Acid Treated Montmorillonite—Eco-Friendly Clay as Catalyst in Carvone Isomerization to Carvacrol
Abstract
1. Introduction
2. Materials and Methods
2.1. Modification of Montmorillonite K10
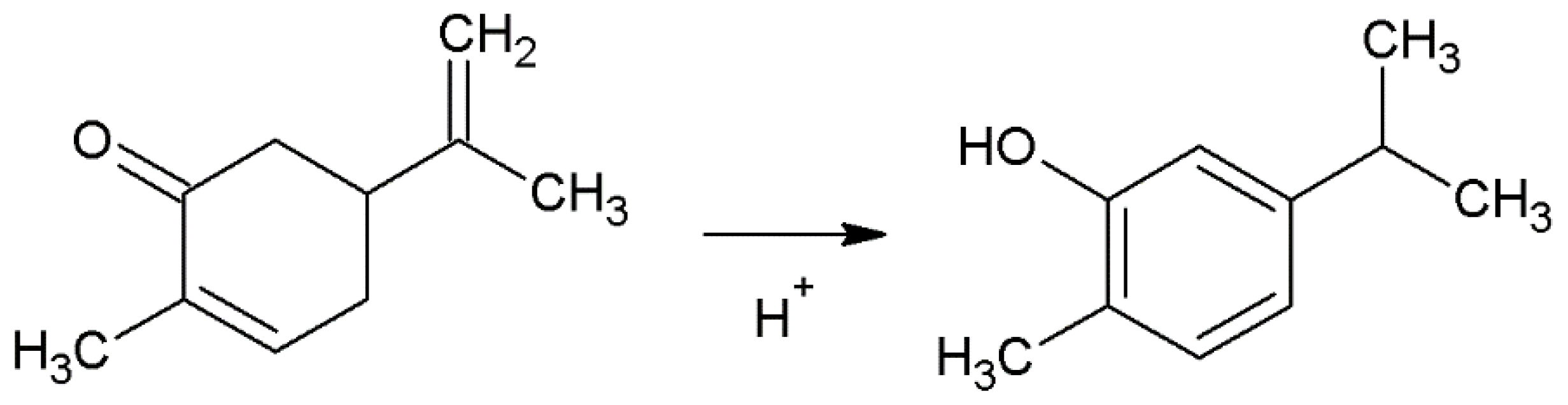
2.2. Isomerization of Carvone to Carvacrol
2.3. Characterization of Materials
3. Results
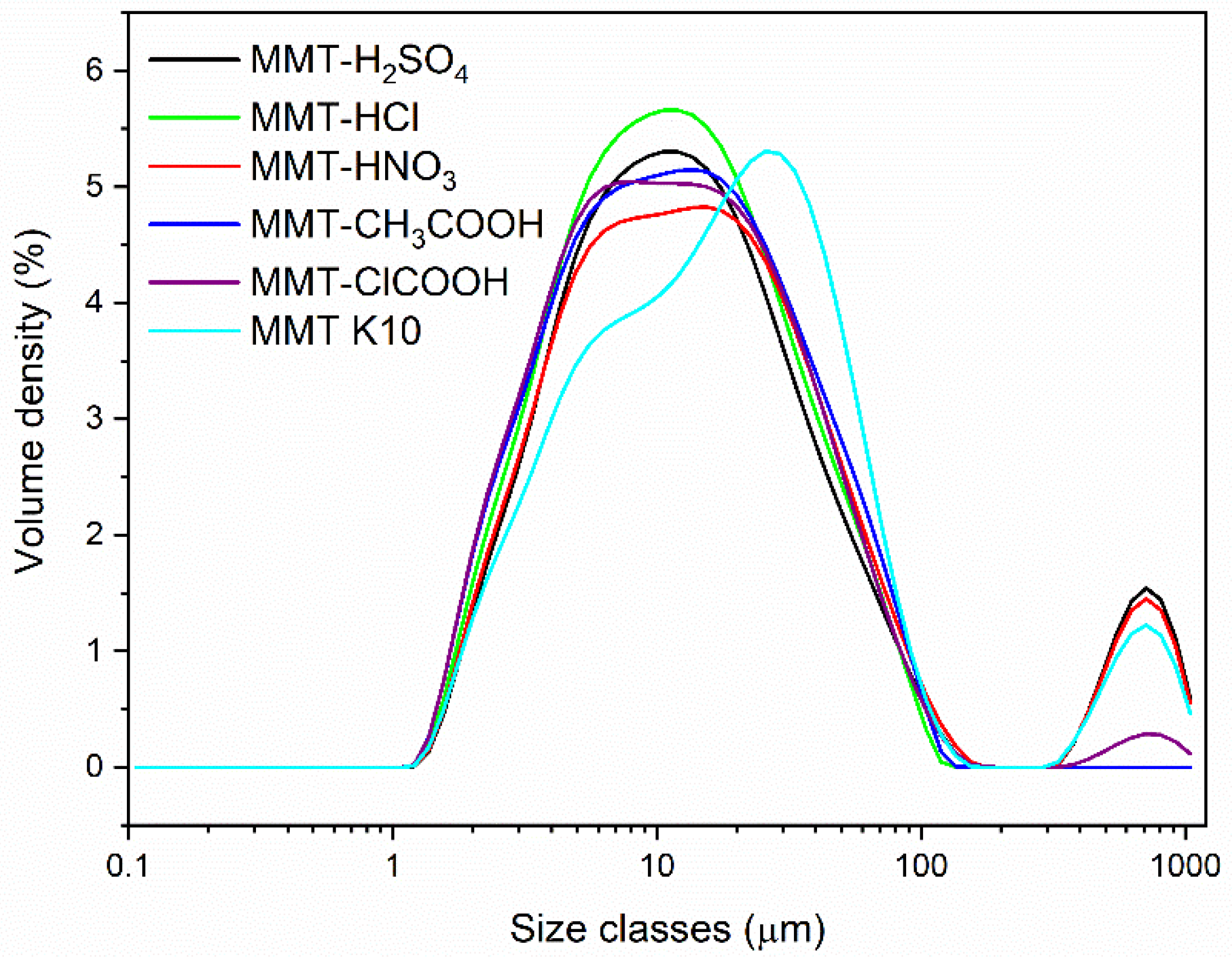
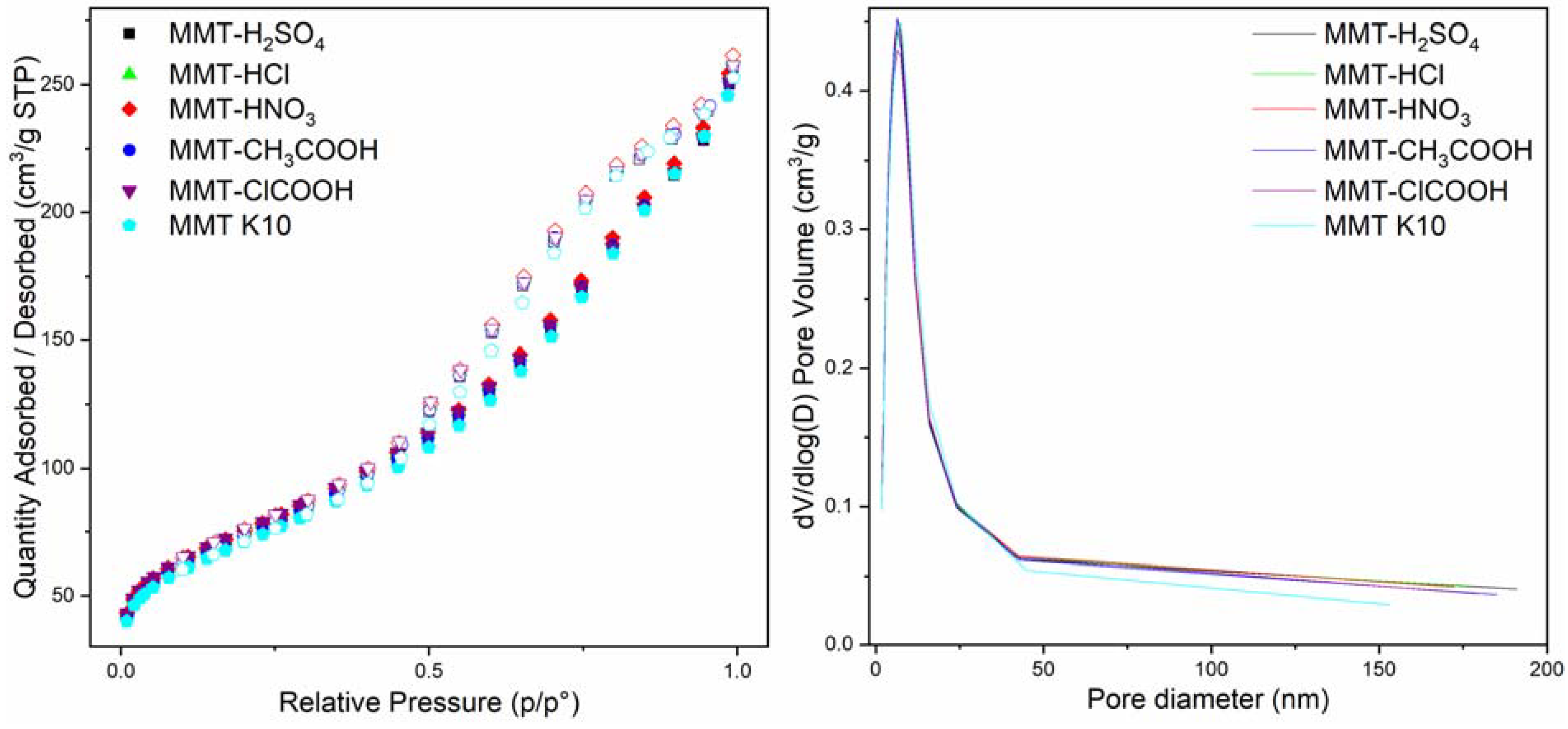
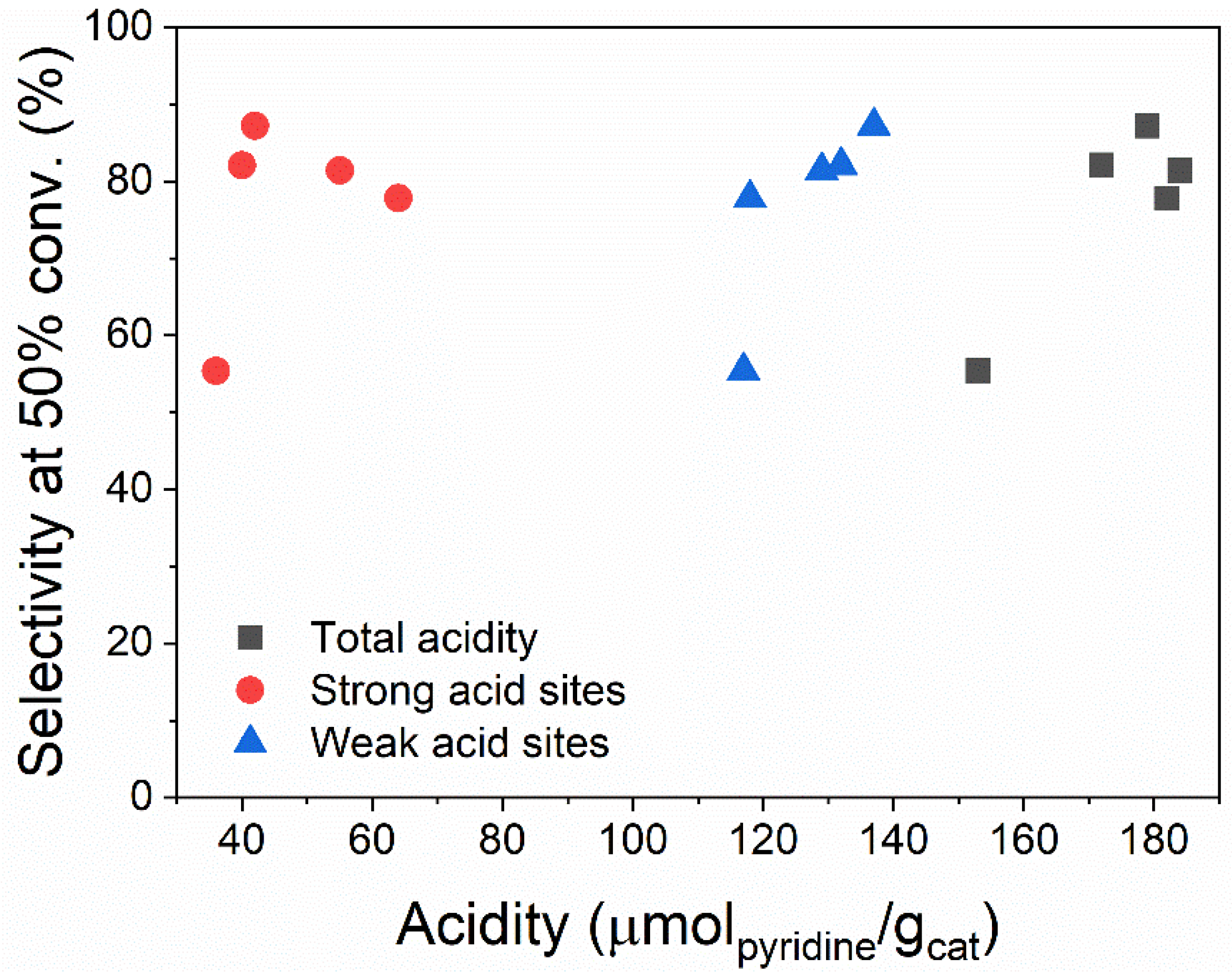
3.1. Material Characterization
3.2. Catalytic Tests
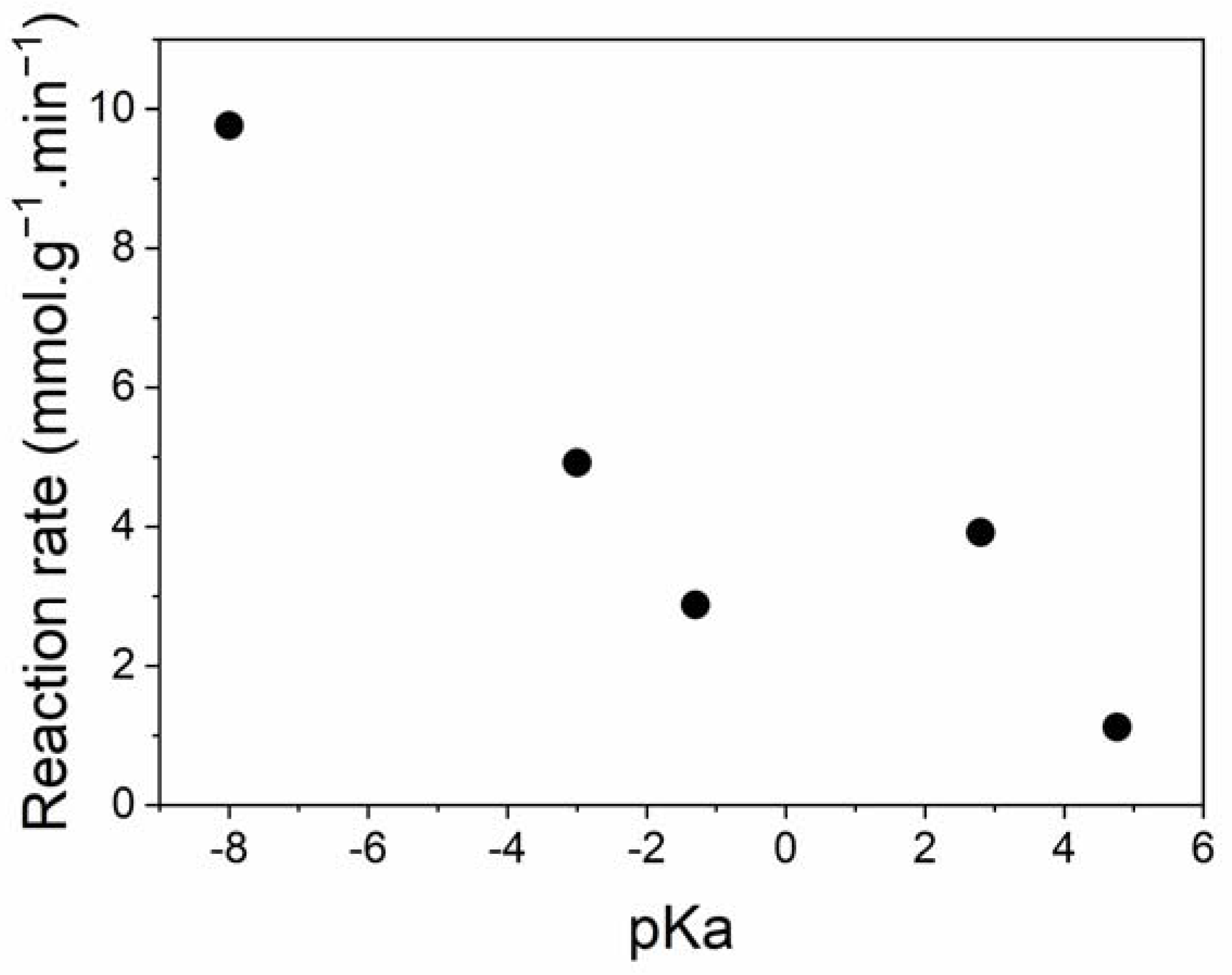
3.2.1. Influence of Acid Treatment Type
3.2.2. Influence of the Solvent
3.2.3. The Influence of the Temperature and Catalyst Amount
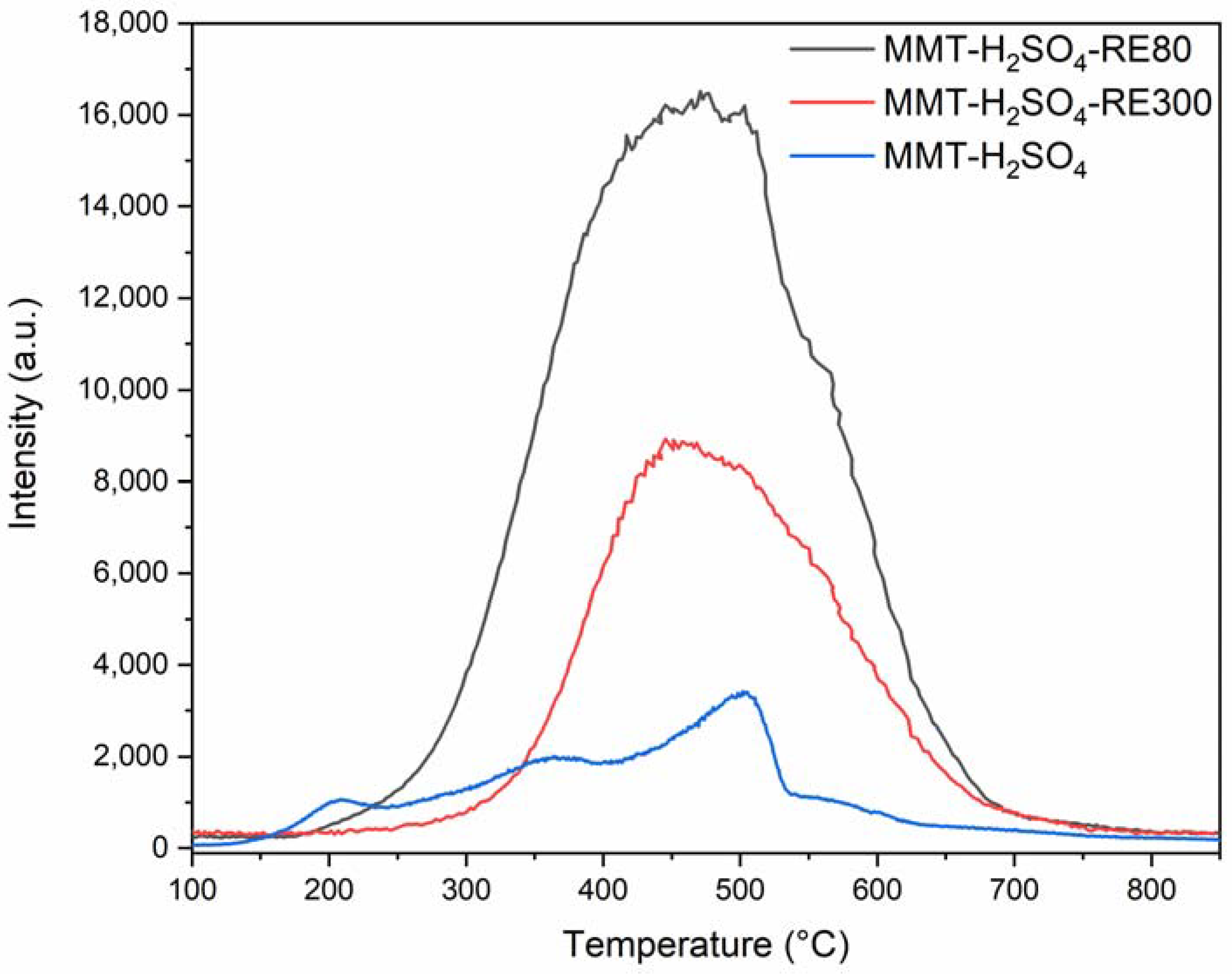
3.2.4. The Catalyst Reuse
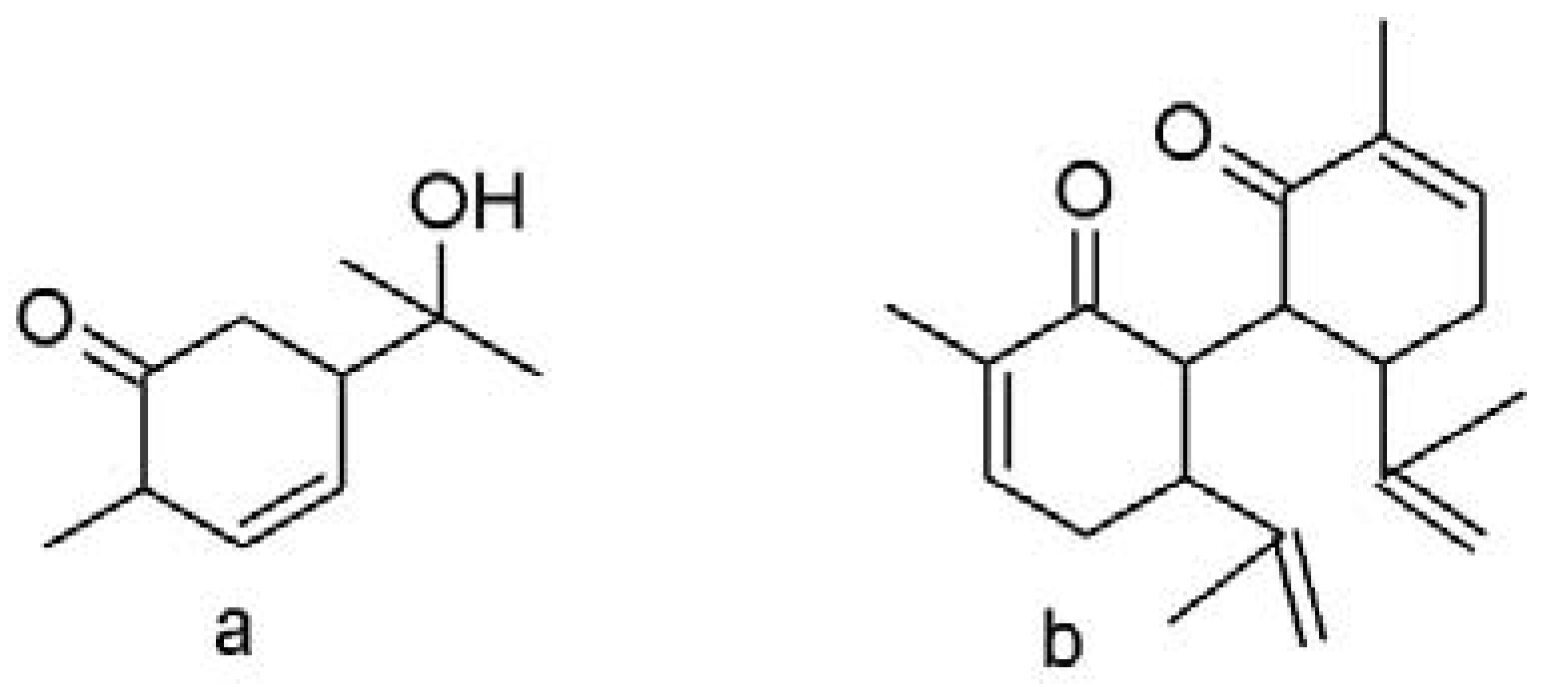
3.2.5. Proposed Mechanism of Carvone Isomerization to Carvacrol
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Conflicts of Interest
References
- Wróblewska, A.; Retajczyk, M. The Isomerization of S-Carvone over the Natural Clinoptilolite as the Catalyst: The Influence of Reaction Time, Temperature and Catalyst Content. React. Kinet. Mech. Catal. 2020, 130, 273–288. [Google Scholar] [CrossRef]
- Alokam, R.; Jeankumar, V.U.; Sridevi, J.P.; Matikonda, S.S.; Peddi, S.; Alvala, M.; Yogeeswari, P.; Sriram, D. Identification and Structure–Activity Relationship Study of Carvacrol Derivatives as Mycobacterium Tuberculosis Chorismate Mutase Inhibitors. J. Enzyme Inhib. Med. Chem. 2014, 29, 547–554. [Google Scholar] [CrossRef] [PubMed]
- Yadav, G.D.; Kamble, S.B. Synthesis of Carvacrol by Friedel–Crafts Alkylation of o-Cresol with Isopropanol Using Superacidic Catalyst UDCaT-5. J. Chem. Technol. Biotechnol. 2009, 84, 1499–1508. [Google Scholar] [CrossRef]
- Marinelli, L.; Di Stefano, A.; Cacciatore, I. Carvacrol and its derivatives as antibacterial agents. Phytochem. Rev. 2018, 17, 903–921. [Google Scholar] [CrossRef]
- Kjonaas, R.A.; Mattingly, S.P. Acid-Catalyzed Isomerization of Carvone to Carvacrol. J. Chem. Educ. 2005, 82, 1813. [Google Scholar] [CrossRef]
- De Carvalho, C.C.C.R.; da Fonseca, M.M.R. Carvone: Why and How Should One Bother to Produce This Terpene. Food Chem. 2006, 95, 413–422. [Google Scholar] [CrossRef]
- Fahlbusch, K.-G.; Hammerschmidt, F.-J.; Panten, J.; Pickenhagen, W.; Schatkowski, D.; Bauer, K.; Garbe, D.; Surburg, H. Ullmann’s Encyclopedia of Industrial Chemistry II Flavors and Fragrances; Wiley-VCH-Verl.: Weinheim, Germany, 2020; p. 105. [Google Scholar]
- Ritter, J.J.; Ginsburg, D. The Action of F-Butyl Hypochlorite on a-Pinene 1,2. J. Am. Chem. Soc. 1950, 72, 2381–2384. [Google Scholar] [CrossRef]
- Gozzi, C.; Convard, A.; Husset, M. Heterogeneous Acid-Catalysed Isomerization of Carvone to Carvacrol. React. Kinet. Catal. Lett. 2009, 97, 301–306. [Google Scholar] [CrossRef]
- Raner, K.D.; Strauss, C.R.; Trainor, R.W.; Thorn, J.S. A New Microwave Reactor for Batchwise Organic Synthesis. J. Org. Chem. 1995, 60, 2456–2460. [Google Scholar] [CrossRef]
- Heckenroth, M.; Khlebnikov, V.; Neels, A.; Schurtenberger, P.; Albrecht, M. Catalytic Hydrogenation Using Abnormal N-Heterocyclic Carbene Palladium Complexes: Catalytic Scope and Mechanistic Insights. ChemCatChem 2011, 3, 167–173. [Google Scholar] [CrossRef][Green Version]
- Benavente, P.; Cárdenas-Lizana, F.; Keane, M.A. Promotional Effect of Water in the Clean Continuous Production of Carvacrol from Carvone. Catal. Today 2018, 308, 45–49. [Google Scholar] [CrossRef]
- Benavente, P.; Cárdenas-Lizana, F.; Keane, M.A. Selective Production of Carvacrol from Carvone over Supported Pd Catalysts. Catal. Commun. 2017, 96, 37–40. [Google Scholar] [CrossRef]
- Jin, X.; Taniguchi, K.; Yamaguchi, K.; Nozaki, K.; Mizuno, N. A Ni–Mg–Al Layered Triple Hydroxide-Supported Pd Catalyst for Heterogeneous Acceptorless Dehydrogenative Aromatization. Chem. Commun. 2017, 53, 5267–5270. [Google Scholar] [CrossRef]
- Liu, X.; Li, S.; Wu, Q.; Wan, M.; Zhang, J.; Liu, Y.; Tang, H. CN CN105523897A: A Synthetic Method of Carvacrol; Huai’an Wanbang Aromatic Chemicals Industry Co., Ltd.: Huaian, China, 2015; Available online: https://patents.google.com/patent/CN105523897A/en (accessed on 10 November 2021).
- Zheng, S.; Zhang, J.; Shen, Z. Platinum(IV)-Catalyzed Synthesis of Unsymmetrical Polysubstituted Benzenes via Intramolecular Cycloaromatization Reaction. Adv. Synth. Catal. 2015, 357, 2803–2808. [Google Scholar] [CrossRef]
- Kim, H.-M.; Oh, J.-M. Physico-chemical interaction between clay minerals and albumin protein according to the type of clay. Minerals 2019, 9, 396. [Google Scholar] [CrossRef]
- Norhayati, M.N.; Uan Noor Maznee, T.I.; Hoong, S.S.; Nurul’Ain, H.; Srihanum, A.; Kosheela Devi, P.P.; Mohd Norhisham, S.; Yeong, S.K.; Hazimah, A.H. Reproducibility of palm-based polyols production. J. Oil Palm Res. 2016, 28, 114–120. [Google Scholar]
- Vyskočilová, E.; Gruberová, A.; Shamzhy, M.; Vrbková, E.; Krupka, J.; Červený, L. Prins cyclization in 4-methyl-2-phenyl-tetrahydro-2Hpyran-4-ol preparation using smectite clay as catalyst. Reac. Kinet. Mech. Cat. 2018, 124, 711–725. [Google Scholar] [CrossRef]
- Sekerová, L.; Černá, H.; Vyskočilová, E.; Vrbková, E.; Červený, L. Preparation of α-Terpineol from Biomass Resource Catalysed by Acid Treated Montmorillonite K10. Catal. Lett. 2021. ahead of print. [Google Scholar] [CrossRef]
- Timofeeva, M.N.; Volcho, K.P.; Mikhalchenko, O.S.; Panchenko, V.N.; Krupskaya, V.V.; Tsybulya, S.V.; Gil, A.; Vicente, M.A.; Salakhutdinov, N.F. Synthesis of octahydro-2H-chromen-4-ol from vanillin and isopulegol over acid modified montmorillonite clays: Effect of acidity on the Prins cyclization. J. Mol. Catal. A Chem. 2015, 398, 26–34. [Google Scholar] [CrossRef]
- Ravichandran, J.; Sivasankar, B. Properties and Catalytic Activity of Acid-Modified Montmorillonite and Vermiculite. Clays Clay Miner. 1997, 45, 854–858. [Google Scholar] [CrossRef]
- Zatta, L.; Paiva, E.J.M.; Corazza, M.L.; Wypych, F.; Ramos, L.P. The Use of Acid-Activated Montmorillonite as a Solid Catalyst for the Production of Fatty Acid Methyl Esters. Energy Fuels 2014, 28, 5834–5840. [Google Scholar] [CrossRef]
- Luna, F.M.T.; Cecilia, J.A.; Saboya, R.M.A.; Barrera, D.; Sapag, K.; Rodríguez-Castellón, E.; Cavalcante, C.L. Natural and Modified Montmorillonite Clays as Catalysts for Synthesis of Biolubricants. Materials 2018, 11, 1764. [Google Scholar] [CrossRef]
- Varadwaj, G.B.B.; Rana, S.; Parida, K.M. Amine functionalized K10 montmorillonite: A solid acid–base catalyst for the Knoevenagel condensation reaction. Dalton Trans. 2013, 42, 5122–5129. [Google Scholar] [CrossRef] [PubMed]
- Vrbková, E.; Šteflová, B.; Vyskočilová, E.; Červený, L. Heterogeneous Mo/W/Zn–SiO2 based catalysts in nopol (2-(6,6-dimethyl-2-bicyclo[3.1.1]hept-2-enyl)ethanol) synthesis. Reac. Kinet. Mech. Cat. 2020, 131, 213–232. [Google Scholar] [CrossRef]
- Vrbková, E.; Šteflová, B.; Sekerová, L.; Krupka, J.; Vyskočilová, E.; Červený, L. Contribution to MoO3–SiO2 and WO3–SiO2 utilization—active catalysts in jasmine aldehyde, 2-hexyl-1,3-dioxolane and methyllaurate synthesis. Reac. Kinet. Mech. Cat. 2020, 129, 645–658. [Google Scholar] [CrossRef]
- Vrbková, E.; Šteflová, B.; Zapletal, M.; Vyskočilová, E.; Červený, L. Tungsten oxide-based materials as effective catalysts in isopulegol formation by intramolecular Prins reaction of citronellal. Res. Chem. Intermed. 2020, 46, 4047–4059. [Google Scholar] [CrossRef]
- Vrbková, E.; Prejza, T.; Lhotka, M.; Vyskočilová, E.; Červený, L. Fe-Modified Zeolite BETA as an Active Catalyst for Intramolecular Prins Cyclization of Citronellal. Catal. Lett. 2021, 151, 1993–2003. [Google Scholar] [CrossRef]
- Velthoen, M.E.Z.; Nab, S.; Weckhuysen, B.M. Probing acid sites in solid catalysts with pyridine UV-Vis spectroscopy. Phys. Chem. Chem. Phys. 2018, 20, 21647–21659. [Google Scholar] [CrossRef]
- Sekerová, L.; Spáčilová, M.; Vyskočilová, E.; Krupka, J.; Červený, L. Acid catalyzed acetalization of aldehydes with diols resulting into the formation of fragrant cyclic acetals. Reac. Kinet. Mech. Cat. 2019, 127, 727–740. [Google Scholar] [CrossRef]
- Liu, Y.; Alessi, D.S.; Flynn, S.L.; Alam, M.S.; Hao, W.; Gingras, M.; Zhao, H.; Konhauser, K.O. Acid-base properties of 23. kaolinite, montmorillonite and illite at marine ionic strength. Chem. Geol. 2018, 483, 191–200. [Google Scholar] [CrossRef]
- Vyskočilová, E.; Vrbková, E.; Trejbal, J.; Vaňková, M.; Červený, L. Eco-Friendly Natural Clay: Montmorillonite Modified with Nickel or Ruthenium as an Effective Catalyst in Gamma-Valerolactone Synthesis. Catal. Lett. 2021. ahead of print. [Google Scholar] [CrossRef]
- Ma, X.; Fan, Y.; Dong, X.; Chen, R.; Li, H.; Sun, D.; Yao, S. Impact of Clay Minerals on the Dewatering of Coal Slurry: An Experimental and Molecular-Simulation Study. Minerals 2018, 8, 400. [Google Scholar] [CrossRef]
- Chimni, S.S.; Singh, S.; Kumar, A. The pH of the reaction controls the stereoselectivity of organocatalyzed direct aldol reactions in water A. Tetrahedron Asymmetry 2009, 20, 1722–1724. [Google Scholar] [CrossRef]
- Vrbková, E.; Vyskočilová, E.; Lhotka, M.; Červený, L. Solvent Influence on Selectivity in α-Pinene Oxide Isomerization Using MoO3-Modified Zeolite BETA. Catalysts 2020, 10, 1244. [Google Scholar] [CrossRef]

| Abbreviation | Material |
|---|---|
| MMT K10 | Nonmodified montmorillonite K10 |
| MMT-H2SO4 | Montmorillonite K10 treated with sulfuric acid |
| MMT-HNO3 | Montmorillonite K10 treated with nitric acid |
| MMT-HCl | Montmorillonite K10 treated with hydrochloric acid |
| MMT-COOH | Montmorillonite K10 treated with acetic acid |
| MMT-ClCOOH | Montmorillonite K10 treated with chloroacetic acid |
| MMT-H2SO4-RE80 | Montmorillonite K10 treated with sulfuric acid recycled using temp. of 80 °C |
| MMT-H2SO4-RE300 | Montmorillonite K10 treated with sulfuric acid recycled using temp. of 300 °C |
| Material | Nitrogen (%) | Carbon (%) | Hydrogen (%) | Sulfur (%) | Chlorine (ppm) |
|---|---|---|---|---|---|
| MMT K10 | - | - | 1.07 | - | - |
| MMT-H2SO4 | - | 0.27 | 1.03 | - | - |
| MMT-HNO3 | - | 0.18 | 1.07 | - | - |
| MMT-HCl | - | 0.18 | 1.12 | - | 217 |
| MMT-COOH | - | 0.21 | 1.08 | - | - |
| MMT-ClCOOH | - | 0.14 | 1.21 | - | 225 |
| Material | ∑ Acid Sites | Weak Acid Sites | Strong Acid Sites |
|---|---|---|---|
| n Pyridine Adsorbed (µmol/gcat) | |||
| MMT K10 | 149 | 149 | - |
| MMT-H2SO4 | 182 | 118 | 64 |
| MMT-HNO3 | 153 | 117 | 36 |
| MMT-HCl | 179 | 137 | 42 |
| MMT-COOH | 184 | 129 | 55 |
| MMT-ClCOOH | 172 | 132 | 40 |
| Material | Dv(10) (µm) | Dv(50) (µm) | Dv(90) (µm) |
|---|---|---|---|
| MMT K10 | 3.69 | 17.1 | 69.1 |
| MMT-H2SO4 | 3.52 | 12.7 | 73.1 |
| MMT-HNO3 | 3.45 | 13.7 | 74.6 |
| MMT-HCl | 3.27 | 11.3 | 41.9 |
| MMT-COOH | 3.10 | 11.7 | 46.9 |
| MMT-ClCOOH | 3.07 | 11.5 | 46.6 |
| Material | SBET (m2/g) | St-Plot (m2/g) | t-Plot Micropore Volume (cm3/g) |
|---|---|---|---|
| MMT K10 | 254 | 246 | 0.0021 |
| MMT-H2SO4 | 263 | 255 | 0.0021 |
| MMT-HNO3 | 271 | 258 | 0.0038 |
| MMT-HCl | 268 | 255 | 0.0046 |
| MMT-COOH | 263 | 257 | 0.0013 |
| MMT-ClCOOH | 271 | 259 | 0.0043 |
| Row | Cat. | Temp. (°C) | Cat. Amount (g) | Solvent | Conversion (24 h, %) | Selectivity (24 h, %) | Selectivity (at 50% Conv., %) | Average Reaction Rate (mmol g−1 min−1) |
|---|---|---|---|---|---|---|---|---|
| 1 | MMT K10 | 80 | 0.5 | toluene | 26.1 | 24.5 | - | 0.24 |
| 2 | MMT-HNO3 | 99.9 | 79.9 | 55.4 | 2.88 | |||
| 3 | MMT-HCl | 99.9 | 84.5 | 87.2 | 9.76 | |||
| 4 | MMT-COOH | 75.9 | 73.6 | 81.4 | 1.12 | |||
| 5 | MMT-ClCOOH | 99.9 | 95.5 | 82.1 | 3.92 | |||
| 6 | MMT-H2SO4 | 99.5 | 83.1 | 77.8 | 4.92 | |||
| 7 | benzonitrile | 11.3 | 45.1 | - | - | |||
| 8 | heptane | 82.2 | 68.6 | 57.8 | 0.76 | |||
| 9 | propan-1-ol | 56.6 | 6.5 | 2.2 | 0.0007 | |||
| 10 | none | 93.8 * | 98.6 * | 78.6 | 5.81 | |||
| 11 | 70 | toluene | 26.6 | 38.0 | - | 0.40 | ||
| 12 | 90 | 100 | 94.1 | 85.4 | 6.44 | |||
| 13 | 80 | 0.075 | 6.8 | 50.1 | - | 0.02 | ||
| 14 | 0.3 | 34.1 | 45.9 | - | 0.36 | |||
| 15 | MMT-H2SO4-RE80 | 0.5 | 67.2 | 99.9 | 97.2 | 1.01 | ||
| 16 | MMT-H2SO4-RE300 | 87.3 | 99.9 | 99.9 | 2.02 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vrbková, E.; Šímová, A.; Vyskočilová, E.; Lhotka, M.; Červený, L. Acid Treated Montmorillonite—Eco-Friendly Clay as Catalyst in Carvone Isomerization to Carvacrol. Reactions 2021, 2, 486-498. https://doi.org/10.3390/reactions2040031
Vrbková E, Šímová A, Vyskočilová E, Lhotka M, Červený L. Acid Treated Montmorillonite—Eco-Friendly Clay as Catalyst in Carvone Isomerization to Carvacrol. Reactions. 2021; 2(4):486-498. https://doi.org/10.3390/reactions2040031
Chicago/Turabian StyleVrbková, Eva, Adéla Šímová, Eliška Vyskočilová, Miloslav Lhotka, and Libor Červený. 2021. "Acid Treated Montmorillonite—Eco-Friendly Clay as Catalyst in Carvone Isomerization to Carvacrol" Reactions 2, no. 4: 486-498. https://doi.org/10.3390/reactions2040031
APA StyleVrbková, E., Šímová, A., Vyskočilová, E., Lhotka, M., & Červený, L. (2021). Acid Treated Montmorillonite—Eco-Friendly Clay as Catalyst in Carvone Isomerization to Carvacrol. Reactions, 2(4), 486-498. https://doi.org/10.3390/reactions2040031






