Presentation of SARS-CoV-2 in a Pediatric Heart Transplant Recipient with Multiple Underlying Comorbidities
Abstract
:1. Introduction
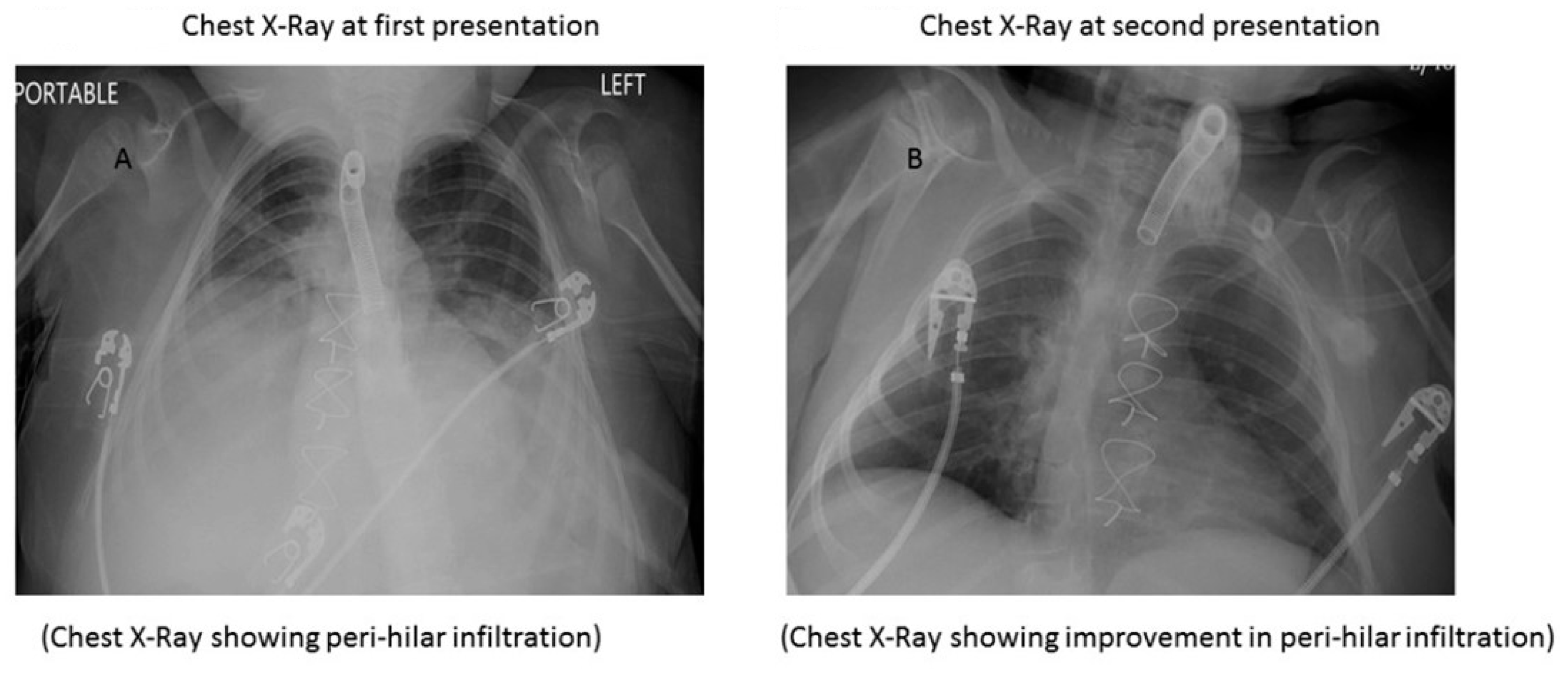
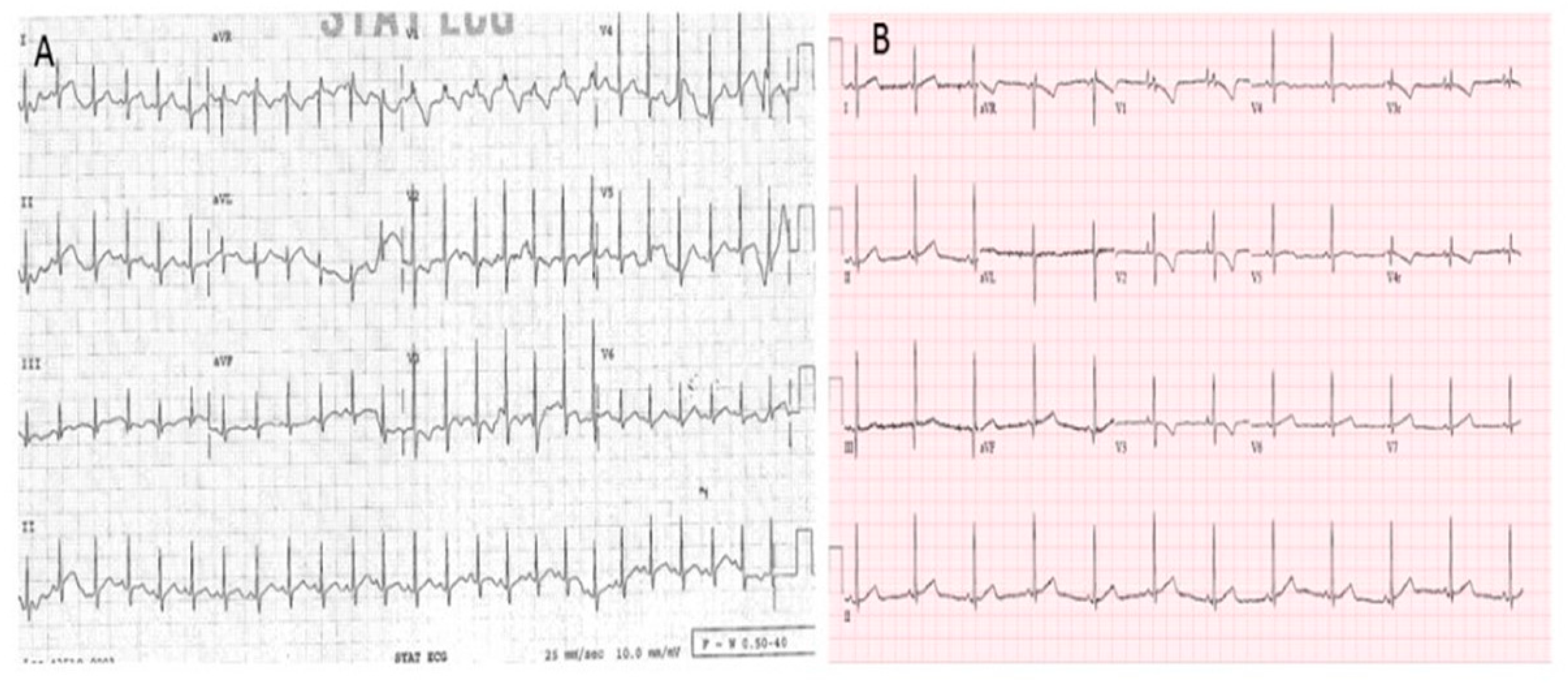
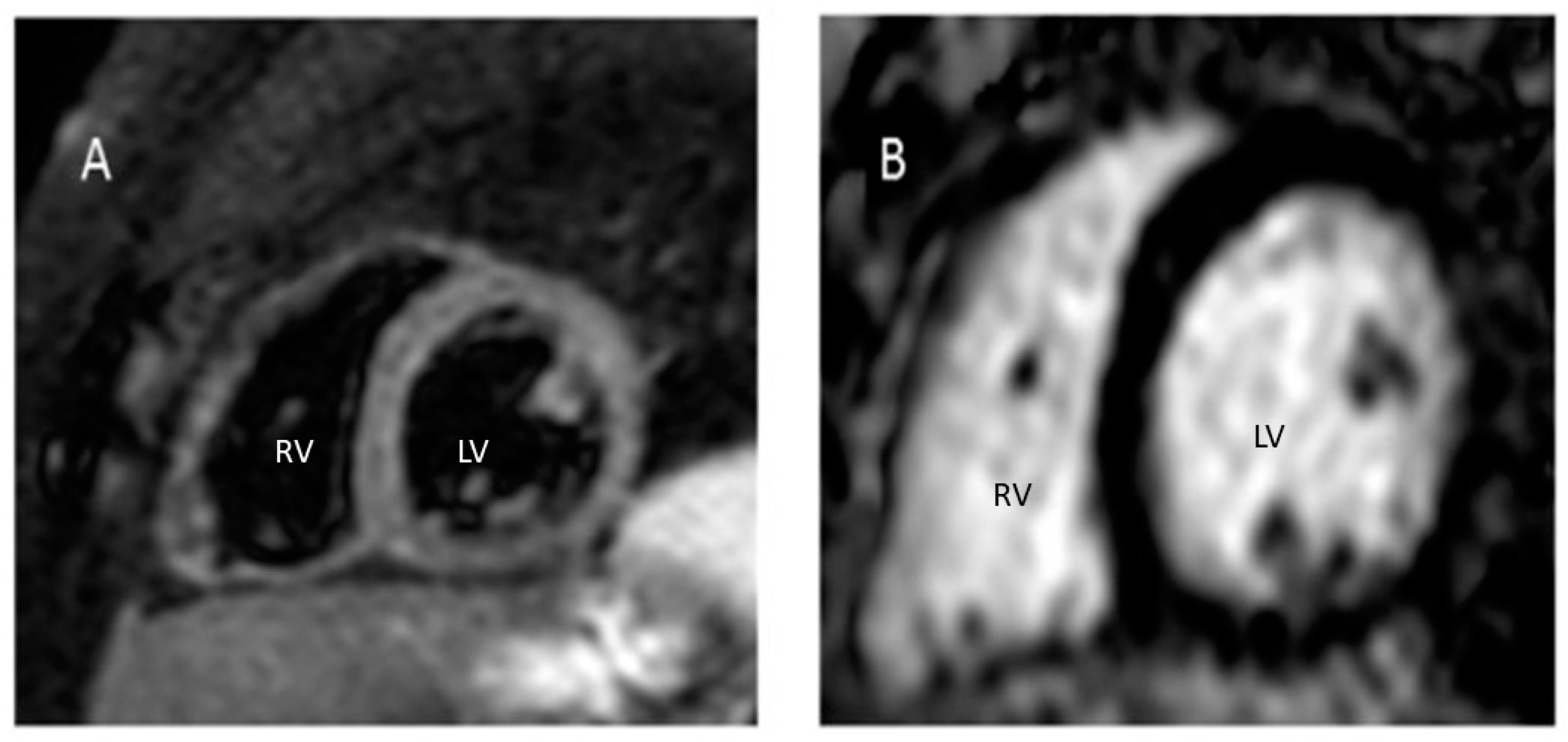
2. Case Report
3. Discussion
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Latif, F.; Farr, M.A.; Clerkin, K.J.; Habal, M.V.; Takeda, K.; Naka, Y.; Restaino, S.; Sayer, G.; Uriel, N. Characteristics and Outcomes of Recipients of Heart Transplant With Coronavirus Disease 2019. JAMA Cardiol. 2020, 5, 1165–1169. [Google Scholar] [CrossRef] [PubMed]
- Lima, B.; Gibson, G.T.; Vullaganti, S.; Malhame, K.; Maybaum, S.; Hussain, S.T.; Shah, S.; Majure, D.T.; Wallach, F.; Jang, K.; et al. COVID-19 in recent heart transplant recipients: Clinicopathologic features and early outcomes. Transpl. Infect. Dis. 2020, 22, e13382. [Google Scholar] [CrossRef] [PubMed]
- Lee, H.; Mantell, B.S.; Richmond, M.E.; Law, S.P.; Zuckerman, W.A.; Addonizio, L.J.; Lee, T.M.; Lytrivi, I.D. Varying presentations of COVID-19 in young heart transplant recipients: A case series. Pediatr. Transplant. 2020, 24, e13780. [Google Scholar] [CrossRef] [PubMed]
- Ketcham, S.W.; Adie, S.K.; Malliett, A.; Abdul-Aziz, A.A.; Bitar, A.; Grafton, G.; Konerman, M.C. Coronavirus disease-2019 in heart transplant recipients in Southeastern Michigan: A case series: COVID-19 in patients with heart transplant. J. Card. Fail. 2020, 26, 457–461. [Google Scholar] [CrossRef] [PubMed]
- Rivinius, R.; Kaya, Z.; Schramm, R.; Boeken, U.; Provaznik, Z.; Heim, C.; Knosalla, C.; Schoenrath, F.; Rieth, A.; Berchtold-Herz, M.; et al. COVID-19 among heart transplant recipients in Germany: A multicenter survey. Clin. Res. Cardiol. 2020, 109, 1531–1539. [Google Scholar] [CrossRef] [PubMed]
- CDC. Multi-System Inflammatory Syndrome in Children (MIS-C) Associated with Coronavirus Disease 2019 (COVID-19). Available online: https://emergency.cdc.gov/han/2020/han00432.asp (accessed on 24 February 2021).
- Bösch, F.; Börner, N.; Kemmner, S.; Lampert, C.; Jacob, S.; Koliogiannis, D.; Stangl, M.; Michel, S.; Kneidinger, N.; Schneider, C.; et al. Attenuated early inflammatory response in solid organ recipients with COVID-19. Clin. Transplant. 2020, 34, e14027. [Google Scholar] [CrossRef] [PubMed]
- Decker, A.; Welzel, M.; Laubner, K.; Grundmann, S.; Kochs, G.; Panning, M.; Thimme, R.; Bode, C.; Wagner, D.; Lother, A. Prolonged SARS-COV-2 shedding and mild course of COVID-19 in a patient after recent heart transplantation. Am. J. Transpl. 2020, 20, 3239–3245. [Google Scholar] [CrossRef] [PubMed]
- Johnson, K.M.; Belfer, J.J.; Peterson, G.R.; Boelkins, M.R.; Dumkow, L.E. Managing COVID-19 in Renal Transplant Recipients: A Review of Recent Literature and Case Supporting Corticosteroid-sparing Immunosuppression. Pharmacother. J. Hum. Pharmacol. Drug Ther. 2020, 40, 517–524. [Google Scholar] [CrossRef] [PubMed]
- Fung, M.; Chiu, C.Y.; DeVoe, C.; Doernberg, S.B.; Schwartz, B.S.; Langelier, C.; Henrich, T.J.; Yokoe, D.; Davis, J.; Hays, S.R.; et al. Clinical outcomes and serologic response in solid organ transplant recipients with COVID-19: A case series from the United States. Arab. Archaeol. Epigr. 2020, 20, 3225–3233. [Google Scholar] [CrossRef] [PubMed]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the author. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Das, B.B. Presentation of SARS-CoV-2 in a Pediatric Heart Transplant Recipient with Multiple Underlying Comorbidities. Transplantology 2021, 2, 87-91. https://doi.org/10.3390/transplantology2010009
Das BB. Presentation of SARS-CoV-2 in a Pediatric Heart Transplant Recipient with Multiple Underlying Comorbidities. Transplantology. 2021; 2(1):87-91. https://doi.org/10.3390/transplantology2010009
Chicago/Turabian StyleDas, Bibhuti B. 2021. "Presentation of SARS-CoV-2 in a Pediatric Heart Transplant Recipient with Multiple Underlying Comorbidities" Transplantology 2, no. 1: 87-91. https://doi.org/10.3390/transplantology2010009
APA StyleDas, B. B. (2021). Presentation of SARS-CoV-2 in a Pediatric Heart Transplant Recipient with Multiple Underlying Comorbidities. Transplantology, 2(1), 87-91. https://doi.org/10.3390/transplantology2010009





