Usefulness of Transanal Irrigation and Colon Hydrotherapy in the Treatment of Chronic Constipation and Beyond: A Review with New Perspectives for Bio-Integrated Medicine
Abstract
1. Introduction
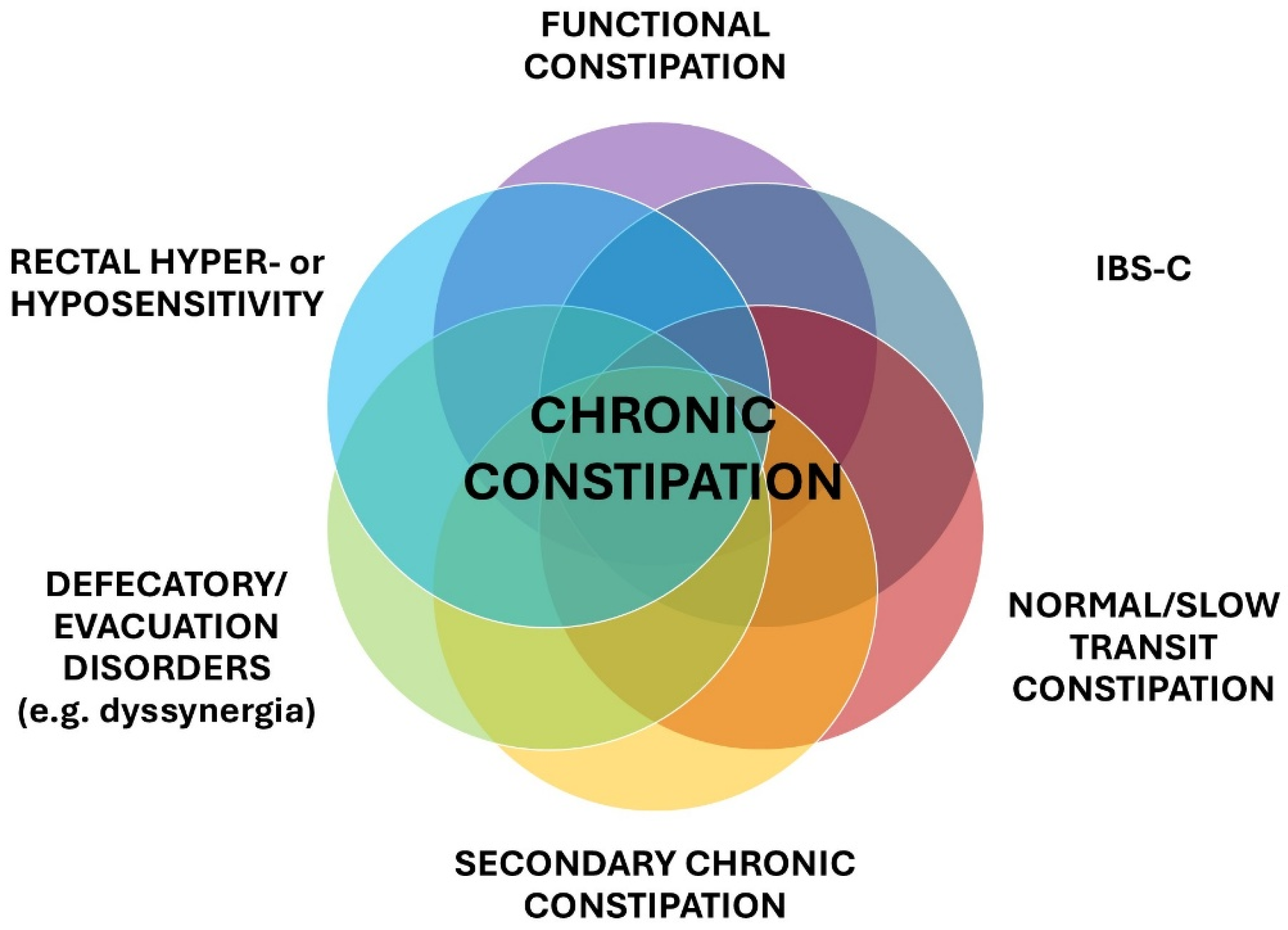
1.1. Intestinal Disorders and Chronic Constipation
- (1)
- Normal transit constipation: This is the most common form, with >50% of constipated patients confirmed by radiopaque marker study, and it is often indistinguishable from IBS-C. Colonic transit falls within the normal range (20–72 h) and show normal ano-rectal function, but patients refer subjective and/or objective symptoms of constipation, mostly associated with bloating and abdominal pain. There may be disturbances in stool consistency, visceral hyposensitivity, or hypersensitivity and alterations in the Enteric Nervous System (ENS). Moreover, significantly higher scores for psychological distress, anxiety, or mood symptoms, which may affect perception of bowel function, are often reported [3].
- (2)
- Slow transit constipation (STC): This is a subtype of chronic primary constipation characterized by abnormally delayed colonic transit in response to meals, in the absence of (or in addition to) outlet obstruction. The estimated prevalence in the general population is about 2–4%. It is more common in women and often presents in younger to middle-aged adults. Because stool remains for longer in the colon, additional absorption of water occurs, resulting in harder stools and infrequent defecation [4].
- (3)
- Defecatory/evacuation disorders: These are the second-most common type of chronic constipation and defecation is impaired due to functional, anatomical, or both causes. In these patients, radiological examinations reveal normal passage of radiopaque markers into the proximal colon, but they then stagnate in the rectum.
- Dietary Changes: Excluding abnormal dietary habits, increasing fiber intake and providing adequate hydration, avoiding trigger foods, or adopting a specific diet (e.g., a low-Fermentable OligoSaccharides, Disaccharides, Monosaccharides, And Polyols (FODMAP) diet, a low-lactose diet, a low-histamine diet, or a gluten-free diet for celiac disease) [9].
- Lifestyle Adjustments: Regular exercise, stress management techniques, adequate sleep, and adequate time and an optimal environment to defecate.
- Medications: Exclude drug side-effects. Treatments to manage symptoms, reduce inflammation, or address underlying causes. In case of hard stools, the main treatments include macrogol, bulking agents, and in case of failure stimulant laxatives (Bisacodyl), prokinetic agents (Prucalopride), secretagogues (Linaclotide and Plecanatide), and bile acid transporter inhibitors (Elobixibat).
- Surgery: In more severe cases, procedures or surgery may be necessary, such as adhesiolysis, colectomy, ventropexy, and rectal resection [10].
- Other Treatments: Biofeedback techniques, particularly indicated for dyssynergic defecation, and recto-colonic water irrigation, which are still being studied by the scientific community today.
1.2. The Physiology of Defecation
- Colonic motility and fecal transport: The process begins with the movement of fecal material from the sigmoid colon into the rectum, facilitated by haustral contractions, slow segmenting movements that mix and propel content, and mass peristalsis, powerful, high-amplitude peristaltic waves that occur a few times a day, often in response to gastrocolic reflex.
- Rectal distension and afferent signaling: As feces enter the rectum, the rectal walls stretch, activating mechanoreceptors in the rectal mucosa. This triggers afferent signals transmitted via the pelvic splanchnic nerves (S2–S4) to the sacral spinal cord. This initiates the recto-anal inhibitory reflex—the internal anal sphincter (IAS), composed of smooth muscle and controlled involuntarily by the autonomic nervous system (ANS), reflexively relaxes and this allows for a small amount of fecal material to contact the anal canal, which is richly innervated and helps the brain assess the kind of contents (solid, liquid, or gas).
- Central integration and conscious perception: The somatosensory cortex and prefrontal cortex interpret the signals and determine the appropriate time and place to defecate. Social and environmental cues influence this decision. If defecation is not appropriate, voluntary contraction of the EAS and puborectalis muscle (innervated by the pudendal nerve) prevents fecal passage.
- Voluntary Defecation Reflex: When defecation is deemed appropriate, the following occurs:
- Relaxation of the EAS and puborectalis muscle, straightening the anorectal angle.
- Increased intra-abdominal pressure via contraction of abdominal muscles and closure of the glottis (the Valsalva maneuver).
- Enhanced rectal contractions, increasing intrarectal pressure.
- These coordinated actions lead to the expulsion of feces through the anal canal.
- Post-defecation recovery: After defecation the rectum returns to baseline tone, IAS and EAS resume contraction, the anal canal closes, and feedback to the brain reduces the sense of urgency [12].
1.3. Neurogenic Bowel Dysfunction: Overview
1.4. Bowel Dysfunction Due to Behavioral or Environmental Factors
- Desensitization of rectal stretch receptors: Repeated inhibition leads to reduced sensitivity of mechanoreceptors in the rectal wall and this increases rectal compliance (the rectum accommodates larger volumes without triggering the urge) [18].
- Disruption of recto-anal coordination (dyssynergic defecation): A learned maladaptive pattern arises where the pelvic floor and EAS contract instead of relaxing during straining. This inevitably leads to functional outlet obstruction [19].
- Fecal retention and hardening: Feces remain in the colon for longer periods, with more water reabsorbed, causing the formation of hard, dry stools and making evacuation more difficult, even when the urge is present [20].
- Habituation and loss of sensory urge: The urge to defecate weakens or disappears over time (a form of “sensory habituation”), especially common in school-aged children and elderly people with mobility or cognitive limitations [21].
2. Biofeedback
- Dyssynergic defecation (functional outlet obstruction), in which patients inappropriately contract the anal sphincter or pelvic floor during defecation. Biofeedback teaches patients to relax these muscles and coordinate abdominal pushing with anal relaxation. Randomized controlled trials have shown that >70% of patients with dyssynergic defecation benefit from biofeedback therapy [24].
- Fecal incontinence: Biofeedback strengthens the EAS and improves rectal sensory discrimination. Patients learn to recognize the urge to defecate earlier and contract the sphincters more effectively [23].
- Urinary incontinence and pelvic floor dysfunction: Often used alongside physical therapy to treat mixed pelvic floor disorders. It appears to enhance muscle strength, timing, and coordination, although there are also conflicting opinions in scientific literature on this topic [25].
3. Transanal Irrigation (TAI)
3.1. TAI Definition
3.2. TAI Mechanism of Action and Indications
3.3. TAI Classification
- Irrigation mechanism: Lukewarm water is introduced into the rectum and colon by means of pressure generated through either a manual or electrically driven pump, ensuring consistent flow and optimal distribution of the irrigation fluid.
- Delivery system: The irrigation medium is administered via a rectal cone or a balloon-retained rectal catheter. Inflation of the balloon serves to secure the catheter in place and minimize leakage during the procedure, particularly in individuals with impaired anal sphincter control secondary to neurological lesions or anatomical alterations of the anorectal complex. There are also TAI systems for children, which can adapt to the anatomy, comfort, and independence of the patient and include low-volume/mini systems, with a cone tip that is inserted gently into the rectum (less invasive and more suitable for younger, smaller, and more sensitive children), or balloon-anchored rectal catheters, which permit more controlled fluid delivery and may support greater autonomy in older children.
- Flow rate regulation: The irrigation flow should be maintained within a range of 200–300 mL/min. This rate has been arbitrarily established to avoid excessive intraluminal pressure that may induce abdominal cramping or reflex rectal contractions, while ensuring sufficient colonic distension to stimulate peristalsis effectively.
- Irrigation volume: The total volume of irrigation fluid typically averages 700 mL, although individual requirements may vary between 250 and 1500 mL, depending on patient tolerance, colonic capacity, and clinical objectives. In pediatric cases, volumes are generally low and weight-dependent.
- Practical implementation: The procedure is designed to be performed by the patient in a seated position, usually on a toilet, enabling a high degree of autonomy and integration into daily bowel care routines. Exceptions include pediatric patients and individuals with significant physical or cognitive limitations, who may require assistance from a caregiver or healthcare professional.
3.4. TAI Benefits
- Relief and prevention of constipation: This provides for the emptying of the lower bowel, reducing constipation and/or fecal impaction, especially in case of the failure of conservative therapies (laxatives or biofeedback training) or pelvic surgery. It may be indicated also for patients who use enemas, rectoclysis, digitation, or other manual maneuvers. TAI seems to reduce symptoms resulting from constipation or inadequate bowel movement with fecal retention, especially bloating and abdominal pain. In addition, it may also possibly reduce or prevent inflammatory conditions of the intestinal mucosa [34,35].
- Relief and prevention of fecal urgency or incontinence: By effectively emptying the bowel, TAI helps prevent uncontrolled bowel movements [36]. TAI may also be indicated for patients with idiopathic rectal hyposensitivity (not due to neurogenic bowel) presenting both symptoms of constipation and fecal incontinence [34].
- Possible improvement of gut microbiota: To date, very few studies have been conducted on the effects of TAI on the microbiota, and they have all been performed on highly selected populations. However, an increase in gut microbiota diversity and a reduction in the proportions of pro-inflammatory taxa been observed in patients with multiple sclerosis and bowel dysfunction, together with significant improvements in gastrointestinal symptoms and quality of life [37]. Positive effects have been registered also in pediatric patients affected by spina bifida who underwent TAI [38].
- Improvement of quality of life: Patients can manage their bowel function and choose the time and place for bowel movements. TAI reduces excessive time spent on bowel care, as well as reliance on toilet access. Moreover, it can give physical and psychological independence and improve quality of social life [34,39].
3.5. TAI Contraindications and Adverse Effects
- (1)
- Minor complications: Abdominal cramps or discomfort, anorectal pain, minor anal/rectal bleeding, leakage of irrigation fluid, or nonspecific or vasovagal symptoms such as nausea, sweating/hot flushes, chills, flushing, headache. Reviews have shown that adverse event rates in TAI patients are similar to those in conservatively managed patients, indicating that some symptoms may stem from the underlying bowel dysfunction rather than the irrigation itself [42].
- (2)
- Procedural complications: Physiological discomfort, catheter or balloon expulsion, bursting of rectal balloons, leakage around device, difficulty in catheter insertion/removal/handling, incomplete bowel evacuation, or residual fluid retention [42].
- (3)
- Rare/serious adverse events: Bowel perforation (estimated approximately 1 in 50.000 irrigations), anorectal fibrosis/stricture due to repeated microtrauma, and autonomic dysreflexia, especially in patients with high spinal cord injury above T6 [43].
4. Colon Hydrotherapy (CHT)
4.1. CHT Benefits
4.2. CHT Possible Risks and Critiques
- Potential adverse effects: As in classic TAI, patients can experience mild symptoms such as cramping, abdominal pain, fullness, bloating, nausea, vomiting, perianal irritation, and soreness. On the other hand, serious adverse events including abscesses, air emboli, rectal perforations, coffee enema-associated colitis, and septicaemia are reported in some very rare cases, especially when there is a lack of adequate anamnestic collection of clinical data (e.g., regarding possible physical/anatomical, pathological, pharmacological, and psychological issues) [47].
- Potential bacterial translocation risk: Water introduced by CHT may disturb the normal flora. However, in clinical practice, intestinal cleansing appears to be effective at rebalancing the intestinal flora, especially in intestinal bacterial overgrowth, even in the absence of supplemental therapeutic support such as rifaximin or probiotics/prebiotics. Regarding the alteration of the mucosal layer, dissemination and absorption of toxins and bacteria into the body [48], this eventuality could occur very rarely and potentially only in very fragile subjects, not ideal candidates for treatment and therefore excluded a priori.
- Potential non-physiological volumes/pressures: Some CHT systems may use large volumes of water under pressures which may be more than what the colon normally experiences. This might risk barotrauma or intestinal perforation if not carried out by non-specialized personnel and/or in the absence of safety systems [49]. However, most devices today are also equipped with safety systems that open the fecal discharge system and release pressure when it increases too much or too suddenly in the colon.
- Potential electrolyte/fluid shifts: Given the exposure to large volumes of incoming water, as well as the active and passive transport of ions by the colon, alterations in fluid and electrolyte balance are possible, especially in sessions that are too frequent.
- Placebo/subjective effects: Some of the reported improvements (reduced bloating, abdominal pain, improved stool frequency and consistency, and general satisfaction) in small studies may be influenced by the placebo effect, a change in diet/hydration, or other concurrent interventions. Rigorous trials are few in number [46].
- Lack of long-term data: At the moment, there are no long-term randomized controlled trials that can confirm some of the claimed benefits.
- Lack of standardization: There is large variability in how the procedure is performed (the volume of water, the temperature, frequency, additives, equipment, and training of the practitioner), making generalization difficult.
5. Gut–Brain Axis Integrated into a Psycho-Neuro-Endocrino-Immuno-Genetic Model
5.1. Gut–Brain Axis in Developmental Phases
5.2. Gut, Neurotransmission, and Neuromodulation
5.3. Gut and Memory
5.4. Intestine and Stress: Irritable Bowel Syndrome
5.5. Psychodynamics and Intestinal Symbolism
5.6. Colon Hydrotherapy for Gastrointestinal Disorders According to Psycho-Neuro-Endocrino-Immunology (PNEI) Model
6. Discussion
7. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ANS | Autonomic Nervous System |
| CHT | Colon Hydrotherapy |
| CMMCs | Colonic Migrating Motor Complexes |
| EAS | External Anal Sphincter |
| EMDR | Eye Movement Desensitization and Reprocessing |
| ENS | Enteric Nervous System |
| FODMAPs | Fermentable Oligo-saccharides, Disaccharides, Monosaccharides, and Polyols |
| GABA | Gamma-Aminobutyric Acid |
| IAS | Internal Anal Sphincter |
| IBS-C | Irritable Bowel Syndrome with Constipation |
| NBD | Neurogenic Bowel Dysfunction |
| PNEI | Psycho-Neuro-Endocrino-Immunology |
| SNS | Somatic Nervous System |
| STC | Slow Transit constipation |
| TAI | Transanal Irrigation |
References
- Barbara, G.; Cremon, C.; Bellini, M.; Corsetti, M.; Di Nardo, G.; Falangone, F.; Fuccio, L.; Galeazzi, F.; Iovino, P.; Sarnelli, G.; et al. Italian guidelines for the management of irritable bowel syndrome: Joint Consensus from the Italian Societies of: Gastroenterology and Endoscopy (SIGE), Neurogastroenterology and Motility (SINGEM), Hospital Gastroenterologists and Endoscopists (AIGO), Digestive Endoscopy (SIED), General Medicine (SIMG), Gastroenterology, Hepatology and Pediatric Nutrition (SIGENP) and Pediatrics (SIP). Dig. Liver Dis. 2023, 55, 187–207. [Google Scholar] [PubMed]
- Siah, K.T.; Wong, R.K.; Whitehead, W.E. Chronic Constipation and Constipation-Predominant IBS: Separate and Distinct Disorders or a Spectrum of Disease? Gastroenterol. Hepatol. 2016, 12, 171–178. [Google Scholar]
- Lembo, A.; Camilleri, M. Chronic constipation. N. Engl. J. Med. 2003, 349, 1360–1368. [Google Scholar] [CrossRef] [PubMed]
- Vlismas, L.J.; Wu, W.; Ho, V. Idiopathic Slow Transit Constipation: Pathophysiology, Diagnosis, and Management. Medicina 2024, 60, 108. [Google Scholar] [CrossRef]
- Bassotti, G.; Chistolini, F.; Nzepa, F.S.; Morelli, A. Colonic propulsive impairment in intractable slow-transit constipation. Arch. Surg. 2003, 138, 1302–1304. [Google Scholar] [CrossRef][Green Version]
- Vermorken, A.J.; Andrès, E.; Cui, Y. Bowel movement frequency, oxidative stress and disease prevention. Mol. Clin. Oncol. 2016, 5, 339–342. [Google Scholar] [CrossRef]
- Han, K.; Kuo, B.; Khalili, H.; Staller, K. Metagenomics Analysis Reveals Unique Gut Microbiota Signature of Slow-Transit Constipation. Clin. Transl. Gastroenterol. 2024, 15, e1. [Google Scholar] [CrossRef]
- Bharucha, A.E.; Lacy, B.E. Mechanisms, Evaluation, and Management of Chronic Constipation. Gastroenterology 2020, 158, 1232–1249.e3. [Google Scholar] [CrossRef]
- Borghini, R.; Spagnuolo, A.; Donato, G.; Borghini, G. Gluten-Free Diet for Fashion or Necessity? Review with New Speculations on Irritable Bowel Syndrome-like Disorders. Nutrients 2024, 16, 4236. [Google Scholar] [CrossRef]
- Vitton, V.; Damon, H.; Benezech, A.; Bouchard, D.; Brardjanian, S.; Brochard, C.; Coffin, B.; Fathallah, N.; Higuero, T.; Jouët, P.; et al. Clinical practice guidelines from the French National Society of Coloproctology in treating chronic constipation. Eur. J. Gastroenterol. Hepatol. 2018, 30, 357–363. [Google Scholar] [CrossRef]
- Santucci, N.R.; Velez, A. Physiology of lower gastrointestinal tract. Aliment. Pharmacol. Ther. 2024, 60, S1–S19. [Google Scholar] [CrossRef]
- Bharucha, A.E.; Pemberton, J.H.; Locke, G.R., 3rd. American Gastroenterological Association technical review on constipation. Gastroenterology 2013, 144, 218–238. [Google Scholar] [CrossRef] [PubMed]
- Emmanuel, A. Neurogenic bowel dysfunction. F1000Research 2019, 8, F1000 Faculty Rev-1800. [Google Scholar] [CrossRef] [PubMed]
- Preziosi, G.; Gordon-Dixon, A.; Emmanuel, A. Neurogenic bowel dysfunction in patients with multiple sclerosis: Prevalence, impact, and management strategies. Degener. Neurol. Neuromuscul. Dis. 2018, 8, 79–90. [Google Scholar] [CrossRef] [PubMed]
- Fasano, A.; Visanji, N.P.; Liu, L.W.; Lang, A.E.; Pfeiffer, R.F. Gastrointestinal dysfunction in Parkinson’s disease. Lancet Neurol. 2015, 14, 625–639. [Google Scholar] [CrossRef]
- Camilleri, M. Gastrointestinal motility disorders in neurologic disease. J. Clin. Investig. 2021, 131, e143771. [Google Scholar] [CrossRef]
- Whitehead, W.E.; Borrud, L.; Goode, P.S.; Meikle, S.; Mueller, E.R.; Tuteja, A.; Weidner, A.; Weinstein, M.; Ye, W. Pelvic Floor Disorders Network. Fecal incontinence in US adults: Epidemiology and risk factors. Gastroenterology 2009, 137, 512–517. [Google Scholar] [CrossRef]
- Rao, S.S. Constipation: Evaluation and treatment of colonic and anorectal motility disorders. Gastroenterol. Clin. N. Am. 2007, 36, 687–711. [Google Scholar] [CrossRef]
- Bharucha, A.E.; Rao, S.S. An update on anorectal disorders for gastroenterologists. Gastroenterology 2014, 146, 37–45.e2. [Google Scholar] [CrossRef]
- Camilleri, M.; Ford, A.C.; Mawe, G.M.; Dinning, P.G.; Rao, S.S.; Chey, W.D.; Simrén, M.; Lembo, A.; Young-Fadok, T.M.; Chang, L. Chronic constipation. Nat. Rev. Dis. Primers 2017, 3, 17095. [Google Scholar] [CrossRef]
- Benninga, M.A.; Faure, C.; Hyman, P.E.; St James Roberts, I.; Schechter, N.L.; Nurko, S. Childhood Functional Gastrointestinal Disorders: Neonate/Toddler. Gastroenterology 2016, 130, 1519–1526. [Google Scholar] [CrossRef]
- Chiarioni, G.; Whitehead, W.E.; Pezza, V.; Morelli, A.; Bassotti, G. Biofeedback is superior to laxatives for normal transit constipation due to pelvic floor dyssynergia. Gastroenterology 2006, 130, 657–664. [Google Scholar] [CrossRef] [PubMed]
- Heymen, S.; Scarlett, Y.; Jones, K.; Ringel, Y.; Drossman, D.; Whitehead, W.E. Randomized controlled trial shows biofeedback to be superior to pelvic floor exercises for fecal incontinence. Dis. Colon Rectum 2009, 52, 1730–1737. [Google Scholar] [CrossRef] [PubMed]
- Rao, S.S.; Welcher, K.D.; Leistikow, J.S. Obstructive defecation: A failure of rectoanal coordination. Am. J. Gastroenterol. 1998, 93, 1042–1050. [Google Scholar] [CrossRef] [PubMed]
- Nunes, E.F.C.; Sampaio, L.M.M.; Biasotto-Gonzalez, D.A.; Nagano, R.C.D.R.; Lucareli, P.R.G.; Politti, F. Biofeedback for pelvic floor muscle training in women with stress urinary incontinence: A systematic review with meta-analysis. Physiotherapy 2019, 105, 10–23. [Google Scholar] [CrossRef]
- Rao, S.S.; Patcharatrakul, T. Diagnosis and Treatment of Dyssynergic Defecation. J. Neurogastroenterol. Motil. 2016, 22, 423–435. [Google Scholar] [CrossRef] [PubMed]
- Bassotti, G.; Usai Satta, P.; Bellini, M. Chronic Idiopathic Constipation in Adults: A Review on Current Guidelines and Emerging Treatment Options. Clin. Exp. Gastroenterol. 2021, 14, 413–428. [Google Scholar] [CrossRef]
- Rodriguez, G.; Kirshblum, S.; Thao, M.; McArthur, M.; Camilleri, M. The Functional Implications of Transanal Irrigation: Insights from Pathophysiology and Clinical Studies of Neurogenic Bowel Dysfunction. J. Clin. Med. 2024, 13, 1527. [Google Scholar] [CrossRef]
- Costa, M.; Wiklendt, L.; Simpson, P.; Spencer, N.J.; Brookes, S.J.; Dinning, P.G. Neuromechanical factors involved in the formation and propulsion of fecal pellets in the guinea-pig colon. Neurogastroenterol. Motil. 2015, 27, 1466–1477. [Google Scholar] [CrossRef]
- Spencer, N.J.; Dinning, P.G.; Brookes, S.J.; Costa, M. Insights into the mechanisms underlying colonic motor patterns. J. Physiol. 2016, 594, 4099–4116. [Google Scholar] [CrossRef]
- Chey, W.Y.; Chang, V.; Hoellrich, C.M.; Lee, K.Y.; Lubkin, M.; Corcoran, D.; Chang, T.M.; Chey, W.D. Mega-Cecum: An Unrecognized Cause of Symptoms in Some Female Patients with Uro-Gynecological Symptoms and Severe Slow Transit Constipation. Dig. Dis. Sci. 2017, 62, 217–223. [Google Scholar] [CrossRef] [PubMed]
- Chey, W.Y.; Chang, V.; Lubukin, M.; Lee, K.Y.; Hoellrich, C.M.; Chey, W.D. Mega-cecum: A novel clinical entity which explains overlapping gastrointestinal and urogynecologic symptoms in women with chronic constipation. Am. J. Gastroenterol. 2013, 108, 1665–1666. [Google Scholar] [CrossRef] [PubMed]
- Emmanuel, A.; Krogh, K.; Perrouin-Verbe, B.; Krassiukov, A.; Koch, S.; Mosiello, G.; Bazzocchi, G.; Christensen, P.; Rodriguez, G.; Brugaletta, C. Consensus on Safe Initiation and Monitoring of Transanal Irrigation to Optimize Adherence with Therapy. Neurogastroenterol. Motil. 2025, 37, e15011. [Google Scholar] [CrossRef] [PubMed]
- Bazzocchi, G.; Corazziari, E.S.; Staiano, A.; Bassotti, G.; Bellini, M.; Chiarioni, G.; D’Alba, L.; Scarpato, E. Position paper on transanal irrigation in chronic non-organic constipation. Dig. Liver Dis. 2024, 56, 770–777. [Google Scholar] [CrossRef]
- Raahave, D.; Christensen, E.; Loud, F.B.; Knudsen, L.L. Correlation of bowel symptoms with colonic transit, length, and faecal load in functional faecal retention. Dan. Med. Bull. 2009, 56, 83–88. [Google Scholar]
- Vollebregt, P.F.; Dekker, L.; Han-Geurts, I.J.M.; Felt-Bersma, R.J.F. Prospective cohort study of high-volume transanal irrigation in patients with constipation and/or faecal incontinence. Colorectal Dis. 2023, 25, 1658–1670. [Google Scholar] [CrossRef]
- Ascanelli, S.; Bombardini, C.; Chimisso, L.; Carcoforo, P.; Turroni, S.; D’Amico, F.; Caniati, M.L.; Baldi, E.; Tugnoli, V.; Morotti, C.; et al. Trans-Anal irrigation in patients with multiple sclerosis: Efficacy in treating disease-related bowel dysfunctions and impact on the gut microbiota: A monocentric prospective study. Mult. Scler. J. Exp. Transl. Clin. 2022, 8, 20552173221109771. [Google Scholar] [CrossRef]
- Furuta, A.; Suzuki, Y.; Takahashi, R.; Jakobsen, B.P.; Kimura, T.; Egawa, S.; Yoshimura, N. Effects of Transanal Irrigation on Gut Microbiota in Pediatric Patients with Spina Bifida. J. Clin. Med. 2021, 10, 224. [Google Scholar] [CrossRef]
- Rosen, H.; Robert-Yap, J.; Tentschert, G.; Lechner, M.; Roche, B. Transanal irrigation improves quality of life in patients with low anterior resection syndrome. Colorectal Dis. 2011, 13, e335–e338. [Google Scholar] [CrossRef]
- Bildstein, C.; Melchior, C.; Gourcerol, G.; Boueyre, E.; Bridoux, V.; Vérin, E.; Leroi, A.M. Predictive factors for compliance with transanal irrigation for the treatment of defecation disorders. World J. Gastroenterol. 2017, 23, 2029–2036. [Google Scholar] [CrossRef]
- Chesnel, C.; Hentzen, C.; Haddad, R.; Charlanes, A.; Le Breton, F.; Turmel, N.; Amarenco, G. Adherence to transanal irrigation in older adults: First-year assessment. Tech. Coloproctol. 2021, 25, 1055–1063. [Google Scholar] [CrossRef] [PubMed]
- Christensen, P.; Krogh, K.; Buntzen, S.; Payandeh, F.; Laurberg, S. Long-term outcome and safety of transanal irrigation for constipation and fecal incontinence. Dis. Colon Rectum 2009, 52, 286–292. [Google Scholar] [CrossRef] [PubMed]
- Morini, A.; Fabozzi, M.; Zanelli, M.; Sanguedolce, F.; Palicelli, A.; Annicchiarico, A.; Bonelli, C.; Zizzo, M. Transanal Irrigation in Patients with Low Anterior Resection Syndrome After Rectal-Sphincter-Preserving Surgery for Oncological and Non-Oncological Disease: A Systematic Review. Surg. Tech. Dev. 2024, 13, 409–425. [Google Scholar] [CrossRef]
- Zhang, D.; Zhao, C.; Tao, Y.; Zhang, J.; Zhang, Q.; Li, D.; Ye, P.; Yu, X.; Chen, C. Remedial colon hydrotherapy device enema as a salvage strategy for inadequate bowel preparation for colonoscopy: A retrospective cohort study. PLoS ONE 2025, 20, e0319493. [Google Scholar] [CrossRef]
- Taffinder, N.J.; Tan, E.; Webb, I.G.; McDonald, P.J. Retrograde commercial colonic hydrotherapy. Colorectal Dis. 2004, 6, 258–260. [Google Scholar] [CrossRef]
- Hsu, H.H.; Leung, W.H.; Hu, G.C. Treatment of irritable bowel syndrome with a novel colonic irrigation system: A pilot study. Tech. Coloproctol. 2016, 20, 551–557. [Google Scholar] [CrossRef]
- Stern, B.; Chintanaboina, J. Water-Only Colon Cleansing Causing Acute Colitis: 1401. Am. J. Gastroenterol. 2016, 111, S632–S633. [Google Scholar] [CrossRef]
- Seow-Choen, F. The physiology of colonic hydrotherapy. Colorectal Dis. 2009, 11, 686–688. [Google Scholar] [CrossRef]
- Rzouq, F.S.; Hatoum, H.H. Commercial Colon Hydrotherapy: A Common Practice with Serious Complications: 939. Am. J. Gastroenterol. 2010, 105, S340. [Google Scholar] [CrossRef]
- Scalabrini, A.; Mucci, C.; Northoff, G. The nested hierarchy of self and its trauma: In search for a synchronic dynamic and topographical re-organization. Front. Hum. Neurosci. 2022, 16, 980353. [Google Scholar] [CrossRef]
- Averina, O.V.; Danilenko, V.N. Human Intestinal Microbiota: Role in Development and Functioning of the Nervous System. Mikrobiologiia 2017, 86, 5–24. [Google Scholar] [CrossRef] [PubMed]
- Cryan, J.F.; O’Riordan, K.J.; Cowan, C.S.M.; Sandhu, K.V.; Bastiaanssen, T.F.S.; Boehme, M.; Codagnone, M.G.; Cussotto, S.; Fulling, C.; Golubeva, A.V.; et al. The Microbiota-Gut-Brain Axis. Physiol. Rev. 2019, 99, 1877–2013. [Google Scholar] [CrossRef] [PubMed]
- Gajić Bojić, M.; Aranđelović, J.; Škrbić, R.; Savić, M.M. Peripheral GABAA receptors—Physiological relevance and therapeutic implications. Pharmacol. Ther. 2025, 266, 108759. [Google Scholar] [CrossRef] [PubMed]
- Greenamyre, J.T. The role of glutamate in neurotransmission and in neurologic disease. Arch. Neurol. 1986, 43, 1058–1063. [Google Scholar] [CrossRef]
- Yassin, L.K.; Nakhal, M.M.; Alderei, A.; Almehairbi, A.; Mydeen, A.B.; Akour, A.; Hamad, M.I.K. Exploring the microbiota-gut-brain axis: Impact on brain structure and function. Front. Neuroanat. 2025, 19, 1504065. [Google Scholar] [CrossRef]
- Scalabrini, A.; Mucci, C.; Northoff, G. Is Our Self Related to Personality? A Neuropsychodynamic Model. Front. Hum. Neurosci. 2018, 12, 346. [Google Scholar] [CrossRef]
- McCoy, K.D.; Burkhard, R.; Geuking, M.B. The microbiome and immune memory formation. Immunol. Cell Biol. 2019, 97, 625–635. [Google Scholar] [CrossRef]
- Zheng, D.; Liwinski, T.; Elinav, E. Interaction between microbiota and immunity in health and disease. Cell Res. 2020, 30, 492–506. [Google Scholar] [CrossRef]
- Lin, X.; Han, H.; Wang, N.; Wang, C.; Qi, M.; Wang, J.; Liu, G. The Gut Microbial Regulation of Epigenetic Modification from a Metabolic Perspective. Int. J. Mol. Sci. 2024, 25, 7175. [Google Scholar] [CrossRef]
- Qin, H.Y.; Cheng, C.W.; Tang, X.D.; Bian, Z.X. Impact of psychological stress on irritable bowel syndrome. World J. Gastroenterol. 2014, 20, 14126–14131. [Google Scholar] [CrossRef]
- Lacy, B.E.; Pimentel, M.; Brenner, D.M.; Chey, W.D.; Keefer, L.A.; Long, M.D.; Moshiree, B. ACG Clinical Guideline: Management of Irritable Bowel Syndrome. Am. J. Gastroenterol. 2021, 116, 17–44. [Google Scholar] [CrossRef] [PubMed]
- Császár-Nagy, N.; Bókkon, I. Hypnotherapy and IBS: Implicit, long-term stress memory in the ENS? Heliyon 2022, 9, e12751. [Google Scholar] [CrossRef] [PubMed]
- Ferrari, S.; Poloni, N.; Diefenbacher, A.; Barbosa, A.; Cosci, F. From hysteria to somatic symptom disorders: Searching for a common psychopathological ground. J. Psychopathol. 2015, 21, 372–379. [Google Scholar]
- Radziwon, C.D.; Lackner, J.M. Cognitive Behavioral Therapy for IBS: How Useful, How Often, and How Does It Work? Curr. Gastroenterol. Rep. 2017, 19, 49. [Google Scholar] [CrossRef]
- Brook, A. Bowel distress and emotional conflict. J. R. Soc. Med. 1991, 84, 39–42. [Google Scholar] [CrossRef]
- Grifoni, J.; Pagani, M.; Persichilli, G.; Bertoli, M.; Bevacqua, M.G.; L’Abbate, T.; Flamini, I.; Brancucci, A.; Cerniglia, L.; Paulon, L.; et al. Auditory Personalization of EMDR Treatment to Relieve Trauma Effects: A Feasibility Study [EMDR+]. Brain Sci. 2023, 13, 1050. [Google Scholar] [CrossRef]
- Byrne, C.M.; Sharma, A.; Telford, K. Trans-anal irrigation should be offered to patients with severe bowel dysfunction. Br. J. Gen. Pract. 2019, 69, 136. [Google Scholar] [CrossRef]
- Ausili, E.; Marte, A.; Brisighelli, G.; Midrio, P.; Mosiello, G.; La Pergola, E.; Lombardi, L.; Iacobelli, B.D.; Caponcelli, E.; Meroni, M.; et al. Short versus mid-long-term outcome of transanal irrigation in children with spina bifida and anorectal malformations. Childs Nerv. Syst. 2018, 34, 2471–2479. [Google Scholar] [CrossRef]
- Ji, Y.; Ji, J.E.; Kim, B.; Han, S.W.; Lee, Y.S.; Kim, S.W.; Choi, E.K. Long-term outcome of transanal irrigation for individuals with spina bifida: A 12-year experience study. Tech. Coloproctol. 2024, 28, 159. [Google Scholar] [CrossRef]
- Loftus, C.; Wallace, E.; McCaughey, M.; Smith, E. Transanal irrigation in the management of neurogenic bowel dysfunction. Ir. Med. J. 2012, 105, 241–243. [Google Scholar]
- Mahishale, A.; Tahiliani, S.; Nayak, S. Pelvic Floor Rehabilitation in Chronic Constipation Associated with Dyspareunia: A Case Report. Altern. Ther. Health Med. 2025, 31, AT11585. [Google Scholar] [PubMed]
- Gold, D.M.; Swash, M.; Farag, A.; Ding, S.; Santoro, G.; Dodi, G. A brief physiology and pathophysiology of the anorectum based on the Integral Theory paradigm. Ann. Transl. Med. 2024, 12, 25. [Google Scholar] [CrossRef] [PubMed]
- Magri, V.; Boltri, M.; Cai, T.; Colombo, R.; Cuzzocrea, S.; De Visschere, P.; Giuberti, R.; Granatieri, C.M.; Latino, M.A.; Larganà, G.; et al. Multidisciplinary approach to prostatitis. Arch. Ital. Urol. Androl. 2019, 90, 227–248. [Google Scholar] [CrossRef] [PubMed]
- Podzemny, V.; Pescatori, L.C.; Pescatori, M. Management of obstructed defecation. World J. Gastroenterol. 2015, 21, 1053–1060. [Google Scholar] [CrossRef]
- Orlandi, S.; Bocus, P.; Geccherle, A.; Ruffo, G.; Ceccaroni, M. Use of transanal irrigation (TAI) in the treatment of persistent bowel disorders in patients with endometriosis: A retrospective study. Int. J. Colorectal Dis. 2024, 39, 182. [Google Scholar] [CrossRef]
- Brown, A.C.; Hairfield, M.; Richards, D.G.; McMillin, D.L.; Mein, E.A.; Nelson, C.D. Medical nutrition therapy as a potential complementary treatment for psoriasis—Five case reports. Altern. Med. Rev. 2004, 9, 297–307. [Google Scholar]





| NEUROLOGIC AND MOTILITY DISORDERS | MEDICATIONS |
|---|---|
| Amyloidosis | Anticholinergics |
| Diabetes | Anticonvulsants |
| Hirschprung Disease | Antidepressants |
| Hypothyroidism | Antipsychotics |
| Multiple Sclerosis | Antispasmodics |
| Parkinson Disease | Calcium channel blockers |
| Spinal cord injury | Opioids |
| Spinal or ganglionic tumors | |
| Stroke | |
| DISEASE IN WHICH TREATMENT CAN CAUSE CONSTIPATION | OTHER CAUSES |
| Bipolar disorder | Chagas disease |
| Chronic pain | Conversion disorder |
| Depression | Decreased fluid intake |
| Parkinson disease | Hypercalcemia |
| Schizophrenia | Hyperparathyroidism |
| Low-fiber diet | |
| Mechanical obstruction | |
| IBS-like disorders (e.g., celiac disease, non-celiac gluten sensitivity, SIBO, FODMAPs, nickel allergy, histamine intolerance, and lactose intolerance) |
| NEUROGENIC BOWEL DYSFUNCTION | MECHANISM OF IMPAIRMENT | SYMPTOMS | |
| Spinal Cord Injuries | Suprasacral lesions (above S2–S4) | reflexive bowel intact, loss of voluntary control, hyperreflexia, slow colonic transit, outlet dysfunction | constipation, reflex defecation |
| Sacral or infrasacral lesions (S2–S4) | areflexic bowel, reduced motility, pelvic splanchnic nerves and pudendal nerves damaged, weak or absent peristalsis in distal colon and rectum, flaccid anal sphincters | fecal incontinence, absent rectal tone, constipation | |
| Neurological Disorders Affecting Defecation | Multiple Sclerosis | central coordination impaired, voluntary and reflex bowel control possibly damaged | constipation, fecal incontinence |
| Parkinson’s Disease | central coordination impaired, delayed colonic transit, delayed colonic transit, reduced rectal contraction and impaired coordination | constipation | |
| Stroke | central coordination impaired, voluntary control possibly impaired | fecal incontinence, constipation | |
| Diabetic Autonomic Neuropathy | autonomic nerves damaged (including pelvic splanchnic), colonic hypomotility | fecal incontinence, rectal hyposensitivity, constipation | |
| Spina Bifida and Myelomeningocele | sacral nerves affected, areflexic bowel | fecal incontinence, costipation | |
| Pelvic Nerve Injury | Pudendal Neuropathy | external anal sphincter/puborectalis muscle damaged | fecal incontinence, incomplete emptying (dyssynergia) |
| Pelvic Splanchnic Nerve Damage (S2–S4) | parasympathetic input to rectum and sigmoid colon disrupted, motility reduced, impaired internal sphincter reflex | constipation, fecal incontinence | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Borghini, R.; Borghini, F.; Spagnuolo, A.; Borghini, A.; Borghini, G. Usefulness of Transanal Irrigation and Colon Hydrotherapy in the Treatment of Chronic Constipation and Beyond: A Review with New Perspectives for Bio-Integrated Medicine. Gastrointest. Disord. 2026, 8, 6. https://doi.org/10.3390/gidisord8010006
Borghini R, Borghini F, Spagnuolo A, Borghini A, Borghini G. Usefulness of Transanal Irrigation and Colon Hydrotherapy in the Treatment of Chronic Constipation and Beyond: A Review with New Perspectives for Bio-Integrated Medicine. Gastrointestinal Disorders. 2026; 8(1):6. https://doi.org/10.3390/gidisord8010006
Chicago/Turabian StyleBorghini, Raffaele, Francesco Borghini, Alessia Spagnuolo, Agnese Borghini, and Giovanni Borghini. 2026. "Usefulness of Transanal Irrigation and Colon Hydrotherapy in the Treatment of Chronic Constipation and Beyond: A Review with New Perspectives for Bio-Integrated Medicine" Gastrointestinal Disorders 8, no. 1: 6. https://doi.org/10.3390/gidisord8010006
APA StyleBorghini, R., Borghini, F., Spagnuolo, A., Borghini, A., & Borghini, G. (2026). Usefulness of Transanal Irrigation and Colon Hydrotherapy in the Treatment of Chronic Constipation and Beyond: A Review with New Perspectives for Bio-Integrated Medicine. Gastrointestinal Disorders, 8(1), 6. https://doi.org/10.3390/gidisord8010006






