Abstract
The inverse association between the groups of odd-chain (OCFA) and branched-chain (BCFA) and the development of diseases in humans have generated interest in the scientific community. In experiment 1, the extent of the passage of odd- and branched-chain fatty acids (OBCFA) from milk fat to fresh cheese fat was studied in sheep and goats. Milk collected in two milk processing plants in west Sardinia (Italy) was sampled every 2 weeks during spring (March, April and May). In addition, a survey was carried out to evaluate the seasonal variation of the OBCFA concentrations in sheep and goats’ cheeses during all lactation period from January to June. Furthermore, to assess the main differences among the sheep and goat cheese, principal component analysis (PCA) was applied to cheese fatty acids (FA) profile. Concentrations of OBCFA in fresh cheese fat of both species were strongly related to the FA content in the unprocessed raw milk. The average contents of OBCFA were 4.12 and 4.13 mg/100 mg of FA in sheep milk and cheese, respectively, and 3.12 and 3.17 mg/100 mg of FA in goat milk and cheese, respectively. The OBCFA concentration did no differed between milk and cheese in any species. The content of OBCFA was significantly higher in sheep than goats’ dairy products. The OBCFA composition of the cheese was markedly affected by the period of sampling in both species: odd and branched FA concentrations increased from March to June. The seasonal changes of OBCFA in dairy products were likely connected to variations in the quality of the diet. The PCA confirmed the higher nutritional quality of sheep cheese for beneficial FA, including OBCFA compared to the goat one, and the importance of the period of sampling in the definition of the fatty acids profile.
1. Introduction
The fatty acid (FA) composition of dairy products has assumed considerable interest in consumers from a nutritional and healthy point of view. Indeed, specific FA of dairy products can affect human health and can have an important role in the prevention of metabolic diseases. This increasing attention is also demonstrated by recent studies that investigated the feasibility of improving milk FA profile in sheep and goats though breeding schemes [1,2,3]. A significant amount of attention has been focused in the last decade on polyunsaturated fatty acid of the omega3 family (PUFA n-3) and conjugated linoleic acid (CLA) concentrations of dairy products. Moreover, the groups of odd-chain fatty acids (OCFA) and branched-chain fatty acids (BCFA), long neglected due to their low incidence on the total amount of FA, have sparked interest in the scientific community due to an inverse relationship with the development of human diseases.
In particular, the group of BCFA comprises mainly saturated fatty acids characterized by the presence of one or more methyl groups in the iso or anteiso position. Such molecules represent the main FAs in some microorganisms (e.g., Bacilli and Lactobacilli), and they can be also observed in mammal tissues. In laboratory animals these FA evidenced anti-inflammatory properties [4], reduced the incidence of necrotizing enterocolitis and altered the ecology of the gastrointestinal microorganisms in a neonatal rat model [5].
The OCFA, which include pentadecanoic (C15:0) and heptadecanoic (C17:0) acids and their isomeric forms, are mostly derived from ruminal bacteria cell wall and then partitioned firstly to organs and tissues and, therefore, detectable in ruminant-derived foods. Thus, dairy foods or ruminant fats represent the major dietary source of OCFA for humans.
Moreover, the milk concentrations of C15:0 and C17:0 are considered biomarkers of rumen microbial fermentation and microbial de novo lipogenesis [6]. In addition, the mammary gland plays a role in their synthesis by using propionate [7], and the subcutaneous adipose tissue and, therefore, after their mobilization, can be incorporated into milk fat [8].
Humans are not able to synthetize C15:0 and C17:0 so they could be considered valuable markers of dairy fat intake [9,10]. The role of these FA in human health has recently been reinforced in view of new biological and nutritional observations. The C15:0 and C17:0 have been inversely associated with cardiovascular disease [11,12,13,14] and incidence of type 2 diabetes [15,16,17,18]. The direct role of C15:0 in attenuating pro-inflammatory state, cytotoxicity in human cell lines, anemia, and dyslipidemia lowering glucose and cholesterol in vivo has been recently evidenced [19].
Among the different factors influencing the OBCFA concentration in milk and dairy products, animal diet is the main one. Indeed, the diet of animals can modulate microbial growth in the rumen, de novo microbial FA synthesis and the uptake of blood circulating FA by the mammary gland. Variations in milk OBCFA have been observed in response to lipid supplementation [20,21,22], change in forage to concentrate ratio [23] and use of by-products rich in bioactive compounds [24]. Changes in the OBCFA concentration in milk has been also observed with lactation stage [25] and with energy balance of animals [26], probably as a consequence of the fat mobilization from adipose tissue and the extent of mammary uptake for milk fat synthesis [27].
In Mediterranean areas, almost all sheep milk is processed into cheese, and milk fatty acid profile has important effects on cheese fat quality under nutritional point of view. The total transfer from milk into cheese of fatty acids of nutritional interest, as PUFA n-3 and CLA, has been reported in sheep [28], but effects of cheese-making technology has been also evidenced [29]. However, the mechanisms influencing the relationship between OBCFA concentrations in unprocessed milk and the derived dairy products are still unclear. The presence of FA in the FA composition of different microorganism can be of significant importance for the dairy industry where different microbial cultures, as starters, are widely used for making different products. The consequence could be the change of the native OCFA and BCFA milk concentrations after processing in cheese or other dairy products.
The aims of this study were (a) to evaluate the extent of OBCFA transfer from ovine and caprine milk to cheese (Experiment 1); (b) to describe the seasonal variation of OBCFA in sheep and goat cheeses (Experiment 2). Both were sequentially investigated with two independent surveys.
2. Materials and Methods
In Experiment 1, bulk tank milk of Sarda breed sheep and Sardinian goats and cheeses were collected from processing plants in North Sardinia for sheep (n. 2) and in west Sardinia for goats (n. 2). Samples were taken every two weeks during spring (March, April and May) for a total of 12 milk samples per species. The transfer of OBCFA from milk to fresh cheeses after 24 h from processing was evaluated.
In Experiment 2, mild sheep and goat cheeses, with a ripening time of about 20–30 days, were sampled monthly from January to July from nine sheep cheese-making plants located in different areas of Sardinia (Guspini, Nurri, Marrubiu, Macomer, Dorgali, Oliena, Onifai, Thiesi, Berchidda), five plants of which produced also goat cheese. The samples collection started in January, which corresponds to the begin of lactation both in sheep and goats reared in Sardinia’s breeding system. The goats cheese samples were from four lots/month from January to April and three different lots/month from May to July for a total of 25 goat cheese samples. The sheep cheese samples were Pecorino Sardo PDO type from 8 lots/month from January to May, and from four different lots/month in June and July for a total of 48 sheep cheese samples.
The determination of FA concentrations in milk and cheese samples was carried out by a gas chromatographic method, as previously described [20]. Briefly, after the lipid extraction, a base-catalyzed trans-esterification FIL-IDF standard procedure [30] was used to prepare FA methyl esters (FAME). Identification of individual FAME was allowed by comparing their retention time with that of a series of analytical standards. Those included the Supelco 37 component FAME MIX (Supelco, Bellefonte, PA, USA), the GLC-110 MIX (Matreya Inc. Pleasant Gap, PA, USA) and some individual BCFA (Matreya Inc. Pleasant Gap, PA, USA). Identification of OBCFA was also supported by the consultation of previous studies [31,32]. The FA concentration was reported as mg/100 mg of total FAME.
Using R software (R Core Team, 2020), general linear model (GLM) was applied to data on OBCFA and others main nutritional FA (R Core Team, 2020). The considered fixed effects were species (sheep and goat) and product type (milk and cheese) for experiment 1, whereas species and month for Experiment 2. Significative differences were declared at p < 0.05.
Principal component analysis (PCA) was applied on sixty-three FA profile of sheep and goats’ cheeses using prcomp R function. A total of 42 FA was considered and, therefore, 42 principal components were extracted. Before PCA, all considered FA were scaled to ensure unit variance.
3. Results and Discussion
Means (±SE) of fat concentration in milk and cheese of sheep and goat, respectively were 6.16% (±0.26) and 5.42% (±0.03), 28.81% (±0.65) and 21.17% (±0.54).
3.1. Experiment 1: Fatty Acid Transfer from Milk to Cheese
FA composition in milk and cheese for sheep and goats is reported in Table 1. In total, 10 OBCFA were identified in both types of milk (Table 1), including 3 OCFA (C13:0, C15:0 and C17:0), 4 isoBCFA (isoC14:0, isoC15:0, isoC16:0, and isoC17:0) and 3 anteisoBCFA (anteisoC13:0, anteisoC15:0 and anteisoC17:0).

Table 1.
Odd and branched chain fatty acid in milk and cheese of sheep and goat species.
Among them, C15:0 and C17:0 were the most abundant fatty acids, in agreement with other research on sheep [33], goats and cows [34,35]; these OCFA accounted for 29% and 16% of the total concentration of OBCFA, respectively. In our study, C13:0 represents only 2.3%, value that agrees with the observations in dairy cows in which it represents 2% of OCFA [34] or it has not been found [36], but lower than values previously reported in sheep’s milk, where accounted for 6.3% of total OCFA [33].
Concentrations of individual OBCFA in the fat of fresh cheeses did not differ from that of raw unprocessed milk (p > 0.05) in both species: it means that overall, OBCFA content of milk is not significantly modified by cheese making processes, both in sheep and goats.
In sheep milk, the content of total OBCFA is higher than that observed in goat milk, due to a higher content of isoBCFA form of C15:0, C16:0 and C17:0, and anteiso C15:0 and C17:0 (p < 0.05). No differences among species have been observed only for isoC13:0 and isoC14:0. The differences between species could be related to their different feeding regimen, as OBCFA in milk may change according to the bacterial population present in the rumen and, particularly, because of the differences in their relative abundance. Differences in OBCFA concentrations between these the species can occur also because of different extents of de novo synthesis of these FA in the mammary gland, differential efficiency of intestinal absorption, and differential storage of some OBCFA in adipose tissue, followed by their release during periods of fat mobilization.
The C17:1c9 was not detected in sheep and it was found in negligible amount in goat milk and fresh cheese. In the mammary gland, the delta-9 desaturase activity can promote the metabolization of some odd-chain fatty acids; only the conversion of C17:0 to C17:1cis9 has been found of quantitative importance in cows [37]. It has also been reported with a mean concentration of 0.2% in milk of Assaf ewes raised in controlled and intensive farming system [33]. Therefore, the lack of C17:1c9 in Sarda dairy ewes might be related to specific environmental condition related to the typical extensive breeding system of Sarda dairy ewes and Sardinian goats.
The total content of OBCFA in sheep milk is higher in this experiment than that reported for cows [36] and sheep (3.19 g/100 g of total FA) [33]. Differences could be related to different feeding strategies at farm level, as grazing pasture, or vegetable lipid supplementation, forage to concentrate ratio or other dietary factors, that deserve to be further investigated.
3.2. Experiment 2: Seasonal Variation of Branched and Odd Chain FA
Seasonal variation of individual and group of branched and odd chain fatty acid has been investigated by collecting cheese samples throughout all lactation period of sheep and goats, that in Sardinia, as in all Mediterranean environmental conditions, occurs from January (start of lactation) to July (when animals are dry off).
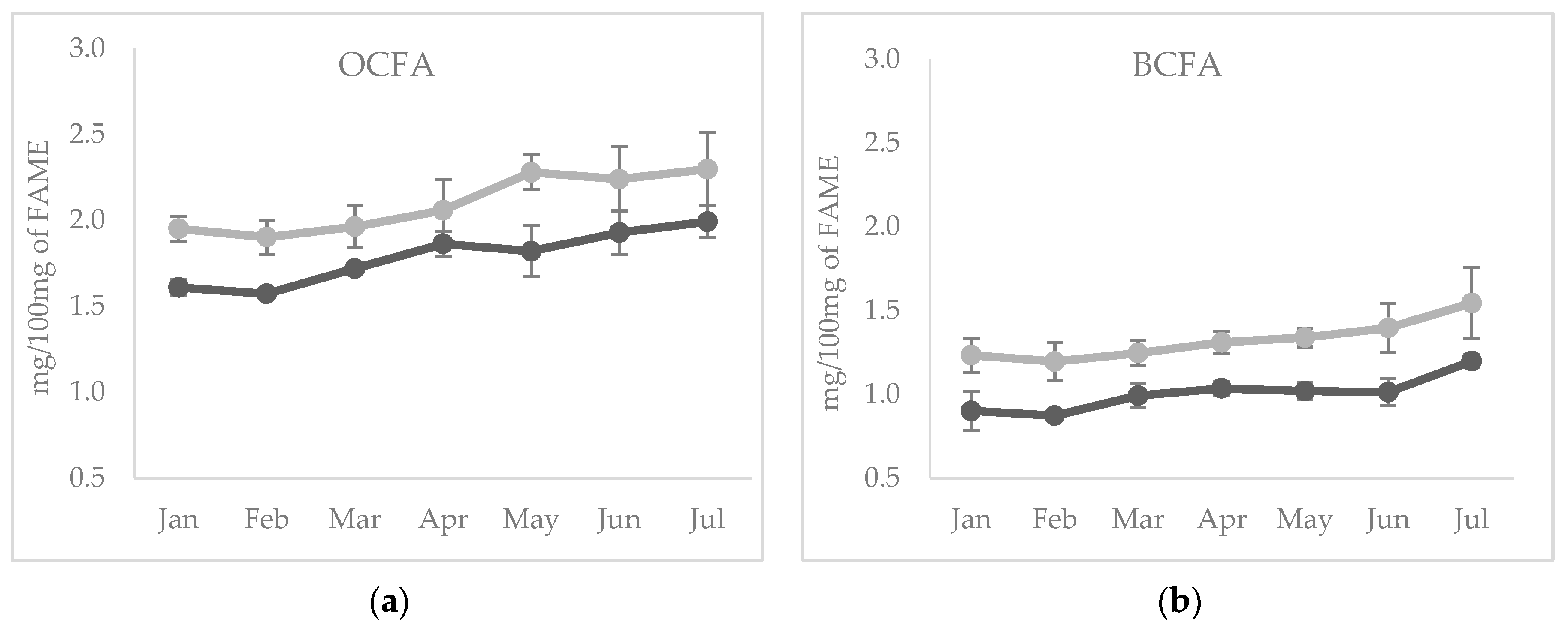
The temporal evolution of these FA in cheese showed an increase of total OCFA and BCFA in both species (p < 0.05) across all the sampling period (Figure 1a,b); the highest contents of these FA were observed in the cheese produced with milk in advanced lactation, which corresponds also to the hot season. The increase of OBCFA across lactation has been previously reported in dairy cows [34].
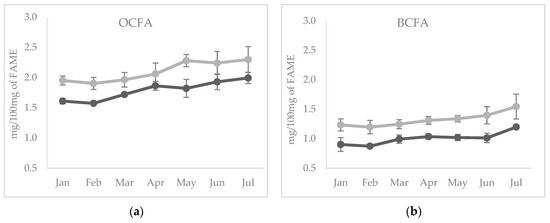
Figure 1.
Temporal evolution of (a) odd chain fatty acids (FA) and (b) branched chain FA from January to July in cheeses from sheep (light grey line) and goats (dark grey line) milk.
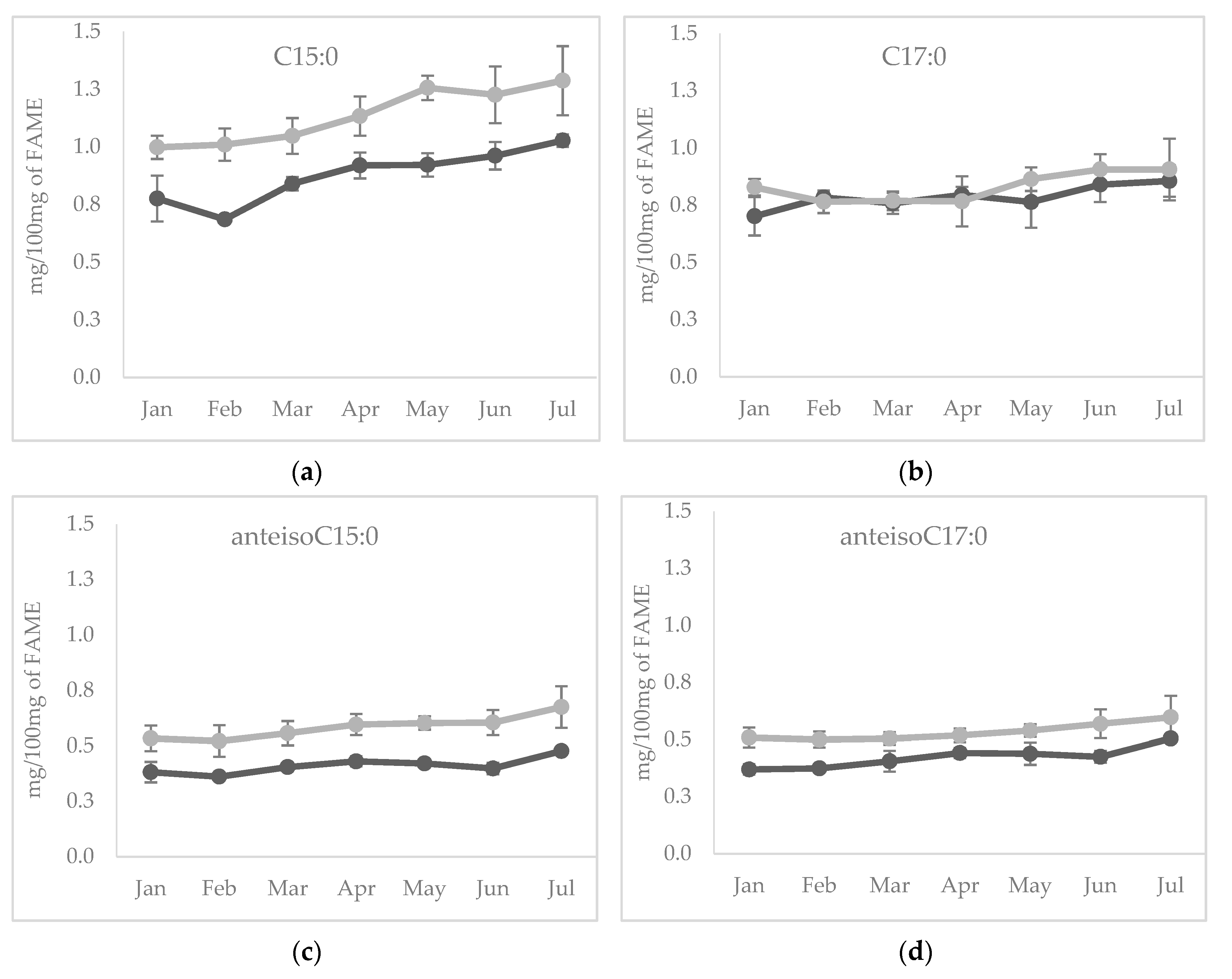
Among individual OCFA, the content of odd C15:0 during lactation followed the same pattern of the total OCFA, being the main representative of this group of FA (Figure 2a), whereas C17:0 showed constant concentration during the early and mid-lactation, then increased at the end of lactation, in June and July for both species (Figure 2b). As far as the changes of individual BCFA during lactation are concerned, both anteisoC15:0 and anteisoC17:0 (Figure 2c,d) followed a similar pattern observed for total BCFA.
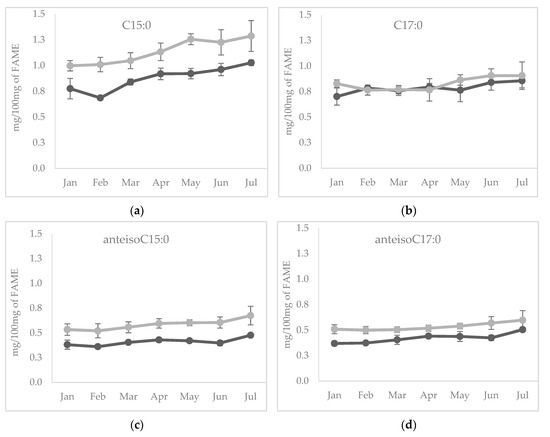
Figure 2.
Temporal evolution of (a) C15:0, (b) C17:0, (c) anteisoC15:0) and (d) anteisoC17:0 from January to July in sheep (light grey line) and in goats (dark grey line) cheeses.
Changes in the animals’ diet during the period under investigation, and in particular the progressive modification of pasture grazed by animals, can be the most probable explanation for the observed FA patterns. In fact, the typical lactation curve of the Sarda sheep breed follows the natural availability, both in quantitative and qualitative terms, of pastures [38]. As lactation progress, there is a worsening of nutritional quality of pasture characterized by increase of fiber and a reduction of protein and FA contents, especially alpha-linolenic acid (C18:3n3), which concentrations decrease in mature grass [39,40]. This is supported by evolution of C18:3in cheeses samples as lactation progress (data not reported), that confirmed the pattern of Sarda ewes previously observed [28].
Different mechanisms could be hypothesized to explain this pattern. One mechanism could be related to the decrease of polyunsaturated fatty acids (PUFA) in the pasture that could have reduced the toxic effect of unsaturated lipids on microbial growth [41,42], especially of cellulolytic bacteria [43]. This could increase the rumen microbial abundance, followed by higher rumen outflow of fatty acid of microbial origin. A second possible explanation could be the positive association between OBCFA and dietary fiber as with progress of lactation there is an increase of neutral detergent fiber (NDF), especially acid detergent lignin (ADL) in pasture. This is supported by previously observation in dairy goats where dietary NDF was found the most important factor of variation in lipid composition of bacteria; and in dairy cows where the proportion of odd- and branched-chain FA increased and those of even-chain saturated FA decreased with increasing forage [44].
The contents of almost all OBCFA in goat cheese differed from that of sheep, confirming the findings of the experiment 1. It is noteworthy that the goat’s milk processed into cheese in this Mediterranean area comes from animals raised in extensive system, characterized by natural pastures rich also in shrubs and essences of the Mediterranean maquis, which contains tannins. These different pasture conditions among species could be a possible explanation of the different concentrations in OBCFA observed between sheep and goats [45].
In order to assess the overall differences among sheep and goat cheese in terms of FA composition, a multivariate statistical analysis was carried out on the detailed FA profile. In particular, PCA was used because it is a useful instrument to reduce the complexity of a multivariate space, such as that of FA of cheese, and has demonstrated to be able to separate samples (e.g., of different origin, dietary treatment, season of production, lactation stage), based exclusively on the FA composition [46,47,48]. An important application of PCA could be, as an example, the authentication of dairy products according to their lipid profile [49].
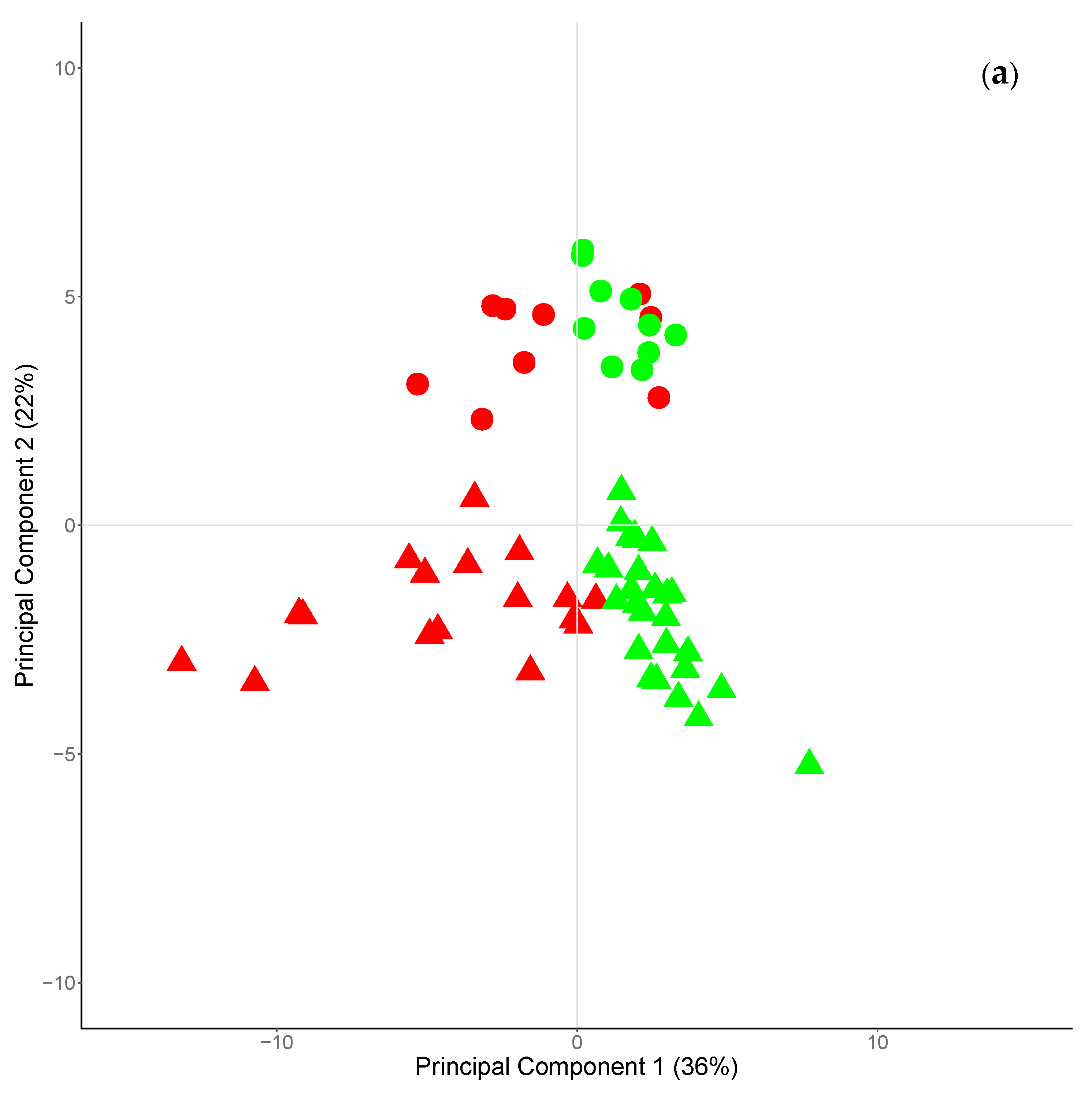
The first five principal components (PCs) explained the 80% of the total variance, with the two first PCs accounting for more than 50% (PC1 and PC2, 36% and 22%, respectively). The plot of the scores of the PC1 and PC2 (Figure 3a) allows the clustering of cheeses according to the season of milk production (PC1) or to their species of origin (PC2). Cheeses produced during winter–early spring season had positive scores for PC1, whereas those produced during late-spring–summer season had negative scores. Regarding the PC2, goat and sheep cheese had positive and negative scores, respectively. In the same plot, can be also observed that the Fa profile of sheep cheese was characterized by a larger variability compared to goat one, across the seasons of production.
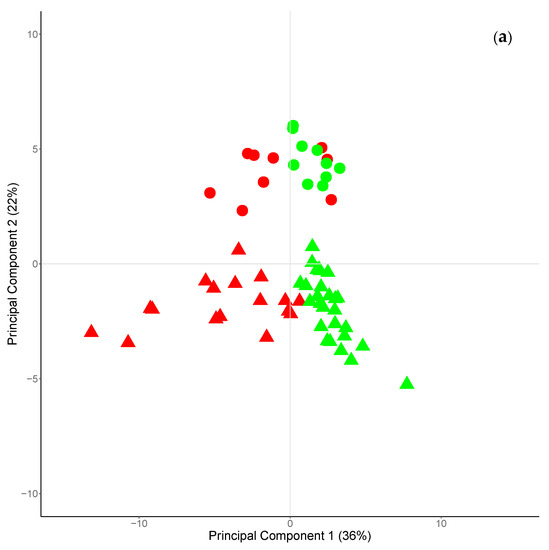
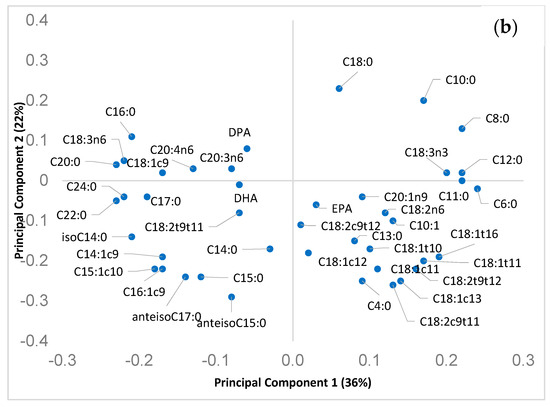
Figure 3.
Scores (a) and loadings (b) plots of the two first principal components explaining 36 and 22% of the total variance, respectively. In Figure 3a, circle identifies goat cheese, triangle identifies sheep cheese; green symbols identified cheese produced in winter early spring, red symbols identified cheese produced in early spring–summer.
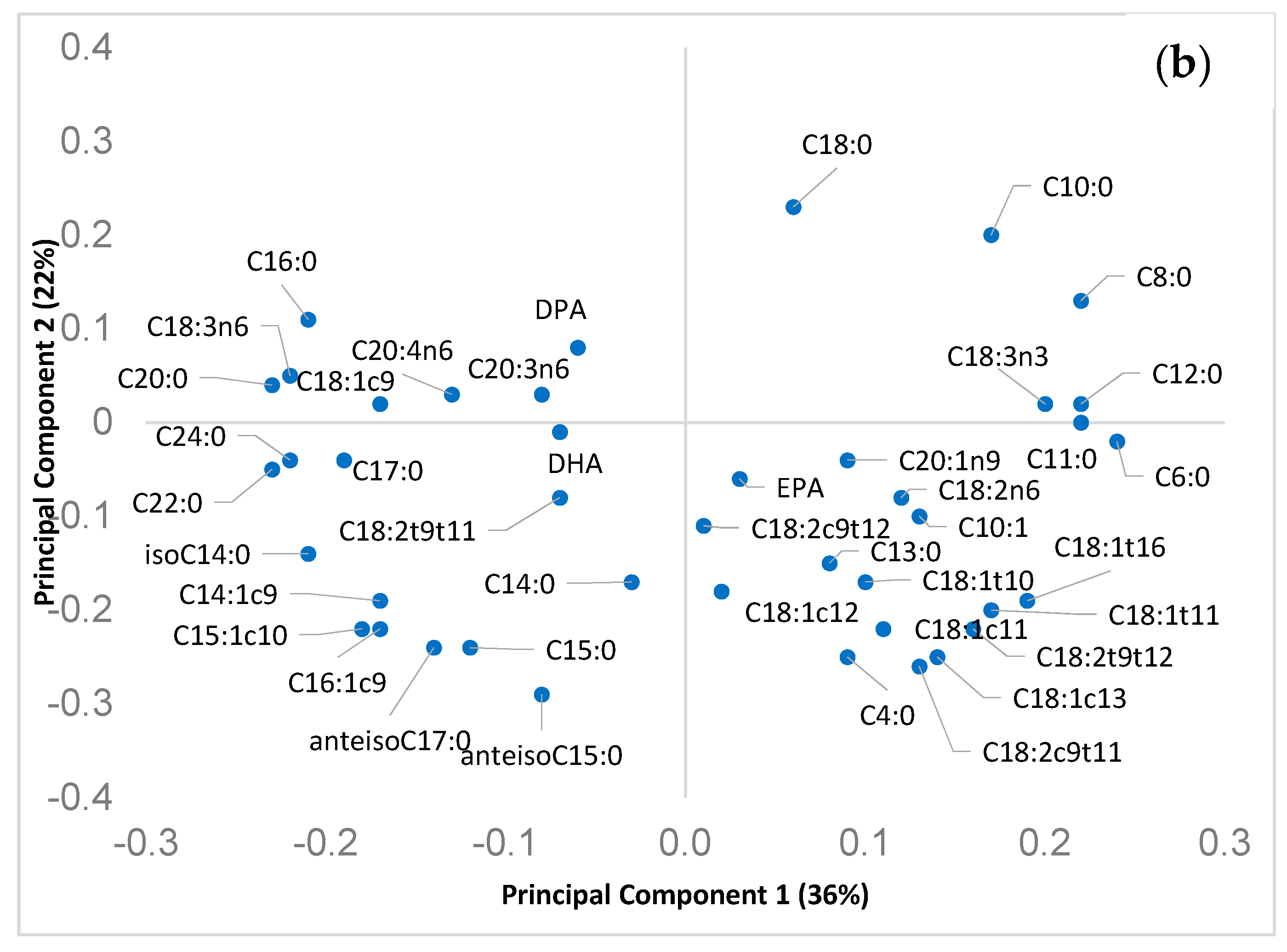
The loadings of the first two PCs are plotted in Figure 3b. PC loadings can be used to describe and explain the main FA differences among the clusters of cheese observed in Figure 3a, i.e., the plot of the scores (cheese produced in different season or from milk of different species).
The original variables (i.e., single fatty acids) exhibiting the highest loading values for the 2 PCs (both negatives and positives) can be used to describe the main differences, in terms of FA, among samples belonging to the cheese of the two species and, also, produced in different seasons.
PC1 had high positive loadings for short chain FA (C4:0, C6:0, C8:0, C10:0), alpha linolenic acid, vaccenic and rumenic acid. High negative loadings for PC1 were observed for different long chain saturated FA (C20:0, C22:0, C24:0) and for the C18:1cis-9. This pattern was quite consistent with the milk (and cheese) FA variation during seasons, due to the worsening of pasture quality: in particular, the decrease of alpha-linolenic, vaccenic and rumenic acids in dairy products is correlated to the decrease in pasture of alpha-linolenic acid content from winter to summer. In addition, PC1 loadings can be also related to the lactation progress (high short chain FA in early lactation and increase in C18:1 cis-9 in mid and late lactation, in response to the energy requirement). This double effect of pasture quality and animal lactation stage on the FA composition of cheese (particularly in sheep cheese) was recently reported by [48]. As aforementioned, it is likely to be a consequence of the typical seasonality of lambing of Sarda dairy sheep.
PC2 had larger negative loadings for rumenic acid, C4:0 and some OBCFA, in particular, anteisoC15:0, C15:0, and some isomer of C18:1, including vaccenic acid. Higher positive loadings of PC2 were observed for C18:0 and C10:0. This pattern suggested a better FA composition of sheep cheese, from a nutritional point of view, considering that FA with negative loadings for PC2 (including some OBCFA), and, therefore, mostly related to sheep cheese, have been associated to beneficial aspect on human health, that were the subject of this work. Due to its nutritional importance, the rumenic acid (most abundant and important CLA isomer) should be mentioned. This FA has been received particular attention due to the important beneficial healthy effects shown in vitro, animal and human as well [50,51]. The higher concentration in sheep milk (and cheese) compared to that of goat has previously been reported [45].
4. Conclusions
Results of the present survey showed that odd and branched chain fatty acids concentrations in the fat of fresh cheese is related to their content in both sheep and goats’ milk. The seasonal evolution evidenced that OBCFA concentrations in milk fat increase with advancing lactation, probably due to variation in feeding technique typical of extensive system of Mediterranean area characterized by animal grazing on pasture. As lactation progress, the pasture availability and quality gets worse, leading to a reduction in the content of PUFA and protein and to an increase in fiber content. These results showed that cheese could be an important source of OBCFA, but the concentration could vary according to the lactation stage. The use of PCA on the detailed FA profile of cheese confirmed the higher nutritional quality of sheep cheese for beneficial FA including OBCFA compared to the goat one, and the importance of the period of sampling in the definition of the fatty acids profile.
Author Contributions
Individual contributions: A.N.: Conceptualization, Statistics and Writing. F.C.: Methodology and Analysis. A.C.: Software and PCA analysis. G.P.: Supervision and Writing. G.B.: Writing Original Draft, Preparation. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by “Fondo di Ateneo per la ricerca 2019 of University of Sassari”.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
The data presented in this study are available on request from the corresponding author.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Dagnachew, B.S.; Meuwissen, T.H.E.; Ådnøy, T. Genetic components of milk Fourier-transform infrared spectra used to predict breeding values for milk composition and quality traits in dairy goats. J. Dairy Sci. 2013, 96, 5933–5942. [Google Scholar] [CrossRef] [PubMed]
- Maroteau, C.; Palhière, I.; Larroque, H.; Clément, V.; Ferrand, M.; Tosser-Klopp, G.; Rupp, R. Genetic parameter estimation for major milk fatty acids in Alpine and Saanen primiparous goats. J. Dairy Sci. 2014, 97, 3142–3155. [Google Scholar] [CrossRef] [PubMed]
- Cesarani, A.; Gaspa, G.; Correddu, F.; Cellesi, M.; Dimauro, C.; Macciotta, N.P.P. Genomic selection of milk fatty acid composition in Sarda dairy sheep: Effect of different phenotypes and relationship matrices on heritability and breeding value accuracy. J. Dairy Sci. 2019, 102, 3189–3203. [Google Scholar] [CrossRef] [PubMed]
- Ran-Ressler, R.R.; Bae, S.; Lawrence, P.; Wang, D.H.; Brenna, J.T. Branched-chain fatty acid content of foods and estimated intake in the USA. Br. J. Nutr. 2014, 112, 565–572. [Google Scholar] [CrossRef] [PubMed]
- Ran-Ressler, R.R.; Khailova, L.; Arganbright, K.M.; Adkins-Rieck, C.K.; Jouni, Z.E.; Koren, O.; Ley, R.E.; Brenna, J.T.; Dvorak, B. Branched chain fatty acids reduce the incidence of necrotizing enterocolitis and alter gastrointestinal microbial ecology in a neonatal rat model. PLoS ONE 2011, 6, e29032. [Google Scholar] [CrossRef]
- Vlaeminck, B.; Fievez, V.; Tamminga, S.; Dewhurst, R.J.; Van Vuuren, A.; De Brabander, D.; Demeyer, D. Milk odd-and branched-chain fatty acids in relation to the rumen fermentation pattern. J. Dairy Sci. 2006, 89, 3954–3964. [Google Scholar] [CrossRef]
- Massart-Leën, A.M.; Peeters, G.; Vandeputte-Van Messom, G.; Roets, E.; Burvenich, C. Effects of valerate and isobutyrate on fatty acid secretion by the isolated perfused mammary gland of the lactating goat. Reprod. Nutr. Dev. 1986, 26, 801–814. [Google Scholar] [CrossRef]
- Berthelot, V.; Bas, P.; Pottier, E.; Normand, J. The effect of maternal linseed supplementation and/or lamb linseed supplementation on muscle and subcutaneous adipose tissue fatty acid composition of indoor lambs. Meat Sci. 2012, 90, 548–557. [Google Scholar] [CrossRef]
- Brevik, A.; Veierød, M.B.; Drevon, C.A.; Andersen, L.F. Evaluation of the odd fatty acids 15: 0 and 17: 0 in serum and adipose tissue as markers of intake of milk and dairy fat. Eur. J. Clin. Nutr. 2005, 59, 1417–1422. [Google Scholar] [CrossRef]
- Albani, V.; Celis-Morales, C.; O’Donovan, C.B.; Walsh, M.C.; Woolhead, C.; Forster, H.; Fallaize, R.; Macready, A.L.; Marsaux, C.F.M.; Navas-Carretero, S.; et al. Within-person reproducibility and sensitivity to dietary change of C15: 0 and C17: 0 levels in dried blood spots: Data from the European Food4Me Study. Mol. Nutr. Food. Res. 2017, 61, 1700142. [Google Scholar] [CrossRef]
- Khaw, K.T.; Friesen, M.D.; Riboli, E.; Luben, R.; Wareham, N. Plasma phospholipid fatty acid concentration and incident coronary heart disease in men and women: The EPIC-Norfolk prospective study. PLoS Med. 2012, 9, e1001255. [Google Scholar] [CrossRef] [PubMed]
- Jenkins, B.; West, J.A.; Koulman, A. A review of odd-chain fatty acid metabolism and the role of pentadecanoic Acid (c15:0) and heptadecanoic Acid (c17:0) in health and disease. Molecules 2015, 20, 2425–2444. [Google Scholar] [CrossRef]
- Warensjö, E.; Jansson, J.H.; Cederholm, T.; Boman, K.; Eliasson, M.; Hallmans, G.; Johansson, I.; Sjögren, P. Biomarkers of milk fat and the risk of myocardial infarction in men and women: A prospective, matched case-control study. Am. J. Clin. Nutr. 2010, 92, 194–202. [Google Scholar] [CrossRef] [PubMed]
- Sun, Q.; Ma, J.; Campos, H.; Hu, F.B. Plasma and erythrocyte biomarkers of dairy fat intake and risk of ischemic heart disease. Am. J. Clin. Nutr. 2007, 86, 929–937. [Google Scholar] [CrossRef]
- Krachler, B.; Norberg, M.; Eriksson, J.W.; Hallmans, G.; Johansson, I.; Vessby, B.; Weinehall, L.; Lindahl, B. Fatty acid profile of the erythrocyte membrane preceding development of Type 2 diabetes mellitus. Nutr. Metab. Cardiovasc. Dis. 2008, 18, 503–510. [Google Scholar] [CrossRef]
- Hodge, A.M.; English, D.R.; O’Dea, K.; Sinclair, A.J.; Makrides, M.; Gibson, R.A.; Giles, G.G. Plasma phospholipid and dietary fatty acids as predictors of type 2 diabetes: Interpreting the role of linoleic acid. Am. J. Clin. Nutr. 2007, 86, 189–197. [Google Scholar] [CrossRef]
- Forouhi, N.G.; Koulman, A.; Sharp, S.J.; Imamura, F.; Kröger, J.; Schulze, M.B.; Crowe, F.L.; Huerta, J.M.; Guevara, M.; Beulens, J.W.; et al. Differences in the prospective association between individual plasma phospholipid saturated fatty acids and incident type 2 diabetes: The EPIC-InterAct case-cohort study. Lancet Diabetes Endocrinol. 2014, 2, 810–818. [Google Scholar] [CrossRef]
- Imamura, F.; Fretts, A.; Marklund, M.; Ardisson Korat, A.V.; Yang, W.S.; Lankinen, M.; Qureshi, W.; Helmer, C.; Chen, T.A.; Wong, K.; et al. Fatty acid biomarkers of dairy fat consumption and incidence of type 2 diabetes: A pooled analysis of prospective cohort studies. PLoS Med. 2018, 15, e1002670. [Google Scholar] [CrossRef]
- Venn-Watson, S.; Lumpkin, R.; Dennis, E.A. Efficacy of dietary odd-chain saturated fatty acid pentadecanoic acid parallels broad associated health benefits in humans: Could it be essential? Sci. Rep. 2020, 10, 8161. [Google Scholar] [CrossRef] [PubMed]
- Correddu, F.; Gaspa, G.; Pulina, G.; Nudda, A. Grape seed and linseed, alone and in combination, enhance unsaturated fatty acids in the milk of Sarda dairy sheep. J. Dairy Sci. 2016, 99, 1725–1735. [Google Scholar] [CrossRef]
- Baumann, E.; Chouinard, P.Y.; Lebeuf, Y.; Rico, D.E.; Gervais, R. Effect of lipid supplementation on milk odd- and branched-chain fatty acids in dairy cows. J. Dairy Sci. 2016, 99, 6311–6323. [Google Scholar] [CrossRef] [PubMed]
- Vazirigohar, M.; Dehghan-Banadaky, M.; Rezayazdi, K.; Nejati-Javaremi, A.; Mirzaei-Alamouti, H.; Patra, A.K. Short communication: Effects of diets containing supplemental fats on ruminal fermentation and milk odd- and branched-chain fatty acids in dairy cows. J. Dairy Sci. 2018, 101, 6133–6141. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Liu, K.; Hao, X.; Xin, H. The relationships between odd-and branched-chain fatty acids to ruminal fermentation parameters and bacterial populations with different dietary ratios of forage and concentrate. J. Anim. Physiol. Anim. Nutr. 2017, 101, 1103–1114. [Google Scholar] [CrossRef] [PubMed]
- Correddu, F.; Nudda, A.; Battacone, G.; Boe, R.; Francesconi, A.H.D.; Pulina, G. Effects of grape seed supplementation, alone or associated with linseed, on ruminal metabolism in Sarda dairy sheep. Anim. Feed. Sci. Technol. 2015, 199, 61–72. [Google Scholar] [CrossRef]
- Stoop, W.M.; Bovenhuis, H.; Heck, J.M.; van Arendonk, J.A. Effect of lactation stage and energy status on milk fat composition of Holstein-Friesian cows. J. Dairy Sci. 2009, 92, 1469–1478. [Google Scholar] [CrossRef]
- De Souza, J.; Leskinen, H.; Lock, A.L.; Shingfield, K.J.; Huhtanen, P. Between-cow variation in milk fatty acids associated with methane production. PLoS ONE 2020, 15, e0235357. [Google Scholar] [CrossRef]
- Craninx, M.; Steen, A.; Van Laar, H.; Van Nespen, T.; Martin-Tereso, J.; De Baets, B.; Fievez, V. Effect of lactation stage on the odd-and branched-chain milk fatty acids of dairy cattle under grazing and indoor conditions. J. Dairy Sci. 2008, 91, 2662–2677. [Google Scholar] [CrossRef]
- Nudda, A.; McGuire, M.A.; Battacone, G.; Pulina, G. Seasonal variation in conjugated linoleic acid and vaccenic acid in milk fat of sheep and its transfer to cheese and ricotta. J. Dairy Sci. 2005, 88, 1311–1319. [Google Scholar] [CrossRef]
- Santillo, A.; Caroprese, M.; Marino, R.; d’Angelo, F.; Sevi, A.; Albenzio, M. Fatty acid profile of milk and Cacioricotta cheese from Italian Simmental cows as affected by dietary flaxseed supplementation. J. Dairy Sci. 2016, 99, 2545–2551. [Google Scholar] [CrossRef]
- IDF Standard 182:1999. Milk Fat: Preparation of Fatty Acid Methyl Esters; IDF: Brussels, Belgium, 1999. [Google Scholar]
- Gómez-Cortés, P.; Rodríguez-Pino, V.; Juárez, M.; de la Fuente, M.A. Optimization of milk odd and branched-chain fatty acids analysis by gas chromatography using an extremely polar stationary phase. Food Chem. 2017, 231, 11–18. [Google Scholar] [CrossRef]
- Vetter, W.; Gaul, S.; Thurnhofer, S.; Mayer, K. Stable carbon isotope ratios of methyl-branched fatty acids are different to those of straight-chain fatty acids in dairy products. Anal. Bioanal. Chem. 2007, 389, 597–604. [Google Scholar] [CrossRef]
- Toral, P.G.; Hervás, G.; Della Badia, A.; Gervais, R.; Frutos, P. Effect of dietary lipids and other nutrients on milk odd- and branched-chain fatty acid composition in dairy ewes. J. Dairy Sci. 2020, 103, 11413–11423. [Google Scholar] [CrossRef] [PubMed]
- Bainbridge, M.L.; Cersosimo, L.M.; Wright, A.D.; Kraft, J. Content and Composition of Branched-Chain Fatty Acids in Bovine Milk Are Affected by Lactation Stage and Breed of Dairy Cow. PLoS ONE 2016, 11, e0150386. [Google Scholar] [CrossRef]
- Ran-Ressler, R.R.; Sim, D.; O’Donnell-Megaro, M.; Bauman, D.E.; Barbano, D.M.; Brenna, J.T. Branched chain fatty acid content of United States retail cow’s milk and implications for dietary intake. Lipids 2011, 46, 569–576. [Google Scholar] [CrossRef] [PubMed]
- Xin, H.; Xu, Y.; Chen, Y.; Chen, G.; Steele, M.A.; Guan, L.L. Short communication: Odd-chain and branched-chain fatty acid concentrations in bovine colostrum and transition milk and their stability under heating and freezing treatments. J. Dairy Sci. 2020, 103, 11483–11489. [Google Scholar] [CrossRef] [PubMed]
- Fievez, V.; Vlaeminck, B.; Dhanoa, M.S.; Dewhurst, R.J. Use of principal component analysis to investigate the origin of heptadecenoic and conjugated linoleic acids in milk. J. Dairy Sci. 2003, 86, 4047–4053. [Google Scholar] [CrossRef]
- Macciotta, N.P.P.; Cappio-Borlino, A.; Pulina, G. Analysis of environmental effects on test day milk yields of Sarda dairy ewes. J. Dairy Sci. 1999, 82, 2212–2217. [Google Scholar] [CrossRef]
- Cabiddu, A.; Decandia, M.; Addis, M.; Piredda, G.; Pirisi, A.; Molle, G. Managing Mediterranean pastures in order to enhance the level of beneficial fatty acids in sheep milk. Small Rumin. Res. 2005, 59, 169–180. [Google Scholar] [CrossRef]
- Chilliard, Y.; Ferlay, A.; Mansbridge, R.M.; Doreau, M. Ruminant milk fat plasticity: Nutritional control of saturated, polyunsaturated, trans and conjugated fatty acids. Ann. Zootech. 2000, 49, 181–205. [Google Scholar] [CrossRef]
- Lourenço, M.; Ramos-Morales, E.; Wallace, R.J. The role of microbes in rumen lipolysis and biohydrogenation and their manipulation. Animal 2010, 4, 1008–1023. [Google Scholar] [CrossRef] [PubMed]
- Enjalbert, F.; Combes, S.; Zened, A.; Meynadier, A. Rumen microbiota and dietary fat: A mutual shaping. J. Appl. Microbiol. 2017, 123, 782–797. [Google Scholar] [CrossRef] [PubMed]
- Maia, M.R.; Chaudhary, L.C.; Figueres, L.; Wallace, R.J. Metabolism of polyunsaturated fatty acids and their toxicity to the microflora of the rumen. Antonie Van Leeuwenhoek 2007, 91, 303–314. [Google Scholar] [CrossRef] [PubMed]
- Bas, P.; Archimède, H.; Rouzeau, A.; Sauvant, D. Fatty acid composition of mixed-rumen bacteria: Effect of concentration and type of forage. J. Dairy Sci. 2003, 86, 2940–2948. [Google Scholar] [CrossRef]
- Nudda, A.; Cannas, A.; Correddu, F.; Atzori, A.S.; Lunesu, M.F.; Battacone, G.; Pulina, G. Sheep and Goats Respond Differently to Feeding Strategies Directed to Improve the Fatty Acid Profile of Milk Fat. Animals 2020, 10, 1290. [Google Scholar] [CrossRef]
- Kadegowda, A.K.G.; Piperova, L.S.; Erdman, R.A. Principal component and multivariate analysis of milk long-chain fatty acid composition during diet-induced milk fat depression. J. Dairy Sci. 2008, 91, 749–759. [Google Scholar] [CrossRef]
- Chung, I.M.; Kim, J.K.; Lee, K.J.; Son, N.Y.; An, M.J.; Lee, J.H.; An, Y.J.; Kim, S.H. Discrimination of organic milk by stable isotope ratio, vitamin E, and fatty acid profiling combined with multivariate analysis: A case study of monthly and seasonal variation in Korea for 2016–2017. Food Chem. 2018, 261, 112–123. [Google Scholar] [CrossRef]
- Correddu, F.; Murgia, M.A.; Mangia, N.P.; Lunesu, M.F.; Cesarani, A.; Deiana, P.; Pulina, G.; Nudda, A. Effect of altitude of flock location, season of milk production and ripening time on the fatty acid profile of Pecorino Sardo cheese. Int. J. Dairy Technol. 2021, 113, 104895. [Google Scholar] [CrossRef]
- Gómez-Cortés, P.; Cívico, A.; de la Fuente, M.A.; Núñez Sánchez, N.; Peña Blanco, F.; Martínez Marín, A.L. Effects of dietary concentrate composition and linseed oil supplementation on the milk fatty acid profile of goats. Animal 2018, 12, 2310–2317. [Google Scholar] [CrossRef]
- Yang, B.; Chen, H.; Stanton, C.; Ross, R.P.; Zhang, H.; Chen, Y.Q.; Chen, W. Review of the roles of conjugated linoleic acid in health and disease. J. Funct. Foods 2015, 15, 314–325. [Google Scholar] [CrossRef]
- Ferlay, A.; Bernard, L.; Meynadier, A.; Malpuech-Brugère, C. Production of trans and conjugated fatty acids in dairy ruminants and their putative effects on human health: A review. Biochimie 2017, 141, 107–120. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).