Recovery Sleep Immediately after Prolonged Sleep Deprivation Stimulates the Transcription of Integrated Stress Response-Related Genes in the Liver of Male Rats
Abstract
1. Introduction
2. Results
3. Discussion
4. Materials and Methods
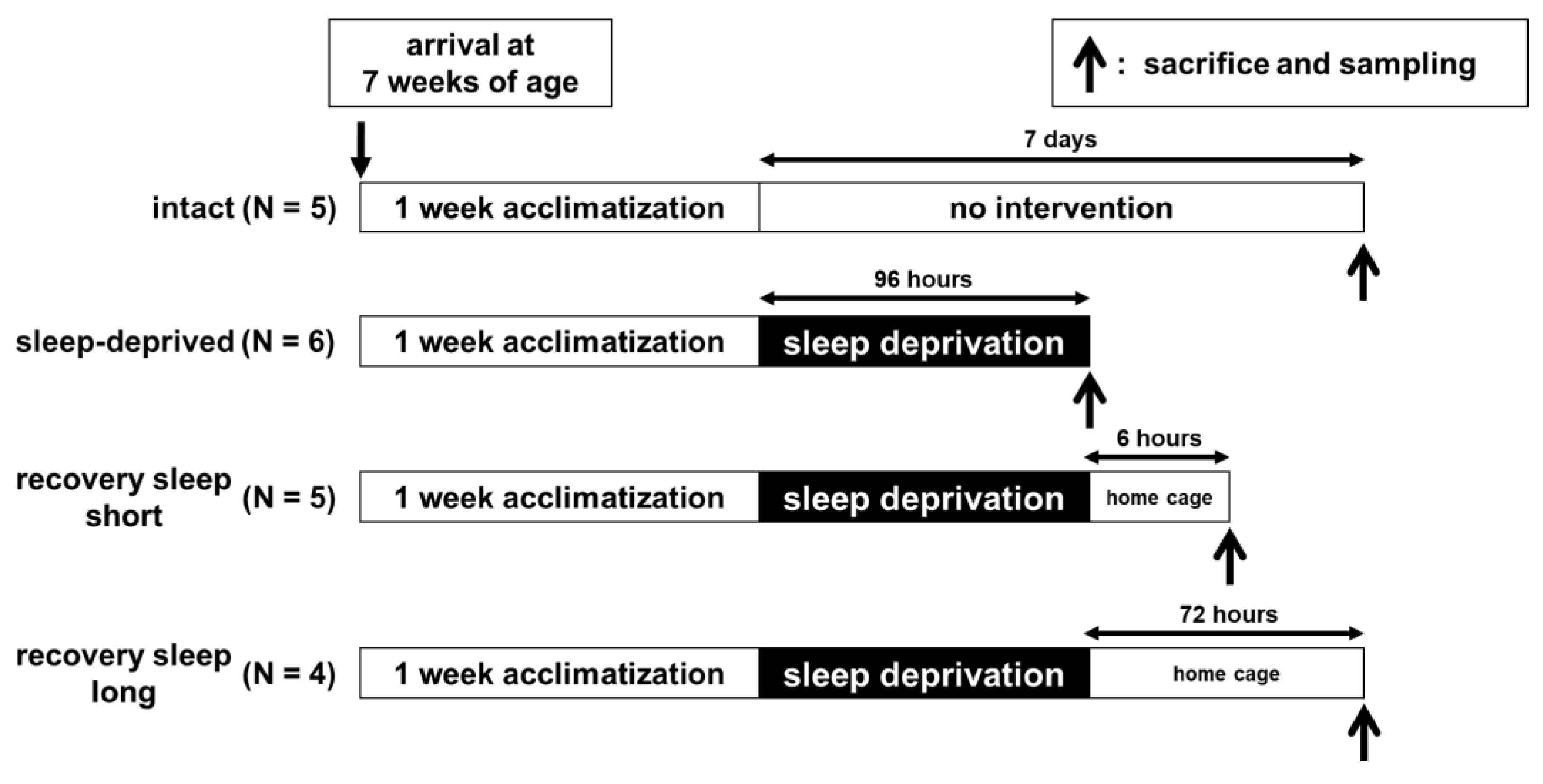
4.1. Animals
4.2. Sleep Deprivation
4.3. Experimental Procedure
4.4. Sample Collection, Total RNA Preparation, cDNA Synthesis and Quantification of mRNA Level by RT-qPCR
4.5. Statistical Analysis
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Hsieh, S.; Cheng, I.C.; Tsai, L.L. Immediate error correction process following sleep deprivation. J. Sleep Res. 2007, 16, 137–147. [Google Scholar] [CrossRef] [PubMed]
- Jackson, M.L.; Raj, S.; Croft, R.J.; Hayley, A.C.; Downey, L.A.; Kennedy, G.A.; Howard, M.E. Slow eyelid closure as a measure of driver drowsiness and its relationship to performance. Traffic Inj. Prev. 2016, 17, 251–257. [Google Scholar] [CrossRef] [PubMed]
- Previc, F.H.; Lopez, N.; Ercoline, W.R.; Daluz, C.M.; Workman, A.J.; Evans, R.H.; Dillon, N.A. The Effects of Sleep Deprivation on Flight Performance, Instrument Scanning, and Physiological Arousal in Pilots. Int. J. Aviat. Psychol. 2009, 19, 326–346. [Google Scholar] [CrossRef]
- Gunzelmann, G.; Gross, J.B.; Gluck, K.A.; Dinges, D.F. Sleep deprivation and sustained attention performance: Integrating mathematical and cognitive modeling. Cogn. Sci. 2009, 33, 880–910. [Google Scholar] [CrossRef] [PubMed]
- Paech, G.M.; Banks, S.; Pajcin, M.; Grant, C.; Johnson, K.; Kamimori, G.H.; Vedova, C.B. Caffeine administration at night during extended wakefulness effectively mitigates performance impairment but not subjective assessments of fatigue and sleepiness. Pharmacol. Biochem. Behav. 2016, 145, 27–32. [Google Scholar] [CrossRef] [PubMed]
- Philip, P.; Sagaspe, P.; Prague, M.; Tassi, P.; Capelli, A.; Bioulac, B.; Commenges, D.; Taillard, J. Acute versus chronic partial sleep deprivation in middle-aged people: Differential effect on performance and sleepiness. Sleep 2012, 35, 997–1002. [Google Scholar] [CrossRef]
- Chuah, Y.M.; Venkatraman, V.; Dinges, D.F.; Chee, M.W. The neural basis of interindividual variability in inhibitory efficiency after sleep deprivation. J. Neurosci. 2006, 26, 7156–7162. [Google Scholar] [CrossRef]
- Franzen, P.L.; Siegle, G.J.; Buysse, D.J. Relationships between affect, vigilance, and sleepiness following sleep deprivation. J. Sleep Res. 2008, 17, 34–41. [Google Scholar] [CrossRef]
- Pigeau, R.; Naitoh, P.; Buguet, A.; McCann, C.; Baranski, J.; Taylor, M.; Thompson, M.; MacK, I. Modafinil, d-amphetamine and placebo during 64 hours of sustained mental work. I. Effects on mood, fatigue, cognitive performance and body temperature. J. Sleep Res. 1995, 4, 212–228. [Google Scholar] [CrossRef]
- Loge, J.H.; Ekeberg, O.; Kaasa, S. Fatigue in the general Norwegian population: Normative data and associations. J. Psychosom. Res. 1998, 45, 53–65. [Google Scholar] [CrossRef]
- Farmer, A.; Fowler, T.; Scourfield, J.; Thapar, A. Prevalence of chronic disabling fatigue in children and adolescents. Br. J. Psychiatry 2004, 184, 477–481. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Ishii, A.; Watanabe, Y. Neural mechanism of facilitation system during physical fatigue. PLoS ONE 2013, 8, e80731. [Google Scholar] [CrossRef]
- Aoki, R.; Kobayashi, N.; Suzuki, G.; Kuratsune, H.; Shimada, K.; Oka, N.; Takahashi, M.; Yamadera, W.; Iwashita, M.; Tokuno, S.; et al. Human herpesvirus 6 and 7 are biomarkers for fatigue, which distinguish between physiological fatigue and pathological fatigue. Biochem. Biophys. Res. Commun. 2016, 478, 424–430. [Google Scholar] [CrossRef] [PubMed]
- Kondo, K.; Sashihara, J.; Shimada, K.; Takemoto, M.; Amo, K.; Miyagawa, H.; Yamanishi, K. Recognition of a novel stage of betaherpesvirus latency in human herpesvirus 6. J. Virol. 2003, 77, 2258–2264. [Google Scholar] [CrossRef] [PubMed]
- Baker, B.M.; Nargund, A.M.; Sun, T.; Haynes, C.M. Protective coupling of mitochondrial function and protein synthesis via the eIF2α kinase GCN-2. PLoS Genet. 2012, 8, e1002760. [Google Scholar] [CrossRef]
- Clementi, E.; Inglin, L.; Beebe, E.; Gsell, C.; Garajova, Z.; Markkanen, E. Persistent DNA damage triggers activation of the integrated stress response to promote cell survival under nutrient restriction. BMC. Biol. 2020, 18, 36. [Google Scholar] [CrossRef]
- Harding, H.P.; Zhang, Y.; Bertolotti, A.; Zeng, H.; Ron, D. Perk is essential for translational regulation and cell survival during the unfolded protein response. Mol. Cell. 2000, 5, 897–904. [Google Scholar] [CrossRef]
- Sun, X.; Liu, J.; Crary, J.F.; Malagelada, C.; Sulzer, D.; Greene, L.A.; Levy, O.A. ATF4 protects against neuronal death in cellular Parkinson's disease models by maintaining levels of parkin. J. Neurosci. 2013, 33, 2398–2407. [Google Scholar] [CrossRef]
- Ly, S.; Lee, D.A.; Strus, E.; Prober, D.A.; Naidoo, N. Evolutionarily Conserved Regulation of Sleep by the Protein Translational Regulator PERK. Curr. Biol. 2020, 30, 1639–1648.e1633. [Google Scholar] [CrossRef]
- Pandey, A.; Oliver, R.; Kar, S.K. Differential Gene Expression in Brain and Liver Tissue of Wistar Rats after Rapid Eye Movement Sleep Deprivation. Clocks Sleep 2020, 2, 442–465. [Google Scholar] [CrossRef]
- Pandey, A.; Kar, S.K. Rapid Eye Movement sleep deprivation of rat generates ROS in the hepatocytes and makes them more susceptible to oxidative stress. Sleep Sci. 2018, 11, 245–253. [Google Scholar] [CrossRef] [PubMed]
- Pandey, A.K.; Kar, S.K. REM sleep deprivation of rats induces acute phase response in liver. Biochem. Biophys. Res. Commun. 2011, 410, 242–246. [Google Scholar] [CrossRef] [PubMed]
- Korneeva, N.L. Integrated Stress Response in Neuronal Pathology and in Health. Biochemistry 2022, 87, S111–S127. [Google Scholar] [CrossRef]
- Baird, T.D.; Wek, R.C. Eukaryotic initiation factor 2 phosphorylation and translational control in metabolism. Adv. Nutr. 2012, 3, 307–321. [Google Scholar] [CrossRef] [PubMed]
- Pakos-Zebrucka, K.; Koryga, I.; Mnich, K.; Ljujic, M.; Samali, A.; Gorman, A.M. The integrated stress response. EMBO Rep. 2016, 17, 1374–1395. [Google Scholar] [CrossRef]
- Wek, R.C.; Cavener, D.R. Translational control and the unfolded protein response. Antioxid. Redox. Signal. 2007, 9, 2357–2371. [Google Scholar] [CrossRef]
- Jiang, H.Y.; Wek, S.A.; McGrath, B.C.; Lu, D.; Hai, T.; Harding, H.P.; Wang, X.; Ron, D.; Cavener, D.R.; Wek, R.C. Activating transcription factor 3 is integral to the eukaryotic initiation factor 2 kinase stress response. Mol. Cell. Biol. 2004, 24, 1365–1377. [Google Scholar] [CrossRef]
- Mungrue, I.N.; Pagnon, J.; Kohannim, O.; Gargalovic, P.S.; Lusis, A.J. CHAC1/MGC4504 is a novel proapoptotic component of the unfolded protein response, downstream of the ATF4-ATF3-CHOP cascade. J. Immunol. 2009, 182, 466–476. [Google Scholar] [CrossRef]
- Szegezdi, E.; Duffy, A.; O'Mahoney, M.E.; Logue, S.E.; Mylotte, L.A.; O'brien, T.; Samali, A. ER stress contributes to ischemia-induced cardiomyocyte apoptosis. Biochem. Biophys. Res. Commun. 2006, 349, 1406–1411. [Google Scholar] [CrossRef]
- Dey, S.; Sayers, C.M.; Verginadis, I.I.; Lehman, S.L.; Cheng, Y.; Cerniglia, G.J.; Tuttle, S.W.; Feldman, M.D.; Zhang, P.J.; Fuchs, S.Y.; et al. ATF4-dependent induction of heme oxygenase 1 prevents anoikis and promotes metastasis. J. Clin. Investig. 2015, 125, 2592–2608. [Google Scholar] [CrossRef]
- He, C.H.; Gong, P.; Hu, B.; Stewart, D.; Choi, M.E.; Choi, A.M.; Alam, J. Identification of activating transcription factor 4 (ATF4) as an Nrf2-interacting protein. Implication for heme oxygenase-1 gene regulation. J. Biol. Chem. 2001, 276, 20858–20865. [Google Scholar] [CrossRef] [PubMed]
- Novoa, I.; Zeng, H.; Harding, H.P.; Ron, D. Feedback inhibition of the unfolded protein response by GADD34-mediated dephosphorylation of eIF2alpha. J. Cell. Biol. 2001, 153, 1011–1022. [Google Scholar] [CrossRef] [PubMed]
- Tabas, I.; Ron, D. Integrating the mechanisms of apoptosis induced by endoplasmic reticulum stress. Nat. Cell. Biol. 2011, 13, 184–190. [Google Scholar] [CrossRef] [PubMed]
- Dey, S.; Baird, T.D.; Zhou, D.; Palam, L.R.; Spandau, D.F.; Wek, R.C. Both transcriptional regulation and translational control of ATF4 are central to the integrated stress response. J. Biol. Chem. 2010, 285, 33165–33174. [Google Scholar] [CrossRef]
- Ingolia, N.T.; Ghaemmaghami, S.; Newman, J.R.; Weissman, J.S. Genome-wide analysis in vivo of translation with nucleotide resolution using ribosome profiling. Science 2009, 324, 218–223. [Google Scholar] [CrossRef]
- Galluzzi, L.; Yamazaki, T.; Kroemer, G. Linking cellular stress responses to systemic homeostasis. Nat. Rev. Mol. Cell. Biol. 2018, 19, 731–745. [Google Scholar] [CrossRef]
- Kültz, D. Molecular and evolutionary basis of the cellular stress response. Annu. Rev. Physiol. 2005, 67, 225–257. [Google Scholar] [CrossRef]
- Andreone, B.J.; Larhammar, M.; Lewcock, J.W. Cell Death and Neurodegeneration. Cold Spring. Harb. Perspect. Biol. 2020, 12, a036434. [Google Scholar] [CrossRef]
- Metcalf, M.G.; Higuchi-Sanabria, R.; Garcia, G.; Tsui, C.K.; Dillin, A. Beyond the cell factory: Homeostatic regulation of and by the UPR. Sci. Adv. 2020, 6, eabb9614. [Google Scholar] [CrossRef]
- Cox, J.S.; Shamu, C.E.; Walter, P. Transcriptional induction of genes encoding endoplasmic reticulum resident proteins requires a transmembrane protein kinase. Cell. 1993, 73, 1197–1206. [Google Scholar] [CrossRef]
- Harding, H.P.; Zhang, Y.; Ron, D. Protein translation and folding are coupled by an endoplasmic-reticulum-resident kinase. Nature 1999, 397, 271–274. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Shen, J.; Arenzana, N.; Tirasophon, W.; Kaufman, R.J.; Prywes, R. Activation of ATF6 and an ATF6 DNA binding site by the endoplasmic reticulum stress response. J. Biol. Chem. 2000, 275, 27013–27020. [Google Scholar] [CrossRef]
- Mackiewicz, M.; Naidoo, N.; Zimmerman, J.E.; Pack, A.I. Molecular mechanisms of sleep and wakefulness. Ann. N. Y. Acad. Sci. 2008, 1129, 335–349. [Google Scholar] [CrossRef] [PubMed]
- Naidoo, N. Roles of endoplasmic reticulum and energetic stress in disturbed sleep. Neuromolecular Med. 2012, 14, 213–219. [Google Scholar] [CrossRef] [PubMed]
- Naidoo, N. Cellular stress/the unfolded protein response: Relevance to sleep and sleep disorders. Sleep. Med. Rev. 2009, 13, 195–204. [Google Scholar] [CrossRef] [PubMed]
- Anafi, R.C.; Pellegrino, R.; Shockley, K.R.; Romer, M.; Tufik, S.; Pack, A.I. Sleep is not just for the brain: Transcriptional responses to sleep in peripheral tissues. BMC. Genomics 2013, 14, 362. [Google Scholar] [CrossRef]
- Koban, M.; Swinson, K.L. Chronic REM-sleep deprivation of rats elevates metabolic rate and increases UCP1 gene expression in brown adipose tissue. Am. J. Physiol. Endocrinol. Metab. 2005, 289, E68–E74. [Google Scholar] [CrossRef]
- da Costa Souza, A.; Ribeiro, S. Sleep deprivation and gene expression. Curr. Top. Behav. Neurosci. 2015, 25, 65–90. [Google Scholar] [CrossRef]
- Sei, H.; Saitoh, D.; Yamamoto, K.; Morita, K.; Morita, Y. Differential effect of short-term REM sleep deprivation on NGF and BDNF protein levels in the rat brain. Brain. Res. 2000, 877, 387–390. [Google Scholar] [CrossRef]
- Taishi, P.; Sanchez, C.; Wang, Y.; Fang, J.; Harding, J.W.; Krueger, J.M. Conditions that affect sleep alter the expression of molecules associated with synaptic plasticity. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2001, 281, R839–R845. [Google Scholar] [CrossRef]
- Cirelli, C.; Tononi, G. Gene expression in the brain across the sleep-waking cycle. Brain. Res. 2000, 885, 303–321. [Google Scholar] [CrossRef]
- Guzman-Marin, R.; Ying, Z.; Suntsova, N.; Methippara, M.; Bashir, T.; Szymusiak, R.; Gomez-Pinilla, F.; McGinty, D. Suppression of hippocampal plasticity-related gene expression by sleep deprivation in rats. J. Physiol. 2006, 575, 807–819. [Google Scholar] [CrossRef] [PubMed]
- Guindalini, C.; Andersen, M.L.; Alvarenga, T.; Lee, K.; Tufik, S. To what extent is sleep rebound effective in reversing the effects of paradoxical sleep deprivation on gene expression in the brain? Behav. Brain. Res. 2009, 201, 53–58. [Google Scholar] [CrossRef] [PubMed]
- Tanaka, M.; Nakamura, F.; Mizokawa, S.; Matsumura, A.; Nozaki, S.; Watanabe, Y. Establishment and assessment of a rat model of fatigue. Neurosci. Lett. 2003, 352, 159–162. [Google Scholar] [CrossRef] [PubMed]
- Khalyfa, A.; Qiao, Z.; Gileles-Hillel, A.; Khalyfa, A.A.; Akbarpour, M.; Popko, B.; Gozal, D. Activation of the Integrated Stress Response and Metabolic Dysfunction in a Murine Model of Sleep Apnea. Am. J. Respir. Cell. Mol. Biol. 2017, 57, 477–486. [Google Scholar] [CrossRef]
- Léger, D.; Stepnowsky, C. The economic and societal burden of excessive daytime sleepiness in patients with obstructive sleep apnea. Sleep. Med. Rev. 2020, 51, 101275. [Google Scholar] [CrossRef]
- Murata, Y.; Oka, A.; Iseki, A.; Mori, M.; Ohe, K.; Mine, K.; Enjoji, M. Prolonged sleep deprivation decreases cell proliferation and immature newborn neurons in both dorsal and ventral hippocampus of male rats. Neurosci. Res. 2018, 131, 45–51. [Google Scholar] [CrossRef]
- Kim, K.; Kim, Y.H.; Lee, S.H.; Jeon, M.J.; Park, S.Y.; Doh, K.O. Effect of exercise intensity on unfolded protein response in skeletal muscle of rat. Korean. J. Physiol. Pharmacol. 2014, 18, 211–216. [Google Scholar] [CrossRef]
- Li, J.; Xu, J.; Xu, W.; Qi, Y.; Lu, Y.; Qiu, L.; Hu, Z.; Chu, Z.; Chai, Y.; Zhang, J. Protective Effects of Hong Shan Capsule against Lethal Total-Body Irradiation-Induced Damage in Wistar Rats. Int. J. Mol. Sci. 2015, 16, 18938–18955. [Google Scholar] [CrossRef]
- Pinto, B.A.; Melo, T.M.; Flister, K.F.; França, L.M.; Kajihara, D.; Tanaka, L.Y.; Laurindo, F.R.; Paes, A.M. Early and sustained exposure to high-sucrose diet triggers hippocampal ER stress in young rats. Metab. Brain. Dis. 2016, 31, 917–927. [Google Scholar] [CrossRef]
- Souza, R.B.; Gomes, F.I.F.; Pereira, K.M.A.; Dutra, P.G.P.; da Cunha, R.M.S.; Chaves, H.V.; Bezerra, M.M. Strontium Ranelate Elevates Expression of Heme Oxygenase-1 and Decreases Alveolar Bone Loss in Rats. J. Oral. Maxillofac. Res. 2018, 9, e4. [Google Scholar] [CrossRef] [PubMed]
- Audet, M.C.; Jacobson-Pick, S.; Wann, B.P.; Anisman, H. Social defeat promotes specific cytokine variations within the prefrontal cortex upon subsequent aggressive or endotoxin challenges. Brain. Behav. Immun. 2011, 25, 1197–1205. [Google Scholar] [CrossRef] [PubMed]
- Bustin, S.A.; Benes, V.; Garson, J.A.; Hellemans, J.; Huggett, J.; Kubista, M.; Mueller, R.; Nolan, T.; Pfaffl, M.W.; Shipley, G.L.; et al. The MIQE guidelines: Minimum information for publication of quantitative real-time PCR experiments. Clin. Chem. 2009, 55, 611–622. [Google Scholar] [CrossRef]
- Pfaffl, M.W. A new mathematical model for relative quantification in real-time RT-PCR. Nucleic. Acids. Res. 2001, 29, e45. [Google Scholar] [CrossRef] [PubMed]
- Team, R.D.C. A Language and Environment for Statistical Computing, version 4.2.1; R Foundation for Statistical Computing: Vienna, Austria, 2022. [Google Scholar]

| Gene Name | GenBank Accession No. | Primer Sequence (5'->3') | Amplicon Size (bp) | Efficiency | R2 Value | References | |
|---|---|---|---|---|---|---|---|
| Actb | NM_031144 | Forward | AAGACAGCACGCTAATAATGC | 115 | 97.84 | 0.999 | [62] |
| Reverse | TTGGAAGGCCGGTTAATTTTC | ||||||
| Atf3 | NM_012912 | Forward | GCACAACATTGGCGTGATTTT | 71 | 97.98 | 0.999 | [58] |
| Reverse | TGGCAGACCCCCAAACTCT | ||||||
| Atf4 | NM_024403 | Forward | TCAGACACCGGCAAGGAG | 134 | 99.75 | 0.999 | [59] |
| Reverse | GTGGCCAAAAGCTCATCTG | ||||||
| Atf6 | NM_001107196 | Forward | TTCTCTGATGGCCGTGCAT | 65 | 93.69 | 0.999 | [60] |
| Reverse | TGAAGATGACCCACAGAACCAA | ||||||
| Ddit3 | NM_001109986 | Forward | TGGCACAGCTTGCTGAAGAG | 54 | 91.09 | 1 | [60] |
| Reverse | TCAGGCGCTCGATTTCCT | ||||||
| Eif2ak3 | NM_031599 | Forward | GGCTGGTGAGGGATGGTAAA | 64 | 86.68 | 0.998 | [60] |
| Reverse | TTGGCTGTGTAACTTGTGTCATCA | ||||||
| Ern1 | NM_001191926 | Forward | GGATGTGAGTGACCGAATAGAAAA | 60 | 89.78 | 0.997 | [60] |
| Reverse | TCCAACTGCCGCACGAT | ||||||
| Hmox1 | NM_012580 | Forward | ACAGCACTACGTAAAGCGTCTCCA | 136 | 89.52 | 0.998 | [61] |
| Reverse | CATGGCCTTCTGCGCAATCTTCTT | ||||||
| Ppp1r15a | NM_133546 | Forward | CCCAGCATTGTCTACCAGT | 84 | 96.47 | 0.999 | originally designed using Primer3 |
| Reverse | CAGGTAAATAGAAGGCCACCT | ||||||
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fukuoka, K.; Murata, Y.; Otomaru, T.; Mori, M.; Ohe, K.; Mine, K.; Enjoji, M. Recovery Sleep Immediately after Prolonged Sleep Deprivation Stimulates the Transcription of Integrated Stress Response-Related Genes in the Liver of Male Rats. Clocks & Sleep 2022, 4, 623-632. https://doi.org/10.3390/clockssleep4040048
Fukuoka K, Murata Y, Otomaru T, Mori M, Ohe K, Mine K, Enjoji M. Recovery Sleep Immediately after Prolonged Sleep Deprivation Stimulates the Transcription of Integrated Stress Response-Related Genes in the Liver of Male Rats. Clocks & Sleep. 2022; 4(4):623-632. https://doi.org/10.3390/clockssleep4040048
Chicago/Turabian StyleFukuoka, Keisuke, Yusuke Murata, Tomomi Otomaru, Masayoshi Mori, Kenji Ohe, Kazunori Mine, and Munechika Enjoji. 2022. "Recovery Sleep Immediately after Prolonged Sleep Deprivation Stimulates the Transcription of Integrated Stress Response-Related Genes in the Liver of Male Rats" Clocks & Sleep 4, no. 4: 623-632. https://doi.org/10.3390/clockssleep4040048
APA StyleFukuoka, K., Murata, Y., Otomaru, T., Mori, M., Ohe, K., Mine, K., & Enjoji, M. (2022). Recovery Sleep Immediately after Prolonged Sleep Deprivation Stimulates the Transcription of Integrated Stress Response-Related Genes in the Liver of Male Rats. Clocks & Sleep, 4(4), 623-632. https://doi.org/10.3390/clockssleep4040048





