Ultraviolet Absorption Spectra of Benzene and Chlorobenzene in Water-Ice Solutions at Temperatures Between 78 K and 273 K
Abstract
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
2.2.1. Ultraviolet Spectrometer
2.2.2. Cryostat and Cell
3. Results
3.1. Benzene
3.2. Chlorobenzene
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dawes, A.; Pascual, N.; Hoffmann, S.V.; Jones, N.C.; Mason, N.J. Vacuum ultraviolet photo absorption spectroscopy of crystalline and amorphous benzene. Phys. Chem. Chem. Phys. 2017, 19, 27544–27555. [Google Scholar] [CrossRef] [PubMed]
- Dawes, A.; Pascual, N.; Mason, N.J.; Gärtner, S.; Hoffmann, S.V.; Jones, N.C. Probing the interaction between solid benzene and water using vacuum ultraviolet and infrared spectroscopy. Phys. Chem. Chem. Phys. 2018, 20, 15273–15287. [Google Scholar] [CrossRef]
- Spangler, J.D.; Kilmer, N.G. Electronic spectra of benzene in cyclohexane at 77°K. J. Chem. Phys. 1968, 48, 698–714. [Google Scholar] [CrossRef]
- Bernstein, E.R.; Lee, J. Absorption spectrum of the 2600 Å transition of benzene in cryogenic liquids. J. Chem. Phys. 1981, 74, 3159–3163. [Google Scholar] [CrossRef][Green Version]
- Bayliss, N.S.; Hulme, L. Solvent effects in the spectra of benzene, toluene, and chlorobenzene at 2600 and 2000 Å. Aust. J. Chem. 1953, 6, 257–277. [Google Scholar] [CrossRef]
- Sponer, H.; Nordheim, G.; Sklar, A.L.; Teller, E. Analysis of the near ultraviolet electronic transition of benzene. J. Chem. Phys. 1939, 7, 207–220. [Google Scholar] [CrossRef]
- Garforth, F.M.; Ingold, C.K.; Poole, H.G. Excited states of benzene. Part I. Introductory consideration of some spectral properties of vibrations accompanying electronic transitions of benzene and deuterated benzenes. J. Chem. Soc. 1948, 95, 406–417. [Google Scholar] [CrossRef]
- Bernhardsson, A.; Forsberg, N.; Malmqvist, P.A.; Roos, B.O.; Serrano-Andres, L. A theoretical study of the 1B2u and 1B1u vibronic bands in benzene. J. Chem. Phys. 2000, 112, 2798–2809. [Google Scholar] [CrossRef]
- Li, J.; Lin, C.K.; Li, X.Y.; Zhu, C.Y.; Lin, S.H. Symmetry forbidden vibronic spectra and internal conversion in benzene. Phys. Chem. Chem. Phys. 2010, 12, 14967–14976. [Google Scholar] [CrossRef]
- Pantos, E.; Philis, J.; Bolovinos, A. The extinction coefficient of benzene vapor in the region 4.6 to 36 eV. J. Mol. Spectrosc. 1978, 72, 36–43. [Google Scholar] [CrossRef]
- Palmer, M.H.; Ridley, T.; Hoffmann, S.V.; Jones, N.C.; Coreno, M.; de Simone, M.; Grazioli, C.; Zhang, T.; Biczysko, M.; Baiardi, A.; et al. Combined theoretical and experimental study of the valence, Rydberg, and ionic states of chlorobenzene. J. Chem. Phys. 2016, 144, 124302. [Google Scholar] [CrossRef]
- Scharping, H.; Zetzch, C. Substituent effects In the VUV absorption spectra of monochlorobenzene and o-, m-, and p-dichlorobenzene. J. Mol. Spectrosc. 1985, 112, 8–17. [Google Scholar] [CrossRef]
- Andrejeva, A.; Tuttle, W.D.; Harris, J.P.; Wright, T.G. Assignment of the vibrations of the S, S1, and D+ states of perhydrogenated and perdeuterated isotopologues of chlorobenzene. J. Chem. Phys. 2015, 143, 104312. [Google Scholar] [CrossRef]
- Wright, T.G.; Panov, S.E.; Miller, T.A. Vibrational spectroscopy of the chlorobenzene cation using zero kinetic energy photoelectron spectroscopy. J. Chem. Phys. 1995, 102, 4793–4803. [Google Scholar] [CrossRef]
- Wilson, E.B., Jr. The normal modes and frequencies of vibration of the regular plane hexagon model of the benzene molecule. Phys. Rev. 1934, 45, 706–714. [Google Scholar] [CrossRef]
- Bist, H.D.; Sarin, V.N.; Ojha, A.; Jain, Y.S. The 2699 A electronic band system of chlorobenzene. The in-plane vibrational modes in the excited state. Appl. Spectrosc. 1970, 24, 292–294. [Google Scholar] [CrossRef]
- Jain, J.S.; Bist, H.D. The out-of-plane vibrational modes of chlorobenzene in its ground and first singlet excited states. J. Mol. Spectrosc. 1973, 47, 126–133. [Google Scholar] [CrossRef]
- Albert, S.; Keppler, K.; Lerch, P.; Quack, M.; Wokaun, A. Synchrotron-based highest resolution FTIR spectroscopy of chlorobenzene. J. Mol. Spectrosc. 2015, 315, 92–101. [Google Scholar] [CrossRef]
- Sponer, H.; Kirby-Smith, J.S. The Raman spectra of mono- and dichlorobenzenes. J. Chem. Phys. 1941, 9, 667–672. [Google Scholar] [CrossRef]
- Cvitaš, T.; Hollas, J.M. Rotational band contour analysis in the 2700 Å system of chlorobenzene. Mol. Phys. 1970, 18, 101–111. [Google Scholar] [CrossRef]
- Spencer, J.R.; Pearl, J.C.; Segura, M.; Flasar, F.M.; Mamoutkine, A.; Romani, P.; Buratti, B.J.; Hendrix, A.R.; Spilker, L.J.; Lopes, R.M.C. Cassini Encounters Enceladus: Background and the Discovery of a South Polar Hot Spot. Science 2006, 311, 1401–1405. [Google Scholar] [CrossRef]
- Miller, K.E.; Eigenbrode, J.L.; Freissinet, C.; Glavin, D.P.; Kotrc, B.; Francois, P.; Summons, R.E. Potential precursor compounds for chlorohydrocarbons detected in Gale Crater, Mars, by the SAM instrument suite on the Curiosity Rover. J. Geophys. Res. Planets. 2016, 121, 296–308. [Google Scholar] [CrossRef]
- Klán, P.; Alena Ansorgová, A.; Del Favero, D.; Holoubek, I. Photochemistry of chlorobenzene in ice. Tetrahedron Lett. 2000, 41, 7785–7789. [Google Scholar] [CrossRef]
- Park, H.-R.; Yang, I.-J.; Kim, M.-S. Photolysis of chlorobenzene in aqueous solution. Bull. Korean Chem. Soc. 1998, 19, 1265–1268. [Google Scholar]
- Liu, P.-Y.; Zheng, M.-H.; Zhang, B.; Xu, X.-B. Mechanism of PCBs formation from the pyrolysis of chlorobenzenes. Chemosphere 2001, 43, 783–785. [Google Scholar] [CrossRef]
- Saudermann, W. Polychlorierte aromatische verbindungen als umweltgifte. Naturwissenschaften 1974, 61, 207–213. [Google Scholar] [CrossRef] [PubMed]
- Herbert, B.M.J.; Villa, S.; Halsall, C.J. Chemical interactions with snow: Understanding the behavior and fate of semi-volatile organic compounds in snow. Ecotoxicol. Environ. Saf. 2006, 63, 3–16. [Google Scholar] [CrossRef]
- Imhof, P.; Kleinermanns, K. Dispersed fluorescence spectra of chlorobenzene. Chem. Phys. 2001, 270, 227–236. [Google Scholar] [CrossRef]
- Kronenberger, A. Über die Absorption und Lumineszenz von Benzol und Benzolderivaten bei −259°. Z. Phys. 1930, 63, 494–532. [Google Scholar] [CrossRef]
- Sponer, H.; Wollman, S.H. Analysis of the near ultraviolet absorption spectrum of monochlorobenzene. J. Chem. Phys. 1941, 9, 816–825. [Google Scholar] [CrossRef]
- Postberg, F.; Khawaja, N.; Abel, B.; Choblet, G.; Glein, C.R.; Gudipati, M.S.; Henderson, B.L.; Hsu, H.W.; Kempf, S.; Klenner, F.; et al. Macromolecular organic compounds from the depths of Enceladus. Nature 2018, 558, 564–568. [Google Scholar] [CrossRef] [PubMed]
- Famá, M.; Loeffler, M.J.; Raut, U.; Baragiola, R.A. Radiation-induced amorphization of crystalline ice. Icarus 2010, 207, 314–319. [Google Scholar] [CrossRef]
- Varnes, E.S.; Jakosky, B.M. Lifetime of organic molecules at the surface of Europa. In Proceedings of the 30th Annual Lunar and Planetary Science Conference, Houston, TX, USA, 15–29 March 1999; Lunar and Planetary Institute: Houston, TX, USA, 1999; p. 1082. [Google Scholar]
- Johnke, B.; Menke, D.; Böske, J. WHO Revision of the toxic equivalency factors for dioxins and furans and its impact on the emission of waste incineration plants in Germany. In Newsletter of the WHO Collaborating Center for Air Quality Management and Air Pollution Control; German Environment Agency: Berlin, Germany, 2003; Volume 31, p. 5. [Google Scholar]
- Anders Borg, O.; Karlsson, D.; Isomaki-Krondal, M.; Davidsson, J.; Lunell, S. Predissociation of chlorobenzene, beyond the pseudo-diatomic model. Chem. Phys. Lett. 2008, 456, 123–126. [Google Scholar] [CrossRef]




| Benzene Transition 11 | ||||
| λ nm | σ cm−1 | Diff. cm−1 | Tentative assgs. cm−1 | Spectrosc. notation |
| 192.25 | 52,016 | 4591 | 521 + 4 × 923 + 2 × 238 | |
| 195.31 | 51,201 | 3776 | 521 + 3 × 923 + 2 × 238 | |
| 199.75 | 50,063 | 2638 | 521 + 2 × 923 + 2 × 148 | |
| 203.00 | 49,261 | 1836 | 521 + 923 + 2 × 238 | |
| 208.56 | 47,948 | 523 | 521 | |
| 210.86 | 47,425 | 0 | 0 | origin |
| Benzene Transition 11 | ||||
| λ nm | σ cm−1 | Diff. cm−1 | Tentative assgs. cm−1 | Spectrosc. notation |
| 226.88 | 44,076 | 6047 | 521 + 6 × 923 | |
| 230.01 | 43,477 | 5448 | 6 × 923 | |
| 232.76 | 42,963 | 4934 | 5 × 923 + 2 × 238 | |
| 236.32 | 42,315 | 4286 | 521 + 4 × 923 | |
| 238.76 | 41,883 | 3854 | 4 × 923 | |
| 241.76 | 41,363 | 3334 | 521 + 3 × 923 | |
| 244.51 | 40,897 | 2868 | 3 × 923 | |
| 247.20 | 40,453 | 2424 | 521 + 2 × 923 | |
| 250.02 | 39,997 | 1968 | 2 × 923 | |
| 253.14 | 39,503 | 1474 | 521 + 923 | |
| 256.14 | 39,041 | 1012 | 923 | |
| 259.14 | 38,589 | 560 | 521 | |
| 260.89 | 38,330 | 301 | 2 × 148 | |
| 262.96 | 38,029 | 0 | 0 | origin |
| 266.96 | 37,459 | −570 | −521 | |
| λ nm | σ cm−1 | Diff. cm−1 | Tentative assgs. cm−1 | Spectrosc. notation |
| Chlorobenzene Transition 1A1 1A1 | ||||
| 192.85 | 51,854 | 6880 | 378 + 7 × 931 | |
| 196.25 | 50,955 | 5981 | 378 + 6 × 931 | |
| 200.10 | 49,975 | 5001 | 378 + 5 × 931 | |
| 203.70 | 49,092 | 4118 | 378 + 4 × 931 | |
| 207.80 | 48,123 | 3149 | 378 + 3 × 931 | |
| 212.90 | 46,970 | 1996 | 378 + 2 × 931 | |
| 215.75 | 46,350 | 1376 | 378 + 931 | |
| 219.55 | 45,548 | 574 | 2 × 287 | |
| 222.35 | 44,974 | 0 | 0 | origin |
| Chlorobenzene Transition 1B2 1A1 | ||||
| λ nm | σ cm−1 | Diff. cm−1 | Tentative assgs. cm−1 | Spectrosc. notation |
| 233.40 | 42,845 | 6848 | 378 + 7 × 931 | |
| 236.35 | 42,310 | 6313 | 2 × 378 + 6 × 931 | |
| 239.95 | 41,675 | 5678 | 1065 + 5 × 931 | |
| 242.20 | 41,288 | 5291 | 1564 + 4 × 931 | |
| 252.90 | 39,541 | 3544 | 2 × 378 + 3 × 931 | |
| 257.15 | 38,888 | 2891 | 1065 + 2 × 931 | |
| 259.75 | 38,499 | 2502 | 671 + 2 × 931 | |
| 263.05 | 38,016 | 2019 | 1065 + 931 | |
| 266.30 | 37,552 | 1555 | 521 + 1065 | |
| 270.80 | 36,928 | 931 | 931 | |
| 273.21 | 36,603 | 606 | 521 | |
| 277.82 | 35,997 | 0 | 0 | origin |
| Peak Position | Shift | Integrated Absorbance | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Compound | Band | 292 K nm | 78 K nm | Δλ nm | 292 (K) | 260 (K) | 230 (K) | 200 (K) | 170 (K) | 140 (K) | 110 (K) | 78 (K) |
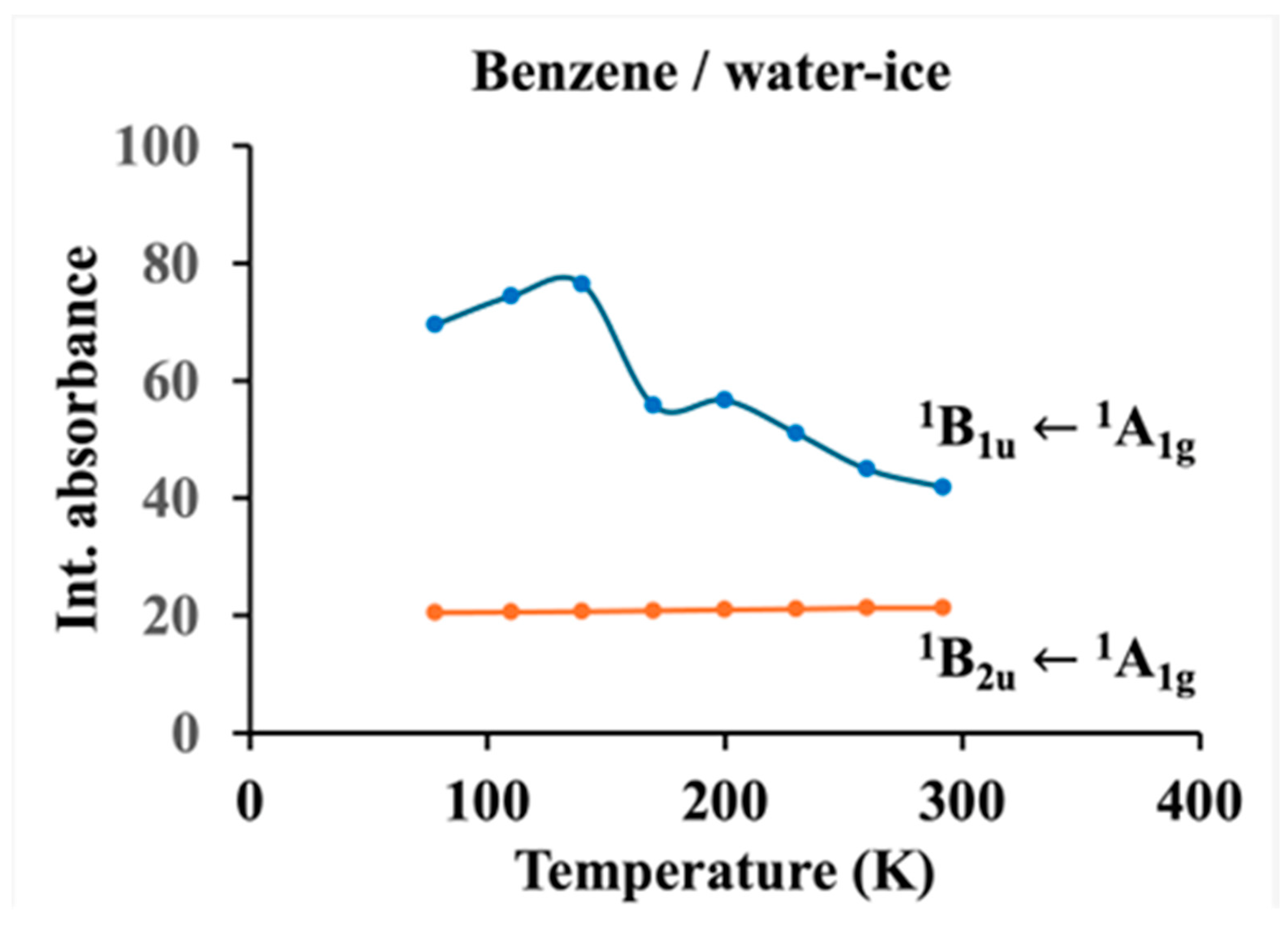
| C6H6 | 1B2u 1A1g | 253.8 | 253.1 | 0.7 | 41.84 | 44.91 | 51.06 | 56.67 | 55.75 | 76.42 | 74.37 | 69.55 |
| 1B1u1A1g | 206.1 | 203.8 | 2.3 | 21.26 | 21.26 | 21.01 | 20.92 | 20.78 | 20.62 | 20.50 | 20.42 | |
| C6H5-Cl | 1B2 1A1 | 263.4 | 266.3 | -2.9 | 79.98 | 79.61 | 78.87 | 78.54 | 74.31 | 70.75 | 68.54 | 65.23 |
| 1A1 1A1 | 199.7 | 203.7 | -4.0 | 51.98 | 51.88 | 51.77 | 51.59 | 51.44 | 51.30 | 51.18 | 51.08 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Sunuwar, S.; Manzanares, C.E. Ultraviolet Absorption Spectra of Benzene and Chlorobenzene in Water-Ice Solutions at Temperatures Between 78 K and 273 K. Chemistry 2026, 8, 9. https://doi.org/10.3390/chemistry8010009
Sunuwar S, Manzanares CE. Ultraviolet Absorption Spectra of Benzene and Chlorobenzene in Water-Ice Solutions at Temperatures Between 78 K and 273 K. Chemistry. 2026; 8(1):9. https://doi.org/10.3390/chemistry8010009
Chicago/Turabian StyleSunuwar, Suresh, and Carlos E. Manzanares. 2026. "Ultraviolet Absorption Spectra of Benzene and Chlorobenzene in Water-Ice Solutions at Temperatures Between 78 K and 273 K" Chemistry 8, no. 1: 9. https://doi.org/10.3390/chemistry8010009
APA StyleSunuwar, S., & Manzanares, C. E. (2026). Ultraviolet Absorption Spectra of Benzene and Chlorobenzene in Water-Ice Solutions at Temperatures Between 78 K and 273 K. Chemistry, 8(1), 9. https://doi.org/10.3390/chemistry8010009






