Nutrition, Exercise, and Stress Management for Treatment and Prevention of Psychiatric Disorders. A Narrative Review Psychoneuroendocrineimmunology-Based
Abstract
:1. Introduction
2. A New Psychopathological Model
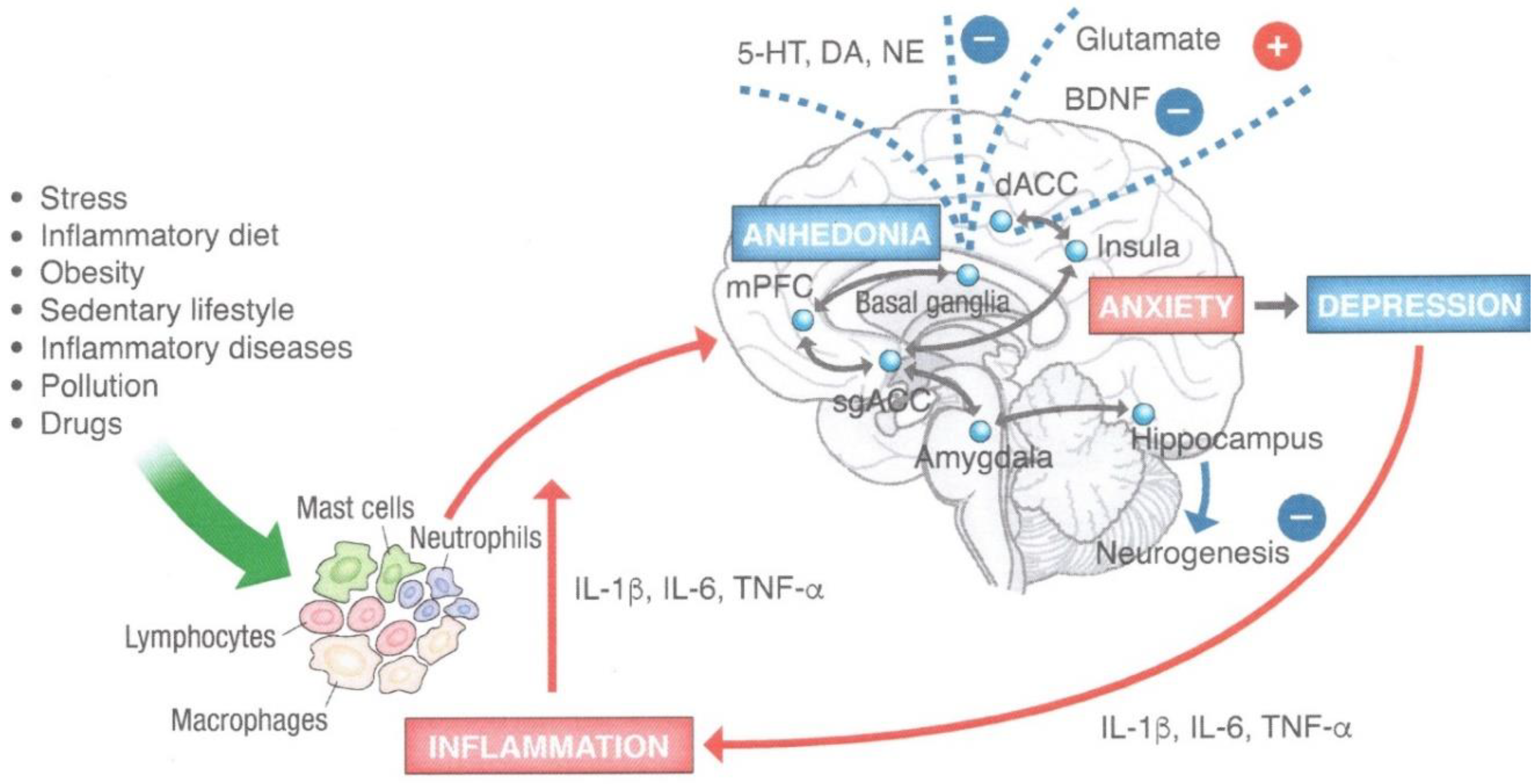
3. Inflamed Mind
4. Nutrition
5. Nutraceutical and Diet Supplements
5.1. Probiotics
5.2. Fish Oil
5.3. Zinc
5.4. N-Acetylcysteine (NAC)
5.5. Vitamin D
6. Physical Activity
7. Stress Management and Psychological Interventions
8. Conclusions
Author Contributions
Funding
Conflicts of Interest
References
- American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 5th ed.; APA: Washington, DC, USA, 2013. [Google Scholar] [CrossRef]
- Angell, M. The epidemic of mental illness: Why? N. Y. Rev. 2011, 58, 20–22. [Google Scholar]
- Gardner, C.; Kleinman, A. Medicine and the Mind—The Consequences of Psychiatry’s Identity Crisis. N. Engl. J. Med. 2019, 381, 1697–1699. [Google Scholar] [CrossRef] [PubMed]
- Voineskos, A.N.; Mulsant, B.H.; Dickie, E.W.; Neufeld, N.H.; Rothschild, A.J.; Whyte, E.M.; Meyers, B.S.; Alexopoulos, G.S.; Hoptman, M.J.; Lerch, J.P.; et al. Effects of Antipsychotic Medication on Brain Structure in Patients with Major Depressive Disorder and Psychotic Features. JAMA Psychiatry 2020, 77, 674–683. [Google Scholar] [CrossRef] [PubMed]
- Anderson, K.N.; Lind, J.N.; Simeone, R.M.; Bobo, W.V.; Mitchell, A.A.; Riehle-Colarusso, T.; Polen, K.N.; Reefhuis, J. Maternal Use of Specific Antidepressant Medications During Early Pregnancy and the Risk of Selected Birth Defects. JAMA Psychiatry 2020, 77, 1246–1255. [Google Scholar] [CrossRef]
- Skottnik, L.; Linden, D.E.J. Mental Imagery and Brain Regulation—New Links Between Psychotherapy and Neuroscience. Front. Psychiatry 2019, 10. [Google Scholar] [CrossRef]
- Jiménez, J.P.; Botto, A.; Herrera, L.; Leighton, C.; Rossi, J.L.; Quevedo, Y.; Silva, J.R.; Martinez, F.; Assar, R.; Salazar, L.A.; et al. Psychotherapy and Genetic Neuroscience: An Emerging Dialog. Front. Genet. 2018, 9, 257. [Google Scholar] [CrossRef]
- Dossett, M.L.; Fricchione, G.L.; Benson, H. A New Era for Mind-Body Medicine. N. Engl. J. Med. 2020, 382, 1390–1391. [Google Scholar] [CrossRef]
- Bottaccioli, F.; Bottaccioli, A.G. Psychoneuroendocrineimmunology and Science of Integrated Care. The Manual; Edra: Palm Beach, FL, USA, 2020. [Google Scholar]
- Sarris, J.; Logan, A.C.; Akbaraly, T.N.; Amminger, G.P.; Balanzá-Martínez, V.; Freeman, M.P.; Hibbeln, J.; Matsuoka, Y.; Mischoulon, D.; Mizoue, T.; et al. Nutritional medicine as mainstream in psychiatry. Lancet Psychiatry 2015, 2, 271–274. [Google Scholar] [CrossRef]
- Sarris, J. Nutritional Psychiatry: From Concept to the Clinic. Drugs 2019, 79, 929–934. [Google Scholar] [CrossRef]
- Mandolesi, L.; Polverino, A.; Montuori, S.; Foti, F.; Ferraioli, G.; Sorrentino, P.; Sorrentino, G.; Mandolesi, L.; Polverino, A.; Montuori, S.; et al. Effects of Physical Exercise on Cognitive Functioning and Wellbeing: Biological and Psychological Benefits. Front. Psychol. 2018, 9, 509. [Google Scholar] [CrossRef]
- Armstrong, K.; Asch, D.A. Bridging Polarization in Medicine—From Biology to Social Causes. N. Engl. J. Med. 2020, 382, 888–889. [Google Scholar] [CrossRef] [PubMed]
- Szyf, M. The epigenetics of perinatal stress. Dialogues Clin. Neurosci. 2019, 21, 369–378. [Google Scholar] [CrossRef] [PubMed]
- Tong, J.; Satyanarayanan, S.K.; Su, H. Nutraceuticals and probiotics in the management of psychiatric and neurological disorders: A focus on microbiota-gut-brain-immune axis. Brain Behav. Immun. 2020, 90, 403–419. [Google Scholar] [CrossRef]
- Khan, A.; Plana-Ripoll, O.; Antonsen, S.; Brandt, J.; Geels, C.; Landecker, H.; Sullivan, P.F.; Pedersen, C.B.; Rzhetsky, A. Environmental pollution is associated with increased risk of psychiatric disorders in the US and Denmark. PLoS Biol. 2019, 17, e3000353. [Google Scholar] [CrossRef]
- Mattson, M.P. An Evolutionary Perspective on Why Food Overconsumption Impairs Cognition. Trends Cogn. Sci. 2019, 23, 200–212. [Google Scholar] [CrossRef]
- Zhai, L.; Zhang, Y.; Zhang, D. Sedentary behaviour and the risk of depression: A meta-analysis. Br. J. Sports Med. 2014, 49, 705–709. [Google Scholar] [CrossRef] [PubMed]
- McDowell, C.P.; Dishman, R.K.; Gordon, B.; Herring, M.P. Physical Activity and Anxiety: A Systematic Review and Meta-analysis of Prospective Cohort Studies. Am. J. Prev. Med. 2019, 57, 545–556. [Google Scholar] [CrossRef] [PubMed]
- Stewart-Brown, S.; Samaraweera, P.C.; Taggart, F.; Kandala, N.-B.; Stranges, S. Socioeconomic gradients and mental health: Implications for public health. Br. J. Psychiatry 2015, 206, 461–465. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Giebel, C.; Corcoran, R.; Goodall, M.; Campbell, N.; Gabbay, M.; Daras, K.; Barr, B.; Wilson, T.; Kullu, C. Do people living in disadvantaged circumstances receive different mental health treatments than those from less disadvantaged backgrounds? BMC Public Health 2020, 20, 1–10. [Google Scholar] [CrossRef]
- Shoaff, J.R.; Coull, B.; Weuve, J.; Bellinger, D.C.; Calafat, A.M.; Schantz, S.L.; Korrick, S.A. Association of Exposure to Endocrine-Disrupting Chemicals During Adolescence with Attention-Deficit/Hyperactivity Disorder–Related Behaviors. JAMA Netw. Open 2020, 3, e2015041. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; Qi, W.; Xu, Q.; Wang, Z.; Cao, X.; Zhou, L.; Ye, L. The role of epigenetics in the reproductive toxicity of environmental endocrine disruptors. Environ. Mol. Mutagen. 2020, 62, 78–88. [Google Scholar] [CrossRef]
- Pariante, C.M. Why are depressed patients inflamed? A reflection on 20 years of research on depression, glucocorticoid resistance and inflammation. Eur. Neuropsychopharmacol. 2017, 27, 554–559. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bauer, M.E.; Teixeira, A.L. Inflammation in psychiatric disorders: What comes first? Ann. N. Y. Acad. Sci. 2018, 1437, 57–67. [Google Scholar] [CrossRef] [PubMed]
- Osimo, E.F.; Baxter, L.J.; Lewis, G.; Jones, P.B.; Khandaker, G.M. Prevalence of low-grade inflammation in depression: A systematic review and meta-analysis of CRP levels. Psychol. Med. 2019, 49, 1958–1970. [Google Scholar] [CrossRef] [PubMed]
- Osimo, E.F.; Pillinger, T.; Rodriguez, I.M.; Khandaker, G.M.; Pariante, C.M.; Howes, O.D. Inflammatory markers in depression: A meta-analysis of mean differences and variability in 5166 patients and 5083 controls. Brain Behav. Immun. 2020, 87, 901–909. [Google Scholar] [CrossRef] [PubMed]
- Jeppesen, R.; Christensen, R.H.; Pedersen, E.M.; Nordentoft, M.; Hjorthøj, C.; Köhler-Forsberg, O.; Benros, M.E. Efficacy and safety of anti-inflammatory agents in treatment of psychotic disorders—A comprehensive systematic review and meta-analysis. Brain Behav. Immun. 2020, 90, 364–380. [Google Scholar] [CrossRef] [PubMed]
- Harrison, N.A.; Cercignani, M.; Voon, V.; Critchley, H.D. Effects of Inflammation on Hippocampus and Substantia Nigra Responses to Novelty in Healthy Human Participants. Neuropsychopharmacology 2014, 40, 831–838. [Google Scholar] [CrossRef]
- Harrison, N.A.; Voon, V.; Cercignani, M.; Cooper, E.A.; Pessiglione, M.; Critchley, H. A Neurocomputational Account of How Inflammation Enhances Sensitivity to Punishments Versus Rewards. Biol. Psychiatry 2016, 80, 73–81. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Köhler, O.; Petersen, L.V.; Mors, O.; Gasse, C. Inflammation and depression: Combined use of selective serotonin reuptake inhibitors and NSAIDs or paracetamol and psychiatric outcomes. Brain Behav. 2015, 5, e00338. [Google Scholar] [CrossRef] [PubMed]
- Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D.W.; Fasano, A.; Miller, G.W.; et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019, 25, 1822–1832. [Google Scholar] [CrossRef] [PubMed]
- WHO; FAO Expert Consultation. Diet, Nutrition and the Prevention of Chronic Diseases; WHO: Geneva, Switzerland, 2013.
- Firth, J.; Stubbs, B.; Teasdale, S.; Ward, P.; Veronese, N.; Shivappa, N.; Hebert, J.R.; Berk, M.; Yung, A.; Sarris, J. Diet as a hot topic in psychiatry: A population-scale study of nutritional intake and inflammatory potential in severe mental illness. World Psychiatry 2018, 17, 365–367. [Google Scholar] [CrossRef]
- Zhang, X.; Zhang, G.; Zhang, H.; Karin, M.; Bai, H.; Cai, D. Hypothalamic IKKβ/NF-kB and ER stress Link Overnutrition. Cell 2009, 135, 61–73. [Google Scholar] [CrossRef] [Green Version]
- Marx, W.; Lane, M.; Hockey, M.; Aslam, H.; Berk, M.; Walder, K.; Borsini, A.; Firth, J.; Pariante, C.M.; Berding, K.; et al. Diet and depression: Exploring the biological mechanisms of action. Mol. Psychiatry 2020, 26, 134–150. [Google Scholar] [CrossRef]
- Nucci, D.; Fatigoni, C.; Amerio, A.; Odone, A.; Gianfredi, V. Red and Processed Meat Consumption and Risk of Depression: A Systematic Review and Meta-Analysis. Int. J. Environ. Res. Public Health 2020, 17, 6686. [Google Scholar] [CrossRef] [PubMed]
- Fu, Y.; Kaneko, K.; Lin, H.-Y.; Mo, Q.; Xu, Y.; Suganami, T.; Ravn, P.; Fukuda, M. Gut Hormone GIP Induces Inflammation and Insulin Resistance in the Hypothalamus. Endocrinology 2020, 161. [Google Scholar] [CrossRef]
- Damasio, A. The Strange Order of Things; Vintage: New York, NY, USA, 2018. [Google Scholar]
- Rao, M.; Gershon, M.D. The bowel and beyond: The enteric nervous system in neurological disorders. Nat. Rev. Gastroenterol. Hepatol. 2016, 13, 517–528. [Google Scholar] [CrossRef] [Green Version]
- Long-Smith, C.; O’Riordan, K.J.; Clarke, G.; Stanton, C.; Dinan, T.G.; Cryan, J.F. Microbiota-Gut-Brain Axis: New Therapeutic Opportunities. Annu. Rev. Pharmacol. Toxicol. 2020, 60, 477–502. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ojeda, J.; Ávila, A.; Vidal, P. Gut Microbiota Interaction with the Central Nervous System throughout Life. J. Clin. Med. 2021, 10, 1299. [Google Scholar] [CrossRef]
- Łoniewski, I.; Misera, A.; Skonieczna-Żydecka, K.; Kaczmarczyk, M.; Kaźmierczak-Siedlecka, K.; Misiak, B.; Marlicz, W.; Samochowiec, J. Major Depressive Disorder and gut microbiota—Association not causation. A scoping review. Prog. Neuro Psychopharmacol. Biol. Psychiatry 2021, 106, 110111. [Google Scholar] [CrossRef] [PubMed]
- Leclercq, S.; Le Roy, T.; Furgiuele, S.; Coste, V.; Bindels, L.B.; Leyrolle, Q.; Neyrinck, A.M.; Quoilin, C.; Amadieu, C.; Petit, G.; et al. Gut Microbiota-Induced Changes in β-Hydroxybutyrate Metabolism Are Linked to Altered Sociability and Depression in Alcohol Use Disorder. Cell Rep. 2020, 33, 108238. [Google Scholar] [CrossRef]
- Lassale, C.; Batty, G.; Baghdadli, A.; Jacka, F.; Villegas, A.S.; Kivimäki, M.; Akbaraly, T. Healthy dietary indices and risk of depressive outcomes: A systematic review and meta-analysis of observational studies. Mol. Psychiatry 2018, 24, 965–986. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dinan, T.G.; Stanton, C.; Cryan, J. Psychobiotics: A Novel Class of Psychotropic. Biol. Psychiatry 2013, 74, 720–726. [Google Scholar] [CrossRef] [PubMed]
- Sarkar, A.; Lehto, S.; Harty, S.; Dinan, T.; Cryan, J.F.; Burnet, P.W. Psychobiotics and the Manipulation of Bacteria–Gut–Brain Signals. Trends Neurosci. 2016, 39, 763–781. [Google Scholar] [CrossRef] [Green Version]
- Cheng, L.-H.; Liu, Y.-W.; Wu, C.-C.; Wang, S.; Tsai, Y.-C. Psychobiotics in mental health, neurodegenerative and neurodevelopmental disorders. J. Food Drug Anal. 2019, 27, 632–648. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Allen, A.P.; Hutch, W.; Borre, Y.; Kennedy, P.J.; Temko, A.; Boylan, G.; Murphy, E.F.; Cryan, J.F.; Dinan, T.G.; Clarke, G. Bifidobacterium longum 1714 as a translational psychobiotic: Modulation of stress, electrophysiology and neurocognition in healthy volunteers. Transl. Psychiatry 2016, 6, e939. [Google Scholar] [CrossRef] [Green Version]
- Gualtieri, P.; Marchetti, M.; Cioccoloni, G.; De Lorenzo, A.; Romano, L.; Cammarano, A.; Colica, C.; Condò, R.; Di Renzo, L. Psychobiotics Regulate the Anxiety Symptoms in Carriers of Allele A of IL-1βGene: A Randomized, Placebo-Controlled Clinical Trial. Mediat. Inflamm. 2020, 2020, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Chao, L.; Liu, C.; Sutthawongwadee, S.; Li, Y.; Lv, W.; Chen, W.; Yu, L.; Zhou, J.; Guo, A.; Li, Z.; et al. Effects of Probiotics on Depressive or Anxiety Variables in Healthy Participants Under Stress Conditions or with a Depressive or Anxiety Diagnosis: A Meta-Analysis of Randomized Controlled Trials. Front. Neurol. 2020, 11, 421. [Google Scholar] [CrossRef] [PubMed]
- Zhang, N.; Zhang, Y.; Li, M.; Wang, W.; Liu, Z.; Xi, C.; Huang, X.; Liu, J.; Huang, J.; Tian, D.; et al. Efficacy of probiotics on stress in healthy volunteers: A systematic review and meta-analysis based on randomized controlled trials. Brain Behav. 2020, 10, e01699. [Google Scholar] [CrossRef]
- Lin, P.-Y.; Huang, S.-Y.; Su, K.-P. A Meta-Analytic Review of Polyunsaturated Fatty Acid Compositions in Patients with Depression. Biol. Psychiatry 2010, 68, 140–147. [Google Scholar] [CrossRef]
- Su, K.-P. Nutrition, psychoneuroimmunology and depression: The therapeutic implications of omega-3 fatty acids in interferon-α-induced depression. Biomedicine 2015, 5, 1–7. [Google Scholar] [CrossRef]
- Appleton, K.M.; Sallis, H.M.; Perry, R.; Ness, A.R.; Churchill, R. Omega-3 fatty acids for depression in adults. Cochrane Database Syst. Rev. 2015, 11. [Google Scholar] [CrossRef] [PubMed]
- Guu, T.-W.; Mischoulon, D.; Sarris, J.; Hibbeln, J.; McNamara, R.K.; Hamazaki, K.; Freeman, M.P.; Maes, M.; Matsuoka, Y.J.; Belmaker, R.; et al. A multi-national, multi-disciplinary Delphi consensus study on using omega-3 polyunsaturated fatty acids (n-3 PUFAs) for the treatment of major depressive disorder. J. Affect. Disord. 2020, 265, 233–238. [Google Scholar] [CrossRef]
- Xu, F.; Fan, W.; Wang, W.; Tang, W.; Yang, F.; Zhang, Y.; Cai, J.; Song, L.; Zhang, C. Effects of omega-3 fatty acids on metabolic syndrome in patients with schizophrenia: A 12-week randomized placebo-controlled trial. Psychopharmacology 2018, 236, 1273–1279. [Google Scholar] [CrossRef]
- Tang, W.; Wang, Y.; Xu, F.; Fan, W.; Zhang, Y.; Fan, K.; Wang, W.; Zhang, Y.; Zhang, C. Omega-3 fatty acids ameliorate cognitive dysfunction in schizophrenia patients with metabolic syndrome. Brain Behav. Immun. 2020, 88, 529–534. [Google Scholar] [CrossRef] [PubMed]
- Bozzatello, P.; De Rosa, M.; Rocca, P.; Bellino, S. Effects of Omega 3 Fatty Acids on Main Dimensions of Psychopathology. Int. J. Mol. Sci. 2020, 21, 6042. [Google Scholar] [CrossRef] [PubMed]
- Amminger, G.P.; Schäfer, M.R.; Papageorgiou, K.; Klier, C.; Cotton, S.; Harrigan, S.; Mackinnon, A.; McGorry, P.D.; Berger, G.E. Long-Chain ω-3 Fatty Acids for Indicated Prevention of Psychotic Disorders. Arch. Gen. Psychiatry 2010, 67, 146–154. [Google Scholar] [CrossRef]
- Amminger, G.P.; Schäfer, M.R.; Schlögelhofer, M.; Klier, C.; McGorry, P.D. Longer-term outcome in the prevention of psychotic disorders by the Vienna omega-3 study. Nat. Commun. 2015, 6, 7934. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- McGorry, P.D.; Nelson, B.; Markulev, C.; Yuen, H.P.; Schäfer, M.R.; Mossaheb, N.; Schlögelhofer, M.; Smesny, S.; Hickie, I.; Berger, G.E.; et al. Effect of ω-3 Polyunsaturated Fatty Acids in Young People at Ultrahigh Risk for Psychotic Disorders. JAMA Psychiatry 2017, 74, 19–27. [Google Scholar] [CrossRef]
- Berger, M.; Nelson, B.; Markulev, C.; Yuen, H.P.; Schäfer, M.R.; Mossaheb, N.; Schlögelhofer, M.; Smesny, S.; Hickie, I.B.; Berger, G.E.; et al. Relationship Between Polyunsaturated Fatty Acids and Psychopathology in the NEURAPRO Clinical Trial. Front. Psychiatry 2019, 10. [Google Scholar] [CrossRef]
- Chasapis, C.T.; Ntoupa, P.-S.A.; Spiliopoulou, C.A.; Stefanidou, M.E. Recent aspects of the effects of zinc on human health. Arch. Toxicol. 2020, 94, 1443–1460. [Google Scholar] [CrossRef]
- Yasuda, H.; Tsutsui, T. Infants and elderlies are susceptible to zinc deficiency. Sci. Rep. 2016, 6, 21850. [Google Scholar] [CrossRef] [Green Version]
- Jung, A.; Spira, D.; Steinhagen-Thiessen, E.; Demuth, I.; Norman, K. Zinc Deficiency Is associated with Depressive Symptoms—Results from the Berlin Aging Study II. J. Gerontol. Ser. A Boil. Sci. Med. Sci. 2016, 72, 1149–1154. [Google Scholar] [CrossRef]
- Petrilli, M.A.; Kranz, T.M.; Kleinhaus, K.; Joe, P.; Getz, M.; Johnson, P.; Chao, M.; Malaspina, D. The Emerging Role for Zinc in Depression and Psychosis. Front. Pharmacol. 2017, 8, 414. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nowak, G. Zinc, future mono/adjunctive therapy for depression: Mechanisms of antidepressant action. Pharmacol. Rep. 2015, 67, 659–662. [Google Scholar] [CrossRef] [PubMed]
- Portbury, S.D.; Adlard, P.A. Zinc Signal in Brain Diseases. Int. J. Mol. Sci. 2017, 18, 2506. [Google Scholar] [CrossRef] [Green Version]
- Zalachoras, I.; Hollis, F.; Ramos-Fernández, E.; Trovo, L.; Sonnay, S.; Geiser, E.; Preitner, N.; Steiner, P.; Sandi, C.; Morató, L. Therapeutic potential of glutathione-enhancers in stress-related psychopathologies. Neurosci. Biobehav. Rev. 2020, 114, 134–155. [Google Scholar] [CrossRef]
- Paydary, K.; Akamaloo, A.; Ahmadipour, A.; Pishgar, F.; Emamzadehfard, S.; Akhondzadeh, S. N-acetylcysteine augmentation therapy for moderate-to-severe obsessive-compulsive disorder: Randomized, double-blind, placebo-controlled trial. J. Clin. Pharm. Ther. 2016, 41, 214–219. [Google Scholar] [CrossRef] [PubMed]
- Costa, D.L.C.; Diniz, J.B.; Requena, G.; Joaquim, M.A.; Pittenger, C.; Bloch, M.H.; Miguel, E.C.; Shavitt, R.G. Randomized, Double-Blind, Placebo-Controlled Trial of N-Acetylcysteine Augmentation for Treatment-Resistant Obsessive-Compulsive Disorder. J. Clin. Psychiatry 2017, 78, e766–e773. [Google Scholar] [CrossRef]
- Fineberg, N.A.; Hollander, E.; Pallanti, S.; Walitza, S.; Grünblatt, E.; Dell’Osso, B.M.; Albert, U.; Geller, D.A.; Brakoulias, V.; Reddy, Y.J.; et al. Clinical advances in obsessive-compulsive disorder. Int. Clin. Psychopharmacol. 2020, 35, 173–193. [Google Scholar] [CrossRef]
- Berk, M.; Dean, O.M.; Cotton, S.M.; Jeavons, S.; Tanious, M.; Kohlmann, K.; Hewitt, K.; Moss, K.; Allwang, C.; Schapkaitz, I.; et al. The Efficacy of AdjunctiveN-Acetylcysteine in Major Depressive Disorder. J. Clin. Psychiatry 2014, 75, 628–636. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Deepmala; Slattery, J.; Kumar, N.; Delhey, L.; Berk, M.; Dean, O.; Spielholz, C.; Frye, R. Clinical trials of N-acetylcysteine in psychiatry and neurology: A systematic review. Neurosci. Biobehav. Rev. 2015, 55, 294–321. [Google Scholar] [CrossRef] [Green Version]
- Kishi, T.; Miyake, N.; Okuya, M.; Sakuma, K.; Iwata, N. N-acetylcysteine as an adjunctive treatment for bipolar depression and major depressive disorder: A systematic review and meta-analysis of double-blind, randomized placebo-controlled trials. Psychopharmacology 2020, 237, 3481–3487. [Google Scholar] [CrossRef]
- Chakraborty, S.; Tripathi, S.J.; Raju, T.R.; Rao, B.S. Mechanisms underlying remediation of depression-associated anxiety by chronic N-acetyl cysteine treatment. Psychopharmacology 2020, 237, 2967–2981. [Google Scholar] [CrossRef]
- Von Känel, R.; Fardad, N.; Steurer, N.; Horak, N.; Hindermann, E.; Fischer, F.; Gessler, K. Vitamin D Deficiency and Depressive Symptomatology in Psychiatric Patients Hospitalized with a Current Depressive Episode: A Factor Analytic Study. PLoS ONE 2015, 10, e0138550. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sepehrmanesh, Z.; Kolahdooz, F.; Abedi, F.; Mazroii, N.; Assarian, A.; Asemi, Z.; Esmaillzadeh, A. Retracted: Vitamin D Supplementation Affects the Beck Depression Inventory, Insulin Resistance, and Biomarkers of Oxidative Stress in Patients with Major Depressive Disorder: A Randomized, Controlled Clinical Trial. J. Nutr. 2015, 146, 243–248. [Google Scholar] [CrossRef] [Green Version]
- Ronaldson, A.; De La Torre, J.A.; Gaughran, F.; Bakolis, I.; Hatch, S.L.; Hotopf, M.; Dregan, A. Prospective associations between vitamin D and depression in middle-aged adults: Findings from the UK Biobank cohort. Psychol. Med. 2020, 1–9. [Google Scholar] [CrossRef]
- Amini, S.; Jafarirad, S.; Amani, R. Postpartum depression and vitamin D: A systematic review. Crit. Rev. Food Sci. Nutr. 2018, 59, 1514–1520. [Google Scholar] [CrossRef]
- Barbour, K.A.; Edenfield, T.M.; Blumenthal, J.A. Exercise as a treatment for depression and other psychiatric disorders: A review. J. Cardiopulm. Rehabil. Prev. 2007, 27, 359–367. [Google Scholar] [CrossRef]
- RCP. No Health without Public Mental Health: The Case for Action; Position Statement PS4/2010; Royal College of Psychiatrists: London, UK, 2010. [Google Scholar]
- Stubbs, B.; Vancampfort, D.; Hallgren, M.; Firth, J.; Veronese, N.; Solmi, M.; Brand, S.; Cordes, J.; Malchow, B.; Gerber, M.; et al. EPA guidance on physical activity as a treatment for severe mental illness: A meta-review of the evidence and Position Statement from the European Psychiatric Association (EPA), supported by the International Organization of Physical Therapists in Mental Health (IOPTMH). Eur. Psychiatry 2018, 54, 124–144. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Firth, J.; Solmi, M.; Wootton, R.E.; Vancampfort, D.; Schuch, F.B.; Hoare, E.; Gilbody, S.; Torous, J.; Teasdale, S.B.; Jackson, S.E.; et al. A meta-review of “lifestyle psychiatry”: The role of exercise, smoking, diet and sleep in the prevention and treatment of mental disorders. World Psychiatry 2020, 19, 360–380. [Google Scholar] [CrossRef]
- Ashdown-Franks, G.; Firth, J.; Carney, R.; Carvalho, A.F.; Hallgren, M.; Koyanagi, A.; Rosenbaum, S.; Schuch, F.; Smith, L.; Solmi, M.; et al. Exercise as Medicine for Mental and Substance Use Disorders: A Meta-review of the Benefits for Neuropsychiatric and Cognitive Outcomes. Sports Med. 2019, 50, 151–170. [Google Scholar] [CrossRef]
- Russell, T.A.; Arcuri, S.M. A Neurophysiological and Neuropsychological Consideration of Mindful Movement: Clinical and Research Implications. Front. Hum. Neurosci. 2015, 9, 282. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kong, J.; Wilson, G.; Park, J.; Pereira, K.; Walpole, C.; Yeung, A. Treating Depression with Tai Chi: State of the Art and Future Perspectives. Front. Psychiatry 2019, 10, 237. [Google Scholar] [CrossRef] [Green Version]
- Liegro, D.; Schiera, G.; Proia, P.; Di Liegro, C.M.; Di Liegro, I. Physical Activity and Brain Health. Genes 2019, 10, 720. [Google Scholar] [CrossRef] [Green Version]
- Raglin, S.J. Exercise and Mental Health. Sport Med. 1990, 9, 323–329. [Google Scholar] [CrossRef]
- Salmon, P. Effects of physical exercise on anxiety, depression, and sensitivity to stress: A unifying theory. Clin. Psychol. Rev. 2001, 21, 33–61. [Google Scholar] [CrossRef]
- Mikkelsen, K.; Stojanovska, L.; Polenakovic, M.; Bosevski, M.; Apostolopoulos, V. Exercise and mental health. Maturitas 2017, 106, 48–56. [Google Scholar] [CrossRef]
- Riva, A.; Falbo, M.; Passoni, P.; Polizzi, S.; Cattoni, A.; Nacinovich, R. High levels of physical activity in female adolescents with anorexia nervosa: Medical and psychopathological correlates. Eat. Weight Disord. 2021, 1–12. [Google Scholar] [CrossRef]
- Balciuniene, V.; Jankauskiene, R.; Baceviciene, M. Effect of an education and mindfulness-based physical activity intervention for the promotion of positive body image in Lithuanian female students. Eat. Weight Disord. 2021, 1–15. [Google Scholar] [CrossRef]
- Fossati, M.; Amati, F.; Painot, D.; Reiner, M.; Haenni, C.; Golay, A. Cognitive-behavioral therapy with simultaneous nutritional and physical activity education in obese patients with binge eating disorder. Eat. Weight Disord. Stud. Anorexia Bulim. Obes. 2004, 9, 134–138. [Google Scholar] [CrossRef]
- McEwen, B.S.; Akil, H. Revisiting the Stress Concept: Implications for Affective Disorders. J. Neurosci. 2020, 40, 12–21. [Google Scholar] [CrossRef] [Green Version]
- McEwen, B.S.; Nasca, C.; Gray, J.D. Stress Effects on Neuronal Structure: Hippocampus, Amygdala, and Prefrontal Cortex. Neuropsychopharmacology 2015, 41, 3–23. [Google Scholar] [CrossRef] [Green Version]
- Slavich, G.M. Psychoneuroimmunology of Stress and Mental Health. In The Oxford Handbook of Stress and Mental Health; Oxford Handbooks Online; Oxford University Press: Oxford, UK, 2020. [Google Scholar] [CrossRef] [Green Version]
- McEwen, B.S. Neurobiological and Systemic Effects of Chronic Stress. Chronic Stress 2017, 1. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schiele, M.; Gottschalk, M.; Domschke, K. The applied implications of epigenetics in anxiety, affective and stress-related disorders—A review and synthesis on psychosocial stress, psychotherapy and prevention. Clin. Psychol. Rev. 2020, 77, 101830. [Google Scholar] [CrossRef]
- Bey, K.; Campos-Martin, R.; Klawohn, J.; Reuter, B.; Grützmann, R.; Riesel, A.; Wagner, M.; Ramirez, A.; Kathmann, N. Hypermethylation of the oxytocin receptor gene (OXTR) in obsessive-compulsive disorder: Further evidence for a biomarker of disease and treatment response. Epigenetics 2021, 1–11. [Google Scholar] [CrossRef] [PubMed]
- Bottaccioli, A.G.; Bottaccioli, F.; Minelli, A. Stress and the psyche-brain-immune network in psychiatric diseases based on psychoneuroendocrineimmunology: A concise review. Ann. N. Y. Acad. Sci. 2018, 1437, 31–42. [Google Scholar] [CrossRef]
- Miller, C.W.T. Epigenetic and Neural Circuitry Landscape of Psychotherapeutic Interventions. Psychiatry J. 2017, 2017, 1–38. [Google Scholar] [CrossRef] [Green Version]
- Yehuda, R.; Daskalakis, N.P.; Desarnaud, F.; Makotkine, I.; Lehrner, A.L.; Koch, E.; Flory, J.D.; Buxbaum, J.D.; Meaney, M.J.; Bierer, L.M. Epigenetic Biomarkers as Predictors and Correlates of Symptom Improvement Following Psychotherapy in Combat Veterans with PTSD. Front. Psychiatry 2013, 4, 118. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vinkers, C.H.; Geuze, E.; van Rooij, S.J.H.; Kennis, M.; Schür, R.R.; Nispeling, D.M.; Smith, A.K.; Nievergelt, C.M.; Uddin, M.; Rutten, B.P.F.; et al. Successful treatment of post-traumatic stress disorder reverses DNA methylation marks. Mol. Psychiatry 2019, 26, 1264–1271. [Google Scholar] [CrossRef]
- Bower, J.E.; Irwin, M.R. Mind–body therapies and control of inflammatory biology: A descriptive review. Brain Behav. Immun. 2016, 51, 1–11. [Google Scholar] [CrossRef] [Green Version]
- Bottaccioli, F.; Carosella, A.; Cardone, R.; Mambelli, M.; Cemin, M.; D’Errico, M.M.; Ponzio, E.; Bottaccioli, A.G.; Minelli, A. Brief Training of Psychoneuroendocrinoimmunology-Based Meditation (PNEIMED) Reduces Stress Symptom Ratings and Improves Control on Salivary Cortisol Secretion Under Basal and Stimulated Conditions. Explor. J. Sci. Health 2014, 10, 170–179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dasanayaka, N.N.; Sirisena, N.D.; Samaranayake, N. The effects of meditation on length of telomeres in healthy individuals: A systematic review. Syst. Rev. 2021, 10, 1–6. [Google Scholar] [CrossRef] [PubMed]
- Shields, G.S.; Spahr, C.M.; Slavich, G.M. Psychosocial Interventions and Immune System Function. JAMA Psychiatry 2020, 77, 1031. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Bottaccioli, F.; Bottaccioli, A.G.; Marzola, E.; Longo, P.; Minelli, A.; Abbate-Daga, G. Nutrition, Exercise, and Stress Management for Treatment and Prevention of Psychiatric Disorders. A Narrative Review Psychoneuroendocrineimmunology-Based. Endocrines 2021, 2, 226-240. https://doi.org/10.3390/endocrines2030022
Bottaccioli F, Bottaccioli AG, Marzola E, Longo P, Minelli A, Abbate-Daga G. Nutrition, Exercise, and Stress Management for Treatment and Prevention of Psychiatric Disorders. A Narrative Review Psychoneuroendocrineimmunology-Based. Endocrines. 2021; 2(3):226-240. https://doi.org/10.3390/endocrines2030022
Chicago/Turabian StyleBottaccioli, Francesco, Anna Giulia Bottaccioli, Enrica Marzola, Paola Longo, Andrea Minelli, and Giovanni Abbate-Daga. 2021. "Nutrition, Exercise, and Stress Management for Treatment and Prevention of Psychiatric Disorders. A Narrative Review Psychoneuroendocrineimmunology-Based" Endocrines 2, no. 3: 226-240. https://doi.org/10.3390/endocrines2030022
APA StyleBottaccioli, F., Bottaccioli, A. G., Marzola, E., Longo, P., Minelli, A., & Abbate-Daga, G. (2021). Nutrition, Exercise, and Stress Management for Treatment and Prevention of Psychiatric Disorders. A Narrative Review Psychoneuroendocrineimmunology-Based. Endocrines, 2(3), 226-240. https://doi.org/10.3390/endocrines2030022





