Polymer-Drug Conjugates as Nanotheranostic Agents
Abstract
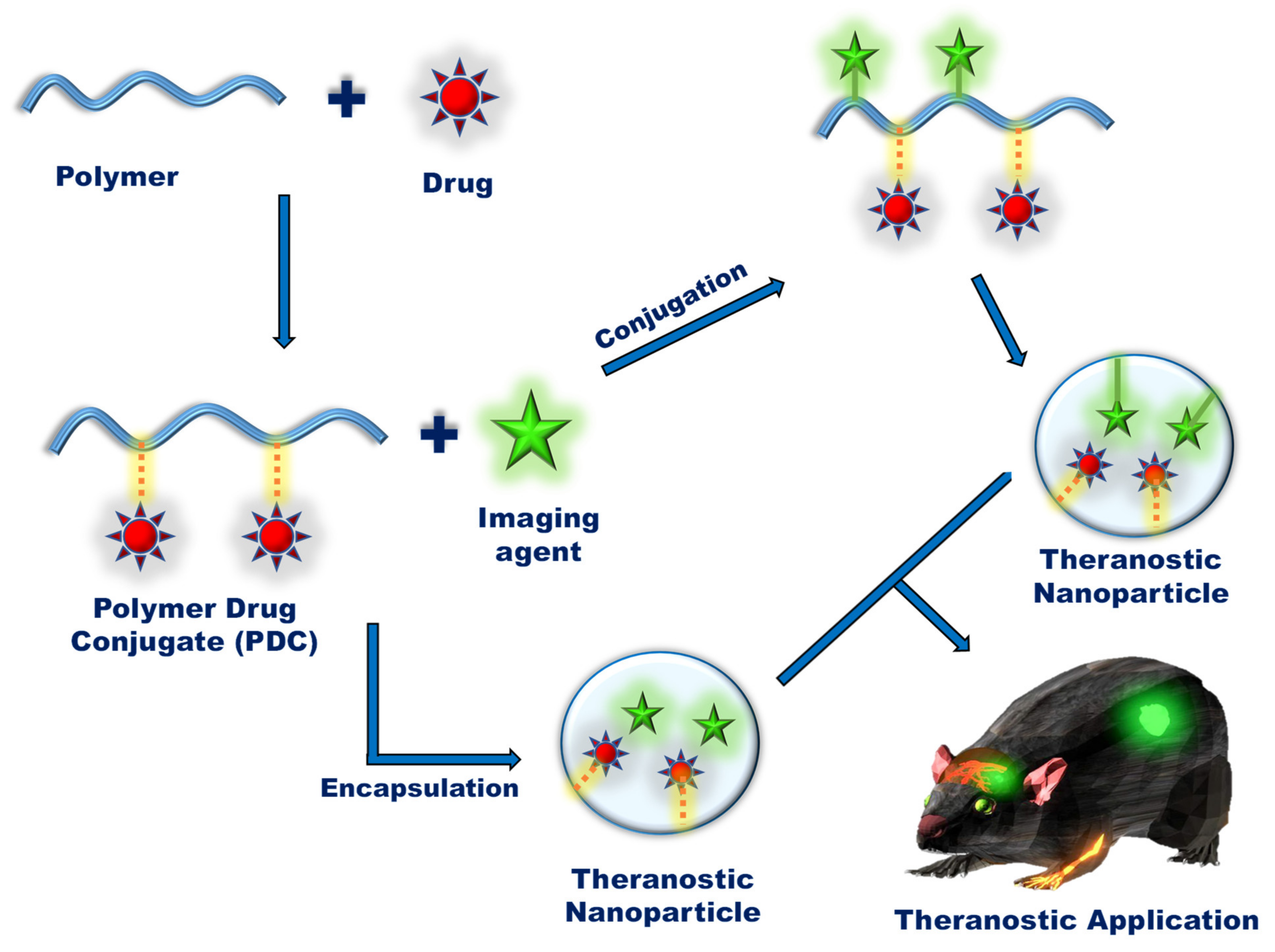
1. Introduction
2. Polymers Used in Nanotheranostics
2.1. Synthetic Polymers in Nanotheranostic Agent Design
2.1.1. Poly N-(2-Hydroxypropyl)-methacrylamide
2.1.2. Polyethylene Glycol
2.1.3. Polylactic Acid
2.1.4. Poly(Lactic-co-glycolic Acid)
2.1.5. Polyphosphazene
2.1.6. Polyphosphoesters
2.1.7. Polyglycerol
2.1.8. Polyglutamic Acid
2.2. Natural Polymers in Nanotheranostic Agent Design
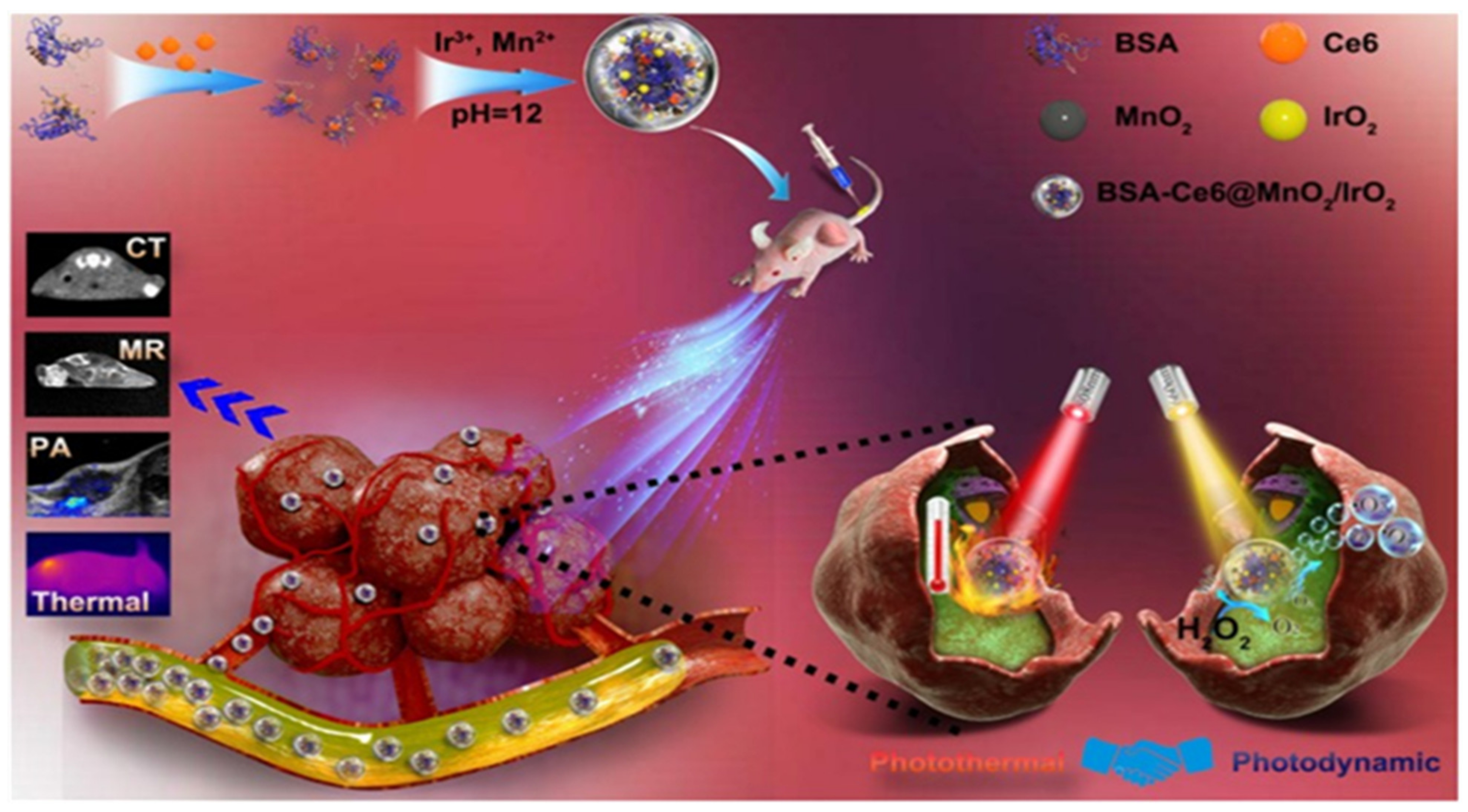
2.2.1. Albumin
2.2.2. Gelatin
2.2.3. Alginate
2.2.4. Chitosan
2.2.5. Hyaluronic Acid
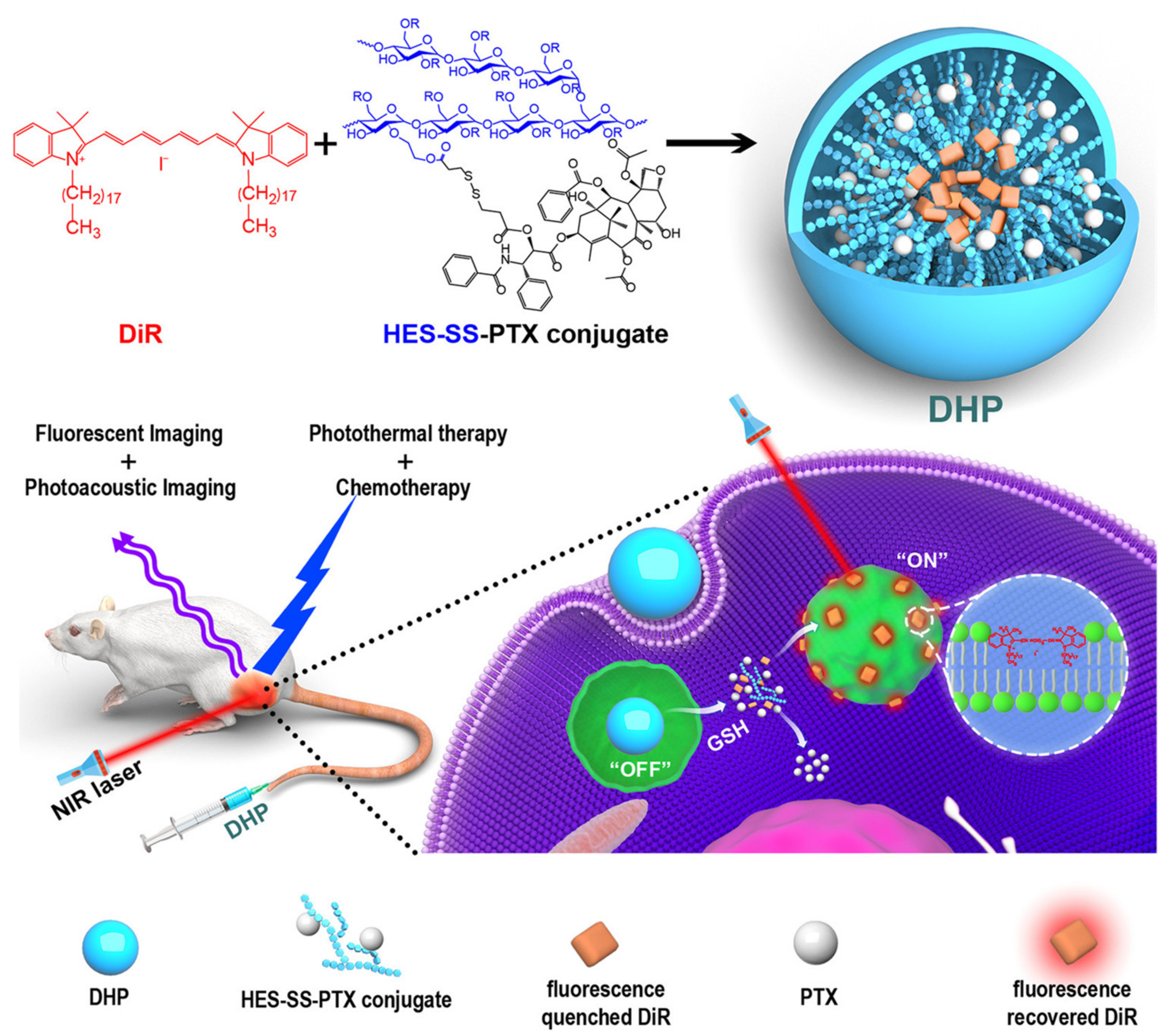
2.2.6. Hydroxyethyl Starch
3. Outlook
Author Contributions
Funding
Conflicts of Interest
References
- Qian, C.G.; Chen, Y.L.; Feng, P.J.; Xiao, X.Z.; Dong, M.; Yu, J.C.; Hu, Q.Y.; Shen, Q.D.; Gu, Z. Conjugated polymer nanomaterials for theranostics. Acta Pharmacol. Sin. 2017, 38, 764–781. [Google Scholar] [CrossRef] [PubMed]
- Larson, N.; Ghandehari, H. Polymeric conjugates for drug delivery. Chem. Mater. 2012, 24, 840–853. [Google Scholar] [CrossRef] [PubMed]
- Chang, M.; Zhang, F.; Wei, T.; Zuo, T.; Guan, Y.; Lin, G.; Shao, W. Smart linkers in polymer-drug conjugates for tumor-targeted delivery. J. Drug Target. 2016, 24, 475–491. [Google Scholar] [CrossRef] [PubMed]
- Das, S.S.; Bharadwaj, P.; Bilal, M.; Barani, M.; Rahdar, A.; Taboada, P.; Bungau, S.; Kyzas, G.Z. Stimuli-responsive polymeric nanocarriers for drug delivery, imaging, and theragnosis. Polymers 2020, 12, 1397. [Google Scholar] [CrossRef]
- Feng, Q.; Tong, R. Anticancer nanoparticulate polymer-drug conjugate. Bioeng. Transl. Med. 2016, 1, 277–296. [Google Scholar] [CrossRef]
- Alven, S.; Nqoro, X.; Buyana, B.; Aderibigbe, B.A. Polymer-Drug Conjugate, a Potential Therapeutic to Combat Breast and Lung Cancer. Pharmaceutics 2020, 12, 406. [Google Scholar] [CrossRef]
- Thakor, P.; Bhavana, V.; Sharma, R.; Srivastava, S.; Bala Singh, S.; Kumar Mehra, N. Polymer–drug conjugates: Recent advances and future perspectives. Drug Discov. Today 2020, 25, 1718–1726. [Google Scholar] [CrossRef]
- Ekladious, I.; Colson, Y.L.; Grinstaff, M.W. Polymer–drug conjugate therapeutics: Advances, insights and prospects. Nat. Rev. Drug Discov. 2019, 18, 273–294. [Google Scholar] [CrossRef]
- Chawla, S.P.; Goel, S.; Chow, W.; Braiteh, F.; Singh, A.S.; Olson, J.E.G.; Osada, A.; Bobe, I.; Riedel, R.F. A Phase 1b Dose Escalation Trial of NC-6300 (Nanoparticle Epirubicin) in Patients with Advanced Solid Tumors or Advanced, Metastatic, or Unresectable Soft-tissue Sarcoma. Clin. Cancer Res. 2020, 26, 4225–4232. [Google Scholar] [CrossRef] [PubMed]
- Peng, H.; Liu, X.; Wang, G.; Li, M.; Bratlie, K.M.; Cochran, E.; Wang, Q. Polymeric multifunctional nanomaterials for theranostics. J. Mater. Chem. B 2015, 3, 6856–6870. [Google Scholar] [CrossRef]
- Zhao, J.; Chen, J.; Ma, S.; Liu, Q.; Huang, L.; Chen, X.; Lou, K.; Wang, W. Recent developments in multimodality fluorescence imaging probes. Acta Pharm. Sin. B 2018, 8, 320–338. [Google Scholar] [CrossRef]
- Wallyn, J.; Anton, N.; Akram, S.; Vandamme, T.F. Biomedical Imaging: Principles, Technologies, Clinical Aspects, Contrast Agents, Limitations and Future Trends in Nanomedicines. Pharm. Res. 2019, 36, 1–31. [Google Scholar] [CrossRef]
- Denk, W.; Strickler, J.H.; Webb, W.W. Two-photon laser scanning fluorescence microscopy. Science 1990, 248, 73–76. [Google Scholar] [CrossRef] [PubMed]
- Zou, C.; Wu, B.; Dong, Y.; Song, Z.; Zhao, Y.; Ni, X.; Yang, Y.; Liu, Z. Biomedical photoacoustics: Fundamentals, instrumentation and perspectives on nanomedicine. Int. J. Nanomed. 2016, 12, 179–195. [Google Scholar] [CrossRef]
- Krasia-Christoforou, T.; Georgiou, T.K. Polymeric theranostics: Using polymer-based systems for simultaneous imaging and therapy. J. Mater. Chem. B 2013, 1, 3002–3025. [Google Scholar] [CrossRef]
- Duncan, R.; Vicent, M.J. Polymer therapeutics-prospects for 21st century: The end of the beginning. Adv. Drug Deliv. Rev. 2013, 65, 60–70. [Google Scholar] [CrossRef] [PubMed]
- Atkinson, S.P.; Andreu, Z.; Vicent, M.J. Polymer therapeutics: Biomarkers and new approaches for personalized cancer treatment. J. Pers. Med. 2018, 8, 6. [Google Scholar] [CrossRef]
- Lee, H.J.; Ponta, A.; Bae, Y. Polymer nanoassemblies for cancer treatment and imaging. Ther. Deliv. 2010, 1, 803–817. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Jiang, W.; Shen, Y.; Li, H.; Sun, C.; Ouahab, A.; Tu, J. A Poly (g,L-glutamic acid)-citric acid based nanoconjugate for cisplatin delivery. Biomaterials 2012, 33, 7182–7193. [Google Scholar] [CrossRef] [PubMed]
- Bansal, K.; Sasso, L.; Makwana, H.; Awwad, S.; Brocchini, S.; Alexander, C. Nanopharmacy: Exploratory Methods for Polymeric Materials. In Pharmaceutical Nanotechnology: Innovation and Production; Wiley-VCH Verlag GmbH & Co. KGaA: Weinheim, Germany, 2016; pp. 231–270. [Google Scholar]
- Bhatia, S.; Bhatia, S. Natural Polymers vs Synthetic Polymer. In Natural Polymer Drug Delivery Systems; Springer International Publishing: Cham, Switzerland, 2016; pp. 95–118. [Google Scholar]
- Duncan, R.; Vicent, M.J. Do HPMA copolymer conjugates have a future as clinically useful nanomedicines? A critical overview of current status and future opportunities. Adv. Drug Deliv. Rev. 2010, 62, 272–282. [Google Scholar] [CrossRef] [PubMed]
- Knop, K.; Hoogenboom, R.; Fischer, D.; Schubert, U.S. Poly(ethylene glycol) in Drug Delivery: Pros and Cons as Well as Potential Alternatives. Angew. Chemie Int. Ed. 2010, 49, 6288–6308. [Google Scholar] [CrossRef] [PubMed]
- Shiraishi, K.; Yokoyama, M. Toxicity and immunogenicity concerns related to PEGylated-micelle carrier systems: A review. Sci. Technol. Adv. Mater. 2019, 20, 324–336. [Google Scholar] [CrossRef]
- Casalini, T.; Rossi, F.; Castrovinci, A.; Perale, G. A Perspective on Polylactic Acid-Based Polymers Use for Nanoparticles Synthesis and Applications. Front. Bioeng. Biotechnol. 2019, 7, 259. [Google Scholar] [CrossRef]
- Bansal, K.K.; Kakde, D.; Purdie, L.; Irvine, D.J.; Howdle, S.M.; Mantovani, G.; Alexander, C. New biomaterials from renewable resources-amphiphilic block copolymers from δ-decalactone. Polym. Chem. 2015, 6, 7196–7210. [Google Scholar] [CrossRef]
- Sharma, S.; Parmar, A.; Kori, S.; Sandhir, R. PLGA-based nanoparticles: A new paradigm in biomedical applications. Trends Anal. Chem. 2016, 80, 30–40. [Google Scholar] [CrossRef]
- Bansal, K.K.; Rosenholm, J.M. Synthetic polymers from renewable feedstocks: An alternative to fossil-based materials in biomedical applications. Ther. Deliv. 2020, 11, 297–300. [Google Scholar] [CrossRef]
- Ullah, R.S.; Wang, L.; Yu, H.; Abbasi, N.M.; Akram, M.; Ul-Abdin, Z.; Saleem, M.; Haroon, M.; Khan, R.U. Synthesis of polyphosphazenes with different side groups and various tactics for drug delivery. RSC Adv. 2017, 7, 23363–23391. [Google Scholar] [CrossRef]
- Rothemund, S.; Teasdale, I. Preparation of polyphosphazenes: A tutorial review. Chem. Soc. Rev. 2016, 45, 5200–5215. [Google Scholar] [CrossRef]
- Pelosi, C.; Tinè, M.R.; Wurm, F.R. Main-chain water-soluble polyphosphoesters: Multifunctional polymers as degradable PEG-alternatives for biomedical applications. Eur. Polym. J. 2020, 141, 110079. [Google Scholar] [CrossRef]
- Steinbach, T.; Wurm, F.R. Poly(phosphoester)s: A New Platform for Degradable Polymers. Angew. Chemie Int. Ed. 2015, 54, 6098–6108. [Google Scholar] [CrossRef]
- Jafari, M.; Abolmaali, S.S.; Najafi, H.; Tamaddon, A.M. Hyperbranched polyglycerol nanostructures for anti-biofouling, multifunctional drug delivery, bioimaging and theranostic applications. Int. J. Pharm. 2020, 576, 118959. [Google Scholar] [CrossRef] [PubMed]
- Hu, M.; Chen, M.; Li, G.; Pang, Y.; Wang, D.; Wu, J.; Qiu, F.; Zhu, X.; Sun, J. Biodegradable hyperbranched polyglycerol with ester linkages for drug delivery. Biomacromolecules 2012, 13, 3552–3561. [Google Scholar] [CrossRef]
- Bajaj, I.; Singhal, R. Poly (glutamic acid)—An emerging biopolymer of commercial interest. Bioresour. Technol. 2011, 102, 5551–5561. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.; Taylor, W.D. Controlled Synthesis of Polyglutamic Acid. European Patent WO2011075483A1, 23 June 2011. [Google Scholar]
- Wall, A.; Nicholls, K.; Caspersen, M.B.; Skrivergaard, S.; Howard, K.A.; Karu, K.; Chudasama, V.; Baker, J.R. Optimised approach to albumin-drug conjugates using monobromomaleimide-C-2 linkers. Org. Biomol. Chem. 2019, 17, 7870–7873. [Google Scholar] [CrossRef]
- Karimi, M.; Bahrami, S.; Ravari, S.B.; Zangabad, P.S.; Mirshekari, H.; Bozorgomid, M.; Shahreza, S.; Sori, M.; Hamblin, M.R. Albumin nanostructures as advanced drug delivery systems. Expert Opin. Drug Deliv. 2016, 13, 1609–1623. [Google Scholar] [CrossRef]
- Elzoghby, A.O. Gelatin-based nanoparticles as drug and gene delivery systems: Reviewing three decades of research. J. Control. Release 2013, 172, 1075–1091. [Google Scholar] [CrossRef]
- Szekalska, M.; Puciłowska, A.; Szymańska, E.; Ciosek, P.; Winnicka, K. Alginate: Current Use and Future Perspectives in Pharmaceutical and Biomedical Applications. Int. J. Polym. Sci. 2016, 2016, 7697031. [Google Scholar] [CrossRef]
- Bellich, B.; D’Agostino, I.; Semeraro, S.; Gamini, A.; Cesàro, A. “The good, the bad and the ugly” of chitosans. Mar. Drugs 2016, 14, 99. [Google Scholar] [CrossRef]
- Elgadir, M.A.; Uddin, M.S.; Ferdosh, S.; Adam, A.; Chowdhury, A.J.K.; Sarker, M.Z.I. Impact of chitosan composites and chitosan nanoparticle composites on various drug delivery systems: A review. J. Food Drug Anal. 2015, 23, 619–629. [Google Scholar] [CrossRef]
- Huang, G.; Huang, H. Application of hyaluronic acid as carriers in drug delivery. Drug Deliv. 2018, 25, 766–772. [Google Scholar] [CrossRef]
- Fallacara, A.; Baldini, E.; Manfredini, S.; Vertuani, S. Hyaluronic Acid in the Third Millennium. Polymers 2018, 10, 701. [Google Scholar] [CrossRef]
- Tan, R.; Wan, Y.; Yang, X. Hydroxyethyl starch and its derivatives as nanocarriers for delivery of diagnostic and therapeutic agents towards cancers. Biomater. Transl. 2020, 1, 46. [Google Scholar]
- Zhu, X.; Anquillare, E.L.B.; Farokhzad, O.C.; Shi, J. Polymer- and Protein-Based Nanotechnologies for Cancer Theranostics. In Cancer Theranostics; Elsevier Inc.: Amsterdam, The Netherlands, 2014; pp. 419–436. [Google Scholar]
- Koziolová, E.; Goel, S.; Chytil, P.; Janoušková, O.; Barnhart, T.E.; Cai, W.; Etrych, T. A tumor-targeted polymer theranostics platform for positron emission tomography and fluorescence imaging. Nanoscale 2017, 9, 10906–10918. [Google Scholar] [CrossRef]
- Zhang, Y.; Hong, H.; Cai, W. PET Tracers Based on Zirconium-89. Curr. Radiopharm. 2011, 4, 131–139. [Google Scholar] [CrossRef]
- Yang, J.; Zhang, R.; Radford, D.C.; Kopeček, J. FRET-trackable biodegradable HPMA copolymer-epirubicin conjugates for ovarian carcinoma therapy. J. Control. Release 2015, 218, 36–44. [Google Scholar] [CrossRef]
- Fang, J.; Šubr, V.; Islam, W.; Hackbarth, S.; Islam, R.; Etrych, T.; Ulbrich, K.; Maeda, H. N-(2-hydroxypropyl)methacrylamide polymer conjugated pyropheophorbide-a, a promising tumor-targeted theranostic probe for photodynamic therapy and imaging. Eur. J. Pharm. Biopharm. 2018, 130, 165–176. [Google Scholar] [CrossRef]
- Cai, H.; Wang, X.; Zhang, H.; Sun, L.; Pan, D.; Gong, Q.; Gu, Z.; Luo, K. Enzyme-sensitive biodegradable and multifunctional polymeric conjugate as theranostic nanomedicine. Appl. Mater. Today 2018, 11, 207–218. [Google Scholar] [CrossRef]
- Hou, W.; Xia, F.; Alves, C.S.; Qian, X.; Yang, Y.; Cui, D. MMP2-Targeting and Redox-Responsive PEGylated Chlorin e6 Nanoparticles for Cancer Near-Infrared Imaging and Photodynamic Therapy. ACS Appl. Mater. Interfaces 2016, 8, 1447–1457. [Google Scholar] [CrossRef]
- Kumar, A.; Mastren, T.; Wang, B.; Hsieh, J.T.; Hao, G.; Sun, X. Design of a Small-Molecule Drug Conjugate for Prostate Cancer Targeted Theranostics. Bioconjug. Chem. 2016, 27, 1681–1689. [Google Scholar] [CrossRef]
- Qi, R.; Wang, Y.; Bruno, P.M.; Xiao, H.; Yingjie, Y.; Li, T.; Lauffer, S.; Wei, W.; Chen, Q.; Kang, X.; et al. Nanoparticle conjugates of a highly potent toxin enhance safety and circumvent platinum resistance in ovarian cancer. Nat. Commun. 2017, 8, 2166. [Google Scholar] [CrossRef] [PubMed]
- Hu, X.; Wang, R.; Yue, J.; Liu, S.; Xie, Z.; Jing, X. Targeting and anti-tumor effect of folic acid-labeled polymer—Doxorubicin conjugates with pH-sensitive hydrazone linker. J. Mater. Chem. 2012, 22, 13303–13310. [Google Scholar] [CrossRef]
- Chatterjee, M.; Maity, R.; Das, S.; Mahata, N.; Basu, B.; Chanda, N. Electrospray-based synthesis of fluorescent poly(d,l-lactide-co-glycolide) nanoparticles for the efficient delivery of an anticancer drug and self-monitoring its effect in drug-resistant breast cancer cells. Mater. Adv. 2020, 1, 3033–3048. [Google Scholar] [CrossRef]
- Jun, Y.J.; Park, J.H.; Avaji, P.G.; Park, K.S.; Lee, K.E.; Lee, H.J.; Sohn, Y.S. Design of theranostic nanomedicine (Ii): Synthesis and physicochemical properties of a biocompatible polyphosphazene–docetaxel conjugate. Int. J. Nanomedicine 2017, 12, 5373–5386. [Google Scholar] [CrossRef]
- Pei, P.; Sun, C.; Tao, W.; Li, J.; Yang, X.; Wang, J. ROS-sensitive thioketal-linked polyphosphoester-doxorubicin conjugate for precise phototriggered locoregional chemotherapy. Biomaterials 2019, 188, 74–82. [Google Scholar] [CrossRef]
- Nagel, G.; Tschiche, H.R.; Wedepohl, S.; Calderón, M. Modular approach for theranostic polymer conjugates with activatable fluorescence: Impact of linker design on the stimuli-induced release of doxorubicin. J. Control. Release 2018, 285, 200–211. [Google Scholar] [CrossRef]
- Dai, X.-H.; Yang, W.-H.; Wu, C.; Jin, H.; Chang, D.-D.; Dai, Y.-R.; Pan, J.-M.; Yan, Y.-S. Synthesis and Characterization of Star-Shaped Porphyrin–cored Poly (Glutamic Acid) Conjugates as Highly Efficient Photosensitizers. J. Photopolym. Sci. Technol. 2016, 29, 823–832. [Google Scholar] [CrossRef][Green Version]
- Panwar, R.; Bhattarai, P.; Patil, V.; Gada, K.; Majewski, S.; Khaw, B.A. Imaging doxorubicin and polymer-drug conjugates of doxorubicin-induced cardiotoxicity with bispecific anti-myosin-anti-DTPA antibody and Tc-99m-labeled polymers. J. Nucl. Cardiol. 2018, 26, 1327–1344. [Google Scholar] [CrossRef] [PubMed]
- Wu, J.; Williams, G.R.; Niu, S.; Yang, Y.; Li, Y.; Zhang, X.; Zhu, L.M. Biomineralized bimetallic oxide nanotheranostics for multimodal imaging-guided combination therapy. Theranostics 2020, 10, 841–855. [Google Scholar] [CrossRef]
- Hafner, S.; Raabe, M.; Wu, Y.; Wang, T.; Zuo, Z.; Rasche, V.; Syrovets, T.; Weil, T.; Simmet, T. High-Contrast Magnetic Resonance Imaging and Efficient Delivery of an Albumin Nanotheranostic in Triple-Negative Breast Cancer Xenografts. Adv. Ther. 2019, 2, 1900084. [Google Scholar] [CrossRef]
- Kirar, S.; Thakur, N.S.; Laha, J.K.; Banerjee, U.C. Porphyrin Functionalized Gelatin Nanoparticle-Based Biodegradable Phototheranostics: Potential Tools for Antimicrobial Photodynamic Therapy. Am. Chem. Soc. 2019, 2, 4202–4212. [Google Scholar] [CrossRef]
- Deshpande, N.; Ramesh, A.; Nandi, D.; Nguyen, A.; Brouillard, A.; Kulkarni, A. Nanotheranostics Supramolecular Polysaccharide Nanotheranostics that Inhibit Cancer Cells Growth and Monitor Targeted Therapy Response. Nanotheranostics 2020, 4, 156–172. [Google Scholar] [CrossRef]
- Jia, X.; Pei, M.; Zhao, X.; Tian, K.; Zhou, T.-T.; Liu, P. PEGylated oxidized alginate-DOX prodrug conjugate nanoparticles crosslinked with fluorescent carbon dots for tumor theranostics. ACS Biomater. Sci. Eng. 2016, 2, 1641–1648. [Google Scholar] [CrossRef]
- Wu, B.; Zhao, N. A Targeted Nanoprobe Based on Carbon Nanotubes-Natural Biopolymer Chitosan Composites. Nanomaterials 2016, 6, 216. [Google Scholar] [CrossRef]
- Surendran, P.S.; Thomas, R.G.; Moon, M.J.; Park, R.; Lee, J.H.; Jeong, Y.Y. A bilirubin-conjugated chitosan nanotheranostics system as a platform for reactive oxygen species stimuli-responsive hepatic fibrosis therapy. Acta Biomater. 2020, 116, 356–367. [Google Scholar] [CrossRef]
- Hu, R.; Zheng, H.; Cao, J.; Davoudi, Z.; Wang, Q. Synthesis and in vitro characterization of carboxymethyl chitosan-CBA-Doxorubicin conjugate nanoparticles as pH-Sensitive drug delivery systems. J. Biomed. Nanotechnol. 2017, 13, 1097–1105. [Google Scholar] [CrossRef]
- Dubey, R.D.; Klippstein, R.; Wang, J.T.-W.; Hodgins, N.; Mei, K.-C.; Sosabowski, J.; Hider, R.C.; Abbate, V.; Gupta, P.N.; Al-jamal, K.T. Novel Hyaluronic Acid Conjugates for Dual Nuclear Imaging and Therapy in CD44-Expressing Tumors in Mice In Vivo. Nanotheranostics 2017, 1, 49–79. [Google Scholar] [CrossRef]
- Yang, Y.; Jing, L.; Li, X.; Lin, L.; Yue, X.; Dai, Z. Hyaluronic Acid Conjugated Magnetic Prussian Blue@Quantum Dot Nanoparticles for Cancer Theranostics. Theranostics 2017, 7, 466–481. [Google Scholar] [CrossRef] [PubMed]
- Li, Y.; Wu, Y.; Chen, J.; Wan, J.; Xiao, C.; Guan, J.; Song, X.; Li, S.; Zhang, M.; Cui, H.; et al. A Simple Glutathione-Responsive Turn-On Theranostic Nanoparticle for Dual-Modal Imaging and Chemo-Photothermal Combination Therapy. Nano Lett. 2019, 19, 5806–5817. [Google Scholar] [CrossRef]
- Yu, C.; Liu, C.; Wang, S.; Li, Z.; Hu, H.; Wan, Y.; Yang, X. Hydroxyethyl starch-based nanoparticles featured with redox-sensitivity and chemo-photothermal therapy for synergized tumor eradication. Cancers 2019, 11, 207. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Nakamura, H.; Maeda, H. The EPR effect: Unique features of tumor blood vessels for drug delivery, factors involved, and limitations and augmentation of the effect. Adv. Drug Deliv. Rev. 2011, 63, 136–151. [Google Scholar] [CrossRef] [PubMed]
- Shi, Y.; van der Meel, R.; Chen, X.; Lammers, T. The EPR effect and beyond: Strategies to improve tumor targeting and cancer nanomedicine treatment efficacy. Theranostics 2020, 10, 7921–7924. [Google Scholar] [CrossRef]
- Banerjee, S.S.; Aher, N.; Patil, R.; Khandare, J. Poly(ethylene glycol)-Prodrug Conjugates: Concept, Design, and Applications. J. Drug Deliv. 2012, 2012, 103973. [Google Scholar] [CrossRef]
- Gou, P.; Liu, W.; Mao, W.; Tang, J.; Shen, Y.; Sui, M. Self-assembling doxorubicin prodrug forming nanoparticles for cancer chemotherapy: Synthesis and anticancer study in vitro and in vivo. J. Mater. Chem. B 2013, 1, 284–292. [Google Scholar] [CrossRef]
- Zhu, L.; Wang, T.; Perche, F.; Taigind, A.; Torchilin, V.P. Enhanced anticancer activity of nanopreparation containing an MMP2-sensitive PEG-drug conjugate and cell-penetrating moiety. Proc. Natl. Acad. Sci. USA 2013, 110, 17047–17052. [Google Scholar] [CrossRef]
- Rezvantalab, S.; Drude, N.I.; Moraveji, M.K.; Güvener, N.; Koons, E.K.; Shi, Y.; Lammers, T.; Kiessling, F. PLGA-based nanoparticles in cancer treatment. Front. Pharmacol. 2018, 9, 1–19. [Google Scholar] [CrossRef]
- Elzeny, H.; Zhang, F.; Ali, E.N.; Fathi, H.A.; Zhang, S.; Li, R.; El-Mokhtar, M.A.; Hamad, M.A.; Wooley, K.L.; Elsabahy, M. Polyphosphoester nanoparticles as biodegradable platform for delivery of multiple drugs and siRNA. Drug Des. Devel. Ther. 2017, 11, 483–496. [Google Scholar] [CrossRef]
- Frey, H.; Haag, R. Dendritic polyglycerol: A new versatile biocompatible material. Rev. Mol. Biotechnol. 2002, 90, 257–267. [Google Scholar] [CrossRef]
- Li, C. Poly(L-glutamic acid)–anticancer drug conjugates. Adv. Drug Deliv. Rev. 2002, 54, 695–713. [Google Scholar] [CrossRef]
- Hale, J.P.; Lewis, I.J. Anthracyclines: Cardiotoxicity and its prevention. Arch. Dis. Child. 1994, 71, 457–462. [Google Scholar] [CrossRef]
- Rahimizadeh, P.; Yang, S.; Lim, S.I. Albumin: An Emerging Opportunity in Drug Delivery. Biotechnol. Bioprocess Eng. 2020, 25, 985–995. [Google Scholar] [CrossRef]
- Zhang, Y.; Wan, Y.; Chen, Y.; Blum, N.T.; Lin, J.; Huang, P. Ultrasound-Enhanced Chemo-Photodynamic Combination Therapy by Using Albumin “Nanoglue”-Based Nanotheranostics. Am. Chem. Soc. Nano 2020, 14, 5560–5569. [Google Scholar] [CrossRef]
- Zhou, Z.; Song, J.; Nie, L.; Xiaoyuan, C. Reactive oxygen species generating systems meeting challenges of photodynamic cancer therapy. Chem. Soc. Rev. 2016, 45, 6597–6626. [Google Scholar] [CrossRef] [PubMed]
- Ferlay, J.; Soerjomataram, I.; Dikshit, R.; Eser, S.; Mathers, C.; Rebelo, M.; Parkin, D.M.; Forman, D.; Bray, F. Cancer incidence and mortality worldwide: Sources, methods and major patterns in GLOBOCAN 2012. Int. J. Cancer 2015, 136, E359–E386. [Google Scholar] [CrossRef] [PubMed]
- Collignon, J.; Lousberg, L.; Schroeder, H.; Jerusalem, G. Triple-negative breast cancer: Treatment challenges and solutions. Breast Cancer Targets Ther. 2016, 8, 93–107. [Google Scholar]
- Foox, M.; Zilberman, M. Drug delivery from gelatin-based systems. Expert Opin. Drug Deliv. 2015, 12, 1547–1563. [Google Scholar] [CrossRef] [PubMed]
- Liu, Y.; Qin, R.; Zaat, S.A.J.; Breukink, E.; Heger, M. Antibacterial photodynamic therapy: Overview of a promising approach to fight antibiotic-resistant bacterial infections. J. Clin. Transl. Res. 2015, 1, 140–167. [Google Scholar]
- Guarino, V.; Caputo, T.; Altobelli, R.; Ambrosio, L. Degradation properties and metabolic activity of alginate and chitosan polyelectrolytes for drug delivery and tissue engineering applications. AIMS Mater. Sci. 2015, 2, 497–502. [Google Scholar] [CrossRef]
- Ravi Kumar, M.N.V.; Muzzarelli, R.A.A.; Muzzarelli, C.; Sashiwa, H.; Domb, A.J. Chitosan Chemistry and Pharmaceutical Perspectives. Chem. Rev. 2004, 104, 6017–6084. [Google Scholar] [CrossRef]
- Gabano, E.; do Queantal, L.; Perin, E.; Silva, F.; Raposinho, P.; António, P.; Ravera, M. Pt (IV)/Re (I) Chitosan Conjugates as a Flexible Platform for the Transport of Therapeutic and/or Diagnostic Anticancer Agents. Inorganics 2018, 6, 4. [Google Scholar] [CrossRef]
- Dosio, F.; Arpicco, S.; Stella, B.; Fattal, E. Hyaluronic acid for anticancer drug and nucleic acid delivery. Adv. Drug Deliv. Rev. 2016, 97, 204–236. [Google Scholar] [CrossRef]
- Reid, J.M.; Qu, W.; Safgren, S.L.; Ames, M.M.; Krailo, M.D.; Seibel, N.L.; Kuttesch, J.; Holcenberg, J. Phase I Trial and Pharmacokinetics of Gemcitabine in Children With Advanced Solid Tumors. J. Clin. Oncol. 2004, 22, 2445–2451. [Google Scholar] [CrossRef] [PubMed]
- Sleightholm, R.; Yang, B.; Yu, F.; Xie, Y.; Oupický, D. Chloroquine-Modified Hydroxyethyl Starch as a Polymeric Drug for Cancer Therapy. Biomacromolecules 2018, 18, 2247–2257. [Google Scholar] [CrossRef] [PubMed]
- Gulin-Sarfaz, T.; Pryazhnikov, E.; Zhang, J.; Rosenholm, J.M. Chemical and photonic interactions in vitro and in vivo between fluorescent tracer and nanoparticle-based scavenger for enhanced molecular imaging. Mater. Today Bio 2019, 2, 100010. [Google Scholar] [CrossRef] [PubMed]

| S. No. | Polymer Name | Structure | Advantages | Disadvantages | References |
|---|---|---|---|---|---|
| 1 | poly-HPMA |  | Biocompatible, hydrophilic, non-immunogenic | Non-biodegradable, broad polydispersity index | [22] |
| 2 | PEG | 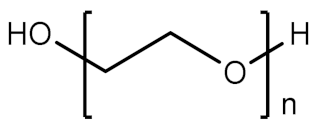 | Biocompatible and hydrophilic, soluble in common organic solvents, polymer with stealth behavior | Non-biodegradable, immunogenic, limited conjugation sites | [23,24] |
| 3 | PLA | 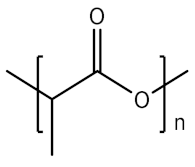 | Biodegradable, biocompatible, renewable | Slow degradation, acidic degradation product, hydrophobic, limited conjugation sites, tissue reaction at the injection site | [25,26] |
| 4 | PLGA |  | Biodegradable, biocompatible, renewable with tunable properties | limited conjugation sites, acidic degradation product, tissue reaction at the injection site | [27,28] |
| 5 | Polyphosphazene |  | Biocompatible, biodegradable, multifunctional | Complicated synthesis procedure of functional polymers, high production cost | [29,30] |
| 6 | Polyphosphoesters | 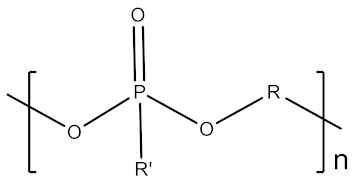 | Biocompatible, biodegradable, stealth property, hydrophilic, multifunctional | High production cost, limited in vivo data | [31,32] |
| 7 | Polyglycerol | 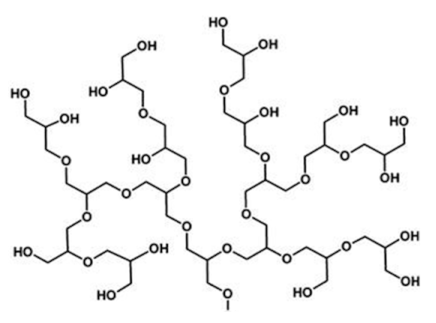 | Hydrophilic, biocompatible, low toxicity and immunogenicity, antifouling property | Difficult to control the degree of branching, molecular weight, and polydispersity, non-biodegradable | [33,34] |
| 8 | PGA | 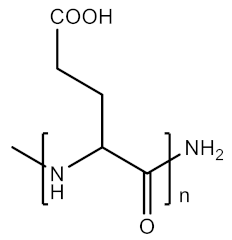 | Hydrophilic, biocompatible, biodegradable | Insoluble in common organic solvents, poor control on molecular weight and polydispersity, high production cost. | [35,36] |
| 9 | Albumin |  | Biocompatible, biodegradable, non-toxic, low cost | Thermolabile, complex structure | [37,38] |
| 10 | Gelatin |  | Biocompatible, biodegradable | Batch-to-batch variation, high polydispersity | [39] |
| 11 | Alginate | 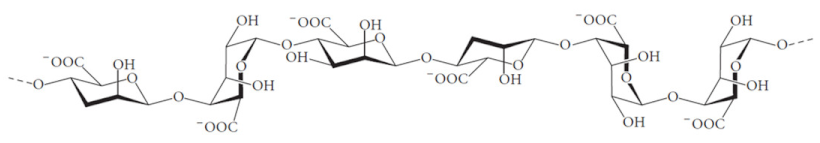 | Biodegradable, biocompatible, non-toxic, low cost | Poor stability, increases the viscosity of the solution, batch-to-batch variation | [40] |
| 12 | Chitosan | 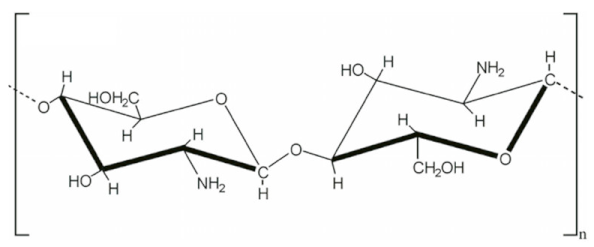 | Non-toxic, biodegradable, biocompatible, inexpensive | Soluble in a limited number of solvents, poor solubility at neutral ph, increases the viscosity of the solution, batch-to-batch variation | [41,42] |
| 13 | HA |  | Hydrophilic, biocompatible, biodegradable, non-immunogenic, targeting ligand | Soluble in a limited number of solvents, increases the viscosity of the solution, immunosuppressive, batch-to-batch variation | [43,44] |
| 14 | HES |  | Hydrophilic, biocompatible, biodegradable, low hypersensitivity | Difficulty in regulating the length and substitution degree of the hydrophobic side-chain, batch-to-batch variation | [45] |
| Polymer | Drug Used | Method of Drug Conjugation | Imaging Agent | Indication | Reference |
|---|---|---|---|---|---|
| poly-HPMA | Doxorubicin | pH-sensitive hydrazone bond formation | Zirconium 89 C and dye(DY-633 andDY-676) C | Cancer | [47] |
| poly-HPMA | Epirubicin | Copolymerization of HPMA with GFLG-EPI | Cy 5 Fluorophore C | Ovarian cancer | [49] |
| poly-HPMA | Pyropheophorbide | Amide bond | Pyropheophorbide C | Cancer | [50] |
| poly-HPMA | Paclitaxel | GFLG linker | Cyanine 5.5 and Gadolinium (III) C | Breast cancer | [51] |
| PEG | Chlorine e6 | Disulfide linker | Chlorine e6 C | Cancer | [52] |
| PEG | Mertansine | Succinimidyl-3-(2-pyridyldithio)-propionate (SPDP) linker | Gallium 68 C | Prostate cancer | [53] |
| Methoxy PEG | Monomethyl auristatin E | Reductive sensitive and self-immolative linker | Cyanine 7.5 C | Cancer | [54] |
| PLA | Doxorubicin | Hydrazone linker | Rhodamine B (RhB) C | Cancer | [55] |
| PLGA | Methotrexate | Ethylenediamine linker | 1-pyrenebutyric acid C | Cancer | [56] |
| Polyphosphazene | Docetaxel | Aconitic acid linker | Cyanine dye C | Cancer | [57] |
| Polyphsphoesters | Doxorubicin | Thioketal linker | Chlorin e6 E | Cancer | [58] |
| Polyglycerol | Doxorubicin | pH-sensitive linker/enzyme sensitive linker | Doxorubicin C | Cancer | [59] |
| PGA | Porphyrin | Ring-opening polymerization (amide bond) | Porphyrin C | Cancer | [60] |
| PGA | Doxorubicin | Carbodiimide coupling | Tc-99m C | Cancer | [61] |
| Albumin | Chlorine e6 | Carbodiimide coupling | Iridium oxide, manganese dioxide E | Cancer | [62] |
| Albumin | Doxorubicin | Maleimide-sulfhydryl/Schiff base | Gadolinium (III) C | Triple-negative breast cancer | [63] |
| Gelatin | Porphyrin | Amide coupling | Porphyrin C | Antibiotic resistance | [64] |
| Alginate | Kinase inhibitor (PI103) | Adamantane-Inclusion complex | 5FAM C | Cancer | [65] |
| Alginate | Doxorubicin | Acid-labile Schiff base | Fluorescent carbon dots C | Cancer | [66] |
| Chitosan | Pyropheophorbide | Carbodiimide coupling | Fluorescein isothiocyanate C | Cancer | [67] |
| Chitosan | Bilirubin, losartan | Carbodiimide coupling | MHI-148 C | Hepatic fibrosis | [68] |
| Chitosan | Doxorubicin | p-carboxybenzaldehyde linker | Doxorubicin | Cancer | [69] |
| HA | Gemcitabine | Carbodiimide coupling | THP C | Cancer | [70] |
| HA | Prussian Blue | Carbodiimide coupling | Fe3O4 E Quantom dots C | Cancer | [71] |
| HES | Paclitaxel | Disulfide linker | DiR E | Cancer | [72] |
| HES | Doxorubicin | Disulfide linker | ICG E | Cancer | [73] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Manandhar, S.; Sjöholm, E.; Bobacka, J.; Rosenholm, J.M.; Bansal, K.K. Polymer-Drug Conjugates as Nanotheranostic Agents. J. Nanotheranostics 2021, 2, 63-81. https://doi.org/10.3390/jnt2010005
Manandhar S, Sjöholm E, Bobacka J, Rosenholm JM, Bansal KK. Polymer-Drug Conjugates as Nanotheranostic Agents. Journal of Nanotheranostics. 2021; 2(1):63-81. https://doi.org/10.3390/jnt2010005
Chicago/Turabian StyleManandhar, Sajana, Erica Sjöholm, Johan Bobacka, Jessica M. Rosenholm, and Kuldeep K. Bansal. 2021. "Polymer-Drug Conjugates as Nanotheranostic Agents" Journal of Nanotheranostics 2, no. 1: 63-81. https://doi.org/10.3390/jnt2010005
APA StyleManandhar, S., Sjöholm, E., Bobacka, J., Rosenholm, J. M., & Bansal, K. K. (2021). Polymer-Drug Conjugates as Nanotheranostic Agents. Journal of Nanotheranostics, 2(1), 63-81. https://doi.org/10.3390/jnt2010005









