Antagonism of Estrogen Receptor α-Driven Transcription Mediated by AP-1 in Breast Cancer Therapy
Abstract
1. Introduction
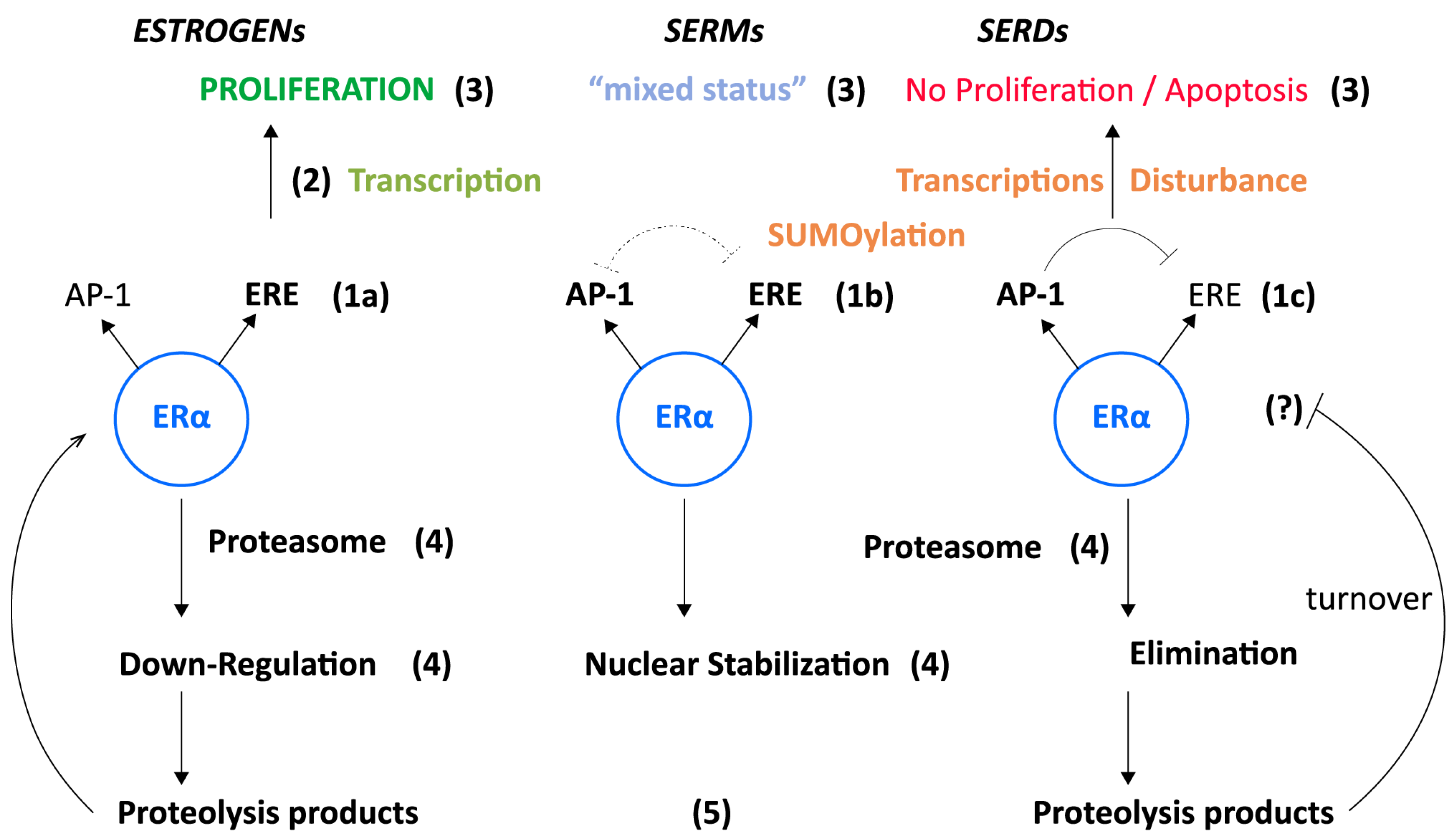
2. ERα-Mediated Transcriptions
3. Ligand Insertion within the ERα Binding Pocket
4. Assessing the Activity of 11β Substituted Estradiol Derivatives
4.1. Nature and Origin of the Investigated Compounds
4.2. Experimental Procedures
4.3. Results
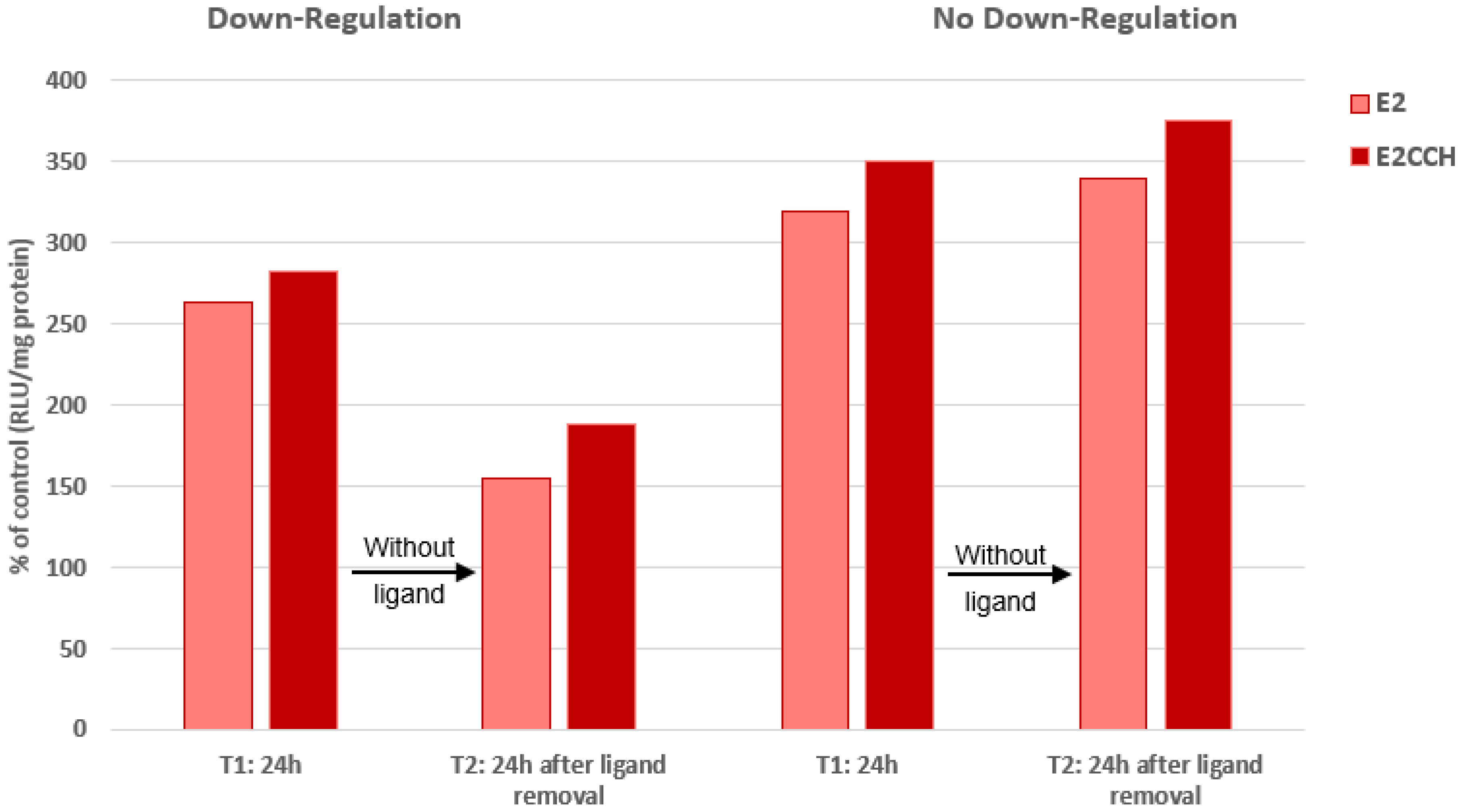
4.3.1. Global View of Our Investigation
4.3.2. Small-Size 11β Substituents
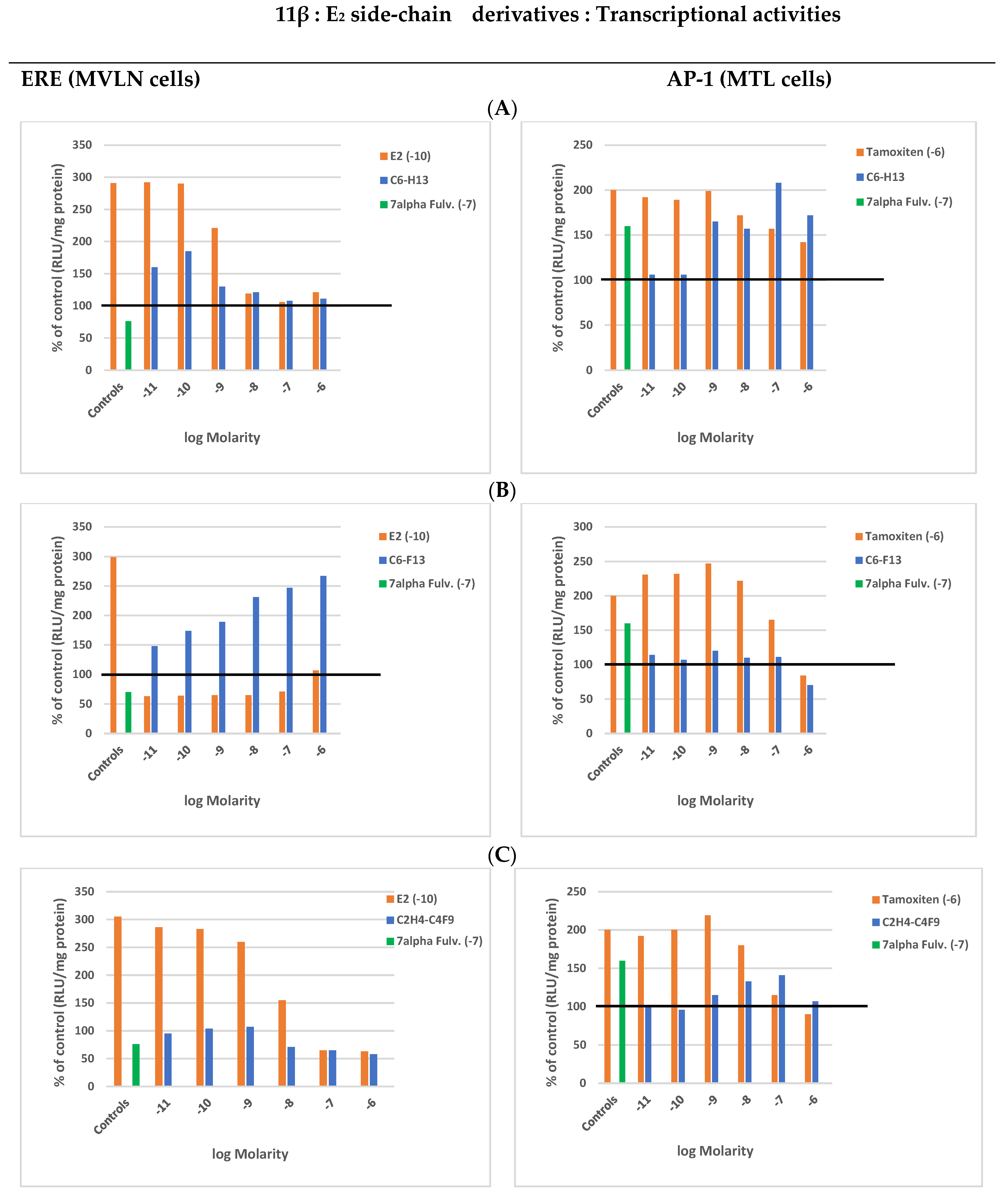
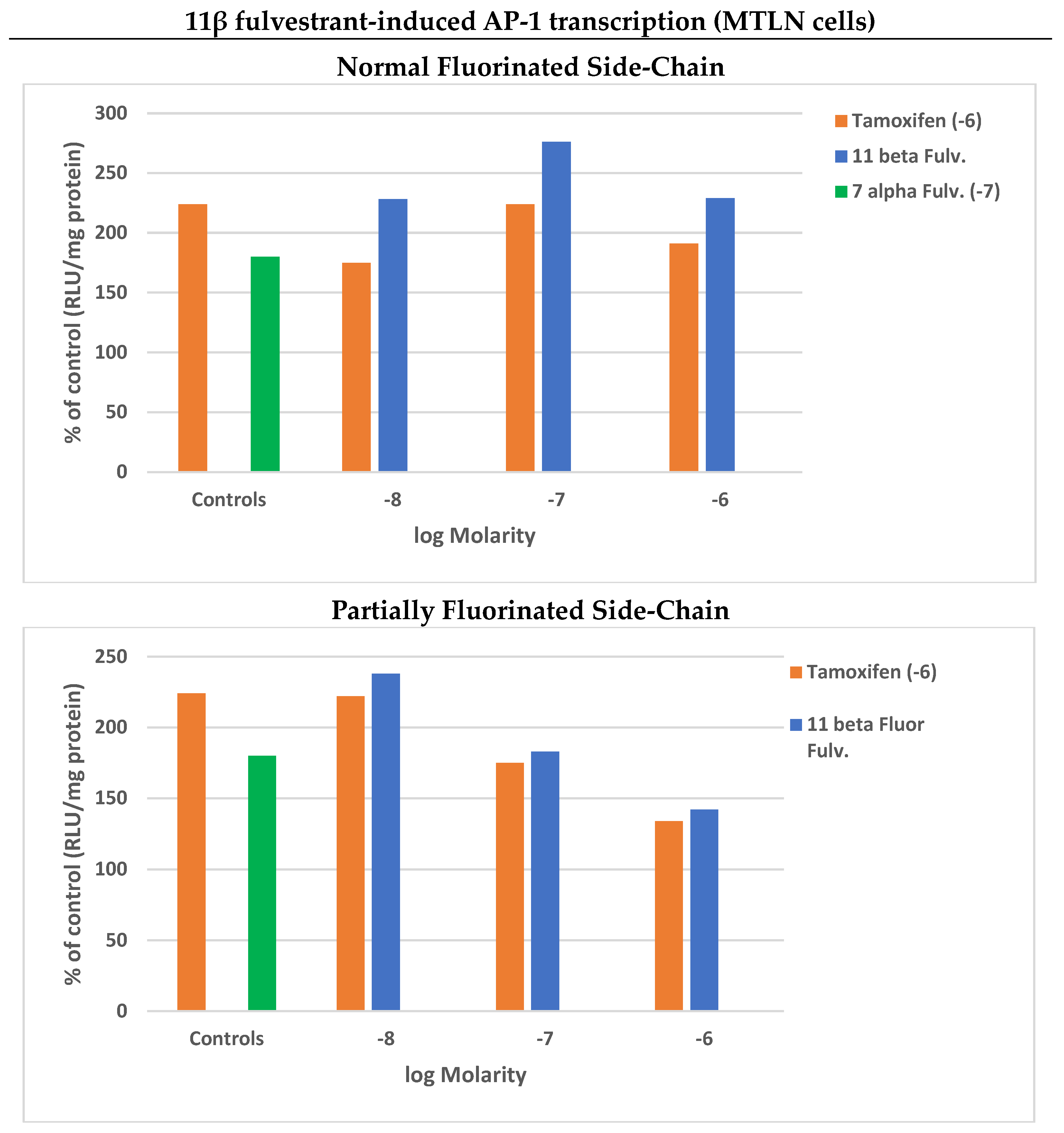
4.3.3. 11β Side-Chains
SERMs
SERDs
5. Concluding Remarks
Supplementary Materials
Funding
Acknowledgments
Conflicts of Interest
References
- Katzenellenbogen, J.A. Stringing along the estrogen receptor to engage DNA. Proc. Natl. Acad. Sci. USA 2023, 120, e230060812. [Google Scholar] [CrossRef] [PubMed]
- Chen, X.; Jin, S.; Chen, M.; Bueno, C.; Wolynes, P.G. The marionette mechanism of domain-domain communication in the antagonist, agonist, and coactivator response of the estrogen receptor. Proc. Natl. Acad. Sci. USA 2023, 120, e2216906120. [Google Scholar] [CrossRef] [PubMed]
- Arao, Y.; Korach, K.S. The physiological role of estrogen receptor functional domains. Essays Biochem. 2021, 65, 867–875. [Google Scholar] [CrossRef]
- Perissi, V.; Rosenfeld, M.G. Controlling nuclear receptors: The circular logic of cofactor cycles. Nat. Rev. Mol. Cell. Biol. 2005, 6, 542–554. [Google Scholar] [CrossRef]
- Tecalco-Cruz, A.C.; Ramirez-Jarquin, J.O.; Cruz-Ramos, E. Estrogen Receptor Alpha and its Ubiquitination in Breast Cancer. Curr. Drug Targets 2019, 20, 690–704. [Google Scholar] [CrossRef]
- Habara, M.; Shimala, M. Estrogen receptor a revised: Expression, function and stability. BioEssays 2022, 44, e2200148. [Google Scholar] [CrossRef] [PubMed]
- Arnal, J.-F.; Lenfant, F.; Metivierr, R.; Flouriot, G.; Henrion, D.; Adlanmerini, M.; Gourdy, P.; Chambon, P.; Katzenellenbogen, B.; Katzenellenbogen, J.A.; et al. Membrane and nuclear estrogen receptor alpha actions: From tissue specificity to medical implications. Physiol. Rev. 2017, 17, 1045–1087. [Google Scholar] [CrossRef]
- Winkeldfeld, S.R.; Lin, F.D.E. Communication between genomic and non-genomic signaling events coordinate steroid hormone actions. Steroids 2018, 133, 2–7. [Google Scholar]
- Leclercq, G. Pathological Maintenance and Evolution of Breast Cancer: The Convergence of Irreversible Biological Actions of ER Alpha. Endocrines 2021, 2, 1–14. [Google Scholar] [CrossRef]
- Marino, M.; Ascenzi, P.; Acconcia, F. S-palmitoylation modulates estrogen receptor alpha localisation and function. Steroids 2006, 71, 298–303. [Google Scholar] [CrossRef]
- Razandi, M.; Pedram, A.; Levin, E.R. Heat shock protein 27 is required for sex steroid receptor trafficking to and functioning at the plasma membrane. Mol. Cell. Biol. 2010, 30, 3249–3261. [Google Scholar] [CrossRef]
- Acconcia, A.; Fiocchetti, M.; Busnero, C.; Fernandez, V.S.; Montalesi, E.; Cipoletti, M.; Pallottini, V.; Marino, M. The extra-nuclear interactome of the estrogen receptors: Implications for the physiological functions. Molec. Cell. Endocrinol. 2021, 538, 111452. [Google Scholar] [CrossRef] [PubMed]
- Safe, S.; Kim, K. Non Classical ER/Sp and AR/AP-1 signaling Pathways. J. Mol. Endocrinol. 2008, 41, 263–275. [Google Scholar] [CrossRef] [PubMed]
- Webb, P.; Nguyen, P.; Valentine, C.; Lopez, G.N.; Kwok, R.; Mclnerney, E.; Katzenellenbogen, B.S.; Enmark, E.; Gustafsson, A.; Nilsson, S.; et al. The estrogen receptor enhances AP-1 activity by two distinct mechanisms with different requirements for receptor transactivation functions. Mol. Endocrinol. 1999, 13, 1672–1685. [Google Scholar] [CrossRef]
- Kushner, P.J.; Agard, D.A.; Greene, G.L.; Scanlan, T.S.; Shiau, A.K.; Uth, R.M.; Webb, P. Estrogen receptor pathways to A-P-1. J. Steroid Biochem. Mol. Biol. 2000, 74, 311–317. [Google Scholar] [CrossRef] [PubMed]
- Acramel, A.; Jacquot, Y. Deciphering of a Putative GPER Recognition Domain in ERα and ERα36. Front. Endocrinol. 2022, 13, 943343. [Google Scholar] [CrossRef]
- Buzon, V.; Carbo, L.R.; Estruch, S.B.; Fletterick, R.J.; Estebanez-Perpina, E.A. A conserved surface on the ligand binding domain of nuclear receptors for allosteric controls. Mol. Cell. Endocrinol. 2012, 348, 394–402. [Google Scholar] [CrossRef]
- Fischer, A.; Smiesko, M. Allosteric Binding Sites On Nuclear Receptors: Focus on Drug Efficacy and Selectivity. Int. J. Mol. Sci. 2020, 21, 534. [Google Scholar] [CrossRef]
- Gallo, D.; Haddad, I.; Duvillier, H.; Jacquemotte, F.; Laïos, I.; Laurent, G.; Jacquot, I.; Vinh, H.; Leclercq, G. Trophic effect in MCF-7cells of ERalpha17p, a peptide corresponding to a platform regulatory motif of the estrogen receptor alpha—Underlying mechanism. J. Steroid Mol. Biol. 2008, 109, 138–149. [Google Scholar] [CrossRef]
- Métivier, R.; Penot, G.; Flouriot, G.; Pakdel, P. Synergism between ER alpha transactivation function 1 (AF-1) and AF-2 mediated by steroid receptor coactivator protein-1: Requirement for AF-I alpha-helical core and for a direct interaction between the N- and C- terminal domains. Mol. Endocrinol. 2001, 15, 1953–1970. [Google Scholar]
- Zwart, W.; de Leeuw, R.; Rondaij, M.; Neefjes, J.; Mancini, M.A.; Michalides, R. The hinge region of the human estrogen receptor determines functional energy between AF-1 and AF-2 in the quantitative response to estradiol and tamoxifen. J. Cell Sci. 2010, 123, 1253–1261. [Google Scholar] [CrossRef][Green Version]
- Yang, Y.; Singleton, D.W.; Shaughnessy, S.A.; Khan, S.A. The F-domain of estrogen receptor-alpha inhibits ligand induced receptor dimerization. Mol. Cell Endocrinol. 2008, 295, 94–100. [Google Scholar] [CrossRef]
- Carlson, K.E.; Choi, I.; Gee, G.; Katzenellenbogen, B.S.; Katzenellenbogen, J.A. Altered Ligand binding properties and enhanced stability of a constitutively active estrogen receptor: Evidence that an open pocket conformation is required for ligand interaction. Biochemistry 1997, 36, 14897–14905. [Google Scholar] [CrossRef]
- El Khisiin, A.; Leclercq, G. Exchange of bound estrogens and antiestrogens In MCF-7 cell: Evidence for ligand-induced stable configurations of the estrogen receptor. Steroids 1998, 63, 565–574. [Google Scholar] [CrossRef]
- Le Romancer, M.; Poulard, C.; Cohen, P.; Sentis, S.; Renoir, J.-M.; Corbo, L. Cracking the estrogen receptors posttranscriptional code in breast tumors. Endocr. Rev. 2011, 32, 597–622. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Zhang, Y.; Hedman, A.C.; Ames, J.B.; Sacks, D.B. Calmodulin lobes facilitate dimerization and activation of estrogen receptor-α. J. Biol. Chem. 2017, 292, 4614–4622. [Google Scholar] [CrossRef] [PubMed]
- Bourgouin-Voillard, S.; Fournier, F.; Alfonso, C.; Jacquot, Y.; Leclercq, G.; Tabet, J.-C. Calmodulin association with the synthetic ERα 17p peptide investigated by Mass Spectrometry. Int. J. Mass Spectrom. 2011, 305, 87–94. [Google Scholar] [CrossRef]
- Berry, N.B.; Fan, M.; Nephew, K.P. Estrogen Recceptor-α Hinge- Region Lysines 302 and 303 RegulateReceptor Degadation by the prteasome. Mol. Endocrinol. 2008, 22, 1535–1551. [Google Scholar] [CrossRef] [PubMed]
- Katzenellenbogen, J.A. The 2010 Philip S. Portoghese Medicinal Chemistry Lecture: Addressing the 3 core Issue in the Design of Estrogen Receptor Ligands. J. Med. Chem. 2011, 54, 5271–5282. [Google Scholar] [CrossRef]
- Amstead, G.M.; Carson, K.M.; Katzenellenbogen, J.A. The estradiol pharmacophore: Ligand structure-estrogen receptor binding affinity relationships and a model for the receptor binding site. Steroids 1997, 62, 268–303. [Google Scholar] [CrossRef] [PubMed]
- Kekenes-Huskey, P.M.; Megge, I.; von Rauch, M.; Gust, R. Knapp. A molecular Docking study of Estrrogenically active componds with 1,2-diarylethane and 1,é-diarylethenePharmacophore. Biorg. Med. Chem. 2004, 12, 6527–6537. [Google Scholar] [CrossRef]
- Ogawa, T.; Otha, K.; Ijima, T.; Suzuki, T.; Otha, S.; Endo, Y. Synthesis and biological evaluation of p-carborane bispheols and their derivatives: Structure-activity relationship for estrogenic activity. Biorg. Med. Chem. 2009, 17, 1109–1117. [Google Scholar] [CrossRef]
- Lorand, T.; Vigh, E.; Garai, J. Hormonal action of plant derived and anthropogenic non-steroidal estrogenic compounds: Phytoestrogens and xenoestrogens. Curr. Med. Chem. 2010, 17, 2632–2653. [Google Scholar] [CrossRef]
- Jordan, V.C.; Schafer, J.M.; Levenson, A.S.; Liu, H.; Pease, K.M.; Simons, L.A.; Zapf, J.W. Molecular classificatin of estrogens. Cancer Res. 2001, 61, 6619–6623. [Google Scholar]
- Zhang, J.-X.; Labaree, D.C.; Hochberg, R.B. Nonpolar and short side chain groups at C-11beta of estradiol result in antiestrogens. J. Med. Chem. 2005, 48, 1428–1447. [Google Scholar] [CrossRef] [PubMed]
- Hanson, R.N.; Hua, E.; Hendricks, A.; Labaree, D.; Hochberg, R.B. Synthesis and evaluation of 11β-(4-substituted phenyl) estradiol analogs: Transition from estrogen receptor agonist to antagonist. Biorg. Med. Chem. 2012, 20, 3768–3780. [Google Scholar] [CrossRef] [PubMed]
- Agouridas, V.; Blazejewski, J.-C.; Cleeren, A.; Laïos, I.; Leclercq, G.; Magnier, E. Fluorous tolerance of the estrogen receptor alpha as probed by 11-fluoroalkylestradiol derivatives. Steroids 2008, 73, 320–327. [Google Scholar] [CrossRef] [PubMed]
- Bourgouin-Voillard, S.; Fournier, F.; Alfonso, C.; Zins, E.-L.; Jacquot, Y.; Pèpe, C.; Leclercq, G.; Tabet, J.-C. Electronic effect of 11βsubstituted 17β-estradiol and instrumental effects on the relative gas phase acidity. J. Am. Soc. Mass Spectrom. 2012, 23, 2167–2177. [Google Scholar] [CrossRef]
- Jakaka, M.; Ito, M.; Weis, J.; Chien, P.; Gehem, B.D.; Jameson, J.L. Estrogen receptor binding to DNA is not required for its activity through-h the nonclassical AP1 pathway. J. Biol. Chem. 2001, 276, 13615–13621. [Google Scholar] [CrossRef] [PubMed]
- Heldring, N.; Isaacs, G.D.; Diehl, A.G.; Sun, M.; Cheung, E.; Ranish, J.A.; Lee Kraus, W. Multiple sequence-specific DNA-binding proteins mediate estrogen receptor signaling through a tethering pathway. Mol. Endocrinol. 2011, 25, 564–574. [Google Scholar] [CrossRef] [PubMed]
- He, H.; Sinha, I.; Fan, R.; Haldosen, L.-A.; Zhao, C.; Dalman-Wright, K. c-Jun/AP-1 overexpression reprograms ERα signaling related to tamoxifen response in in ERα -positive breast cancer. Oncogene 2018, 19, 2586–2600. [Google Scholar] [CrossRef] [PubMed]
- Song, D.; Lian, Y.; Zhang, L. The potential activator protein 1 (AP-1)in breast cancer targeted therapy. Front. Immunol. 2023, 14, 1224892. [Google Scholar] [CrossRef] [PubMed]
- Cheung, E.; Acevedo, M.L.; Cole, P.A.; Lee Kraus, W. Altered pharmacology and distinct coactivators usage for estrogen receptor-dependent transcription through activating protein -1. Proc. Natl. Acad. Sci. USA 2005, 102, 559–564. [Google Scholar] [CrossRef]
- Maaroufi, Y.; Leclercq, G. Importance of AB and C Domains of the Estrogen Receptor for its Adsorption to Hydroxylapatite. J. Steroid Biochem. Mol. Biol. 1994, 48, 155–1631. [Google Scholar] [CrossRef]
- Spera, D.; Cabrera, G.; Fiashi, R.; Carlson, K.E.; Katzenellenbogen, J.A.; Napolitano, E. Estradiol derivatives bearing sulfur-containing substituents at the 11β or7α positions: Versatile reagents for the preparation of estrogen conjugates. Biorg. Med. Chem. 2004, 12, 4393–4401. [Google Scholar] [CrossRef]
- Pons, M.; Gagne, D.; Nicolas, J.-C.; Methalli, M.A. A new cellular model of response to estrogens: A bioluminescent test to characterize (anti)estrogen molecules. Biotechniques 1990, 9, 450–459. [Google Scholar]
- Astruc, M.E.; Chabret, C.; Ball, P. Prolonged Treatment of breast cancer cells with antiestrogens increases the activating propein-1-mediated response: Involvement of the estrogen receptor. Endocrinology 1995, 136, 824–832. [Google Scholar] [CrossRef] [PubMed]
- Bourgoin-Voillard, S.; Gallo, D.; Laïos, I.; Cleeren, A.; El Bali, L.; Jacquot, Y.; Nonclercq, D.; Laurent, G.; Tabet, J.-C.; Leclercq, G. Capacity of Type I and II to confer to estrogen receptor alpha an appropriate conformation for the recruitment of coactivators containing a LxxLL motif—Relationship with the regulation of receptor level and ERE-dependent transcription in MCF-7 cells. Biochem. Pharmacol. 2010, 79, 746–757. [Google Scholar] [CrossRef][Green Version]
- Otto, A.M. A one minute pulse of estradiol to MCF-7 breast cancer cells changes estrogen receptor binding properties and commits cells to induce estrogenic responses. J. Steroid Biochem. Mol. Biol. 1995, 54, 39–46. [Google Scholar] [CrossRef]
- Lippman, M.E.; Dickson, R.B.; Kasid, A.; Gelmann, E.; Davidson, N.; McManaway, M.; Huff, K.; Bronzert, D.; Bates, S.; Swain, S.J. Autocrine and pararacrine growth regulation of breast cancer. J. Steroid Biochem. 1986, 24, 147–154. [Google Scholar] [CrossRef] [PubMed]
- Levenson, S.A.; Mac Gregor Schafer, J.I.; Bentrem, D.J.; Pease, K.M.; Jordan, V.C. Control of the estrogen-like actions of the tamoxifen-estrogen receptor complex by the surface amino acid at position 351. J. Steroid Biochem. Mol. Bol. 2001, 76, 61–70. [Google Scholar] [CrossRef] [PubMed]
- Vallet, A.; El Ezzy, M.; Diennet, M.; Haidar, S.; Bouvier, M.; Mader, S. The AF-2 cofactor binding region is a key for the selective SUMOylation of estrogen receptor alpha by antiestrogens. J. Biol. Chem. 2023, 229, 102757. [Google Scholar] [CrossRef] [PubMed]
- Traboulsi, T.; El Ezzy, M.; Dumeaux, V.; Audemard, E.; Mader, S. Role of SUMOylation in differential ERα transcriptional repression by tamoxifen and fulvestrant in breast cancer cells. Oncogene 2019, 38, 1019–1037. [Google Scholar] [CrossRef]
- Himi, K.; Hussein, N.; Mendoza-Sanchez, R.; El-Ezzy, M.; Ismail, H.; Durette, C.; Bail, M.; Rozendaal, M.J.; Bouvier, M.; Thibaut, P.; et al. Role of SUMOylation in Full Antiestrogenic. Mol. Cell. Biol. 2012, 32, 3823–3837. [Google Scholar]
- Agouridas, V.; Magnier, E.; Blazejewski, J.-C.; Laïos, I.; Cleeren, A.; Nonclercq, D.; Laurent, G.; Leclercq, G. Effect of Fluorination on the Pharmacological Profile of Fulvestrant in Breast Carcinoma Cells. J. Med. Chem. 2009, 52, 883–887. [Google Scholar] [CrossRef] [PubMed]
- Jin, L.; Borras, M.; Lacroix, M.; Legros, N.; Leclercq, G. Antiestrogenic activity of two 11β-estradiol derivatives on MCF-7 breast cancer cells. Steroids 1995, 60, 512–518. [Google Scholar] [CrossRef]
- Tempé, D.; Vives, E.; Brocky, F.; Brooks, H.S.; De Rossi, S.; PLechaczyk, M.; Bossis, G. SUMOylation of the inductible (c-Fois:c-Jun)/AP-1 transcription complex occurs on target promoters to limit Transcriptional activation. Oncogene 2014, 33, 921–927. [Google Scholar] [CrossRef]
- Wang, Q.; Zhang, N.; Yang, X.; Feng, S.; Wang, F.; Zhang, W.; He, Z. ERα promotes SUMO1- mediated protein SUMOylation in breast cancer. Gland Surg. 2023, 12, 963–973. [Google Scholar] [CrossRef]
- Guan, J.; Zhou, W.; Hafner, M.; Blake, R.A.; Chalouni, C.; Chen, I.P.; De Bruyn, T.; Giltnane, J.M.; Hartman, S.J.; Heidersbach, A.; et al. Therapeutic Ligands Antagonize Estrogen Receptor Function By Impairing Its Mobility. Cell 2019, 178, 949–963. [Google Scholar] [CrossRef]
- Scherbacov, A.M.; Sorokin, D.V.; Omelchuk, O.A.; Shchekotikhin, A.E.; Krasil’nikov, M.A. Glucose starvation greatly enhance antiproliferative and antiestrogenic potency of oligomycin A in MCF-7 breast cancer. Biochimie 2021, 186, 51–58. [Google Scholar] [CrossRef]
- Klinge, C.M. Estrogens regulates life and death in mitochondria. J. Bioenerg. 2017, 49, 307–324. [Google Scholar] [CrossRef] [PubMed]
- Maximov, P.Y.; Curpan, P.; Jordan, V.C. Estrogen Receptor Complex to Trigger or Delay Estrogen-Induced Apoptosis in Long Term Estrogen Deprived Breast Cancer. Front. Endocrinol. 2022, 13, 869562. [Google Scholar] [CrossRef] [PubMed]
- Garcia, M.; Derocq, D.; Freiss, G.; Rochefort, H. Activation of estrogen receptor transfected into a receptor-negative breast cancer cell line decreases the metastatic and invasive potential of the cells. Proc. Natl. Acad. Sci. USA 1992, 89, 11518–11542. [Google Scholar] [CrossRef] [PubMed]
- Zheng, Y.; Murphy, L.C.L. Regulation of steroid hormones receptors and co regulators during cycle highlights potential novel functions in addition to transcription factors. Nuclear Recept. Signal 2016, 14, e001. [Google Scholar]
- JavanModoghadam, S.; Weihua, Z.; Hunt, K.K.; Keyomarsi, K. Estrogen receptor alpha is cell cycle-regulated and regulates cell cycle in a ligand-dependent fashion. Cell Cycle 2016, 15, 1579–1590. [Google Scholar] [CrossRef] [PubMed]
- Sun, X.; Kaufman, P.D. Ki-67:More than a prokiferation marker. Chromosoma 2018, 127, 175–186. [Google Scholar] [CrossRef] [PubMed]
- Nettles, K.W.; Bruning, J.B.; Gil, G.; O’Neil, E.E.; Nowak, J.; Guo, J.; Kim, Y.; De Sombre, R.; Dilis, R.; Hanson, R.N.; et al. Structural plasticity in the estrogen receptor ligand-binding domain. Look. Nucl. Recept. New Rep. 2007, 8, 563–568. [Google Scholar]
- Khan, S.H.; Braet, S.M.; Koehler, S.J.; Elacua, E.; Anand, G.S.; Okafor, C.D. Ligand-induced shifs in conformational ensembles that describe transcriptioal activation. Elife 2022, 11, e80140. [Google Scholar] [CrossRef]
- Helsen, C.; Claessens, F. Looking at nuclear receptors from a new angle. Mol. Cell. Endocrinol. 2014, 382, 97–106. [Google Scholar] [CrossRef]
- WeiKum, E.R.; Liu, X.; Ortlund, E.A. The nuclear receptor superfamily: A structural Perspective. Protein Sci. 2018, 27, 1876–1892. [Google Scholar] [CrossRef]
- Bourgoin-Voillard, S.A.; Zins, E.-E.; Fournier, F.; Jacquot, Y.; Alfonso, C.; Pèpe, C.; Leclercq, G.; Tabet, J.-C. Stereochemical Effects During [M-H]-Dissociations of Epimeric 11-OH-17β Estradiols at Distant Electronic Effects of substituents at Position C(11) on Gas Phase Acidity. J. Am. Soc. Mass Spectrom. 2009, 20, 3318–3333. [Google Scholar] [CrossRef] [PubMed][Green Version]
- French, A.N.; Napolitano, E.; VanBroklin, R.N.; Hanson, R.N.; Welch, R.J.; Katenellenbogen, J.A. Synthesis, radiolabeling and tissue distribution of 11 beta- fluoroalkyl- and 11 beta fluoroalkoxy- substituted estrogens: Target tissue uptake activity and defluorination of a homologous series of fluorine-18-labeled estrogens. Nucl. Med. Biol. 1993, 20, 31–47. [Google Scholar] [CrossRef] [PubMed]
- Eignerova, B.; Sedlak, D.; Drcinsky, M.; Bartunek, P.; Kotora, M. Synthesis and Biochemical Characterization of 17α-Perfluoro alkylated Estradiols as Selective Ligands for Estrogen Receptor α. J. Med. Chem. 2010, 53, 6947–6953. [Google Scholar] [CrossRef] [PubMed]
| Binding | LxxLL Recruitment | Transcription | ERα Level | |||
|---|---|---|---|---|---|---|
| 0 | 25 (°C) | ERE | AP-1 | |||
| estradiol | 100 | 100 | 100/−7 | 100/−10 | 30/−6 | 60/−9 |
| tamoxifen | 5 | 40 | −25/−6 | 25/−6 | 100/−6 | 500/−7 |
| 7a-fulvestrant | 10 | 100 | −40/−8 | 15/−6 | 80/−7 | 35/−7 |
| A. Small-size substituents (estrogens) | ||||||
| -CH3 | 100 | 250 | 100/−8 | 100/−11 | ? | 50/−9 |
| -CH2Cl | 100 | 200 | 100/−8 | 100/−11 | ? | 40/−9 |
| -CCH (ethynyl) | 30 | 170 | 100/−8 | 285/−11 | ? | 45/−9 |
| B. Side-chain substituents (* estrogens, ** SERMs, *** SERDs) | ||||||
| -C6H13 ** | 10 | 100 | −40/−7 | 65/−10 | 120/−7 | 225/−8 |
| -C6F13 * | 3 | 3 | 15/−8 | 90/−6 | 55/−9 | 45/−9 |
| -C2H4CF9 ** | 10 | 10 | −30/−7 | 30/−9 | 65/−8 | 140/−7 |
| -(CH2)9-X *** | 6 | 80 | −40/−8 | 15/−6 | 125/−7 | 35/−7 |
| -(CH2)3(CF2)4(CH2)2 -X *** | 2 | 10 | −40/−8 | 65/−6 | 110/−8 | 80/−6 |
| X = -S(O)(CH3)3C2F5 | ||||||
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2024 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Leclercq, G. Antagonism of Estrogen Receptor α-Driven Transcription Mediated by AP-1 in Breast Cancer Therapy. Endocrines 2024, 5, 102-115. https://doi.org/10.3390/endocrines5010007
Leclercq G. Antagonism of Estrogen Receptor α-Driven Transcription Mediated by AP-1 in Breast Cancer Therapy. Endocrines. 2024; 5(1):102-115. https://doi.org/10.3390/endocrines5010007
Chicago/Turabian StyleLeclercq, Guy. 2024. "Antagonism of Estrogen Receptor α-Driven Transcription Mediated by AP-1 in Breast Cancer Therapy" Endocrines 5, no. 1: 102-115. https://doi.org/10.3390/endocrines5010007
APA StyleLeclercq, G. (2024). Antagonism of Estrogen Receptor α-Driven Transcription Mediated by AP-1 in Breast Cancer Therapy. Endocrines, 5(1), 102-115. https://doi.org/10.3390/endocrines5010007






