1921–2021: A Century of Renewable Ammonia Synthesis
Abstract
:1. Introduction
2. Early 1920s: Development and Small-Scale Technology
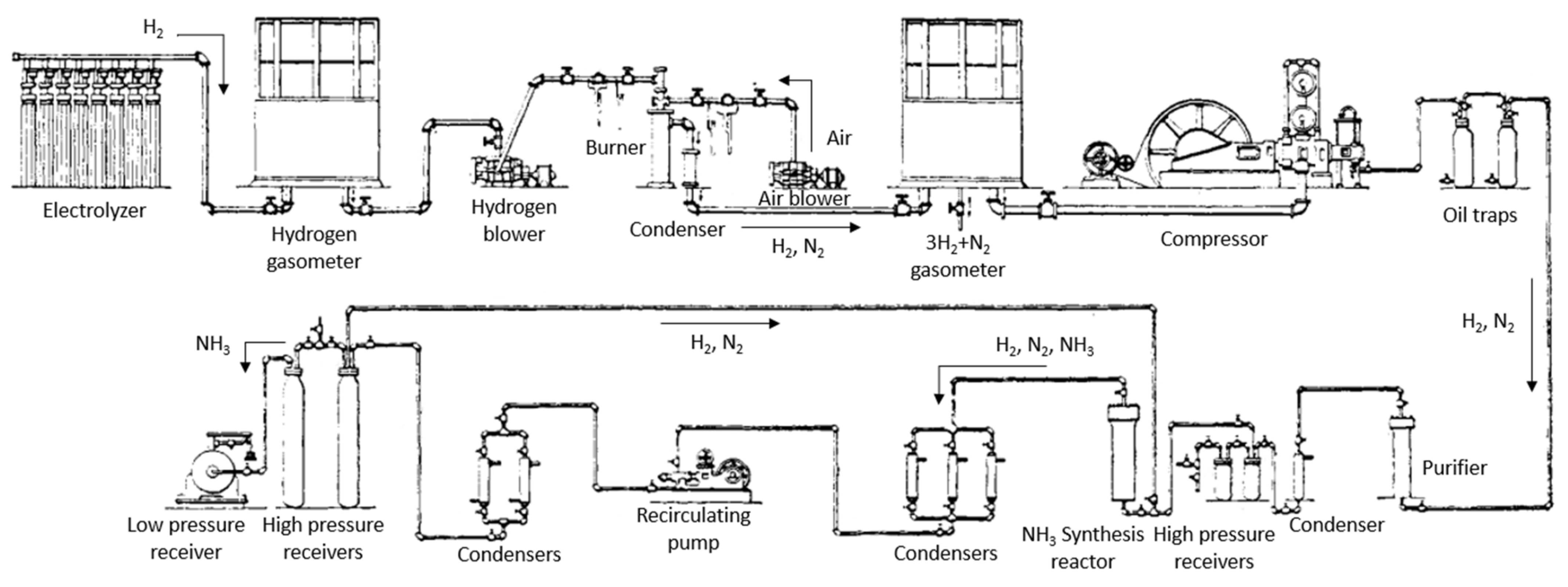
2.1. Renewable Ammonia Synthesis Technology
2.2. Italy
2.3. Other European Countries
2.4. Japan
2.5. Canada
2.6. United States
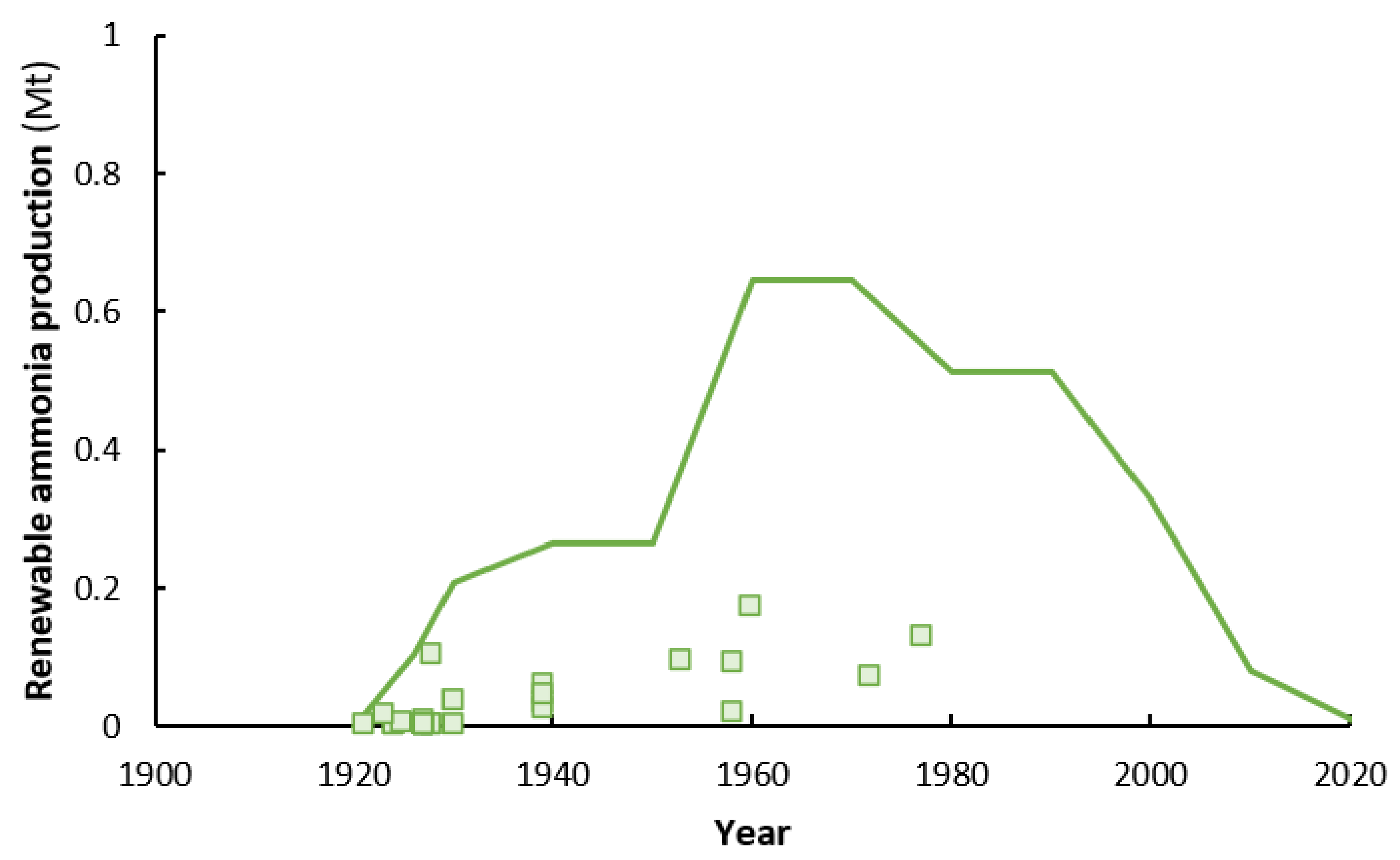
3. Late 1920s–1960s: Scale-Up of Renewable Ammonia and Competition from Fossil Technology
3.1. Norway
3.2. Egypt
3.3. India
3.4. Peru
3.5. Zimbabwe
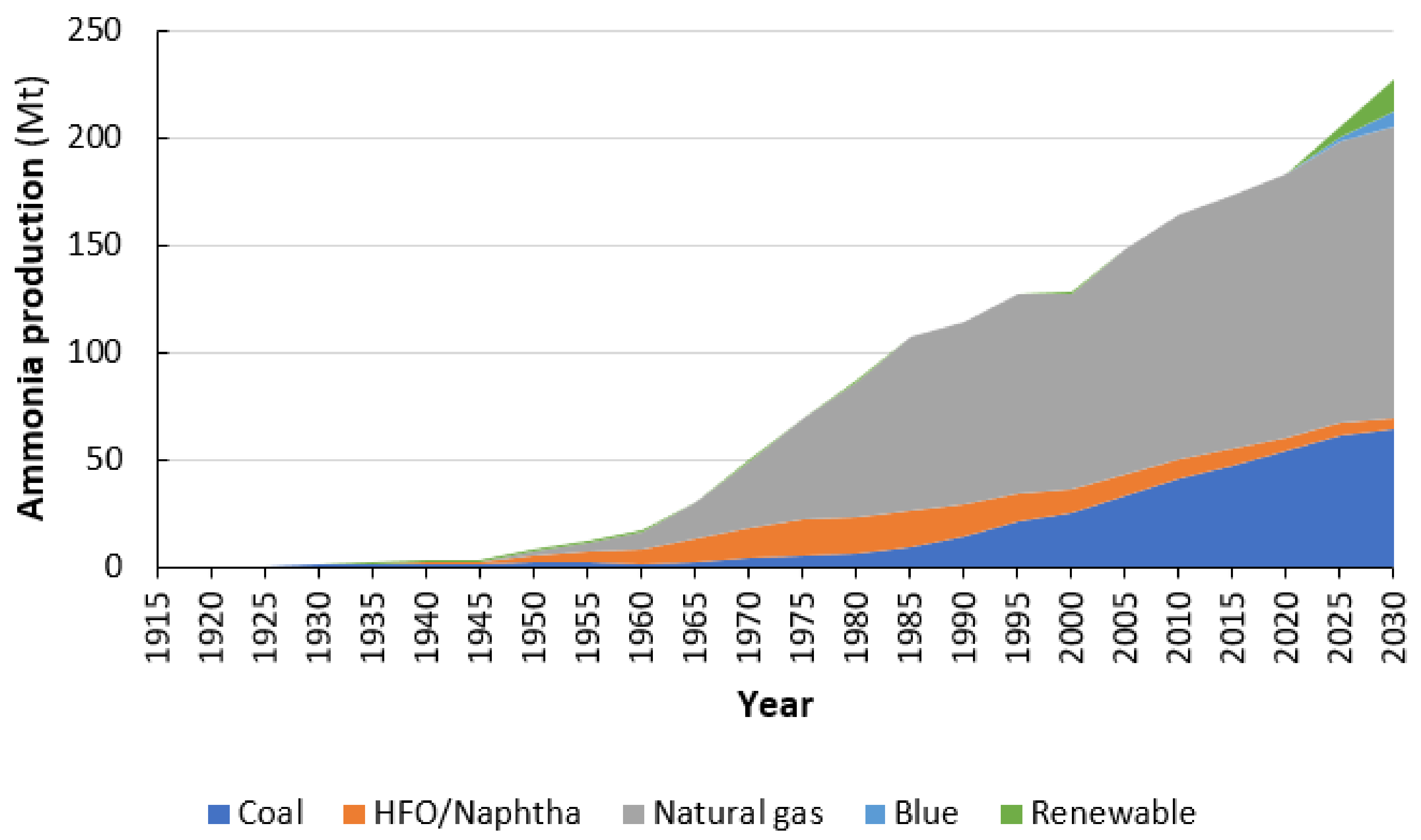
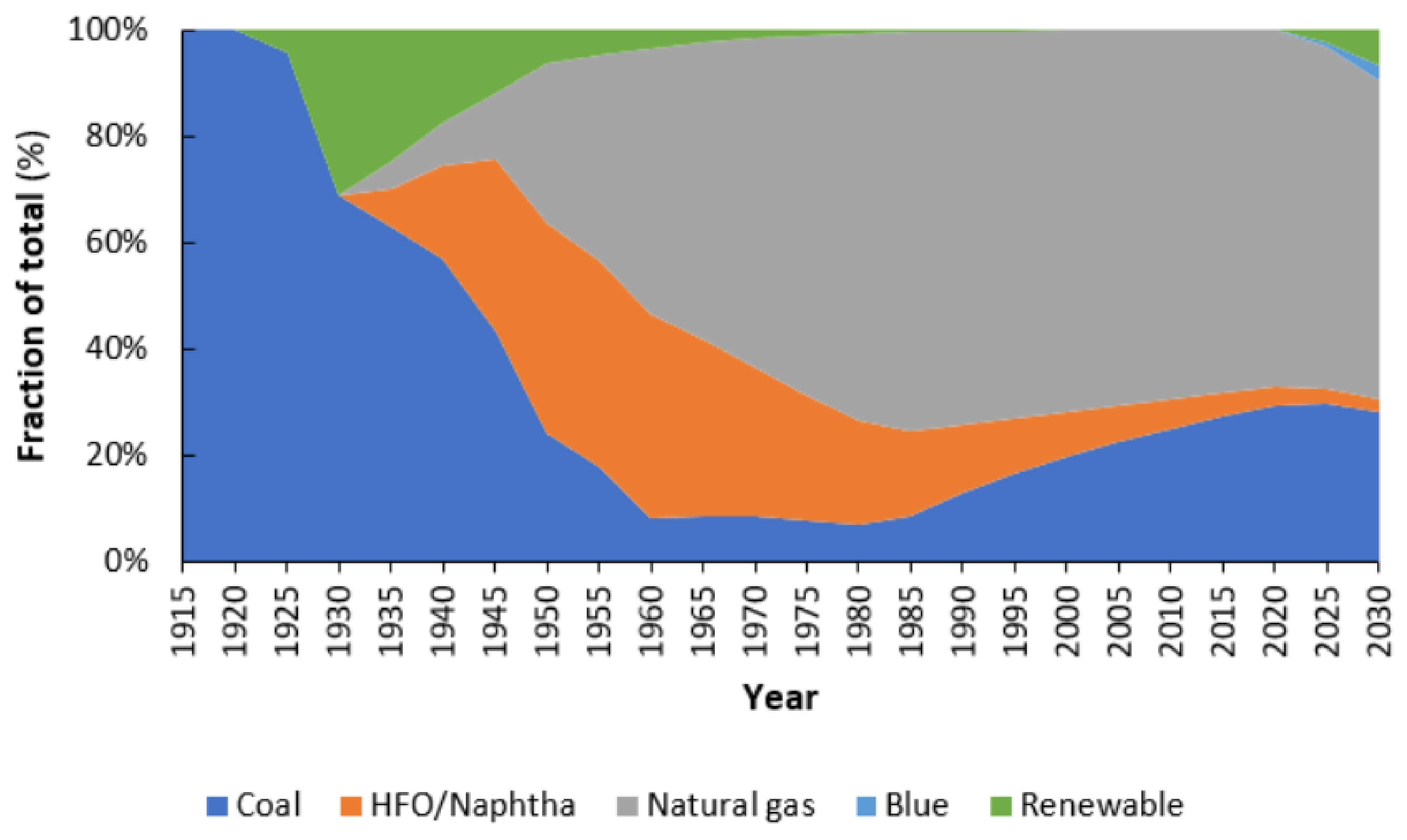
4. 1960s–2021: Natural Gas Outcompetes Renewable Ammonia Production on a Large Scale
- Technology improvements for fossil-based hydrogen production, especially for natural gas-based hydrogen production (Section 4.1);
- Cost reductions and availability of fossil-based feedstocks, especially natural gas (Section 4.2);
- Better cost-scaling of fossil fuel-based technologies (Section 4.3);
- Globalization of the fertilizer trade (Section 4.4).
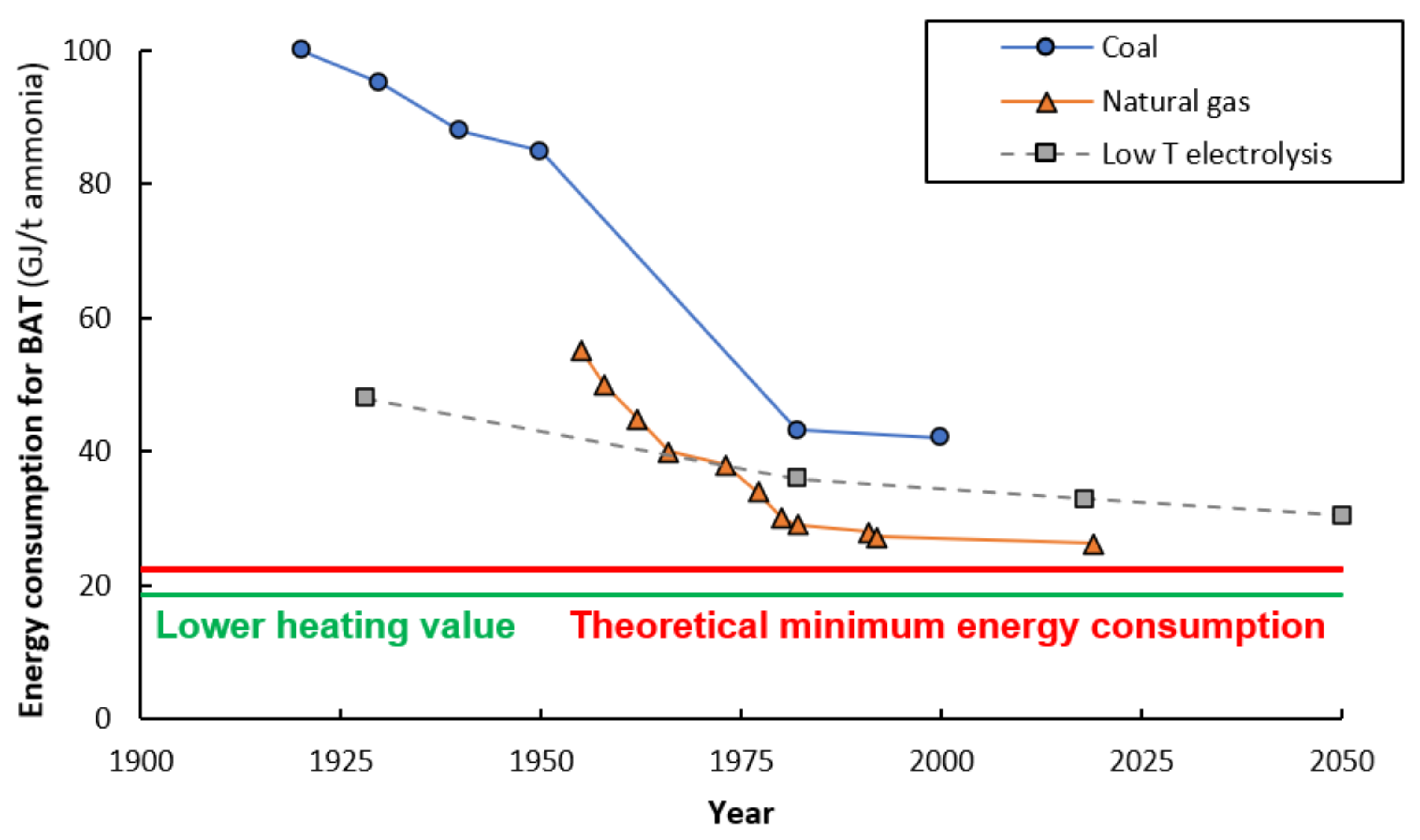
4.1. Technology Improvements for Fossil Fuel-based Hydrogen Production
4.2. Cost Reductions of Fossil Feedstocks
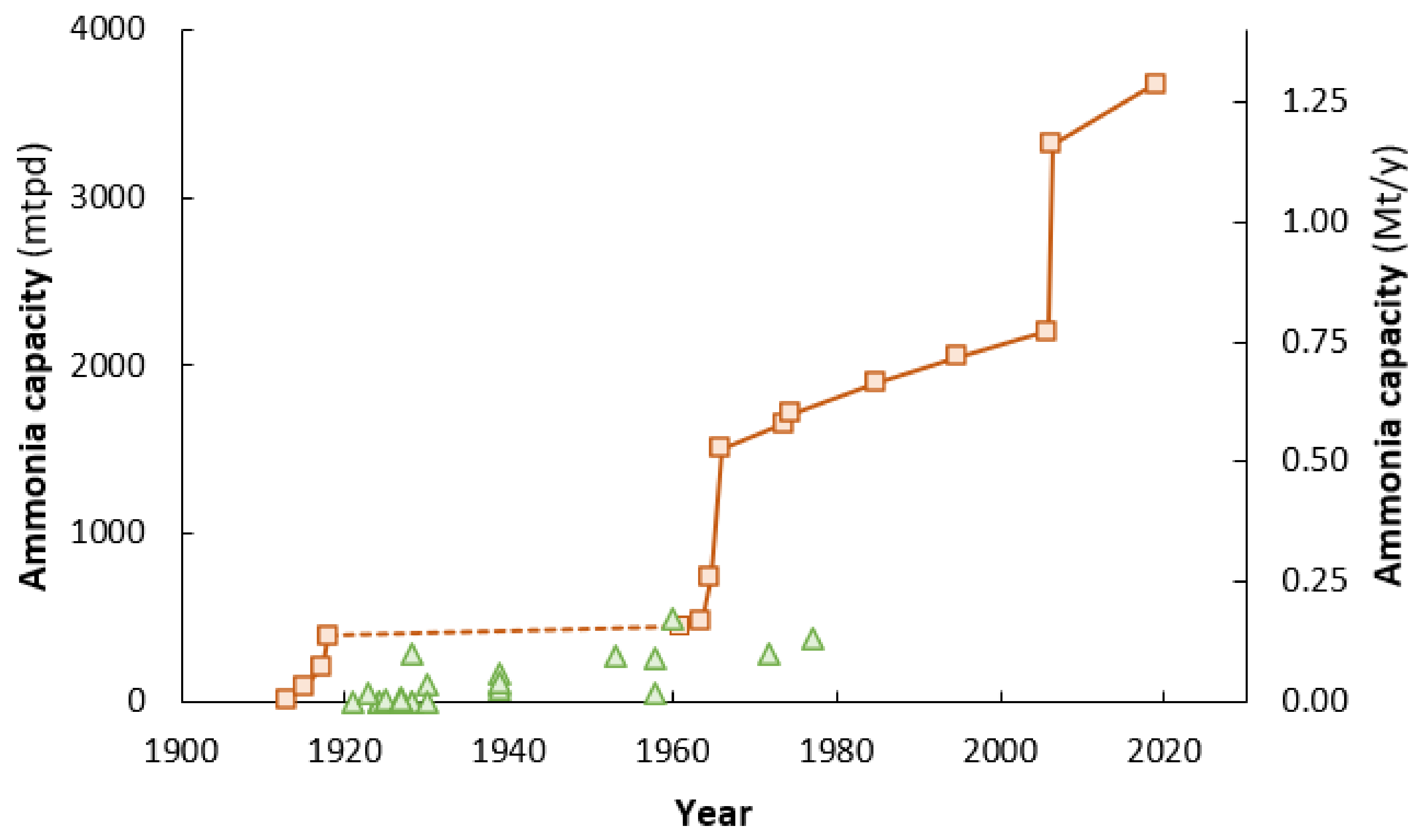
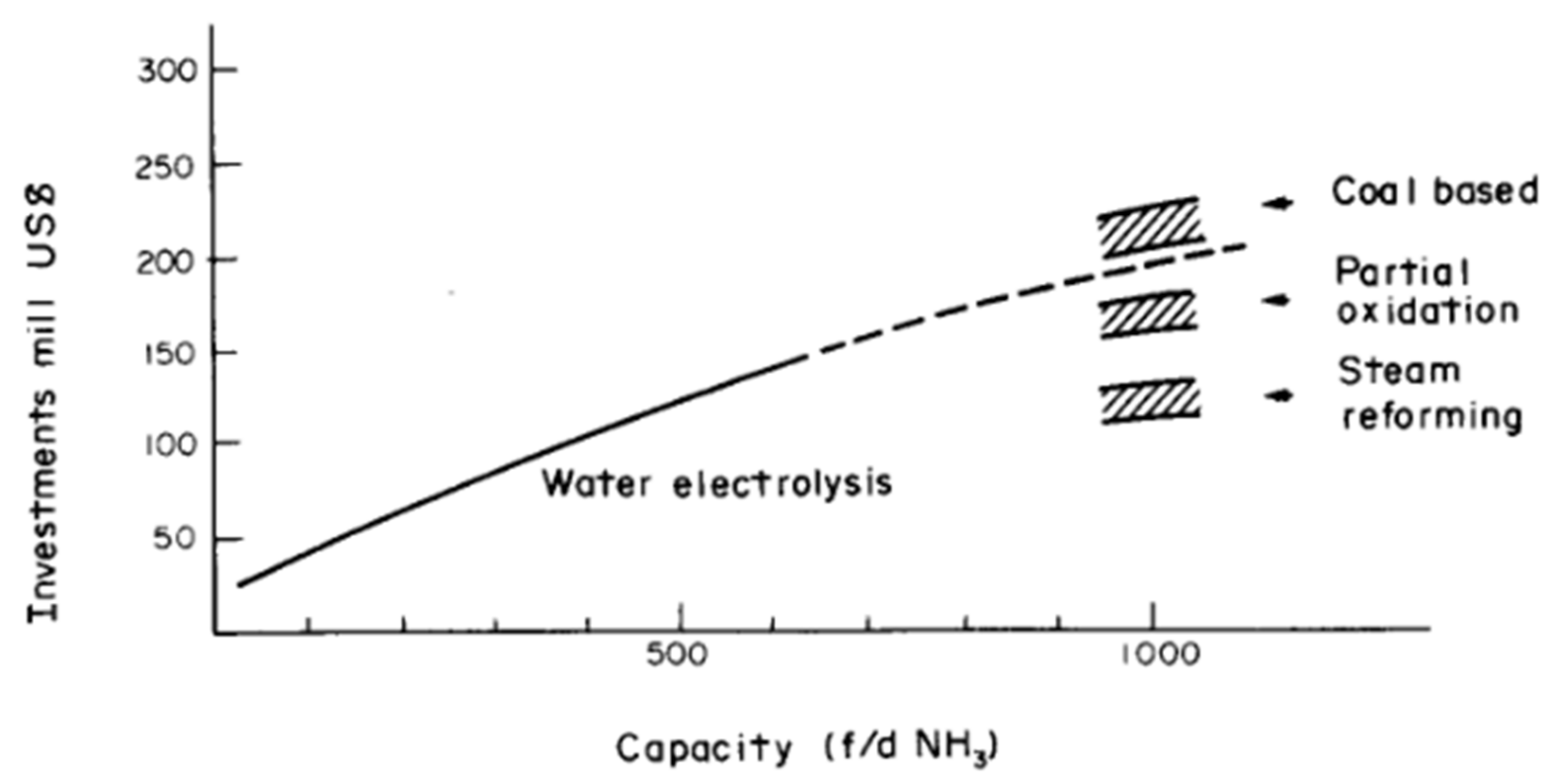
4.3. Better Cost-Scaling of Fossil Fuel-Based Technologies
4.4. Globalization of the Fertilizer Trade
5. 2021 and Beyond: Renewed Interest in Renewable Ammonia
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Erisman, J.W.; Sutton, M.A.; Galloway, J.; Klimont, Z.; Winiwarter, W. How a century of ammonia synthesis changed the world. Nat. Geosci. 2008, 1, 636–639. [Google Scholar] [CrossRef]
- Smil, V. Detonator of the population explosion. Nature 1999, 400, 415. [Google Scholar] [CrossRef]
- Smil, V. Enriching the Earth: Fritz Haber, Carl Bosch, and the Transformation of World Food Production; MIT Press: Cambridge, MA, USA, 2004. [Google Scholar]
- Liu, H. Ammonia synthesis catalyst 100 years: Practice, Enlightenment and Challenge. Chin. J. Catal. 2014, 35, 1619–1640. [Google Scholar] [CrossRef]
- Rouwenhorst, K.H.R.; Krzywda, P.M.; Benes, N.E.; Mul, G.; Lefferts, L. Ammonia Production Technologies. In Techno-Economic Challenges of Green Ammonia as Energy Vector; Bañares-Alcántara, R., Valera-Medina, A., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2020; pp. 41–84. [Google Scholar] [CrossRef]
- Hatfield, O. A Review of Global Ammonia Supply. 2020. Available online: https://www.ammoniaenergy.org/wp-content/uploads/2020/12/Oliver-Hatfield.pdf (accessed on 19 March 2021).
- International Renewable Energy Agency (IRENA). Ammonia Energy Association. In Production, Market Status and Future Prospects of Renewable Ammonia; IRENA: Masdar City, United Arab Emirates, 2022. [Google Scholar]
- The Royal Society. Ammonia: Zero-Carbon Fertiliser, Fuel and Energy Store. 2020. Available online: https://royalsociety.org/-/media/policy/projects/green-ammonia/green-ammonia-policy-briefing.pdf (accessed on 19 March 2021).
- Krishnan, S.; Fairlie, M.; Andres, P.; De Groot, T.; Kramer, G.J. Power to gas (H2): Alkaline electrolysis. In Technological Learning in the Transition to a Low-Carbon Energy System: Conceptual Issues, Empirical Findings, and Use, in Energy Modeling; Elsevier Inc: Amsterdam, The Netherlands, 2020; pp. 165–187. [Google Scholar] [CrossRef]
- Ernst, F.A. Industrial Chemical Monographs: Fixation of Atmospheric Nitrogen; Chapman & Hall, Ltd.: London, UK, 1928. [Google Scholar]
- Scott, E.K. Nitrates and Ammonia from Atmospheric Nitrogen. Lectures III. J. R. Soc. Arts 1923, 71, 900–917. [Google Scholar]
- Travis, A.S. Nitrogen Capture: The Growth of an International Industry (1900–1940); Springer: Cham, Switzerland, 2018. [Google Scholar] [CrossRef]
- Rouwenhorst, K.H.R.; Jardali, F.; Bogaerts, A.; Lefferts, L. From the Birkeland-Eyde process towards energy-efficient plasma-based NOX synthesis: A Techno-Economic Analysis. Energy Environ. Sci. 2021, 14, 2520–2534. [Google Scholar] [CrossRef]
- Patil, B.S.; Wang, Q.; Hessel, V.; Lang, J. Plasma N2-fixation: 1900–2014. Catal. Today 2015, 256, 49–66. [Google Scholar] [CrossRef]
- Brightling, J.R. Ammonia and the fertiliser industry: The Development of Ammonia at Billingham. Johns. Matthey Technol. Rev. 2018, 62, 32–47. [Google Scholar] [CrossRef]
- Travis, A.S. Globalising Synthetic Nitrogen: The Interwar Inauguration of a New Industry. Ambix 2017, 64, 1–28. [Google Scholar] [CrossRef]
- Travis, A.S. The Synthetic Nitrogen Industry in World War I: Its Emergence and Expansion; Springer: Cham, Switzerland, 2015. [Google Scholar] [CrossRef]
- Van Rooij, A. Engineering Contractors in the Chemical Industry. The Development of Ammonia Processes, 1910–1940. Hist. Technol. 2005, 21, 345–366. [Google Scholar] [CrossRef]
- Ernst, F.A.; Reed, F.C.; Edwards, W.L. A Direct Synthetic Ammonia Plant. Ind. Eng. Chem. 1925, 17, 775–788. [Google Scholar] [CrossRef]
- LeRoy, R.L. Industrial water electrolysis: Present and Future. Int. J. Hydrog. Energy 1983, 8, 401–417. [Google Scholar] [CrossRef]
- International Renewable Energy Agency (IRENA). Green Hydrogen Cost Reduction: Scaling up Electrolysers to Meet the 1.5 °C Climate Goal. 2020. Available online: https://irena.org/-/media/Files/IRENA/Agency/Publication/2020/Dec/IRENA_Green_hydrogen_cost_2020.pdf (accessed on 19 March 2021).
- Ernst, F.A.; Sherman, M.S. The World’s Inorganic Nitrogen Industry. Ind. Eng. Chem. 1927, 19, 196–204. [Google Scholar] [CrossRef]
- Sánchez, A.; Martín, M. Scale up and scale down issues of renewable ammonia plants: Towards Modular Design. Sustain. Prod. Consum. 2018, 16, 176–192. [Google Scholar] [CrossRef]
- West, J.H. The Claude synthetic ammonia process and plant. J. Soc. Chem. Ind. 1921, 40, R420–R424. [Google Scholar] [CrossRef]
- The Claude process for ammonia synthesis. Nature 1921, 107, 765. [CrossRef]
- Rouwenhorst, K.H.R.; Van der Ham, A.G.J.; Lefferts, L. Beyond Haber-Bosch: The Renaissance of the Claude Process. Int. J. Hydrog. Energy 2021, 46, 21566–21579. [Google Scholar] [CrossRef]
- Slade, R.E. The Nitrogen Industry. J. Soc. Dye. Colour. 1928, 44, 265–268. [Google Scholar] [CrossRef]
- Norris Shreve, R. The Chemical Process Industries; McGraw Hill: New York, NY, USA, 1945. [Google Scholar]
- Conti, P.P.G. The Development of the Chemical Industry in Italy. Ind. Eng. Chem. 1926, 18, 999–1002. [Google Scholar] [CrossRef]
- Bates, H.R. Economic Relationships between Nitrogen and Fertilizers. Ind. Eng. Chem. 1928, 20, 1133–1139. [Google Scholar] [CrossRef]
- Vieira da Rosa, A. Fundamentals of Renewable Energy Processes, 2nd ed.; Academic Press: Burlington, MA, USA, 2009. [Google Scholar] [CrossRef]
- Brown, T.; Renewable Ammonia from Biomass: SynGest, BioNitrogen, Agrebon. NH3 Fuel Association. 2013. Available online: https://nh3fuelassociation.org/2013/04/24/ammonia-from-biomass/ (accessed on 19 March 2021).
- Laskin, J.B.; Feldwick, R.D. Recent development of large electrolytic hydrogen generators. Int. J. Hydrog. Energy 1978, 3, 311–320. [Google Scholar] [CrossRef]
- Tilak, B.V.; Lu, P.W.T.; Colman, J.E.; Srinivasan, S. Electrolytic Production of Hydrogen. In Comprehensive Treatise of Electrochemistry; Bockris, J.O., Conway, B.E., Yeager, E., White, R.E., Eds.; Springer: Boston, MA, USA, 1981; pp. 1–104. [Google Scholar] [CrossRef]
- Grundt, T.; Christiansen, K. Hydrogen by water electrolysis as basis for small scale ammonia production. A comparison with hydrocarbon based technologies. Int. J. Hydrog. Energy 1982, 7, 247–257. [Google Scholar] [CrossRef]
- Birkeland, K. On the oxidation of atmospheric nitrogen in electric arcs. Trans. Faraday Soc. 1906, 2, 98–116. [Google Scholar] [CrossRef] [Green Version]
- Eyde, S. The Manufacture of nitrates from the atmosphere by the electric arc—Birkeland-Eyde Process. J. R. Soc. Arts 1909, 57, 568–576. [Google Scholar]
- Hine, F. Electrode Processes and Electrochemical Engineering; Springer: Cham, Switzerland, 1985. [Google Scholar]
- Egyptian Natural Gas Holding Company. History of the Natural Gas Industry. 2019. Available online: https://www.egas.com.eg/history-natural-gas-industry (accessed on 18 August 2021).
- Kasahara, S. Water Electrolysis. In Nuclear Hydrogen Production Handbook, 1st ed.; Yan, X.L., Hino, R., Eds.; CRC Press: Boca Raton, FL, USA, 2011; pp. 83–97. [Google Scholar]
- National Renewable Energy Laboratory. Current (2009) State-of-the-Art Hydrogen Production Cost Estimate Using Water Electrolysis. Available online: https://www.nrel.gov/docs/fy10osti/46676.pdf (accessed on 21 March 2021).
- Brown, T.; Green Ammonia Plants in Chile, Australia, New Zealand. Ammonia Energy. 2019. Available online: https://www.ammoniaenergy.org/articles/green-ammonia-plants-in-chile-australia-new-zealand/ (accessed on 13 April 2021).
- Vogt, U.F.; Schlupp, M.; Burnat, D.; Züttel, A. Novel Developments in Alkaline Water Electrolysis. In Proceedings of the 8th International Symposium Hydrogen & Energy, Zhaoquing, China, 16–21 February 2014; pp. 1–27. Available online: http://www.elygrid.com/wp-content/uploads/2021/07/event_8th-international-symposium-hydrogen-and-energy.pdf (accessed on 10 January 2022).
- Bhatia, R.; Malik, R.P.S.; Bhatia, M. Direct and indirect economic impacts of the Bhakra multipurpose dam, India. Irrig. Drain. 2007, 56, 195–206. [Google Scholar] [CrossRef]
- Chhabra, O.N. Replacement of Electrolysis Plant with Steam Naptha Rreformation Plant, Its Integration with Old Heavy Water Plant. 1992. Available online: https://inis.iaea.org/collection/NCLCollectionStore/_Public/24/053/24053969.pdf (accessed on 10 January 2022).
- National Fertilizers Limited. xBrief Summary—Installation of GTG cum HRSG under Energy Reduction Scheme at Nangal Unit. 2018. Available online: http://environmentclearance.nic.in/writereaddata/Online/TOR/09_Jan_2018_12043508069CZE2TLBriefDescriptionofProject-Nangal.pdf (accessed on 10 January 2022).
- ThyssenKrupp. Hydrogen from Large-Scale Electrolysis. 2018. Available online: https://ucpcdn.thyssenkrupp.com/_legacy/UCPthyssenkruppBAISUhdeChlorineEngineers/assets.files/products/water_electrolysis/tk_19_0820_hydrogen_broschuere_2019_03.pdf (accessed on 19 October 2021).
- Smith, C.; Hill, A.K.; Torrente-Murciano, L. Current and future role of Haber–Bosch ammonia in a carbon-free energy landscape. Energy Environ. Sci. 2020, 13, 331–344. [Google Scholar] [CrossRef]
- Nielsen, A. Ammonia: Catalysis and Manufacture, 1st ed.; Nielsen, A., Ed.; Springer: Berlin/Heidelberg, Germany, 1995. [Google Scholar]
- Liu, H. Ammonia Synthesis Catalysts: Innovation and Practice; World Scientific: Singapore, 2013. [Google Scholar] [CrossRef] [Green Version]
- Appl, M. Ammonia: Principles and Industrial Practice, 1st ed.; Wiley-VCH: Weinheim, Germany, 1999. [Google Scholar] [CrossRef]
- Jennings, J.M. Catalytic Ammonia Synthesis: Fundamentals and Practice, 1st ed.; Plenum Press: New York, NY, USA, 1992. [Google Scholar]
- International Renewable Energy Agency (IRENA). Renewable Power Generation Costs in 2019. 2020. Available online: https://www.irena.org/-/media/Files/IRENA/Agency/Publication/2020/Jun/IRENA_Power_Generation_Costs_2019.pdf (accessed on 20 October 2021).
- Axelrod, L. The Technology of Ammonia Plants. Catal. Rev. 1981, 23, 53–65. [Google Scholar] [CrossRef]
- Armijo, J.; Philibert, C. Flexible production of green hydrogen and ammonia from variable solar and wind energy: Case Study of Chile and Argentina. Int. J. Hydrog. Energy 2020, 45, 1541–1558. [Google Scholar] [CrossRef]
- ThyssenKrupp. Making the World’s Largest Ammonia Plant Even Larger. 2019. Available online: https://insights.thyssenkrupp-industrial-solutions.com/story/making-the-worlds-largest-ammonia-plant-even-larger/ (accessed on 30 May 2021).
- Morgan, E.R.; Manwell, J.F.; McGowan, J.G. Sustainable Ammonia Production from U.S. Offshore Wind Farms: A Techno-Economic Review. ACS Sustain. Chem. Eng. 2017, 5, 9554–9567. [Google Scholar] [CrossRef]
- Nayak-Luke, R.M.; Forbes, C.; Cesaro, Z.; Bañares-Alcántara, R.; Rouwenhorst, K.H.R. Techno-Economic Aspects of Production, Storage and Distribution of Ammonia. In Techno-Economic Challenges of Green Ammonia as Energy Vector; Bañares-Alcántara, R., Valera-Medina, A., Eds.; Elsevier Inc.: Amsterdam, The Netherlands, 2020; pp. 191–208. [Google Scholar] [CrossRef]
- Salmon, N.; Bañares-Alcántara, R. Green ammonia as a spatial energy vector: A Review. Sustain. Energy Fuels 2021, 2021, 2814–2839. [Google Scholar] [CrossRef]
- Ewing, R. Global Ammonia Prices Soar on String of Spot Deals and Shutdowns. 2021. Available online: https://www.icis.com/explore/resources/news/2021/02/12/10606213/global-ammonia-prices-jump-as-key-plants-go-offline/ (accessed on 10 January 2022).
- Stamicarbon. Maire Tecnimont Group Starts Preliminary Work on a Renewable Power-to-Fertilizer Plant in Kenya. Retrieved. 2021. Available online: https://www.stamicarbon.com/press-release/maire-tecnimont-group-starts-preliminary-work-renewable-power-fertilizer-plant-kenya (accessed on 25 May 2021).
- Green, L. An ammonia energy vector for the hydrogen economy. Int. J. Hydrog. Energy 1982, 7, 355–359. [Google Scholar] [CrossRef]
- Avery, W.H. A Role for Ammonia in the Hydrogen Economy. Int. J. Hydrog. Energy 1988, 13, 761–773. [Google Scholar] [CrossRef]
- Christensen, C.H.; Johannessen, T.; Sørensen, R.Z.; Nørskov, J.K. Towards an ammonia-mediated hydrogen economy? Catalysis Today 2006, 111, 140–144. [Google Scholar] [CrossRef]
- Thomas, G.; Parks, G. Potential Roles of Ammonia in a Hydrogen Economy: A Study of Issues Related to the Use Ammonia for On-Board Vehicular Hydrogen Storage; U.S. Department of Energy: Washington, DC, USE, 2006. [Google Scholar]
- Morlanés, N.; Katikaneni, S.P.; Paglieri, S.N.; Harale, A.; Solami, B.; Sarathy, S.M.; Gascon, J. A Technological Roadmap to the Ammonia Energy Economy: Current State and Missing Technologies. Chem. Eng. J. 2020, 408, 127310. [Google Scholar] [CrossRef]
- MacFarlane, D.R.; Cherepanov, P.V.; Choi, J.; Suryanto, B.H.R.; Hodgetts, R.Y.; Bakker, J.M.; Simonov, A.N. A Roadmap to the Ammonia Economy. Joule 2020, 4, 1186–1205. [Google Scholar] [CrossRef]
- Sousa Cardoso, J.; Silva, V.; Rocha, R.C.; Hall, M.C.; Costa, M.; Eusébio, D. Ammonia as an energy vector: Current and future prospects for low-carbon fuel applications in internal combustion engines. J. Clean. Prod. 2021, 296, 126562. [Google Scholar] [CrossRef]
- Guo, J.; Chen, P. Catalyst: NH3 as an Energy Carrier. Chem 2017, 3, 709–712. [Google Scholar] [CrossRef]
- Rouwenhorst, K.H.R.; Van Der Ham, A.G.J.; Mul, G.; Kersten, S.R.A. Islanded ammonia power systems: Technology Review & Conceptual Process Design. Renew. Sustain. Energy Rev. 2019, 114, 109339. [Google Scholar] [CrossRef]
- Alfa Laval; Hafnia; Haldor Topsøe; Vestas; Siemens Gamesa. Ammonfuel—An Industrial View of Ammonia as a Marine Fuel. 2020. Available online: https://hafniabw.com/news/ammonfuel-an-industrial-view-of-ammonia-as-a-marine-fuel/ (accessed on 20 January 2021).
- International Renewable Energy Agency (IRENA). Renewable Power Generation Costs in 2020. 2021. Available online: https://www.irena.org/-/media/Files/IRENA/Agency/Publication/2021/Jun/IRENA_Power_Generation_Costs_2020.pdf (accessed on 15 October 2021).
- Saygin, D.; Gielen, D. Zero-Emission Pathway for the Global Chemical and Petrochemical Sector. Energies 2021, 14, 3772. [Google Scholar] [CrossRef]
- Singh, A.R.; Rohr, B.A.; Statt, M.J.; Schwalbe, J.A.; Cargnello, M.; Nørskov, J.K. Strategies toward Selective Electrochemical Ammonia Synthesis. ACS Catalysis 2019, 9, 8316–8324. [Google Scholar] [CrossRef]
- Kyriakou, V.; Garagounis, I.; Vasileiou, E.; Vourros, A.; Stoukides, M. Progress in the Electrochemical Synthesis of Ammonia. Catalysis Today 2017, 286, 2–13. [Google Scholar] [CrossRef]
- McPherson, I.J.; Sudmeier, T.; Fellowes, J.; Tsang, S.C.E. Materials for electrochemical ammonia synthesis. Dalton Trans. 2019, 48, 1562–1568. [Google Scholar] [CrossRef]
- Giddey, S.; Badwal, S.P.S.; Kulkarni, A. Review of electrochemical ammonia production technologies and materials. Int. J. Hydrog. Energy 2013, 38, 14576–14594. [Google Scholar] [CrossRef]
- Rouwenhorst, K.H.R.; Engelmann, Y.; Van ’t Veer, K.; Postma, R.S.; Bogaerts, A.; Lefferts, L. Plasma-driven catalysis: Green Ammonia Synthesis with Intermittent Electricity. Green Chem. 2020, 22, 6258–6287. [Google Scholar] [CrossRef]
- Hong, J.; Prawer, S.; Murphy, A.B. Plasma Catalysis as an Alternative Route for Ammonia Production: Status, Mechanisms, and Prospects for Progress. ACS Sustain. Chem. Eng. 2018, 6, 15–31. [Google Scholar] [CrossRef]
- Peng, P.; Chen, P.; Schiappacasse, C.; Zhou, N.; Anderson, E.; Chen, D.; Ruan, R. A review on the non-thermal plasma-assisted ammonia synthesis technologies. J. Clean. Prod. 2018, 177, 597–609. [Google Scholar] [CrossRef]
- Carreon, M.L. Plasma catalytic ammonia synthesis: State of the Art and Future Directions. J. Phys. D Appl. Phys. 2019, 52, 483001. [Google Scholar] [CrossRef]
- Kibsgaard, J.; Nørskov, J.K.; Chorkendorff, I. The Difficulty of Proving Electrochemical Ammonia Synthesis. ACS Energy Lett. 2019, 2986–2988. [Google Scholar] [CrossRef] [Green Version]
- Dražević, E.; Skúlason, E. Are There Any Overlooked Catalysts for Electrochemical NH3 Synthesis—New Insights from Analysis of Thermochemical Data. iScience 2020, 23, 101803. [Google Scholar] [CrossRef] [PubMed]
- Choi, J.; Suryanto, B.H.R.; Wang, D.; Du, H.-L.; Hodgetts, R.Y.; Vallana, F.M.F.; MacFarlane, D.R.; Simonov, A.N. Identification and elimination of false positives in electrochemical nitrogen reduction studies. Nat. Commun. 2020, 11, 5546. [Google Scholar] [CrossRef] [PubMed]
- Rouwenhorst, K.H.R.; Lefferts, L. Feasibility study of plasma-catalytic ammonia synthesis for energy storage applications. Catalysts 2020, 10, 999. [Google Scholar] [CrossRef]
- Hollevoet, L.; De Ras, M.; Roeffaers, M.; Hofkens, J.; Martens, J.A. Energy-Efficient Ammonia Production from Air and Water Using Electrocatalysts with Limited Faradaic Efficiency. ACS Energy Lett. 2020, 5, 1124–1127. [Google Scholar] [CrossRef] [Green Version]
- Hollevoet, L.; Jardali, F.; Gorbanev, Y.; Creel, J.; Bogaerts, A.; Martens, J.A. Towards Green Ammonia Synthesis through Plasma-Driven Nitrogen Oxidation and Catalytic Reduction. Angew. Chem. 2020, 132, 24033–24037. [Google Scholar] [CrossRef]
- Del Pozo, C.A.; Cloete, S. Techno-economic assessment of blue and green ammonia as energy carriers in a low-carbon future. Energy Convers. Manag. 2022, 255, 115312. [Google Scholar] [CrossRef]
- Iberdrola. The First 5 Green Hydrogen Storage Tanks Arrive in Puertollano. 2021. Available online: https://www.iberdrola.com/press-room/news/detail/storage-tanks-green-hydrogen-puertollano (accessed on 17 March 2021).
- Sanchez, A.; Martín, M.; Vega, P. Biomass Based Sustainable Ammonia Production: Digestion vs. Gasification. ACS Sustain. Chem. Eng. 2019, 7, 9995–10007. [Google Scholar] [CrossRef]
- International Energy Agency.The Future of Hydrogen: Seizing today’s opportunities. Available online: https://www.iea.org/events/the-future-of-hydrogen-seizing-todays-opportunities (accessed on 19 March 2021).
- De Pee, A.; Pinner, D.; Roelofsen, O.; Somers, K.; Speelman, E.; Witteveen, M. Decarbonization of industrial sectors: The next frontier. Available online: https://www.mckinsey.com/~/media/mckinsey/business%20functions/sustainability/our%20insights/how%20industry%20can%20move%20toward%20a%20low%20carbon%20future/decarbonization-of-industrial-sectors-the-next-frontier.pdf (accessed on 17 March 2021).
- Moya, J.A.; Boulamanti, A. Production Costs from Energy Intensive Industries in the EU and Third Countries; Publications Office of the European Union: Luxembourg, 2016. [Google Scholar]
- Bicer, Y.; Dincer, I.; Zamfirescu, C.; Vezina, G.; Raso, F. Comparative life cycle assessment of various ammonia production methods. J. Clean. Prod. 2016, 135, 1379–1395. [Google Scholar] [CrossRef]
- Fertilizer.org. Ammonia production: Moving towards maximum efficiency and lower GHG emissions. Available online: https://www.fertilizer.org/images/Library_Downloads/2014_ifa_ff_ammonia_emissions_july.pdf (accessed on 17 March 2021).
- Appl, M. Ammonia, 2. Production Processes. In Ullmann’s Encyclopedia of Industrial Chemistry; Wiley-VCH: Weinheim, Germany, 2012. [Google Scholar] [CrossRef]
- Fisher, J.C.; Pry, R.H. A simple substitution model of technological change. Technol. Forecast. Soc. Chang. 1971, 3, 75–88. [Google Scholar] [CrossRef]
- Santos, D.M.F.; Sequeira, C.A.C.; Figueiredo, J.L. Hydrogen production by alkaline water electrolysis. Química Nova 2013, 36, 1176–1193. [Google Scholar] [CrossRef]
- Aalborg University. Technology data for high temperature solid oxide electrolyser cells, alkali and PEM electrolysers. Available online: https://www.osti.gov/etdeweb/servlets/purl/22128040 (accessed on 17 March 2021).
- Smolinka, T.; Günther, M.; Garche, J. NOW-Studie “Stand und Entwicklungspotenzial der Wasserelektrolyse zur Herstellung von Wasserstoff aus regenerativen Energien”. Available online: https://www.now-gmbh.de/wp-content/uploads/2020/09/now-studie-wasserelektrolyse-2011.pdf (accessed on 17 March 2021).
- Miolati, A. Synthetic Ammonia and the Casale Process. In Amplified Edition of a Lecture Delivered the 27th February 1927 at the Institute of Chemistry of the Polytechnic School of Prague; “L’Universale” Tipografia Poliglotta/Ammonia Casale SA: Rome, Italy, 1927; pp. 51–52. [Google Scholar]
- Casale, S.A. Capacité des usines à amoniaque synthétique procéedé Casale. Available online: https://www.casale.ch/ (accessed on 19 March 2021).
- Dannenbaum, W. Walter Dannenbaum, Vice President, to Board of Directors; Pacific Nitrogen Corporation: Wilmington, DE, USA, 1928. [Google Scholar]
- Cope, W.C. Ammonia. Part I. US Production facilities. Chem. Ind. 1949, 64, 920–925. [Google Scholar]
- Davenport, C.H. Boom Still on for U.S. Ammonia. Pet. Refin. 1995, 35, 141–148. [Google Scholar]
- Water Electrolysis. Solar-Hydrogen Energy Systems; Elsevier: Amsterdam, The Netherlands, 1979; pp. 35–58. [Google Scholar]







| Process | Year | Temperature (°C) | Pressure (atm) | Single Pass Conversion (%) |
|---|---|---|---|---|
| Haber–Bosch (Germany) * | 1913 | 550 | 200 | 7–8 |
| Casale (Italy) | 1921 | 500 | 800–850 | 15–18 |
| Claude (France) | 1921–1922 | 500–650 | 900–1000 | 40 ** |
| Fauser (Italy) | 1921–1922 | 500 | 250–300 | 12–23 |
| General Chemical/Allied (United States) | 1921 | 500 | 200 | 20–22 *** |
| Nitrogen Engineering Corporation (United States) | 1926 | 500 | 200–300 | 20–22 *** |
| Mont Cenis (France) | 1925–1926 | 400–425 | 100 | 9–20 *** |
| Showa Fertilizer (Japan) | 1931 | - | - | - |
| Location | Ammonia Technology | Year | Capacity (kt-NH3 y−1) |
|---|---|---|---|
| Terni/Nera Montoro * | Casale | 1921–1923 | 0.7 |
| 1924 | 2.6 | ||
| 1926 | 7.0 | ||
| 1927 | 10.5 | ||
| Bussi | Claude | 1923 ** | 2.5 |
| Belluno (Mas) | Fauser | 1924 | - |
| Merano | Fauser | 1925 | 37.8 |
| 1930s | 35.0 | ||
| Agordo | - | 1926 *** | 3.5 |
| Novara | Fauser | 1926 | 7.5 |
| Coghinas | Fauser | 1927 | 3.5–7.0 |
| Crotone | Fauser | 1927 | 7.0 |
| 1930s | 24.5 | ||
| San Giuseppe di Cairo | Fauser | N/A | 17.5 |
| 1930s | 44.8 | ||
| Taranto | Fauser | - | - |
| Massina | Fauser | - | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rouwenhorst, K.H.R.; Travis, A.S.; Lefferts, L. 1921–2021: A Century of Renewable Ammonia Synthesis. Sustain. Chem. 2022, 3, 149-171. https://doi.org/10.3390/suschem3020011
Rouwenhorst KHR, Travis AS, Lefferts L. 1921–2021: A Century of Renewable Ammonia Synthesis. Sustainable Chemistry. 2022; 3(2):149-171. https://doi.org/10.3390/suschem3020011
Chicago/Turabian StyleRouwenhorst, Kevin H. R., Anthony S. Travis, and Leon Lefferts. 2022. "1921–2021: A Century of Renewable Ammonia Synthesis" Sustainable Chemistry 3, no. 2: 149-171. https://doi.org/10.3390/suschem3020011
APA StyleRouwenhorst, K. H. R., Travis, A. S., & Lefferts, L. (2022). 1921–2021: A Century of Renewable Ammonia Synthesis. Sustainable Chemistry, 3(2), 149-171. https://doi.org/10.3390/suschem3020011








