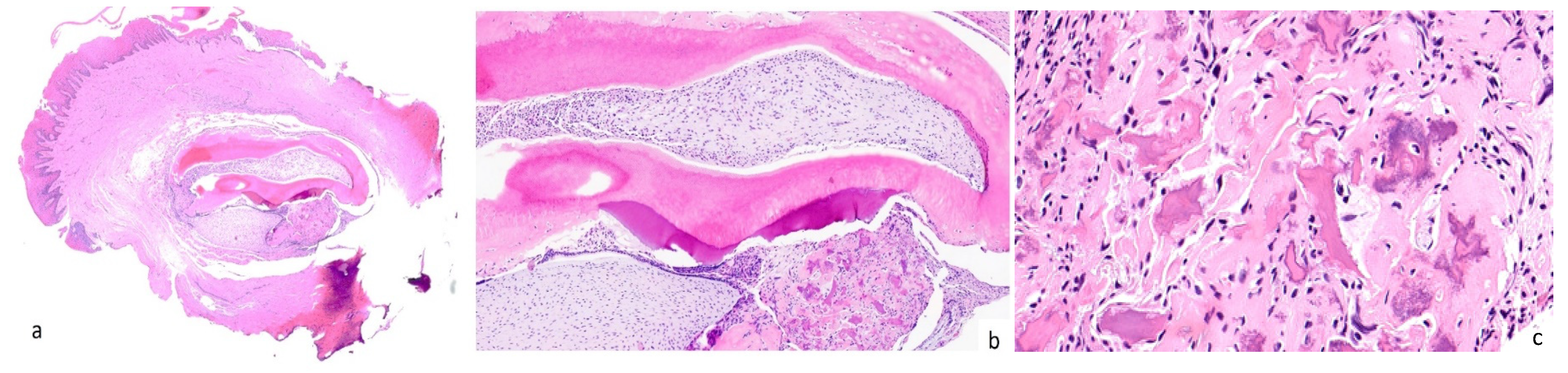
Paracetamol (500 mg twice a day) was suggested for pain management during the 3–4 days following surgery. The specimen was fixed in 10% formalin and sent for histological examination at the Unit of Pathological Anatomy. Upon Hematoxylin and Eosin staining, the lesion was found to be composed of a peripheral rim of fibroblasts, immersed in a dense fibrous connective tissue with many collagen fibres. In the central part, tubular dentin, dental follicle, dental papilla, cementum-like calcifications, and ameloblast-like cells were observable. On the basis of typical histopathological features, the final diagnosis was peripheral developing odontoma. (Figure 3).
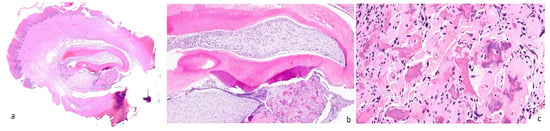
Figure 3.
A well-demarcated mucosal lesion characterized by an overlying reactive hyperkeratotic epithelium and a fibroblastic proliferation in a collagenous background (a). Centrally, the lesion presents multiple components such as the tubular dentin, dental follicle, dental papilla, cementum-like calcifications, and ameloblast-like cells (b). Magnification (400×) of the cementum-like calcifications (c).
No adjunctive immunohistochemical investigation was necessary to confirm the diagnosis. No artifacts or alterations related to the diode laser were reported by the pathologist, confirming the absence of morpho-structural or cytological changes induced by the thermal effect as a result of tissue–laser light interaction.
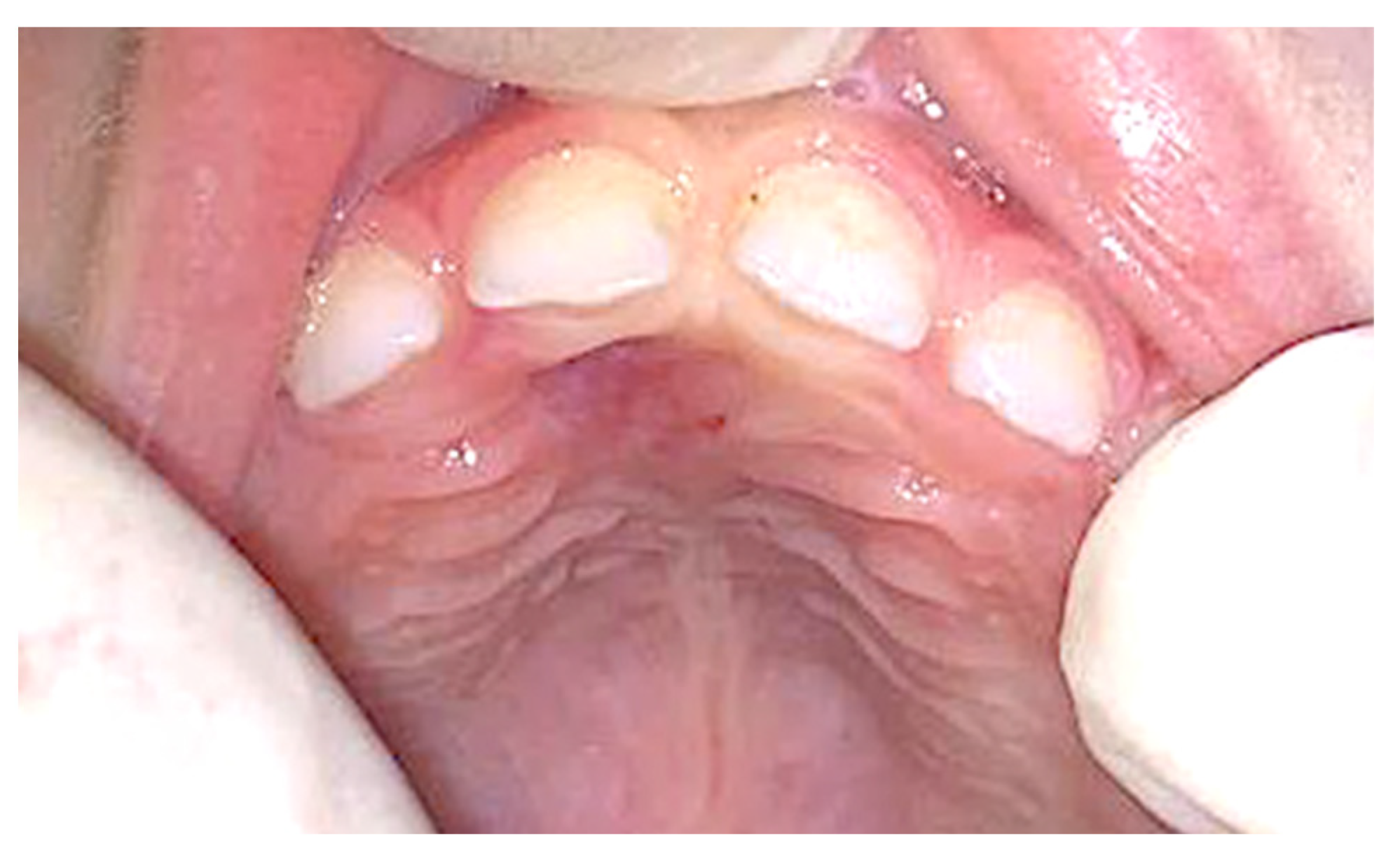
No post-operative complication was observed, and no recurrence was detected after 8 months of follow-up (Figure 4).
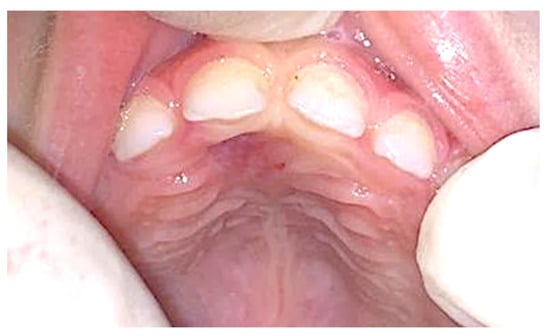
Figure 4.
Clinical appearance 8 months later, showing no sign of recurrence.
4. Data Collection from the Literature
In order to compare the management, we proposed, in the current case of peripheral developing odontoma occurring in a 4-year-old child, to use the guidelines available in the recent literature. We searched in Google Scholar, PubMed, Scopus, and SciELO, using the following keywords: “peripheral odontoma” AND “pediatric” OR “infant” OR “adult”. Only case reports or case series of specific peripheral odontomas from 1990 to 2024 were included, which provided the case description with age, gender, localization, clinical diagnosis, treatment, histopathological diagnosis, and eventual recurrence. A synoptic table (Table 1) was set up with all the included studies, reporting every described aspect.

Table 1.
Data from the relevant literature of the last 34 years about PO in pediatric and adult patients.
5. Discussion
Herein, after having reported the rare occurrence of peripheral developing odontoma involving the palatal mucosa in a pediatric patient, as well as its management from diagnosis to follow-up, we compare our case with data emerging from the pertinent literature.
6. Epidemiology
Odontogenic lesions are usually divided into peripheral and central, according to their primary localization. The relative frequency of peripheral odontogenic tumors is rare and there is lack of consensus of their frequency in the literature [1]. Saghravanian et al. reported that only 4.3% of odontogenic tumors were diagnosed as peripheral. Peripheral odontogenic fibroma was the most common peripheral odontogenic tumor followed by peripheral ameloblastoma [27,28]. Due to important differences in the sample size and the infrequency, there are some discrepancies in epidemiologic data, gender, age, and localization about peripheral odontogenic tumors [28].
Clinically, odontomas are reported to be central (intraosseous), peripheral (extraosseous), or erupted, with the central being the most frequent, followed by the erupted, while the erupted PO is extremely rare [29].
In Table 1, 21 PO cases have been listed as described in the literature. Summarized data show that PO is mostly diagnosed in males (52.4%), and mostly in pediatric patients with 14 from the age of 1 to 18 (70%), 3 from 18 to 39 years (15%), and 3 in newborns (<1 year, 15%). The maxilla was the most frequent location (80.9%), with a higher prevalence in the palate region, followed by the gingiva. Data on age, localization, clinical presentation, and size of the lesion are confirmed by the findings of the current case.
7. Etiopathological Theories
The etiopathogenesis of peripheral odontoma is not yet well described and is still very complex. It is believed that PO development and manifestation, as well as other peripheral odontogenic tumors, are associated with the inclusion of the epithelial dental lamina remains (rests of Serres) located in the gingiva that lose the ability to interact with the mesenchyme.
However, there is no consensus about the stimuli that could activate the epithelial remnants and initiate the proliferation for PO. Trauma, infection, hereditary influence, and genetic alterations associated or otherwise with hereditary conditions (Gardner syndrome and Hermanns syndrome) are also possible triggers [11,12,13,14,15,16,17,18,19,20,21,22,23,24,25].
Various morphologic types in PO might be seen depending on the developmental stage of the tooth germ, which are described as mixed odontogenic lesions without dental hard tissue, developing odontoma, denticles, and hard tissue masses without enamel organ. Another possible theory is that PO arises from the basal layers of the surface epithelium or ectopic neural crest cells with the ability to develop tooth germ [26].
8. Clinical–Radiological Features and Differential Diagnosis
In most of the reported cases, the lesions were clinically described as asymptomatic swelling, with firm consistency, normally colored overlaying mucosa, and slow growth (limited to 0.25–1.5 cm) [3,4,5,7,10,11]. In our case, the patient complained about a slow and progressive growth that caused discomfort and difficulties in speech and feeding, but without pain or bleeding, while clinically, a solitary nodular swelling on the palate measuring approximately 0.5 × 0.5 cm in size and covered by a normally colored mucosa was detectable.
Consequently, considering the clinical–pathological features, as a localized slow-growth of keratinized gingival tissue with a nodular aspect, covered by normally colored mucosa and asymptomatic, and which may be ulcerated due to secondary trauma, the differential diagnosis includes a large spectrum of benign gingival lesions involving the teeth (e.g., pyogenic granulomas, peripheral fibro-osseous lesions, peripheral odontogenic tumors and peripheral giant cell granuloma or fibromas, peripheral ossifying fibroma), malignancies (e.g., osseous, salivary glands, epithelial malignancy), and hereditary diseases (e.g., hereditary gingival fibromatosis, plasminogen deficiency, mucopolysaccharidosis II, and Gardner syndrome) [3,4,5,7,10,11,20]). Differential diagnosis is considered very challenging due to the rarity of this lesion and must also include the misplaced tooth germ and supernumerary teeth that do not grow and cause swelling [30,31].
Also in the literature, it is reported to be difficult to differentially diagnose ameloblastic fibro-odontoma from developing complex odontoma. Both belong to mixed odontogenic tumors group and are characterized by the odontogenic epithelium and ectomesenchyme. Some authors considered the two lesions to be part of a range in children [26]. However, the ameloblastic fibro-odontoma is a non-neoplastic type of ameloblastic fibroma which consists of odontogenic epithelium and odontogenic ectomesenchyme, with mature dentin and enamel. Developing odontoma is almost entirely characterized by dental hard tissues, although it depends on the developmental stage of tooth germ [26,27,28,29,30,31,32,33].
Radiological images could provide the first hint; intralesional calcifications without findings of osteolysis could guide the surgeon and pathologist toward a benign lesion such as a fibro-osseous or odontogenic tumor [30]. The most prescribed first-level exam, orthopantomography, is generally not helpful in showing and defining little calcifications because of the possible overlapping with hard tissues (teeth or bone). A periapical radiograph may occasionally be adequate. The second-level exams, CT and CT–Cone Beam, undoubtedly represent the radiological exams more fit for diagnosis because they allow us to study each aspect with the possibility of switching to different axes of visualization to identify the proper dimension, the possible involvement of adjacent anatomical structures and the small spots of calcification, due to high sensitivity [29]. Also, MRI could be helpful, but it is considered a more expensive and invasive exam with a lower definition than CT [31].
Clinical, histopathologic, and radiographic images are fundamental features for clinicians to be familiar with in order to prevent incorrect diagnosis.
As for the case reported herein, an adjunctive challenge was the low grade of compliance due to the age of the patient, which made the general anesthesia to perform the CT at the time of pre-operative investigations mandatory. As such, the radiograph demonstrated the real extent of the lesion, the absence of bone involvement, and a little intralesional calcification, resulting in a pre-operative diagnosis of a benign lesion.
9. Peripheral Developing Odontoma in Pediatric and Adult Patients: Analysis of the Relevant Literature
Considering the rarity of peripheral odontogenic lesions, especially regarding peripheral developing odontomas, we decided to carry out a review of the relevant literature on PO, and the results are presented as follows.
Friedrich et al. described an interesting case of peripheral developing odontoma of the maxilla in a 3-year-old male patient. The patient clinically showed a small painless nodular neoformation on the right palatal region. At the radiological investigations, an alteration of the dentition without a delineated lesion in the interested region was observable. Surgical treatment consisted of a conventional surgical excision, hemostasis, and suture, without recurrence at a 3 year follow-up [2]. Soluk-Tekkeşin et al. described a rare case of the right palatal incisors in a 12-year-old girl presenting a non-ulcerated painless swelling with radiological signs of radiopaque masses that were entirely located in the soft tissues and with no bone relation. The tumor was completely removed, without recurrence at a 10 month follow-up, by scalpel excision and diagnosed as “Peripheral Compound Odontoma” [3]. Mikami et al. described a rare case of congenital PO in a 9-month-old male infant, presenting as two asymptomatic swellings in the anterior palate, covered by a normal mucosa and present since birth. Both masses were surgically removed at the age of 28 months with the patient under general anesthesia. The diagnosis was “peripheral developing odontoma accompanied by teratomatous fibroma”. No recurrences were observed at a 10 year follow-up [22]. Silva et al. reported two cases of peripheral developing odontoma in a newborn (8-month-old male patient) with an asymptomatic, slowly growing congenital nodule on the alveolar mucosa of the palate, and a 5-month-old male with a nodule of the buccal aspect of the incisive papilla, both without bone involvement and treated with surgical excision and no sign of recurrence [13]. In addition, de Oliveira et al. reported a rare case of erupted PO in a 30-year-old male patient, localized on the buccal side of the alveolar ridge and erupted almost 18 months before the clinical diagnosis and described as asymptomatic calcified mass. Radiographic images showed multiple radiopaque masses and an incisional biopsy was performed due to the patient’s refusal to completely remove the lesion [25]. Moghadam et al. described a case of a peripheral developing odontoma in a healthy 8-year-old girl, presenting as an isolated and ulcerated soft tissue mass of the palatal gingiva in the region of the right canine and deciduous first molar. On the periapical radiograph, no osseous involvement was detected, and the mass was excised under local anesthesia, along with a clinical diagnosis of reactive soft tissue lesion (pyogenic granuloma or peripheral ossifying fibroma). The histopathological examination diagnosed the lesion as peripheral developing complex odontoma or peripheral ameloblastic fibro-odontoma. In this case, the authors also described the importance for oral pathologists and clinicians to be aware of the challenging differential diagnosis of these lesions and be familiar with the clinical and histopathological features of peripheral odontoma [26]. Furthermore, da Silva et al. also reported a case of a peripheral compound odontoma in an 11-year-old female patient with an asymptomatic exophytic, sessile, nodular lesion in the anterior region of the palate, characterized by no alterations at the radiographic examination. An excisional biopsy was performed, and no recurrences observed at a 6 month follow-up. The authors affirmed that the treatment of choice for PO is local excision and that, at the time of their study, no recurrence cases have been reported after complete lesion removal [4]. Kintarak et al. in their study presented a 13-year-old female with a 5 month history of a slow growing, asymptomatic nodular mass on her palatal interdental papilla between the maxillary right central and lateral incisors. A surgical excision was performed and no recurrences observable at an 18 month follow-up [19].
All data mentioned are summarized in Table 1.
Considering the most relevant literature on this topic, we found 20 case reports in the last 34 years, confirming a relatively rare occurrence in patients or a low publication rate; 14 were from the age of 1 to 18 (70%), 3 from 18 to 39 years (15%), and 3 in newborn (<1 year, 15%). Of particular interest are data about sex distribution, including 11 cases (55%) which occurred in males and 10 (45%) in females, confirming the male predilection of PO. Besides the age range, all cases show common histopathologic features such as para-keratinized epithelium with a chronic inflammatory infiltrate, which delimitates a spindle fibroblast stroma, and calcification spots without areas of cellular atypia. Considering that PO occurrence is very rare, the sample of this narrative review is too small to carry out any statistical analysis, but several considerations might still be deduced. First of all, the distribution of sex and localization is consistent with the previous studies, with a slight predominance in males [13,14,15,16]. In more than half of the cases, PO was localized in the maxilla (85%), then the mandible (15%), especially in the lingual posterior region. It is relevant to highlight that the cases presented in the mandibula did not allow clinicians to perform a clinical diagnosis; however, such an association is persistent but not statistically demonstrated [5,6,7,8,9,10,11,12,13,14,15,16,17]. The case we presented was in accord with the literature, as the young patients presented a PO localization in the palatal region of the maxilla. In fact, as reported by Soluk-Tekkeşin et al. [3], the palatal occurrence further expands the differential diagnosis including reactive lesions as well neoplastic lesions both odontogenic and salivary. As often happens in such cases, in our patient the challenge was to predict the lesion’s nature without a pre-operative biopsy. In this, we were helped only by a CT scan that, although performed under general anesthesia, showed no cortical bone erosion and no tooth displacement, leading the doctor to exclude malignancy. In addition, CT was very useful in evaluating a little calcification spot in the lesion center, directing us toward an ossifying/cementifying/odontogenic lesion.
As found in literature, the pre-operative assessment of similar lesions, especially in children, lacks consensus. In the studies considered in this review, the most used radiograph was the periapical radiograph followed by OPT. Both are first-level exams that must be executed if the patient is compliant, they are very useful for central lesions (lesions with exclusive bony involvement) but useless for peripheral lesions because of the overlapping of healthy hard tissues such as teeth and bone [33]. The radiological exam suggestable for this type of lesion is the CT or CT Cone Beam [34].
The operative regime must be deepened. In the reported cases, the authors did not specify the operative anesthesiologic regime used for the surgical biopsy. Local anaesthesia is the safest and most practiced regimen but requires the patient’s complete compliance. It is interesting to report that all newborns were treated in general anesthesia because of the lack of patient collaboration. In our case, thanks to the anesthesiologist, the patient was treated under conscious sedation and local anesthesia, avoiding an intra-operative general anesthesia. The advantages of conscious sedation in dental procedures are well established in the literature [35], so it should be preferred when compliance is reduced and general anesthesia is not suggested, especially in pediatric patients.
10. Therapeutic Strategies
As regards the therapy, data from the literature confirm that surgery represents the eligible treatment for POs. Surgical removal should allow for complete excision, often involving the periosteum. The removal of local irritative factors is mandatory; hence, surgery is frequently associated with adjacent teeth scaling and root planning when involved or substituting over contoured restorations. In addition, surgery is usually performed without a preliminary incisional biopsy, especially in small lesions, and as a consequence, without a pre-operative diagnosis. The possibility to obtain a pre-operative diagnosis is hugely relevant in order to more or less plan the invasive surgical procedure; therefore, in such cases, clinicians face the challenge of being resolutive in removing a lesion only clinically diagnosed as PO, while considering a wide panel of possible differential diagnosis and, at the same time, trying to be as conservative as possible. Resective surgery is generally not indicated for such lesions. The most reported and performed surgery is by scalpel, followed by bone debridement, tooth scaling, and root planning, with an approach that is as conservative as possible [17,18,19,20,21,22,23,24,25,26]. Of the 18 studies conducted on pediatric and adult patients that we considered in this review, almost all the authors have preferred this conventional treatment strategy because of its well-established reliability [17,18,19,20,21,22,23,24,25,26]. At our acknowledgement, this is the first reported study of a PO surgically treated using a diode laser.
An alternative surgical approach is represented by the diode laser thanks to the possibility of reduced comorbidity, with no suture needed, a reduced rate of superinfections, and a reduced drug intake for post-operative pain management [36]. Diode laser use in dentistry and oral surgery is widely supported by the literature, mainly for soft tissue surgery (both for benign and malignant lesions) [37,38], allowing for fast and non-bleeding lesion removal with (usually) secondary intention healing of the covering mucosa, as well as the transmucosal or intralesional photocoagulation of vascular malformations (thus avoiding invasive and unpredictable surgical procedures as in the past years) or lesions with high risk of intraoperative bleeding before the surgical removal [39]. It is relevant to underline that the use of diode laser was debated due to the risk of thermal alterations at the histologic examination, but it is fully demonstrated that the thermal effect is usually very low and does not affect the histological diagnosis [40]. Therefore, diode laser excision is highly suggestable for patients with reduced compliance, including children, and with lesions similar to those reported herein [41]. In our case, the patient was a 4-year-old child, thus with very low compliance, so the use of diode laser was very useful because the suture was unnecessary, the post-operative course was without complications, and the palatal mucosa fully recovered in 10 days through secondary intention healing. This therapeutical choice is also strictly in accordance with the suggestions of the American Academy of Pediatric Dentistry regarding the policy on the use of lasers from 2013 [42] to the latest version of 2022 [43], precisely recognizing “Lasers as an alternative and complementary method of providing soft and hard tissue dental procedures for infants, children, adolescents, and persons with special health care needs”.
11. Limitations and Future Directions
Some limitations are present in this narrative review including the prominent lack of data in the literature regarding PO in patients as it accounts for only a small number of reported or published cases of odontogenic lesions. It did not allow us to perform any statistical analysis nor draw definitive conclusions. The lack of articles differing from case reports or short case series is due to the rarity of this odontogenic lesion. Our intention was to report on the diagnostic–therapeutic pathway that we followed with good outcomes, comparing it with the past literature, and finally suggesting the possible best option in such a delicate situation. Based on the case description, and supported by the latest literature, the authors would further encourage the use of diode laser to induce rapid and complete healing with reduced comorbidities, especially in pediatric patients. Moreover, this approach is also supported by the use of pre-operative conscious sedation, providing the opportunity to carry out the appropriate surgical procedure, which is often complicated by the proximity of teeth and/or periodontal tissues. Lastly, the use of diode laser for such interventions, when performed by an expert surgeon with the most adequate laser setting, is helpful in collecting a surgical specimen for the subsequent histological examination, without any morphological alterations limiting the final diagnosis.
Author Contributions
Conceptualization, S.C., M.F. and G.B.; methodology, G.F., A.M. and A.D.A.; investigation, S.C., G.F., M.C. and G.I.; writing—original draft preparation, M.F. and S.C.; writing—review and editing, S.C. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Institutional Review Board Statement
Internal Ethical Committee; study number 7390-protocol number 0069684-04/08/2022.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Vered, M.; Wright, J.M. Update from the 5th Edition of the World Health Organization Classification of Head and Neck Tumors: Odontogenic and Maxillofacial Bone Tumours. Head Neck Pathol. 2022, 16, 63–75. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Friedrich, R.E.; Fuhrmann, A.; Scheuer, H.A.; Zustin, J. Small peripheral developing odontoma of the maxilla in a 3-year-old patient depicted on cone-beam tomograms. In Vivo 2010, 24, 895–898. [Google Scholar] [PubMed]
- Soluk-Tekkeşin, M.; Balkan, B.; Akatay, D.K.; Atalay, B. A Rare Case of Peripheral Compound Odontoma and Review of the Literature. Head Neck Pathol. 2022, 16, 913–917. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- da Silva Rocha, O.K.M.; da Silva Barros, C.C.; da Silva, L.A.B.; de Souza Júnior, E.F.; de Morais, H.H.A.; da Costa Miguel, M.C. Peripheral compound odontoma: A rare case report and literature review. J. Cutan. Pathol. 2020, 47, 720–724. [Google Scholar] [CrossRef] [PubMed]
- Ide, F.; Mishima, K.; Saito, I.; Kusama, K. Rare peripheral odontogenic tumors: Report of 5 cases and comprehensive review of the literature. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2008, 106, e22–e28. [Google Scholar] [CrossRef] [PubMed]
- Ide, F.; Shimoyama, T.; Horie, N. Gingival peripheral odontoma in an adult: Case report. J. Periodontol. 2000, 71, 830–832. [Google Scholar] [CrossRef] [PubMed]
- Jones, A.V.; Franklin, C.D. An analysis of oral and maxillofacial pathology found in children over a 30-year period. Int. J. Paediatr. Dent. 2006, 16, 19–30. [Google Scholar] [CrossRef] [PubMed]
- Gardner, D.G. The mixed odontogenic tumors. Oral Surg. Oral Med. Oral Pathol. 1984, 58, 166–168. [Google Scholar] [CrossRef] [PubMed]
- Bilodeau, E.A.; Collins, B.M. Odontogenic Cysts and Neoplasms. Surg. Pathol. Clin. 2017, 10, 177–222. [Google Scholar] [CrossRef] [PubMed]
- Soluk Tekkesin, M.; Pehlivan, S.; Olgac, V.; Aksakallı, N.; Alatli, C. Clinical and histopathological investigation of odontomas: Review of the literature and presentation of 160 cases. J. Oral Maxillofac. Surg. 2012, 70, 1358–1361. [Google Scholar] [CrossRef] [PubMed]
- Manor, Y.; Mardinger, O.; Katz, J.; Taicher, S.; Hirshberg, A. Peripheral odontogenic tumours--differential diagnosis in gingival lesions. Int. J. Oral Maxillofac. Surg. 2004, 33, 268–273. [Google Scholar] [CrossRef] [PubMed]
- Satish, V.; Prabhadevi, M.C.; Sharma, R. Odontome: A Brief Overview. Int. J. Clin. Pediatr. Dent. 2011, 4, 177–185. [Google Scholar] [CrossRef] [PubMed]
- Silva, A.R.S.; Carlos-Bregni, R.; Vargas, P.A.; de Almeida, O.P.; Lopes, M.A. Peripheral developing odontoma in newborn. Report of two cases and literature review. Med. Oral Patol. Oral Cir. Bucal. 2009, 14, e612–e615. [Google Scholar] [CrossRef] [PubMed]
- Hanemann, J.A.; Oliveira, D.T.; Garcia, N.G.; Santos, M.R.; Pereira, A.A. Peripheral compound odontoma erupting in the gingiva. Head Face Med. 2013, 9, 15. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Giunta, J.L.; Kaplan, M.A. Peripheral, soft tissue odontomas. Two case reports. Oral Surg. Oral Med. Oral Pathol. 1990, 69, 406–411. [Google Scholar] [CrossRef] [PubMed]
- Anuradha, B.R.; Penumarty, S.; Charan, C.R.; Swati, M. Application of 810-Nm Diode Laser in the Management of Peripheral Ossifying Fibroma. J. Indian Soc. Periodontol. 2015, 19, 224–226. [Google Scholar] [CrossRef] [PubMed]
- Castro, G.W.; Houston, G.; Weyrauch, C. Peripheral odontoma: Report of case and review of literature. ASDC J. Dent. Child. 1994, 61, 209–213. [Google Scholar] [PubMed]
- Ledesma-Montes, C.; Perez-Bache, A.; Garcés-Ortíz, M. Gingival compound odontoma. Int. J. Oral Maxillofac. Surg. 1996, 25, 296–297. [Google Scholar] [CrossRef] [PubMed]
- Kintarak, S.; Kumplanont, P.; Kietthubthew, S.; Chungpanich, S. A nodular mass of the anterior palatal gingiva. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. Endod. 2006, 102, 3–6. [Google Scholar] [CrossRef] [PubMed]
- Ilief-Ala, M.A.; Eisenberg, E.; Mathieu, G. Peripheral complex odontoma in a pediatric dental patient: A case report. J. Mass Dent. Soc. 2008, 56, 24–26. [Google Scholar] [PubMed]
- de Fátima Bernardes, V.; de Miranda Cota, L.O.; de Oliveira Costa, F.; Mesquita, R.A.; Gomez, R.S.; Aguiar, M.C.F. Gingival peripheral odontoma in a child: Case report of an uncommon lesion. Braz. J. Oral Sci. 2008, 7, 1624–1626. [Google Scholar] [CrossRef]
- Mikami, T.; Yagi, M.; Mizuki, H.; Takeda, Y. Congenital peripheral developing odontoma accompanied by congenital teratomatous fibroma in a 9-month-old boy: A case report. J. Oral Sci. 2013, 55, 89–91. [Google Scholar] [CrossRef] [PubMed]
- Koneru, A.; Surekha, R.; Vanishree, M.; Hamsini, A.; Hunasgi, S. Rare gingival odontoma: Report of a case and review of literature. J. Dr. NTR Univ. Health Sci. 2014, 3, 133–135. [Google Scholar] [CrossRef]
- Custódio, M.; Araujo, J.P.; Gallo, C.B.; Trierveiler, M. Gingival complex odontoma: A rare case report with a review of the literature. Autops. Case Rep. 2018, 8, e2018009. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- de Oliveira, M.A.; Reis, B.; Pallos, D.; Kim, Y.J.; Braz-Silva, P.H.; Martins, F. The Importance of Histopathological Examination to the Final Diagnosis of Peripheral Odontogenic Tumors: A Case Report of a Peripheral Odontoma. Case Rep. Dent. 2019, 2019, 9712816. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Atarbashi Moghadam, S.; Mokhtari, S. Peripheral Developing Odontoma or Peripheral Ameloblastic Fibroodontoma: A Rare Challenging Case. Case Rep Dent. 2016, 2016, 9379017. [Google Scholar] [CrossRef] [PubMed] [PubMed Central]
- Saghravanian, N.; Jafarzadeh, H.; Bashardoost, N.; Pahlavan, N.; Shirinbak, I. Odontogenic tumors in an Iranian population: A 30-year evaluation. J. Oral Sci. 2010, 52, 391–396. [Google Scholar] [CrossRef] [PubMed]
- Buchner, A.; Merrell, P.W.; Carpenter, W.M. Relative frequency of peripheral odontogenic tumors: A study of 45 new cases and comparison with studies from the literature. J. Oral Pathol. Med. 2006, 35, 385–391. [Google Scholar] [CrossRef] [PubMed]
- Barba, L.T.; Campos, D.M.; Rascón, M.M.N.; Barrera, V.A.R.; Rascón, A.N. Descriptive aspects of odontoma: Literature review. Rev. Odont. Mex. 2016, 20, e265–e269. [Google Scholar] [CrossRef]
- Mortazavi, H.; Safi, Y.; Baharvand, M.; Rahmani, S.; Jafari, S. Peripheral Exophytic Oral Lesions: A Clinical Decision Tree. Int. J. Dent. 2017, 2017, 9193831. [Google Scholar] [CrossRef] [PubMed]
- Yu, J.L.; Kapur, R.P.; Susarla, S.M. Recurrent Gingival Lesions in a Pediatric Patient. Plast. Reconstr. Surg. Glob. Open 2022, 10, e4382. [Google Scholar] [CrossRef] [PubMed]
- Philipsen, H.P.; Reichart, P.A.; Prætorius, F. Mixed odontogenic tumours and odontomas; Considerations on interrelationship. Review of the literature and presentation of 134 new cases of odontomas. Eur. J. Cancer B Oral Oncol. 1997, 33, 86–99. [Google Scholar] [CrossRef] [PubMed]
- Crusoé-Rebello, I.; Torres, M.; Burgos, V.; Oliveira, C.; dos Santos, J.; Azevedo, R.; Campos, P. Hybrid Lesion: Central Giant Cell Granuloma and Benign Fibro-Osseous Lesion. Dentomaxillofacial Radiol. 2009, 38, 421–425. [Google Scholar] [CrossRef] [PubMed]
- Missias, E.M.; Nascimento, E.; Pontual, M.; Pontual, A.A.; Freitas, D.Q.; Perez, D.; Ramos-Perez, F. Prevalence of Soft Tissue Calcifications in the Maxillofacial Region Detected by Cone Beam CT. Oral Dis. 2018, 24, 628–637. [Google Scholar] [CrossRef] [PubMed]
- Galeotti, A.; Garret Bernardin, A.; D’Antò, V.; Ferrazzano, G.F.; Gentile, T.; Viarani, V.; Cassabgi, G.; Cantile, T. Inhalation Conscious Sedation with Nitrous Oxide and Oxygen as Alternative to General Anesthesia in Precooperative, Fearful, and Disabled Pediatric Dental Patients: A Large Survey on 688 Working Sessions. Biomed Res. Int. 2016, 2016, 7289310. [Google Scholar] [CrossRef] [PubMed]
- Ortega-Concepción, D.; Cano-Durán, J.A.; Peña-Cardelles, J.-F.; Paredes-Rodríguez, V.-M.; González-Serrano, J.; López-Quiles, J. The Application of Diode Laser in the Treatment of Oral Soft Tissues Lesions. A Literature Review. J. Clin. Exp. Dent. 2017, 9, e925–e928. [Google Scholar] [CrossRef] [PubMed]
- Capodiferro, S.; Loiudice, A.M.; Pilolli, G.; Lajolo, C.; Giuliani, M.; Maiorano, E.; Favia, G. Diode Laser Excision of Chondroid Lipoma of the Tongue with Microscopic (Conventional and Confocal Laser Scanning) Analysis. Photomed. Laser Surg. 2009, 27, 683–687. [Google Scholar] [CrossRef] [PubMed]
- Limongelli, L.; Capodiferro, S.; Tempesta, A.; Sportelli, P.; Dell’Olio, F.; Angelelli, G.; Maiorano, E.; Favia, G. Early Tongue Carcinomas (Clinical Stage I and II): Echo-Guided Three-Dimensional Diode Laser Mini-Invasive Surgery with Evaluation of Histological Prognostic Parameters. A Study of 85 Cases with Prolonged Follow-Up. Lasers Med. Sci. 2020, 35, 751–758. [Google Scholar] [CrossRef] [PubMed]
- Capodiferro, S.; Limongelli, L.; Tempesta, A.; Maiorano, E.; Favia, G. Diode Laser Treatment of Venous Lake of the Lip. Clin. Case Rep. 2018, 6, 1923–1924. [Google Scholar] [CrossRef]
- Tenore, G.; Mohsen, A.; Nuvoli, A.; Palaia, G.; Rocchetti, F.; Di Gioia, C.R.T.; Cicconetti, A.; Romeo, U.; Del Vecchio, A. The Impact of Laser Thermal Effect on Histological Evaluation of Oral Soft Tissue Biopsy: Systematic Review. Dent. J. 2023, 11, 28. [Google Scholar] [CrossRef]
- Tavares, T.S.; Da Costa, A.A.S.; Freire-Maia, F.B.; Souza, L.N.; Zarzar, P.M.; Martins-Júnior, P.A.; Aguiar, M.C.F.; Mesquita, R.A.; Caldeira, P.C. Unusual Exophytic Gingival Lesion in a Newborn Treated with Diode Laser. Oral Surg. Oral Med. Oral Pathol. Oral Radiol. 2020, 130, e74–e79. [Google Scholar] [CrossRef] [PubMed]
- AAPD. Policy on Use of Lasers for Pediatric Dental Patients; AAPD Reference Manual 36; AAPD: Chicago, IL, USA, 2013; pp. 75–77. [Google Scholar]
- American Academy of Pediatric Dentistry. Policy on the Use of Lasers for Pediatric Dental Patients; The Reference Manual of Pediatric Dentistry; American Academy of Pediatric Dentistry: Chicago, IL, USA, 2022; pp. 131–134. [Google Scholar]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).