Clinical Characteristics, Treatments and Outcomes of 18 Lung Transplant Recipients with COVID-19
Abstract
1. Introduction
2. Methods
2.1. Study Population, Setting, and Clinical Data
2.2. Laboratory and Radiology Assessment
2.3. Statistical Analysis
2.4. Ethical Consideration
3. Results
3.1. Clinical Patient Characteristics
3.2. Laboratory and Radiological Features
3.3. Therapeutic Intervention
3.4. Remdesivir in Patients with Impaired Renal Function
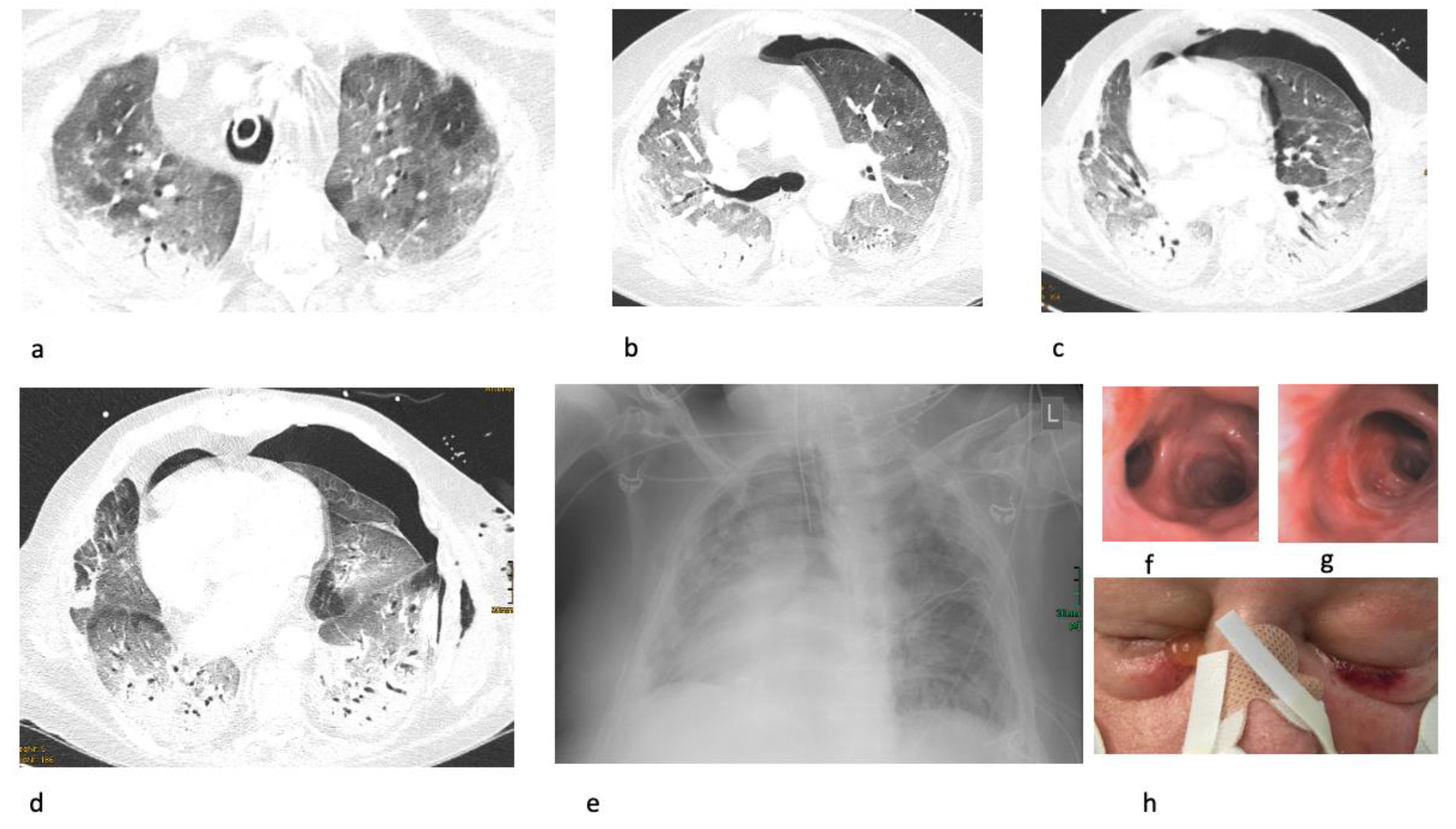
3.5. Autopsy in a Patient with COVID-19 with Siddiqi Stage III Disease
4. Discussion
4.1. COVID-19 Severity: The Siddiqi Stages
4.2. SARS-CoV-2 Infection: Extrapulmonary Manifestations
4.3. SARS-CoV-2 Infection: Pulmonary Manifestations
4.4. Liver Function Test Abnormalities
4.5. CRP as Marker of Disease Activity in COVID-19
4.6. COVID-19 and ABO Blood Group
4.7. AIFELL Score
4.8. Remdesivir in Patients with Impaired Renal Function
4.9. COVID-19 Convalescent Plasma (CCP)
4.10. Respiratory Co-Infections
4.11. Immunosuppressive Therapy during COVID-19
4.12. Hospital Admission Rate
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Limitation of Study
References
- Chambers, D.C.; Cherikh, W.S.; Harhay, M.O.; Hayes, D.; Hsich, E.; Khush, K.K.; Meiser, B.; Potena, L.; Rossano, J.W.; Toll, A.E.; et al. The International Thoracic Organ Transplant Registry of the International Society for Heart and Lung Transplantation: Thirty-sixth adult lung and heart-lung transplantation Report-2019; Focus theme: Donor and recipient size match. J. Heart Lung Transplant. 2019, 38, 1042–1055. [Google Scholar] [CrossRef]
- Pereira, M.R.; Mohan, S.; Cohen, D.J.; Husain, S.A.; Dube, G.K.; Ratner, L.E.; Arcasoy, S.; Aversa, M.M.; Benvenuto, L.J.; Dadhania, D.M.; et al. COVID-19 in solid organ transplant recipients: Initial report from the US epicenter. Am. J. Transplant. 2020, 20, 1800–1808. [Google Scholar] [CrossRef]
- Verleden, G.M.; Godinas, L.; Lorent, N.; Van Bleyenbergh, P.; Dupont, L.; Delcroix, M.; Yserbyt, J.; Dooms, C.; Vos, R. COVID-19 in lung transplant patients: A case series. Am. J. Transplant. 2020, 20, 3234–3238. [Google Scholar] [CrossRef]
- Bennett, D.; De Vita, E.; Ventura, V.; Bernayyali, S.; Fossi, A.; Paladini, P.; Luzzi, L.; Maccherini, M.; Valente, S.; Bargagli, E.; et al. Impact of SARS-CoV-2 outbreak on heart and lung transplant: A patient-perspective survey. Transpl. Infect. Dis. 2021, 23, e13428. [Google Scholar] [CrossRef] [PubMed]
- Siddiqi, H.K.; Mehra, M.R. COVID-19 illness in native and immunosuppressed states: A clinical-therapeutic staging proposal. J. Heart Lung Transplant. 2020, 39, 405–407. [Google Scholar] [CrossRef]
- Levenfus, I.; Ullmann, E.; Battegay, E.; Schuurmans, M.M. Triage tool for suspected COVID-19 patients in the emergency room: AIFELL score. Braz. J. Infect. Dis. 2020, 24, 458–461. [Google Scholar] [CrossRef]
- Guidance from the International Society of Heart and Lung Transplantation Regarding the SARS CoV-2 Pandemic. Available online: www.https://ishlt.org/ishlt/media/documents/SARS-CoV-2_Guidance-for-Cardiothoracic-Transplant-and-VAD-center.pdf. (accessed on 3 April 2021).
- Sardu, C.; Gambardella, J.; Morelli, M.B.; Wang, X.; Marfella, R.; Santulli, G. Hypertension, Thrombosis, Kidney Failure, and Diabetes: Is COVID-19 an Endothelial Disease? A Comprehensive Evaluation of Clinical and Basic Evidence. J. Clin. Med. 2020, 9, 1417. [Google Scholar] [CrossRef] [PubMed]
- Pons, S.; Fodil, S.; Azoulay, E.; Zafrani, L. The vascular endothelium: The cornerstone of organ dysfunction in severe SARS-CoV-2 infection. Crit. Care. 2020, 24, 353. [Google Scholar] [CrossRef] [PubMed]
- Bernard, I.; Limonta, D.; Mahal, L.K.; Hobman, T.C. Endothelium Infection and Dysregulation by SARS-CoV-2: Evidence and Caveats in COVID-19. Viruses 2020, 13, 29. [Google Scholar] [CrossRef] [PubMed]
- Rios, C.I.; Cassatt, D.R.; Hollingsworth, B.A.; Satyamitra, M.M.; Tadesse, Y.S.; Taliaferro, L.P.; Winters, T.A.; DiCarlo, A.L. Commonalities Between COVID-19 and Radiation Injury. Radiat. Res. 2021, 195, 1–24. [Google Scholar] [PubMed]
- Crisan-Dabija, R.; Pavel, C.A.; Popa, I.V.; Tarus, A.; Burlacu, A. “A Chain Only as Strong as Its Weakest Link”: An Up-to-Date Literature Review on the Bidirectional Interaction of Pulmonary Fibrosis and COVID-19. J. Proteome Res. 2020, 19, 4327–4338. [Google Scholar] [CrossRef] [PubMed]
- Weber, S.; Hellmuth, J.C.; Scherer, C.; Muenchhoff, M.; Mayerle, J.; Gerbes, A.L. Liver function test abnormalities at hospital admission are associated with severe course of SARS-CoV-2 infection: A prospective cohort study. Gut 2021. [Google Scholar] [CrossRef]
- Youssef, M.; Hussein, M.; Attia, A.S.; Elshazli, R.M.; Omar, M.; Zora, G.; Farhoud, A.S.; Elnahla, A.; Shirabi, A.; Toraih, E.A.; et al. COVID-19 and liver dysfunction: A systematic review and meta-analysis of retrospective studies. J. Med. Virol. 2020, 92, 1825–1833. [Google Scholar] [CrossRef] [PubMed]
- Jothimani, D.; Venugopal, R.; Abedin, M.F.; Kaliamoorthy, I.; Rela, M. COVID-19 and the liver. J. Hepatol. 2020, 73, 1231–1240. [Google Scholar] [CrossRef] [PubMed]
- Saad, M.; Omrani, A.S.; Baig, K.; Bahloul, A.; Elzein, F.; Matin, M.A.; Selim, M.A.A.; Al Mutairi, M.; Al Nakhli, D.A.; Al Aidoroos, A.Y.; et al. Clinical aspects and outcomes of 70 patients with Middle East respiratory syndrome coronavirus infection: A single-center experience in Saudi Arabia. Int. J. Infect. Dis. 2014, 29, 301–306. [Google Scholar] [CrossRef]
- Al-Hameed, F.; Wahla, A.S.; Siddiqui, S.; Ghabashi, A.; Al-Shomrani, M.; Al-Thaqafi, A.; Tashkandi, Y. Characteristics and Outcomes of Middle East Respiratory Syndrome Coronavirus Patients Admitted to an Intensive Care Unit in Jeddah, Saudi Arabia. J. Intensive Care Med. 2016, 31, 344–348. [Google Scholar] [CrossRef]
- Chang, H.-L.; Chen, K.-T.; Lai, S.-K.; Kuo, H.W.; Su, I.J.; Lin, R.S.; Sung, F.C. Hematological and Biochemical Factors Predicting SARS Fatality in Taiwan. J. Formos. Med. Assoc. 2006, 105, 439–450. [Google Scholar] [CrossRef]
- Zhang, B.; Zhou, X.; Qiu, Y.; Song, Y.; Feng, F.; Feng, J.; Song, Q.; Qingzhu, J.; Wang, J. Clinical characteristics of 82 cases of death from COVID-19. PLoS ONE 2020, 15, e0235458. [Google Scholar] [CrossRef]
- Guan, W.J.; Ni, Z.Y.; Hu, Y.; Liang, W.; Ou, C.; He, J.; Shan, H.; Lei, C.; Hui, D.S.C.; Du, B.; et al. Clinical Characteristics of Coronavirus Disease 2019 in China. N. Engl. J. Med. 2020, 382, 1708–1720. [Google Scholar] [CrossRef] [PubMed]
- Xu, L.; Liu, J.; Lu, M.; Yang, D.; Zheng, X. Liver injury during highly pathogenic human coronavirus infections. Liver Int. 2020, 40, 998–1004. [Google Scholar] [CrossRef]
- Little, P. Non-steroidal anti-inflammatory drugs and covid-19. BMJ 2020, 368, m1185. [Google Scholar] [CrossRef]
- Gordon, C.J.; Tchesnokov, E.P.; Feng, J.Y.; Porter, D.P.; Gotte, M. The antiviral compound remdesivir potently inhibits RNA-dependent RNA polymerase from Middle East respiratory syndrome coronavirus. J. Biol. Chem. 2020, 295, 4773–4779. [Google Scholar] [CrossRef]
- Fan, Z.; Chen, L.; Li, J.; Tian, C.; Zhang, Y.; Huang, S.; Liu, Z.; Cheng, J. Clinical features of COVID-19 related liver damage. medRxiv 2020. [Google Scholar] [CrossRef]
- Wang, Y.; Zhang, D.; Du, G.; Du, R.; Zhao, J.; Jin, Y.J.; Fu, S.; Gao, L.; Cheng, Z.; Lu, Q.; et al. Remdesivir in adults with severe COVID-19: A randomised, double-blind, placebo-controlled, multicentre trial. Lancet 2020, 395, 1569–1578. [Google Scholar] [CrossRef]
- Tan, C.; Huang, Y.; Shi, F.; Tan, K.; Ma, Q.; Chen, Y.; Jiang, X.; Li, X. C-reactive protein correlates with computed tomographic findings and predicts severe COVID-19 early. J. Med. Virol. 2020, 92, 856–862. [Google Scholar] [CrossRef]
- Kermali, M.; Khalsa, R.K.; Pillai, K.; Ismail, Z.; Harky, A. The role of biomarkers in diagnosis of COVID-19—A systematic review. Life Sci. 2020, 254, 117788. [Google Scholar] [CrossRef]
- Qin, C.; Zhou, L.; Hu, Z.; Zhang, S.; Yang, S.; Tao, Y.; Xie, C.; Ma, K.; Shang, K.; Wang, W.; et al. Dysregulation of Immune Response in Patients with Coronavirus 2019 (COVID-19) in Wuhan, China. Clin. Infect. Dis. 2020, 71, 762–768. [Google Scholar] [CrossRef] [PubMed]
- Liu, F.; Li, L.; Xu, M.; Wu, J.; Luo, D.; Zhu, Y.; Li, B.; Song, X.; Zhou, X. Prognostic value of interleukin-6, C-reactive protein, and procalcitonin in patients with COVID-19. J. Clin. Virol. 2020, 127, 104370. [Google Scholar] [CrossRef] [PubMed]
- Herold, T.; Jurinovic, V.; Arnreich, C.; Lipworht, B.J.; Hellmuth, J.C.; von Bergwelt-Baildon, M.; Klein, M.; Weinberger, T. Elevated levels of IL-6 and CRP predict the need for mechanical ventilation in COVID-19. J. Allergy Clin. Immunol. 2020, 146, 128–136.e4. [Google Scholar] [CrossRef] [PubMed]
- Kim, M.Y.; Brennan, D.C.; Shah, P. General Approach to the Clinical Care of Solid Organ Transplant Recipients with COVID-19 Infection: Management for Transplant Recipients. Curr. Transpl. Rep. 2020, 7, 366–378. [Google Scholar] [CrossRef]
- Li, J.; Wang, X.; Chen, J.; Cai, Y.; Deng, A.; Yang, M. Association between ABO blood groups and risk of SARS-CoV-2 pneumonia. Br. J. Haematol. 2020, 190, 24–27. [Google Scholar] [CrossRef]
- Zhao, J.; Yang, Y.; Huang, H.; Li, D.; Gu, D.F.; Lu, X.F.; Zhang, Z.; Liu, L.; Liu, T.; Liu, Y.K.; et al. Relationsho between the ABO Blood Group and the COVID-19 Susceptibility. medRxiv 2020. Available online: https://www.medrxiv.org/content/medrxiv/early/2020/03/27/2020.03.11.20031096.full.pdf (accessed on 19 April 2021). [CrossRef]
- Zietz, M.; Zucker, J.; Tatonetti, N.P. Associations between blood type and COVID-19 infection, intubation, and death. Nat. Commun. 2020, 11, 5761. [Google Scholar] [CrossRef] [PubMed]
- Gerard, C.; Maggipinto, G.; Minon, J.M. COVID-19 and ABO blood group: Another viewpoint. Br. J. Haematol. 2020, 190, e93–e94. [Google Scholar] [CrossRef] [PubMed]
- Wu, B.B.; Gu, D.Z.; Yu, J.N.; Yang, J.; Shen, W.Q. Association between ABO blood groups and COVID-19 infection, severity and demise: A systematic review and meta-analysis. Infect. Genet. Evol. 2020, 84, 104485. [Google Scholar] [CrossRef]
- Malin, J.J.; Suarez, I.; Priesner, V.; Fatkenheuer, G.; Rybniker, J. Remdesivir against COVID-19 and Other Viral Diseases. Clin. Microbiol. Rev. 2020, 34, e00162-20. [Google Scholar] [CrossRef]
- WHO Solidarity Trial Consortium. Repurposed Antiviral Drugs for Covid-19—Interim WHO Solidarity Trial Results. N. Engl. J. Med. 2021, 384, 497–511. [Google Scholar] [CrossRef]
- Beigel, J.H.; Tomashek, K.M.; Dodd, L.E.; Mehta, A.K.; Zingman, B.S.; Kalil, A.C.; Hohmann, E.; Chu, H.Y.; Luetkemeyer, A.; Kline, S.; et al. Remdesivir for the Treatment of Covid-19—Final Report. N. Engl. J. Med. 2020, 383, 1813–1826. [Google Scholar] [CrossRef] [PubMed]
- Update on Remdesivir—EMA Will Evaluate New Data from Solidarity Trial. Available online: https://www.ema.europa.eu/en/news/update-remdesivir-ema-will-evaluate-new-data-solidarity-trial (accessed on 14 February 2021).
- Roberto, P.; Francesco, L.; Emanuela, C.; Giorgia, G.; Pasquale, N.; Sara, D. Current treatment of COVID-19 in renal patients: Hope or hype? Intern. Emerg. Med. 2020, 15, 1389–1398. [Google Scholar] [CrossRef]
- Ackley, T.W.; McManus, D.; Topal, J.E.; Cicali, B.; Shah, S. A Valid Warning or Clinical Lore: An Evaluation of Safety Outcomes of Remdesivir in Patients with Impaired Renal Function from a Multicenter Matched Cohort. Antimicrob. Agents Chemother. 2021, 65, e02290-20. [Google Scholar] [CrossRef] [PubMed]
- Le, M.P.; Le Hingrat, Q.; Jaquet, P.; Wicky, P.H.; Bunel, V.; Massias, L.; Visseaux, B.; Messika, J.; Descamps, D.; Mal, H.; et al. Removal of Remdesivir’s Metabolite GS-441524 by Hemodialysis in a Double Lung Transplant Recipient with COVID-19. Antimicrob. Agents Chemother. 2020, 64, e01521-20. [Google Scholar] [CrossRef]
- Williamson, E.J.; Walker, A.J.; Bhaskaran, K.; Bacon, S.; Bates, C.; Morton, C.E.; Curtis, H.J.; Mehrkar, A.; Evans, D.; Inglesby, P.; et al. Factors associated with COVID-19-related death using OpenSAFELY. Nature 2020, 584, 430–436. [Google Scholar] [CrossRef]
- Harvala, H.; Mehew, J.; Robb, M.L.; Harvala, H. Convalescent plasma treatment for SARS-CoV-2 infection: Analysis of the first 436 donors in England, 22 April to 12 May 2020. Eurosurveillance 2020, 25. [Google Scholar] [CrossRef] [PubMed]
- Duan, K.; Liu, B.; Li, C.; Zhang, H.; Yu, T.; Qu, J.; Zhou, M.; Chen, L.; Meng, S.; Hu, Y.; et al. Effectiveness of convalescent plasma therapy in severe COVID-19 patients. Proc. Natl. Acad. Sci USA 2020, 117, 9490–9496. [Google Scholar] [CrossRef] [PubMed]
- Hegerova, L.; Gooley, T.A.; Sweerus, K.A.; Maree, C.; Bailey, N.; Bailey, M.; Dunleavy, V.; Patel, K.; Alcorn, K.; Haley, R.; et al. Use of convalescent plasma in hospitalized patients with COVID-19: Case series. Blood 2020, 136, 759–762. [Google Scholar] [CrossRef] [PubMed]
- Joyner, M.; Wright, R.S.; Fairweather, D.; Senefeld, J.W.; Bruno, K.A.; Klassen, S.A.; Carter, R.E.; Klompas, A.M.; Wiggins, C.C.; Shepherd, J.R.A.; et al. Early Safety Indicators of COVID-19 Convalescent Plasma in 5000 Patients. J. Clin. Investig. 2020, 130, 4791–4797. [Google Scholar] [CrossRef]
- Shen, C.; Wang, Z.; Zhao, F.; Yang, Y.; Li, J.; Yuan, J.; Wang, F.; Li, D.; Yang, M.; Xing, L.; et al. Treatment of 5 Critically Ill Patients With COVID-19 With Convalescent Plasma. JAMA 2020, 323, 1582–1589. [Google Scholar] [CrossRef] [PubMed]
- Casadevall, A.; Henderson, J.P.; Joyner, M.J.; Pirofski, L.A. SARS-CoV-2 variants and convalescent plasma: Reality, fallacies, and opportunities. J. Clin. Investig. 2021, 131. [Google Scholar] [CrossRef]
- Kemp, S.A.; Collier, D.A.; Datir, R.P.; Ferreira, I.A.T.M.; Gayed, S.; Jahun, A.; Hosmillo, M.; Rees-Spear, C.; Mlcochova, P.; Lumb, I.U.; et al. SARS-CoV-2 evolution during treatment of chronic infection. Nature 2021, 592, 277–282. [Google Scholar] [CrossRef] [PubMed]
- Andreano, E.; Piccini, G.; Licastro, D.; Casakubi, L.; Johnson, N.V.; Paciello, I.; Dal Monego, S.; Pantano, E.; Manganaro, N.; Manenti, A.; et al. SARS-CoV-2 escape in vitro from a highly neutralizing COVID-19 convalescent plasma. bioRxiv 2020. [Google Scholar] [CrossRef]
- Kossyvakis, A.; Mentis, A.A.; Tryfinopoulou, K.; Kaliaropulos, A.; Antalis, E.; Lytras, T.; Meijer, A.; Tsiodras, S.; Karakitsos, P.; Mentis, A.F. Antiviral susceptibility profile of influenza A viruses; keep an eye on immunocompromised patients under prolonged treatment. Eur. J. Clin. Microbiol. Infect. Dis. 2017, 36, 361–371. [Google Scholar] [CrossRef] [PubMed]
- Lai, C.C.; Wang, C.Y.; Hsueh, P.R. Co-infections among patients with COVID-19: The need for combination therapy with non-anti-SARS-CoV-2 agents? J. Microbiol. Immunol. Infect. 2020, 53, 505–512. [Google Scholar] [CrossRef] [PubMed]
- Zhu, X.; Ge, Y.; Wu, T.; Zhao, K.; Chen, Y.; Wu, B.; Zhu, F.; Zhu, B.; Cui, L. Co-infection with respiratory pathogens among COVID-2019 cases. Virus Res. 2020, 285, 198005. [Google Scholar] [CrossRef]
- Timsit, J.F.; Sonneville, R.; Kalil, A.C.; Basseti, M.; Ferrer, R.; Jaber, S.; Lanternier, F.; Luyt, C.E.; Machado, F.; Mikulska, M.; et al. Diagnostic and therapeutic approach to infectious diseases in solid organ transplant recipients. Intensive Care Med. 2019, 45, 573–591. [Google Scholar] [CrossRef] [PubMed]
- Raja, M.A.; Mendoza, M.A.; Villavicencio, A.; Anjan, S.; Reynolds, J.M.; Kittipibul, V.; Fernandez, A.; Guerra, G.; Camargo, J.F.; Simkins, J.; et al. COVID-19 in solid organ transplant recipients: A systematic review and meta-analysis of current literature. Transpl. Rev. 2021, 35, 100588. [Google Scholar] [CrossRef]
- Aversa, M.; Benvenuto, L.; Anderson, M.; Shah, L.; Robbins, H.; Pereira, M.; Carroll, M.; Hum, J.; Nolan, M.; Reilly, G. COVID-19 in lung transplant recipients: A single center case series from New York City. Am. J. Transplant. 2020, 20, 3072–3080. [Google Scholar] [CrossRef]
- Song, S.H.; Chen, T.L.; Deng, L.P.; Zhang, Y.Z.; Mo, P.Z.; Gao, S.C.; Hu, W.J.; Xiong, Y.; Ma, Z.Y. Clinical characteristics of four cancer patients with SARS-CoV-2 infection in Wuhan, China. Infect. Dis. Poverty 2020, 9, 82. [Google Scholar] [CrossRef]
- Morlacchi, L.C.; Rossetti, V.; Gigli, L.; Amati, F.; Rosso, L.; Aliberti, S.; Nosotti, M.; Blasi, F. COVID-19 in lung transplant recipients: A case series from Milan, Italy. Transpl. Infect. Dis. 2020, 22, e13356. [Google Scholar] [CrossRef]
- Mezquida, B. Covid-19 Pneumonia in Bilateral Lung Transplant Recipient: Case Report. Int. J. Transpl. Res. Med. 2020, 6, 1–3. [Google Scholar] [CrossRef]
| Mild (Siddiqi I) n = 7 | Moderate (Siddiqi IIA,B) n = 9 | Severe (Siddiqi III) n = 2 | |
|---|---|---|---|
| Age, mean yrs., (range) | 41.3 (19–64) | 54.7 (28–68) | 58.5 (56–61) |
| Male sex (%) | 3 (43%) | 7 (78%) | 2 (100%) |
| BMI, mean (kg/m2) | 22.4 | 26.3 | 31.2 |
| Pretransplant diagnosis | |||
| Cystic fibrosis | 4 (57%) | 2 (22%) | 0 |
| COPD | 1 (14%) | 4 (44%) | 0 |
| ILD | 2 (29%) | 2 (22%) | 2 (100%) |
| PAH | 0 | 1 (11%) | 0 |
| Comorbidities (%) | |||
| Hypertension | 2 (29%) | 6 (56%) | 2 (100%) |
| Chronic kidney disease | 2 (29%) | 4 (44%) | 2 (100%) |
| Diabetes | 2 (29%) | 3 (33%) | 1 (50%) |
| Cardiovascular disease | 1 (14%) | 3 (33%) | 2 (100%) |
| Malignancy | 0 | 4 (44%) | 1 (50%) |
| Mild (Siddiqi I) n = 7 | Moderate (Siddiqi IIA,B) n = 9 | Severe (Siddiqi III) n = 2 | |
|---|---|---|---|
| Symptoms | |||
| Fever | 2 (29%) | 7 (78%) | 0 |
| Cough | 1 (14%) | 5 (56%) | 0 |
| Dyspnea | 0 | 4 (44%) | 2 (100%) |
| Sore throat | 0 | 0 | 0 |
| Fatigue | 1 (14%) | 4 (44%) | 1 (50%) |
| Anorexia | 1 (14%) | 2 (22%) | 0 |
| Diarrhea | 1 (14%) | 2 (22%) | 0 |
| Nausea/vomiting | 3 (43%) | 3 (33%) | 1 (50%) |
| Altered sense of smell | 0 | 0 | 0 |
| Altered sense of taste | 0 | 1 (11%) | 0 |
| Headache | 2 (29%) | 3 (33%) | 0 |
| Rhinorrhea | 1 (14%) | 1 (11%) | 0 |
| Vital signs | |||
| Temperature (°C) | 38.4 (37.9–38.8) | 37.3 (35.9–39.2) | 38.3 (36.8–39.8) |
| Heart rate (bpm) | 118 | 82 (69–129) | 88 (81–95) |
| Oxygen saturation, % | 96 | 96.4 (95–99) | 92 (90–93) |
| Laboratory values, mean (range) | |||
| CRP (mg/l) | 29.8 (4–77) | 58.2 (4.8–140) | 302 (199–406) |
| Hemoglobin (g/l) | 135 (104–172) | 121 (96–152) | 113 (99–127) |
| Thrombocytes (G/l) | 201 (153–313) | 186 (126–365) | 174 (172–176) |
| Leucocytes (G/l) | 6.41 (3.67–10.6) | 6.67 (4.99–9.38) | 11.6 (5.9–17.3) |
| Neutrophils (G/l) | 4.89 (3.26–8.03) | 5.29 (2.04–7.49) | 10.4 (4.1–16.1) |
| Eosinophils (G/l) | 0.067 (0–0.14) | 0.024 (0–0.08) | 0 (0–0) |
| Lymphocytes (G/l) | 0.95 (0.17–1.25) | 0.60 (0.18–1.58) | 1.29 (0.47–2.1) |
| ASAT (U/l) | 42.7 (18–69) | 29.9 (14–44) | 44 (39–49) |
| ALAT (U/l) | 47.3 (11–90) | 24.7 (12–39) | 16 (9–23) |
| LDH (U/l) | 383 (309–450) | 486 (5–636) | 801 (500–1101) |
| Bilirubin (µmol/l) | 17.7 (5–34) | 8.1 (4–15) | N/A |
| Creatinine (µmol/l) | 119 (90–166) | 231 (17–809) | 208 (202–213) |
| Creatinin kinase (U/l) | 46 (43–49) | 113 (25–525) | 92 (69–114) |
| Blood Group (A, B, AB, 0) | |||
| A (%) | 3 (43%) | 7 (78%) | 0 |
| B (%) | 0 | 1 (11%) | 0 |
| AB (%) | 0 | 0 | 0 |
| 0 (%) | 4 (57%) | 1 (11%) | 2 (100%) |
| AIFELL Score at presentation | 1.5 (1–2) | 3.2 (2–4) | 4.5 (4–5) |
| Mild (Siddiqi I) n = 7 | Moderate (Siddiqi IIA,B) n = 9 | Severe (Siddiqi III) n = 2 | |
|---|---|---|---|
| Immunosuppression | |||
| Prednisone | 7 (100%) | 9 (100%) | 2 (100%) |
| Mycophenolate mofetil | 6 (86%) | 7 (78%) | 2 (100%) |
| Cyclosporine A | 1 (14%) | 3 (33%) | 2 (100%) |
| Tacrolimus | 6 (86%) | 4 (44%) | 0 |
| Everolimus | 0 | 1 (11%) | 0 |
| Rapamycin | 1 (14%) | 0 | 0 |
| Treatment | |||
| Remdesivir | 0 | 6 (67%) | 0 |
| Augmentin | 3 (43%) | 0 | 1 (50%) |
| Ceftriaxone | 0 | 0 | 0 |
| Tazobactam/piperacillin | 1 (14%) | 3 (33%) | 1 (50%) |
| Meropenem | 0 | 3 (33%) | 1 (50%) |
| Vancomycin | 0 | 1 (11%) | 0 |
| Dexamethasone | 0 | 3 (33%) | 2 (100%) |
| Treatment setting | |||
| Ambulant | 6 (86%) | 0 | 0 |
| Hospital, normal ward | 1 (14%) | 7 (78%) | 0 |
| Hospital, intermediate care | 0 | 0 | 0 |
| Hospital, intensive care | 0 | 2 (22%) | 2 (100%) |
| Hospitalization (days) | 7 (7–7) | 22 (3–44) | 20 (19–20) |
| Oxygenation | |||
| Normal, room air | 7 (100%) | 5 (56%) | 0 |
| Oxygen, nasal cannula | 0 | 3 (33%) | |
| Oxygen, non-rebreather | 0 | 0 | 0 |
| Oxygen, HFOT | 0 | 1 (11%) | 0 |
| Non-invasive ventilation | 0 | 0 | 0 |
| Mechanical ventilation | 0 | 0 | 2 (100%) |
| ECMO | 0 | 0 | 0 |
| Outcome | |||
| Alive | 7 (100%) | 9 (100%) | 0 |
| Dead | 0 | 0 | 2 (100%) |
| Patient Number | 1 | 2 | 3 | 4 | 5 | 6 | 7 | 8 | 9 | 10 | 11 | 12 | 13 | 14 | 15 | 16 | 17 | 18 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Date of COVID-19 | 10/ 2020 | 10/ 2020 | 03/ 2020 | 10/ 2020 | 10/ 2020 | 11/ 2020 | 11/ 2020 | 11/ 2020 | 12/ 2020 | 05/ 2020 | 11/ 2020 | 12/ 2020 | 01/ 2021 | 12/ 2020 | 01/ 2021 | 01/ 2021 | 01/ 2021 | 01/ 2021 |
| Demographic | ||||||||||||||||||
| Age (years) | 56 | 28 | 56 | 48 | 38 | 22 | 68 | 66 | 27 | 64 | 49 | 68 | 64 | 34 | 19 | 61 | 67 | 63 |
| male/female | m | m | f | m | m | f | m | m | m | m | f | m | f | m | f | m | f | m |
| BMI, kg/m2 | 31 | 27.4 | 21.9 | 19.3 | 19.6 | 20.3 | 42.8 | 27.1 | 19.5 | 27.7 | 18.9 | 22.4 | 27.4 | 14.9 | 15.2 | 31.4 | 37.5 | 31.8 |
| Transplant Data | ||||||||||||||||||
| Transplant year | 2019 | 2019 | 2019 | 2019 | 2006 | 2019 | 2015 | 2020 | 2014 | 2018 | 2016 | 2012 | 2016 | 2016 | 2020 | 2016 | 2010 | 2018 |
| Previous disease | ILD | PAH | COPD | CF | CF | CF | COPD | IPF | CF | IPF | Pl.par.Fib | COPD | COPD | CF | CF | ILD | ILD | COPD |
| Comorbidities | ||||||||||||||||||
| Hypertension | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | ||||||||
| Diabetes | 1 | 1 | 1 | 1 | 1 | 1 | ||||||||||||
| Cardiovascular disease | 1 | 1 | 1 | 1 | 1 | 1 | ||||||||||||
| Malignancy | 1 | 1 | 1 | 1 | 1 | |||||||||||||
| Chronic kidney disease | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | ||||||||||
| Immunosuppression | ||||||||||||||||||
| Prednison | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 |
| MMF | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | |||
| Ciclosporine | 1 | 1 | 1 | 1 | 1 | 1 | ||||||||||||
| Tacrolimus | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | ||||||||
| Everolimus | 1 | |||||||||||||||||
| Certican/rapamycin | 1 | |||||||||||||||||
| Risk stratification | ||||||||||||||||||
| Siddiqi Stage | III | IIA | I | IIB | IIB | I | IIA | IIA | I | I | IIA | IIB | IIB | I | I | III | I | IIA |
| AIFELL Score | 4 | 1 | 2 | 4 | 3 | 3 | 4 | 2 | 4 | 4 | 1 | 5 | ||||||
| Radiology | ||||||||||||||||||
| Examination (CT/CXR) | CT | CT | CT | CT | CT | CT | CT | CT | CT | CT | CT | CXR | ||||||
| GGO | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | |||||||
| Consolidation | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | |||||||||
| Pleural Effusion | 1 | 1 | ||||||||||||||||
| Pneumothorax | 1 | |||||||||||||||||
| Treatment | ||||||||||||||||||
| Antiviral | ||||||||||||||||||
| Remdesivir | 1 | 1 HD dose | 1 | 1 HD dose | 1 | 1 | ||||||||||||
| Antibiotics | ||||||||||||||||||
| Azithromycin | ||||||||||||||||||
| Augmentin | 1 | 1 | 1 | 1 | ||||||||||||||
| Ceftriaxon | 1 | 1 | ||||||||||||||||
| Tazobac | 1 | 1 | 1 | 1 | 1 | |||||||||||||
| Meropenem | 1 | 1 | 1 | 1 | ||||||||||||||
| Vancomycin | 1 | |||||||||||||||||
| Corticosteroids | ||||||||||||||||||
| Prednison | 1 | |||||||||||||||||
| Dexamethasone | 1 | 1 | 1 | 1 | 1 | |||||||||||||
| CCP | 1 | |||||||||||||||||
| Treatment Setting | ||||||||||||||||||
| Ambulant | 1 | 1 | 1 | 1 | 1 | 1 | ||||||||||||
| Hosp. normal ward | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | ||||||||||
| Hosp. ICU | 1 | 1 | 1 | 1 | ||||||||||||||
| No. of Hosp. Days | 20 | 3 | 7 | 43 | 44 | 9 | 26 | 3 | 36 | 7 | 19 | 23 | ||||||
| Oxygenation | ||||||||||||||||||
| Normal breathing no O2 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | ||||||
| Oxygen nasal cannula | 1 | 1 | 1 | |||||||||||||||
| Oxygen HFOT | 1 | |||||||||||||||||
| Mechanical ventilation | 1 | 1 | ||||||||||||||||
| Outcome | ||||||||||||||||||
| Alive | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | 1 | ||
| Dead | 1 | 1 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hage, R.; Steinack, C.; Gautschi, F.; Pfister, S.; Inci, I.; Schuurmans, M.M. Clinical Characteristics, Treatments and Outcomes of 18 Lung Transplant Recipients with COVID-19. Transplantology 2021, 2, 229-245. https://doi.org/10.3390/transplantology2020022
Hage R, Steinack C, Gautschi F, Pfister S, Inci I, Schuurmans MM. Clinical Characteristics, Treatments and Outcomes of 18 Lung Transplant Recipients with COVID-19. Transplantology. 2021; 2(2):229-245. https://doi.org/10.3390/transplantology2020022
Chicago/Turabian StyleHage, René, Carolin Steinack, Fiorenza Gautschi, Susan Pfister, Ilhan Inci, and Macé M. Schuurmans. 2021. "Clinical Characteristics, Treatments and Outcomes of 18 Lung Transplant Recipients with COVID-19" Transplantology 2, no. 2: 229-245. https://doi.org/10.3390/transplantology2020022
APA StyleHage, R., Steinack, C., Gautschi, F., Pfister, S., Inci, I., & Schuurmans, M. M. (2021). Clinical Characteristics, Treatments and Outcomes of 18 Lung Transplant Recipients with COVID-19. Transplantology, 2(2), 229-245. https://doi.org/10.3390/transplantology2020022









