Exploring the Role of DARPP-32 in Addiction: A Review of the Current Limitations of Addiction Treatment Pathways and the Role of DARPP-32 to Improve Them
Abstract
:1. Introduction
2. Current Pharmacological Treatment
2.1. Pharmacological Treatment of Alcohol Addiction
2.2. Pharmacological Treatment of Opioid Addiction
2.3. Pharmacological Treatment of Nicotine Addiction
2.4. Pharmacological Treatment of Cannabinoid Addiction
2.5. Pharmacological Treatment of Psychostimulant Addiction
2.6. Attempts to Improve Pharmacological Treatment Options
3. The Neurobiology of Addiction
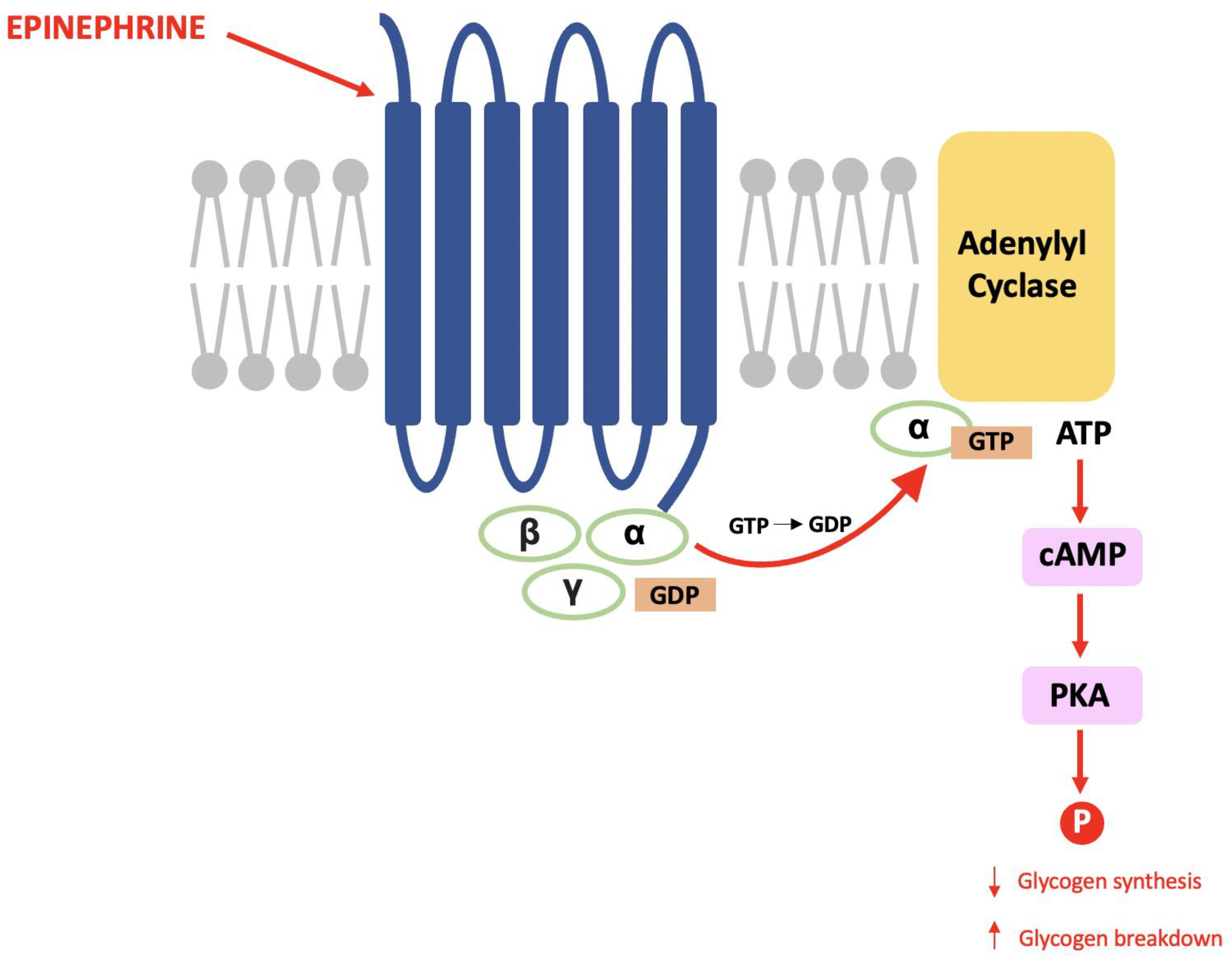
4. Dopamine- and cAMP-Regulated Phosphoprotein, Mr 32 kDa (DARPP-32)
4.1. DARPP-32 Discovery
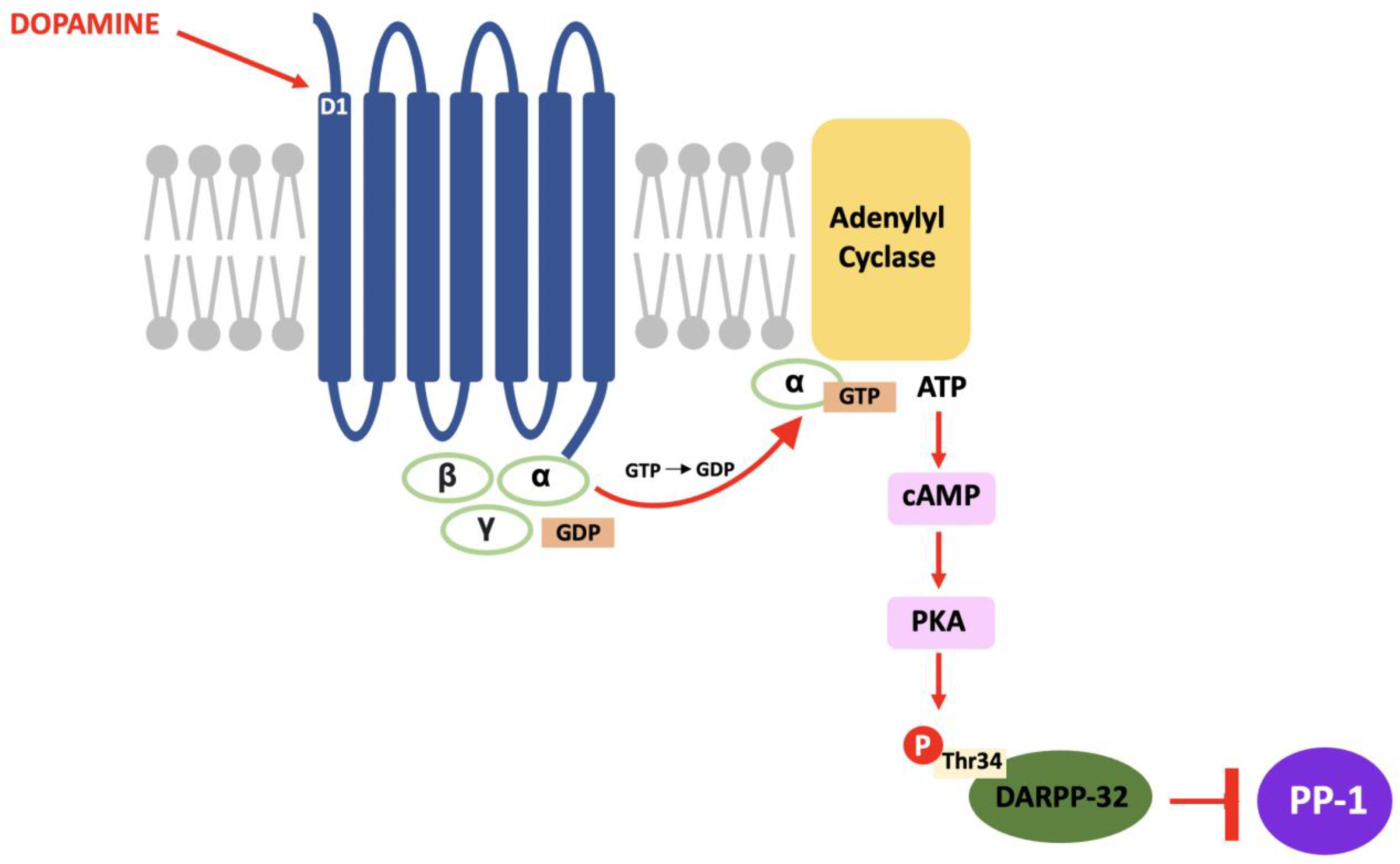
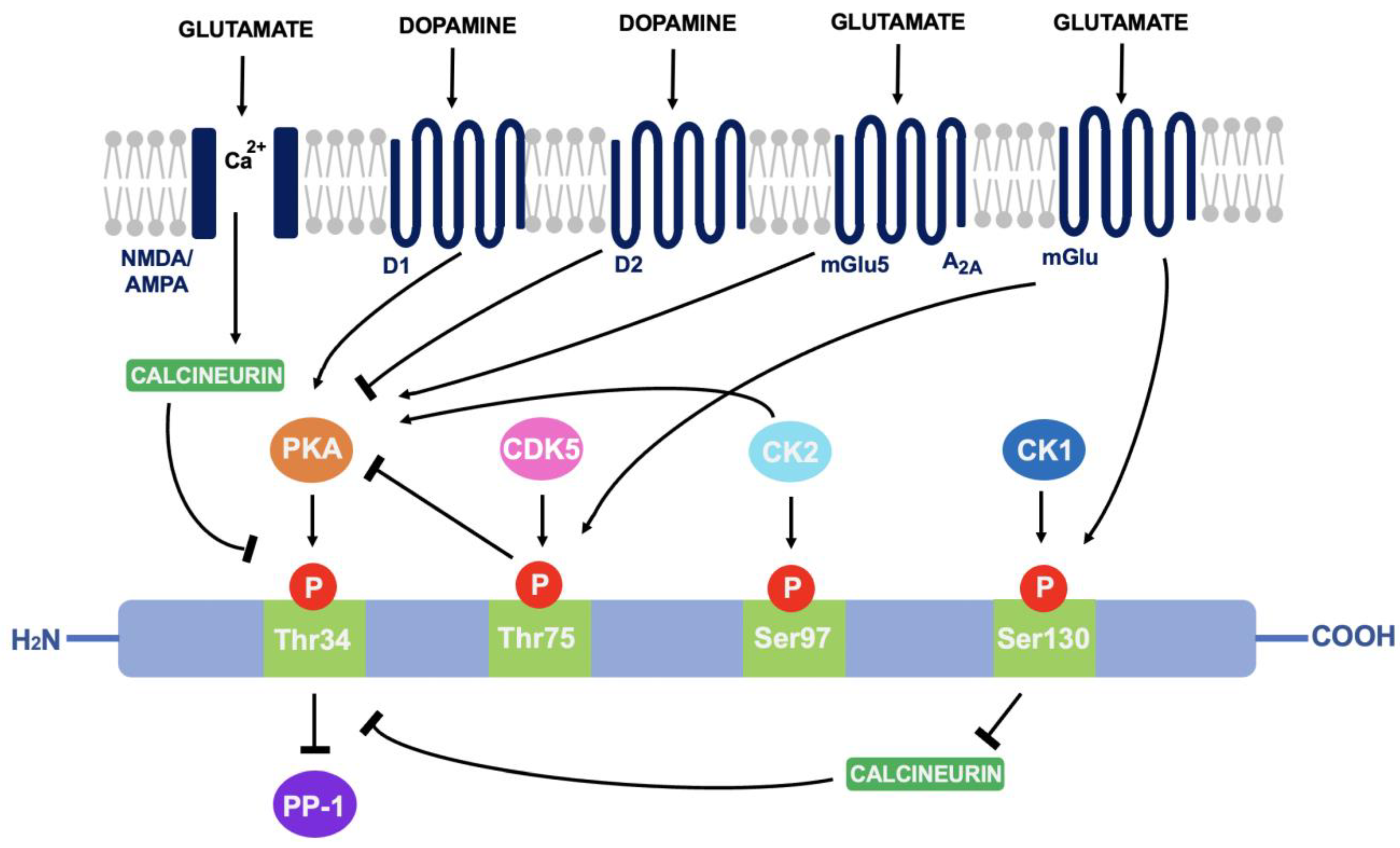
4.2. DARPP-32 Phosphorylation
4.3. DARPP-32 Localisation
4.4. DARPP-32 and Neuroplasticity
4.5. DARPP-32 and Substances of Abuse
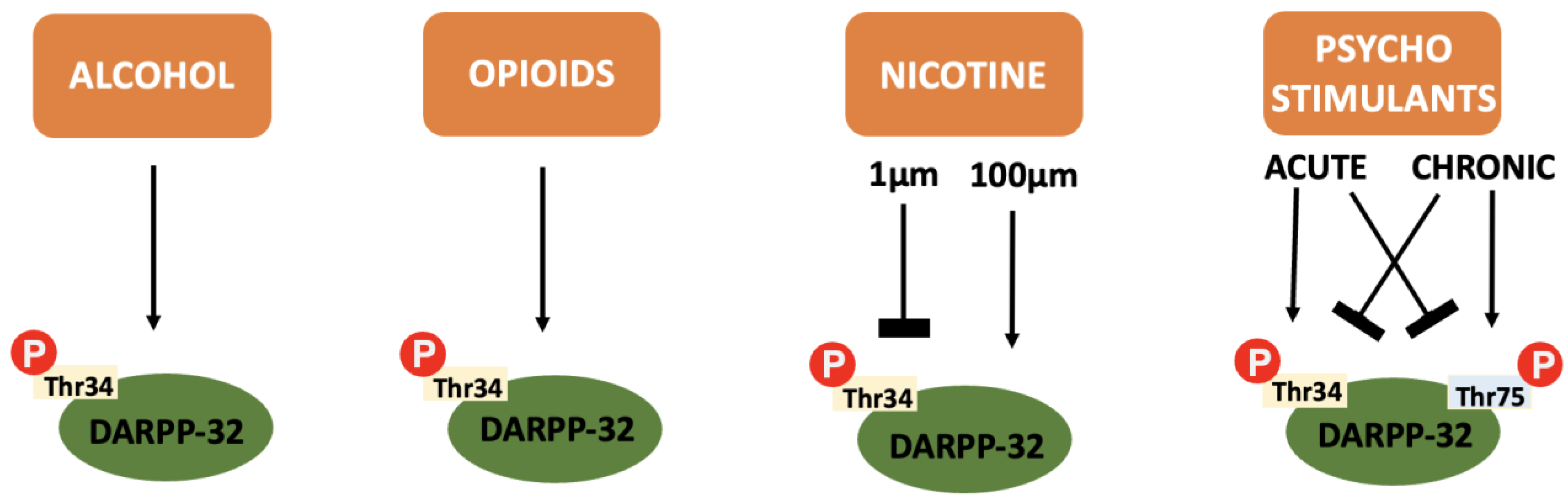
4.5.1. DARPP-32 and Alcohol
4.5.2. DARPP-32 and Opioids
4.5.3. DARPP-32 and Nicotine
4.5.4. DARPP-32 and Cannabinoids
4.5.5. DARPP-32 and Psychostimulants
5. Discussion
6. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- UN Office on Drugs and Crime. World Drug Report. WHO 2021. Available online: https://www.unodc.org/unodc/en/data-and-analysis/wdr2021.html (accessed on 10 December 2021).
- World Health Organization. Global Status Report on Alcohol and Health. WHO 2018. Available online: https://www.who.int/publications/i/item/9789241565639 (accessed on 10 December 2021).
- World Health Organisation. Tobacco Fact Sheet. WHO 2021. Available online: https://www.who.int/news-room/fact-sheets/detail/tobacco (accessed on 10 December 2021).
- Dubey, M.J.; Ghosh, R.; Chatterjee, S.; Biswas, P.; Chatterjee, S.; Dubey, S. COVID-19 and addiction. Diabetes Metab. Syndr. Clin. Res. Rev. 2020, 14, 817–823. [Google Scholar] [CrossRef]
- Chiappini, S.; Guirguis, A.; John, A.; Corkery, J.M.; Schifano, F. COVID-19: The Hidden Impact on Mental Health and Drug Addiction. Front. Psychiatry 2020, 11, 767. [Google Scholar] [CrossRef] [PubMed]
- Richter, L.; Vuolo, L.; Salmassi, M. Stigma and Addiction Treatment. In The Stigma of Addiction; Springer: Cham, Switzerland, 2019. [Google Scholar] [CrossRef]
- Roberts, E.; Hotopf, M.; Strang, J.; Marsden, J.; White, M.; Eastwood, B.; Drummond, C. The hospital admission profile of people presenting to specialist addiction services with problematic use of alcohol or opioids: A national retrospective cohort study in England. Lancet Reg. Health 2021, 3, 100036. [Google Scholar] [CrossRef]
- McGinty, E.E.; Barry, C.L. Stigma Reduction to Combat the Addiction Crisis—Developing an Evidence Base. N. Engl. J. Med. 2020, 382, 1291–1292. [Google Scholar] [CrossRef]
- NIDA. Principles of Effective Treatment. 2020. Available online: https://www.drugabuse.gov/publications/principles-drug-addiction-treatment-research-based-guide-third-edition/principles-effective-treatment (accessed on 11 December 2021).
- Witkiewitz, K.; Litten, R.; Leggio, L. Advances in the science and treatment of alcohol use disorder. Sci. Adv. 2019, 5, eaax4043. [Google Scholar] [CrossRef] [PubMed]
- BNF British National Formulary—NICE. 2021. Available online: https://bnf.nice.org.uk/treatment-summaries/alcohol-dependence/ (accessed on 11 December 2021).
- Jørgensen, C.H.; Pedersen, B.; Tønnesen, H. The Efficacy of Disulfiram for the Treatment of Alcohol Use Disorder. Alcohol. Clin. Exp. Res. 2011, 35, 1749–1758. [Google Scholar] [CrossRef]
- Kalra, G.; De Sousa, A.; Shrivastava, A. Disulfiram in the management of alcohol dependence: A comprehensive clinical review. Open J. Psychiatry 2014, 4, 43–52. [Google Scholar] [CrossRef]
- Littleton, J.; Zieglgänsberger, W. Pharmacological Mechanisms of Naltrexone and Acamprosate in the Prevention of Relapse in Alcohol Dependence. Am. J. Addict. 2003, 12, s3–s11. [Google Scholar] [CrossRef]
- Anton, R.F.; O’Malley, S.S.; Ciraulo, D.; Cisler, R.A.; Couper, D.; Donovan, D.; Gastfriend, D.R.; Hosking, J.D.; Johnson, B.A.; LoCastro, J.S.; et al. Combined Pharmacotherapies and Behavioral Interventions for Alcohol Dependence. JAMA 2006, 295, 2003–2017. [Google Scholar] [CrossRef] [Green Version]
- Brasser, S.M.; McCaul, M.E.; Houtsmuller, E.J. Alcohol effects during acamprosate treatment: A dose-response study in humans. Alcohol. Clin. Exp. Res. 2004, 28, 1074–1083. [Google Scholar] [CrossRef]
- Rösner, S.; Hackl-Herrwerth, A.; Leucht, S.; Lehert, P.; Vecchi, S.; Soyka, M. Acamprosate for alcohol dependence. Cochrane Database Syst. Rev. 2010, 8, 45–53. [Google Scholar] [CrossRef]
- Swift, R.; Aston, E. Pharmacotherapy for Alcohol Use Disorder. Harv. Rev. Psychiatry 2015, 23, 122–133. [Google Scholar] [CrossRef]
- Jalali, M.; Botticelli, M.; Hwang, R.; Koh, H.; McHugh, R. The opioid crisis: A contextual, social-ecological framework. Health Res. Policy Syst. 2020, 18, 87. [Google Scholar] [CrossRef] [PubMed]
- Tetrault, J.; Fiellin, D. Current and Potential Pharmacological Treatment Options for Maintenance Therapy in Opioid-Dependent Individuals. Drugs 2012, 72, 217–228. [Google Scholar] [CrossRef]
- Mattick, R.; Breen, C.; Kimber, J.; Davoli, M. Methadone maintenance therapy versus no opioid replacement therapy for opioid dependence. Cochrane Database Syst. Rev. 2009, 2009, CD002209. [Google Scholar] [CrossRef]
- Lobmaier, P.; Gossop, M.; Waal, H.; Bramness, J. The pharmacological treatment of opioid addiction—A clinical perspective. Eur. J. Clin. Pharmacol. 2010, 66, 537–545. [Google Scholar] [CrossRef] [PubMed]
- Stotts, A.; Dodrill, C.; Kosten, T. Opioid dependence treatment: Options in pharmacotherapy. Expert Opin. Pharmacother. 2009, 10, 1727–1740. [Google Scholar] [CrossRef] [PubMed]
- Carlsen, S.; Lunde, L.; Torsheim, T. Opioid and Polydrug Use Among Patients in Opioid Maintenance Treatment. Subst. Abus. Rehabil. 2020, 11, 9–18. [Google Scholar] [CrossRef] [PubMed]
- Bell, J.; Butler, B.; Lawrance, A.; Batey, R.; Salmelainen, P. Comparing overdose mortality associated with methadone and buprenorphine treatment. Drug Alcohol Depend. 2009, 104, 73–77. [Google Scholar] [CrossRef]
- Jaffe, J. From morphine clinics to buprenorphine: Regulating opioid agonist treatment of addiction in the United States. Drug Alcohol Depend. 2003, 70, S3–S11. [Google Scholar] [CrossRef]
- Strang, J.; Hall, W.; Hickman, M.; Bird, S. Impact of supervision of methadone consumption on deaths related to methadone overdose (1993–2008): Analyses using OD4 index in England and Scotland. BMJ 2010, 341, c4851. [Google Scholar] [CrossRef]
- Umezu, T. Unusual Effects of Nicotine as a Psychostimulant on Ambulatory Activity in Mice. ISRN Pharmacol. 2012, 2012, 170981. [Google Scholar] [CrossRef] [PubMed]
- Favrod-Coune, T.; Broers, B. The Health Effect of Psychostimulants: A Literature Review. Pharmaceuticals 2010, 3, 2333–2361. [Google Scholar] [CrossRef] [PubMed]
- Aubin, H.; Luquiens, A.; Berlin, I. Pharmacotherapy for smoking cessation: Pharmacological principles and clinical practice. Br. J. Clin. Pharmacol. 2014, 77, 324–336. [Google Scholar] [CrossRef] [PubMed]
- Molyneux, A. Nicotine replacement therapy. BMJ 2004, 328, 454–456. [Google Scholar] [CrossRef]
- Hartmann-Boyce, J.; Chepkin, S.; Ye, W.; Bullen, C.; Lancaster, T. Nicotine replacement therapy versus control for smoking cessation. Cochrane Database Syst. Rev. 2018, 2019, CD000146. [Google Scholar] [CrossRef]
- Zwar, N.; Mendelsohn, C.; Richmond, R. Supporting smoking cessation. BMJ 2014, 348, f7535. [Google Scholar] [CrossRef]
- Amin, M.; Ali, D. Pharmacology of Medical Cannabis. Adv. Exp. Med. Biol. 2019, 1162, 151–165. [Google Scholar] [CrossRef]
- Connor, J.; Stjepanović, D.; Le Foll, B.; Hoch, E.; Budney, A.; Hall, W. Cannabis use and cannabis use disorder. Nat. Rev. Dis. Prim. 2021, 7, 16. [Google Scholar] [CrossRef]
- Karila, L.; Roux, P.; Rolland, B.; Benyamina, A.; Reynaud, M.; Aubin, H.; Lancon, C. Acute and Long-Term Effects of Cannabis Use: A Review. Curr. Pharm. Des. 2014, 20, 4112–4118. [Google Scholar] [CrossRef]
- Lowe, D.; Sasiadek, J.; Coles, A.; George, T. Cannabis and mental illness: A review. Eur. Arch. Psychiatry Clin. Neurosci. 2018, 269, 107–120. [Google Scholar] [CrossRef] [PubMed]
- Brezing, C.; Levin, F. The Current State of Pharmacological Treatments for Cannabis Use Disorder and Withdrawal. Neuropsychopharmacology 2017, 43, 173–194. [Google Scholar] [CrossRef] [PubMed]
- Sherman, B.; McRae-Clark, A. Treatment of Cannabis Use Disorder: Current Science and Future Outlook. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2016, 36, 511–535. [Google Scholar] [CrossRef] [PubMed]
- Rømer Thomsen, K.; Thylstrup, B.; Kenyon, E.A.; Lees, R.; Baandrup, L.; Feldstein Ewing, S.W.; Freeman, T.P. Cannabinoids for the treatment of Cannabis Use Disorder: New avenues for reaching and helping youth? Neurosci. Biobehav. Rev. 2022, 132, 169–180. [Google Scholar] [CrossRef] [PubMed]
- Hall, W.; Stjepanović, D.; Caulkins, J.; Lynskey, M.; Leung, J.; Campbell, G.; Degenhardt, L. Public health implications of legalising the production and sale of cannabis for medicinal and recreational use. Lancet 2019, 394, 1580–1590. [Google Scholar] [CrossRef]
- Olive, M.; Taylor, S.B.; Lewis, C.R. The neurocircuitry of illicit psychostimulant addiction: Acute and chronic effects in humans. Subst. Abus. Rehabil. 2013, 4, 29–43. [Google Scholar]
- Karila, L.; Gorelick, D.; Weinstein, A.; Noble, F.; Benyamina, A.; Coscas, S.; Blecha, L.; Lowenstein, W.; Martinot, J.L.; Reynaud, M.; et al. New treatments for cocaine dependence: A focused review. Int. J. Neuropsychopharmacol. 2007, 11, 425–438. [Google Scholar] [CrossRef]
- Pierce, R.; Fant, B.; Swinford-Jackson, S.; Heller, E.; Berrettini, W.; Wimmer, M. Environmental, genetic and epigenetic contributions to cocaine addiction. Neuropsychopharmacology 2018, 43, 1471–1480. [Google Scholar] [CrossRef]
- Farrell, M.; Martin, N.; Stockings, E.; Bórquez, A.; Cepeda, J.; Degenhardt, L. Responding to global stimulant use: Challenges and opportunities. Lancet 2019, 394, 1652–1667. [Google Scholar] [CrossRef]
- Phillips, K.; Epstein, D.; Preston, K. Psychostimulant addiction treatment. Neuropharmacology 2014, 87, 150–160. [Google Scholar] [CrossRef] [Green Version]
- Tardelli, V.; Bisaga, A.; Arcadepani, F.; Gerra, G.; Levin, F.; Fidalgo, T. Prescription psychostimulants for the treatment of stimulant use disorder: A systematic review and meta-analysis. Psychopharmacology 2020, 237, 2233–2255. [Google Scholar] [CrossRef] [PubMed]
- Lappin, J.; Sara, G. Psychostimulant use and the brain. Addiction 2019, 114, 2065–2077. [Google Scholar] [CrossRef] [PubMed]
- Hersey, M.; Bacon, A.K.; Bailey, L.G.; Coggiano, M.A.; Newman, A.H.; Leggio, L.; Tanda, G. Psychostimulant Use Disorder, an Unmet Therapeutic Goal: Can Modafinil Narrow the Gap? Front. Neurosci. 2021, 15, 656475. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Gandía, M.; Rodríguez-Arias, M. Pharmacological treatments for opiate and alcohol addiction: A historical perspective of the last 50 years. Eur. J. Pharmacol. 2018, 836, 89–101. [Google Scholar] [CrossRef]
- Perez-Macia, V.; Martinez- Cortes, M.; Mesones, J.; Segura-Trepichio, M.; Garcia-Fernandez, L. Monitoring and Improving Naltrexone Adherence in Patients with Substance Use Disorder. Patient Prefer. Adherence 2021, 15, 999–1015. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, D.C.; Cortes-Briones, J.; Creatura, G.; Bluez, G.; Thurnauer, H.; Deaso, E.; Bielen, K.; Surti, T.; Radhakrishnan, R.; Gupta, A.; et al. Efficacy and safety of a fatty acid amide hydrolase inhibitor (PF-04457845) in the treatment of cannabis withdrawal and dependence in men: A double-blind, placebo-controlled, parallel group, phase 2a single-site randomised controlled trial. Lancet Psychiatry 2019, 6, 35–45. [Google Scholar] [CrossRef]
- Spanagel, R. Cannabinoids and the endocannabinoid system in reward processing and addiction: From mechanisms to interventions. Cannabinoids 2020, 22, 241–250. [Google Scholar] [CrossRef]
- Ozgen, M.; Blume, S. The continuing search for an addiction vaccine. Vaccine 2019, 37, 5485–5490. [Google Scholar] [CrossRef]
- Kuhn, B.; Kalivas, P.; Bobadilla, A. Understanding Addiction Using Animal Models. Front. Behav. Neurosci. 2019, 13, 262. [Google Scholar] [CrossRef]
- Koob, G.; Volkow, N. Neurocircuitry of Addiction. Neuropsychopharmacology 2009, 35, 217–238. [Google Scholar] [CrossRef] [Green Version]
- Volkow, N.; Boyle, M. Neuroscience of Addiction: Relevance to Prevention and Treatment. Am. J. Psychiatry 2018, 175, 729–740. [Google Scholar] [CrossRef] [PubMed]
- Uhl, G.; Koob, G.; Cable, J. The neurobiology of addiction. Ann. N. Y. Acad. Sci. 2019, 1451, 5–28. [Google Scholar] [CrossRef] [PubMed]
- Volkow, N.; Koob, G.; McLellan, A. Neurobiological Advances from the Brain Disease Model of Addiction. N. Engl. J. Med. 2016, 374, 363–371. [Google Scholar] [CrossRef] [PubMed]
- Koob, G. Neurobiology of Addiction: Toward the Development of New Therapies. Ann. N. Y. Acad. Sci. 2006, 909, 170–185. [Google Scholar] [CrossRef]
- Volkow, N.; Michaelides, M.; Baler, R. The Neuroscience of Drug Reward and Addiction. Physiol. Rev. 2019, 99, 2115–2140. [Google Scholar] [CrossRef]
- Tomkins, D.; Sellers, E. Addiction and the brain: The role of neurotransmitters in the cause and treatment of drug dependence. CMAJ 2001, 164, 817–821. [Google Scholar]
- Tzschentke, T.; Schmidt, W. Glutamatergic mechanisms in addiction. Mol. Psychiatry 2003, 8, 373–382. [Google Scholar] [CrossRef]
- Robinson, T. The neural basis of drug craving: An incentive-sensitization theory of addiction. Brain Res. Rev. 1993, 18, 247–291. [Google Scholar] [CrossRef]
- Kalivas, P.; LaLumiere, R.; Knackstedt, L.; Shen, H. Glutamate transmission in addiction. Neuropharmacology 2009, 56, 169–173. [Google Scholar] [CrossRef]
- Nairn, A.; Svenningsson, P.; Nishi, A.; Fisone, G.; Girault, J.; Greengard, P. The role of DARPP-32 in the actions of drugs of abuse. Neuropharmacology 2004, 47, 14–23. [Google Scholar] [CrossRef]
- Hemmings, H.; Greengard, P.; Tung, H.; Cohen, P. DARPP-32, a dopamine-regulated neuronal phosphoprotein, is a potent inhibitor of protein phosphatase-1. Nature 1984, 310, 503–505. [Google Scholar] [CrossRef]
- Feher, J. ATP Production I. In Quantitative Human Physiology; Elsevier: Amsterdam, The Netherlands, 2017. [Google Scholar] [CrossRef]
- Wang, H.; Farhan, M.; Xu, J.; Lazarovici, P.; Zheng, W. The involvement of DARPP-32 in the pathophysiology of schizophrenia. Oncotarget 2017, 8, 53791–53803. [Google Scholar] [CrossRef]
- Saidy, B.; Kotecha, S.; Butler, A.; Rakha, E.A.; Ellis, I.O.; Green, A.R.; Martin, S.G.; Storr, S.J. PP1, PKA and DARPP-32 in breast cancer: A retrospective assessment of protein and mRNA expression. J. Cell. Mol. Med. 2021, 25, 5015–5024. [Google Scholar] [CrossRef]
- Fernandez, É.; Schiappa, R.; Girault, J.; Novère, N. DARPP-32 Is a Robust Integrator of Dopamine and Glutamate Signals. PLoS Comput. Biol. 2006, 2, e176. [Google Scholar] [CrossRef]
- Svenningsson, P.; Lindskog, M.; Rognoni, F.; Fredholm, B.; Greengard, P.; Fisone, G. Activation of adenosine A2A and dopamine D1 receptors stimulates cyclic AMP-dependent phosphorylation of DARPP-32 in distinct populations of striatal projection neurons. Neuroscience 1998, 84, 223–228. [Google Scholar] [CrossRef]
- Nishi, A.; Snyder, G.; Greengard, P. Bidirectional Regulation of DARPP-32 Phosphorylation by Dopamine. J. Neurosci. 1997, 17, 8147–8155. [Google Scholar] [CrossRef]
- Dagda, R.; Das Banerjee, T. Role of protein kinase A in regulating mitochondrial function and neuronal development: Implications to neurodegenerative diseases. Rev. Neurosci. 2015, 26, 359–370. [Google Scholar] [CrossRef]
- Bibb, J.A.; Snyder, G.L.; Nishi, A.; Yan, Z.; Meijer, L.; Fienberg, A.A.; Tsai, L.-H.; Kwon, Y.T.; Girault, J.-A.; Czernik, A.J.; et al. Phosphorylation of DARPP-32 by Cdk5 modulates dopamine signalling in neurons. Nature 1999, 402, 669–671. [Google Scholar] [CrossRef]
- Svenningsson, P.; Nishi, A.; Fisone, G.; Girault, J.-A.; Nairn, A.C.; Greengard, P. DARPP-32: An Integrator of Neurotransmission. Annu. Rev. Pharmacol. Toxicol. 2004, 44, 269–296. [Google Scholar] [CrossRef]
- Bibb, J.A.; Nishi, A.; O’Callaghan, J.P.; Ule, J.; Lan, M.; Snyder, G.L.; Horiuchi, A.; Saito, T.; Hisanaga, S.-I.; Czernik, A.J.; et al. Phosphorylation of Protein Phosphatase Inhibitor-1 by Cdk5. J. Biol. Chem. 2001, 276, 14490–14497. [Google Scholar] [CrossRef] [Green Version]
- Girault, J.; Hemmings, H.; Williams, K.; Nairn, A.; Greengard, P. Phosphorylation of DARPP-32, a dopamine- and cAMP-regulated phosphoprotein, by casein kinase II. J. Biol. Chem. 1989, 264, 21748–21759. [Google Scholar] [CrossRef]
- Desdouits, F.; Siciliano, J.; Greengard, P.; Girault, J. Dopamine- and cAMP-regulated phosphoprotein DARPP-32: Phosphorylation of Ser-137 by casein kinase I inhibits dephosphorylation of Thr-34 by calcineurin. Proc. Natl. Acad. Sci. USA 1995, 92, 2682–2685. [Google Scholar] [CrossRef]
- Nishi, A.; Snyder, G.; Nairn, A.; Greengard, P. Role of Calcineurin and Protein Phosphatase-2A in the Regulation of DARPP-32 Dephosphorylation in Neostriatal Neurons. J. Neurochem. 2016, 72, 2015–2021. [Google Scholar] [CrossRef]
- Nishi, A.; Watanabe, Y.; Higashi, H.; Tanaka, M.; Nairn, A.; Greengard, P. Glutamate regulation of DARPP-32 phosphorylation in neostriatal neurons involves activation of multiple signaling cascades. Proc. Natl. Acad. Sci. USA 2005, 102, 1199–1204. [Google Scholar] [CrossRef]
- Belkhiri, A.; Zhu, S.; El-Rifai, W. DARPP-32: From neurotransmission to cancer. Oncotarget 2016, 7, 17631–17640. [Google Scholar] [CrossRef] [PubMed]
- Ouimet, C.; Miller, P.; Hemmings, H.; Walaas, S.; Greengard, P. DARPP-32, a dopamine- and adenosine 3′:5′-monophosphate-regulated phosphoprotein enriched in dopamine-innervated brain regions. III. Immunocytochemical localization. J. Neurosci. 1984, 4, 111–124. [Google Scholar] [CrossRef] [PubMed]
- Dupre, K. A Potential Neuroanatomical Dissociation of DARPP-32 in Striatal ERK Signaling. J. Neurosci. 2008, 28, 10783–10785. [Google Scholar] [CrossRef] [PubMed]
- Greengard, P.; Allen, P.; Nairn, A. Beyond the Dopamine Receptor. Neuron 1999, 23, 435–447. [Google Scholar] [CrossRef]
- Impey, S.; Obrietan, K.; Storm, D. Making New Connections. Neuron 1999, 23, 11–14. [Google Scholar] [CrossRef]
- Wiegert, J.; Bading, H. Activity-dependent calcium signaling and ERK-MAP kinases in neurons: A link to structural plasticity of the nucleus and gene transcription regulation. Cell Calcium 2011, 49, 296–305. [Google Scholar] [CrossRef]
- Russo, S.; Dietz, D.; Dumitriu, D.; Morrison, J.; Malenka, R.; Nestler, E. The addicted synapse: Mechanisms of synaptic and structural plasticity in nucleus accumbens. Trends Neurosci. 2010, 33, 267–276. [Google Scholar] [CrossRef] [PubMed]
- Gould, T.; Manji, H. DARPP-32: A molecular switch at the nexus of reward pathway plasticity. Proc. Natl. Acad. Sci. USA 2005, 102, 253–254. [Google Scholar] [CrossRef]
- Valjent, E.; Pascoli, V.; Svenningsson, P.; Paul, S.; Enslen, H.; Corvol, J.-C.; Stipanovich, A.; Caboche, J.; Lombroso, P.J.; Nairn, A.C.; et al. From the Cover: Regulation of a protein phosphatase cascade allows convergent dopamine and glutamate signals to activate ERK in the striatum. Proc. Natl. Acad. Sci. USA 2004, 102, 491–496. [Google Scholar] [CrossRef]
- Svenningsson, P.; Nairn, A.; Greengard, P. DARPP-32 mediates the actions of multiple drugs of abuse. AAPS J. 2005, 7, E353–E360. [Google Scholar] [CrossRef] [PubMed]
- Snyder, G.; Fienberg, A.; Huganir, R.; Greengard, P. A Dopamine/D1 Receptor/Protein Kinase A/Dopamine- and cAMP-Regulated Phosphoprotein (Mr32 kDa)/Protein Phosphatase-1 Pathway Regulates Dephosphorylation of the NMDA Receptor. J. Neurosci. 1998, 18, 10297–10303. [Google Scholar] [CrossRef]
- Hoffman, P.; Rabe, C.; Grant, K.; Valverius, P.; Hudspith, M.; Tabakoff, B. Ethanol and the NMDA receptor. Alcohol 1990, 7, 229–231. [Google Scholar] [CrossRef]
- Maldve, R.E.; Zhang, T.A.; Ferrani-Kile, K.; Schreiber, S.S.; Lippmann, M.J.; Snyder, G.L.; Fienberg, A.A.; Leslie, S.W.; Gonzales, R.A.; Morrisett, R.A. DARPP-32 and regulation of the ethanol sensitivity of NMDA receptors in the nucleus accumbens. Nat. Neurosci. 2002, 5, 641–648. [Google Scholar] [CrossRef] [PubMed]
- Risinger, F.; Freeman, P.; Greengard, P.; Fienberg, A. Motivational Effects of Ethanol in DARPP-32 Knock-Out Mice. J. Neurosci. 2001, 21, 340–348. [Google Scholar] [CrossRef]
- Nuutinen, S.; Kiianmaa, K.; Panula, P. DARPP-32 and Akt regulation in ethanol-preferring AA and ethanol-avoiding ANA rats. Neurosci. Lett. 2011, 503, 31–36. [Google Scholar] [CrossRef]
- Abrahao, K.; Oliveira Goeldner, F.; Souza-Formigoni, M. Individual Differences in Ethanol Locomotor Sensitization Are Associated with Dopamine D1 Receptor Intra-Cellular Signaling of DARPP-32 in the Nucleus Accumbens. PLoS ONE 2014, 9, e98296. [Google Scholar] [CrossRef] [Green Version]
- Borgkvist, A.; Usiello, A.; Greengard, P.; Fisone, G. Activation of the cAMP/PKA/DARPP-32 Signaling Pathway is Required for Morphine Psychomotor Stimulation but not for Morphine Reward. Neuropsychopharmacology 2007, 32, 1995–2003. [Google Scholar] [CrossRef] [PubMed]
- Yger, M.; Girault, J. DARPP-32, Jack of All Trades? Master of Which? Front. Behav. Neurosci. 2011, 5, 56. [Google Scholar] [CrossRef] [PubMed]
- Konsolaki, E.; Skaliora, I. Motor vs. cognitive elements of apparent “hyperlocomotion”: A conceptual and experimental clarification. Proc. Natl. Acad. Sci. USA 2014, 112, E3–E4. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Kong, Q. Locomotor activity: A distinctive index in morphine self-administration in rats. PLoS ONE 2017, 12, e0174272. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.; Greene-Naples, J.; Lyle, M.; Iordanou, J.; Felder, J. The Effects of Repeated Opioid Administration on Locomotor Activity: I. Opposing Actions of μ and κ Receptors. J. Pharmacol. Exp. Ther. 2009, 330, 468–475. [Google Scholar] [CrossRef]
- Lindskog, M.; Svenningsson, P.; Fredholm, B.; Greengard, P.; Fisone, G. μ- and δ-opioid receptor agonists inhibit DARPP-32 phosphorylation in distinct populations of striatal projection neurons. Eur. J. Neurosci. 1999, 11, 2182–2186. [Google Scholar] [CrossRef]
- Scheggi, S.; Crociani, A.; De Montis, M.; Tagliamonte, A.; Gambarana, C. Dopamine D1 receptor-dependent modifications in the dopamine and cAMP-regulated phosphoprotein of Mr 32 kDa phosphorylation pattern in striatal areas of morphine-sensitized rats. Neuroscience 2009, 163, 627–639. [Google Scholar] [CrossRef]
- Hamada, M.; Higashi, H.; Nairn, A.; Greengard, P.; Nishi, A. Differential regulation of dopamine D1 and D2 signaling by nicotine in neostriatal neurons. J. Neurochem. 2004, 90, 1094–1103. [Google Scholar] [CrossRef]
- Lee, A.; Picciotto, M. Effects of nicotine on DARPP-32 and CaMKII signaling relevant to addiction. A Tribute to Paul Greengard (1925–2019). Adv. Pharmacol. 2021, 90, 89–115. [Google Scholar] [CrossRef]
- Zhu, H.; Lee, M.; Guan, F.; Agatsuma, S.; Scott, D.; Fabrizio, K.; Fienberg, A.A.; Hiroi, N. DARPP-32 Phosphorylation Opposes the Behavioral Effects of Nicotine. Biol. Psychiatry 2005, 58, 981–989. [Google Scholar] [CrossRef]
- Borgkvist, A.; Marcellino, D.; Fuxe, K.; Greengard, P.; Fisone, G. Regulation of DARPP-32 phosphorylation by Δ9-tetrahydrocannabinol. Neuropharmacology 2008, 54, 31–35. [Google Scholar] [CrossRef]
- Polissidis, A.; Chouliara, O.; Galanopoulos, A.; Rentesi, G.; Dosi, M.; Hyphantis, T.; Marselos, M.; Papadopoulou-Daifoti, Z.; Nomikos, G.G.; Spyraki, C.; et al. Individual differences in the effects of cannabinoids on motor activity, dopaminergic activity and DARPP-32 phosphorylation in distinct regions of the brain. Int. J. Neuropsychopharmacol. 2009, 13, 1175–1191. [Google Scholar] [CrossRef]
- Andersson, M. Cannabinoid Action Depends on Phosphorylation of Dopamine- and cAMP-Regulated Phosphoprotein of 32 kDa at the Protein Kinase A Site in Striatal Projection Neurons. J. Neurosci. 2005, 25, 8432–8438. [Google Scholar] [CrossRef] [PubMed]
- Chiang, Y.; Lo, Y.; Chen, J. Crosstalk between Dopamine D2 receptors and cannabinoid CB1 receptors regulates CNR 1 promoter activity via ERK1/2 signaling. J. Neurochem. 2013, 127, 163–176. [Google Scholar] [CrossRef] [PubMed]
- Girault, J.; Nairn, A. DARPP-32 40 years later. A Tribute to Paul Greengard (1925–2019). Adv. Pharmacol. 2021, 90, 67–87. [Google Scholar] [CrossRef] [PubMed]
- Zachariou, V.; Sgambato-Faure, V.; Sasaki, T.; Svenningsson, P.; Berton, O.; Fienberg, A. Phosphorylation of DARPP-32 at Threonine-34 is Required for Cocaine Action. Neuropsychopharmacology 2005, 31, 555–562. [Google Scholar] [CrossRef] [PubMed]
- Sun, W.; Zhou, L.; Hazim, R.; Quinones-Jenab, V.; Jenab, S. Effects of acute cocaine on ERK and DARPP-32 phosphorylation pathways in the caudate-putamen of Fischer rats. Brain Res. 2007, 1178, 12–19. [Google Scholar] [CrossRef]
- Tropea, T.; Kosofsky, B.; Rajadhyaksha, A. Enhanced CREB and DARPP-32 phosphorylation in the nucleus accumbens and CREB, ERK, and GluR1 phosphorylation in the dorsal hippocampus is associated with cocaine-conditioned place preference behavior. J. Neurochem. 2008, 106, 1780–1790. [Google Scholar] [CrossRef]
- Engmann, O.; Giralt, A.; Gervasi, N.; Marion-Poll, L.; Gasmi, L.; Filhol, O.; Picciotto, M.R.; Gilligan, D.; Greengard, P.; Nairn, A.C.; et al. DARPP-32 interaction with adducin may mediate rapid environmental effects on striatal neurons. Nat. Commun. 2015, 6, 10099. [Google Scholar] [CrossRef]
- Scheggi, S.; Raone, A.; De Montis, M.; Tagliamonte, A.; Gambarana, C. Behavioral expression of cocaine sensitization in rats is accompanied by a distinct pattern of modifications in the PKA/DARPP-32 signaling pathway. J. Neurochem. 2007, 103, 1168–1183. [Google Scholar] [CrossRef] [Green Version]
- Mahajan, S.; Aalinkeel, R.; Reynolds, J.; Nair, B.; Sykes, D.; Hu, Z. Therapeutic Targeting of “DARPP-32”: A Key Signaling molecule in the Dopiminergic Pathway for the Treatment of Opiate Addiction. Int. Rev. Neurobiol. 2009, 88, 199–222. [Google Scholar] [CrossRef] [PubMed]
- Ignatowski, T.A.; Aalinkeel, R.; Reynolds, J.L.; Nair, B.B.; Sykes, D.E.; Gleason, C.P.K.; Law, W.C.; Mammen, M.J.; Prasad, P.N.; Schwartz, S.A.; et al. Nanotherapeutic Approach for Opiate Addiction Using DARPP-32 Gene Silencing in an Animal Model of Opiate Addiction. J. Neuroimmune Pharmacol. 2015, 10, 136–152. [Google Scholar] [CrossRef]
- Bonoiu, A.; Mahajan, S.; Ding, H.; Roy, L.; Yong, K.; Kumar, R. Nanotechnology approach for drug addiction therapy: Gene silencing using delivery of gold nanorod-siRNA nanoplex in dopaminergic neurons. Proc. Natl. Acad. Sci. USA 2009, 106, 5546–5550. [Google Scholar] [CrossRef] [PubMed]
- Kopwitthaya, A.; Cheung Law, W.; Vathy, A.L.; Aalinkeel, R.; Reynolds, L.J.; Nair, B.; Mahajan, S. Evaluating the Efficacy of Gene Silencing in Dopaminergic Neuronal Cells in-vitro Using Gold Nanorods (GNR) with Different Surface Properties Complexed to DARPP-32 siRNA. J. Adv. Pharm. Sci. Technol. 2014, 1, 36–50. [Google Scholar] [CrossRef]
- Souza, R.P.; Soares, E.C.; Rosa, D.V.; Souza, B.R.; Gomes, K.M.; Valvassori, S.S.; Réus, G.Z.; Inácio, C.G.; Martins, M.R.; Gomez, M.V.; et al. Cerebral DARPP-32 expression after methylphenidate administration in young and adult rats. Int. J. Dev. Neurosci. 2008, 27, 1–7. [Google Scholar] [CrossRef]
- Di Luca, M.; Cimino, M.; Cattabeni, F. The phosphorylation state of DARPP-32, a third messenger for dopamine, is regulated by in vivo pharmacological treatments. Neurochem. Int. 1992, 20, 165–170. [Google Scholar] [CrossRef]
- Kohut, S. Interactions between nicotine and drugs of abuse: A review of preclinical findings. Am. J. Drug Alcohol Abus. 2016, 43, 155–170. [Google Scholar] [CrossRef] [Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Greener, M.R.; Storr, S.J. Exploring the Role of DARPP-32 in Addiction: A Review of the Current Limitations of Addiction Treatment Pathways and the Role of DARPP-32 to Improve Them. NeuroSci 2022, 3, 494-509. https://doi.org/10.3390/neurosci3030035
Greener MR, Storr SJ. Exploring the Role of DARPP-32 in Addiction: A Review of the Current Limitations of Addiction Treatment Pathways and the Role of DARPP-32 to Improve Them. NeuroSci. 2022; 3(3):494-509. https://doi.org/10.3390/neurosci3030035
Chicago/Turabian StyleGreener, Megan R., and Sarah J. Storr. 2022. "Exploring the Role of DARPP-32 in Addiction: A Review of the Current Limitations of Addiction Treatment Pathways and the Role of DARPP-32 to Improve Them" NeuroSci 3, no. 3: 494-509. https://doi.org/10.3390/neurosci3030035
APA StyleGreener, M. R., & Storr, S. J. (2022). Exploring the Role of DARPP-32 in Addiction: A Review of the Current Limitations of Addiction Treatment Pathways and the Role of DARPP-32 to Improve Them. NeuroSci, 3(3), 494-509. https://doi.org/10.3390/neurosci3030035






