Abstract
Pediatric acute-onset neuropsychiatric syndrome (PANS) is a condition characterized by the abrupt, dramatic onset of obsessive–compulsive disorder (OCD) or eating restriction accompanied by equally abrupt and severe comorbid neuropsychiatric symptoms. PANDAS (pediatric autoimmune neuropsychiatric disorder associated with streptococcal infection) is a heterogeneous syndrome identified as post-Streptococcus pyogenes infection (β-hemolytic Streptococcus group A) complications regarding the central nervous system with specific involvement of neuropsychiatric and behavioral skills. In the first part of our study, we share our experience in the treatment of a group of extreme-grade (according to CY-BOCS severity scale) symptomatic patients with intravenous immunoglobulin (IVIG), following the most recent studies regarding the dosage of the drug. Our contribution is to share our experience made on a sample of 55 patients all in the highest level of a severity grade. In the second part of our study, we also analyze the literature on PANS/PANDAS rehabilitation therapy, since in the literature there is no discussion of union and comparison on this method. Objective: This study aims to evaluate the clinical features of the patients observed from different Italian cohorts, with the attempt at evaluating clinical response to IVIG treatment in children with an extreme severity grade of PANS/PANDAS disease. Furthermore, after having analyzed the literature, we propose rehabilitation therapy as an added value to the pharmacological treatment. Materials and Methods: A total of 55 patients with a diagnosis of PANS/PANDAS, who belonged to an extreme grade of disease, were enrolled. All patients were administered with IVIG treatment at 2 g/kg per day for two consecutive days. Results From our study, a noticeable improvement (until complete remission) of symptoms was evident for at least one year in 47 out of 55 (85%) observed children, while 11 out of these 43 (25%) showed an evident symptoms remission in a single attempt and the remaining 32 (75%) required a second administration to notice a lasting symptomatic improvement.
1. Introduction
PANDAS (pediatric autoimmune neuropsychiatric disorder associated with streptococcal infection) collects a specific specter of disturbances linked to group A beta-hemolytic streptococcal infection (GABHS). It is a post-infectious complication affecting the central nervous system, similarly to Sydenham chorea or rheumatic fever, but with specific involvement of neuropsychiatric and behavioral skills. Swedo et al. firstly described these disturbances in 1998 [1], reporting five clinical features required to diagnose these disorders, including the presence of neuropsychiatric abnormalities, mainly obsessive–compulsive disorder (OCD) and tic, with onset in the pre-pubertal age (3–14 years) and a relapsing/remitting course, clearly linked to a GABHS previous infection. Hyperactivity, choreiform movements, and Tourette syndrome (TS) are also possible diagnostic elements. From the several cases that came out from literature over time [2,3,4], it is evident that there is a great variety of phenotypes, so allowing to widen the edges of these new disorders including CANS (childhood acute neuropsychiatric symptoms), defined by the presence of OCD with abrupt onset and PANS (pediatric acute-onset neuropsychiatric syndrome), of which PANDAS represents a subgroup of disorders [5,6].
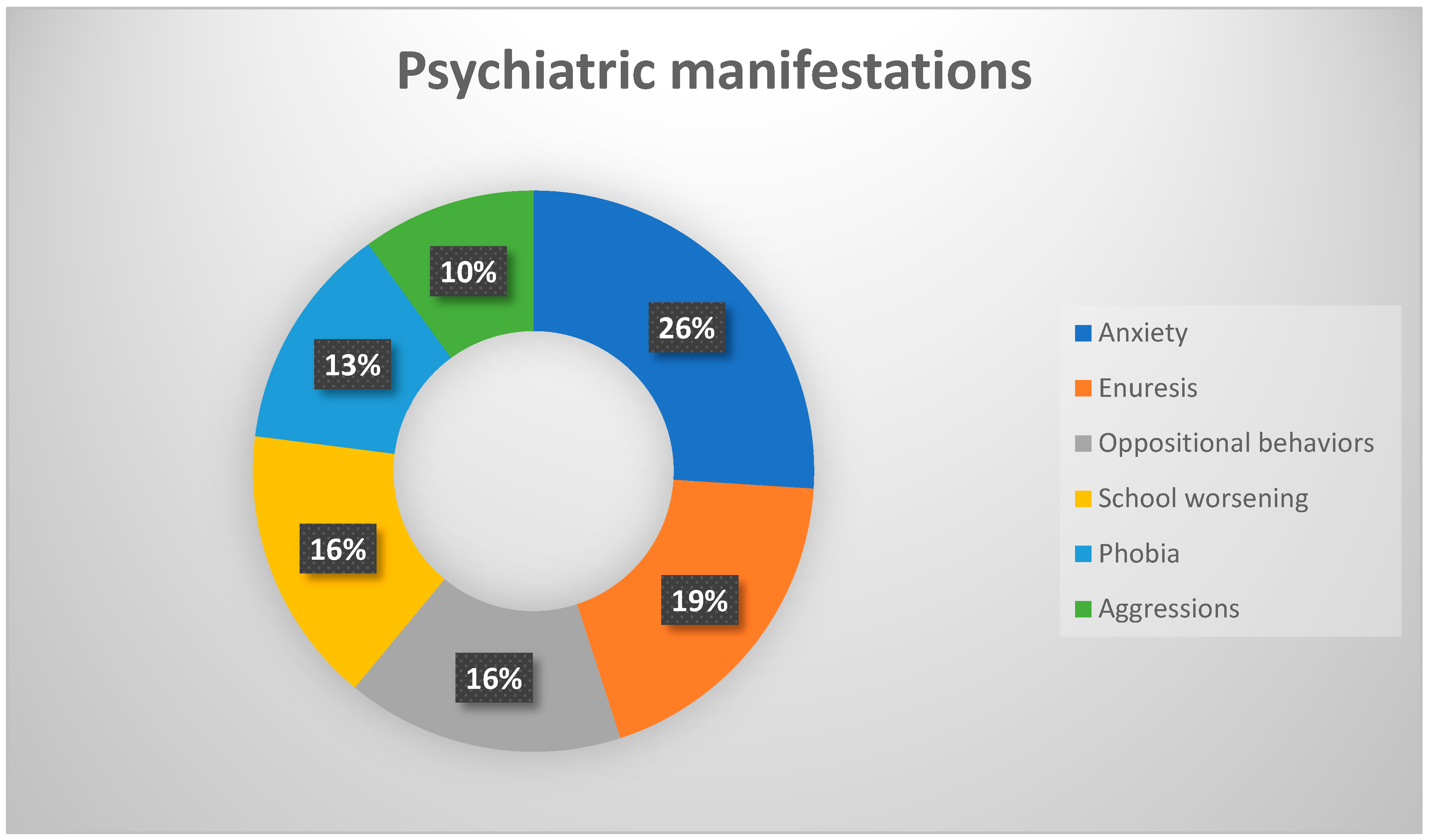
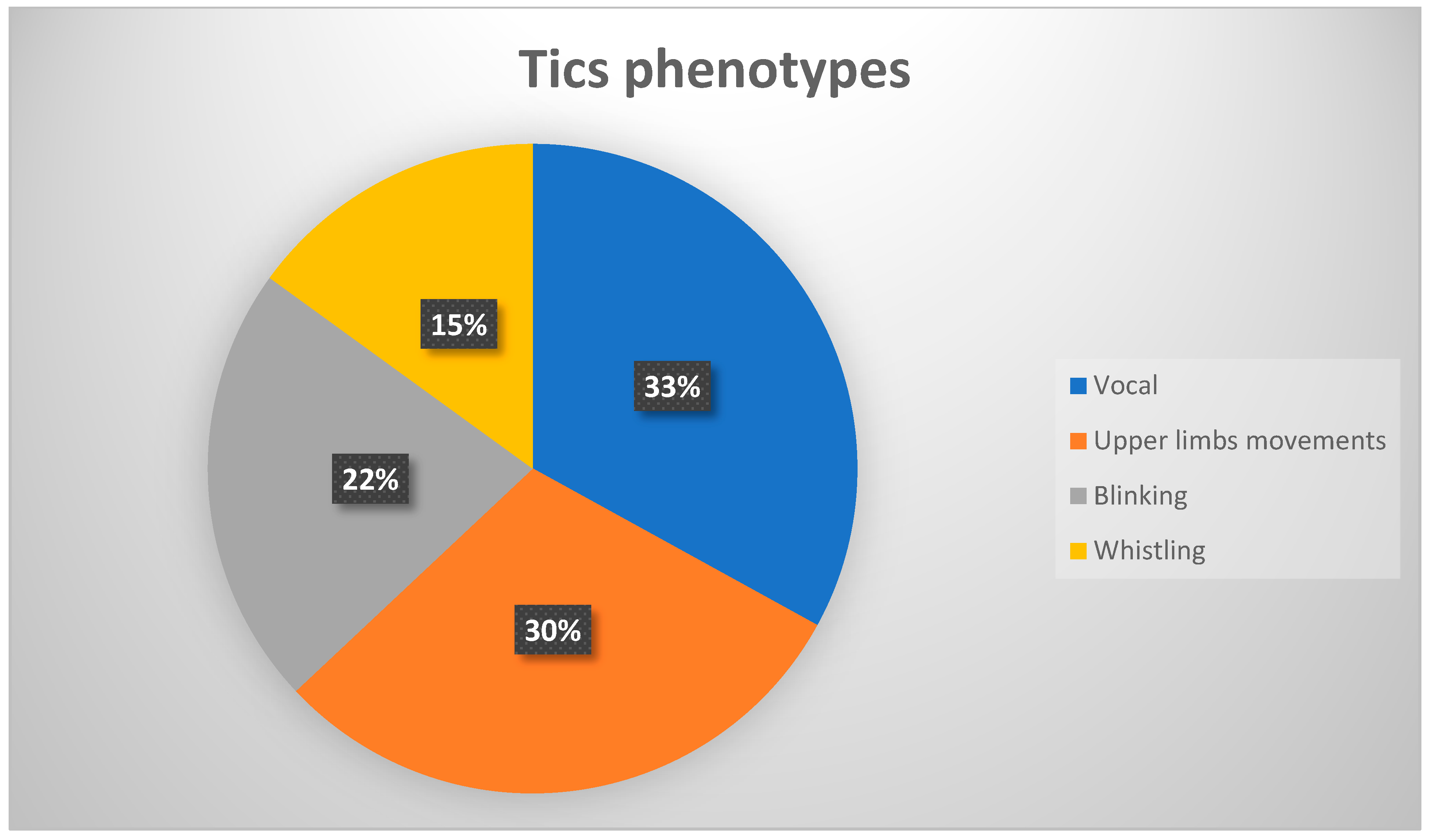
Despite different pathogenic causes evaluated [7], such as previous infections, immune impairment, or environmental factors, a precise triggering event is not always detectable. Defining criteria for PANS [8] is the presence of sudden-onset OCD or food restriction behavior together with at least two associated manifestations including behavioral disturbances (irritability, aggression, oppositional behavior), anxiety, depression, deterioration in school performance, and other neurologic signs like a motor or sensory impairment, sleep disturbances with enuresis, or incontinence (Figure 1 and Figure 2).
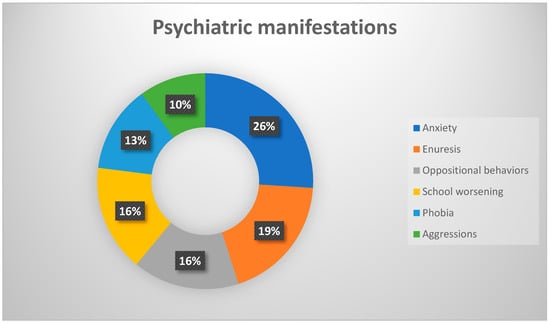
Figure 1.
Percentages frequency of the main psychiatric manifestations.
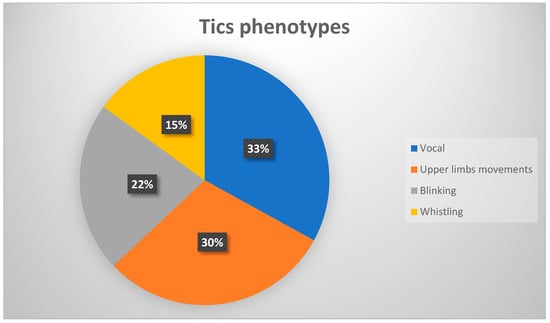
Figure 2.
Percentages frequency of the main tics phenotypes.
The pathogenic mechanism is now held responsible for an autoimmune impairment, consisting of molecular mimicry between antigens in the bacterial cell and antigens. Particularly, the production of autoantibodies has been shown against neurotransmitter receptors D1 or D2 of dopamine, responsible for neuropsychiatric impairment of these patients. Also, the presence of antibasal ganglia antibodies in sera of PANDAS patients is a highly specific and sensitive marker in comparison to control, nonaffected groups. Antineuronal antibodies that may contribute to the progression of PANDAS symptoms include antipyruvate kinase antibodies, antidopamine receptor antibody, and antilysoganglioside (GM1) antibody [9]. However, these findings seem not to be a specific marker of PANS, resulting also in Sydenham chorea or TS. A clear etiologic factor is not always detectable, so it is currently difficult to establish a standardized protocol to manage the treatment of these disorders. A recent PANS Conference Consensus proposed orienting the treatment according to the grade of symptoms.
These guidelines [10] suggest an initial approach based on antibiotics together with corticosteroids for the light and mild forms, while for more severe forms, an equally severe treatment as intravenous immunoglobulins infusion (IVIG) or plasmapheresis or biologic drugs such as rituximab can be required. Another possible approach taken into consideration is the surgical removal of tonsils. However, tonsillectomy has not shown any improvement in delaying the onset of symptoms or reducing the grade, as confirmed in a previous paper of Pavone et al. [11]. Complementary therapies to suppress the autoimmune response causing the symptoms consist of the administration of corticosteroids, therapeutic plasma exchange, intravenous immunoglobulin (IVIG), or rituximab [12]. Among these different options, a surely advised treatment is represented by IVIG. One of the first efficacy evaluations of this approach was performed in 1999 by Perlmutter’s team [13], who made a comparison between a subgroup of patients affected by OCD and tics treated with five single-volume plasma exchanges (PEX) and another treated with IVIG at 2 g/kg daily on two consecutive days, both compared to the same number of placebo controls; they noticed important clinical improvements either for tics or for OCD disturbances with good maintenance even 12 months after the treatment. Another study to evaluate the efficacy and safety of IVIG treatment in PANDAS children was performed by Williams et al. [14], who evaluated a group of 35 children rated as having a moderate–severe grade of OCD manifestations. The treatment was revealed to be well tolerated after a follow-up performed up to 24 months later. Further, other studies have been performed to assess the potential role of IVIG treatment for PANDAS patients, revealing a potential role in addition to antibiotics therapy [15] to prevent reinfection from etiologic agents, with a clear improvement of symptoms and prolonging the period of clinical wellness. Nevertheless, since dealing with an expensive procedure not lacking in risk, its application should be limited to those serious–severe cases, according to precise indications [16].
This study aims to assess the clinical features of the patients observed from different Italian cohorts, and to evaluate clinical response to IVIG treatment in children with an extreme severity grade of PANDAS/PANS disease. The outcome is to verify the decrease in tics and OCD using the Children’s Yale–Brown Obsessive–Compulsive Scale (CY-BOCS) severity score. We will consider a satisfactory clinical response to treatment a lowering of at least one degree of the CY-BOCS scale, which persists for at least one year of observation.
Besides, we conducted a review of disability and rehabilitation programs in PANS/PANDAS. We described the importance of rehabilitation, as occupational therapy, logopedic treatment and psychologic support, to prepare to recognize and help the child with PANS/PANDAS.
2. Materials and Methods
We enrolled a total of 55 patients with a diagnosis of PANS/PANDAS, who belonged to the extreme grade of disease and were never treated with IVIG/other therapy before, except for the antibiotic one. That group had been followed for one year since the onset of disease from our and other participating centers. Our sample (n = 55) was composed of 30 males (54%) and 25 females (46%), with an average age of 8.9 years old. At the diagnosis, all patients were assessed according to Swedo et al.’s (2012) parameters for PANS disorders [17]: abrupt, dramatic onset of OCD or eating restriction accompanied by equally abrupt and severe comorbid neuropsychiatric symptoms, which include anxiety, emotional lability, depression, irritability, aggression, oppositionality, deterioration in school performance, behavioral (developmental) regression, sensory amplification, movement abnormalities, sleep disturbance, and urinary frequency. Psychiatric manifestations were present in all patients, and all of them presented anxiety, enuresis, or oppositional behavior. OCD was present, especially in the form of food behavioral restrictions, and other psychiatric disturbances such as psychosis or self-harm were noticed as well. As concerns movement disturbances, instead, tics were the most frequent manifestation reported with wide variety among simple and complex forms, either vocal or motor type, while choreic movements were described in a smaller subgroup of patients (n = 4), with the main involvement of the upper limbs.
All of them were submitted to the CY-BOCS based on five items including questions on obsession and five items including questions on compulsion (0 to 4 points for each item); the items were administered by our team of psychologists who only enrolled the patients with a Total Severity Score ≥ 32 (Table 1). Presentation findings were assessed, including either clinical features (tics, chorea, OCD) or laboratory data (pharyngeal swab, antistreptolysin O titer, antideoxyribonuclease titer, Epstein–Barr virus, chlamydia, mycoplasma, toxoplasmosis, other infections, rubella, cytomegalovirus, and herpes simplex virus-2 and autoimmunity panel), together with brain MRI and EEG findings.

Table 1.
Total CY-BOCS score: range of severity for patients who have both obsessions and compulsion.
All patients enrolled were administered, after informing their parents about the possible although rare risks of allergic reactions and after having them sign the proper consent form, with the IVIG treatment at the dosage of 2 g/kg per day for two consecutive days.
Pavone et al. (2018) [18] reported on a study conducted in 34 children affected by PANDAS, all of them treated with an IVIG dosage of 2 g/kg day for two consecutive days, having a good final result with evident clinical improvement. Following this and previous studies that used the same dosage, we decided to follow the same posology.
The study protocol conformed to the ethical guidelines of the 1975 Declaration of Helsinki as revised in 2000 and was approved by the ethic committee of the University Hospital Vittorio Emanuele of Catania, Italy (nd 1394 04/04/2017).
For the review about rehabilitation feature and PANDAS, we searched PubMed for the following terms and keywords: “PANS/PANDAS” and “rehabilitation” and “PANS/PANDAS” and “disability”. The database searches yielded nine references from 2000 to 2020. Thus, only nine titles and abstracts were screened and then six remained for full-text screening; of these, three met the inclusion criteria.
3. Results
3.1. Clinical
In our study, we have analyzed a total of 55 extreme-grade PANS/PANDAS children, treated with IVIG at the dosage of 2 g/kg per day for two consecutive days. From 3 to 30 days after therapy in 47 out of 55 of the cases treated (85%), a long-term (1 yr) clinical benefit was noticed (n = 12 from extreme grade to no symptoms, n = 22 from extreme to mild, n = 9 from extreme to moderate, n = 4 from extreme to severe of CY-BOCS severity ratings), whereas in a minor percentage (15%, n = 8 cases), we noticed a reappearance of symptoms within 1 to 6 months after treatment, after an only temporary improvement (Table 2).

Table 2.
Distribution on severity scale after therapy (1–2 IVIG cycles) after 1 year of observation.
3.2. Laboratory
A positive result in pharyngeal swab was present in n = 37 (67%) children, versus a number of 14 (25%) negatives. In 4 (8%) cases, swab results were not available due to a laboratory error. ASO titer (measured in the whole sample) and Anti-DNase B titer (29/50 patients) provided a wide range of value, spanning from 296 to 2141 UI for ASO (982.7 ± 457.9 UI), and from 299 to 1160 UI (539.8 ± 214.1 U/mL) for DNase B. The discordance from the values here obtained and those obtained in other centers could be ascribable to the different bacterial stains involved. All the other laboratory parameters searched were negative and not worth further study. EEG and ECG were normal in all patients analyzed with not-pathological waves to the tracks, and MRI data revealed anomalies, such as bilateral enlargement in perioptic sheath in one case and mild asymmetry of the lateral ventricles in two patients; both findings had no clinical relevance and no correlation with the pathology.
All patients, affected from clinical manifestation defined nonresponsive to antimicrobial treatment, were administered intravenous immunoglobulin (IVIG) treatment at 2 g/kg per day for two consecutive days.
3.3. Other Findings
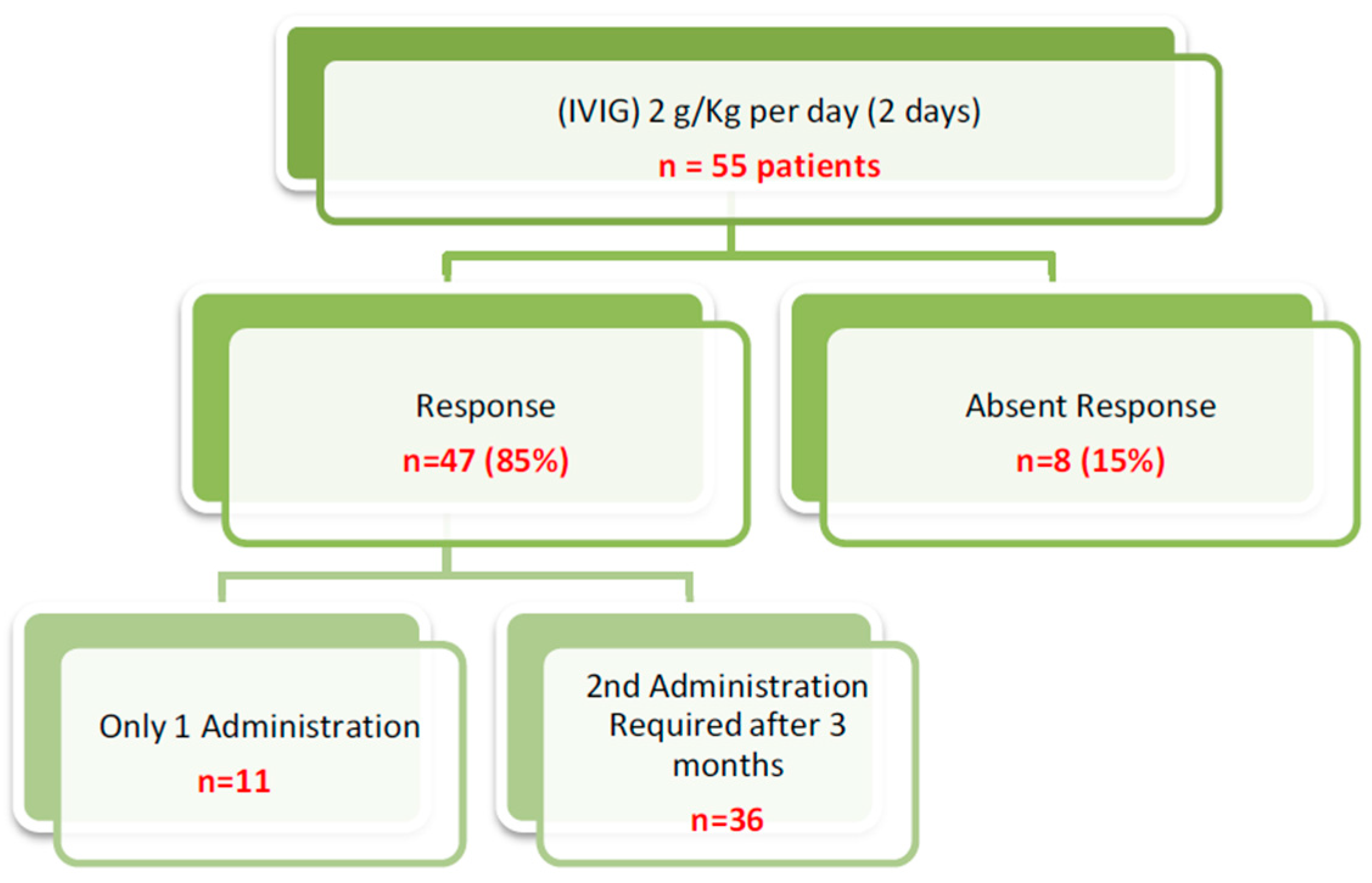
What comes out from our study is a noticeable improvement (transition from extreme symptom grade to lower grade) in 47 out 55 children, and in some cases (12 out 47), a complete remission of symptoms for at least one year in observed children. A total of 11 patients (4 males and 7 females) out of the 47 responders required only one cycle of IVIG therapy (2 g/kg per day for two consecutive days), showing clear clinical improvement of symptoms with no further relapse, whereas in the remaining 36 patients, a second cycle led to clinical well-being for at least one year as in the following months they had manifested resumption of symptoms. In a small percentage (15%, n = 8 cases) after an initial response, symptoms reappeared requiring a third further cycle one year after the first administration, without obtaining a significant result. Of the eight nonresponder cases, five also had temporo-mandibular joint disorders. We applied exercises that consisted of 10 repetitions, twice a day, of protrusion–retrusion of the mandible with a spacer (cotton roll) between the incisors and lateralization of the jaw with a spacer between the canines. After a month of exercises, the painful symptoms improved in all of them along with the stress deriving from this condition. This was one of the reasons that prompted us to carry out a small review on rehabilitation therapies, a weapon often underestimated but that can be useful as a support to other therapies (Figure 3).
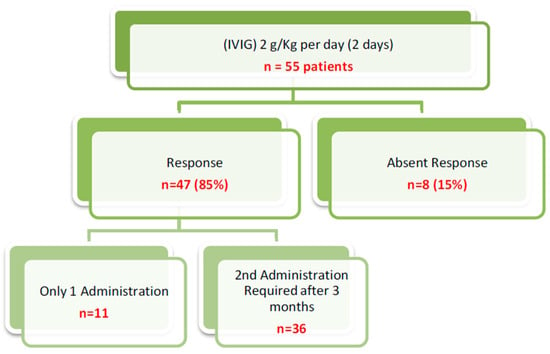
Figure 3.
Outcome after administration of IVIG therapy.
4. Discussion
PANDAS are possible post-infectious disorders of pediatric age in which, after a previous pharyngeal infection of GABHS, children present with sudden-onset symptoms including behavioral disturbances together with movement disorders such as tics or OCD, with possible relapsing–remitting clinical course, consequent to new infective processes [1,2,3,4]. Similarly to Sydenham chorea [17], another condition related to GABHS infection, it is universally thought that in both conditions, a molecular mimicry between streptococcal and cellular antigens leads to an antibody production that would be responsible for the clinical manifestation, so representing the previous GABHS (or from other etiological agents, as in PANS) as a mere trigger for an autoimmune process. Sometimes the symptomatology may retrace the typical Tourette syndrome phenotype, at the point that precise differential diagnosis between the two forms is often difficult, and so leading some authors to consider the two conditions a different manifestation of the same disease. This pathogenetic hypothesis is nowadays limited by the lack of evidence regarding etiological causes of TS, nevertheless, a possible interaction has been observed by Spinello et al. [19], who documented that after a repeated GABHS infection, an immune response was generated which was associated with neurological and behavioral phenotypes similar to Tourette syndrome. Considering the wide heterogeneity of clinical manifestations and the possibility of overlap with any other neuropsychiatric disturbance, reaching the diagnosis of PANS/PANDAS can represent a real clinical challenge, requiring the support of biochemical and laboratory assays, together with complementary instrumental surveys. In the specific case of our sample, we selected a cluster of extreme-grade patients whose clinical manifestations were composed of a wide range of tics movements, together with equally severe OCD subtypes, including suicidal behavior. Quite informative have been, in the same way, laboratory assays, which gave positive results for cultural pharyngeal swabs in 58% of cases (n = 32 out 55 patients), and negative in 14 cases in which false negativity could not be excluded due to previous antibiotics treatment assumed shortly before the patient was placed in our study.
Varying from literature evidence that admits the presence of abnormal brain activity in EEG in PANS/PANDAS patients (together with obstructive apnea syndrome, sleeping disturbances, or parasomnias) [20], in our sample, EEG pattern was normal in nearly all the observed patients.
The MRI had no significant result either; the isolated anomalies revealed in the three cases observed are probably not related to symptomatology, as in the literature there are no comparisons between the abnormalities found and the clinical finding of PANS/PANDAS. Despite morphologic imaging, a better comprehension of neuropathogenesis of clinical features could be provided by functional imaging assays. An initial approach was provided by Citak et al. [21], who performed an HMPAO-SPECT (hexamethyl propylene amine oxime single-photon missions computed tomography) on a group of patients affected by Sydenham chorea or PANDAS, revealing a hypoperfusion pattern, with special involvement of thalamus or basal ganglia (striatum); a further contribution in explaining the pathophysiology of behavioral and movement impairment of these patients come from Kumar’s [22] team, who demonstrate the presence of a neuroinflammation pattern involving bilaterally lentiform nuclei and caudate nuclei in observed cases and not in controls, suggesting that this distribution pattern of neuroinflammation could be related to the clinical manifestation.
Administering the first cycle of IVIG therapy has already given us good results 3–30 days after administration. It is very important to underline that in cases of symptomatology reappearing within the year, administering the second course of therapy was important and led us to obtain a lasting improvement in symptoms (at least one year). The importance of the second cycle after the failure of the first is, therefore, an important aspect to consider. Administering the third cycle to those who did not respond to the second one did not lead us to obtain relevant clinical results, at which point we used other support therapies, such as rehabilitation, helping the patient to obtain a slight improvement in some aspects.
As regards therapeutic approaches, as support to accompany the IVIG treatment, as an initial option to control neuropsychiatric disturbances, an initial approach would consist of cognitive behavior therapy together with pharmacologic support with selective serotonin reuptake inhibitors (SSRIs) [23] for treating OCD manifestations, while for tic disorders, initial behavioral interventions are similarly recommended together with psychopharmacological treatments [24]. Nevertheless, after an initial high percentage of response, most patients showed lifelong treatment resistance [20], following the autoimmune etiology suspected, thus requiring in most cases the adoption of other therapeutic strategies.
Besides standard psychiatric treatments, an alternative therapy could include a treatment/prevention approach consisting of antibiotics or tonsillectomy [19] to prevent the recurrence of GABHS infections.
Considering the evidence that emerged from the literature and the intrinsic limitations of our study, such as the lack of a control group and a too-small period of follow-up, we cannot affirm a sure conclusion about the efficacy of IVIG treatment in PANDAS. In our opinion, anyway, it represents a safe and effective accessory approach in the clinical management of patients with serious–severe type of disturbances, agreeing with the PANS Research Consortium (PRC) [25]. Tics, movement disorders such as motoric hyperactivity, dysgraphia, speech disorders, and OCD are the main causes of disability in many aspects of daily living [26]. The performance skills in activities of daily living (ADL) of the patients, like personal hygiene and grooming, bathing, extracurricular activities, and participation in all aspects of leisure activities are other features that the physician team must evaluate.
Pharmacologic treatment is the most common treatment for the disorder. The rehabilitation could be an added value. Several studies highlight the importance of different therapeutic approaches, but without a specific description [27,28]. In patients with an abnormal neurological examination, muscle weakness, abnormal reflexes, or chorea, a further workup is indicated. In these cases, an individualized rehabilitation treatment could avoid repetitive and nonrhythmic motor movements. In the study of Cocuzza et al. [29], PANDAS patients with temporomandibular joint disorders have undergone rehabilitation treatment. The exercises were explained in the hospital to correctly execute them, then the patients executed mirror exercises at home. The exercises consisted of 10 repetitions, twice a day, of protrusion–retrusion of the mandible with a spacer (cotton roll) between the incisors, lateralization of the jaw with a spacer between the canines positioned homolaterally to the direction of the lateral displacement, maximum opening and closing of the mouth. The four PANDAS patients with oromandibular dystonia achieved an improvement of the algic symptoms through the self-rehabilitation program, with a reduction of the hyperkinetic movements. No disturbances or worsening have been reported after the self-rehabilitation treatment was performed. Sokol [30] described eating compulsions, as an OCD, in four patients. He supposed a temporal relationship between antibiotic treatment and decreased eating disorder symptoms. According to his research, the compulsions progressively decreased with antibiotics and with rehabilitation treatment.
Tona et al. [31] described activities of daily living, math, handwriting, extracurricular activities, free play, organized sports, community and family social participation, higher-level thinking, attention, memory, sequencing, emotional coping, and energy and drive were commonly affected during exacerbations. During exacerbations, children often required assistance and adaptation to remain functional or were unable to function at a typical level.
A personalized rehabilitation program could be applied to subjects with this disorder to reduce pain, intensity and number of hyperkinetic movements, consequent awareness, and disability in ADL. Obsessive–compulsive disorders, included eating disorders, could be treated with individual therapy, group counseling and skills training, cognitive behavioral therapy, psychodynamic therapy, mindfulness-based approaches, and yoga therapy. Occupational therapy, introduced early, improves fine and gross motor skills and motor planning.
5. Conclusions
During our study, we have seen how the proposed IVIG treatment has benefited our patients, although not all of them, being in line with the studies already present in the literature from which we took inspiration and used the same immunoglobulin dosages.
Considering the variability of a disease still under study, we have taken as a good result the simple clinical improvement of at least one degree of decrease (according to the CY-BOCS), lasting at least one year. Considering the great impairment during their lives that extreme-grade patients have, moving to a lower grade brings enormous improvements in daily living. Considering the heterogeneity of the studied group, the short observation time, and the still unclear pathogenic mechanisms of PANS/PANDAS, we cannot establish the correct definitive therapy; however, the encouraging data from this and other studies can tell us that we are not far from a better understanding of this disease.
Surely, more precise knowledge about the pathogenic mechanisms at the basis of neuropsychiatric impairment involved in these disturbances could help researchers to identify more effective and shared therapeutic approaches and confirm the accuracy of IVIG treatment. However, from the review, we did try to create a clear idea on the importance of rehabilitation therapy; it is clear that this therapy is eligible to be an added value of proven utility associated with drug and other therapy in PANS/PANDAS. A rehabilitation program must be tailored to a child’s specific needs, and the exercises must build specific weak skills. Personalized rehabilitation training could improve the movement disorders of patients with PANS/PANDAS.
Author Contributions
Conceptualization, G.C., R.F., and P.P.; methodology, A.S., A.F.; investigation, R.C., R.L.; data curation, G.M.; L.S., S.S.; writing—original draft preparation, P.P., G.C., C.O., M.G.P.; writing—review and editing, S.C., A.M., L.M., M.V.; supervision, P.P., M.V. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding
Acknowledgments
The authors would like to thank AME (USA) Editor American manuscript Editors, for editing the manuscript.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Swedo, S.E.; Leonard, H.L.; Garvey, M.; Mittleman, B.; Allen, A.J.; Perlmutter, S.; Lougee, L.; Dow, S.; Zamkoff, J.; Dubbert, B.K. Pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections: Clinical description of the first 50 cases. Am. J. Psychiatry. 1998, 155, 264–271. [Google Scholar] [CrossRef]
- Frankovich, J.; Thienemann, M.; Pearlstein, J.; Crable, A.; Brown, K.; Chang, K. Multidisciplinary clinic dedicated to treating youth with pediatric acute-onset neuropsychiatric syndrome: Presenting characteristics of the first 47 consecutive patients. J. Child Adolesc. Psychopharmacol. 2015, 25, 38–47. [Google Scholar] [CrossRef]
- Swedo, S.E.; Seidlitz, J.; Kovacevic, M.; Latimer, M.E.; Hommer, R.; Lougee, L.; Grant, P. Clinical presentation of pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections in research and community settings. J. Child Adolesc. Psychopharmacol. 2015, 25, 26–30. [Google Scholar] [CrossRef]
- Macerollo, A.; Martino, D. Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal Infections (PANDAS): An Evolving Concept. Tremor Other Hyperkinet Mov. 2013, 3. [Google Scholar] [CrossRef]
- Pavone, P.; Parano, E.; Rizzo, R.; Trifiletti, R.R. Autoimmune neuropsychiatric disorders associated with streptococcal infection: Sydenham chorea, PANDAS, and PANDAS variants. J. Child Neurol. 2006, 21, 727–736. [Google Scholar] [CrossRef] [PubMed]
- Singer, H.S.; Gilbert, D.L.; Wolf, D.S.; Mink, J.W.; Kurlan, R. Moving from PANDAS to CANS. J. Pediatr. 2012, 160, 725–731. [Google Scholar] [CrossRef] [PubMed]
- Cunningham, M.W. Post-streptococcal autoimmune sequelae: Rheumatic fever and beyond. In Streptococcus Pyogenes: Basic Biology to Clinical Manifestations; Ferretti, J.J., Stevens, D.L., Fischetti, V.A., Eds.; University of Oklahoma Health Sciences Center: Oklahoma City, OK, USA, 2016. [Google Scholar]
- Swedo, S.E.; Leckman, J.; Rose, N. From research subgroup to clinical syndrome: Modifying the PANDAS criteria to describe PANS (pediatric acute-onset neuropsychiatric syndrome). Pediatr. Ther. 2012, 2, 1–8. [Google Scholar] [CrossRef]
- Baj, J.; Sitarz, S.; Forma, A.; Wròblewska, K.; Juchnowicz, H.K. Alterations in the Nervous System and Gut Microbiota after B-Hemolytic Streptococcus Group A Infection—Characteristics and Diagnostic Criteria of PANDAS Recognition. Int. J. Mol. Sci. 2020, 21, 1476. [Google Scholar] [CrossRef] [PubMed]
- Chang, K.; Frankovich, J.; Cooperstock, M.; Cunningham, M.W.; Latimer, M.E.; Murphy, T.K.; Pasternack, M.; Thienemann, M.; Williams, K.; Walter, J.; et al. PANS Collaborative Consortium. Clinical evaluation of youth with pediatric acute-onset neuropsychiatric syndrome (PANS): Recommendations from the 2013 PANS Consensus Conference. J. Child Adolesc. Psychopharmacol. 2015, 3–13. [Google Scholar] [CrossRef]
- Pavone, P.; Rapisarda, V.; Serra, A.; Nicita, F.; Spalice, A.; Parano, E.; Rizzo, R.; Maiolino, L.; Di Mauro, P.; Vitaliti, G.; et al. Pediatric autoimmune neuropsychiatric disorder associated with group a streptococcal infection: The role of surgical treatment. Int. J. Immunopathol. Pharm. 2014, 27, 371–378. [Google Scholar]
- Sigra, S.; Hesselmark, E.; Bejerot, S. Treatment of PANDAS and PANS: A systematic review. Neurosci. Biobehav. Rev. 2018, 86, 51–65. [Google Scholar] [CrossRef] [PubMed]
- Perlmutter, S.J.; Leitman, S.F.; Garvey, M.A.; Hamburger, S.; Feldman, E.; Leonard, H.L.; Swedo, S.E. Therapeutic plasma exchange and intravenous immunoglobulin for obsessive-compulsive disorder and tic disorders in childhood. Lancet 1999, 354, 1153–1158. [Google Scholar] [CrossRef]
- Williams, K.A.; Swedo, S.E.; Farmer, C.A.; Grantz, H.; Grant, P.J.; D’Souza, P.; Hommer, R.; Katsovich, L.; King, R.A.; Leckman, J.F. Randomized, Controlled Trial of Intravenous Immunoglobulin for Pediatric Autoimmune Neuropsychiatric Disorders Associated with Streptococcal Infections. J. Am. Acad. Child Adolesc. Psychiatry. 2016, 55, 860–867. [Google Scholar] [CrossRef] [PubMed]
- Kovacevic, M.; Grant, P.; Swedo, S.E. Use of intravenous immunoglobulin in the treatment of twelve youths with pediatric autoimmune neuropsychiatric disorders associated with streptococcal infections. J. Child Adolesc. Psychopharmaco 2015, 25, 65–69. [Google Scholar] [CrossRef] [PubMed]
- Frankovich, J.; Swedo, S.; Murphy, T. Clinical Management of Pediatric Acute-Onset Neuropsychiatric Syndrome: Part II-Use of Immunomodulatory Therapies. J. Child. Adolesc. Psychopharmacol. 2017, 27, 574–593. [Google Scholar] [CrossRef]
- Swedo, S.E.; Leonard, H.L.; Schapiro, M.B.; Casey, B.J.; Mannheim, G.B.; Lenane, M.C. Sydenham’s chorea: Physical and psychological symptoms of St Vitus dance. Pediatrics 1993, 91, 706–713. [Google Scholar] [PubMed]
- Pavone, P.; Falsaperla, R.; Nicita, F.; Zecchini, A.; Battaglia, C.; Spalice, A.; Iozzi, L.; Parano, E.; Vitaliti, G.; Verrotti, A.; et al. Pediatric Autoimmune Neuropsychiatric Disorder Associated with Streptococcal Infection (PANDAS): Clinical Manifestations, IVIG Treatment Outcomes, Results from a Cohort of Italian Patients. NNBN 2018, 8, 854–860. [Google Scholar] [CrossRef]
- Spinello, C.; Laviola, G.; Macri, S. Pediatric Autoimmune Disorders Associated with Streptococcal Infections and Tourette’s Syndrome in Preclinical Studies. Front. Neuroendocrinol. 2016, 10, 310. [Google Scholar] [CrossRef]
- Hommer, R.E.; Buckley, A.; Swedo, S.E. New onset sleep disturbances and PSG findings in children with acute or subacute neuro-psychiatric changes. In Abstract Submitted to 2014 AACAP Annual Meeting; AACAP: Washington, DC, USA, 2014. [Google Scholar]
- Citak, E.C.; Gucuyener, K.; Karabacak, N.I.; Serdaroğlu, A.; Okuyaz, C.; Aydin, K. Functional brain imaging in Sydenham’s chorea and streptococcal tic disorders. J. Child. Neurol. 2004, 19, 387–390. [Google Scholar] [CrossRef]
- Kumar, A.; Williams, M.T.; Chugani, H.T. Evaluation of basal ganglia and thalamic inflammation in children with pediatric autoimmune neuropsychiatric disorders associated with streptococcal infection and Tourette syndrome: A positron emission tomographic (PET) study using 11C-[R]-PK11195. J. Child Neurol. 2015, 30, 749–756. [Google Scholar] [CrossRef]
- Pediatric OCD Treatment Study (POTS) Team. Cognitive-behavior therapy, sertraline, and their combination for children and adolescents with obsessive-compulsive disorder: The Pediatric OCD Treatment Study (POTS) randomized controlled trial. JAMA 2004, 292, 1969–1976. [Google Scholar] [CrossRef] [PubMed]
- Hollis, C.; Pennant, M.; Cuenca, J.; Glazebrook, C.; Kendall, T.; Whittington, C.; Stockton, S.; Larsson, L.; Bunton, P.; Dobson, S.; et al. Clinical effectiveness and patient perspectives of different treatment strategies for tics in children and adolescents with Tourette syndrome: A systematic review and qualitative analysis. Health Technol. Assess. Rep. 2016, 20, 1–450. [Google Scholar] [CrossRef] [PubMed]
- Grant, J.E. Clinical practice: Obsessive-compulsive disorder. N. Engl. J. Med. 2014, 371, 646–653. [Google Scholar] [CrossRef] [PubMed]
- Murphy, T.K.; Kurlan, R.; Leckman, J. The immunobiology of Tourette’s disorder, pediatric autoimmune neuropsychiatric disorders associated with Streptococcus, and related disorders: A way forward. J. Child Adolesc. Psychopharmacol. 2010, 20, 317–331. [Google Scholar] [CrossRef] [PubMed]
- Gamucci, A.; Uccella, S.; Sciarretta, L.; D’Apruzzo, M.; Calevo, M.G.; Mancardi, M.M.; Veneselli, E.; De Grandis, E. PANDAS and PANS: Clinical, Neuropsychological, and Biological Characterization of a Monocentric Series of Patients and Proposal for a Diagnostic Protocol. J. Child Adolesc. Psychopharmacol. 2019, 29, 305–312. [Google Scholar] [CrossRef]
- Okumura, R.; Yamazaki, S.; Ohashi, T.; Magara, S.; Tohyama, J.; Sakuma, H.; Hayashi, M.; Saitoh, A. Neuropsychiatric Disorder Associated with Group G Streptococcus Infection. Case. Rep. Pediatr. 2018, 23. [Google Scholar] [CrossRef]
- Cocuzza, S.; Marino, S.; Gulino, A.; Pustorino, E.; Murabito, P.; Maniaci, A.; Sabino, L.; Taibi, R.; Di Luca, M.; Falsaperla, R.; et al. ENT involvement and orobuccal movements’ disorders in Pandas patients: Assessment and rehabilitations tools. Eur. Rev. Med. Pharm. Sci. 2019, 23, 4110–4117. [Google Scholar]
- Sokol, M.S. Infection-triggered anorexia nervosa in children: Clinical description of four cases. J. Child Adolesc. Psychopharmacol. 2000, 10, 133–145. [Google Scholar] [CrossRef]
- Tona, J.T.; Bhattacharjya, S.; Calaprice, D. Impact, of PANS and PANDAS exacerbations on occupational performance: A mixed-methods study. Am. J. Occup. Ther. 2017, 71, 7103220020P1–7103220020P9. [Google Scholar] [CrossRef]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).