Motivational and Control Mechanisms Underlying Adolescent versus Adult Alcohol Use
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants
2.2. Questionnaires on Substance Use and Mental Health
2.3. Motivational and Cognitive Control Measures
2.4. Procedure
2.5. Data Preparation and Analysis
3. Results
3.1. Group Comparison between Adolescents and Adults
3.2. Associations of Alcohol Use Measures with Motivational and Control Processes
3.3. Does Age Moderate the Relation between Alcohol Use and Cognition?
4. Discussion
5. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- Crone, E.A.; Dahl, R.E. Understanding adolescence as a period of social–affective engagement and goal flexibility. Nat. Rev. Neurosci. 2012, 13, 636–650. [Google Scholar] [CrossRef] [PubMed]
- Chassin, L.; Fora, D.B.; King, K.M. Trajectories of alcohol and drug use and dependence from adolescence to adulthood: The effects of familial alcoholism and personality. J. Abnorm Psychol. 2004, 113, 483–498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lee, M.R.; Boness, C.L.; McDowell, Y.E.; Vergés, A.; Steinley, D.L.; Sher, K.J. Desistance and Severity of Alcohol Use Disorder: A Lifespan-Developmental Investigation. Clin. Psychol. Sci. 2018, 6, 90–105. [Google Scholar] [CrossRef] [PubMed]
- Johnston, L.D.; Miech, R.A.; O’Malley, P.M.; Bachman, J.G.; Schulenberg, J.E.; Patrick, M.E. Monitoring the Future National Survey Results on Drug Use, 1975–2017: Overview key findings on adolescent drug use 2018. Available online: https://files.eric.ed.gov/fulltext/ED589762.pdf (accessed on 21 September 2020).
- Carroll, L.J.; Cassidy, J.D.; Peloso, P.M.; Borg, J.; von Holst, H.; Holm, L.; Paniak, C.; Pepin, M. Prognosis for mild traumatic brain injury: Results of the WHO Collaborating Centre Task Force on Mild Traumatic Brain Injury. J. Rehabil Med. 2004, 84–105. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Uhl, G.R.; Koob, G.F.; Cable, J. The neurobiology of addiction. Ann. N. Y. Acad. Sci. 2019, 62, 118–127. [Google Scholar] [CrossRef]
- Asbridge, M.; Cartwright, J.; Wilson, K.; Langille, D. Age at first drink, experiences of drunkenness, and alcohol-related problems in Canadian youth: Is early onset bad if you are a moderate drinker? J. Stud. Alcohol Drugs 2016, 77, 974–979. [Google Scholar] [CrossRef]
- DeWit, D.J.; Adlaf, E.M.; Offord, D.R.; Ogborne, A.C. Age at first alcohol use: A risk factor for the development of alcohol disorders. Am. J. Psychiatry 2000, 157, 745–750. [Google Scholar] [CrossRef]
- Liang, W.; Chikritzhs, T. Age at first use of alcohol and risk of heavy alcohol use: A population-based study. Biomed Res. Int. 2013, 2013. [Google Scholar] [CrossRef] [Green Version]
- Pitkänen, T.; Lyyra, A.L.; Pulkkinen, L. Age of onset of drinking and the use of alcohol in adulthood: A follow-up study from age 8–42 for females and males. Addiction 2005, 100, 652–661. [Google Scholar] [CrossRef]
- Steinberg, L. Cognitive and affective development in adolescence. Trends Cogn. Sci. 2005, 9, 69–74. [Google Scholar] [CrossRef]
- Davidow, J.Y.; Foerde, K.; Galván, A.; Shohamy, D. An upside to reward sensitivity: The hippocampus supports enhanced reinforcement learning in adolescence. Neuron 2016, 92, 93–99. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Cousijn, J.; Luijten, M.; Feldstein Ewing, S.W. Adolescent resilience to addiction: A social plasticity hypothesis. Lancet Child Adolesc. Heal. 2018, 2, 69–78. [Google Scholar] [CrossRef]
- Squeglia, L.M.; Jacobus, J.; Tapert, S.F. The effect of alcohol use on human adolescent brain structures and systems. In Handbook of Clinical Neurology; Elsevier: Edinburgh, UK, 2014; Volume 125, pp. 501–510. [Google Scholar]
- Spear, L.P. Adolescent alcohol exposure: Are there separable vulnerable periods within adolescence? Physiol. Behav. 2015, 148, 122–130. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McAteer, A.M.; Hanna, D.; Curran, D. Age-related differences in alcohol attention bias: A cross-sectional study. Psychopharmacology 2018, 235, 2387–2393. [Google Scholar] [CrossRef]
- Scheel, J.F.; Schielke, K.; Lautenbacher, S.; Aust, S.; Kremer, S.; Wolstein, J. Low-Dose Alcohol Effects on Attention in Adolescents. Zeitschrift für Neuropsychol. 2013, 24, 103–111. [Google Scholar] [CrossRef]
- Rooke, S.E.; Hine, D.W. A dual process account of adolescent and adult binge drinking. Addict. Behav. 2011, 36, 341–346. [Google Scholar] [CrossRef] [PubMed]
- Wiers, R.W.; Bartholow, B.D.; Van den Wildenberg, E.; Thush, C.; Engels, R.C.M.E.; Sher, K.J.; Grenard, J.; Ames, S.L.; Stacy, A.W. Automatic and controlled processes and the development of addictive behaviors in adolescents: A review and a model. Pharmacol. Biochem. Behav. 2007, 86, 263–283. [Google Scholar] [CrossRef] [PubMed]
- Conrod, P.; Nikolaou, K. Annual Research Review: On the developmental neuropsychology of substance use disorders. J. Child Psychol. Psychiatry 2016, 57, 371–394. [Google Scholar] [CrossRef]
- Saunders, J.B.; Aasland, O.G.; Babor, T.F.; de la Fuente, J.R.; Grant, M. Development of the Alcohol Use Disorders Identification Test (AUDIT): WHO Collaborative Project on Early Detection of Persons with Harmful Alcohol Consumption--II. Addiction 1993, 88, 791–804. [Google Scholar] [CrossRef]
- Babor, T.F.; Robaina, K. The Alcohol Use Disorders Identification Test (AUDIT): A review of graded severity algorithms and national adaptations. Int. J. Alcohol Drug Res. 2016, 5, 17. [Google Scholar] [CrossRef]
- Heatherton, T.F.; Kozlowski, L.T.; Frecher, R.C.; Fagerstrom, K.O. The Fagerström Test for Nicotine Dependence: A revision of the Fagerstrom Tolerance Questionnaire. Br. J. Addict. 1991, 86, 1119–1127. [Google Scholar] [CrossRef] [PubMed]
- Beck, A.T.; Steer, R.A.; Ball, R.; Ranieri, W.F. Comparison of Beck depression inventories -IA and -II in psychiatric outpatients. J. Pers. Assess. 1996, 67, 588–597. [Google Scholar] [CrossRef] [PubMed]
- Spielberger, C.D.; Gorsuch, R.L.; Lushene, R.E. The State Trait Anxiety Inventory; Consulting Psychologists Press: Palo Alto, CA, USA, 1970. [Google Scholar]
- Cousijn, J.; van Benthem, P.; van der Schee, E.; Spijkerman, R. Motivational and control mechanisms underlying adolescent cannabis use disorders: A prospective study. Dev. Cogn. Neurosci. 2015, 16, 36–45. [Google Scholar] [CrossRef] [PubMed]
- Ataya, A.F.; Adams, S.; Mullings, E.; Cooper, R.M.; Attwood, A.S.; Munafò, M.R. Internal reliability of measures of substance-related cognitive bias. Drug Alcohol Depend. 2012, 121, 148–151. [Google Scholar] [CrossRef]
- Cooper, M.L. Motivations for alcohol use among adolescents: Development and validation of a four-factor model. Psychol. Assess. 1994, 6, 117–128. [Google Scholar] [CrossRef]
- Patton, J.H.; Stanford, M.S.; Barratt, E.S. Factor structure of the barratt impulsiveness scale. J. Clin. Psychol. 1995, 51, 768–774. [Google Scholar] [CrossRef]
- Hammes, J.G.W. De Stroop Kleur-Woord Test Handleiding; Swets en Zeitlinger: Lisse, The Netherlands, 1971. [Google Scholar]
- Figner, B.; Mackinlay, R.J.; Wilkening, F.; Weber, E.U. Affective and Deliberative Processes in Risky Choice: Age Differences in Risk Taking in the Columbia Card Task. J. Exp. Psychol. Learn. Mem. Cogn. 2009, 35, 709–730. [Google Scholar] [CrossRef] [Green Version]
- Petrides, M.; Milner, B. Deficits on subject-ordered tasks after frontal- and temporal-lobe lesions in man. Neuropsychologia 1982, 20, 249–262. [Google Scholar] [CrossRef]
- Jeffreys, H. Theory of Probability 3rd edition, 3rd ed.; Clarendon Press: Oxford, UK, 1961. [Google Scholar]
- Carlson, S.R.; Johnson, S.C. Impulsivity is not always associated with student drinking: A moderation study of impulsivity and drinking by positive alcohol expectancies. Addict. Behav. 2012, 37, 556–560. [Google Scholar] [CrossRef]
- Fernie, G.; Cole, J.C.; Goudie, A.J.; Field, M. Risk-taking but not response inhibition or delay discounting predict alcohol consumption in social drinkers. Drug Alcohol Depend. 2010, 112, 54–61. [Google Scholar] [CrossRef]
- Goudriaan, A.E.; Grekin, E.R.; Sher, K.J. Decision making and binge drinking: A longitudinal study. Alcohol. Clin. Exp. Res. 2007, 31, 928–938. [Google Scholar] [CrossRef]
- Von Diemen, L.; Bassani, D.G.; Fuchs, S.C.; Szobot, C.M.; Pechansky, F. Impulsivity, age of first alcohol use and substance use disorders among male adolescents: A population based case-control study. Addiction 2008, 103, 1198–1205. [Google Scholar] [CrossRef] [PubMed]
- Sharma, L.; Markon, K.E.; Clark, L.A. Toward a theory of distinct types of “impulsive” behaviors: A meta-analysis of self-report and behavioral measures. Psychol. Bull. 2014, 140, 374–408. [Google Scholar] [CrossRef] [PubMed]
- Dalley, J.W.; Robbins, T.W. Fractionating impulsivity: Neuropsychiatric implications. Nat. Rev. Neurosci. 2017, 18, 158–171. [Google Scholar] [CrossRef]
- Fineberg, N.A.; Chamberlain, S.R.; Goudriaan, A.E.; Stein, D.J.; Vanderschuren, L.J.M.J.; Gillan, C.M.; Shekar, S.; Gorwood, P.A.P.M.; Voon, V.; Morein-Zamir, S.; et al. New developments in human neurocognition: Clinical, genetic, and brain imaging correlates of impulsivity and compulsivity. CNS Spectr. 2014, 19, 69–89. [Google Scholar] [CrossRef] [PubMed]
- Hershberger, A.R.; Um, M.; Cyders, M.A. The relationship between the UPPS-P impulsive personality traits and substance use psychotherapy outcomes: A meta-analysis. Drug Alcohol Depend. 2017, 178, 408–416. [Google Scholar] [CrossRef]
- Leung, D.; Staiger, P.K.; Hayden, M.; Lum, J.A.G.; Hall, K.; Manning, V.; Verdejo-Garcia, A. Meta-analysis of the relationship between impulsivity and substance-related cognitive biases. Drug Alcohol Depend. 2017, 172, 21–33. [Google Scholar] [CrossRef]
- Quinn, P.D.; Harden, K.P. Differential changes in impulsivity and sensation seeking and the escalation of substance use from adolescence to early adulthood. Dev. Psychopathol. 2013, 25, 223–239. [Google Scholar] [CrossRef] [Green Version]
- Argyriou, E.; Um, M.; Carron, C.; Cyders, M.A. Age and impulsive behavior in drug addiction: A review of past research and future directions. Pharmacol. Biochem. Behav. 2018, 164, 106–117. [Google Scholar] [CrossRef] [Green Version]
- De Wit, H. Impulsivity as a determinant and consequence of drug use: A review of underlying processes. Addict. Biol. 2009, 14, 22–31. [Google Scholar] [CrossRef]
- Kuntsche, E.; Knibbe, R.; Gmel, G.; Engels, R. Why do young people drink? A review of drinking motives. Clin Psychol Rev 2005, 25, 841–861. [Google Scholar] [CrossRef] [PubMed]
- Littlefield, A.K.; Sher, K.J.; Wood, P.K. Do changes in drinking motives mediate the relation between personality change and “maturing out” of problem drinking? J Abnorm Psychol 2010, 119, 93–105. [Google Scholar] [CrossRef] [Green Version]
- Treloar Padovano, H.; Miranda, R. Subjective cannabis effects as part of a developing disorder in adolescents and emerging adults. J. Abnorm. Psychol. 2018, 127, 282–293. [Google Scholar] [CrossRef] [PubMed]
- Spear, L.P. Effects of adolescent alcohol consumption on the brain and behaviour. Nat. Rev. Neurosci. 2018, 19, 197–214. [Google Scholar] [CrossRef] [PubMed]
- Schramm-Sapyta, N.L.; Walker, Q.D.; Caster, J.M.; Levin, E.D.; Kuhn, C.M. Are adolescents more vulnerable to drug addiction than adults? Evidence from animal models. Psychopharmacology 2009, 206, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Crews, F.T.; Braun, C.J.; Hoplight, B.; Switzer, R.C.; Knapp, D.J. Binge ethanol consumption causes differential brain damage in young adolescent rats compared with adult rats. Alcohol. Clin. Exp. Res. 2000, 24, 1712–1723. [Google Scholar] [CrossRef]
- Schindler, A.G.; Tsutsui, K.T.; Clark, J.J. Chronic alcohol intake during adolescence, but not adulthood, promotes persistent deficits in risk-based decision making. Alcohol. Clin. Exp. Res. 2014, 38, 1622–1629. [Google Scholar] [CrossRef]
- Lee, K.M.; Coelho, M.A.; McGregor, H.A.; Solton, N.R.; Cohen, M.; Szumlinski, K.K. Adolescent Mice Are Resilient to Alcohol Withdrawal-Induced Anxiety and Changes in Indices of Glutamate Function within the Nucleus Accumbens. Front. Cell. Neurosci. 2016, 10. [Google Scholar] [CrossRef] [Green Version]
- Pautassi, R.M.; Godoy, J.C.; Molina, J.C. Adolescent rats are resistant to the development of ethanol-induced chronic tolerance and ethanol-induced conditioned aversion. Pharmacol. Biochem. Behav. 2015, 138, 58–69. [Google Scholar] [CrossRef]
- Fernandez, G.M.; Lew, B.J.; Vedder, L.C.; Savage, L.M. Chronic intermittent ethanol exposure leads to alterations in brain-derived neurotrophic factor within the frontal cortex and impaired behavioral flexibility in both adolescent and adult rats. Neuroscience 2017, 348, 324–334. [Google Scholar] [CrossRef] [Green Version]
- Labots, M.; Cousijn, J.; Jolink, L.A.; Leon Kenemans, J.; Vanderschuren, L.J.M.J.; Lesscher, H.M.B. Age-related differences in alcohol intake and control over alcohol seeking in rats. Front. Psychiatry 2018, 9. [Google Scholar] [CrossRef] [PubMed]
- Waber, D.P.; De Moor, C.; Forbes, P.W.; Almli, C.R.; Botteron, K.N.; Leonarf, G.; Molovan, D.; Paus, T.; Rumsey, J. The NIH MRI study of normal brain development: Performance of a population based sample of healthy children aged 6 to 18 years on a neuropsychological battery. J. Int. Neuropsychol. Soc. 2007, 13, 729–746. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Comalli, P.E.; Wapner, S.; Werner, H. Interference effects of stroop color-word test in childhood, adulthood, and aging. J. Genet. Psychol. 1962, 100, 47–53. [Google Scholar] [CrossRef] [PubMed]
- Baddeley, A. Working Memory: Theories, Models, and Controversies. Annu. Rev. Psychol. 2012, 63, 1–29. [Google Scholar] [CrossRef] [Green Version]
- Varlinskaya, E.I.; Spear, L.P. Acute Effects of Ethanol on Social Behavior of Adolescent and Adult Rats: Role of Familiarity of the Test Situation. Alcohol. Clin. Exp. Res. 2002, 26, 1502–1511. [Google Scholar] [CrossRef]
| Adolescents | Adults | |||||||
|---|---|---|---|---|---|---|---|---|
| N | Mean | SD | N | Mean | SD | p | BF01 | |
| Age | 45 | 16.5 | 0.5 | 45 | 31.6 | 1.5 | ||
| Male | 23 | 23 | ||||||
| Alcohol use: | ||||||||
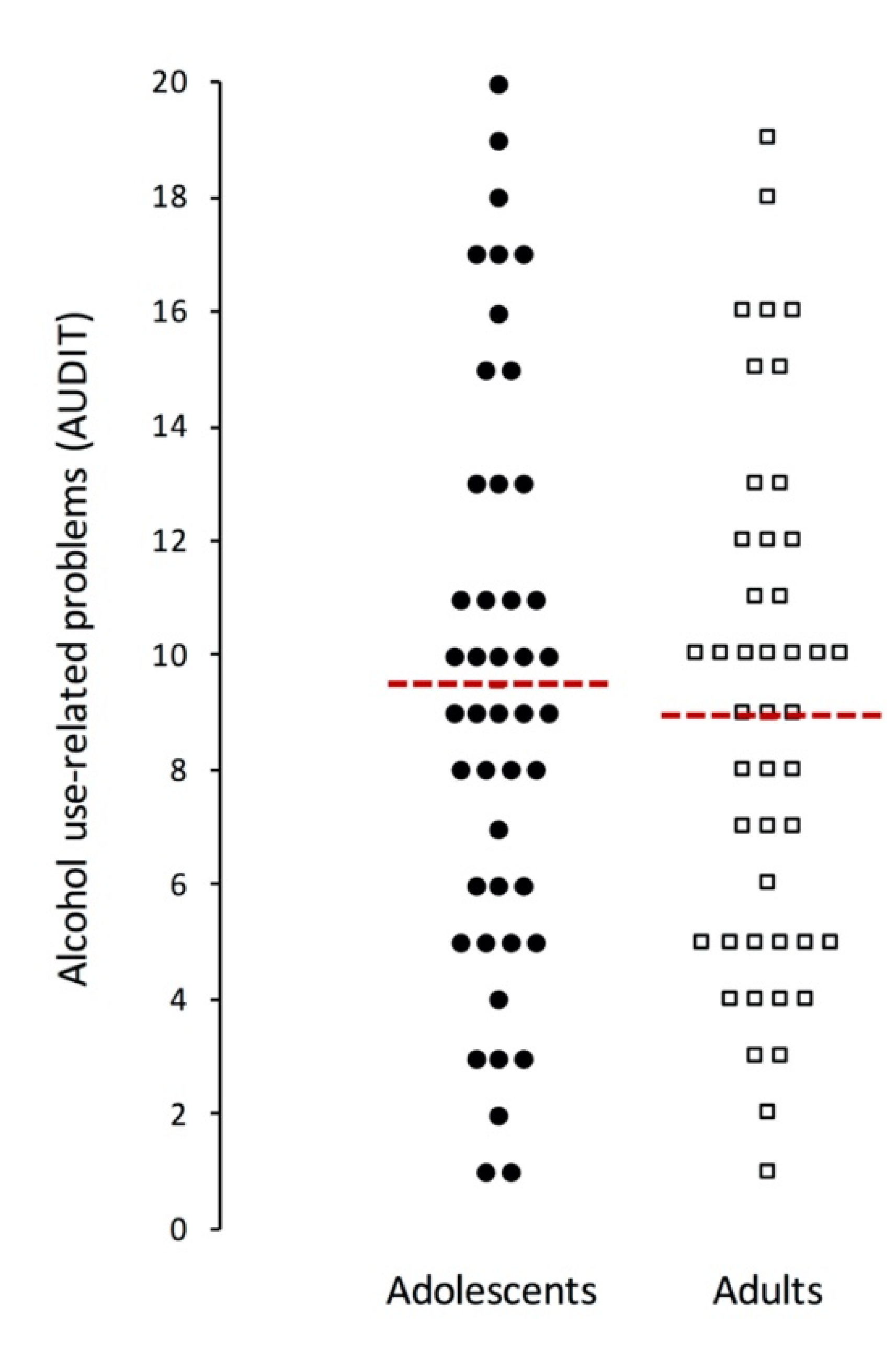
| AUDIT total | 45 | 9.5 | 5.0 | 45 | 8.9 | 4.5 | 0.593 | 4.00 |
| AUDIT problems (sum AUDIT 4–10) | 45 | 3.6 | 3.2 | 45 | 3.4 | 3.5 | 0.622 † | 4.04 |
| Monthly use, (standard units) | 45 | 40.5 | 37.9 | 45 | 40.7 | 30.9 | 0.478 † | 3.92 |
| Monthly use (days) | 45 | 6.1 | 0.7 | 45 | 12.6 | 1.2 | 0.000† | 0.01 |
| Drinks per drinking episode (standard units) | 45 | 6.3 | 0.5 | 45 | 3.3 | 0.3 | 0.000† | 0.00 |
| Age first drink | 45 | 14.2 | 1.2 | 45 | 14.1 | 1.8 | 0.437 † | 3.49 |
| Age first binge | 43 | 15.0 | 1.1 | 45 | 16.4 | 2.5 | 0.001† | 0.07 |
| Age first time drunk | 39 | 15.0 | 0.9 | 44 | 15.8 | 2.6 | 0.047† | 1.23 |
| Nicotine dependence (FTND) | 19 | 1.4 | 1.7 | 18 | 1.7 | 2.2 | 0.964 † | 3.01 |
| Lifetime Illicit substance (episodes) | 44 | 5.0 | 21.1 | 43 | 17.1 | 36.9 | 0.011† | 0.70 |
| Depression (BDI) | 45 | 8.2 | 6.2 | 45 | 5.6 | 4.6 | 0.033† | 0.43 |
| Anxiety (STAI): State | 45 | 32.0 | 7.3 | 45 | 34.4 | 8.8 | 0.168 † | 2.50 |
| Anxiety (STAI): Trait | 45 | 34.9 | 8.2 | 45 | 38.5 | 10.1 | 0.064 | 1.06 |
| Motivational and Control Measures | ||||||||
| Session induced alcohol craving (VAS) | 45 | 0.0 | 1.1 | 45 | 0.0 | 2.1 | 0.454 † | 3.69 |
| Alcohol attentional bias (Alcohol Stroop, s) | 45 | 1.3 | 4.5 | 44 | 1.2 | 3.5 | 0.802 † | 4.45 |
| Cannabis-Alcohol Approach Avoidance task: | ||||||||
| Alcohol approach bias (ms) | 44 | -31.0 | 81.2 | 45 | -10.6 | 83.7 | 0.246 | 2.48 |
| Cannabis approach bias (ms) | 44 | -20.6 | 109.8 | 45 | -17.8 | 79.1 | 0.891 | 4.47 |
| Soda approach bias (ms) | 44 | -17.6 | 70.0 | 45 | -8.4 | 79.9 | 0.565 | 3.89 |
| Neutral approach bias (ms) | 44 | -18.4 | 85.9 | 45 | -14.8 | 76.9 | 0.836 | 4.42 |
| Drinking Motives (DMQ-r): Social | 44 | 11.6 | 5.2 | 44 | 11.8 | 4.1 | 0.957 † | 4.47 |
| Drinking Motives (DMQ-r): Coping | 44 | 3.8 | 4.3 | 44 | 3.4 | 3.1 | 0.836 † | 4.25 |
| Drinking Motives (DMQ-r): Enhancement | 44 | 9.3 | 4.7 | 44 | 6.8 | 3.8 | 0.009† | 0.24 |
| Drinking Motives (DMQ-r): Conformity | 44 | 0.8 | 1.6 | 44 | 1.2 | 1.6 | 0.247 † | 3.25 |
| Impulsivity (BIS-11) | 45 | 66.1 | 11.0 | 45 | 58.6 | 8.7 | 0.001 | 0.02 |
| Interference control (Classical Stroop) | 45 | 0.7 | 0.1 | 45 | 0.7 | 0.1 | 0.597 † | 4.05 |
| Risky decision making (CCT, cards) | 44 | 8.3 | 1.8 | 45 | 8.21 | 2.3 | 0.781 | 4.35 |
| Working memory (SOPT, correct clicks) | 43 | 59.6 | 4.4 | 45 | 59.1 | 4.6 | 0.623 | 4.03 |
| Measure | AUDIT Problems | Monthly Use (Drinking Days) | Drinks per Drinking Episode (Standard Units) | |||
|---|---|---|---|---|---|---|
| rτ | BF01 | rτ | BF01 | rτ | BF01 | |
| Session induced alcohol craving (VAS) | 0.08 | 4.06 | 0.11 | 2.10 | −0.06 | 5.44 |
| Alcohol attentional bias (Alcohol Stroop) | −0.01 | 7.10 | −0.02 | 6.96 | −0.12 | 1.94 |
| Alcohol approach bias (CA-AAT, ms) | −0.08 | 3.90 | 0.08 | 3.92 | −0.14 | 1.05 |
| Social drinking motives (DMQ-r) | 0.26 ** | 0.01 | 0.24 * | 0.04 | 0.19 * | 0.26 |
| Coping drinking motives (DMQ-r) | 0.29 ** | 0.00 | 0.29 ** | 0.00 | 0.04 | 6.06 |
| Enhancement drinking motives (DMQ-r) | 0.41 ** | 0.00 | 0.21 * | 0.13 | 0.37 ** | 0.00 |
| Conformity drinking motives (DMQ-r) | 0.15 | 0.78 | 0.13 | 1.68 | −0.08 | 3.78 |
| Impulsivity (BIS-11) | 0.17 * | 0.47 | −0.06 | 5.32 | 0.15 * | 0.79 |
| Interference control (Classical Stroop) | 0.08 | 3.87 | 0.01 | 7.18 | −0.03 | 6.53 |
| Risky decision making (CCT) | −0.01 | 7.21 | −0.02 | 6.90 | −0.02 | 7.01 |
| Working memory (SOPT) | −0.02 | 6.84 | 0.07 | 4.37 | 0.01 | 7.06 |
| Predictor | Alcohol Use Outcome | ∆R2 | B | 95% CI bca (B) | SE (B) | b | p | BF01 |
|---|---|---|---|---|---|---|---|---|
| Impulsivity (BIS-11) X age | AUDIT problems | 0.01 | 0.55 | −7.23–1.81 | 0.65 | 0.13 | 0.45 | 1.43 |
| Monthly use (drinking days) | 0.03 | 2.83 | −0.47–5.45 | 1.49 | 0.30 | 0.05 | 0.59 | |
| Drinks per drinking episode (standard units) | 0.01 | 0.52 | −0.47–1.51 | 0.50 | 0.13 | 0.29 | 2.50 | |
| Drinking motives (DMQ-r) | ||||||||
| Social X age | AUDIT problems | 0.05 | 1.31 | −0.26–4.14 | 1.09 | 0.34 | 0.04 | 0.69 |
| Monthly use (drinking days) | 0.01 | −1.47 | −3.65–0.85 | 1.15 | −0.17 | 0.24 | 2.13 | |
| Drinks per drinking episode (standard units) | 0.02 | 0.89 | 0.02–1.95 | 0.50 | 0.24 | 0.10 | 1.13 | |
| Coping X age | AUDIT problems | 0.00 | 0.14 | −1.03–1.96 | 0.73 | 0.05 | 0.79 | 2.05 |
| Monthly use (drinking days) | 0.01 | −1.16 | −4.37–1.59 | 1.51 | -0.13 | 0.38 | 2.81 | |
| Enhancement X age | AUDIT problems | 0.01 | 0.39 | −0.84–3.26 | 0.95 | 0.12 | 0.47 | 3.10 |
| Monthly use (drinking days) | 0.01 | −1.82 | −4.13–0.98 | 1.28 | −0.20 | 0.15 | 1.80 | |
| Drinks per drinking episode (standard units) | 0.00 | 0.19 | −0.87–1.25 | 0.54 | 0.05 | 0.73 | 3.86 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cousijn, J.; Green, K.H.; Labots, M.; Vanderschuren, L.J.M.J.; Kenemans, J.L.; Lesscher, H.M.B. Motivational and Control Mechanisms Underlying Adolescent versus Adult Alcohol Use. NeuroSci 2020, 1, 44-58. https://doi.org/10.3390/neurosci1010005
Cousijn J, Green KH, Labots M, Vanderschuren LJMJ, Kenemans JL, Lesscher HMB. Motivational and Control Mechanisms Underlying Adolescent versus Adult Alcohol Use. NeuroSci. 2020; 1(1):44-58. https://doi.org/10.3390/neurosci1010005
Chicago/Turabian StyleCousijn, Janna, Kayla H. Green, Maaike Labots, Louk J. M. J. Vanderschuren, J. Leon Kenemans, and Heidi M. B. Lesscher. 2020. "Motivational and Control Mechanisms Underlying Adolescent versus Adult Alcohol Use" NeuroSci 1, no. 1: 44-58. https://doi.org/10.3390/neurosci1010005
APA StyleCousijn, J., Green, K. H., Labots, M., Vanderschuren, L. J. M. J., Kenemans, J. L., & Lesscher, H. M. B. (2020). Motivational and Control Mechanisms Underlying Adolescent versus Adult Alcohol Use. NeuroSci, 1(1), 44-58. https://doi.org/10.3390/neurosci1010005





