A Demonstration of the Capability of Low-Cost Hyperspectral Imaging for the Characterisation of Coral Reefs
Abstract
:1. Introduction
2. Materials and Methods
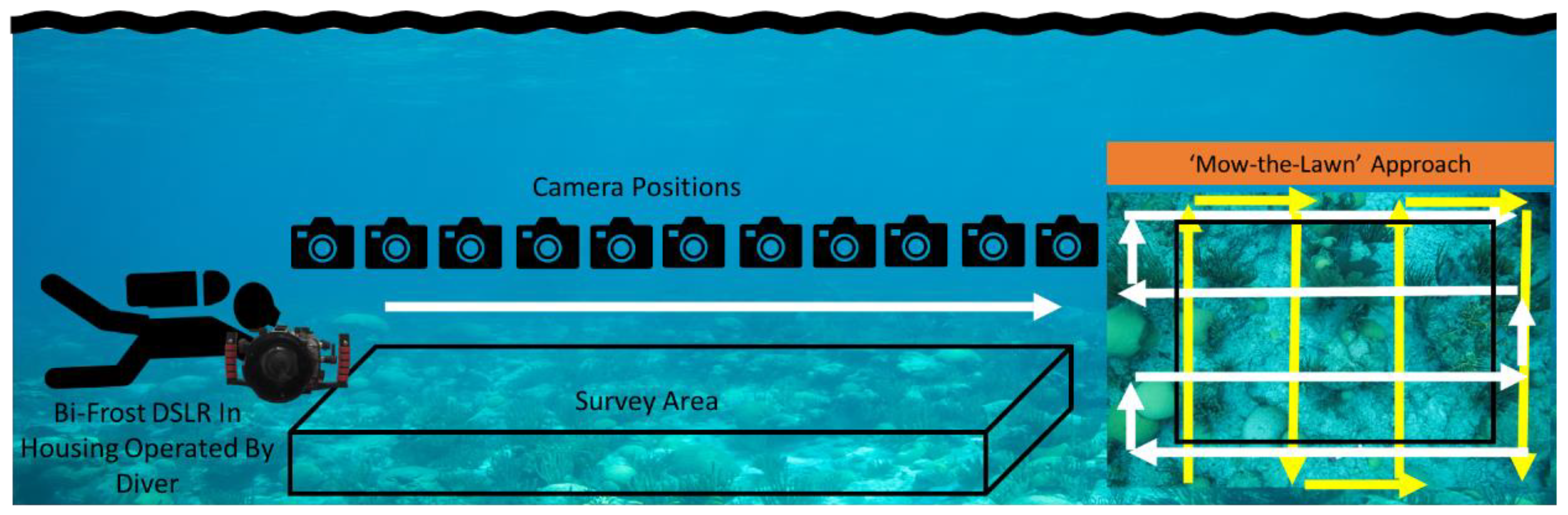
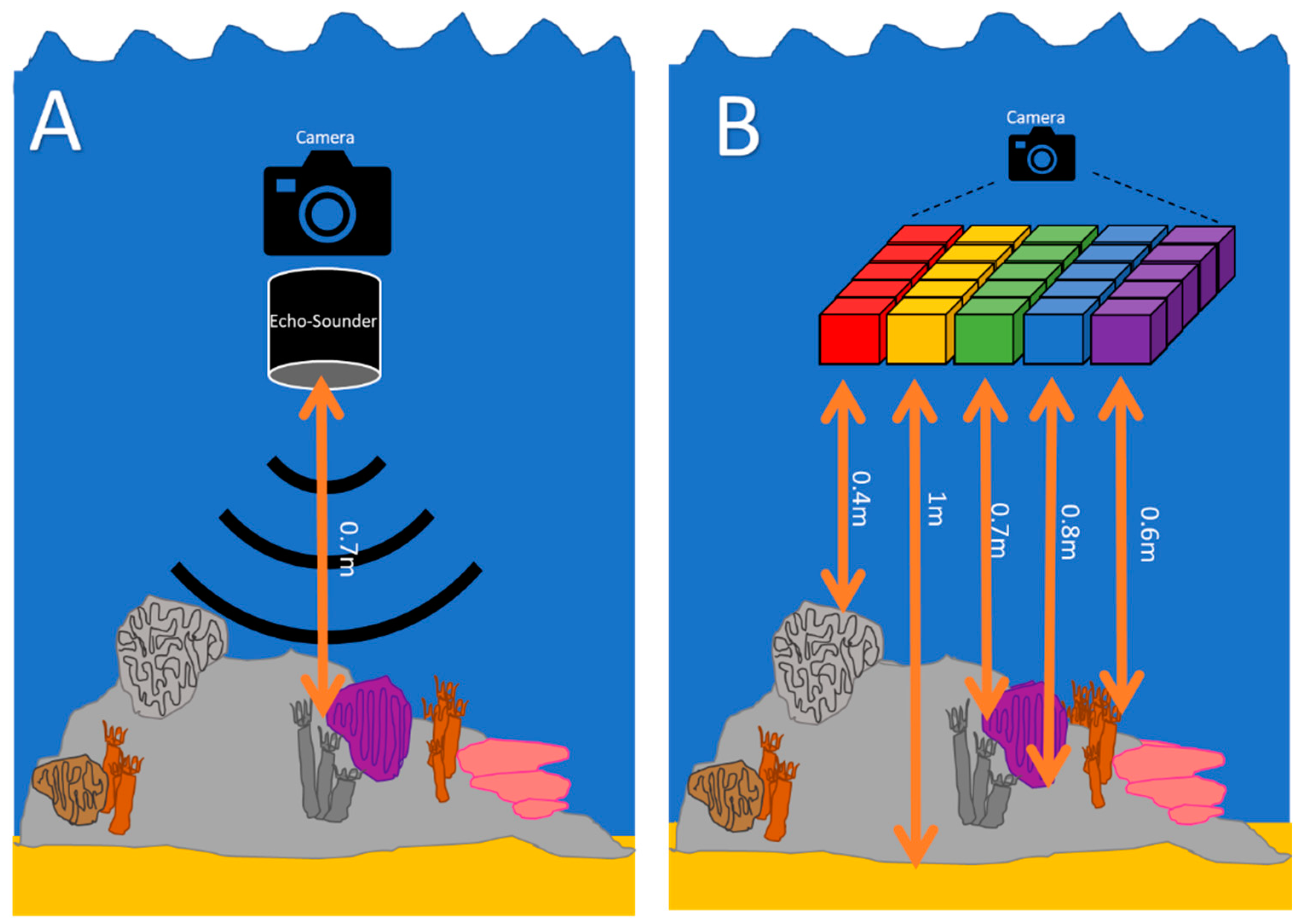
2.1. Hyperspectral Data Collection
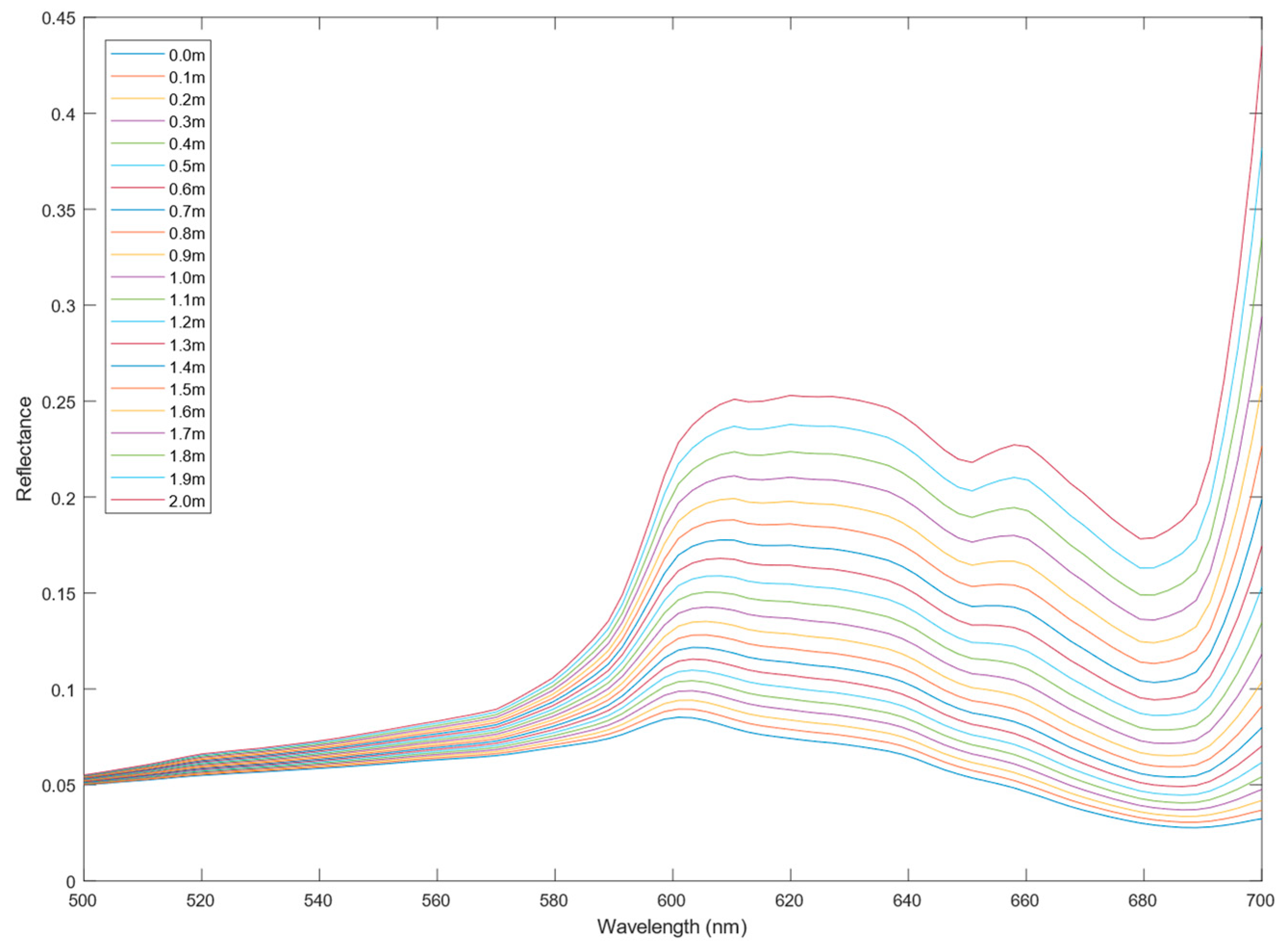
2.2. Hyperspectral Data Correction
2.3. Strucutre-from-Motion (SfM) Photogrammetry
2.4. Spectral Classification
2.5. Survey Area
3. Results
3.1. RGB Represetations of Hyperspectral Data
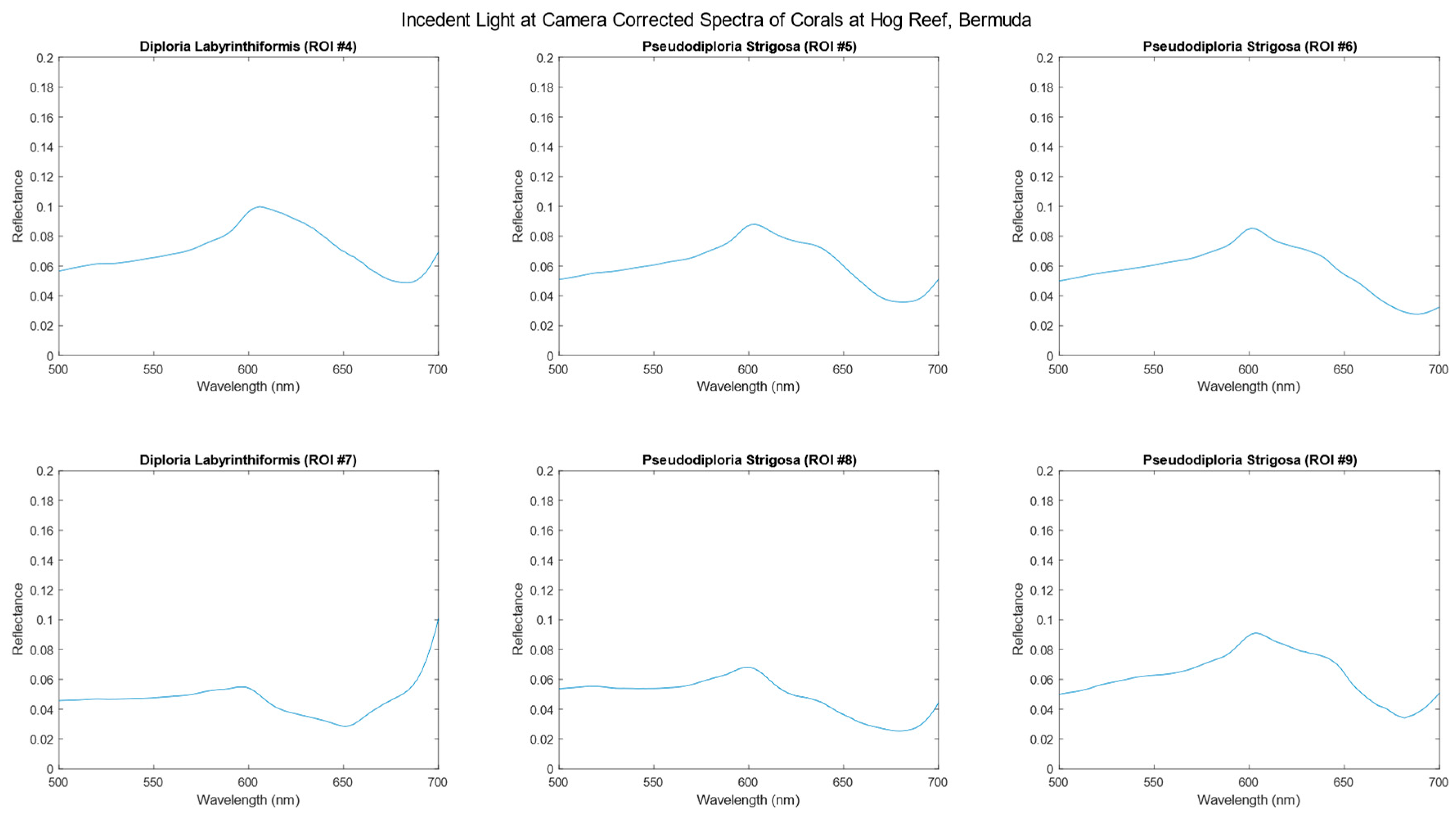
3.2. Incident Light Corrected Hyperspectral Data
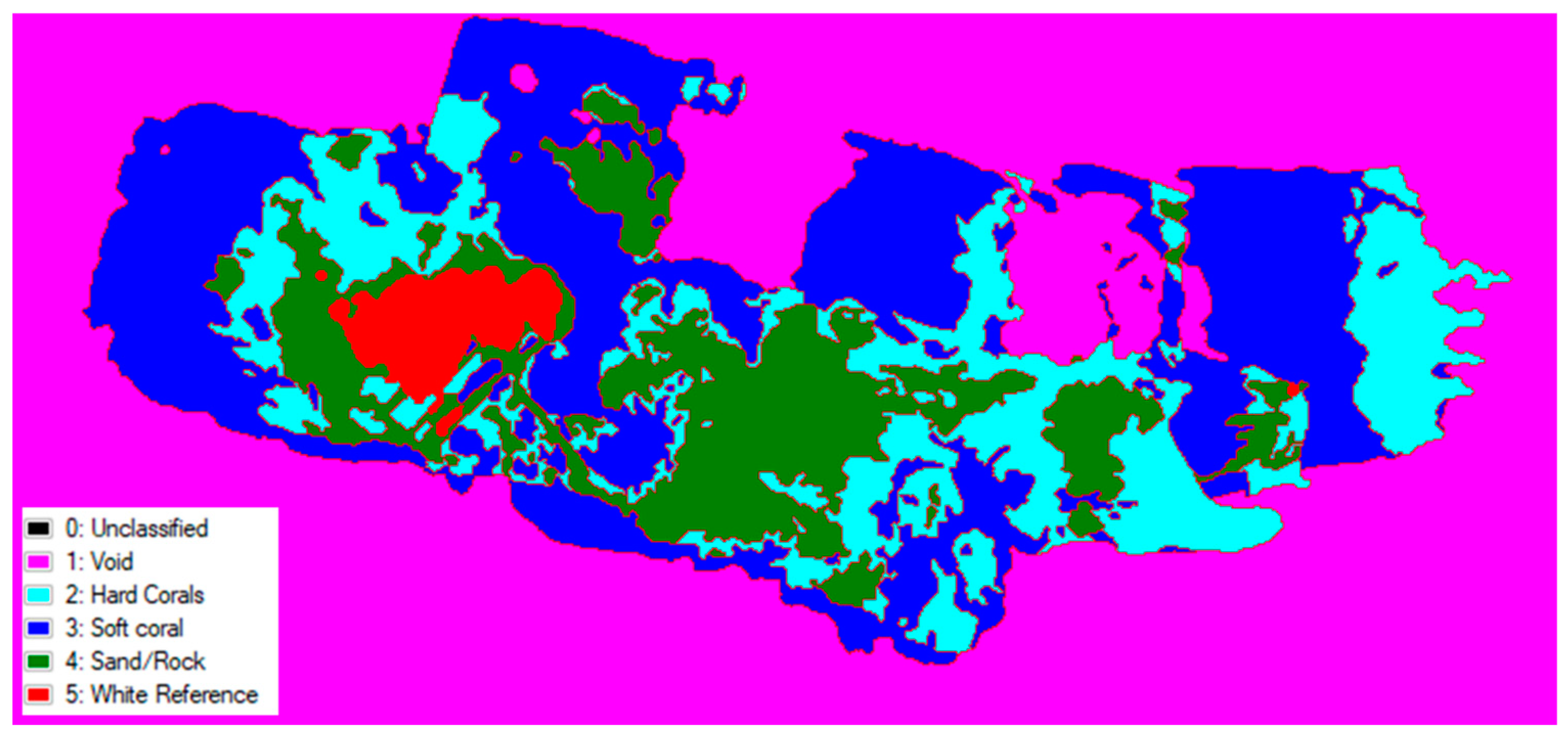
3.3. Simple Spectral Classification
3.4. Digital Elevation Models (DEM) of Reefs from Images Taken by a Hypersepctral Imager
3.5. Physical Information of the ROIs Derived from DEMs
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Ødegård, Ø.; Mogstad, A.A.; Johnsen, G.; Sørensen, A.J.; Ludvigsen, M. Underwater hyperspectral imaging: A new tool for marine archaeology. Appl. Opt. 2018, 57, 3214–3223. [Google Scholar] [CrossRef] [PubMed]
- Johnsen, G.; Ludvigsen, M.; Sørensen, A.; Aas, L.M.S. The use of underwater hyperspectral imaging deployed on remotely operated vehicles—Methods and applications. IFAC-Papers Online 2016, 49, 476–481. [Google Scholar] [CrossRef]
- Mogstad, A.A.; Odegard, O.; Nornes, S.M.; Ludvigsen, M.; Johnsen, G.; Berge, J. Mapping the historical shipwreck Figaro in the high arctic using underwater sensor-carrying robots. Remote Sens. 2020, 12, 997. [Google Scholar] [CrossRef]
- Beck, A.J.; Kaandrop, M.; Hamm, T.; Bogner, B.; Kossel, E.; Lenz, M.; Haeckel, M. Rapid shipboard measurement of net-collected marine microplastic polymer types using near-infrared hyperspectral imaging. Anal. Bioanal. Chem. 2023, 415, 2989–2998. [Google Scholar] [CrossRef] [PubMed]
- Serranti, S.; Palmieri, R.; Bonifazi, G.; Cózar, A. Characterization of microplastic litter from oceans by an innovative approach based on hyperspectral imaging. Waste Manag. 2018, 76, 117–125. [Google Scholar] [CrossRef]
- Freitas, S.; Silva, H.; Silva, E. Hyperspectral Imaging Zero-Shot Learning for Remote Marine Litter Detection and Classification. Remote Sens. 2022, 14, 5516. [Google Scholar] [CrossRef]
- Balsi, M.; Esposito, S.; Moroni, M. Hyperspectral characterization of marine plastic litters. MetroSea 2019, 2, 28–32. [Google Scholar] [CrossRef]
- Garaba, S.P.; Dierssen, H.M. An airborne remote sensing case study of synthetic hydrocarbon detection using short wave infrared absorption features identified from marine-harvested macro- and microplastics. Remote Sens. Environ. 2018, 205, 224–235. [Google Scholar] [CrossRef]
- Teague, J.; Megson-Smith, D.A.; Yannick, V.; Scott, T.B.; Day, J.C.C. Underwater Spectroscopic Techniques for In-Situ Nuclear Waste Characterisation; Waste Management: Phoenix, AZ, USA, 2022; pp. 1–9. [Google Scholar]
- Chennu, A.; Färber, P.; De’ath, G.; De Beer, D.; Fabricius, K.E. A diver-operated hyperspectral imaging and topographic surveying system for automated mapping of benthic habitats. Sci. Rep. 2017, 7, 7122. [Google Scholar] [CrossRef]
- Johnsen, G.; Volent, Z.; Dierssen, H.; Pettersen, R.; Van Ardelan, M.; Søreide, F.; Moline, M. Underwater hyperspectral imagery to create biogeochemical maps of seafloor properties. Subsea Opt. Imaging 2013, 508, 536e–540e. [Google Scholar]
- Teague, J.; Megson-Smith, D.A.; Allen, M.J.; Day, J.C.C.; Scott, T.B. A Review of Current and New Optical Techniques for Coral Monitoring. Oceans 2022, 3, 30–45. [Google Scholar] [CrossRef]
- Gleason, A.C.R.; Reid, R.P.; Voss, K.J. Automated Classification of Underwater Multispectral Imagery for Coral Reef Monitoring. In Proceedings of the OCEANS 2007 Conference, Vancouver, BC, Canada, 29 September–4 October 2007. [Google Scholar] [CrossRef]
- Thompson, D.R.; Hochberg, E.J.; Asner, G.P.; Green, R.O.; Knapp, D.E.; Gao, B.C.; Garcia, R.; Gierach, M.; Lee, Z.; Maritorena, S.; et al. Airborne mapping of benthic reflectance spectra with Bayesian linear mixtures. Remote Sens. Environ. 2017, 200, 18–30. [Google Scholar] [CrossRef]
- Teague, J.; Willans, J.; Allen, M.; Scott, T.; Day, J. Hyperspectral imaging as a tool for assessing coral health utilising natural fluorescence. J. Spectr. Imaging 2019, 8, a7. [Google Scholar] [CrossRef]
- Kok, J.; Bainbridge, S.; Olsen, M.; Rigby, P. Towards Effective Aerial Drone-based Hyperspectral Remote Sensing of Coral Reefs. In Proceedings of the Oceans 2020 Conference, Biloxi, MS, USA, 5–30 October 2020. [Google Scholar] [CrossRef]
- Teague, J.; Willans, J.; Megson-Smith, D.A.; Day, J.C.C.; Allen, M.J.; Scott, T.B. Using Colour as a Marker for Coral ‘Health’: A Study on Hyperspectral Reflectance and Fluorescence Imaging of Thermally Induced Coral Bleaching. Oceans 2022, 3, 547–556. [Google Scholar] [CrossRef]
- Joyce, K.E.; Phinn, S.R. Hyperspectral analysis of chlorophyll content and photosynthetic capacity of coral reef substrates. Limnol. Oceanogr. 2003, 48, 489–496. [Google Scholar] [CrossRef]
- Hochberg, E.J.; Apprill, A.M.; Atkinson, M.J.; Bidigare, R.R. Bio-optical modeling of photosynthetic pigments in corals. Coral Reefs 2006, 25, 99–109. [Google Scholar] [CrossRef]
- Boyd, J. Drones survey the great barrier reef: Aided by AI, hyperspectral cameras can distinguish bleached from unbleached coral—[News]. IEEE Spectr. 2019, 27, 7–9. [Google Scholar] [CrossRef]
- Casella, E.; Collin, A.; Harris, D.; Ferse, S.; Bejarano, S.; Hench, J.L.; Rovere, A. Mapping coral reefs using consumer-grade drones and structure from motion photogrammetry techniques. Coral Reefs 2017, 36, 269–275. [Google Scholar] [CrossRef]
- Palandro, D.; Andréfouët, S.; Muller-Karger, F.E.; Dustan, P.; Hu, C.; Hallock, P. Detection of changes in coral reef communities using Landsat-5 TM and Landsat-7 ETM+ data. Can. J. Remote Sens. 2003, 2, 201–209. [Google Scholar] [CrossRef]
- Joyce, K.E.; Phinn, S.R.; Roelfsema, C.M.; Neil, D.T.; Dennison, W.C. Combining Landsat ETM+ and Reef Check classifications for mapping coral reefs: A critical assessment from the southern Great Barrier Reef, Australia. Coral Reefs 2004, 23, 21–25. [Google Scholar] [CrossRef]
- Mizuochi, H.; Tsuchida, S.; Mizuyama, M.; Yamamoto, S.; Iwao, K. Multi-band bottom index: A novel approach for coastal environmental monitoring using hyperspectral data. Remote Sens. Appl. Soc. Environ. 2022, 27, 100797. [Google Scholar] [CrossRef]
- Foo, S.A.; Asner, G.P. Scaling up coral reef restoration using remote sensing technology. Front. Mar. Sci. 2019, 3, 79. [Google Scholar] [CrossRef]
- Boreman, G.D. Classification of imaging spectrometers for remote sensing applications. Opt. Eng. 2005, 44, 013602. [Google Scholar] [CrossRef]
- Maritorena, S.; Morel, A.; Gentili, B. Diffuse reflectance of oceanic shallow waters: Influence of water depth and bottom albedo. Limnol. Oceanogr. 1994, 39, 1689–1703. [Google Scholar] [CrossRef]
- Buiteveld, H.; Hakvoort, J.H.M.; Donze, M. Optical properties of pure water. Ocean Opt. XII 1994, 2258, 174–183. [Google Scholar] [CrossRef]
- Baker, K.S.; Smith, R.C. Optical properties of the clearest natural waters (200–800 nm). Appl. Opt. 1981, 20, 177–184. [Google Scholar] [CrossRef]
- Teague, J.; Scott, T.B. Underwater Photogrammetry and 3D Reconstruction of Submerged Objects in Shallow Environments by ROV and Underwater GPS. J. Mar. Sci. Res. Technol. 2017, 1, 7. [Google Scholar]
- Hochberg, E.J.; Atkinson, M.J. Spectral discrimination of coral reef benthic communities. Coral Reefs 2000, 19, 164–171. [Google Scholar] [CrossRef]
- Colomina, I.; Molina, P. Unmanned aerial systems for photogrammetry and remote sensing: A review. ISPRS J. Photogramm. Remote Sens. 2014, 92, 79–97. [Google Scholar] [CrossRef]
- Hochberg, E.J. Bermuda Benthic Community Mapping Program Report; Bermuda Department of Environment and Natural ReSources: Paget, Bermuda, 2015.
- Hochberg, E.J.; Atkinson, M.J.; Apprill, A.; Andréfouët, S. Spectral reflectance of coral. Coral Reefs 2004, 23, 84–95. [Google Scholar] [CrossRef]
- Zeng, K.; Xu, Z.; Yang, Y.; Liu, Y.; Zhao, H.; Zhang, Y.; Xie, B.; Zhou, W.; Li, C.; Cao, W. In situ hyperspectral characteristics and the discriminative ability of remote sensing to coral species in the South China Sea. Ocean. Opt. XII 2022, 59, 272–294. [Google Scholar] [CrossRef]




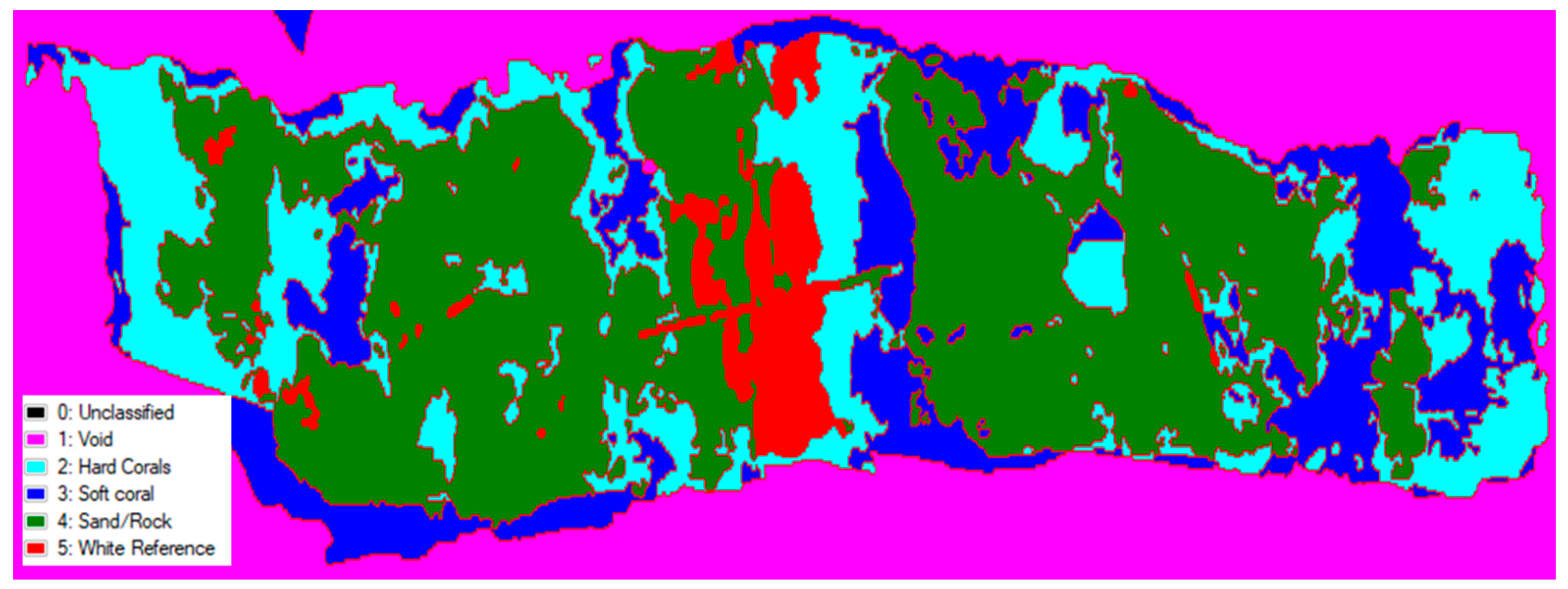


| Class | Pixel Count | Percentage Coverage (%) |
|---|---|---|
| Hard Coral | 30,358 | 25.437 |
| Soft Coral | 25,258 | 21.248 |
| Sand/Rock | 57,513 | 48.190 |
| White Reference | 6116 | 5.125 |
| Class | Pixel Count | Percentage Coverage (%) |
|---|---|---|
| Hard Coral | 46,875 | 30.179 |
| Soft Coral | 81,835 | 52.689 |
| Sand/Rock | 22,951 | 14.777 |
| White Reference | 3657 | 2.355 |
| Label | Species | Perimeter (cm) | Area (cm2) |
|---|---|---|---|
| ROI #4 | DLAB | 52.66 | 1.53 |
| ROI #5 | PSTR | 75.33 | 3.75 |
| ROI #6 | PSTR | 74.83 | 3.70 |
| ROI #7 | DLAB | 83.13 | 2.53 |
| ROI #8 | PSTR | 61.63 | 2.36 |
| ROI #9 | PSTR | 23.22 | 0.35 |
| Label | Species | Perimeter (cm) | Area (cm2) |
|---|---|---|---|
| ROI #5 | MCAV | 20.81 | 0.32 |
| ROI #6 | MCAV | 17.25 | 0.20 |
| ROI #7 | DLAB | 23.84 | 0.32 |
| ROI #8 | PSTR | 12.40 | 0.11 |
| ROI #9 | MCAV | 17.97 | 0.22 |
| ROI #12 | MCAV | 17.04 | 0.21 |
| ROI #13 | DLAB | 30.30 | 0.63 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Teague, J.; Day, J.C.C.; Allen, M.J.; Scott, T.B.; Hochberg, E.J.; Megson-Smith, D. A Demonstration of the Capability of Low-Cost Hyperspectral Imaging for the Characterisation of Coral Reefs. Oceans 2023, 4, 286-300. https://doi.org/10.3390/oceans4030020
Teague J, Day JCC, Allen MJ, Scott TB, Hochberg EJ, Megson-Smith D. A Demonstration of the Capability of Low-Cost Hyperspectral Imaging for the Characterisation of Coral Reefs. Oceans. 2023; 4(3):286-300. https://doi.org/10.3390/oceans4030020
Chicago/Turabian StyleTeague, Jonathan, John C. C. Day, Michael J. Allen, Thomas B. Scott, Eric J. Hochberg, and David Megson-Smith. 2023. "A Demonstration of the Capability of Low-Cost Hyperspectral Imaging for the Characterisation of Coral Reefs" Oceans 4, no. 3: 286-300. https://doi.org/10.3390/oceans4030020
APA StyleTeague, J., Day, J. C. C., Allen, M. J., Scott, T. B., Hochberg, E. J., & Megson-Smith, D. (2023). A Demonstration of the Capability of Low-Cost Hyperspectral Imaging for the Characterisation of Coral Reefs. Oceans, 4(3), 286-300. https://doi.org/10.3390/oceans4030020










