Fluorescent Paints in Contemporary Murals: A Case Study
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
2.2. µ-Raman Spectroscopy and SERS Analysis
2.3. Liquid Chromatography with Spectrophotometric (HPLC-DAD) and Mass Spectrometric Detection (HPLC-MS) Analyses
2.4. Pyrolysis-Gas Chromatography-Mass Spectrometry Analyses
3. Results and Discussion
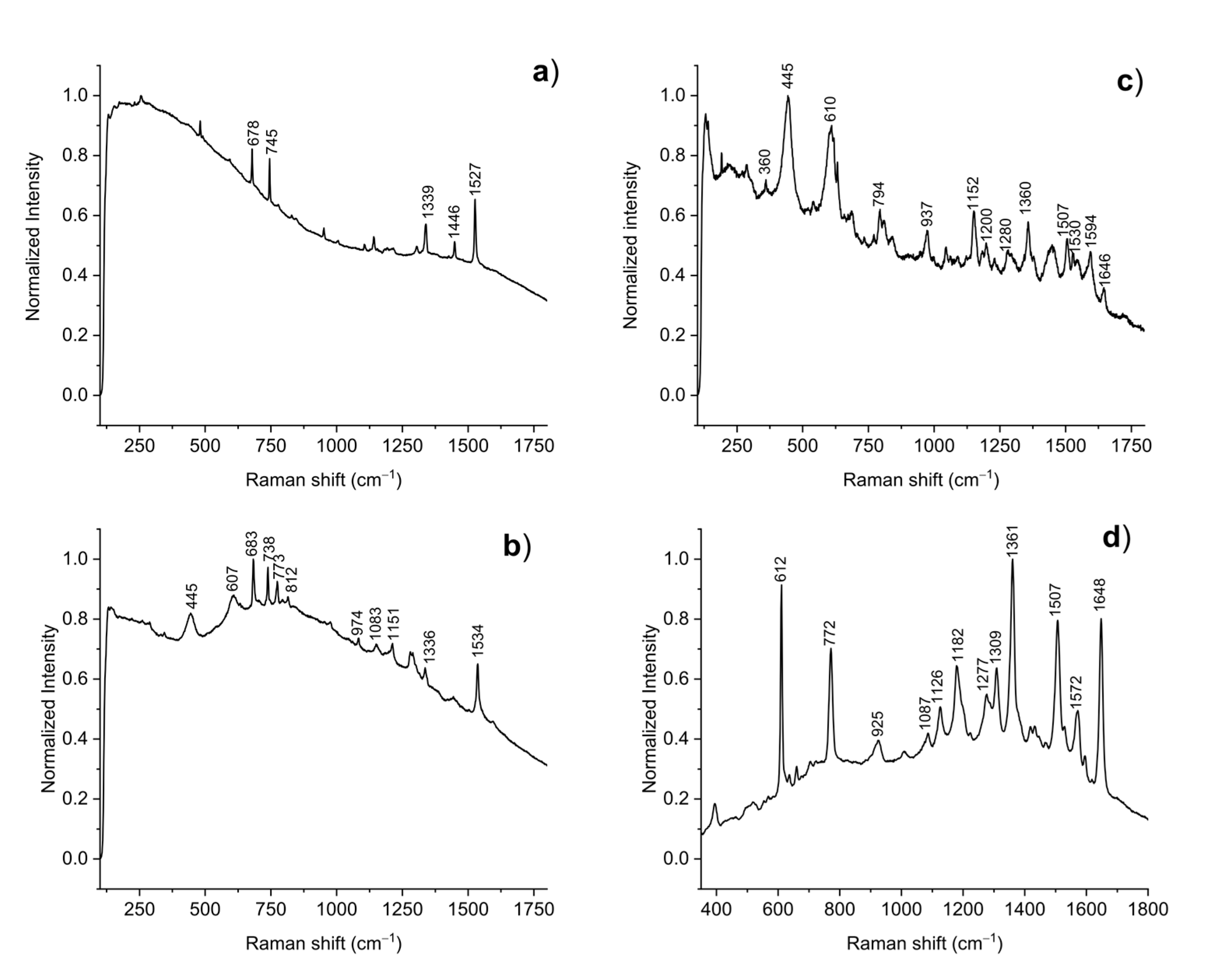
3.1. Raman Spectroscopy and SERS Analysis
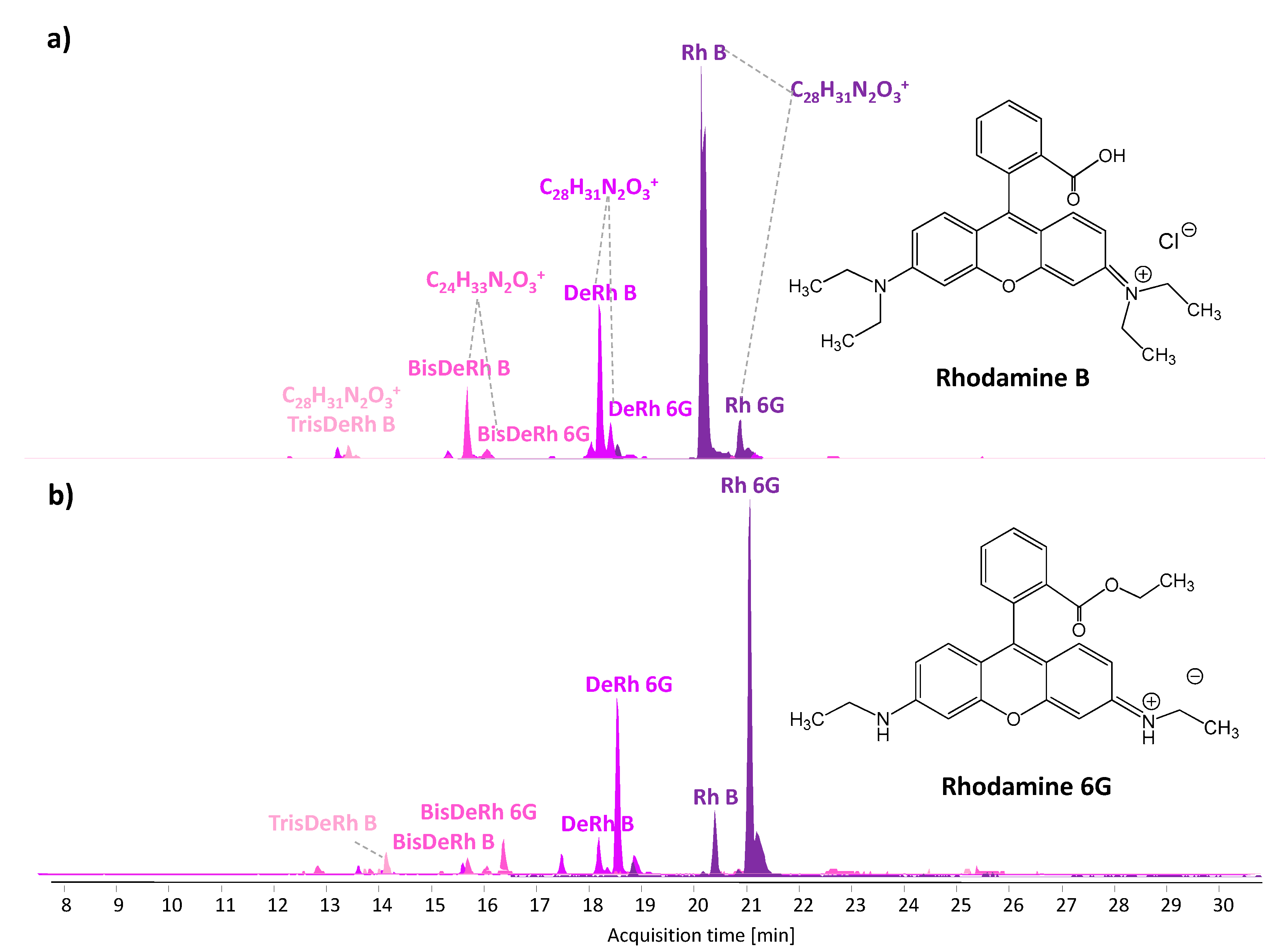
3.2. HPLC-DAD-MS
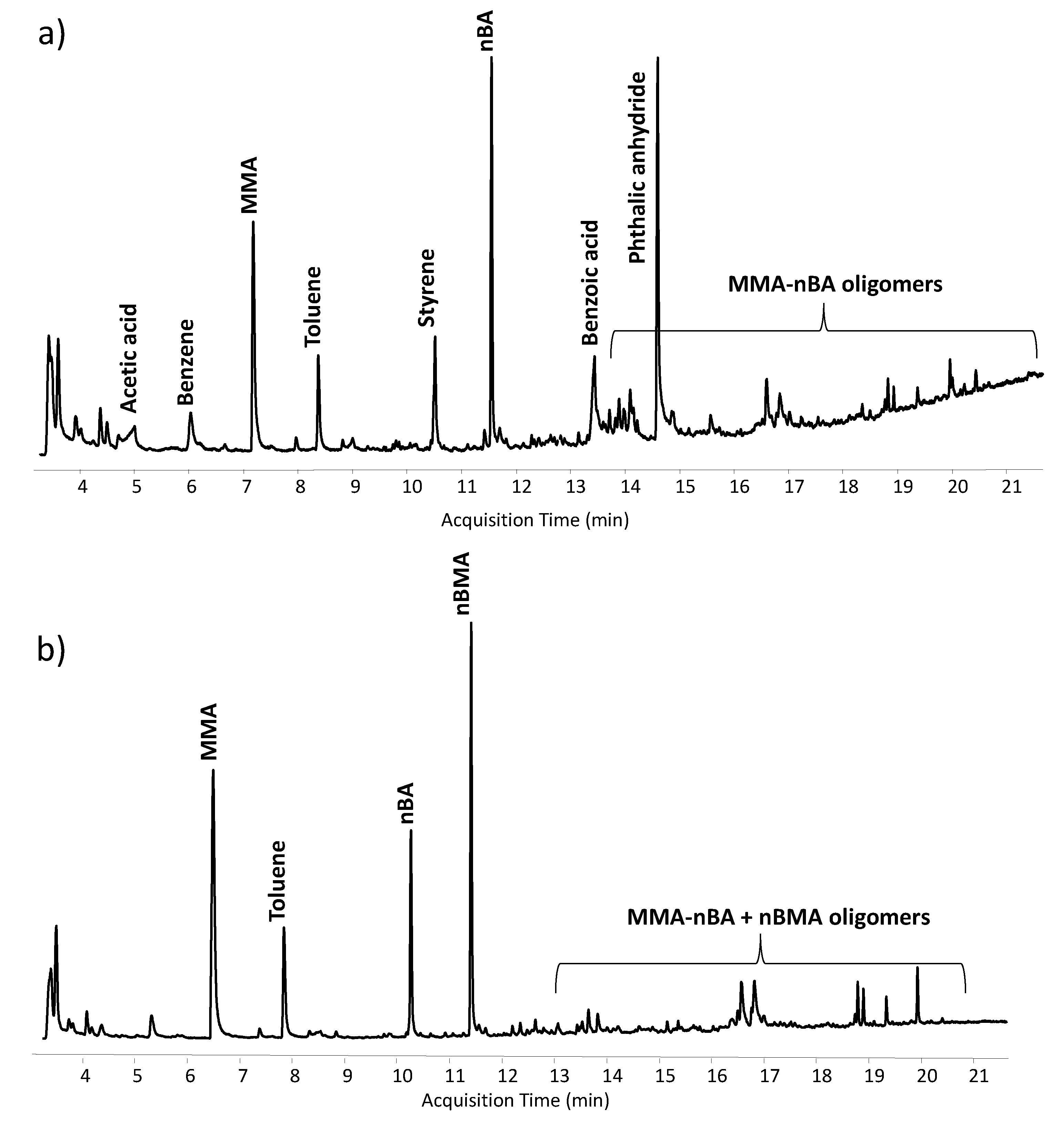
3.3. Py-GC-MS
4. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Learner, T.; Sanchez-Pons, M.; Shank, W. (Eds.) Conservation Issues in Modern and Contemporary Murals; Cambridge Scholars Publishing: Newcastle upon Tyne, UK, 2015. [Google Scholar]
- Cortea, I.M.; Ratoiu, L.; Rădvan, R. Characterization of spray paints used in street art graffiti by a non-destructive multi-analytical approach. Color Res. Appl. 2021, 46, 183–194. [Google Scholar] [CrossRef]
- Germinario, G.; van der Werf, I.D.; Sabbatini, L. Chemical characterisation of spray paints by a multi-analytical (Py/GC–MS, FTIR, μ-Raman) approach. Microchem. J. 2016, 124, 929–939. [Google Scholar] [CrossRef]
- Cimino, D.; Lamuraglia, R.; Saccani, I.; Berzioli, M.; Izzo, F.C. Assessing the (In)Stability of Urban Art Paints: From Real Case Studies to Laboratory Investigations of Degradation Processes and Preservation Possibilities. Heritage 2022, 5, 581–609. [Google Scholar] [CrossRef]
- Pellis, G.; Bertasa, M.; Ricci, C.; Scarcella, A.; Croveri, P.; Poli, T.; Scalarone, D. A multi-analytical approach for precise identification of alkyd spray paints and for a better understanding of their ageing behaviour in graffiti and urban artworks. J. Anal. Appl. Pyrolysis 2022, 165, 105576. [Google Scholar] [CrossRef]
- Connors-Rowe, S.A.; Morris, H.R.; Whitmore, P.M. Evaluation of Appearance and Fading of Daylight Fluorescent Water Colors. J. Am. Inst. Conserv. 2005, 44, 75–94. [Google Scholar] [CrossRef]
- Pintus, V.; Wei, S.; Schreiner, M. UV ageing studies: Evaluation of lightfastness declarations of commercial acrylic paints. Anal. Bioanal. Chem. 2012, 402, 1567–1584. [Google Scholar] [CrossRef]
- Doménech-Carbó, M.T.; Silva, M.F.; Aura-Castro, E.; Fuster-López, L.; Kröner, S.; Martínez-Bazán, M.L.; Más-Barberá, X.; Mecklenburg, M.F.; Osete-Cortina, L.; Doménech, A.; et al. Study of behaviour on simulated daylight ageing of artists’ acrylic and poly(vinyl acetate) paint films. Anal. Bioanal. Chem. 2011, 399, 2921–2937. [Google Scholar] [CrossRef]
- Ciccola, A.; Serafini, I.; Guiso, M.; Ripanti, F.; Domenici, F.; Sciubba, F.; Postorino, P.; Bianco, A. Spectroscopy for contemporary art: Discovering the effect of synthetic organic pigments on UVB degradation of acrylic binder. Polym. Degrad. Stab. 2019, 159, 224–228. [Google Scholar] [CrossRef]
- Rivas, T.; Alonso-Villar, E.M.; Pozo-Antonio, J.S. Forms and factors of deterioration of urban art murals under humid temperate climate; influence of environment and material properties. Eur. Phys. J. Plus 2022, 137, 1257. [Google Scholar] [CrossRef]
- Pozo-Antonio, J.S.; Alonso-Villar, E.M.; Rivas, T.; Márquez, I. Evaluation of a protective acrylic finish applied to surfaces painted with acrylic paints for outdoor or indoor uses. Dye. Pigment. 2023, 212, 111141. [Google Scholar] [CrossRef]
- Alonso-Villar, E.M.; Rivas, T.; Pozo-Antonio, J.S.; Pellis, G.; Scalarone, D. Efficacy of Colour Protectors in Urban Art Paintings under Different Conditions: From a Real Mural to the Laboratory. Heritage 2023, 6, 3475–3498. [Google Scholar] [CrossRef]
- Rousaki, A.; Vandenabeele, P.; Berzioli, M.; Saccani, I.; Fornasini, L.; Bersani, D. An in-and-out-the-lab Raman spectroscopy study on street art murals from Reggio Emilia in Italy. Eur. Phys. J. Plus 2022, 137, 252. [Google Scholar] [CrossRef]
- Bosi, A.; Ciccola, A.; Serafini, I.; Guiso, M.; Ripanti, F.; Postorino, P.; Curini, R.; Bianco, A. Street art graffiti: Discovering their composition and alteration by FTIR and micro-Raman spectroscopy. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2020, 225, 117474. [Google Scholar] [CrossRef] [PubMed]
- La Nasa, J.; Campanella, B.; Sabatini, F.; Rava, A.; Shank, W.; Lucero-Gomez, P.; De Luca, D.; Legnaioli, S.; Palleschi, V.; Colombini, M.P.; et al. 60 years of street art: A comparative study of the artists’ materials through spectroscopic and mass spectrometric approaches. J. Cult. Herit. 2021, 48, 129–140. [Google Scholar] [CrossRef]
- Cucci, C.; Bartolozzi, G.; Vita, M.D.; Marchiafava, V.; Picollo, M.; Casadio, F. The Colors of Keith Haring: A Spectroscopic Study on the Materials of the Mural Painting Tuttomondo and on Reference Contemporary Outdoor Paints. Appl. Spectrosc. 2016, 70, 186–196. [Google Scholar] [CrossRef]
- Magrini, D.; Bracci, S.; Cantisani, E.; Conti, C.; Rava, A.; Sansonetti, A.; Shank, W.; Colombini, M. A multi-analytical approach for the characterization of wall painting materials on contemporary buildings. Spectrochim. Acta Part A Mol. Biomol. Spectrosc. 2017, 173, 39–45. [Google Scholar] [CrossRef]
- Nasa, J.L.; Orsini, S.; Degano, I.; Rava, A.; Modugno, F.; Colombini, M.P. A chemical study of organic materials in three murals by Keith Haring: A comparison of painting techniques. Microchem. J. 2016, 124, 940–948. [Google Scholar] [CrossRef]
- PRIN2020 SUPERSTAR, (n.d.). Available online: https://prin2020superstar.dcci.unipi.it/ (accessed on 19 May 2023).
- CAPuS Project, (n.d.). Available online: http://www.capusproject.eu/ (accessed on 19 May 2023).
- Ricci, C.; Croveri, P.; Scarcella, A.; Sunara, S.M.; Tabak, T.; Bertasa, M.; Scalarone, D. Tools to Document and Disseminate the Conservation of Urban Art. In Document|Archive|Disseminate Graffiti-Scapes, Proceedings of the goINDIGO 2022 International Graffiti Symposium, Vienna, Austria, 11–13 May 2022; Project INDIGO: Vienna, Austria, 2022; pp. 188–202. [Google Scholar]
- Pelaez, M.; Nolan, N.T.; Pillai, S.C.; Seery, M.K.; Falaras, P.; Kontos, A.G.; Dunlop, P.S.M.; Hamilton, J.W.J.; Byrne, J.A.; O’Shea, K.; et al. A review on the visible light active titanium dioxide photocatalysts for environmental applications. Appl. Catal. B Environ. 2012, 125, 331–349. [Google Scholar] [CrossRef]
- Fremout, W.; Saverwyns, S. Identification of synthetic organic pigments: The role of a comprehensive digital Raman spectral library. J. Raman Spectrosc. 2012, 43, 1536–1544. [Google Scholar] [CrossRef]
- Campanella, B.; Botti, J.; Cavaleri, T.; Cicogna, F.; Legnaioli, S.; Pagnotta, S.; Poggialini, F.; Poli, T.; Scalarone, D.; Palleschi, V. The shining brightness of daylight fluorescent pigments: Raman and SERS study of a modern class of painting materials. Microchem. J. 2020, 152, 104292. [Google Scholar] [CrossRef]
- Cañamares, M.V.; Chenal, C.; Birke, R.L.; Lombardi, J.R. DFT, SERS, and Single-Molecule SERS of Crystal Violet. J. Phys. Chem. C 2008, 112, 20295–20300. [Google Scholar] [CrossRef]
- Petroviciu, I.; Teodorescu, I.C.; Vasilca, S.; Albu, F. Transition from Natural to Early Synthetic Dyes in the Romanian Traditional Shirts Decoration. Heritage 2023, 6, 505–523. [Google Scholar] [CrossRef]
- Ferreira, B.R.V.; Correa, D.N.; Eberlin, M.N.; Vendramini, P.H. Fragmentation Reactions of Rhodamine B and 6G as Revealed by High Accuracy Orbitrap Tandem Mass Spectrometry. J. Braz. Chem. Soc. 2017, 28, 136–142. [Google Scholar] [CrossRef]
- Sabatini, F.; Giugliano, R.; Degano, I. Photo-oxidation processes of Rhodamine B: A chromatographic and mass spectrometric approach. Microchem. J. 2018, 140, 114–122. [Google Scholar] [CrossRef]
- Sabatini, F.; Degano, I.; van Bommel, M. Investigating the in-solution photodegradation pathway of Diamond Green G by chromatography and mass spectrometry. Color. Technol. 2021, 137, 456–467. [Google Scholar] [CrossRef]
- Confortin, D.; Neevel, H.; Brustolon, M.; Franco, L.; Kettelarij, A.J.A.J.; Williams, R.M.R.M.; van Bommel, M.R. Crystal violet: Study of the photo-fading of an early synthetic dye in aqueous solution and on paper with HPLC-PDA, LC-MS and FORS. J. Phys. Conf. Ser. 2010, 231, 012011. [Google Scholar] [CrossRef]
- Groeneveld, I.; Bagdonaite, I.; Beekwilder, E.; Ariese, F.; Somsen, G.W.; Van Bommel, M.R. Liquid core waveguide cell with in situ absorbance spectroscopy and coupled to liquid chromatography for studying light-induced degradation. Anal. Chem. 2022, 94, 7647–7654. [Google Scholar] [CrossRef]
- Dunn, J.; Siegel, J.; Allison, J. Photodegradation and Laser Desorption Mass Spectrometry for the Characterization of Dyes Used in Red Pen Inks. Sci. J. Forensic 2003, 48, JFS2002359. [Google Scholar] [CrossRef]
- Lomax, S.Q.; Lomax, J.F.; Graham, T.K.; Moore, T.J.T.; Knapp, C.G. Historical azo pigments: Synthesis and characterization. J. Cult. Herit. 2018, 35, 218–224. [Google Scholar] [CrossRef]
- Stenger, J.; Kwan, E.E.; Eremin, K.; Speakman, S.; Kirby, D.; Stewart, H.; Huang, S.G.; Kennedy, A.R.; Newman, R.; Khandekar, N. Lithol red salts: Characterization and deterioration. e-Preserv. Sci. 2010, 7, 147–157. [Google Scholar]
- Kirby, D.P.; Khandekar, N.; Sutherland, K.; Price, B.A. Applications of laser desorption mass spectrometry for the study of synthetic organic pigments in works of art. Int. J. Mass Spectrom. 2009, 284, 115–122. [Google Scholar] [CrossRef]
- Lomax, S.Q.; Lomax, J.F.; Luca-Westrate, A.D. The use of Raman microscopy and laser desorption ionization mass spectrometry in the examination of synthetic organic pigments in modern works of art. J. Raman Spectrosc. 2014, 45, 448–455. [Google Scholar] [CrossRef]
- Watanabe, T.; Takizawa, T.; Honda, K. Photocatalysis through excitation of adsorbates. 1. Highly efficient N-deethylation of rhodamine B adsorbed to cadmium sulfide. J. Phys. Chem. 1977, 81, 1845–1851. [Google Scholar] [CrossRef]
- Souto, C.S.C.N. Analysis of Early Synthetic Dyes with HPLC-DAD-MS; Universidade de Lisboa: Lisbon, Portugal, 2010. [Google Scholar]
- Weiss, K.D. Paint and coatings: A mature industry in transition. Prog. Polym. Sci. 1997, 22, 203–245. [Google Scholar] [CrossRef]
- Borgioli, L. I Leganti Nell’arte Contemporanea; Nardini Editore: Florence, Italy, 2020. [Google Scholar]
- Jablonski, E.; Learner, T.; Hayes, J.; Golden, M. Conservation concerns for acrylic emulsion paints. Stud. Conserv. 2003, 48, 3–12. [Google Scholar] [CrossRef]
- Ploeger, R.; Scalarone, D.; Chiantore, O. The characterization of commercial artists’ alkyd paints. J. Cult. Herit. 2008, 9, 412–419. [Google Scholar] [CrossRef]
- Colombini, A.; Kaifas, D. Characterization of some orange and yellow organic and fluorescent pigments by Raman spectroscopy. e-Preserv. Sci. 2010, 7, 14–21. [Google Scholar]

| Sample | Coloration | µ-Raman | SERS | HPLC-MS | Py-GC-MS |
|---|---|---|---|---|---|
| S1B | Blue | PB15 | - | PY74 (tr) | Acrylic resin (MMA-nBA) + styrene modified alkyd + PVAc |
| S2G | Green | PB15, PG7 | CV (tr) | - | Acrylic resin (MMA-nBA nBMA) |
| S3P | Pink | Rutile, BR1 (tr) | BR1 | BV10, BR1, PY74 (tr) | Acrylic resin (MMA-nBA nBMA) |
| S4Y | Yellow | Rutile, PG7, BV10 | BR1, CV (tr) | - | Acrylic resin (MMA-nBA) |
| S5O | Orange | Rutile, BV10 | BR1 | BR1, BV10, PY74 (tr) | Acrylic resin (MMA-nBA nBMA) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sabatini, F.; La Nasa, J.; Degano, I.; Campanella, B.; Legnaioli, S.; Saccani, I.; Modugno, F. Fluorescent Paints in Contemporary Murals: A Case Study. Heritage 2023, 6, 5689-5699. https://doi.org/10.3390/heritage6080299
Sabatini F, La Nasa J, Degano I, Campanella B, Legnaioli S, Saccani I, Modugno F. Fluorescent Paints in Contemporary Murals: A Case Study. Heritage. 2023; 6(8):5689-5699. https://doi.org/10.3390/heritage6080299
Chicago/Turabian StyleSabatini, Francesca, Jacopo La Nasa, Ilaria Degano, Beatrice Campanella, Stefano Legnaioli, Ilaria Saccani, and Francesca Modugno. 2023. "Fluorescent Paints in Contemporary Murals: A Case Study" Heritage 6, no. 8: 5689-5699. https://doi.org/10.3390/heritage6080299
APA StyleSabatini, F., La Nasa, J., Degano, I., Campanella, B., Legnaioli, S., Saccani, I., & Modugno, F. (2023). Fluorescent Paints in Contemporary Murals: A Case Study. Heritage, 6(8), 5689-5699. https://doi.org/10.3390/heritage6080299










