Medial Cortical Structures Mediate Implicit Trustworthiness Judgments about Kin Faces, but Not Familiar Faces: A Brief Report
Abstract
:1. Introduction
2. Materials and Methods
2.1. Participants/Stimuli
2.2. fMRI Acquisition
2.3. Behavioral Procedures
2.4. fMRI Trustworthiness Analysis
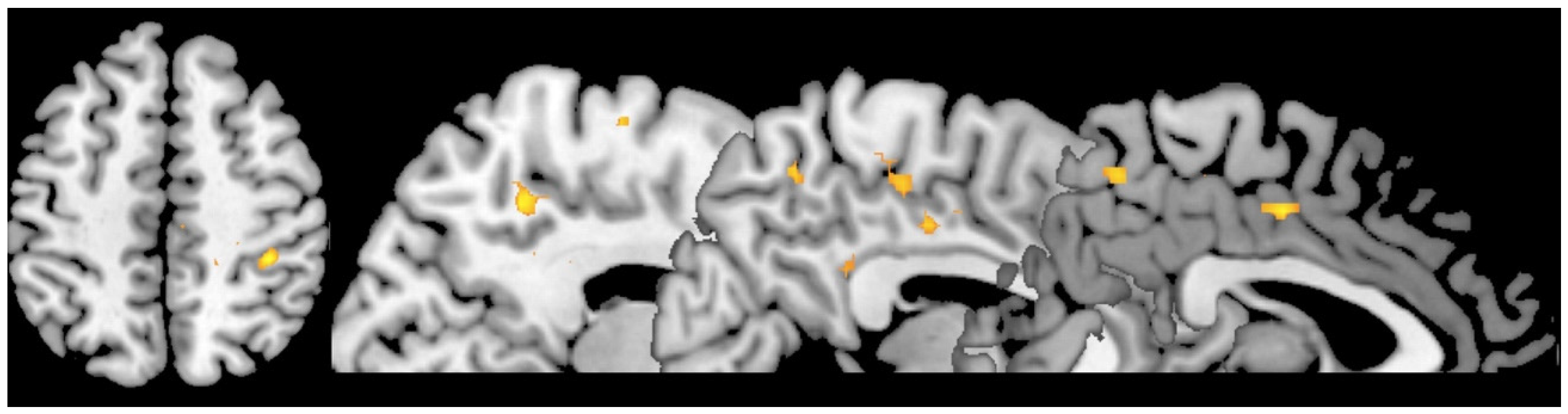
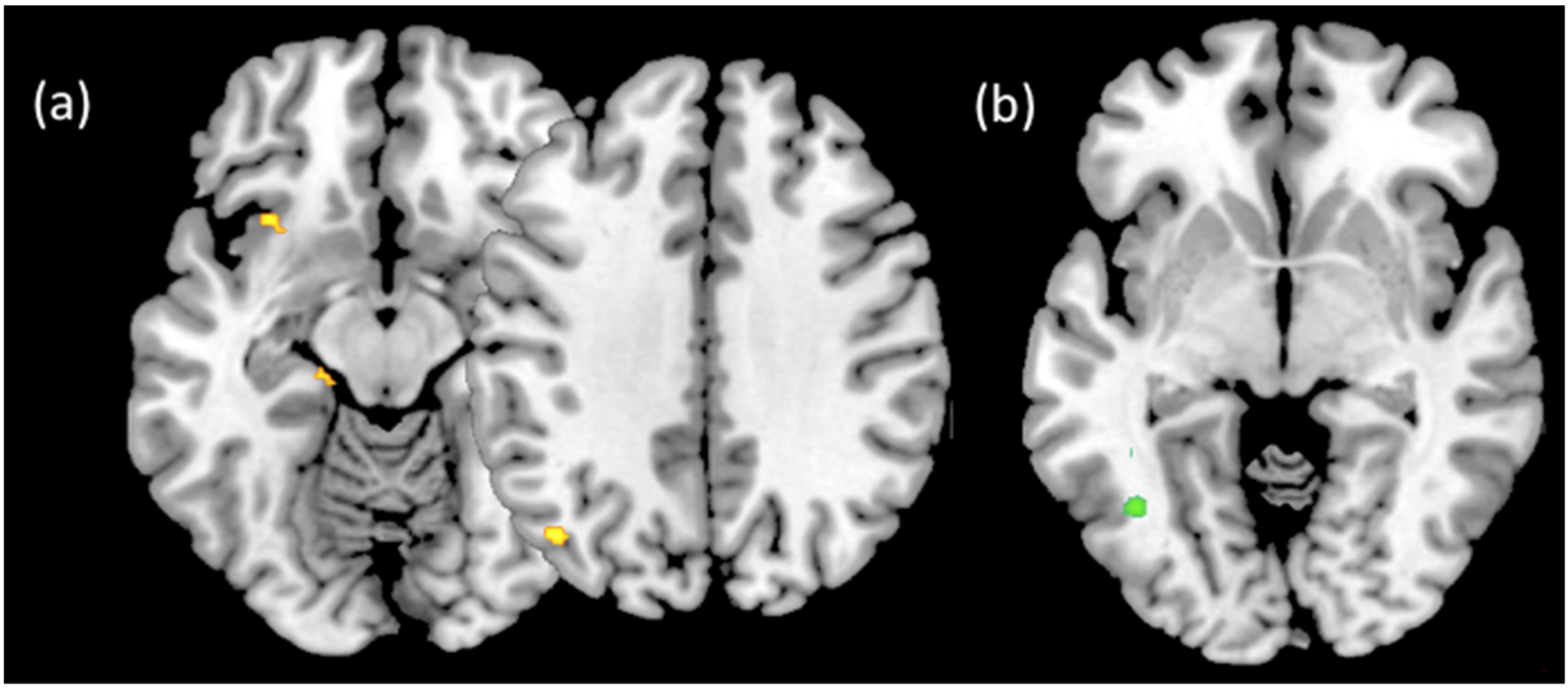
3. Results
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Hamilton, W. The genetical evolution of social behaviour. II. J. Theor. Biol. 1964, 7, 17–52. [Google Scholar] [CrossRef]
- Bourke, A.F.G. Hamilton’s rule and the causes of social evolution. Philos. Trans. R. Soc. B Biol. Sci. 2014, 369, 20130362. [Google Scholar] [CrossRef] [PubMed]
- Gardner, A.; West, S.; Wild, G. The genetical theory of kin selection. J. Evol. Biol. 2011, 24, 1020–1043. [Google Scholar] [CrossRef] [PubMed]
- Gorrell, J.C.; McAdam, A.G.; Coltman, D.W.; Humphries, M.M.; Boutin, S. Adopting kin enhances inclusive fitness in asocial red squirrels. Nat. Commun. 2010, 1, 1–4. [Google Scholar] [CrossRef] [PubMed]
- Leadbeater, E.; Carruthers, J.M.; Green, J.P.; Rosser, N.S.; Field, J. Nest Inheritance Is the Missing Source of Direct Fitness in a Primitively Eusocial Insect. Science 2011, 333, 874–876. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Linksvayer, T.; Wade, M. Theoretical predictions for sociogenomic data: The effects of kin selection and sex-limited expression on the evolution of social insect genomes. Front. Ecol. Evol. 2016, 4, 65. [Google Scholar] [CrossRef]
- Warner, M.; Mikheyev, A.; Linksvayer, T. Genomic signature of kin selection in an ant with obligately sterile workers. Mol. Biol. Evol. 2017, 34, 1780–1787. [Google Scholar] [CrossRef]
- West, S.A.; Gardner, A. Adaptation and Inclusive Fitness. Curr. Biol. 2013, 23, R577–R584. [Google Scholar] [CrossRef] [Green Version]
- Charpentier, M.; Crawford, J.; Boulet, M.; Drea, C. Message ‘scent’: Lemurs detect the genetic relatedness and quality of conspecifics via olfactory cues. Anim. Behav. 2010, 80, 101–108. [Google Scholar] [CrossRef]
- Lehmann, L.; Perrin, N. Altruism, Dispersal, and Phenotype-Matching Kin Recognition. Am. Nat. 2002, 159, 451–468. [Google Scholar] [CrossRef]
- Daly, M.; Wilson, M.I. Whom are newborn babies said to resemble? Evol. Hum. Behav. 1982, 3, 69–78. [Google Scholar] [CrossRef]
- Daly, M.; Wilson, M.I. The Truth about Cinderella: A Darwinian View of Parental Love; Yale University Press: New Haven, CT, USA, 1998. [Google Scholar]
- DeBruine, L.M. Facial resemblance enhances trust. Proc. R. Soc. Lond. Ser. B Biol. Sci. 2002, 269, 1307–1312. [Google Scholar] [CrossRef] [PubMed]
- Debruine, L.; Jones, B.; Perrett, D. Women’s attractiveness judgments of self-resembling faces change across the menstrual cycle. Horm. Behav. 2005, 47, 379–383. [Google Scholar] [CrossRef] [PubMed]
- Hauber, M.E.; Sherman, P.W. Self-referent phenotype matching: theoretical considerations and empirical evidence. Trends Neurosci. 2001, 24, 609–616. [Google Scholar] [CrossRef]
- Lacy, R.C.; Sherman, P.W. Kin Recognition by Phenotype Matching. Am. Nat. 1983, 121, 489–512. [Google Scholar] [CrossRef]
- Lieberman, D.; Tooby, J.; Cosmides, L. The architecture of human kin detection. Nature 2007, 445, 727–731. [Google Scholar] [CrossRef] [Green Version]
- Neff, B.D.; Sherman, P.W. Decision making and recognition mechanisms. Proc. R. Soc. B Biol. Sci. 2002, 269, 1435–1441. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Platek, S.M. Unconcious reactions to children’s faces: the effects of resemblance. Evol. Cogn. 2002, 8, 207–214. [Google Scholar]
- Platek, S.M.; Raines, D.M.; Gallup, J.G.G.; Mohamed, F.B.; Thomson, J.W.; Myers, T.E.; Panyavin, I.S.; Levin, S.L.; Davis, J.A.; Fonteyn, L.C.M.; et al. Reactions to children’s faces: Males are more affected by resemblance than females are, and so are their brains. Evol. Hum. Behav. 2004, 25, 394–405. [Google Scholar] [CrossRef]
- Platek, S.M.; Krill, A.L.; Kemp, S.M. The neural basis of facial resemblance. Neurosci. Lett. 2008, 437, 76–81. [Google Scholar] [CrossRef]
- Platek, S.M.; Kemp, S.M. Is family special to the brain? An event-related fMRI study of familiar, familial, and self-face recognition. Neuropsychologia 2009, 47, 849–858. [Google Scholar] [CrossRef] [PubMed]
- Platek, S.M.; Burch, R.L.; Panyavin, I.S.; Wassserman, B.H., Jr.; Gallup, G.G. Reactions towards children’s faces: Resemblance matters more for females than males. Evol. Hum. Behav. 2002, 23, 159–166. [Google Scholar] [CrossRef]
- Platek, S.M.; Critton, R.L.; Burch, R.L.; Frederick, D.A.; Myers, T.E., Jr.; Gallup, G.G. How much paternal resemblance is enough? Sex differences in the reaction to resemblance but not in ability to detect resemblance. Evol. Hum. Behav. 2003, 24, 81–87. [Google Scholar] [CrossRef]
- Platek, S.M.; Keenan, J.P.; Mohamed, F.B. Sex differences in the neural correlates of child facial resemblance: An event-related fMRI study. NeuroImage 2005, 25, 1336–1344. [Google Scholar] [CrossRef] [PubMed]
- Rushton, J.P.; Russell, R.J.H.; Wells, P.A. Genetic similarity theory: Beyond kin selection. Behav. Genet. 1984, 14, 179–193. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Strassmann, J.; Owen, G.; Queller, D. Kin discrimination and cooperation in microbes. Annu. Rev. Microbiol. 2011, 65, 349–367. [Google Scholar] [CrossRef] [PubMed]
- Volk, A.; Quinsey, V.L. The influence of infant facial cues on adoption preferences. Hum. Nat. 2002, 13, 437–455. [Google Scholar] [CrossRef] [Green Version]
- Volk, A.A.; Quinsey, V.L. Parental Investment and Resemblance: Replications, Refinements, and Revisions. Evol. Psychol. 2007, 5, 1–14. [Google Scholar] [CrossRef]
- Willis, J.; Todorov, A. First impressions: Making up your mind after a 100-ms exposure to a face. Psychol. Sci. 2006, 17, 592–598. [Google Scholar] [CrossRef]
- Bzdok, D.; Langner, R.; Hoffstaedter, F.; Turetsky, B.; Zilles, K.; Eickhoff, S. The modular neuroarchitecture of social judgements on faces. Cereb. Cortex 2011, 22, 951–961. [Google Scholar] [CrossRef]
- Engell, A.D.; Haxby, J.V.; Todorov, A. Implicit Trustworthiness Decisions: Automatic Coding of Face Properties in the Human Amygdala. J. Cogn. Neurosci. 2007, 19, 1508–1519. [Google Scholar] [CrossRef] [PubMed]
- Winston, J.; Strange, B.; O’Doherty, J.; Dolan, R.; Strange, B. Automatic and intentional brain responses during evaluation of trustworthiness of faces. Nat. Neurosci. 2002, 5, 277–283. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bargh, J.; Schwader, K.; Hailey, S.; Dyer, R.; Boothby, E. Automaticity in social-cognitive processes. Trends Cogn. Sci. 2012, 16, 593–605. [Google Scholar] [CrossRef] [PubMed]
- Bargh, J.A.; Chartrand, T.L. The unbearable automaticity of being. Am. Psychol. 1999, 54, 462–479. [Google Scholar] [CrossRef]
- Hassin, R.; Uleman, J.; Bargh, J. The new unconscious; Oxford University Press: New York, NY, USA, 2005. [Google Scholar]
- O’Doherty, J.; Kringelbach, M.L.; Rolls, E.T.; Hornak, J.; Andrews, C.; Kringelbach, M.; Rolls, E. Abstract reward and punishment representations in the human orbitofrontal cortex. Nat. Neurosci. 2001, 4, 95–102. [Google Scholar] [CrossRef]
- O’Doherty, J.; Winston, J.; Critchley, H.; Perrett, D.; Burt, D.; Dolan, R.; Burt, M. Beauty in a smile: The role of medial orbitofrontal cortex in facial attractiveness. Neuropsychologia 2003, 41, 147–155. [Google Scholar] [CrossRef]
- Tsukiura, T.; Cabeza, R. Remembering beauty: Roles of orbitofrontal and hippocampal regions in successful memory encoding of attractive faces. Neuroimage 2011, 54, 653–660. [Google Scholar] [CrossRef] [Green Version]
- Winston, J.S.; O’Doherty, J.; Kilner, J.M.; Perrett, D.I.; Dolan, R.J. Brain systems for assessing facial attractiveness. Neuropsychologia 2007, 45, 195–206. [Google Scholar] [CrossRef] [Green Version]
- Bartels, A.; Zeki, S. The neural correlates of maternal and romantic love. Neuroimage 2004, 21, 1155–1166. [Google Scholar] [CrossRef] [Green Version]
- Platek, S.M.; Krill, A.L. Self-face resemblance attenuates other-race face effect in the amygdala. Brain Res. 2009, 1284, 156–160. [Google Scholar] [CrossRef]
- Santos, A.; Mier, D.; Kirsch, P.; Meyer-Lindenberg, A. Evidence for a general face salience signal in human amygdala. Neuroimage 2011, 54, 3111–3116. [Google Scholar] [CrossRef] [PubMed]
- Bornstein, M.; Aterberry, M.; Mash, C. Differentiated brain activity in response to faces of “own” versus “unfamiliar” babies in primipara mothers: An electrophysiological study. Dev. Neuropsychol. 2013, 38, 365–385. [Google Scholar] [CrossRef] [PubMed]
- Zhu, Q.; Song, Y.; Hu, S.; Li, X.; Tian, M.; Zhen, Z.; Dong, Q.; Kanwisher, N.; Liu, J. Heritability of the Specific Cognitive Ability of Face Perception. Curr. Biol. 2010, 20, 137–142. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Platek, S.M.; Krill, A.L.; Wilson, B. Implicit trustworthiness ratings of self-resembling faces activate brain centers involved in reward. Neuropsychologia 2009, 47, 289–293. [Google Scholar] [CrossRef] [PubMed]
- Minear, M.; Park, D.C. A lifespan database of adult facial stimuli. Behav. Res. Methods Instrum. Comput. 2004, 36, 630–633. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Beckmann, C.F.; Jenkinson, M.; Smith, S.M. General multilevel linear modeling for group analysis in FMRI. Neuroimage 2003, 20, 1052–1063. [Google Scholar] [CrossRef]
- Critchley, H.D.; Tang, J.; Glaser, D.; Butterworth, B.; Dolan, R.J. Anterior cingulate activity during error and autonomic response. Neuroimage 2005, 27, 885–895. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Woolrich, M.W.; Behrens, T.E.; Beckmann, C.F.; Jenkinson, M.; Smith, S.M. Multilevel linear modelling for FMRI group analysis using Bayesian inference. Neuroimage 2004, 21, 1732–1747. [Google Scholar] [CrossRef] [PubMed]
- Worsley, K.J.; Evans, A.C.; Marrett, S.; Neelin, P. A Three-Dimensional Statistical Analysis for CBF Activation Studies in Human Brain. Br. J. Pharmacol. 1992, 12, 900–918. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Aharon, I.; Etcoff, N.; Ariely, D.; Chabris, C.F.; O’Connor, E.; Breiter, H.C. Beautiful faces have variable reward value: fMRI and behavioral evidence. Neuron 2001, 32, 537–551. [Google Scholar]
- Gusnard, D.A.; Akbudak, E.; Shulman, G.L.; Raichle, M.E. Medial prefrontal cortex and self-referential mental activity: Relation to a default mode of brain function. Proc. Natl. Acad. Sci. USA 2001, 98, 4259–4264. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jenkins, A.; Mitchell, J. Medial prefrontal cortex subserves diverse forms of self-reflection. Soc. Neurosci. 2010, 6, 211–218. [Google Scholar] [CrossRef] [PubMed]
- Lou, H.C.; Luber, B.; Crupain, M.; Keenan, J.P.; Nowak, M.; Kjaer, T.W.; Sackeim, H.A.; Lisanby, S.H. Parietal cortex and representation of the mental Self. Proc. Natl. Acad. Sci. USA 2004, 101, 6827–6832. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vogeley, K.; Bussfeld, P.; Newen, A.; Herrmann, S.; Happe, F.; Falkai, P.; Maier, W.; Shah, N.J.; Fink, G.R.; Zilles, K. Mind Reading: Neural Mechanisms of Theory of Mind and Self-Perspective. Neuroimage 2001, 14, 170–181. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kocsor, F.; Rezneki, R.; Juhasz, S.; Bereczkei, T. Preference for facial self-resemblance and attractiveness in human mate choice. Arch. Sex. Behav. 2011, 40, 1263–1270. [Google Scholar] [CrossRef] [PubMed]
- Platek, S.M.; Thomson, J.W. Facial Resemblance Exaggerates Sex-Specific Jealousy-Based Decisions. Evol. Psychol. 2007, 5, 223–231. [Google Scholar] [CrossRef]
- Krill, A.; Platek, S.M. In-Group and Out-Group Membership Mediates Anterior Cingulate Activation to Social Exclusion. Front. Evol. Neurosci. 2009, 1, 1. [Google Scholar] [CrossRef] [PubMed]
- Hrdy, S. Mothers and Others: The Evolutionary Origins of Mutual Understanding; Belknap Press of Harvard University Press: Cambridge, MA, USA, 2009. [Google Scholar]
- Riem, M.; Bakermans-Kranenburg, M.; Pieper, S.; Tops, M.; Boksem, M.; Vermeiren, R.; van Ijzendoorn, M.; Rombouts, S. Oxytocin modulates amygdala, insula, and inferior frontal gyrus responses to infant crying: A randomized controlled trial. Biol. Psychiatry 2011, 70, 291–297. [Google Scholar] [CrossRef] [PubMed]
- Cameron, N.M.; Shahrokh, D.; Del Corpo, A.; Dhir, S.K.; Szyf, M.; Champagne, F.A.; Meaney, M.J.; Cameron, N. Epigenetic Programming of Phenotypic Variations in Reproductive Strategies in the Rat Through Maternal Care. J. Neuroendocr. 2008, 20, 795–801. [Google Scholar] [CrossRef] [PubMed]
- Verosky, S.C.; Todorov, A. Differential neural responses to faces physically similar to the self as a function of their valence. NeuroImage 2010, 49, 1690–1698. [Google Scholar] [CrossRef] [PubMed]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Platek, S.M.; Hendry, J.C. Medial Cortical Structures Mediate Implicit Trustworthiness Judgments about Kin Faces, but Not Familiar Faces: A Brief Report. Psych 2019, 1, 482-490. https://doi.org/10.3390/psych1010037
Platek SM, Hendry JC. Medial Cortical Structures Mediate Implicit Trustworthiness Judgments about Kin Faces, but Not Familiar Faces: A Brief Report. Psych. 2019; 1(1):482-490. https://doi.org/10.3390/psych1010037
Chicago/Turabian StylePlatek, Steven M., and Judson C. Hendry. 2019. "Medial Cortical Structures Mediate Implicit Trustworthiness Judgments about Kin Faces, but Not Familiar Faces: A Brief Report" Psych 1, no. 1: 482-490. https://doi.org/10.3390/psych1010037
APA StylePlatek, S. M., & Hendry, J. C. (2019). Medial Cortical Structures Mediate Implicit Trustworthiness Judgments about Kin Faces, but Not Familiar Faces: A Brief Report. Psych, 1(1), 482-490. https://doi.org/10.3390/psych1010037



