Effects of Inhibitors on Corrosion Resistance of Acrylic–Amino Resin Coatings in Alkaline Solution for Industrial Measuring Tapes
Abstract
1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Sample Preparation
2.3. Characterization
2.4. Hydrophilicity
2.5. Electrochemical Corrosion
2.6. Long-Term Alkaline Immersion
3. Results
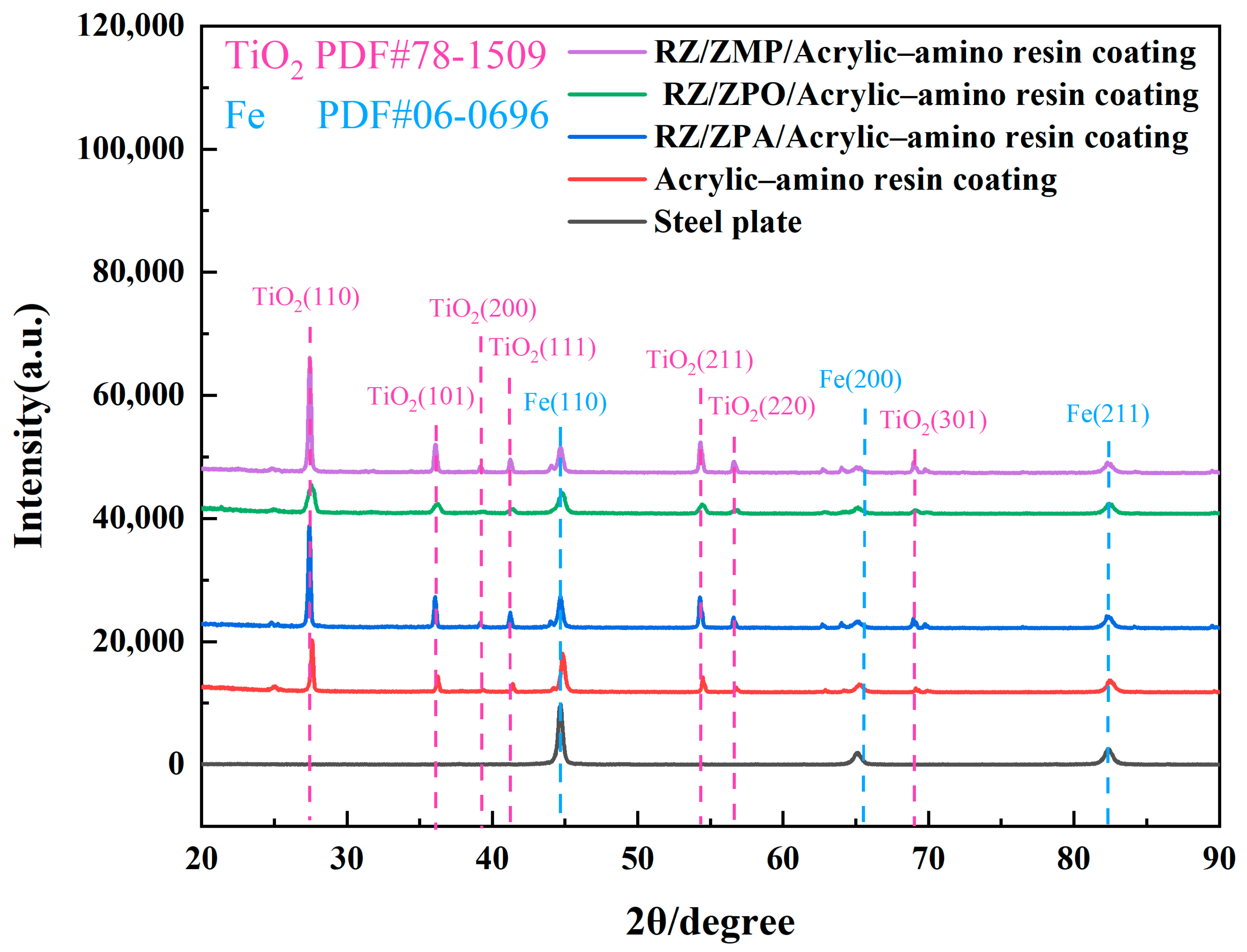
3.1. Characterization
3.2. Long-Term Alkaline Immersion
3.3. Electrochemical Corrosion Resistance
3.4. Corrosion Mechanism
4. Conclusions
Author Contributions
Funding
Data Availability Statement
Conflicts of Interest
References
- Saxena, D.; Vishwakarma, A.; M., V. Use of Primary Steel Rebar and Its New Techniques Used in High-Rise Building in Construction Industries: A Review. IJRASET 2022, 10, 1803–1808. [Google Scholar] [CrossRef]
- Qureshia, T.; Wang, G.; Mukherjeea, S.; Islama, M.A.; Filleter, T.; Singh, C.V.; Panesar, D.K. Graphene-based anti-corrosive coating on steel for reinforced concrete infrastructure applications: Challenges and potential. Constr. Build. Mater. 2022, 351, 128947. [Google Scholar] [CrossRef]
- Zhao, Z.; Mei, Y.; Zhang, A.; Zeng, C.; Li, F.; Huang, L.; Xu, H. Research on Impact Compression Strength and Explosion Resistance of Layered Gradient Rubber Concrete in Underground Engineering. IJST-Trans. Civ. Eng. 2025, 49, 2427–2444. [Google Scholar] [CrossRef]
- Lloyd, R.R.; Provis, J.L.; van Deventer, J.S.J. Pore solution composition and alkali diffusion in inorganic polymer cement. Cem. Concr. Res. 2010, 40, 1386–1392. [Google Scholar] [CrossRef]
- Nürnberger, U. Corrosion of metals in contact with mineral building materials. In Corrosion of Reinforcement in Concrete; Woodhead Publishing: Cambridge, UK, 2007; pp. 1–9. [Google Scholar]
- Ortolan, V.K.; Mancio, M.; Tutikian1, B.F. Evaluation of the influence of the pH of concrete pore solution on the corrosion resistance of steel reinforcement. J. Build. Pathol. Rehabil. 2016, 1, 10. [Google Scholar] [CrossRef]
- Motlatle, A.M.; Ray, S.S.; Ojijo, V.; Scriba, M.R. Polyester-Based Coatings for Corrosion Protection. Polymers 2022, 14, 3413. [Google Scholar] [CrossRef]
- Abadchi, M.R.; Mirzaee, M.; Dorkhani, E.; Zolriasatein, A.; Noori, N.R. Surface modification of acrylic coating with anti-corrosion and anti-UV materials. Chin. Chem. Soc. 2022, 69, 912–924. [Google Scholar] [CrossRef]
- Chopra, I.; Ola, S.K.; Priyanka; Dhayal, V.; Shekhawat, D.S. Recent advances in epoxy coatings for corrosion protection of steel: Experimental and modelling approach—A review. Mater. Today 2022, 63, 1658–1663. [Google Scholar]
- Cao, H.; Lyu, Z.; Dong, W.; Zhao, Z.; Gan, W.; Wang, Y. Corrosion Experimental Research on Local Damage of Epoxy-Coated Steel Bars in Concrete Under Marine Environment. Front. Mater. 2022, 8. [Google Scholar] [CrossRef]
- Ren, K.; Zhang, J.; Liu, Y.; Hou, Q.; Zhang, R.; Krishnamurthy, M.; Zuo, Z.; Zhao, Y.; Yu, Z. Failure mechanisms and corrosion behavior of epoxy-based protective coatings prepared on 20# steel exposed to cooling water. Int. J. Electrochem. Sci. 2025, 20, 101207. [Google Scholar]
- Puig, M.; Gimeno, M.J.; Gracenea, J.J.; Suay, J.J. Anticorrosive properties enhancement in powder coating duplex systems by means of ZMP anticorrosive pigment. Assessment by electrochemical techniques. Prog. Org. Coat. 2014, 77, 1993–1999. [Google Scholar] [CrossRef]
- Gimeno, M.J.; Chamorro, S.; March, R.; Oró, E.; Pérez, P.; Gracenea, J.; Suay, J. Anticorrosive properties enhancement by means of phosphate pigments in an epoxy 2k coating. Assessment by NSS and ACET. Prog. Org. Coat. 2014, 77, 2024–2030. [Google Scholar] [CrossRef]
- Wan, H.; Song, D.; Li, X.; Zhang, D.; Gao, J.; Du, C. Effect of Zinc Phosphate on the Corrosion Behavior of Waterborne Acrylic Coating/Metal Interface. Materials 2017, 10, 654. [Google Scholar] [CrossRef]
- Zhang, W.; Xia, W.; Chen, Z.; Zhang, G.; Qian, S.; Lin, Z. Comparison of the cathodic protection of epoxy resin coating/Zinc-rich coatings on defective areas under atmospheric and immersion conditions: The secondary activation of Zinc Particles. Coatings 2024, 14, 336. [Google Scholar] [CrossRef]
- Zin, I.M.; Lyon, S.B.; Pokhmurskii, V.I. Corrosion control of galvanized steel using a phosphate/calcium ion inhibitor mixture. Corros. Sci. 2003, 45, 777–788. [Google Scholar] [CrossRef]
- Pokhmurs’kyi, V.I.; Zin’, I.M.; Layon, S.B.; Bilyi, L.M. Synergistic effect of phosphate and calcium-containing pigments on the corrosion resistance of galvanized steel. Mater. Sci. 2003, 39, 153–160. [Google Scholar] [CrossRef]
- Samad, U.A.; Alam, M.A.; Abdo, H.S.; Al-Zahrani, S.M. Synergistic effect of nanoparticles: Enhanced mechanical and corrosion protection properties of epoxy coatings incorporated with SiO2 and ZrO2. Polymers 2023, 15, 3100. [Google Scholar] [CrossRef] [PubMed]
- Yang, S.; Huang, J.; Chen, J.; Noël, J.J.; Barker, I.; Henderson, J.D.; He, P.; Zhang, H.; Zhang, H.; Zhu, J. A comparative study on the anti-corrosive performance of zinc phosphate in powder coatings. Coatings 2022, 12, 217. [Google Scholar] [CrossRef]
- GB/T 699-2015; Steel for Mechanical Structure. Standardization Administration of China: Beijing, China, 2015.
- Muthee, D.K.; Dejene, B.F. Effect of annealing temperature on structural, optical, and photocatalytic properties of titanium dioxide nanoparticles. Heliyon 2021, 7, e07269. [Google Scholar] [CrossRef] [PubMed]
- Zhu, L.; Feng, C.; Peng, B.; Hui, X.; Bai, X.; Yu, Z. Performance research of natural mica modified with zirconium-based metal–organic frameworks for an epoxy resin anti-corrosion coating. Molecules 2023, 28, 7106. [Google Scholar] [CrossRef]
- Chen, Y.; Wang, J.; Wen, S.; Zhang, J.; Yu, X.; Mao, Y. Synthesis of rose-like sheet zinc phosphate by the induction-calcination method and its application as a corrosion inhibitor in coatings. Int. J. Electrochem. Sci. 2021, 16, 210456. [Google Scholar] [CrossRef]
- Sørensen, P.A.; Dam-Johansel, K.; Weinell, C.E.; Kiil, S. Cathodic delamination of seawater-immersed anticorrosive coatings: Mapping of parameters affecting the rate. Prog. Org. Coat. 2010, 68, 283–292. [Google Scholar] [CrossRef]
- Deflorian, F.; Rossi, S.; Kamarchik, P.; Fedrizzi, L.; Bonora, P.L. Degradation mechanism of electrodeposited coatings in alkaline solution. Prog. Org. Coat. 2003, 47, 103–111. [Google Scholar] [CrossRef]
- Kasatkin, V.E.; Kasatkina, I.V.; Korosteleva, I.G.; Kornienko, L.P.; Dorofeeva, V.N.; Andreev, N.N. Corrosion behavior and EIS study of steel in an alkaline medium containing chlorides with Catechol inhibitor. Int. J. Corros. Scale Inhib. 2023, 12, 1791–1805. [Google Scholar] [CrossRef]
- Santarelli, M.; Bonse, B.C.; Rocha Poço, J.G. Selection of Crosslinking Agents for Acrylic Resin Used in External Coatings for Aluminum Packaging in the Beverage Industry. Coatings 2024, 14, 585. [Google Scholar] [CrossRef]
- Biryukov, A.I.; Kozaderov, O.A.; Batmanova, T.V. Features of the corrosion of coatings based on zinc alloys: Oxidation products and the selective dissolution of zinc. Review. Condens. Matter Interphases 2024, 26, 25–36. [Google Scholar] [CrossRef] [PubMed]
- Maltseva, A.; Lamaka, S.V.; Yasakau, K.A.; Mei, D.; Kurchavov, D.; Zheludkevich, M.L.; Lefèvre, G.; Volovitch, P. In situ surface film evolution during Mg aqueous corrosion in presence of selected carboxylates. Corros. Sci. 2020, 171, 108484. [Google Scholar] [CrossRef]
- Roscher, J.; Liu, D.; Xie, X.; Holze, R. Aromatic metal corrosion inhibitors. Corros. Mater. Degrad. 2024, 5, 513–560. [Google Scholar] [CrossRef]
- Belkheiri, A.; Dahmani, K.; Mzioud, K.; Khattabi, M.; Kharbouch, O.; Galai, M.; Merroun, Y.; Dkhireche, N.; Benzekri, Z.; Boukhris, S.; et al. Assessment of 14-(4-nitrophenyl)-14H-dibenzo [a, j] xanthene as an effective organic corrosion inhibitor for mild steel in 1 M HCl: Electrochemical, theoretical, and surface analysis. Int. J. Electrochem. Sci. 2025, 20, 100907. [Google Scholar] [CrossRef]
- Naderi, R.; Attar, M.M. The role of zinc aluminum phosphate anticorrosive pigment in protective performance and cathodic disbondment of epoxy coating. Corros. Sci. 2010, 52, 1291–1296. [Google Scholar] [CrossRef]
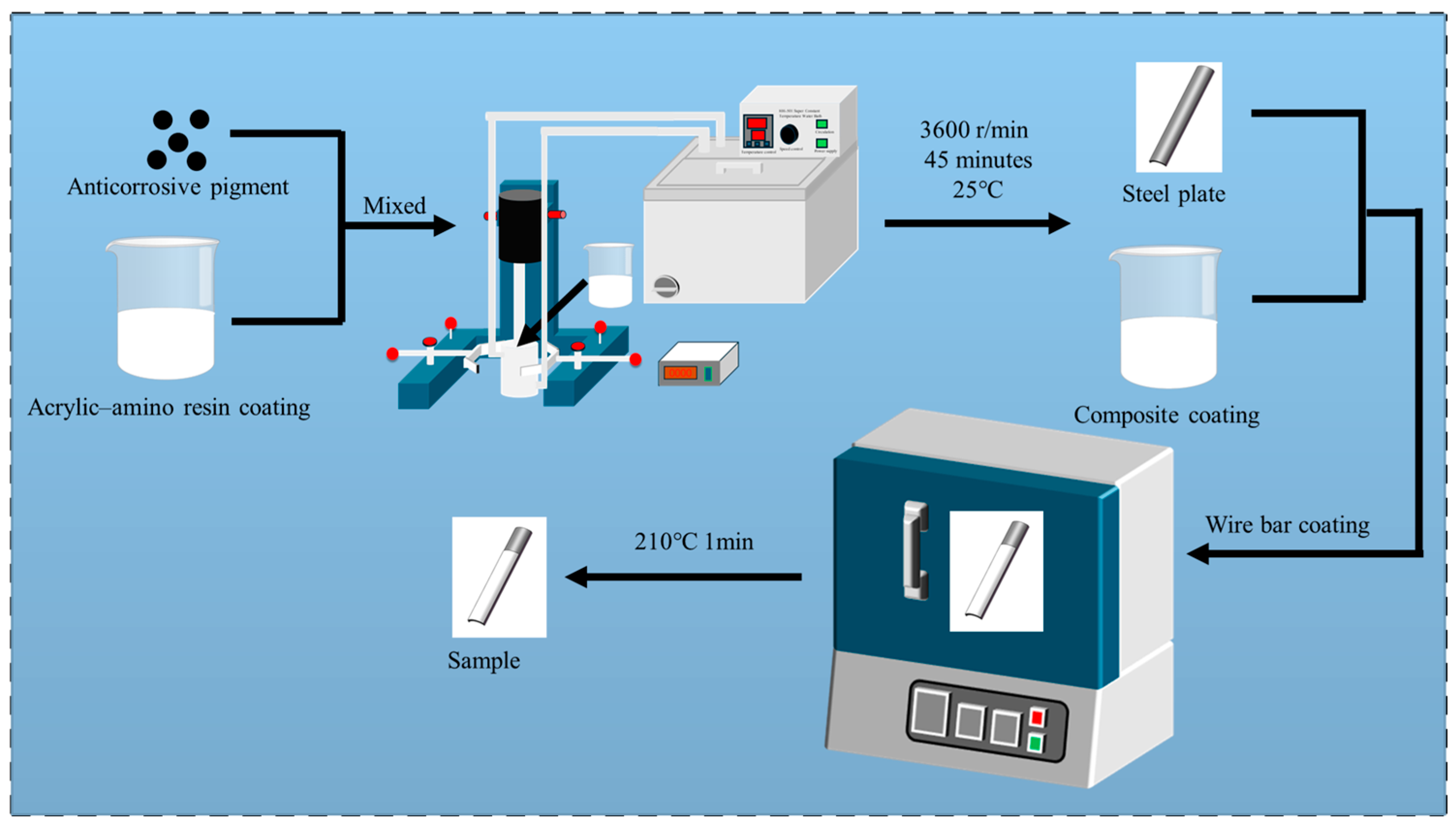






| Number | Coating | Powder Content (wt.%) | Baking Process |
|---|---|---|---|
| #1 | RZ/ZMP/Acrylic–amino resin coating | 0.5 RZ + 2.5 ZMP | 210 °C 1 min |
| #2 | RZ/ZPO/Acrylic–amino resin coating | 0.5 RZ + 2.5 ZPO | 210 °C 1 min |
| #3 | RZ/ZPA/Acrylic–amino resin coating | 3 RZ + 1 ZPA | 210 °C 1 min |
| #4 | Acrylic–amino resin coating | / | 210 °C 1 min |
| #5 | Steel plate | / | / |
| At.% | C | O | P | Zn | Al | Mo |
|---|---|---|---|---|---|---|
| RZ | 25.49 | 15.69 | 0 | 58.82 | / | / |
| ZPA | 11.57 | 57.64 | 10.82 | 15.77 | 4.92 | / |
| ZPO | 5.32 | 55.6 | 11.66 | 27.43 | 0 | / |
| ZMP | 7.92 | 59.76 | 9.35 | 22.38 | 0 | 0.6 |
| Coating | Eocp/V | Ecorr/V | Icorr/A·cm−2 |
|---|---|---|---|
| RZ/ZMP/Acrylic–amino resin coating | −0.1457 | −0.295 | 1.174 × 10−7 |
| RZ/ZPO/Acrylic–amino resin coating | −0.1667 | −0.294 | 1.649 × 10−8 |
| RZ/ZPA/Acrylic–amino resin coating | −0.2424 | −0.305 | 1.477 × 10−8 |
| Acrylic–amino resin coating | −0.1993 | −0.271 | 2.780 × 10−8 |
| Steel plate | −0.2685 | −0.309 | 7.389 × 10−8 |
| Coating | Rs/Ωcm2 | Rct/Ωcm2 | CPEc |
|---|---|---|---|
| RZ/ZMP/Acrylic–amino resin coating | 1.010 × 103 | 4.675 × 106 | 5.982 × 10−6 |
| RZ/ZPO/Acrylic–amino resin coating | 1.276 × 103 | 1.411 × 107 | 8.631 × 10−7 |
| RZ/ZPA/Acrylic–amino resin coating | 2.043 × 104 | 4.309 × 106 | 9.646 × 10−7 |
| Acrylic–amino resin coating | 8.696 × 102 | 3.345 × 106 | 1.012 × 10−6 |
| Steel plate | 2.896 | 7.515 × 104 | 4.434 × 10−5 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2026 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license.
Share and Cite
Hu, X.; Wu, W.; Zhang, A.; An, Y.-A.; Liu, K.; Li, D. Effects of Inhibitors on Corrosion Resistance of Acrylic–Amino Resin Coatings in Alkaline Solution for Industrial Measuring Tapes. Corros. Mater. Degrad. 2026, 7, 12. https://doi.org/10.3390/cmd7010012
Hu X, Wu W, Zhang A, An Y-A, Liu K, Li D. Effects of Inhibitors on Corrosion Resistance of Acrylic–Amino Resin Coatings in Alkaline Solution for Industrial Measuring Tapes. Corrosion and Materials Degradation. 2026; 7(1):12. https://doi.org/10.3390/cmd7010012
Chicago/Turabian StyleHu, Xingwei, Wangping Wu, Ao Zhang, Yu-Ao An, Kunming Liu, and Danfeng Li. 2026. "Effects of Inhibitors on Corrosion Resistance of Acrylic–Amino Resin Coatings in Alkaline Solution for Industrial Measuring Tapes" Corrosion and Materials Degradation 7, no. 1: 12. https://doi.org/10.3390/cmd7010012
APA StyleHu, X., Wu, W., Zhang, A., An, Y.-A., Liu, K., & Li, D. (2026). Effects of Inhibitors on Corrosion Resistance of Acrylic–Amino Resin Coatings in Alkaline Solution for Industrial Measuring Tapes. Corrosion and Materials Degradation, 7(1), 12. https://doi.org/10.3390/cmd7010012






