Abstract
We present an informational study of a spherically confined hydrogen atom, a hydrogenic ion confined in a strongly coupled plasma, a spherically confined harmonic oscillator, and a particle confined in a cage. For this, we have implemented a numerical procedure to obtain information entropies of these confined quantum systems. The procedure is based on the variational formalism that uses the finite element method (FEM) for the expansion of the wavefunction in terms of local base functions. Such a study is carried out in order to analyze what happens in the rigorous confinement regime. In particular, we have shown that the effects of the interaction potential is no longer important for rigorous confinements and the studied systems start to behave just like an electron confined by a impenetrable spherical cage. When possible, we compared our results with those published in the literature.
1. Introduction
The study of confined quantum systems is a field that examines the spatial limitation effects on the physical properties of the electrons, nuclei, atoms, or molecules. The literature contains several theoretical and experimental works indicating that the spatial confinement changes the physical and chemical properties of the systems [1,2,3]. For instance, when the hydrogen atom is confined by impenetrable walls the energy spectrum is altered; moreover, in confined environments some alkali metals present electronic configurations similar to those of transition metals [4,5]. In recent years, with the increases in computational capacity and the development of modern techniques, this question has received considerable attention [6,7,8,9,10].
In the Schrödinger equation that describes the confined system, we can use a model potential that has both nuclear and confinement potential properties [11]. The choice of the potential model depends on the physical system of interest and of the characteristics of the confinement [12,13,14,15,16]. For instance, we can use infinite potential barriers models [17,18] or soft barriers models [19,20].
In particular, strong or rigorous confinement region includes interesting details such as: (a) it has a highly concentrated probability density when compared to the weakly confined regions; (b) the kinetic energy of electrons is large in contrast to their potential energy; and (c) the influence of the confinement barrier becomes greater than the free system potential [21,22,23,24]. The rigorous confinement region is established when the confinement radius of the system goes to zero. Energy studies based on ionization energy have been successfully undertaken in mapping this specific region [25,26,27].
In the atomic, molecular and chemical-physical context we define the informational entropies (or Shannon entropy) on position, ; on momentum, ; spaces; and the entropy sum, , by adding and [28,29]. These informational quantities have been utilized in the study of various confined quantum systems such as the confined hydrogen-like atoms [30,31,32,33,34,35], the confined He-like atoms [36,37], plasma environments [38,39], and in many other systems [40,41,42]. Analysis of basis functions [29,43] and electronic correlation [36,44,45], as well as physical and chemical phenomena [46,47,48,49] are also being approached in the informational field.
Despite of the energy analysis, the information entropies have been used as a powerful tool to study the regions where the effects of confinement are extreme. According to previous studies using entropic quantities, we have that the influence of the one-dimensional harmonic [50] and Coulomb [29] potentials is practically nullified by the presence of strong confinement. Moreover, in this specific region of the system (where the confinement radius tends to zero) the effects of Coulomb correlation in confined helium-like atoms become negligible [36].
The main goal of this work is to analyze how information entropy can contribute to the understanding of the strong confinement regime. For this, we have determined entropic quantities of interest for the spherically confined hydrogen atom, the hydrogenic ion confined in a strongly coupled plasma, and the spherically confined harmonic oscillator and compare them with the results of a particle confined in a cage.
This paper is organized as follows: in Section 2 we present the physical systems of interest, the numerical procedures based on the variational formalism using the finite element method, and beyond the informational formalism adopted; in Section 3 we discuss the results obtained; and, finally, in Section 4 we summarize the central aspects of our investigation.
2. Theoretical Background
In this section, we present the concepts and methods applied in our research. In Section 2.1, we discuss the physical systems and the variational formulation of the problem. Moreover, in Section 2.2, we define the informational quantities of interest.
2.1. System of Interest
The radial Schrödinger equation, using atomic units, is given by
where is the radial wavefunction solution, the energy of the stationary state, and the potential model. The labels n and l refer to the main and angular quantum numbers, respectively. The total wavefunction solution for a particular quantum numbers is given by
where is the correspondent spherical harmonic and is the solid angled.
Here, we consider four different model potentials of the type infinite spherical barrier to confine an electron. For models that contain an atomic nucleus, it is located at the center of the hard sphere. Our starting point is the potential that describes a particle (electron) confined in a spherical cage, that is,
where is the confinement radius. For the r range of values between the particle is free of potential and at the confinement frontier the value of the potential is infinite.
Following, we have the Coulomb potential model of confined hydrogenic-like atoms,
where Z is the nuclear charge. We also consider an electron under influence of the isotropic harmonic potential, given by,
where is the angular frequency.
Finally, we selected to study one electron in a plasmatic environment of the sphere model. For strongly coupled plasma surrounding an ion having a single valence electron, one can define a sphere of radius such that the plasma electrons with density is sufficient to neutralize () of central positive charge:
Under this condition the interaction potential energy of the electron with an ion is
To solve the radial Schrödinger Equation (1) with the potentials (3), (4), (5), or (6) by using the variational principle, for a given angular moment l, it corresponds to find the results that agree with the extreme condition of the energy functional, , where the function must vanish at the origin and at the surface of a sphere of radius due to confinement (boundary conditions).
In variational context, the radial wavefunctions and energy eigenvalues are found by expanding the function in terms of a finite basis set , that is,
where are the coefficients of the expansion. The efficiency of the numerical calculation depends on the choice of the finite basis set. The variational solutions are obtained by solving the generalized eigenvalue eigenvector problem
where is the vector of the expansion coefficients , being
and
where the effective potential is written as .
The variational methodology that we use to solve the problem is the p-version of the finite element method (p-FEM). In the radial case, the p-FEM consist of dividing the range of integration into elements, being the i-th element defined in the range of up to with and , and the radial wavefunction is expanded in local base functions {} satisfying the following property:
where is the j-th polynomial associated with i-th element and is its highest order given by the parameter . Thus, considering the properties (11), the matrix representation of any local operator assumes a block tridiagonal form and the spatial confinement on the radial wavefunction can be imposed easily just removing the basis function of the expansion. For more details, see references [11,51,52].
2.2. Shannon Informational Entropy
The Shannon information entropy in the atomic, molecular, and chemical physics context can be defined in terms of quantum-mechanical wavefunction. Therefore, information entropies on position, , and momentum, , spaces, in atomic units, are recognized as [29]
and
where and are the total position and momentum space wavefunction, respectively. Detailed discussions of the dimensionality of expressions (12) and (13) can be found in Refs. [29,53].
Using the variable separation method the wavefunctions can be written as
where are the spherical harmonics and is the solid angled. Furthermore, the relationship between and is given by [54]
being the spherical Bessel function. Employing the expressions (14) in Equations (12) and (13) we separate the and entropies into two parts, that is [55],
and
The radial parts and that depends on the potential function are
and
The angular contribution known as angular entropy (common for and ) is given by
The quantity is independent of the potential and has a closed form determined by
where is the Legendre function.
The quantity is obtained from entropy sum of . From the entropy sum we can still derive the following entropy uncertainty relation:
This entropy uncertainty relationship is also known as the BBM inequality relation [56], which it is a common theorem for any quantum system. From expression (23) we can obtain Kennard’s uncertainty relationship as discussed in Ref. [56].
3. Results and Discussions
This section presents the data obtained respective analysis of Shannon entropies, in atomic units, for a particle confined in a spherical cage (Equation (3)), the confined hydrogen atom (Equation (4) with ), spherically confined harmonic oscillator (Equation (5) with ), and hydrogenic ion confined in a strongly coupled plasma (Equation (6) with ). For this, we have implemented the procedure based on the theory presented previously using the computer language Fortran. Unless stated otherwise, all curves were fitted with cubic spline interpolation utilizing the free application SciDAVis. Particularly, for the hydrogen atom, our computational implementation obtained values equivalent to accurate results existing in the literature [33].
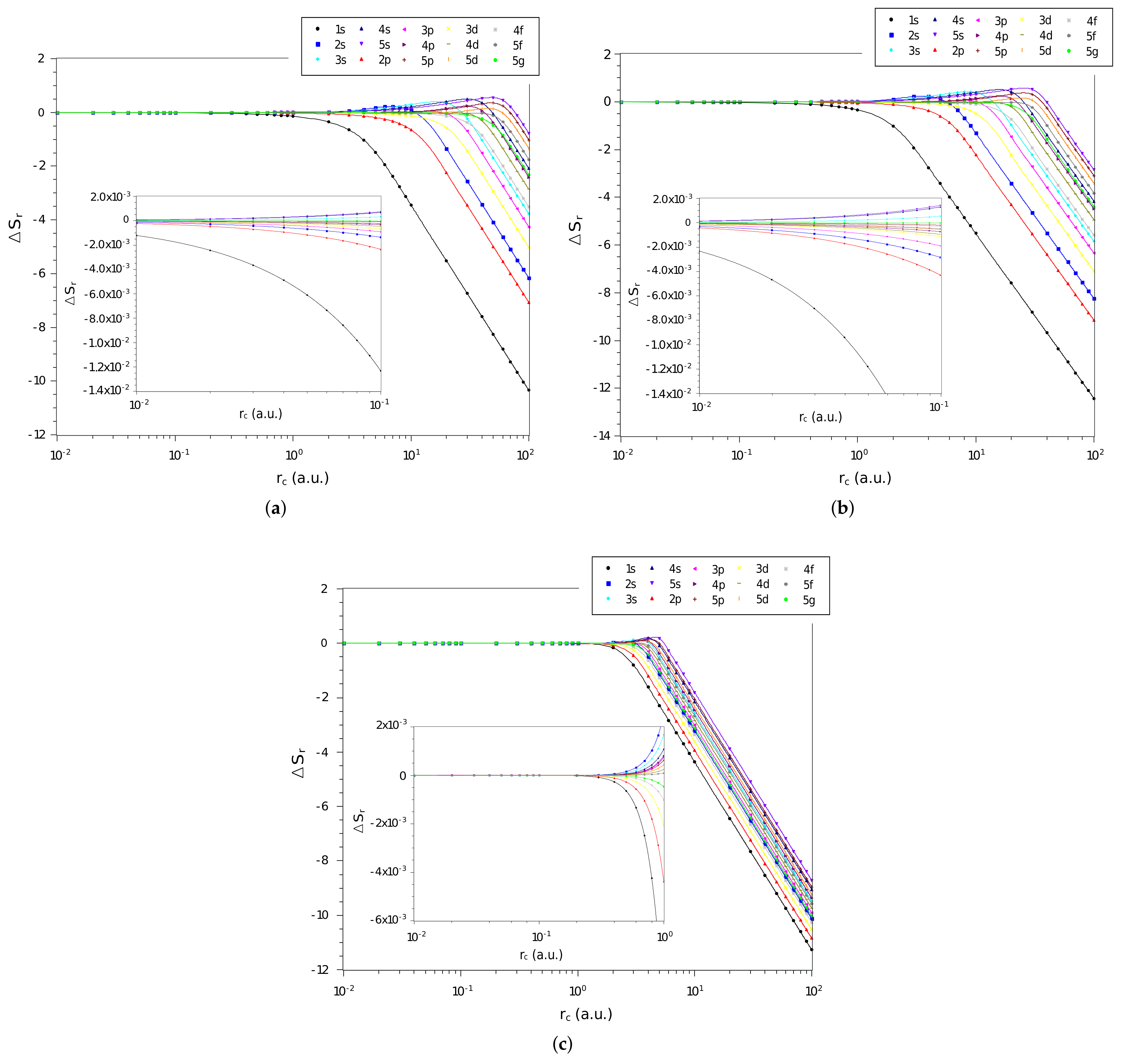
The difference, , between the values of position space entropy of each of the three systems and that of the particle confined in a cage, calculated by Equation (16), as function of confinement radius, , on a logarithmic scale, are shown in Figure 1a–c, for the confined hydrogen atom, the confined ion in a strongly coupled plasma, and the spherically confined harmonic oscillator, respectively. We plot all states with principal quantum numbers and . The first observation is that for the three systems the quantities tend to zero when the confinement radius decreases, indicating that in a rigorous confinement regime the position entropy of the systems tends to behave like that of a particle confined in a cage. The second observation is that, in general, the more excited the state, the faster moves towards zero. This is in agreement with our intuition that since the more excited states are less influenced by the interaction potential, they tend more quickly towards the behavior of a particle in a spherical cage.
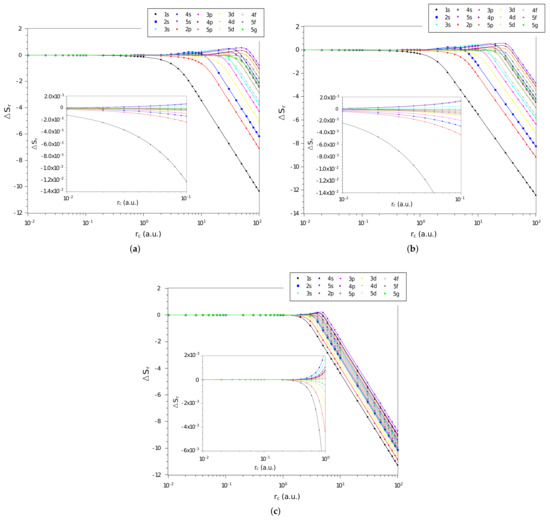
Figure 1.
Values of in different quantum states as a function of on a logarithmic scale for the (a) hydrogen confined, (b) ion-sphere, and (c) confined harmonic oscillator.
Particularly, it can be seen that the behavior of the entropy difference of the hydrogen confined and the ion-sphere are very similar. By zooming the graph in the region of small radii (Figure 1a,b), it is visible that the state is less influenced by confinement when compared to the others states. In turn, by zooming in on the graph (Figure 1c), it can be seen that the values of the position space entropy difference of the harmonic oscillator tend to zero for larger values of than the others systems.
It is also remarkable that some regions of have states with positive entropy differences. This is counterintuitive since the position Shannon entropy indicates the delocalization of density. Considering that one of the systems has an attractive potential, our intuition is that the density of this system would always be less spread out than that of the particle in a cage. The states that present are , , , , , , , and , for the confined hydrogen and the ion-sphere; and, in the case of the harmonic oscillator, the states and are added.
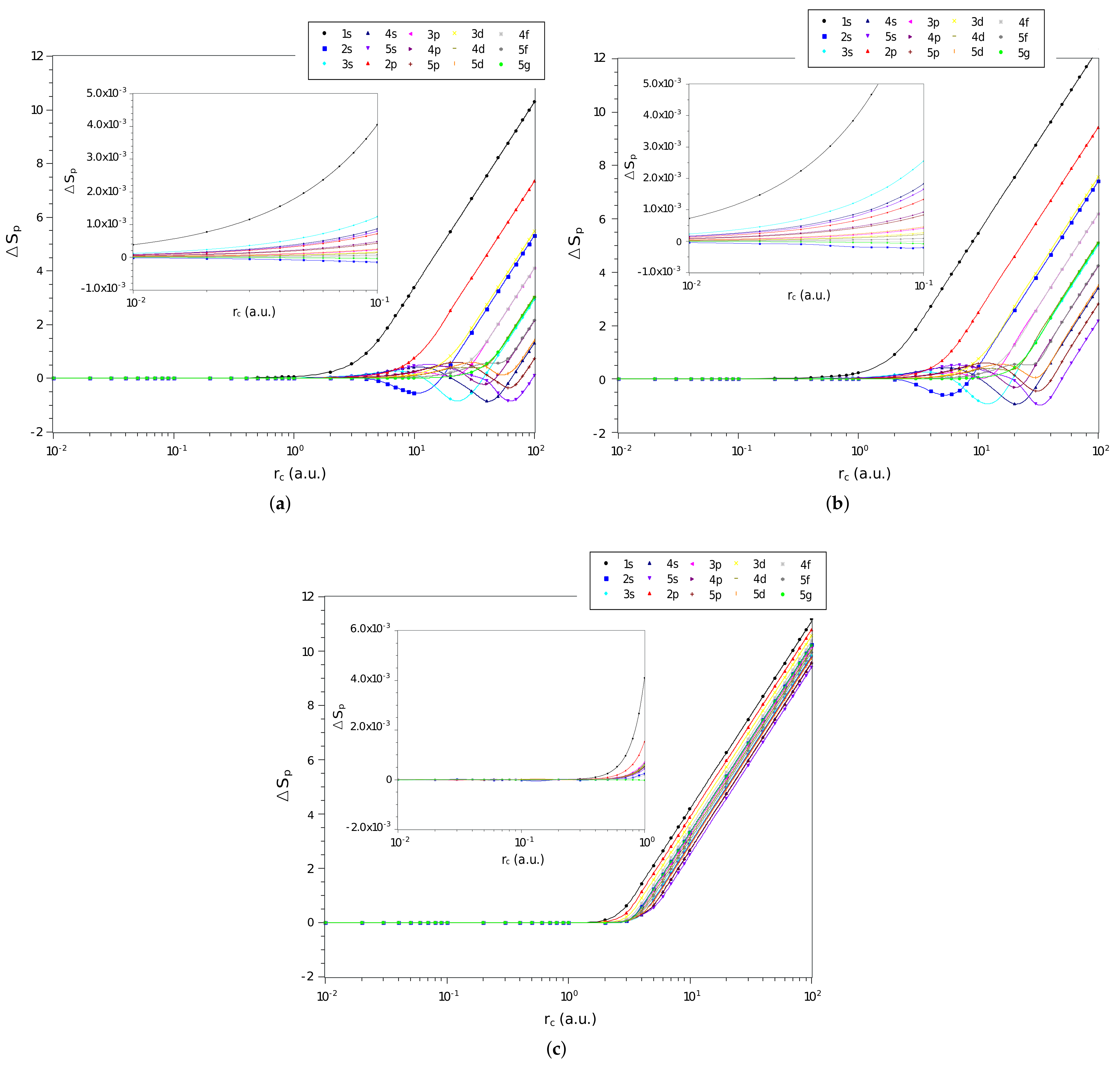
In Figure 2a–c are shown the difference, , between the values of momentum space entropy of each of the three systems and that of the particle confined in a spherical cage, calculated from Equation (17), as function of on a logarithmic scale, for the confined hydrogen atom, the confined ion in a strongly coupled plasma and the spherically confined harmonic oscillator, respectively. We have displayed in Figure all states with principal quantum numbers and . We observe similar behavior to that of position space entropy when the confinement radius tends to zero, decreasing its amplitude gradually to zero. This confirms that the interaction potential is no longer important for rigorous confinements and the system starts to behave like an electron confined in a spherical cage.
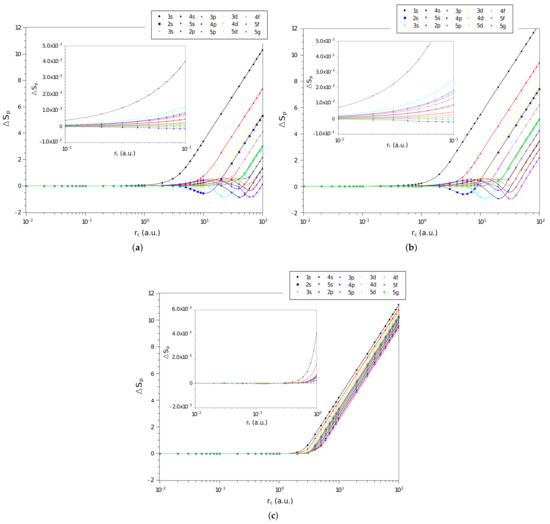
Figure 2.
Values of in different quantum states as a function of on a logarithmic scale for the (a) hydrogen confined, (b) ion-sphere, and (c) confined harmonic oscillator.
In the case of moment space entropy, the comparison between states and systems is quite similar to the one made for position space entropy. The sign change behavior of the entropy difference in some regions of is also noticed for the hydrogen confined and the ion-sphere systems. In particular, the entropy difference of some states seems to oscillate more prominently when compared to the position space ones. The states that present are , , , , , and and , for the confined hydrogen and the ion-sphere, respectively; and, in the case of the harmonic oscillator, there are no regions with sign change.
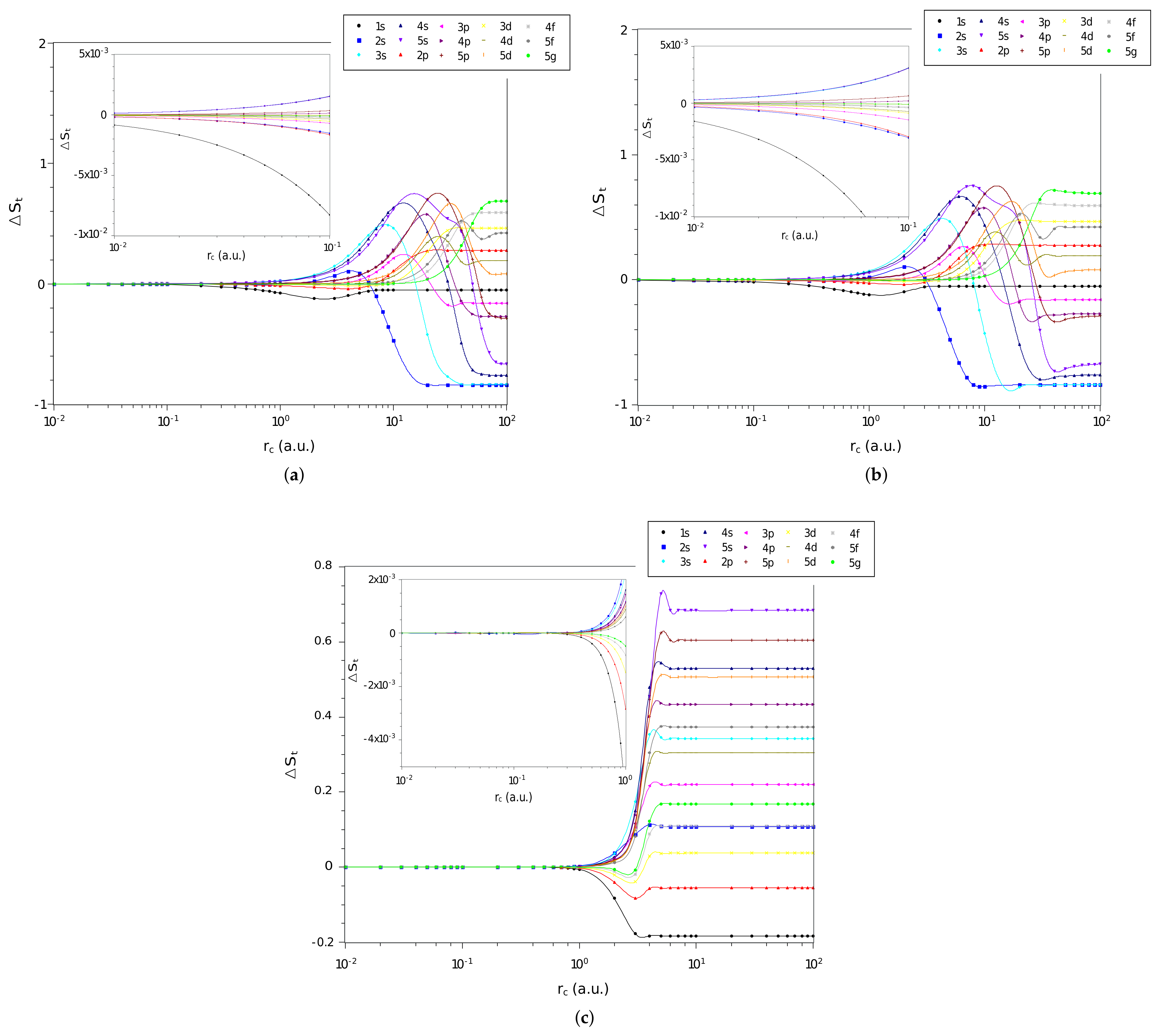
Now, we analyze the difference, , between the values of entropy sum of each of the three systems and that of the particle confined in a cage, calculated by Equation (23), as function of confinement radius, . Specifically, for states with and are shown in Figure 3a–c for the confined hydrogen atom, the confined ion in a strongly coupled plasma and the spherically confined harmonic oscillator, respectively. Again we observe that all , for the three systems, tend to zero when the confinement radius decreases. This indicates that in a rigorous confinement regime the entropy sum tends to behave like that of a particle confined in a spherical cage.
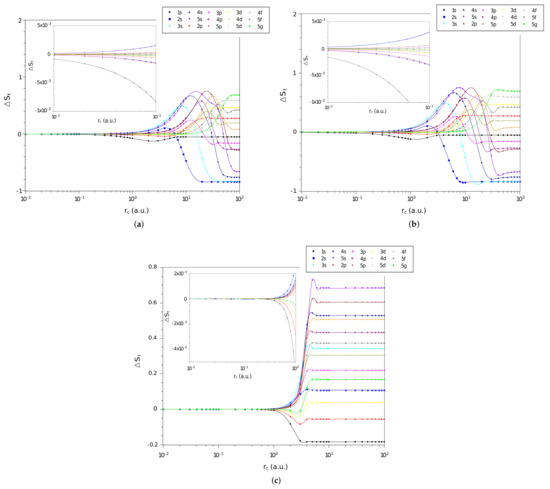
Figure 3.
Values of in different quantum states as a function of on a logarithmic scale for the (a) hydrogen confined, (b) ion-sphere, and (c) confined harmonic oscillator.
However, a different behavior of can be noticed when compared with and for intermediate and large values of . In the entropy sum case, the values of converge asymptotically to constant values. For the and quantities, the values increase (positively or negatively) due to the behavior of and for an electron confined in a spherical cage (see discussion at the end of this section).
Note that a particular state of a given system with () has a larger (smaller) entropy sum than the same state for a particle confined in a cage for a given radius of confinement. That is, several states of the three systems under study have a lower entropy sum when free () than when subjected to strong confinement. For intermediate values of we can observe oscillations in , confirming that confinement affects the position and momentum spaces differently.
The strong or rigorous confinement regime can be defined when the influence of the confining potential becomes greater than the free atomic system potential to specific configurations (or values). Based on the entropy sum, , of the ground state Nascimento et al. [36] defined three regions for confined atomic systems. The intermediate region is formed by values that limit the half well depth in the ground state versus curve. The half well depth is given by
where values are calculated in tending to infinity and in ( value where assumes a minimal value). The strong and weak confinement correspond to the regions where are dominant the confinement () or Coulombian () potentials, respectively. In Table 1, we determine the three regions from each of the three examined systems.

Table 1.
Confinement regions for the hydrogen confined (HC), ion-sphere (IS), and confined harmonic oscillator (HO).
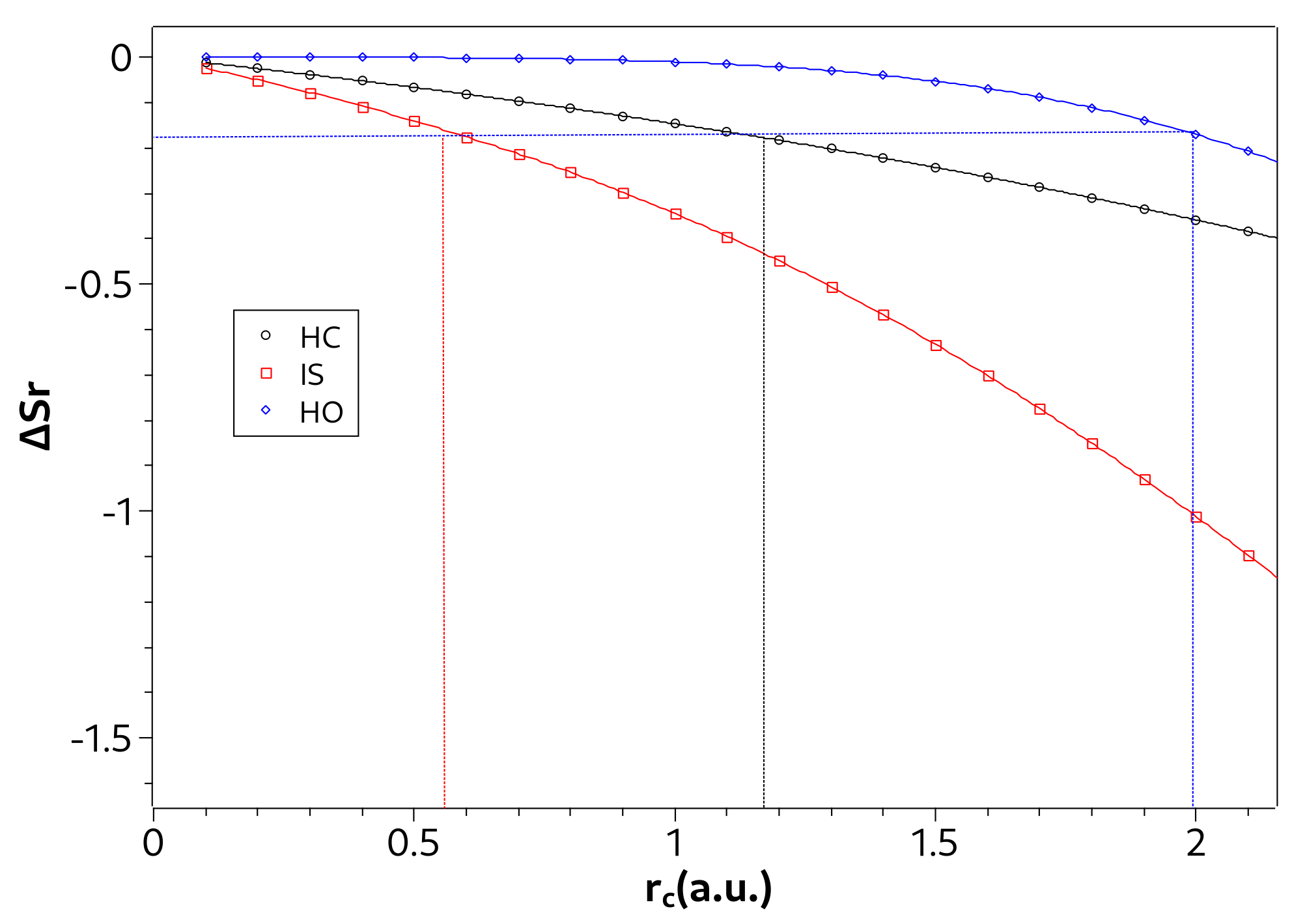
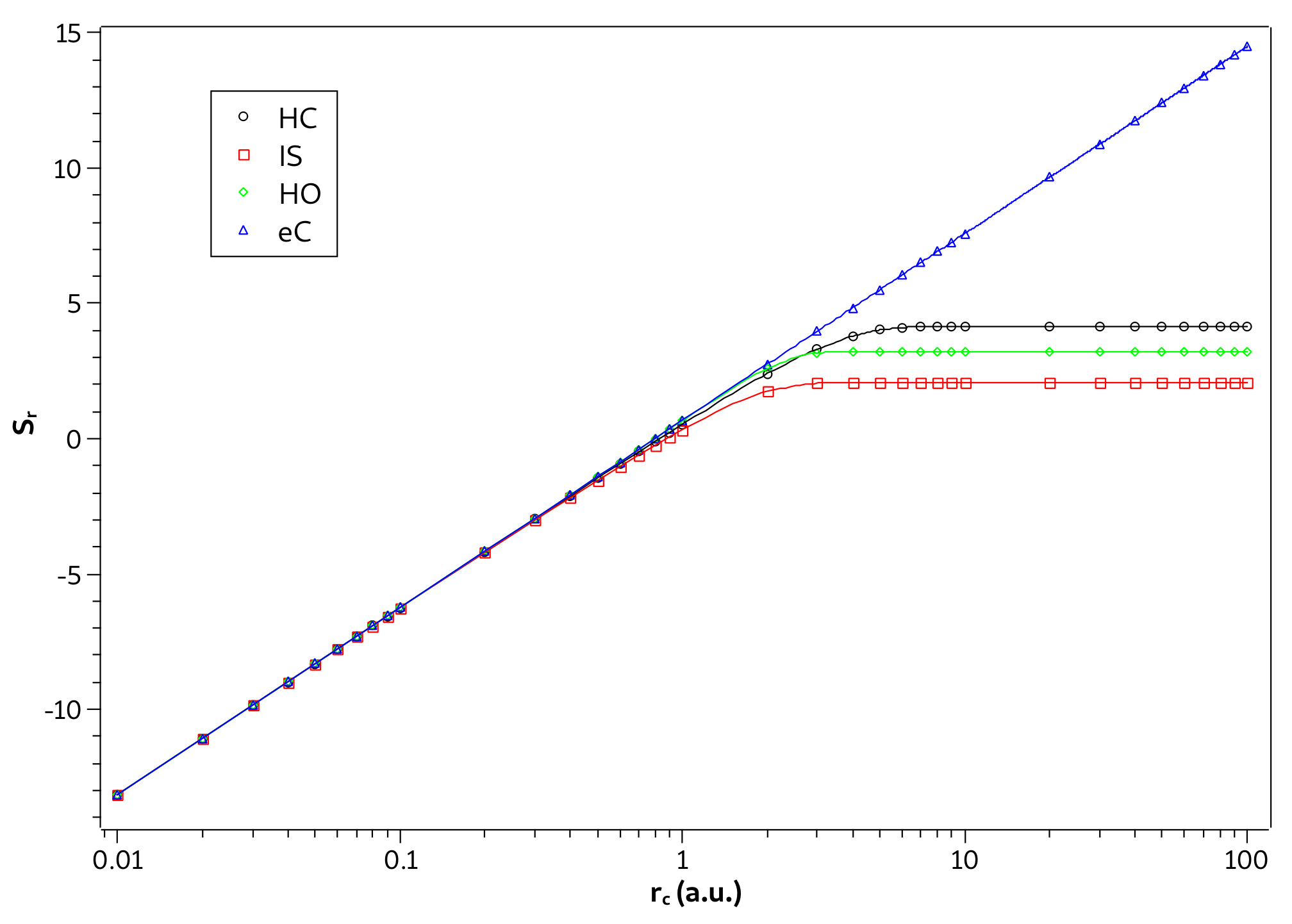
Note in Table 1 that the strong confinement region is smaller in the ion-sphere model than in the confined hydrogen atom. This indicates that, with respect to the confinement radius, the interaction potential of a hydrogen ion in a strongly coupled plasma surrounding is less influenced than the Coulomb potential of hydrogen atom. On the other hand, in the case of the harmonic oscillator, we verified that it was not possible to establish the three confinement regions by Equation (23), since there is no well in the ground state curve of versus . Indeed, as seen in Figure 3c, the influence of the confinement potential is greater even for larger confinement radii. To overcome this difficulty, we propose an additional criterion to deal with the harmonic oscillator case. To propose it, we consider Figure 4 where we show the values of in the ground state as a function of for the three systems. In such a figure we notice that in the confinement radius that defines the beginning of the strong confinement region for the hydrogenoid models. Thus, we propose the value of when to define the strong confinement region of the harmonic oscillator, written in Table 1. Establishing a criterion using can be particularly advantageous in situations where it is difficult to calculate .
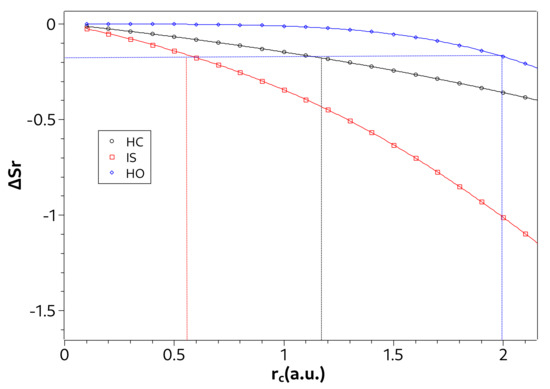
Figure 4.
Values of in ground state as a function of for the hydrogen confined (HC), ion-sphere (IS), and confined harmonic oscillator (HO). The vertical lines delimit the region of strong confinement for each system.
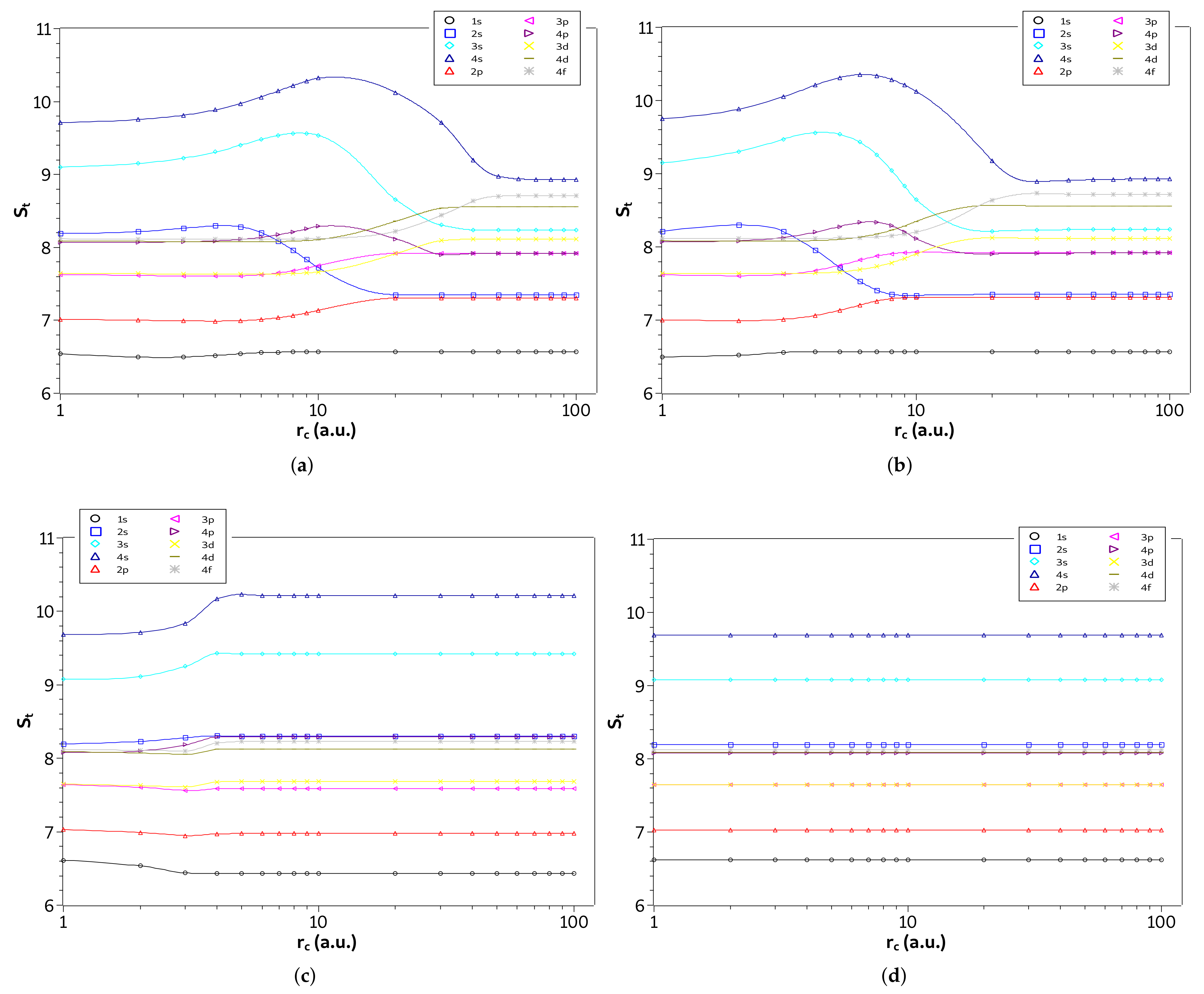
Furthermore, in Figure 5a–d are shown the entropy sum, , of the four systems as function of on a logarithmic scale in the weak and intermediate regions (). We plot all states with principal quantum numbers and . Thus, it becomes clear from the figures for approximately which confinement radius, , a given state of a specific system is in its lowest entropy sum configuration. Specifically, the state is always the lowest entropy sum for all systems studied. On the other hand, some states have the lowest entropy sum for intermediate values of , while others have a lower entropy for the free configuration or in the strong confinement region. Moreover, we see that for the confined hydrogen atom and the ion confined in a strongly coupled plasma the states with angular momentum quantum number s (except the state) are the most affected by confinement, while for the harmonic oscillator the and states, although affected, decrease their entropy sum.
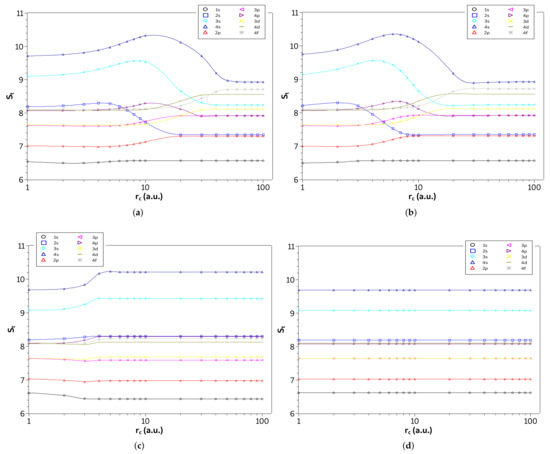
Figure 5.
Values of in different quantum states as a function of on a logarithmic scale in the weak and intermediate regions for the (a) hydrogen confined, (b) ion-sphere, (c) confined harmonic oscillator, and (d) particle confined in a cage.
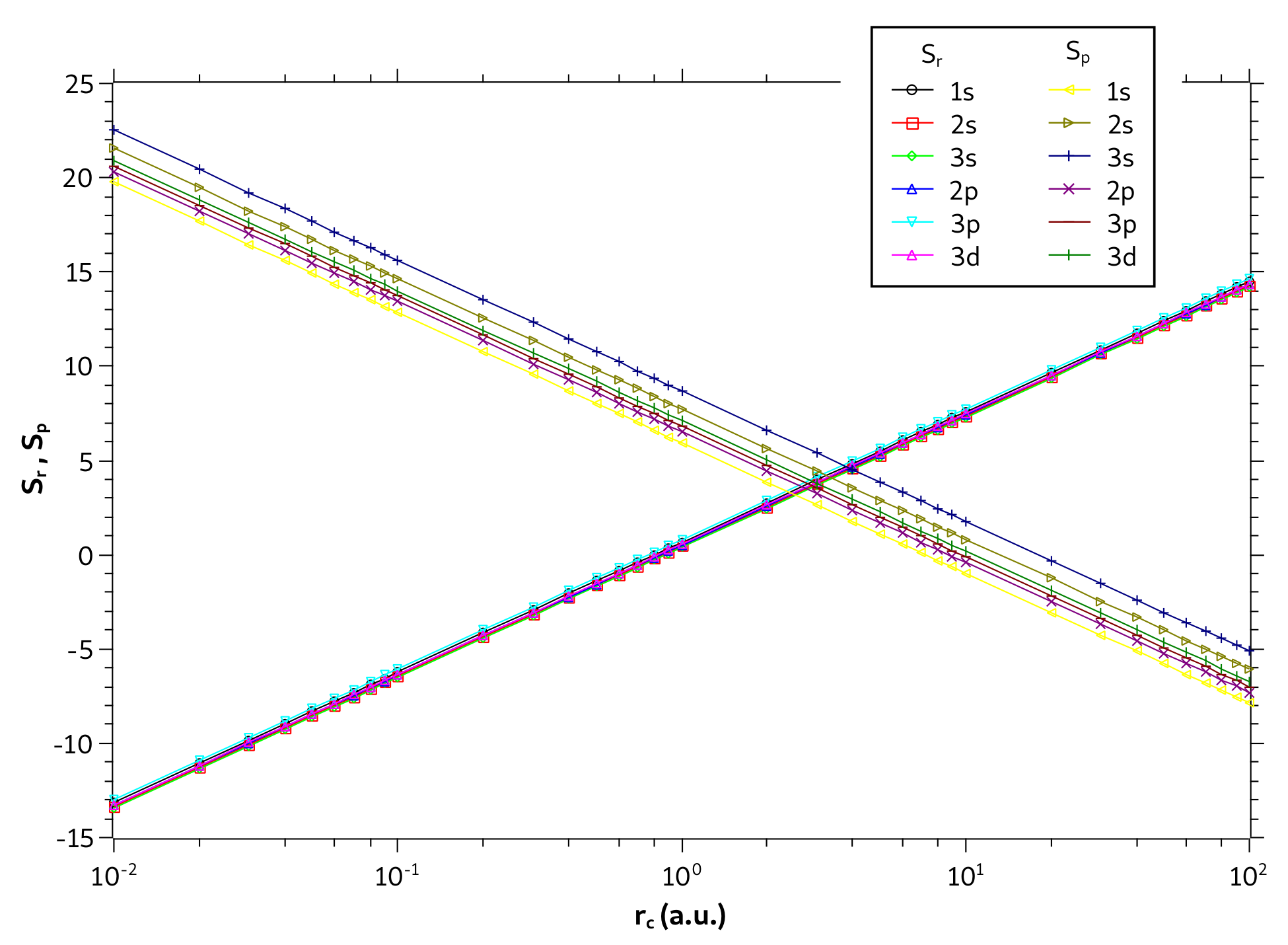
We now investigate only the particle confined in a spherical cage. In Figure 6, are displayed the entropies and as a function as a function of on a logarithmic scale. We plot all states with principal quantum numbers and . Note that the values of are quite close for different quantum states. The same behavior was also observed by Nascimento et al. in [53] for the problem of one-dimensional infinite potential well. They have qualitatively explained such behavior by analyzing the probability densities in the position and momentum spaces. According to their explanation, the probability densities in the position are spread by same range of r values leading to equal values of for all levels while in the momentum space the probability densities are spread for increasing ranges of p values leading to increase in with the level increment.
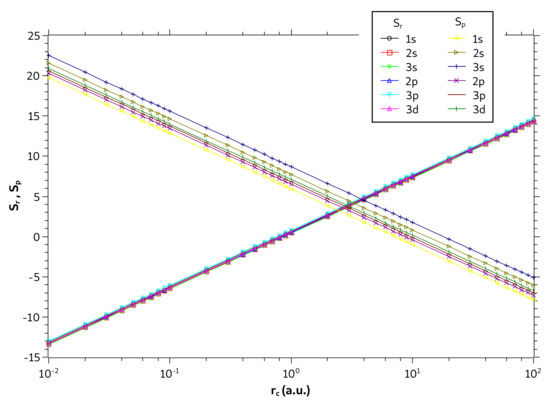
Figure 6.
Values of and in different quantum states as a function of on a logarithmic scale for the particle confined in a cage.
Moreover, Figure 6 also shows that entropies are linear on the monolog axes. In fact, the coefficient of determination gives , demonstrating that the linear model fits perfectly to the sample of points. For the state, for example, the linear regression fit of dataset gives and ; and for state gives and . As expected, the slopes of the straight lines and have opposite values so that in the sum the term with the will be canceled, showing that the entropy sum is constant, as it can be seen in Figure 5d. Note that the entropy curves for states and form two parallel lines (same slope) and that the lines are much closer to each other (nearby y-intercepts) than the lines.
Since it has been shown, in the strong confinement regime, that the three systems previously studied begin to behave like a particle confined in a spherical cage, then this also means that in this regime the entropies of these systems start to have a linear behavior as a function of . Such behavior is evidenced by Figure 7, where we plot the graph of the values of entropy in the ground state as a function of on the logarithmic scale for the four systems in question. We can see in Figure 7 that the changes in behaviors for the linear trend of the confined particle happen almost abruptly near the regions of strong confinement defined in Table 1.
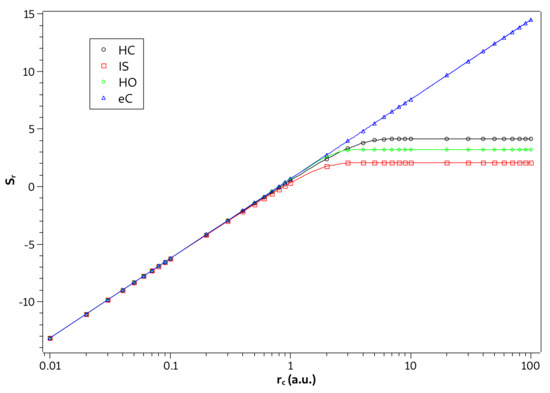
Figure 7.
Values of in ground state as a function of on logarithmic scale for the hydrogen confined (HC), ion-sphere (IS), confined harmonic oscillator (HO), and particle confined (eC).
4. Conclusions
In this work, we presented a comparative study between some confined systems using Shannon’s informational entropies as tools to analyze how the effects of confinement impact in these systems. For that, we implemented a computational code in Fortran based on the finite element method to solve the radial part of the Schorödinger equation. In particular, we determined the position and momentum space entropies and the entropy sum for different states of the following systems: a particle confined in a spherical cage, the spherically confined hydrogen atom, the hydrogenic ion confined in a strongly coupled plasma, and the spherically confined harmonic oscillator.
Our main observation was that the difference between values of entropies for all states of each of the three systems and that of the particle confined in a spherical cage tend to zero when the confinement radius decreases. This confirms that the interaction potential is no longer important for rigorous confinements and the system starts to behave just like an confined electron. Furthermore, we observed that the behavior of these entropies differences for the hydrogen confined and the ion-sphere are very similar, while for the harmonic oscillator they tend to zero faster. A counterintuitive behavior also noticed for the hydrogen confined and the ion-sphere systems was the sign change of the entropy difference on position and momentum spaces in some regions of confinement radius indicating that the density of these systems is more spread out than that of the particle in a cage.
We also determined in this work the three confinement regions of three of the examined systems using a procedure based on entropy sum proposed by Nascimento et al. [36]; and an additional criterion, using the entropy of space, was required to define the strong confinement region of the harmonic oscillator. Lastly, we only investigate the particle confined in a spherical cage. Besides the quite close values of entropy in the position space for different quantum states, we observed that entropies are linear on monolog axis. This means that in strong confinement regime the entropies of the other systems start to behave as a logarithmic function of the confinement radius.
As a final conclusion, with the employment of a very accurate numerical procedure, we have shown significant results on the behavior of strongly confined quantum systems and demonstrated the feasibility of using Shannon’s informational entropies as a tool to analyze such behavior. As a perspective, we intend to extend the method to calculate Shannon’s informational entropy for excited states of two-electron atoms and artificial atoms in the presence of external fields.
Author Contributions
All authors contributed equally to this work. All authors have read and agreed to the published version of the manuscript.
Funding
This research received no external funding.
Data Availability Statement
Data are available from the authors after a reasonable request.
Acknowledgments
We acknowledge the partial financial support provided by the Brazilian “Coordenação de Aperfeiçoamento de Pessoal de Nível Superior” (CAPES)—Finance Code 001. A.J.S. is grateful to “Conselho Nacional de Desenvolvimento Científico e Tecnológico” (CNPq) for grants. F.V.P. is also grateful for the provision of computational time in the Centro Nacional de Computação (CESUP), Universidade Federal do Rio Grande do Sul.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Connerade, J.P. Confining and compressing the atom. Eur. Phys. J. D 2020, 74, 211. [Google Scholar] [CrossRef]
- Sen, K.D. (Ed.) Electronic Structure of Quantum Confined Atoms and Molecules; Springer: Cham, Switzerland, 2014. [Google Scholar] [CrossRef]
- Sabin, J.R.; Brändas, E.; Cruz, S.A. (Eds.) Advances in Quantum Chemistry: Theory of Confined Quantum Systems vol 57 e 58; Academic Press: New York, NY, USA, 2009. [Google Scholar]
- García-Miranda, J.J.; Garza, J.; Ibarra, I.A.; Martínez, A.; Martínez-Sánchez, M.A.; Rivera-Almazo, M.; Vargas, R. Electronic Structure of Systems Confined by Several Spatial Restrictions. In Chemical Reactivity in Confined Systems; John Wiley Sons, Ltd.: Hoboken, NJ, USA, 2021; Chapter 4; p. 69. [Google Scholar] [CrossRef]
- LEY-KOO, E. Recent progress in confined atoms and molecules: Superintegrability and symmetry breakings. Rev. Mex. Fis. 2018, 64, 326. [Google Scholar] [CrossRef]
- El-Gammal, F.N. Confined atoms in plasma environment: Variational Monte Carlo calculations. Mol. Phys. 2021, 119, e1879302. [Google Scholar] [CrossRef]
- Maniero, A.M.; de Carvalho, C.R.; Prudente, F.V.; Jalbert, G. Oscillating properties of a two-electron quantum dot in the presence of a magnetic field. J. Phys. B At. Mol. Opt. Phys. 2020, 53, 185001. [Google Scholar] [CrossRef]
- Saha, S.; Jose, J. Shannon entropy as a predictor of avoided crossing in confined atoms. Int. J. Quantum Chem. 2020, 120, e26374. [Google Scholar] [CrossRef]
- Cruz, E.; Aquino, N.; Prasad, V. Localization–delocalization of a particle in a quantum corral in presence of a constant magnetic field. Eur. Phys. J. D 2021, 75, 106. [Google Scholar] [CrossRef]
- Deshmukh, P.C.; Jose, J.; Varma, H.R.; Manson, S.T. Electronic structure and dynamics of confined atoms. Eur. Phys. J. D 2021, 75, 166. [Google Scholar] [CrossRef]
- Prudente, F.V.; Guimarães, M.N. Confined Quantum Systems Using the Finite Element and Discrete Variable Representation Methods. In Electronic Structure of Quantum Confined Atoms and Molecules; Sen, K.D., Ed.; Springer: Cham, Switzerland, 2014; Chapter 5; pp. 101–143. [Google Scholar]
- Zicovich-Wilson, C.; Planelles, J.H.; Jaskólski, W. Spatially Confined Simple Quantum Mechanical Systems. Int. J. Quantum Chem. 1994, 50, 429. [Google Scholar] [CrossRef]
- Costa, L.S.; Prudente, F.V.; Acioli, P.H.; Neto, J.J.S.; Vianna, J.D.M. A study of confined quantum systems using the Woods-Saxon potential. J. Phys. B At. Mol. Opt. Phys. 1999, 32, 2461. [Google Scholar] [CrossRef]
- Connerade, J.P.; Dolmatov, V.K.; Lakshmi, P.A.; Manson, S.T. Electron structure of endohedrally confined atoms: Atomic hydrogen in an attractive shell. J. Phys. B At. Mol. Opt. Phys. 1999, 32, L239. [Google Scholar] [CrossRef]
- Baltenkov, A.S. Resonances in photoionization cross sections of inner subshells of atoms inside the fullerene cage. J. Phys. B At. Mol. Opt. Phys. 1999, 32, 2745. [Google Scholar] [CrossRef]
- Nascimento, E.M.; Prudente, F.V.; Guimarães, M.N.; Maniero, A.M. A study of the electron structure of endohedrally confined atoms using a model potential. J. Phys. B At. Mol. Opt. Phys. 2011, 44, 015003. [Google Scholar] [CrossRef]
- Salazar, S.J.C.; Laguna, H.G.; Prasad, V.; Sagar, R.P. Shannon-information entropy sum in the confined hydrogenic atom. Int. J. Quantum Chem. 2020, 120, e26188. [Google Scholar] [CrossRef]
- De Morais, G.d.S.T.; Custodio, R. Assessment of a numeric variational method for the solution of confined multielectron atoms. J. Mol. Model. 2021, 27, 212. [Google Scholar] [CrossRef]
- Rodriguez-Bautista, M.; Vargas, R.; Aquino, N.; Garza, J. Electron-density delocalization in many-electron atoms confined by penetrable walls: A Hartree–Fock study. Int. J. Quantum Chem. 2018, 118, e25571. [Google Scholar] [CrossRef]
- Pasteka, L.F.; Helgaker, T.; Saue, T.; Sundholm, D.; Werner, H.J.; Hasanbulli, M.; Major, J.; Schwerdtfeger, P. Atoms and molecules in soft confinement potentials. Mol. Phys. 2020, 118, e1730989. [Google Scholar] [CrossRef]
- Barbosa, T.N.; Almeida, M.M.; Prudente, F.V. A quantum monte carlo study of confined quantum systems: Application to harmonic oscillator and hydrogenic-like atoms. J. Phys. B At. Mol. Opt. Phys. 2015, 48, 055002. [Google Scholar] [CrossRef]
- Prudente, F.V.; Costa, L.S.; Viana, J.D.M. A study of two-electron quantum dot spectrum using discrete variable representation method. J. Chem. Phys. 2005, 123, 224701. [Google Scholar] [CrossRef]
- Bielinska-Waz, D.; Karwowski, J.; Diercksen, G.H.F. Spectra of confined two-electron atoms. J. Phys. B At. Mol. Opt. Phys. 2001, 34, 1987. [Google Scholar] [CrossRef]
- Gueorguiev, V.G.; Rau, A.R.P.; Draayer, J.P. Confined one-dimensional harmonic oscillator as a two-mode system. Am. J. Phys. 2006, 74, 394. [Google Scholar] [CrossRef]
- Montgomery, H.; Aquino, N.; Flores-Riveros, A. The ground state energy of a helium atom under strong confinement. Phys. Lett. A 2010, 374, 2044. [Google Scholar] [CrossRef]
- Aquino, N.; Flores-Riveros, A.; Rivas-Silva, J. The compressed helium atom variationally treated via a correlated Hylleraas wave function. Phys. Lett. A 2003, 307, 326. [Google Scholar] [CrossRef]
- Flores-Riveros, A.; Aquino, N.; Montgomery, H. Spherically compressed helium atom described by perturbative and variational methods. Phys. Lett. A 2010, 374, 1246. [Google Scholar] [CrossRef]
- Sen, K.D. (Ed.) Statistical Complexity: Applications in Electronic Struture; Springer: Dordrecht, The Netherlands, 2011. [Google Scholar] [CrossRef]
- Nascimento, W.S.; Prudente, F.V. Shannon entropy: A study of confined hydrogenic-like atoms. Chem. Phys. Lett. 2018, 691, 401. [Google Scholar] [CrossRef]
- Estañón, C.R.; Aquino, N.; Puertas-Centeno, D.; Dehesa, J.S. Crámer-Rao complexity of the confined two-dimensional hydrogen. Int. J. Quantum Chem. 2021, 121, e26424. [Google Scholar] [CrossRef]
- Mukherjee, N.; Roy, A.K. Information-entropic measures for non-zero l states of confined hydrogen-like ions. Eur. Phys. J. D 2018, 72, 118. [Google Scholar] [CrossRef]
- Majumdar, S.; Mukherjee, N.; Roy, A.K. Various complexity measures in confined hydrogen atom. Chem. Phys. Lett. 2017, 687, 322. [Google Scholar] [CrossRef]
- Jiao, L.; Zan, L.; Zhang, Y.; Ho, Y. Benchmark values of Shannon entropy for spherically confined hydrogen atom. Int. J. Quantum Chem. 2017, 117, e25375. [Google Scholar] [CrossRef]
- Martínez-Flores, C. Shannon entropy and Fisher information for endohedral confined one- and two-electron atoms. Phys. Lett. A 2021, 386, 126988. [Google Scholar] [CrossRef]
- Martínez-Sánchez, M.A.; Vargas, R.; Garza, J. Shannon Entropy for the Hydrogen Atom Confined by Four Different Potentials. Quantum Rep. 2019, 1, 208. [Google Scholar] [CrossRef]
- Nascimento, W.S.; de Almeida, M.M.; Prudente, F.V. Coulomb Correlation and Information Entropies in Confined Helium-Like Atoms. Eur. Phys. J. D 2021, 75, 171. [Google Scholar] [CrossRef]
- Majumdar, S.; Roy, A.K. Shannon Entropy in Confined He-Like Ions within a Density Functional Formalism. Quantum Rep. 2020, 2, 189. [Google Scholar] [CrossRef]
- Lee, M.J.; Jung, Y.D. Characteristics of Shannon’s Information Entropy of Atomic States in Strongly Coupled Plasma. Entropy 2020, 22, 881. [Google Scholar] [CrossRef]
- Zan, L.R.; Jiao, L.G.; Ma, J.; Ho, Y.K. Information-theoretic measures of hydrogen-like ions in weakly coupled Debye plasmas. Phys. Plasmas 2017, 24, 122101. [Google Scholar] [CrossRef]
- Carrillo, R.S.; Gil-Barrera, C.A.; Sun, G.H.; Solaimani, M.; Dong, S.H. Shannon entropies of asymmetric multiple quantum well systems with a constant total length. Eur. Phys. J. Plus 2021, 136, 1060. [Google Scholar] [CrossRef]
- Carrillo, R.S.; Dong, Q.; Sun, G.H.; Silva-Ortigoza, R.; Dong, S.H. Shannon entropy of asymmetric rectangular multiple well with unequal width barrier. Results Phys. 2022, 33, 105109. [Google Scholar] [CrossRef]
- Song, X.D.; Sun, G.H.; Dong, S.H. Shannon information entropy for an infinite circular well. Phys. Lett. A 2015, 379, 1402. [Google Scholar] [CrossRef]
- Gadre, S.R.; Sears, S.B.; Chakravorty, S.J.; Bendale, R.D. Some novel characteristics of atomic information entropies. Phys. Rev. A 1985, 32, 2602. [Google Scholar] [CrossRef] [PubMed]
- Site, L.D. Shannon entropy and many-electron correlations: Theoretical concepts, numerical results, and Collins conjecture. Int. J. Quantum Chem. 2014, 115, 1396. [Google Scholar] [CrossRef]
- Saha, S.; Jose, J. Shannon entropy as an indicator of correlation and relativistic effects in confined atoms. Phys. Rev. A 2020, 102, 052824. [Google Scholar] [CrossRef]
- Sabirov, D.S.; Osawa, E. Information Entropy of Fullerenes. J. Chem. Inf. Model. 2015, 55, 1576. [Google Scholar] [CrossRef]
- Sabirov, D.S. Information entropy of mixing molecules and its application to molecular ensembles and chemical reactions. Comput. Theor. Chem. 2020, 1187, 112933. [Google Scholar] [CrossRef]
- Sabirov, D.S. Information entropy changes in chemical reactions. Comput. Theor. Chem. 2018, 1123, 169. [Google Scholar] [CrossRef]
- Park, K.; Kim, J.; Moon, S.; An, K. Maximal Shannon entropy in the vicinity of an exceptional point in an open microcavity. Sci. Rep. 2020, 10, 12551. [Google Scholar] [CrossRef] [PubMed]
- Nascimento, W.S.; Prudente, F.V. Sobre um estudo da entropia de Shannon no contexto da mecânica quântica: Uma aplicação ao oscilador harmônico livre e confinado. Quim. Nova 2016, 39, 757. [Google Scholar] [CrossRef]
- Guimarães, M.N.; Prudente, F.V. A study of the confined hydrogen atom using the finite element method. J. Phys. B At. Mol. Phys. 2005, 38, 2811. [Google Scholar] [CrossRef]
- Prudente, F.V.; Soares Neto, J.J. Optimized mesh for the finite-element method using a quantum-mechanical procedure. Chem. Phys. Lett. 1999, 302, 43. [Google Scholar] [CrossRef]
- Nascimento, W.S.; de Almeida, M.M.; Prudente, F.V. Information and quantum theories: An analysis in one-dimensional systems. Eur. J. Phys. 2020, 41, 025405. [Google Scholar] [CrossRef]
- Goldman, S.; Joslin, C. Spectroscopic properties of an isotropically compressed hydrogen atom. J. Phys. Chem. 1992, 96, 6021. [Google Scholar] [CrossRef]
- Yáñez, R.J.; Van Assche, W.; González-Férez, R.; Dehesa, J.S. Entropic integrals of hyperspherical harmonics and spatial entropy of D-dimensional central potentials. J. Math. Phys. 1999, 40, 5675. [Google Scholar] [CrossRef]
- Bialynicki-Birula, I.; Mycielski, J. Uncertainty relations for information entropy in wave mechanics. Commun. Math. Phys. 1975, 44, 129. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).