Modeling Volatility Characteristics of Epileptic EEGs using GARCH Models
Abstract
:1. Introduction
2. Methods
Data
3. Conditional Volatility Models
3.1. Autoregressive Moving Average Models
3.2. Generalized Autoregressive Conditional Heteroscedasticity Models
4. Confidence Intervals for Half-Life (HL)
5. Analysis
6. Results
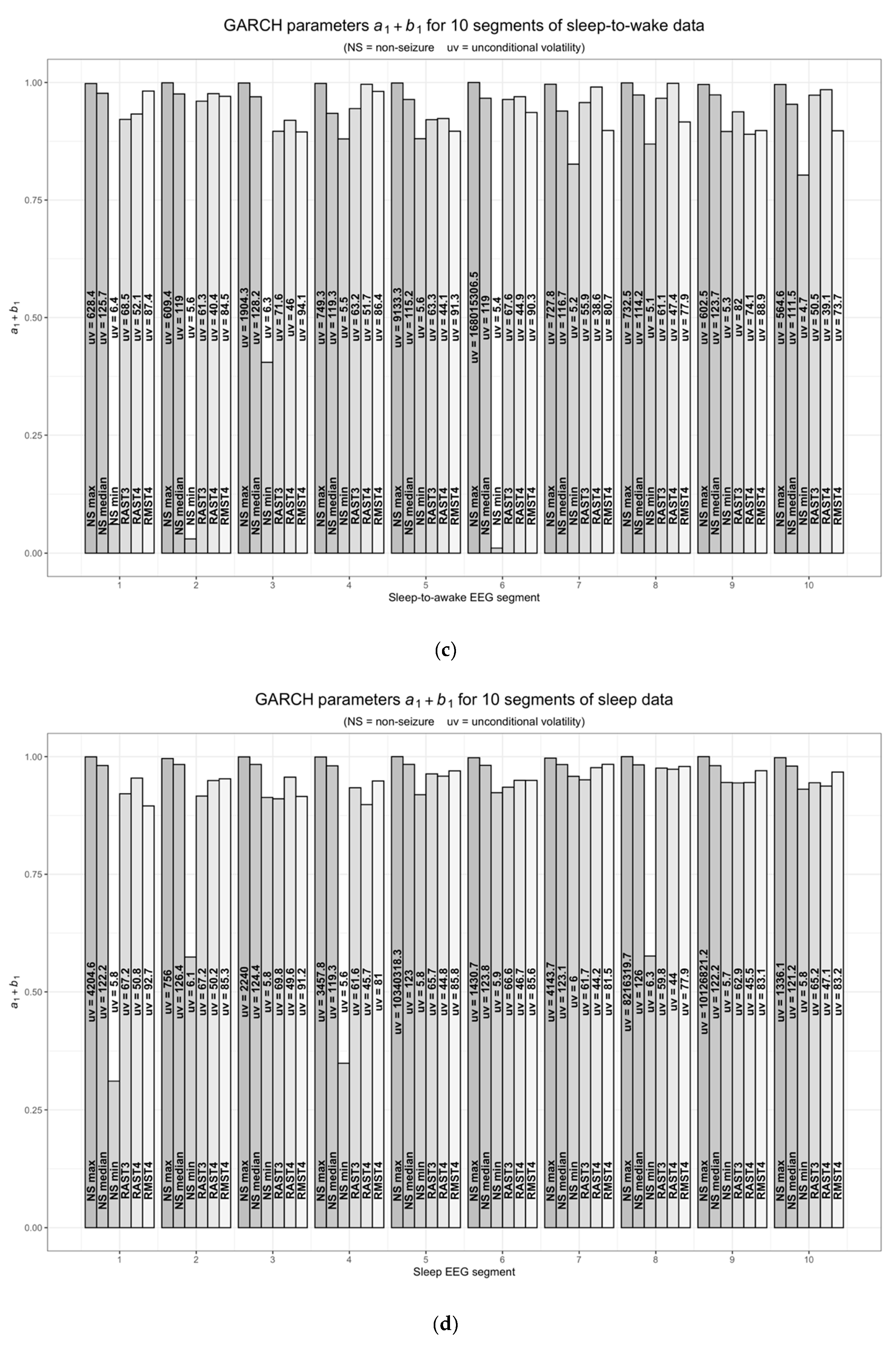
6.1. GARCH Models
6.2. Half-life and Confidence Intervals
7. Random Starting Points
8. Discussion
9. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Rosso, O.A.; Martin, M.; Plastino, A. Brain electrical activity analysis using wavelet-based informational tools. Phys. A Stat. Mech. Appl. 2002, 313, 587–608. [Google Scholar] [CrossRef]
- Adeli, H.; Zhou, Z.; Dadmehr, N. Analysis of EEG records in an epileptic patient using wavelet transform. J. Neurosci. Methods 2003, 123, 69–87. [Google Scholar] [CrossRef]
- Dikanev, T.; Smirnov, D.; Wennberg, R.; Velazquez, J.P.; Bezruchko, B. EEG nonstationarity during intracranially recorded seizures: Statistical and dynamical analysis. Clin. Neurophysiol. 2005, 116, 1796–1807. [Google Scholar] [CrossRef] [PubMed]
- Senhadji, L.; Wendling, F. Epileptic transient detection: Wavelets and time-frequency approaches. Neurophysiol. Clin. Neurophysiol. 2002, 32, 175–192. [Google Scholar] [CrossRef] [Green Version]
- Lehnertz, K.; Mormann, F.; Osterhage, H.; Müller, A.; Prusseit, J.; Chernihovskyi, A.; Staniek, M.; Krug, D.; Bialonski, S.; Elger, C.E. State-of-the-Art of Seizure Prediction. J. Clin. Neurophysiol. 2007, 24, 147–153. [Google Scholar] [CrossRef] [PubMed]
- Osterhage, H.; Lehnertz, K. NONLINEAR TIME SERIES ANALYSIS IN EPILEPSY. Int. J. Bifurc. Chaos 2007, 17, 3305–3323. [Google Scholar] [CrossRef]
- Mormann, F.; Andrzejak, R.G.; Elger, C.E.; Lehnertz, K. Seizure prediction: The long and winding road. Brain 2007, 130, 314–333. [Google Scholar] [CrossRef] [Green Version]
- Acharya, U.R.; Sree, S.V.; Ang, P.C.A.; Yanti, R.; Suri, J.S. Application of Non-Linear and Wavelet Based Features for the Automated Identification of Epileptic Eeg Signals. Int. J. Neural Syst. 2012, 22, 1250002. [Google Scholar] [CrossRef]
- Ahammad, N.; Fathima, T.; Joseph, P. Detection of Epileptic Seizure Event and Onset Using EEG. BioMed Res. Int. 2014, 2014, 1–7. [Google Scholar] [CrossRef]
- Faust, O.; Acharya, U.R.; Adeli, H.; Adeli, A. Wavelet-based EEG processing for computer-aided seizure detection and epilepsy diagnosis. Seizure 2015, 26, 56–64. [Google Scholar] [CrossRef] [Green Version]
- Urigüen, J.A.; Garcia-Zapirain, B.; Artieda, J.; Iriarte, J.; Valencia, M. Comparison of background EEG activity of different groups of patients with idiopathic epilepsy using Shannon spectral entropy and cluster-based permutation statistical testing. PLoS ONE 2017, 12, e0184044. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Jun, Y.-H.; Eom, T.-H.; Kim, Y.-H.; Chung, S.-Y.; Lee, I.G.; Kim, J.-M. Changes in background electroencephalographic activity in benign childhood epilepsy with centrotemporal spikes after oxcarbazepine treatment: A standardized low-resolution brain electromagnetic tomography (sLORETA) study. BMC Neurol. 2019, 19, 3. [Google Scholar] [CrossRef] [PubMed]
- Hu, D.; Cao, J.; Lai, X.; Liu, J. Epileptic State Classification based on Intrinsic Mode Function and Wavelet Packet Decomposition. In Proceedings of the 2019 41st Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Berlin, Germany, 23–27 July 2019; pp. 2382–2385. [Google Scholar]
- Swami, P.; Bhatia, M.; Tripathi, M.; Chandra, P.S.; Panigrahi, B.K.; Gandhi, T.K. Selection of optimum frequency bands for detection of epileptiform patterns. Healthc. Technol. Lett. 2019, 6, 126–131. [Google Scholar] [CrossRef] [PubMed]
- Mormann, F.; Lehnertz, K.; David, P.; Elger, C.E. Mean phase coherence as a measure for phase synchronization and its application to the EEG of epilepsy patients. Phys. D Nonlinear Phenom. 2000, 144, 358–369. [Google Scholar] [CrossRef]
- Le Van Quyen, M.; Martinerie, J.; Navarro, V.; Baulac, M.; Varela, F.J. Characterizing Neurodynamic Changes Before Seizures. J. Clin. Neurophysiol. 2001, 18, 191–208. [Google Scholar] [CrossRef]
- Aschenbrenner-Scheibe, R.; Maiwald, T.; Winterhalder, M.; Voss, H.U.; Timmer, J.; Schulze-Bonhage, A. How well can epileptic seizures be predicted? An evaluation of a nonlinear method. Brain 2003, 126, 2616–2626. [Google Scholar] [CrossRef] [Green Version]
- Li, D.; Zhou, W.; Drury, I.; Savit, R. Non-linear, non-invasive method for seizure anticipation in focal epilepsy. Math. Biosci. 2003, 186, 63–77. [Google Scholar] [CrossRef]
- Mormann, F.; Kreuz, T.; Andrzejak, R.G.; David, P.; Lehnertz, K.; Elger, C.E. Epileptic seizures are preceded by a decrease in synchronization. Epilepsy Res. 2003, 53, 173–185. [Google Scholar] [CrossRef]
- Pardalos, P.M.; Chaovalitwongse, W.; Iasemidis, L.D.; Sackellares, J.C.; Shiau, D.-S.; Carney, P.R.; Prokopyev, O.A.; Yatsenko, V.A. Seizure warning algorithm based on optimization and nonlinear dynamics. Math. Program. 2004, 101, 365–385. [Google Scholar] [CrossRef]
- Bartolomei, F.; Wendling, F.; Regis, J.; Gavaret, M.; Guye, M.; Chauvel, P. Pre-ictal synchronicity in limbic networks of mesial temporal lobe epilepsy. Epilepsy Res. 2004, 61, 89–104. [Google Scholar] [CrossRef]
- Escalona-Morán, M.; Cosenza, M.G.; Guillén, P.; Coutin, P. Synchronization and clustering in electroencephalographic signals. Chaos Solitons Fractals 2007, 31, 820–825. [Google Scholar] [CrossRef] [Green Version]
- Iasemidis, L.; Shiau, D.-S.; Pardalos, P.; Chaovalitwongse, W.; Narayanan, K.; Prasad, A.; Tsakalis, K.; Carney, P.; Sackellares, J. Long-term prospective on-line real-time seizure prediction. Clin. Neurophysiol. 2005, 116, 532–544. [Google Scholar] [CrossRef] [PubMed]
- Kannathal, N.; Choo, M.L.; Acharya, U.R.; Sadasivan, P. Entropies for detection of epilepsy in EEG. Comput. Methods Programs Biomed. 2005, 80, 187–194. [Google Scholar] [CrossRef] [PubMed]
- Kaplan, A.Y.; Fingelkurts, A.A.; Borisov, S.V.; Darkhovsky, B.S. Nonstationary nature of the brain activity as revealed by EEG/MED: Methodological, practical and conceptual challenges. Signal Process. 2005, 85, 2190–2212. [Google Scholar] [CrossRef]
- Le Van Quyen, M.; Soss, J.; Navarro, V.; Robertson, R.; Chávez, M.; Baulac, M.; Martinerie, J. Preictal state identification by synchronization changes in long-term intracranial EEG recordings. Clin. Neurophysiol. 2005, 116, 559–568. [Google Scholar] [CrossRef] [PubMed]
- Li, X.; Ouyang, G. Nonlinear similarity analysis for epileptic seizures prediction. Nonlinear Anal. Theory Methods Appl. 2006, 64, 1666–1678. [Google Scholar] [CrossRef]
- Winterhalder, M.; Schelter, B.; Maiwald, T.; Brandt, A.; Schad, A.; Schulze-Bonhage, A.; Timmer, J. Spatio-temporal patient–individual assessment of synchronization changes for epileptic seizure prediction. Clin. Neurophysiol. 2006, 117, 2399–2413. [Google Scholar] [CrossRef]
- Wendling, F.; Bartolomei, F.; Bellanger, J.; Chauvel, P. Interpretation of interdependencies in epileptic signals using a macroscopic physiological model of the EEG. Clin. Neurophysiol. 2001, 112, 1201–1218. [Google Scholar] [CrossRef]
- Jouny, C.; Franaszczuk, P.J.; Bergey, G.K. Characterization of epileptic seizure dynamics using Gabor atom density. Clin. Neurophysiol. 2003, 114, 426–437. [Google Scholar] [CrossRef]
- Kiymik, M.K.; Subasi, A.; Ozcalık, H.R. Neural Networks with Periodogram and Autoregressive Spectral Analysis Methods in Detection of Epileptic Seizure. J. Med. Syst. 2004, 28, 511–522. [Google Scholar] [CrossRef]
- Shoeb, A.; Edwards, H.; Connolly, J.; Bourgeois, B.; Treves, S.T.; Guttag, J. Patient-specific seizure onset detection. Epilepsy Behav. 2004, 5, 483–498. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Alkan, A.; Koklukaya, E.; Subasi, A. Automatic seizure detection in EEG using logistic regression and artificial neural network. J. Neurosci. Methods 2005, 148, 167–176. [Google Scholar] [CrossRef] [PubMed]
- Güler, I.; Übeyli, E.D. Adaptive neuro-fuzzy inference system for classification of EEG signals using wavelet coefficients. J. Neurosci. Methods 2005, 148, 113–121. [Google Scholar] [CrossRef] [PubMed]
- Päivinen, N.; Lammi, S.; Pitkänen, A.; Nissinen, J.; Penttonen, M.; Grönfors, T. Epileptic seizure detection: A nonlinear viewpoint. Comput. Methods Programs Biomed. 2005, 79, 151–159. [Google Scholar] [CrossRef]
- Subasi, A. Automatic detection of epileptic seizure using dynamic fuzzy neural networks. Expert Syst. Appl. 2006, 31, 320–328. [Google Scholar] [CrossRef]
- Valenti, P.; Cazamajou, E.; Scarpettini, M.; Aizemberg, A.; Silva, W.; Kochen, S. Automatic detection of interictal spikes using data mining models. J. Neurosci. Methods 2006, 150, 105–110. [Google Scholar] [CrossRef]
- Smart, O.; Firpi, H.; Vachtsevanos, G.J. Genetic programming of conventional features to detect seizure precursors. Eng. Appl. Artif. Intell. 2007, 20, 1070–1085. [Google Scholar] [CrossRef] [Green Version]
- Xu, G.; Wang, J.; Zhang, Q.; Zhang, S.; Zhu, J. A spike detection method in EEG based on improved morphological filter. Comput. Boil. Med. 2007, 37, 1647–1652. [Google Scholar] [CrossRef] [PubMed]
- Chan, A.M.; Sun, F.T.; Boto, E.H.; Wingeier, B.M. Automated seizure onset detection for accurate onset time determination in intracranial EEG. Clin. Neurophysiol. 2008, 119, 2687–2696. [Google Scholar] [CrossRef]
- Patnaik, L.; Manyam, O.K. Epileptic EEG detection using neural networks and post-classification. Comput. Methods Programs Biomed. 2008, 91, 100–109. [Google Scholar] [CrossRef]
- Ocak, H. Optimal classification of epileptic seizures in EEG using wavelet analysis and genetic algorithm. Signal Process. 2008, 88, 1858–1867. [Google Scholar] [CrossRef]
- Ocak, H. Automatic detection of epileptic seizures in EEG using discrete wavelet transform and approximate entropy. Expert Syst. Appl. 2009, 36, 2027–2036. [Google Scholar] [CrossRef]
- Kang, X.; Boly, M.; Findlay, G.; Jones, B.; Gjini, K.; Maganti, R.; Struck, A.F. Quantitative spatio-temporal characterization of epileptic spikes using high density EEG: Differences between NREM sleep and REM sleep. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Navarrete, M.; Alvarado-Rojas, C.; Le Van Quyen, M.; Valderrama, M. RIPPLELAB: A Comprehensive Application for the Detection, Analysis and Classification fo High Frequency Oscillations in Electroencephalographic Signlas. PLoS ONE 2016, 11, e0158276. [Google Scholar] [CrossRef]
- Zhou, M.; Tian, C.; Cao, R.; Wang, B.; Niu, Y.; Hu, T.; Guo, H.; Xiang, J. Epileptic Seizure Detection Based on EEG Signals and CNN. Front. Aging Neurosci. 2018, 12. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.; Cao, J.; Wang, J.; Hu, D.; Deng, M. Epileptic Signal Classification with Deep Transfer Learning Feature on Mean Amplitude Spectrum. In Proceedings of the 2019 41st Annual International Conference of the IEEE Engineering in Medicine and Biology Society (EMBC), Berlin, Germany, 23–27 July 2019; pp. 2392–2395. [Google Scholar]
- Zuo, R.; Wei, J.; Li, X.; Li, C.; Zhao, C.; Ren, Z.; Liang, Y.; Geng, X.; Jiang, C.; Yang, X.; et al. Automated Detection of High-Frequency Oscillations in Epilepsy Based on a Convolutional Neural Network. Front. Comput. Neurosci. 2019, 13, 6. [Google Scholar] [CrossRef] [Green Version]
- Jrad, N.; Kachenoura, A.; Merlet, I.; Bartolomei, F.; Nica, A.; Biraben, A.; Wendling, F. Automatic Detection and Classification of High-Frequency Oscillations in Depth-EEG Signals. IEEE Trans. Biomed. Eng. 2017, 64, 2230–2240. [Google Scholar] [CrossRef]
- Guo, J.; Yang, K.; Liu, H.; Yin, C.; Xiang, J.; Li, H.; Ji, R.; Gao, Y. A Stacked Sparse Autoencoder-Based Detector for Automatic Identification of Neuromagnetic High Frequency Oscillations in Epilepsy. IEEE Trans. Med. Imaging 2018, 37, 2474–2482. [Google Scholar] [CrossRef]
- Jiang, C.; Li, X.; Yan, J.; Yu, T.; Wang, X.; Ren, Z.; Li, D.; Liu, C.; Du, W.; Zhou, X.; et al. Determining the Quantitative Threshold of High-Frequency Oscillation Distribution to Delineate the Epileptogenic Zone by Automated Detection. Front. Neurol. 2018, 9, 889. [Google Scholar] [CrossRef] [Green Version]
- Liu, S.; Gürses, C.; Sha, Z.; Quach, M.M.; Sencer, A.; Bebek, N.; Curry, D.J.; Prabhu, S.; Tummala, S.; Henry, T.R.; et al. Stereotyped high-frequency oscillations discriminate seizure onset zones and critical functional cortex in focal epilepsy. Brain 2018, 141, 713–730. [Google Scholar] [CrossRef] [Green Version]
- Medvedev, A.V.; Agoureeva, G.I.; Murro, A.M. A Long Short-Term Memory neural network for the detection of epileptiform spikes and high frequency oscillations. Sci. Rep. 2019, 9, 1–10. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Engle, R.F. Autoregressive Conditional Heteroscedasticity with Estimates of the Variance of United Kingdom Inflation. Economic 1982, 50, 987. [Google Scholar] [CrossRef]
- Bollerslev, T. Generalized autoregressive conditional heteroskedasticity. J. Econ. 1986, 31, 307–327. [Google Scholar] [CrossRef] [Green Version]
- Ruppert, D. Statistics and Finance; Springer Science and Business Media LLC: Berlin, Germany, 2004. [Google Scholar]
- Diggle, P.J. Time Series: A Biostatistical Introduction; Oxford University Press: Oxford, UK, 1990. [Google Scholar]
- Hamilton, M.S. The Sacred and the Secular University. By Jon H. Roberts and James Turner. Princeton: Princeton University Press, 2000. xiv + 184 pp. $24.95 cloth. Church Hist. 2001, 70, 593–594. [Google Scholar] [CrossRef]
- Chatfield, C. The Analysis of Time Series, An Introduction, 6th ed.; Chapman & Hall/CRC: New York, NY, USA, 2004. [Google Scholar]
- Hu, M.Y.; Tsoukalas, C. Conditional volatility properties of sleep-disordered breathing. Comput. Biol. Med. 2006, 36, 303–312. [Google Scholar] [CrossRef] [PubMed]
- Wong, K.F.K.; Galka, A.; Yamashita, O.; Ozaki, T. Modelling non-stationary variance in EEG time series by state space GARCH model. Comput. Boil. Med. 2006, 36, 1327–1335. [Google Scholar] [CrossRef]
- Galka, A.; Yamashita, O.; Ozaki, T.; Biscay, R.; Valdés-Sosa, P. A solution to the dynamical inverse problem of EEG generation using spatiotemporal Kalman filtering. NeuroImage 2004, 23, 435–453. [Google Scholar] [CrossRef]
- Zivot, E. Practical Issues in the Analysis of Univariate GARCH Models; Springer Science and Business Media LLC: Berlin/Heidelburg, Germany, 2009; pp. 113–155. [Google Scholar]
- A Nelson, D.B. Stationarity and Persistence in the GARCH(1,1) Model. Econ. Theory 1990, 6, 318–334. [Google Scholar] [CrossRef]
- Xie, Y. Maximum Likelihood Estimation and Forecasting for GARCH, Markov Switching, and Locally Stationary Wavelet Processes; Department of Forest Economics, Swedish University of Agricultural Sciences: Umeå, Sweden, 2007. [Google Scholar]
- Terasvirta, T. An Introduction to Univariate GARCH Models Handbook of Financial Time Series; Springer: Berlin/Heildelburg, Germany, 2009; pp. 17–42. [Google Scholar]
- Muler, N.; Yohai, V.J. Robust estimates for GARCH models. J. Stat. Plan. Inference 2008, 138, 2918–2940. [Google Scholar] [CrossRef]
- Tsay, R.S. Analysis of Financial Time Series; Wiley: Hoboken, NJ, USA, 2005. [Google Scholar]
- Engle, R. GARCH 101: The Use of ARCH/GARCH Models in Applied Econometrics. J. Econ. Perspect. 2001, 15, 157–168. [Google Scholar] [CrossRef] [Green Version]
- Akter, M.S.; Islam, R.; Iimura, Y.; Sugano, H.; Fukumori, K.; Wang, D.; Tanaka, T.; Cichocki, A. Multiband entropy-based feature-extraction method for automatic identification of epileptic focus based on high-frequency components in interictal iEEG. Sci. Rep. 2020, 10, 1–17. [Google Scholar] [CrossRef] [PubMed]

| Seizure Channels | Non-seizure Channels | |||
|---|---|---|---|---|
| RAST3 | RAST4 | RMST4 | Half Life | |
| Segment | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | Median Min Max |
| 1 | 1.64 (1.29,1.98) 5 | 1.78 (1.55,2.01) 5 | 6.36 (3.24,9.47) 32 | 12.37 6.00 69.59 |
| 2 | 1.83 (1.37,2.29) 5 | 1.49 (1.26,1.72) 5 | N/A | 12.24 4.03 141.25 |
| 3 | 5.24 (4.27,6.22) 22 | 1.93 (1.68,2.18) 3 | 6.49 (3.11,9.88) 26 | 14.09 3.24 452.16 |
| 4 | 1.90 (1.58,2.23) 5 | 1.79 (1.55,2.03) 5 | 6.33 (1.52,11.14) 33 | 18.61 4.44 106.59 |
| 5 | 1.27 (0.98,1.55) 4 | 1.50 (1.30,1.70) 5 | 6.42 (0.10,12.74) 37 | 8.27 2.16 208.17 |
| 6 | 1.61 (1.30,1.91) 2 | 1.98 (1.73,2.23) 2 | 268.82 (0.14,537.50) 53 | 28.11 4.82 83.82 |
| Seizure Channels | Non-seizure Channels | |||
|---|---|---|---|---|
| RAST3 | RAST4 | RMST4 | Half Life | |
| Segment | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | Median Min Max |
| 1 | 1.13 (0.86,1.41) 2 | 1.47 (1.18,1.75) 2 | 6.49 (2.73,10.24) 42 | 7.64 1.16 186.05 |
| 2 | 1.72 (1.20,2.23) 2 | 1.88 (1.47,2.29) 2 | 70.74 (5.59,135.88) 52 | 10.94 4.89 67.61 |
| 3 | 1.75 (1.06,2.44) 7 | 1.71 (1.31,2.11) 5 | 13.65 (3.83,23.47) 53 | 15.25 3.06 115.7 |
| 4 | 1.84 (1.35,2.34) 4 | 1.35 (1.14,1.58) 1 | 39.8 (6.99,72.6) 50 | 12.83 6.13 74.15 |
| 5 | 5.61 (4.30,6.95) 31 | 6.18 (4.94,7.43) 31 | 16.56 (7.42,25.69) 48 | 6.61 2.58 54.82 |
| 6 | 1.67 (1.26,2.09) 3 | 1.55 (1.29,1.80) 3 | 8.98 (3.1,14.86) 45 | 10.68 4.67 49.25 |
| 7 | 5.50 (4.66,6.33) 30 | 1.71 (1.47,1.95) 4 | N/A | 6.84 2.65 187.9 |
| 8 | 1.47 (1.17,1.78) 3 | 1.55 (1.32,1.79) 3 | 6.46 (3.07,9.85) 44 | 6.45 5.68 73.61 |
| 9 | 1.41 (1.12,1.70) 2 | 1.34 (1.15,1.54) 2 | 6.49 (3.28,9.7) 32 | 13.08 4.59 92.17 |
| Seizure Channels | Non-seizure Channels | |||
|---|---|---|---|---|
| RAST3 | RAST4 | RMST4 | Half Life | |
| Segment | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | Median Min Max |
| 1 | 8.43 (6.39,10.47) 3 | 14.86 (9.97,19.75) 23 | 6.26 (4.89,7.64) 0 | 36.04 12.69 96 |
| 2 | 7.93 (5.61,10.25) 2 | 13.28 (7.10,19.47) 18 | 14.38 (7.94,20.82) 21 | 41.09 12.95 85.08 |
| 3 | 7.38 (5.82,8.93) 2 | 15.51 (10.79,20.24) 16 | 7.84 (6.35,9.32) 3 | 39.87 7.64 90.62 |
| 4 | 10.13 (7.92,12.33) 7 | 6.44 (4.98,7.91) 0 | 13.05 (9.92,16.17) 9 | 34.97 13.22 65.6 |
| 5 | 18.57 (14.58,22.55) 23 | 16.33 (10.77,21.88) 22 | 22.54 (16.63,28.45) 37 | 41.25 8.21 151.69 |
| 6 | 10.31 (7.99,12.62) 6 | 13.41 (8.53,18.20) 14 | 13.39 (9.93,16.85) 11 | 37.05 8.69 127.36 |
| 7 | 13.70 (11.47,15.93) 6 | 29.42 (18.56,40.29) 55 | 41.87 (26.61,57.13) 56 | 40.41 16.16 91.14 |
| 8 | 28.15 (19.96,36.34) 56 | 25.51 (14.18,36.84) 56 | 32.90 (20.28,45.51) 60 | 38.86 1.26 94.01 |
| 9 | 12.05 (9.23,14.97) 7 | 12.26 (8.59,15.93) 13 | 22.84 (16.49,29.19) 43 | 32.98 12.27 93.52 |
| 10 | 12.13 (9.97,14.29) 10 | 10.73 (8.26,13.20) 7 | 20.78 (15.07,26.49) 41 | 34.07 9.67 98.84 |
| Seizure Channels | Non-seizure Channels | |||
|---|---|---|---|---|
| RAST3 | RAST4 | RMST4 | Half Life | |
| Segment | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | Median Min Max |
| 1 | 8.47 (5.74,11.21) 22 | 10.00 (7.10,12.90) 23 | 37.68 (12.44,62.93) 49 | 29.9 2.82 308.76 |
| 2 | 17.07 (12.91,21.22) 29 | 29.08 (12.12,46.04) 43 | 23.14 (8.68,37.61) 49 | 30.64 6.32 134 |
| 3 | 6.32 (5.11,7.54) 24 | 8.27 (5.20,11.35) 25 | 6.23 (3.87,8.59) 27 | 21.72 0.77 126.04 |
| 4 | 12.14 (8.40,15.88) 38 | 181.4 (80.7,282.1) 12 | 36.23 (13.53,58.92) 27 | 9.26 5.42 342.14 |
| 5 | 8.41 (6.59,10.24) 36 | 8.68 (4.51,12.85) 41 | 6.33 (3.94,8.72) 33 | 18.84 6.18 178.51 |
| 6 | 18.86 (13.29,24.44) 41 | 22.62 (5.77,39.47) 59 | 10.51 (6.7,14.33) 40 | 20.32 4.56 136.41 |
| 7 | 15.83 (10.98,20.68) 28 | 72.72 (24.29,121.2) 27 | 6.43 (2.43,10.44) 39 | 11.35 3.63 188.24 |
| 8 | 20.30 (14.19,26.40) 36 | 380.8 (134.7,626.9) 3 | 7.92 (1.45,14.39) 41 | 24.55 4.94 98.15 |
| 9 | 10.76 (7.27,14.24) 37 | 5.94 (4.40,7.49) 23 | 6.42 (2.5,10.34) 32 | 25.01 6.29 106.96 |
| 10 | 25.37 (12.71,38.03) 32 | 44.89 (20.83,68.94) 29 | 6.39 (1.64,11.14) 40 | 14.58 3.16 164.74 |
| Seizure Channels | Non-seizure Channels | |||
|---|---|---|---|---|
| RAST3 | RAST4 | RMST4 | Half Life | |
| Segment | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | Median Min Max |
| 1 | 1.64 (1.34,2.05) 0 | 1.78 (1.57,2.04) 0 | 6.36 (4.23,12.16) 29 | 14.24 6 312.3 |
| 2 | 1.83 (1.45,2.41) 1 | 1.49 (1.29,1.75) 0 | N/A | 12.27 4.03 165.01 |
| 3 | 5.25 (4.41,6.44) 21 | 1.93 (1.7,2.21) 0 | 6.49 (4.23,13.17) 27 | 18.42 3.24 452.16 |
| 4 | 1.9 (1.62,2.28) 0 | 1.79 (1.57,2.06) 0 | 6.33 (3.53,23.46) 38 | 19.56 4.44 152.9 |
| 5 | 1.27 (1.02,1.61) 0 | 1.5 (1.32,1.72) 0 | 6.42 (3.15,99.28) 50 | 8.27 2.16 413.78 |
| 6 | 1.61 (1.35,1.97) 0 | 1.98 (1.75,2.26) 1 | 268.82 (134.36,148185.87) 14 | 29.05 4.24 139.58 |
| Seizure Channels | Non-seizure Channels | |||
|---|---|---|---|---|
| RAST3 | RAST4 | RMST4 | Half Life | |
| Segment | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | Median Min Max |
| 1 | 1.13 (0.90,1.47) 1 | 1.47 (1.22,1.80) 1 | 6.49 (4.06,14.82) 42 | 9.33 1.16 283.79 |
| 2 | 1.72 (1.30,2.39) 0 | 1.88 (1.53,2.37) 0 | 70.74 (36.74,850.50) 23 | 11.43 4.89 166.48 |
| 3 | 1.75 (1.22,2.76) 1 | 1.71 (1.38,2.21) 1 | 13.65 (7.88,46.53) 55 | 18.72 3.06 237.25 |
| 4 | 1.84 (1.44,2.48) 0 | 1.36 (1.16,1.61) 0 | 39.8 (21.74,219.32) 39 | 13.06 6.13 153.46 |
| 5 | 5.62 (4.54,7.31) 30 | 6.18 (5.14,7.72) 30 | 16.56 (10.63,36.42) 38 | 6.61 2.58 54.82 |
| 6 | 1.67 (1.33,2.19) 0 | 1.55 (1.32,1.84) 0 | 8.98 (5.37,24.85) 50 | 10.64 4.67 49.25 |
| 7 | 5.5 (4.76,6.48) 28 | 1.71 (1.50,1.98) 0 | N/A | 7.42 2.65 187.9 |
| 8 | 1.47 (1.21,1.84) 0 | 1.55 (1.34,1.82) 0 | 6.46 (4.19,13.20) 45 | 6.45 5.68 92.53 |
| 9 | 1.41 (1.16,1.75) 0 | 1.34 (1.16,1.56) 0 | 6.49 (4.3,12.53) 36 | 13.26 4.59 94.41 |
| Seizure Channels | Non-seizure Channels | |||
|---|---|---|---|---|
| RAST3 | RAST4 | RMST4 | Half Life | |
| Segment | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | Median Min Max |
| 1 | 8.43 (6.78,11.08) 0 | 14.86 (11.16,22.06) 16 | 6.26 (5.12,8) 0 | 26.15 9.94 64.51 |
| 2 | 7.93 (6.12,11.16) 0 | 13.28 (9.03,24.61) 17 | 14.38 (9.9,25.82) 18 | 29.67 1.19 57.56 |
| 3 | 7.38 (6.08,9.32) 0 | 15.51 (11.87,22.25) 11 | 7.84 (6.58,9.65) 0 | 29.51 6.36 55.36 |
| 4 | 10.13 (8.31,12.92) 3 | 6.44 (5.24,8.31) 0 | 13.05 (10.51,17.12) 4 | 26.31 10.65 47.97 |
| 5 | 18.57 (15.27,23.62) 14 | 16.33 (12.16,24.66) 16 | 22.54 (17.85,30.51) 24 | 30.11 7.08 82.57 |
| 6 | 10.31 (8.40,13.27) 0 | 13.41 (9.80,20.97) 5 | 13.39 (10.63,18.01) 1 | 28 7.14 79.1 |
| 7 | 13.7 (11.77,16.35) 1 | 29.43 (21.47,46.53) 50 | 41.87 (30.66,65.77) 53 | 29.67 11 63.35 |
| 8 | 28.15 (21.79,39.64) 48 | 25.51 (17.63,45.67) 50 | 32.9 (23.75,53.22) 53 | 28.22 1.21 64.22 |
| 9 | 12.05 (9.69,15.87) 2 | 12.26 (9.41,17.43) 6 | 22.84 (17.85,31.58) 36 | 25.75 8.99 729.5 |
| 10 | 12.13 (10.29,14.74) 2 | 10.73 (8.71,13.9) 2 | 20.78 (16.28,28.6) 35 | 26.01 7.98 67.47 |
| Seizure Channels | Non-seizure Channels | |||
|---|---|---|---|---|
| RAST3 | RAST4 | RMST4 | Half Life | |
| Segment | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | HL 95% CI Overlapping Interval Count | Median Min Max |
| 1 | 8.48 (6.39,12.43) 12 | 10.00 (7.73,14.02) 14 | 37.68 (22.51,112.78) 47 | 20.12 2.20 181.18 |
| 2 | 17.07 (13.71,22.52) 24 | 29.08 (18.32,69.11) 44 | 23.14 (14.19,60.8) 46 | 18.25 3.08 85.24 |
| 3 | 6.32 (5.29,7.81) 16 | 8.27 (6.01,13.05) 23 | 6.23 (4.50,9.91) 19 | 15.7 0.49 77.54 |
| 4 | 12.14 (9.26,17.48) 34 | 181.38 (116.58,407.3) 15 | 36.23 (22.22,96.03) 26 | 5.92 2.71 201.51 |
| 5 | 8.41 (6.90,10.72) 22 | 8.68 (5.82,16.44) 34 | 6.33 (4.57,10.04) 22 | 11.69 3.09 109.53 |
| 6 | 18.87 (14.55,26.72) 37 | 22.62 (12.90,85.89) 48 | 10.51 (7.69,16.39) 33 | 13.49 2.67 78.44 |
| 7 | 15.83 (12.10,22.75) 28 | 72.72 (43.60,216.39) 26 | 6.43 (3.91,16.19) 33 | 8.3 2.47 99.04 |
| 8 | 20.3 (15.58,28.97) 27 | 380.8 (231.25,1075.45) 6 | 7.92 (4.29,37.49) 47 | 17.17 2.49 54.15 |
| 9 | 10.76 (8.1,15.83) 25 | 5.94 (4.70,7.99) 15 | 6.42 (3.93,15.72) 25 | 16.65 3.03 59.89 |
| 10 | 25.37 (16.88,50.32) 35 | 44.89 (29.18,96.25) 25 | 6.39 (3.60,22.43) 41 | 10.08 2.7 98.01 |
| Signal | Channel | Mean (s.d.) | Min | Q1 | Med | Q3 | Max |
|---|---|---|---|---|---|---|---|
| Seizure | RAST3 | 2.11 (1.31) | 1.18 | 1.45 | 1.70 | 1.89 | 5.56 |
| RAST4 | 1.86 (0.68) | 1.44 | 1.59 | 1.75 | 1.91 | 5.64 | |
| RMST4 | 22.49 (52.72) | 6.28 | 6.39 | 6.45 | 8.11 | 276.91 | |
| Others | 38.33 (73.79) | 0.16 | 6.44 | 17.15 | 42.05 | 2310.14 | |
| Awake | RAST3 | 2.27 (1.49) | 1.15 | 1.46 | 1.71 | 1.91 | 5.70 |
| RAST4 | 2.61 (1.89) | 1.29 | 1.43 | 1.56 | 2.17 | 6.62 | |
| RMST4 | 20.99 (20.92 | 6.17 | 6.51 | 8.51 | 36.66 | 97.28 | |
| Other | 26.53 (83.71) | 0.21 | 6.4 | 10.55 | 28.06 | 3465.39 | |
| Sleep | RAST3 | 12.92 (6.06) | 5.20 | 9.19 | 11.62 | 15.61 | 50.25 |
| RAST4 | 15.76 (6.96) | 5.95 | 10.06 | 14.43 | 19.04 | 33.30 | |
| RMST4 | 18.39 (8.15) | 5.84 | 11.72 | 17.8 | 23.03 | 36.52 | |
| Other | 56.57 (188.78) | 0.16 | 28.29 | 37.32 | 49.16 | 6931.13 | |
| Sleep/Awake | RAST3 | 22.20 (49.86) | 5.35 | 9.05 | 13.93 | 20.34 | 494.76 |
| RAST4 | 52.46 (114.55) | 5.10 | 9.27 | 14.36 | 45.83 | 692.80 | |
| RMST4 | 14.99 (16.58) | 5.87 | 6.38 | 6.44 | 12.24 | 74.99 | |
| Other | 41.48 (152.17) | 0.16 | 6.48 | 20.28 | 42.97 | 6931.13 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Follis, J.L.; Lai, D. Modeling Volatility Characteristics of Epileptic EEGs using GARCH Models. Signals 2020, 1, 26-46. https://doi.org/10.3390/signals1010003
Follis JL, Lai D. Modeling Volatility Characteristics of Epileptic EEGs using GARCH Models. Signals. 2020; 1(1):26-46. https://doi.org/10.3390/signals1010003
Chicago/Turabian StyleFollis, Jack L., and Dejian Lai. 2020. "Modeling Volatility Characteristics of Epileptic EEGs using GARCH Models" Signals 1, no. 1: 26-46. https://doi.org/10.3390/signals1010003
APA StyleFollis, J. L., & Lai, D. (2020). Modeling Volatility Characteristics of Epileptic EEGs using GARCH Models. Signals, 1(1), 26-46. https://doi.org/10.3390/signals1010003




