The Placental Stress Code: Lymphocyte Proliferation, Immune and Metabolic Enzyme Shifts Reveal Oxidative Stress in Preeclampsia and Preeclampsia with Gestational Diabetes Mellitus
Abstract
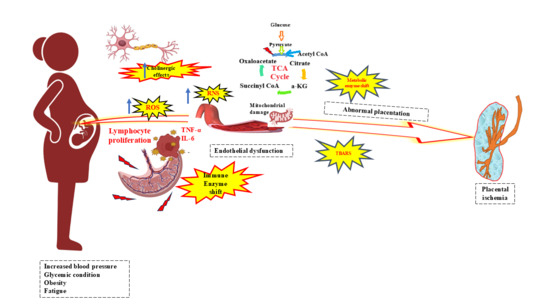
1. Introduction
2. Materials and Methods
2.1. Subjects
2.2. Demographic Profile
2.3. Sample Collection
2.3.1. Fasting Plasma Glucose (FPG) and Oral Glucose Tolerance Test (OGTT)
2.3.2. Albumin Analysis
2.4. Isolation of Peripheral Blood Mononuclear Cells (PBMCs) and Lymphocytes
2.5. PHA-Induced Proliferation of Lymphocytes
2.6. PHA-Induced Cytokine Production
2.7. Metabolic Enzyme Activity
2.7.1. Hexokinase Activity
2.7.2. Pyruvate Kinase Activity
2.7.3. Citrate Synthase Activity
2.7.4. Cytochrome C Oxidase Assay
2.8. Antioxidant Enzyme Assays
2.8.1. Superoxide Dismutase (SOD) Activity
2.8.2. Catalase (CAT) Activity
2.8.3. Glutathione Peroxidase (GPx) Activity
2.8.4. Glutathione-S-Transferase (GST) Activity
2.9. Nitric Oxide Production
2.10. Lipid Peroxidation
2.11. Cholinesterase Activity
2.12. Statistical Analysis
3. Results
3.1. Biochemical Parameters of Pregnant Women
3.2. Lymphocyte Proliferative Activity
3.3. Response of Proinflammatory Cytokine Production
3.4. Effects of Rate-Limiting Metabolic Enzyme Activity
3.5. Effects of Antioxidant Enzyme Activity
3.6. Effects of Reactive Nitrogen Species, Lipid Peroxidation, and Choline Esterase Activities
3.7. Correlations of Anthropometric Variables with Biochemical Parameters
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
Abbreviations
| PE | Preeclampsia |
| GDM | Gestational Diabetes Mellitus |
| NP | Normotensive pregnant |
| FBG | Fasting Blood Glucose |
| PP1 | Postprandial glucose after 1 h |
| PP2 | Postprandial glucose after 2 h |
| HbA1c | Glycated Hemoglobin A1c |
| PHA | phytohemagglutinin |
| NO | nitric oxide |
| TBARS | Thiobarbituric acid reactive substances |
| AChE | acetylcholine esterase |
| IL-6 | Interleukin- 6 |
| TNF-α | Tumor Necrosis Factor alpha |
| FGR | Fetal growth restriction |
| PTB | Preterm birth |
| ROS | reactive oxygen species |
| SOD | superoxide dismutase |
| CAT | catalase |
| GPx | glutathione peroxidase |
| GST | glutathione S-transferase |
| HK | hexokinase |
| CS | citrate synthase |
| PK | pyruvate kinase |
| eNOS | endothelial nitric oxide synthase |
| DIPSI | Diabetes in Pregnancy Study Group India |
| PBMCs | Peripheral Blood Mononuclear Cells |
| MTT | (3-[4,5-dimethylthiazol-2-yl]-2,5-diphenyl tetrazolium bromide |
| DTNB | di-Thio-nitrobenzene |
| CDNB | 1-Chloro-2,4-dinitrobenzene |
| SHS | Suboptimal Health Status |
| IUGR | Intrauterine growth restriction |
References
- Abalos, E.; Cuesta, C.; Carroli, G.; Qureshi, Z.; Widmer, M.; Vogel, J.P.; Souza, J.P. WHO Multicountry Survey on Maternal and Newborn Health Research Network Pre-Eclampsia, Eclampsia and Adverse Maternal and Perinatal Outcomes: A Secondary Analysis of the World Health Organization Multicountry Survey on Maternal and Newborn Health. BJOG Int. J. Obstet. Gynaecol. 2014, 121, 14–24. [Google Scholar] [CrossRef] [PubMed]
- American College of Obstetricians and Gynecologists. Gestational Hypertension and Preeclampsia: ACOG Practice Bulletin, Number 222. Obstet. Gynecol. 2020, 135, e237–e260. [Google Scholar] [CrossRef] [PubMed]
- Choudhury, A.A.; Devi Rajeswari, V. Gestational Diabetes Mellitus–A Metabolic and Reproductive Disorder. Biomed. Pharmacother. 2021, 143, 112183. [Google Scholar] [CrossRef] [PubMed]
- Balu, U.R.; Vasantharekha, R.; Paromita, C.; Ali, K.; Mudgal, G.; Kesari, K.K.; Seetharaman, B. Linking EDC-Laden Food Consumption and Modern Lifestyle Habits with Preeclampsia: A Non-Animal Approach to Identifying Early Diagnostic Biomarkers through Biochemical Alterations. Food Chem. Toxicol. 2024, 194, 115073. [Google Scholar] [CrossRef]
- Pankiewicz, K.; Szczerba, E.; Fijałkowska, A.; Sierdziński, J.; Issat, T.; Maciejewski, T.M. The Impact of Coexisting Gestational Diabetes Mellitus on the Course of Preeclampsia. J. Clin. Med. 2022, 11, 6390. [Google Scholar] [CrossRef]
- Robillard, P.-Y.; Boukerrou, M.; Dekker, G.; Scioscia, M.; Bonsante, F.; Boumahni, B.; Iacobelli, S. Risk Factors for Early and Late Onset Preeclampsia in Reunion Island: Multivariate Analysis of Singleton and Twin Pregnancies. A 20-Year Population-Based Cohort of 2120 Preeclampsia Cases. Reprod. Med. 2021, 2, 131–143. [Google Scholar] [CrossRef]
- Aziz, F.; Khan, M.F.; Moiz, A. Gestational diabetes mellitus, hypertension, and dyslipidemia as the risk factors of preeclampsia. Sci. Rep. 2024, 14, 6182. [Google Scholar] [CrossRef]
- Rana, S.; Lemoine, E.; Granger, J.P.; Karumanchi, S.A. Preeclampsia: Pathophysiology, Challenges, and Perspectives. Circ. Res. 2019, 124, 1094–1112. [Google Scholar] [CrossRef]
- Torres-Torres, J.; Espino-y-Sosa, S.; Martinez-Portilla, R.; Borboa-Olivares, H.; Estrada-Gutierrez, G.; Acevedo-Gallegos, S.; Ruiz-Ramirez, E.; Velasco-Espin, M.; Cerda-Flores, P.; Ramirez-Gonzalez, A.; et al. A Narrative Review on the Pathophysiology of Preeclampsia. Int. J. Mol. Sci. 2024, 25, 7569. [Google Scholar] [CrossRef]
- Cornelius, D.C.; Cottrell, J.; Amaral, L.M.; LaMarca, B. Inflammatory Mediators: A Causal Link to Hypertension during Preeclampsia. Br. J. Pharmacol. 2019, 176, 1914–1921. [Google Scholar] [CrossRef]
- Gelen, V.; Şengül, E.; Atila, G.; Uslu, H.; Makav, M. Association of Gestational Diabetes and Proinflammatory Cytokines (IL-6, TNF-α and IL-1β). J. Embryol. 2017, 1, 6–11. [Google Scholar]
- Zhang, P. Decidual Vasculopathy and Spiral Artery Remodeling Revisited III: Hypoxia and Re-Oxygenation Sequence with Vascular Regeneration. Reprod. Med. 2020, 1, 77–90. [Google Scholar] [CrossRef]
- Cindrova-Davies, T.; Yung, H.-W.; Johns, J.; Spasic-Boskovic, O.; Korolchuk, S.; Jauniaux, E.; Burton, G.J.; Charnock-Jones, D.S. Oxidative Stress, Gene Expression, and Protein Changes Induced in the Human Placenta during Labor. Am. J. Pathol. 2007, 171, 1168–1179. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.-Q.; Zhang, L. Mitochondrial Dysfunction in the Pathogenesis of Preeclampsia. Curr. Hypertens. Rep. 2022, 24, 157–172. [Google Scholar] [CrossRef]
- Ijomone, O.K.; Erukainure, O.L.; Shallie, P.; Naicker, T. Neurotoxicity in Pre-Eclampsia Involves Oxidative Injury, Exacerbated Cholinergic Activity and Impaired Proteolytic and Purinergic Activities in Cortex and Cerebellum. Hum. Exp. Toxicol. 2021, 40, 158–171. [Google Scholar] [CrossRef]
- Petkova-Parlapanska, K.; Kostadinova-Slavova, D.; Angelova, M.; Sadi J Al-Dahwi, R.; Georgieva, E.; Goycheva, P.; Karamalakova, Y.; Nikolova, G. Oxidative Stress and Antioxidant Status in Pregnant Women with Gestational Diabetes Mellitus and Late-Onset Complication of Pre-Eclampsia. Int. J. Mol. Sci. 2025, 26, 3605. [Google Scholar] [CrossRef]
- Yang, Y.; Wu, N. Gestational Diabetes Mellitus and Preeclampsia: Correlation and Influencing Factors. Front. Cardiovasc. Med. 2022, 9, 831297. [Google Scholar] [CrossRef]
- Zhao, Y.; Zhang, X.; Du, N.; Sun, H.; Chen, L.; Bao, H.; Zhao, Q.; Qu, Q.; Ma, D.; Kwak-Kim, J.; et al. Immune Checkpoint Molecules on T Cell Subsets of Pregnancies with Preeclampsia and Gestational Diabetes Mellitus. J. Reprod. Immunol. 2020, 142, 103208. [Google Scholar] [CrossRef]
- Priyanka, H.P.; Sharma, U.; Gopinath, S.; Sharma, V.; Hima, L.; ThyagaRajan, S. Menstrual Cycle and Reproductive Aging Alters Immune Reactivity, NGF Expression, Antioxidant Enzyme Activities, and Intracellular Signaling Pathways in the Peripheral Blood Mononuclear Cells of Healthy Women. Brain Behav. Immun. 2013, 32, 131–143. [Google Scholar] [CrossRef]
- Nikbakht, M.; Pakbin, B.; Nikbakht Brujeni, G. Evaluation of a New Lymphocyte Proliferation Assay Based on Cyclic Voltammetry; an Alternative Method. Sci. Rep. 2019, 9, 4503. [Google Scholar] [CrossRef]
- Priyanka, H.P.; Pratap, U.P.; Nair, R.S.; Vasantharekha, R.; ThyagaRajan, S. Estrogen-Receptor Status Determines Differential Regulation of A1- and A2-Adrenoceptor-Mediated Cell Survival, Angiogenesis, and Intracellular Signaling Responses in Breast Cancer Cell Lines. Med. Oncol. 2024, 41, 92. [Google Scholar] [CrossRef] [PubMed]
- Pratap, U.P.; Hima, L.; Kannan, T.; Thyagarajan, C.; Priyanka, H.P.; Vasantharekha, R.; Pushparani, A.; Thyagarajan, S. Sex-Based Differences in the Cytokine Production and Intracellular Signaling Pathways in Patients with Rheumatoid Arthritis. Arch. Rheumatol. 2020, 35, 545–557. [Google Scholar] [CrossRef] [PubMed]
- Pratap, U.P.; Patil, A.; Sharma, H.R.; Hima, L.; Chockalingam, R.; Hariharan, M.M.; Shitoot, S.; Priyanka, H.P.; ThyagaRajan, S. Estrogen-Induced Neuroprotective and Anti-Inflammatory Effects Are Dependent on the Brain Areas of Middle-Aged Female Rats. Brain Res. Bull. 2016, 124, 238–253. [Google Scholar] [CrossRef] [PubMed]
- Pratap, U.P.; Sharma, H.R.; Mohanty, A.; Kale, P.; Gopinath, S.; Hima, L.; Priyanka, H.P.; ThyagaRajan, S. Estrogen Upregulates Inflammatory Signals through NF-κB, IFN-γ, and Nitric Oxide via Akt/mTOR Pathway in the Lymph Node Lymphocytes of Middle-Aged Female Rats. Int. Immunopharmacol. 2015, 29, 591–598. [Google Scholar] [CrossRef]
- Nair, R.S.; Patel, M.N.; Kannan, T.; Gour, S.; Hariharan, M.M.; Prasanna, V.; Thirumalai, A.; Chockalingam, R.; Vasantharekha, R.; ThyagaRajan, S.; et al. Effects of 17β-Estradiol and Estrogen Receptor Subtype-Specific Agonists on Jurkat E6.1 T-Cell Leukemia Cells. Toxicol. Vitr. 2025, 106, 106057. [Google Scholar] [CrossRef]
- Vasantharekha, R.; Priyanka, H.P.; Nair, R.S.; Hima, L.; Pratap, U.P.; Srinivasan, A.V.; ThyagaRajan, S. Alterations in Immune Responses Are Associated with Dysfunctional Intracellular Signaling in Peripheral Blood Mononuclear Cells of Men and Women with Mild Cognitive Impairment and Alzheimer’s Disease. Mol. Neurobiol. 2024, 61, 2964–2977. [Google Scholar] [CrossRef]
- Li, H.; Yin, B.; Jiang, N.; Zhu, B. Effect of Combined Gestational Diabetes Mellitus and Preeclampsia on Pregnancy Outcomes. Clin. Exp. Obstet. Gynecol. 2025, 52, 27065. [Google Scholar] [CrossRef]
- Alejandro, E.U.; Mamerto, T.P.; Chung, G.; Villavieja, A.; Gaus, N.L.; Morgan, E.; Pineda-Cortel, M.R.B. Gestational Diabetes Mellitus: A Harbinger of the Vicious Cycle of Diabetes. Int. J. Mol. Sci. 2020, 21, 5003. [Google Scholar] [CrossRef]
- Burton, G.J.; Redman, C.W.; Roberts, J.M.; Moffett, A. Pre-Eclampsia: Pathophysiology and Clinical Implications. BMJ 2019, 366, l2381. [Google Scholar] [CrossRef]
- Saito, S.; Nakashima, A.; Shima, T.; Ito, M. Th1/Th2/Th17 and Regulatory T-Cell Paradigm in Pregnancy. Am. J. Reprod. Immunol. 2010, 63, 601–610. [Google Scholar] [CrossRef]
- Darmochwal-Kolarz, D.; Kludka-Sternik, M.; Tabarkiewicz, J.; Kolarz, B.; Rolinski, J.; Leszczynska-Gorzelak, B.; Oleszczuk, J. The Predominance of Th17 Lymphocytes and Decreased Number and Function of Treg Cells in Preeclampsia. J. Reprod. Immunol. 2012, 93, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Mor, G.; Aldo, P.; Alvero, A.B. The Unique Immunological and Microbial Aspects of Pregnancy. Nat. Rev. Immunol. 2017, 17, 469–482. [Google Scholar] [CrossRef] [PubMed]
- Deer, E.; Herrock, O.; Campbell, N.; Cornelius, D.; Fitzgerald, S.; Amaral, L.M.; LaMarca, B. The Role of Immune Cells and Mediators in Preeclampsia. Nat. Rev. Nephrol. 2023, 19, 257–270. [Google Scholar] [CrossRef] [PubMed]
- Harmon, A.C.; Cornelius, D.C.; Amaral, L.M.; Faulkner, J.L.; Cunningham, M.W.; Wallace, K.; LaMarca, B. The Role of Inflammation in the Pathology of Preeclampsia. Clin. Sci. 2016, 130, 409–419. [Google Scholar] [CrossRef]
- Toldi, G.; Rigó, J.; Stenczer, B.; Vásárhelyi, B.; Molvarec, A. Increased Prevalence of IL-17-Producing Peripheral Blood Lymphocytes in Pre-Eclampsia. Am. J. Reprod. Immunol. 2011, 66, 223–229. [Google Scholar] [CrossRef]
- McElwain, C.J.; Tuboly, E.; McCarthy, F.P.; McCarthy, C.M. Mechanisms of Endothelial Dysfunction in Pre-Eclampsia and Gestational Diabetes Mellitus: Windows into Future Cardiometabolic Health? Front. Endocrinol. 2020, 11, 655. [Google Scholar] [CrossRef]
- Yan, M.; Chen, Z.; Tang, J.; Duan, X.; Peng, W.; Liu, R.; Li, W.; Hu, Z.; Liu, Y. Association between Gestational Diabetes Mellitus and Offspring Health: A Two-Sample Mendelian Randomization Study. BMC Pregnancy Childbirth 2025, 25, 321. [Google Scholar] [CrossRef]
- Raghupathy, R. Cytokines as Key Players in the Pathophysiology of Preeclampsia. Med. Princ. Pract. 2013, 22 (Suppl. S1), 8–19. [Google Scholar] [CrossRef]
- Cudihy, D.; Lee, R.V. The Pathophysiology of Pre-Eclampsia: Current Clinical Concepts. J. Obstet. Gynaecol. 2009, 29, 576–582. [Google Scholar] [CrossRef]
- Kristiansen, O.P.; Mandrup-Poulsen, T. Interleukin-6 and Diabetes: The Good, the Bad, or the Indifferent? Diabetes 2005, 54 (Suppl. S2), S114–S124. [Google Scholar] [CrossRef]
- Ferguson, K.K.; Meeker, J.D.; McElrath, T.F.; Mukherjee, B.; Cantonwine, D.E. Repeated Measures of Inflammation and Oxidative Stress Biomarkers in Preeclamptic and Normotensive Pregnancies. Am. J. Obstet. Gynecol. 2017, 216, 527.e1–527.e9. [Google Scholar] [CrossRef] [PubMed]
- Sánchez-Aranguren, L.C.; Prada, C.E.; Riaño-Medina, C.E.; Lopez, M. Endothelial Dysfunction and Preeclampsia: Role of Oxidative Stress. Front. Physiol. 2014, 5, 372. [Google Scholar] [CrossRef] [PubMed]
- Vaka, V.R.; McMaster, K.M.; Cunningham, M.W.; Ibrahim, T.; Hazlewood, R.; Usry, N.; Cornelius, D.C.; Amaral, L.M.; LaMarca, B. Role of Mitochondrial Dysfunction and Reactive Oxygen Species in Mediating Hypertension in the Reduced Uterine Perfusion Pressure Rat Model of Preeclampsia. Hypertension 2018, 72, 703–711. [Google Scholar] [CrossRef] [PubMed]
- Myatt, L.; Cui, X. Oxidative Stress in the Placenta. Histochem. Cell Biol. 2004, 122, 369–382. [Google Scholar] [CrossRef]
- Raijmakers, M.T.M.; Roes, E.M.; Poston, L.; Steegers, E.A.P.; Peters, W.H.M. The Transient Increase of Oxidative Stress during Normal Pregnancy Is Higher and Persists after Delivery in Women with Pre-Eclampsia. Eur. J. Obstet. Gynecol. Reprod. Biol. 2008, 138, 39–44. [Google Scholar] [CrossRef]
- D’Souza, V.; Rani, A.; Patil, V.; Pisal, H.; Randhir, K.; Mehendale, S.; Wagh, G.; Gupte, S.; Joshi, S. Increased Oxidative Stress from Early Pregnancy in Women Who Develop Preeclampsia. Clin. Exp. Hypertens. 2016, 38, 225–232. [Google Scholar] [CrossRef]
- Lappas, M.; Hiden, U.; Desoye, G.; Froehlich, J.; Hauguel-de Mouzon, S.; Jawerbaum, A. The Role of Oxidative Stress in the Pathophysiology of Gestational Diabetes Mellitus. Antioxid. Redox Signal 2011, 15, 3061–3100. [Google Scholar] [CrossRef]
- Coughlan, M.T.; Vervaart, P.P.; Permezel, M.; Georgiou, H.M.; Rice, G.E. Altered Placental Oxidative Stress Status in Gestational Diabetes Mellitus. Placenta 2004, 25, 78–84. [Google Scholar] [CrossRef]
- Madazli, R.; Benian, A.; Gümüştaş, K.; Uzun, H.; Ocak, V.; Aksu, F. Lipid Peroxidation and Antioxidants in Preeclampsia. Eur. J. Obstet. Gynecol. Reprod. Biol. 1999, 85, 205–208. [Google Scholar] [CrossRef]
- Johal, T.; Lees, C.C.; Everett, T.R.; Wilkinson, I.B. The Nitric Oxide Pathway and Possible Therapeutic Options in Pre-Eclampsia. Br. J. Clin. Pharmacol. 2014, 78, 244–257. [Google Scholar] [CrossRef]
- Conrad, K.P.; Benyo, D.F. Placental Cytokines and the Pathogenesis of Preeclampsia. Am. J. Reprod. Immunol. 1997, 37, 240–249. [Google Scholar] [CrossRef]
- Possomato-Vieira, J.S.; Khalil, R.A. Mechanisms of Endothelial Dysfunction in Hypertensive Pregnancy and Preeclampsia. Adv. Pharmacol. 2016, 77, 361–431. [Google Scholar] [CrossRef] [PubMed]
- Borovikova, L.V.; Ivanova, S.; Zhang, M.; Yang, H.; Botchkina, G.I.; Watkins, L.R.; Wang, H.; Abumrad, N.; Eaton, J.W.; Tracey, K.J. Vagus Nerve Stimulation Attenuates the Systemic Inflammatory Response to Endotoxin. Nature 2000, 405, 458–462. [Google Scholar] [CrossRef] [PubMed]
- Klapproth, H.; Reinheimer, T.; Metzen, J.; Münch, M.; Bittinger, F.; Kirkpatrick, C.J.; Höhle, K.D.; Schemann, M.; Racké, K.; Wessler, I. Non-Neuronal Acetylcholine, a Signalling Molecule Synthezised by Surface Cells of Rat and Man. Naunyn Schmiedebergs Arch. Pharmacol. 1997, 355, 515–523. [Google Scholar] [CrossRef] [PubMed]
- Mohammad, F.K.; Mohammed, A.A.; Garmavy, H.M.; Rashid, H.M. Association of Reduced Maternal Plasma Cholinesterase Activity With Preeclampsia: A Meta-Analysis. Cureus 2023, 15, e47220. [Google Scholar] [CrossRef]
- Artyomov, M.N.; Van den Bossche, J. Immunometabolism in the Single-Cell Era. Cell Metab. 2020, 32, 710–725. [Google Scholar] [CrossRef]
- Miller, D.; Motomura, K.; Galaz, J.; Gershater, M.; Lee, E.D.; Romero, R.; Gomez-Lopez, N. Cellular Immune Responses in the Pathophysiology of Preeclampsia. J. Leukoc. Biol. 2022, 111, 237–260. [Google Scholar] [CrossRef]
- Muralimanoharan, S.; Maloyan, A.; Myatt, L. Mitochondrial Function and Glucose Metabolism in the Placenta with Gestational Diabetes Mellitus: Role of miR-143. Clin. Sci. 2016, 130, 931–941. [Google Scholar] [CrossRef]
- Smith, A.N.; Wang, X.; Thomas, D.G.; Tatum, R.E.; Booz, G.W.; Cunningham, M.W. The Role of Mitochondrial Dysfunction in Preeclampsia: Causative Factor or Collateral Damage? Am. J. Hypertens. 2021, 34, 442–452. [Google Scholar] [CrossRef]
- Marín, R.; Chiarello, D.I.; Abad, C.; Rojas, D.; Toledo, F.; Sobrevia, L. Oxidative Stress and Mitochondrial Dysfunction in Early-Onset and Late-Onset Preeclampsia. Biochim. Biophys. Acta Mol. Basis Dis. 2020, 1866, 165961. [Google Scholar] [CrossRef]
- Jahan, F.; Vasam, G.; Green, A.E.; Bainbridge, S.A.; Menzies, K.J. Placental Mitochondrial Function and Dysfunction in Preeclampsia. Int. J. Mol. Sci. 2023, 24, 4177. [Google Scholar] [CrossRef]
- Szlapinski, S.K.; Hill, D.J. Metabolic Adaptations to Pregnancy in Healthy and Gestational Diabetic Pregnancies: The Pancreas–Placenta Axis. Curr. Vasc. Pharmacol. 2021, 19, 141–153. [Google Scholar] [CrossRef]
- Spradley, F.T.; Palei, A.C.; Granger, J.P. Immune Mechanisms Linking Obesity and Preeclampsia. Biomolecules 2015, 5, 3142–3176. [Google Scholar] [CrossRef]
- Bodnar, L.M.; Ness, R.B.; Markovic, N.; Roberts, J.M. The Risk of Preeclampsia Rises with Increasing Prepregnancy Body Mass Index. Ann. Epidemiol. 2005, 15, 475–482. [Google Scholar] [CrossRef]
- Zhao, X.; Lan, Y.; Shao, H.; Peng, L.; Chen, R.; Yu, H.; Hua, Y. Associations between Prepregnancy Body Mass Index, Gestational Weight Gain, and Pregnancy Outcomes in Women with Twin Pregnancies: A Five-Year Prospective Study. Birth 2022, 49, 741–748. [Google Scholar] [CrossRef]
- Catalano, P.M.; Shankar, K. Obesity and Pregnancy: Mechanisms of Short Term and Long Term Adverse Consequences for Mother and Child. BMJ 2017, 356, j1. [Google Scholar] [CrossRef]
- García-morales, A.; Lomas-soria, C.; Granados-higa, G.; García-quiroz, J.; Avila, E.; Olmos-ortiz, A.; Díaz, L. Inflammation in Pregnancy: Key Drivers, Signaling Pathways and Associated Complications. Arch. Med. Res. 2026, 2, 103301. [Google Scholar] [CrossRef] [PubMed]


 NP
NP  PE
PE  PE + GDM.
PE + GDM.
 NP
NP  PE
PE  PE + GDM.
PE + GDM.




| Variables | NP (M ± SD) | PE (M ± SD) | PE + GDM (M ± SD) |
|---|---|---|---|
| FBG (mg/dL) | 83.63 ± 7.52 | 82.89 ± 6.36 | 109 ± 14.26 * |
| PP1 (mg/dL) | 106.5 ± 19.43 | 124 ± 13.66 | 156.9 ± 38.31 * |
| PP2 (mg/dL) | 102 ± 19.01 | 94.6 ± 15.05 | 136.2 ± 13.51 * |
| HbA1c (%) | 4.92 ± 0.19 | 5.24 ± 0.54 | 5.82 ± 0.58 * |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Balu, U.R.; Vasantharekha, R.; Priyanka, H.P.; Nair, R.S.; Mudgal, G.; Barathi, S. The Placental Stress Code: Lymphocyte Proliferation, Immune and Metabolic Enzyme Shifts Reveal Oxidative Stress in Preeclampsia and Preeclampsia with Gestational Diabetes Mellitus. Reprod. Med. 2025, 6, 43. https://doi.org/10.3390/reprodmed6040043
Balu UR, Vasantharekha R, Priyanka HP, Nair RS, Mudgal G, Barathi S. The Placental Stress Code: Lymphocyte Proliferation, Immune and Metabolic Enzyme Shifts Reveal Oxidative Stress in Preeclampsia and Preeclampsia with Gestational Diabetes Mellitus. Reproductive Medicine. 2025; 6(4):43. https://doi.org/10.3390/reprodmed6040043
Chicago/Turabian StyleBalu, Usha Rani, Ramasamy Vasantharekha, Hannah P. Priyanka, Rahul S. Nair, Gaurav Mudgal, and Seetharaman Barathi. 2025. "The Placental Stress Code: Lymphocyte Proliferation, Immune and Metabolic Enzyme Shifts Reveal Oxidative Stress in Preeclampsia and Preeclampsia with Gestational Diabetes Mellitus" Reproductive Medicine 6, no. 4: 43. https://doi.org/10.3390/reprodmed6040043
APA StyleBalu, U. R., Vasantharekha, R., Priyanka, H. P., Nair, R. S., Mudgal, G., & Barathi, S. (2025). The Placental Stress Code: Lymphocyte Proliferation, Immune and Metabolic Enzyme Shifts Reveal Oxidative Stress in Preeclampsia and Preeclampsia with Gestational Diabetes Mellitus. Reproductive Medicine, 6(4), 43. https://doi.org/10.3390/reprodmed6040043