Testosterone Boosters Intake in Athletes: Current Evidence and Further Directions
Abstract
1. Introduction
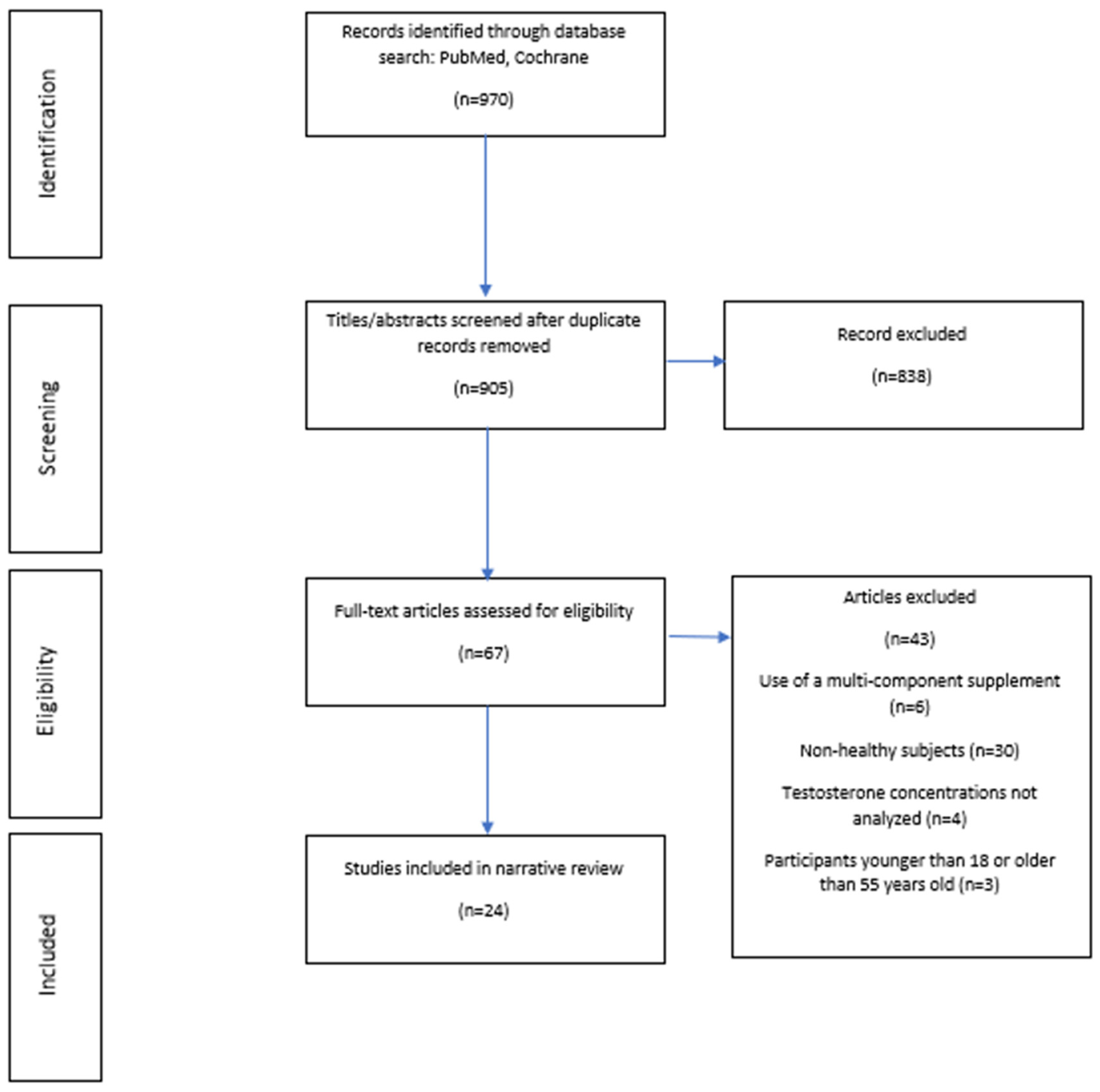
2. Materials and Methods
3. Results
3.1. L-Arginine
3.2. L-Carnitine
3.3. Magnesium
3.4. Selenium
3.5. Shilajit
3.6. Eurycoma Longifolia
3.7. Serenoa Repens
3.8. Boron
3.9. Ashwagandha Root
3.10. Fenugreek
4. Discussion
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Bhasin, S.; Woodhouse, L.; Casaburi, R.; Singh, A.B.; Bhasin, D.; Berman, N.; Chen, X.; Yarasheski, K.E.; Magliano, L.; Dzekov, C.; et al. Testosterone Dose-Response Relationships in Healthy Young Men. Am. J. Physiol. Endocrinol. Metab. 2001, 281, E1172–E1181. [Google Scholar] [CrossRef]
- Finkelstein, J.S.; Lee, H.; Burnett-Bowie, S.-A.M.; Pallais, J.C.; Yu, E.W.; Borges, L.F.; Jones, B.F.; Barry, C.V.; Wulczyn, K.E.; Thomas, B.J.; et al. Gonadal Steroids and Body Composition, Strength, and Sexual Function in Men. N. Engl. J. Med. 2013, 369, 1011–1022. [Google Scholar] [CrossRef]
- Geyer, H.; Schänzer, W.; Thevis, M. Anabolic Agents: Recent Strategies for Their Detection and Protection from Inadvertent Doping. Br. J. Sports Med. 2014, 48, 820–826. [Google Scholar] [CrossRef]
- Hooper, D.R.; Tenforde, A.S.; Hackney, A.C. Treating Exercise-Associated Low Testosterone and Its Related Symptoms. Physician Sportsmed. 2018, 46, 427–434. [Google Scholar] [CrossRef]
- Leitão, A.E.; de Souza Vieira, M.C.; Pelegrini, A.; da Silva, E.L.; de Azevedo Guimarães, A.C. A 6-Month, Double-Blind, Placebo-Controlled, Randomized Trial to Evaluate the Effect of Eurycoma Longifolia (Tongkat Ali) and Concurrent Training on Erectile Function and Testosterone Levels in Androgen Deficiency of Aging Males (ADAM). Maturitas 2021, 145, 78–85. [Google Scholar] [CrossRef]
- Rao, A.; Steels, E.; Inder, W.J.; Abraham, S.; Vitetta, L. Testofen, a Specialised Trigonella Foenum-Graecum Seed Extract Reduces Age-Related Symptoms of Androgen Decrease, Increases Testosterone Levels and Improves Sexual Function in Healthy Aging Males in a Double-Blind Randomised Clinical Study. Aging Male Off. J. Int. Soc. Study Aging Male 2016, 19, 134–142. [Google Scholar] [CrossRef]
- Tambi, M.I.B.M.; Imran, M.K.; Henkel, R.R. Standardised Water-Soluble Extract of Eurycoma Longifolia, Tongkat Ali, as Testosterone Booster for Managing Men with Late-Onset Hypogonadism? Andrologia 2012, 44 (Suppl. 1), 226–230. [Google Scholar] [CrossRef]
- Sudeep, H.V.; Thomas, J.V.; Shyamprasad, K. A Double Blind, Placebo-Controlled Randomized Comparative Study on the Efficacy of Phytosterol-Enriched and Conventional Saw Palmetto Oil in Mitigating Benign Prostate Hyperplasia and Androgen Deficiency. BMC Urol. 2020, 20, 86. [Google Scholar] [CrossRef]
- Koumanov, F.; Bozadjieva, E.; Andreeva, M.; Platonva, E.; Ankov, V. Clinical Trial of Tribestan. Exp. Med. 1982, 4, 211–215. [Google Scholar]
- Pokrywka, A.; Obmiński, Z.; Malczewska-Lenczowska, J.; Fijałek, Z.; Turek-Lepa, E.; Grucza, R. Insights into Supplements with Tribulus Terrestris Used by Athletes. J. Hum. Kinet. 2014, 41, 99–105. [Google Scholar] [CrossRef]
- Chen, C.K.; Ooi, F.K.; Abu Kasim, N.A.; Asari, M.A. Effects of Eurycoma Longifolia Jack Supplementation Combined with Resistance Training on Isokinetic Muscular Strength and Power, Anaerobic Power, and Urinary Testosterone: Epitestosterone Ratio in Young Males. Int. J. Prev. Med. 2019, 10, 118. [Google Scholar] [CrossRef] [PubMed]
- Ferrando, A.A.; Green, N.R. The Effect of Boron Supplementation on Lean Body Mass, Plasma Testosterone Levels, and Strength in Male Bodybuilders. Int. J. Sport Nutr. 1993, 3, 140–149. [Google Scholar] [CrossRef] [PubMed]
- Wankhede, S.; Langade, D.; Joshi, K.; Sinha, S.R.; Bhattacharyya, S. Examining the Effect of Withania Somnifera Supplementation on Muscle Strength and Recovery: A Randomized Controlled Trial. J. Int. Soc. Sports Nutr. 2015, 12, 43. [Google Scholar] [CrossRef]
- Sandhu, J.S.; Shah, B.; Shenoy, S.; Chauhan, S.; Lavekar, G.S.; Padhi, M.M. Effects of Withania Somnifera (Ashwagandha) and Terminalia Arjuna (Arjuna) on Physical Performance and Cardiorespiratory Endurance in Healthy Young Adults. Int. J. Ayurveda Res. 2010, 1, 144–149. [Google Scholar] [CrossRef]
- Clemesha, C.G.; Thaker, H.; Samplaski, M.K. “Testosterone Boosting” Supplements Composition and Claims Are Not Supported by the Academic Literature. World J. Men’s Health 2020, 38, 115–122. [Google Scholar] [CrossRef]
- Balasubramanian, A.; Thirumavalavan, N.; Srivatsav, A.; Yu, J.; Lipshultz, L.I.; Pastuszak, A.W. Testosterone Imposters: An Analysis of Popular Online Testosterone Boosting Supplements. J. Sex. Med. 2019, 16, 203–212. [Google Scholar] [CrossRef]
- McConell, G.K. Effects of L-Arginine Supplementation on Exercise Metabolism. Curr. Opin. Clin. Nutr. Metab. Care 2007, 10, 46–51. [Google Scholar] [CrossRef]
- McRae, M.P. Therapeutic Benefits of L-Arginine: An Umbrella Review of Meta-Analyses. J. Chiropr. Med. 2016, 15, 184–189. [Google Scholar] [CrossRef]
- Abel, T.; Knechtle, B.; Perret, C.; Eser, P.; von Arx, P.; Knecht, H. Influence of Chronic Supplementation of Arginine Aspartate in Endurance Athletes on Performance and Substrate Metabolism—A Randomized, Double-Blind, Placebo-Controlled Study. Int. J. Sports Med. 2005, 26, 344–349. [Google Scholar] [CrossRef]
- Andrade, W.B.; Jacinto, J.L.; da Silva, D.K.; Roveratti, M.C.; Estoche, J.M.; Oliveira, D.B.; Balvedi, M.C.W.; da Silva, R.A.; Aguiar, A.F. L-Arginine Supplementation Does Not Improve Muscle Function during Recovery from Resistance Exercise. Appl. Physiol. Nutr. Metab. Physiol. Appl. Nutr. Metab. 2018, 43, 928–936. [Google Scholar] [CrossRef]
- Moosakhani, A.; Choobineh, S.; Soori, R. Effect of L-Arginine Alpha Ketoglutarate Supplementation on Hormonal Response during Recovery in Male Endurance Swimmers. Gazz. Med. Ital. Arch. Sci. Med. 2018, 177, 65–71. [Google Scholar] [CrossRef]
- Hsu, M.-C.; Chien, K.-Y.; Hsu, C.-C.; Chung, C.-J.; Chan, K.-H.; Su, B. Effects of BCAA, Arginine and Carbohydrate Combined Drink on Post-Exercise Biochemical Response and Psychological Condition. Chin. J. Physiol. 2011, 54, 71–78. [Google Scholar] [CrossRef]
- Zajac, A.; Poprzecki, S.; Zebrowska, A.; Chalimoniuk, M.; Langfort, J. Arginine and Ornithine Supplementation Increases Growth Hormone and Insulin-like Growth Factor-1 Serum Levels after Heavy-Resistance Exercise in Strength-Trained Athletes. J. Strength Cond. Res. 2010, 24, 1082–1090. [Google Scholar] [CrossRef] [PubMed]
- Kraemer, W.J.; Spiering, B.A.; Volek, J.S.; Ratamess, N.A.; Sharman, M.J.; Rubin, M.R.; French, D.N.; Silvestre, R.; Hatfield, D.L.; van Heest, J.L.; et al. Androgenic Responses to Resistance Exercise: Effects of Feeding and L-Carnitine. Med. Sci. Sports Exerc. 2006, 38, 1288–1296. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Kraemer, W.J.; Volek, J.S.; French, D.N.; Rubin, M.R.; Sharman, M.J.; Gómez, A.L.; Ratamess, N.A.; Newton, R.U.; Jemiolo, B.; Craig, B.W.; et al. The Effects of L-Carnitine L-Tartrate Supplementation on Hormonal Responses to Resistance Exercise and Recovery. J. Strength Cond. Res. 2003, 17, 455–462. [Google Scholar] [CrossRef] [PubMed]
- Cinar, V.; Polat, Y.; Baltaci, A.K.; Mogulkoc, R. Effects of Magnesium Supplementation on Testosterone Levels of Athletes and Sedentary Subjects at Rest and after Exhaustion. Biol. Trace Elem. Res. 2011, 140, 18–23. [Google Scholar] [CrossRef] [PubMed]
- Shafiei Neek, L.; Gaeini, A.A.; Choobineh, S. Effect of Zinc and Selenium Supplementation on Serum Testosterone and Plasma Lactate in Cyclist after an Exhaustive Exercise Bout. Biol. Trace Elem. Res. 2011, 144, 454–462. [Google Scholar] [CrossRef]
- El-Bayoumy, K.; Richie, J.P.; Boyiri, T.; Komninou, D.; Prokopczyk, B.; Trushin, N.; Kleinman, W.; Cox, J.; Pittman, B.; Colosimo, S. Influence of Selenium-Enriched Yeast Supplementation on Biomarkers of Oxidative Damage and Hormone Status in Healthy Adult Males. Cancer Epidemiol. Biomark. Prev. 2002, 11, 1459–1465. [Google Scholar]
- Pandit, S.; Biswas, S.; Jana, U.; De, R.K.; Mukhopadhyay, S.C.; Biswas, T.K. Clinical Evaluation of Purified Shilajit on Testosterone Levels in Healthy Volunteers. Andrologia 2016, 48, 570–575. [Google Scholar] [CrossRef]
- Ruan, J.; Li, Z.; Zhang, Y.; Chen, Y.; Liu, M.; Han, L.; Zhang, Y.; Wang, T. Bioactive Constituents from the Roots of Eurycoma Longifolia. Molecules 2019, 24, 3157. [Google Scholar] [CrossRef] [PubMed]
- Low, B.-S.; Choi, S.-B.; Abdul Wahab, H.; Das, P.K.; Chan, K.-L. Eurycomanone, the Major Quassinoid in Eurycoma Longifolia Root Extract Increases Spermatogenesis by Inhibiting the Activity of Phosphodiesterase and Aromatase in Steroidogenesis. J. Ethnopharmacol. 2013, 149, 201–207. [Google Scholar] [CrossRef]
- Henkel, R.R.; Wang, R.; Bassett, S.H.; Chen, T.; Liu, N.; Zhu, Y.; Tambi, M.I. Tongkat Ali as a Potential Herbal Supplement for Physically Active Male and Female Seniors—A Pilot Study. Phytother. Res. PTR 2014, 28, 544–550. [Google Scholar] [CrossRef] [PubMed]
- Leitão, A.E.; Vieira, M.D.C.S.; Gomes, D.A.; Boing, L.; Pelegrini, A.; Luiz, E.; de Azevedo Guimarães, A.C. Exercise Associated or Not to the Intake of Eurycoma Longifolia Improves Strength and Cardiorespiratory Fitness in Men with Androgen Deficiency. Complement. Ther. Clin. Pract. 2021, 42, 101301. [Google Scholar] [CrossRef] [PubMed]
- Talbott, S.M.; Talbott, J.A.; George, A.; Pugh, M. Effect of Tongkat Ali on Stress Hormones and Psychological Mood State in Moderately Stressed Subjects. J. Int. Soc. Sports Nutr. 2013, 10, 28. [Google Scholar] [CrossRef]
- Chan, K.Q.; Stewart, C.; Chester, N.; Hamzah, S.H.; Yusof, A. The Effect of Eurycoma Longifolia on the Regulation of Reproductive Hormones in Young Males. Andrologia 2021, 53, e14001. [Google Scholar] [CrossRef] [PubMed]
- Strauch, G.; Perles, P.; Vergult, G.; Gabriel, M.; Gibelin, B.; Cummings, S.; Malbecq, W.; Malice, M.P. Comparison of Finasteride (Proscar) and Serenoa Repens (Permixon) in the Inhibition of 5-Alpha Reductase in Healthy Male Volunteers. Eur. Urol. 1994, 26, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Bello, M.; Guadarrama-García, C.; Velasco-Silveyra, L.M.; Farfán-García, E.D.; Soriano-Ursúa, M.A. Several Effects of Boron Are Induced by Uncoupling Steroid Hormones from Their Transporters in Blood. Med. Hypotheses 2018, 118, 78–83. [Google Scholar] [CrossRef]
- Naghii, M.R.; Mofid, M.; Asgari, A.R.; Hedayati, M.; Daneshpour, M.-S. Comparative Effects of Daily and Weekly Boron Supplementation on Plasma Steroid Hormones and Proinflammatory Cytokines. J. Trace Elem. Med. Biol. Organ Soc. Miner. Trace Elem. (GMS) 2011, 25, 54–58. [Google Scholar] [CrossRef]
- Naghii, M.R.; Samman, S. The Effect of Boron Supplementation on Its Urinary Excretion and Selected Cardiovascular Risk Factors in Healthy Male Subjects. Biol. Trace Elem. Res. 1997, 56, 273–286. [Google Scholar] [CrossRef]
- Smith, S.J.; Lopresti, A.L.; Teo, S.Y.M.; Fairchild, T.J. Examining the Effects of Herbs on Testosterone Concentrations in Men: A Systematic Review. Adv. Nutr. 2020. [Google Scholar] [CrossRef]
- Lopresti, A.L.; Smith, S.J.; Malvi, H.; Kodgule, R. An Investigation into the Stress-Relieving and Pharmacological Actions of an Ashwagandha (Withania Somnifera) Extract: A Randomized, Double-Blind, Placebo-Controlled Study. Medicine 2019, 98, e17186. [Google Scholar] [CrossRef] [PubMed]
- Nguyen, S.M.; Ko, N.K.; Sattar, A.S.; Ipek, E.G.; Ali, S. Pulmonary Embolism Secondary to Testosterone-Enhancing Herbal Supplement Use. Cureus 2017, 9, e1545. [Google Scholar] [CrossRef]
- Wilborn, C.; Taylor, L.; Poole, C.; Foster, C.; Willoughby, D.; Kreider, R. Effects of a Purported Aromatase and 5α-Reductase Inhibitor on Hormone Profiles in College-Age Men. Int. J. Sport Nutr. Exerc. Metab. 2010, 20, 457–465. [Google Scholar] [CrossRef]
- Rao, A.J.; Mallard, A.R.; Grant, R. Testofen® (Fenugreek Extract) Increases Strength and Muscle Mass Compared to Placebo in Response to Calisthenics. A Randomized Control Trial. Transl. Sports Med. 2020, 3, 374–380. [Google Scholar] [CrossRef]
- Wankhede, S.; Mohan, V.; Thakurdesai, P. Beneficial Effects of Fenugreek Glycoside Supplementation in Male Subjects during Resistance Training: A Randomized Controlled Pilot Study. J. Sport Health Sci. 2016, 5, 176–182. [Google Scholar] [CrossRef]
- Mokashi, M.; Singh-Mokashi, R.; Mohan, V.; Thakurdesai, P. Effects of Glycosides based Fenugreek Seed Extract on Serum Testosterone Levels of Healthy Sedentary Male Subjects: A Exploratory Double Blind, Placebo Controlled, Crossover Study. Asian J. Pharm. Clin. Res. 2014, 7, 177–181. [Google Scholar]
- Maheshwari, A.; Verma, N.; Swaroop, A.; Bagchi, M.; Preuss, H.G.; Tiwari, K.; Bagchi, D. Efficacy of FurosapTM, a Novel Trigonella Foenum-Graecum Seed Extract, in Enhancing Testosterone Level and Improving Sperm Profile in Male Volunteers. Int. J. Med. Sci. 2017, 14, 58–66. [Google Scholar] [CrossRef] [PubMed]
- Mansoori, A.; Hosseini, S.; Zilaee, M.; Hormoznejad, R.; Fathi, M. Effect of Fenugreek Extract Supplement on Testosterone Levels in Male: A Meta-Analysis of Clinical Trials. Phytother. Res. PTR 2020, 34, 1550–1555. [Google Scholar] [CrossRef] [PubMed]
- Shukla, K.K.; Mahdi, A.A.; Ahmad, M.K.; Shankhwar, S.N.; Rajender, S.; Jaiswar, S.P. Mucuna Pruriens Improves Male Fertility by Its Action on the Hypothalamus-Pituitary-Gonadal Axis. Fertil. Steril. 2009, 92, 1934–1940. [Google Scholar] [CrossRef] [PubMed]
- Saylam, B.; Çayan, S. Do Antioxidants Improve Serum Sex Hormones and Total Motile Sperm Count in Idiopathic Infertile Men? Turk. J. Urol. 2020, 46, 442–448. [Google Scholar] [CrossRef]
- Chaing, H.S.; Merino-chavez, G.; Yang, L.L.; Wang, F.N.; Hafez, E.S. Medicinal Plants: Conception/Contraception. Adv. Contracept. Deliv. Syst. CDS 1994, 10, 355–363. [Google Scholar] [PubMed]
- Lopresti, A.L.; Drummond, P.D.; Smith, S.J. A Randomized, Double-Blind, Placebo-Controlled, Crossover Study Examining the Hormonal and Vitality Effects of Ashwagandha (Withania Somnifera) in Aging, Overweight Males. Am. J. Men’s Health 2019, 13, 1557988319835985. [Google Scholar] [CrossRef] [PubMed]
- Al-Qarawi, A.A.; Abdel-Rahman, H.A.; El-Badry, A.A.; Harraz, F.; Razig, N.A.; Abdel-Magied, E.M. The Effect of Extracts of Cynomorium Coccineum and Withania Somnifera on Gonadotrophins and Ovarian Follicles of Immature Wistar Rats. Phytother. Res. PTR 2000, 14, 288–290. [Google Scholar] [CrossRef]
- Mishra, L.C.; Singh, B.B.; Dagenais, S. Scientific Basis for the Therapeutic Use of Withania Somnifera (Ashwagandha): A Review. Altern. Med. Rev. J. Clin. Ther. 2000, 5, 334–346. [Google Scholar]
- Almaiman, A.A. Effect of Testosterone Boosters on Body Functions: Case Report. Int. J. Health Sci. 2018, 12, 86–90. [Google Scholar]
- Wargo, K.A.; Allman, E.; Ibrahim, F. A Possible Case of Saw Palmetto-Induced Pancreatitis. South. Med. J. 2010, 103, 683–685. [Google Scholar] [CrossRef] [PubMed]
- Bui, P.V.; Moualla, M.; Upson, D.J. A Possible Association of Diindolylmethane with Pulmonary Embolism and Deep Venous Thrombosis. Case Rep. Med. 2016, 2016, 7527098. [Google Scholar] [CrossRef] [PubMed]
- Bezuglov, E.; Talibov, O.; Butovskiy, M.; Khaitin, V.; Achkasov, E.; Waśkiewicz, Z.; Lazarev, A. The Inclusion in WADA Prohibited List Is Not Always Supported by Scientific Evidence: A Narrative Review. Asian J. Sports Med. 2021, in press. [Google Scholar] [CrossRef]
- Isenmann, E.; Ambrosio, G.; Joseph, J.F.; Mazzarino, M.; de la Torre, X.; Zimmer, P.; Kazlauskas, R.; Goebel, C.; Botrè, F.; Diel, P.; et al. Ecdysteroids as Non-Conventional Anabolic Agent: Performance Enhancement by Ecdysterone Supplementation in Humans. Arch. Toxicol. 2019, 93, 1807–1816. [Google Scholar] [CrossRef]
- Parr, M.K.; Botrè, F.; Naß, A.; Hengevoss, J.; Diel, P.; Wolber, G. Ecdysteroids: A Novel Class of Anabolic Agents? Biol. Sport 2015, 32, 169–173. [Google Scholar] [CrossRef]
- Gugoasa, L.A.; Staden, R.-I.S. Advanced Methods for the Analysis of Testosterone. Curr. Med. Chem. 2018, 25, 4037–4049. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lazarev, A.; Bezuglov, E. Testosterone Boosters Intake in Athletes: Current Evidence and Further Directions. Endocrines 2021, 2, 109-120. https://doi.org/10.3390/endocrines2020011
Lazarev A, Bezuglov E. Testosterone Boosters Intake in Athletes: Current Evidence and Further Directions. Endocrines. 2021; 2(2):109-120. https://doi.org/10.3390/endocrines2020011
Chicago/Turabian StyleLazarev, Artemii, and Eduard Bezuglov. 2021. "Testosterone Boosters Intake in Athletes: Current Evidence and Further Directions" Endocrines 2, no. 2: 109-120. https://doi.org/10.3390/endocrines2020011
APA StyleLazarev, A., & Bezuglov, E. (2021). Testosterone Boosters Intake in Athletes: Current Evidence and Further Directions. Endocrines, 2(2), 109-120. https://doi.org/10.3390/endocrines2020011






