Discovering Giuseppe Capogrossi: Study of the Painting Materials in Three Works of Art Stored at Galleria Nazionale (Rome)
Abstract
1. Introduction
2. Materials and Methods
2.1. The Paintings
2.2. Diagnostic Approach
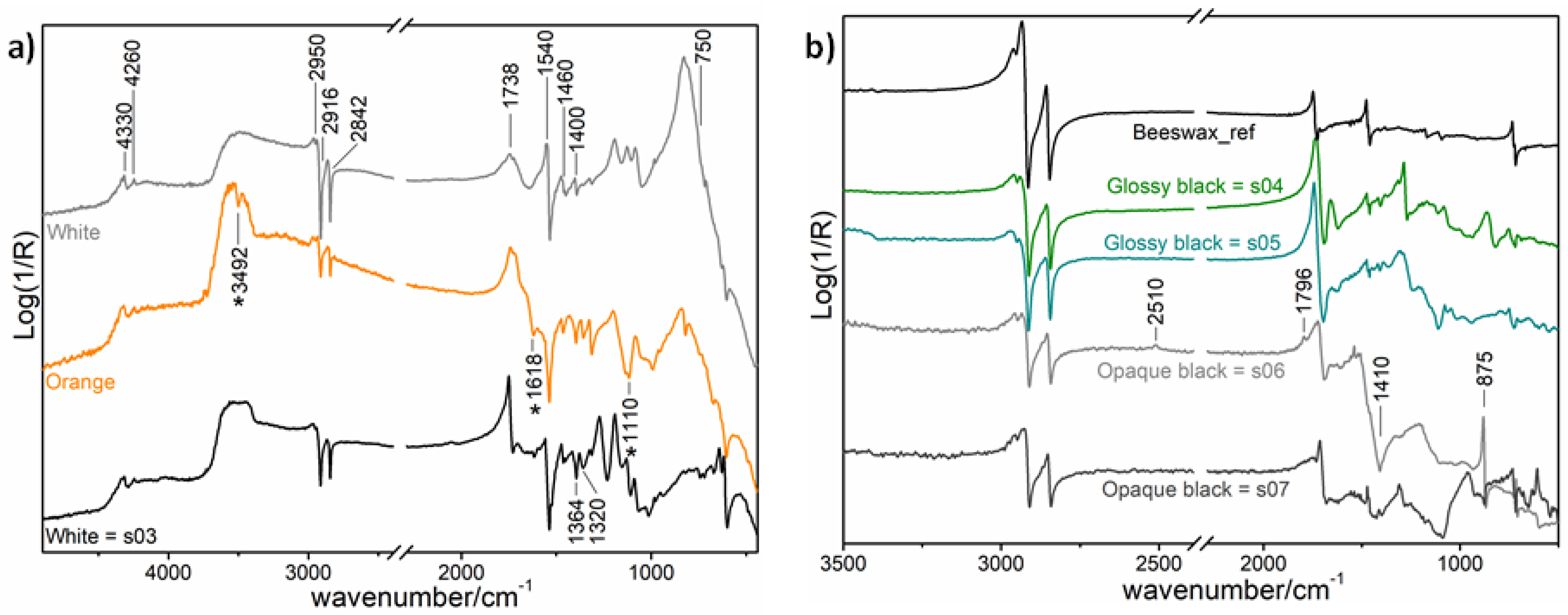
2.2.1. Non-Invasive In Situ Reflection FT-IR Spectroscopy
2.2.2. Sampling for Mass Spectrometric Analysis
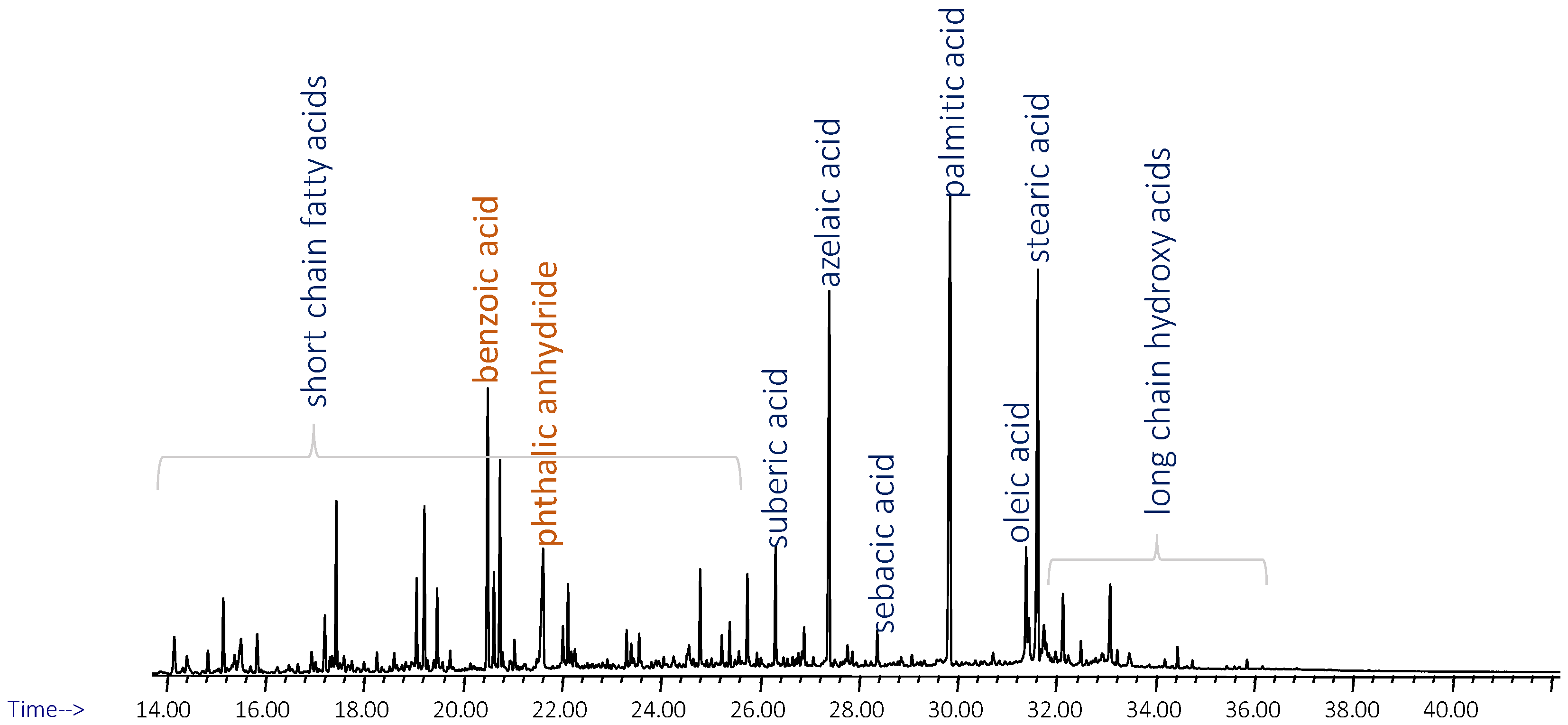
2.2.3. Py-GC-MS
2.2.4. HPLC-ESI-Q-ToF
3. Results
3.1. Superficie 207
3.2. Superficie 538
3.3. Superficie 553
4. Discussion
5. Conclusions
- -
- Superficie 207 (1957): egg yolk and hydrocerussite were identified in the industrial preparation mixed with acrylic resin and paraffin, while titanium white and styrene-acrylic resin were used for the preparation applied by Capogrossi. The background was created using a mixture of alkyd and styrene-acrylic resin, while for the black ideograms only an alkyd binder was used. In addition, on the black ideograms, a varnish layer based on acrylic resin was detected;
- -
- Superficie 538 (1961): a mixture of gypsum and animal glue was identified in the preparation applied by Capogrossi, while a combination of safflower oil and wax was used in the paint;
- -
- Superficie 533 (1965): an industrial preparation based on animal glue, oil and egg yolk was identified; the preparation layer applied by Capogrossi was based on zinc and titanium white in the oil binder and a pictorial film based on oil. The black ideograms were created with ivory black mixed with Prussian blue and beeswax or paraffin.
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Chiantore, O.; Rava, A. Conservare L’arte Contemporanea: Problemi, Metodi, Materiali, Ricerche; Electa: Florence, Italy, 2005. [Google Scholar]
- Learner, T.; Learner, T.J.S.; Smithen, P.; Institute, G.C.; Krueger, J.W.; Art, N.G.O.; Schilling, M.R.; Gallery, T. Modern Paints Uncovered: Proceedings from the Modern Paints Uncovered Symposium; Getty Conservation Institute: Los Angeles, CA, USA, 2007. [Google Scholar]
- van den Berg, K.J.; Bonaduce, I.; Burnstock, A.; Ormsby, B.; Scharff, M.; Carlyle, L.; Heydenreich, G.; Keune, K. Conservation of Modern Oil Paintings; Springer Nature: Cham, Switzerland, 2019. [Google Scholar]
- Van den Berg, K.J.; Burnstock, A.; De Keijzer, M.; Krueger, J.; Learner, T.; De Tagle, A.; Heydenreich, G. Issues in Contemporary Oil Paint; Springer Nature: Cham, Switzerland, 2014. [Google Scholar]
- Jabloiiski, E.; Learner, T.; Hayes, J.; Golden, M. Conservation concerns for acrylic emulsion paints: A literature review. Rev. Conserv. 2003, 4, 3–12. [Google Scholar]
- Learner, T. Analysis of Modern Paints; Getty Publications: Los Angeles, CA, USA, 2004. [Google Scholar]
- Ploeger, R.; Scalarone, D.; Chiantore, O. The characterization of commercial artists’ alkyd paints. J. Cult. Herit. 2008, 9, 412–419. [Google Scholar] [CrossRef]
- Learner, T. The analysis of synthetic resins found in twentieth century paint media. In Proceedings of the Resins Ancient and Modern: Pre-Prints of the SSCR’s 2nd Resins Conference, Aberdeen, UK, 13–14 September 1995; pp. 76–84. [Google Scholar]
- Eastman, P. Characterization of Acrylics at Rohm and Haas. In Proceedings of the Extended Abstracts, the Fifth Annual Infrared and Raman Users Group Conference, Los Angeles, CA, USA, 4–8 March 2002; pp. 88–89. [Google Scholar]
- Digney-Peer, S.; Bumstock, A.; Leamer, T.; Khanjian, H.; Hoogland, F.; Boon, J. The migration of surfactants in acrylic emulsion paint films. Stud. Conserv. 2004, 49, 202–207. [Google Scholar] [CrossRef]
- Ormsby, B.; Learner, T. The effects of wet surface cleaning treatments on acrylic emulsion artists’ paints–A review of recent scientific research. Stud. Conserv. 2009, 54, 29–41. [Google Scholar] [CrossRef]
- La Nasa, J.; Zanaboni, M.; Uldanck, D.; Degano, I.; Modugno, F.; Kutzke, H.; Tveit, E.S.; Topalova-Casadiego, B.; Colombini, M.P. Novel application of liquid chromatography/mass spectrometry for the characterization of drying oils in art: Elucidation on the composition of original paint materials used by Edvard Munch (1863–1944). Anal. Chim. Acta 2015, 896, 177–189. [Google Scholar] [CrossRef]
- La Nasa, J.; Nodari, L.; Nardella, F.; Sabatini, F.; Degano, I.; Modugno, F.; Legnaioli, S.; Campanella, B.; Tufano, M.K.; Zuena, M.; et al. Chemistry of modern paint media: The strained and collapsed painting by Alexis Harding. Microchem. J. 2020, 155, 104659. [Google Scholar] [CrossRef]
- Izzo, F.C.; Balliana, E.; Pinton, F.; Zendri, E. A preliminary study of the composition of commercial oil, acrylic and vinyl paints and their behaviour after accelerated ageing conditions. Conserv. Sci. Cult. Herit. 2014, 14, 353–369. [Google Scholar]
- Stella, E.M.; Bracci, S.; Iannaccone, R.; La Nasa, J.; Colombini, M.P. Violon. Céret by Pablo Picasso: The case of a lost painting. A methodological approach. J. Cult. Herit. 2019, 35, 199–208. [Google Scholar] [CrossRef]
- Hendriks, L.; Hajdas, I.; Ferreira, E.S.; Scherrer, N.C.; Zumbühl, S.; Smith, G.D.; Welte, C.; Wacker, L.; Synal, H.-A.; Günther, D. Uncovering modern paint forgeries by radiocarbon dating. Proc. Natl. Acad. Sci. USA 2019, 116, 13210–13214. [Google Scholar] [CrossRef]
- Modugno, F.; Di Gianvincenzo, F.; Degano, I.; van der Werf, I.D.; Bonaduce, I.; van den Berg, K.J. On the influence of relative humidity on the oxidation and hydrolysis of fresh and aged oil paints. Sci. Rep. 2019, 9, 5533. [Google Scholar] [CrossRef]
- Ciccola, A.; Tozzi, L.; Romani, M.; Serafini, I.; Ripanti, F.; Curini, R.; Vitucci, F.; Guidi, M.C.; Postorino, P. Lucio Fontana and the light: Spectroscopic analysis of the artist’s collection at the National Gallery of Modern and Contemporary Art. Spectrochim. Acta Part A: Mol. Biomol. Spectrosc. 2020, 118319. [Google Scholar] [CrossRef]
- Argan, G.C. Capogrossi, Catalogo Ragionato; Editalia: Florence, Italy, 1967. [Google Scholar]
- Bucarelli, P.; Mantura, B. Giuseppe Capogrossi Catalogo Della Mostra (Roma, Galleria Nazionale d’Arte Moderna, Valle Giulia, 12 Dicembre 1974–2 Febbraio 1975; De Luca: Roma, Italy, 1974. [Google Scholar]
- Mantura, B. Capogrossi Fino al 1948, Catalogo della Mostra (Roma, Complesso Monumentale di San Michele a Ripa, Sala delle Navi, 9 Ottobre–23Novembre 1986; De Luca-Mondadori: Roma, Italy, 1986. [Google Scholar]
- Frezzotti, F.; Italiano, C.; Rosso, A. Galleria Nazionale d’Arte Moderna e Maxxi. Le collezioni 1958–2008; Electa: Florence, Italy, 2009. [Google Scholar]
- Rosi, F.; Cartechini, L.; Sali, D.; Miliani, C. Recent trends in the application of Fourier Transform Infrared (FT-IR) spectroscopy in Heritage Science: From micro-to non-invasive FT-IR. Phys. Sci. Rev. 2019, 4. [Google Scholar] [CrossRef]
- Mancini, D.; Percot, A.; Belot-Gurlet, L.; Carnazza, P.; Colomban, P. Contactless approach to identify binders of German and Italian modern painting through portable vibrational spectroscopy. In Proceedings of the 3rd International Conference on Innovation in Art—INART 2018, Parma, Italy, 26–29 March 2018. [Google Scholar]
- Orsini, S.; La Nasa, J.; Modugno, F.; Colombini, M.P. Characterization of Aquazol polymers using techniques based on pyrolysis and mass spectrometry. J. Anal. Appl. Pyrolysis 2013, 104, 218–225. [Google Scholar] [CrossRef]
- La Nasa, J.; Orsini, S.; Degano, I.; Rava, A.; Modugno, F.; Colombini, M.P. A chemical study of organic materials in three murals by Keith Haring: A comparison of painting techniques. Microchem. J. 2016, 124, 940–948. [Google Scholar] [CrossRef]
- Wei, S.; Pintus, V.; Schreiner, M. A comparison study of alkyd resin used in art works by Py-GC/MS and GC/MS: The influence of aging. J. Anal. Appl. Pyrolysis 2013, 104, 441–447. [Google Scholar] [CrossRef]
- Fardi, T.; Pintus, V.; Kampasakali, E.; Pavlidou, E.; Schreiner, M.; Kyriacou, G. Analytical characterization of artist’s paint systems based on emulsion polymers and synthetic organic pigments. J. Anal. Appl. Pyrolysis 2018, 135, 231–241. [Google Scholar] [CrossRef]
- Learner, T. The analysis of synthetic paints by pyrolysis–gas chromatography–mass spectrometry (PyGCMS). Stud. Conserv. 2001, 46, 225–241. [Google Scholar]
- Chiantore, O.; Scalarone, D.; Learner, T. Characterization of Artists’ Acrylic Emulsion Paints. Int. J. Polym. Anal. Charact. 2003, 8, 67–82. [Google Scholar] [CrossRef]
- Osete-Cortina, L.; Doménech-Carbó, M.T. Analytical characterization of diterpenoid resins present in pictorial varnishes using pyrolysis–gas chromatography–mass spectrometry with on line trimethylsilylation. J. Chromatogr. A 2005, 1065, 265–278. [Google Scholar] [CrossRef]
- Osete-Cortina, L.; Doménech-Carbó, M.T. Characterization of acrylic resins used for restoration of artworks by pyrolysis-silylation-gas chromatography/mass spectrometry with hexamethyldisilazane. J. Chromatogr. A 2006, 1127, 228–236. [Google Scholar] [CrossRef]
- Saliu, F.; Modugno, F.; Orlandi, M.; Colombini, M.P. HPLC–APCI-MS analysis of triacylglycerols (TAGs) in historical pharmaceutical ointments from the eighteenth century. Anal. Bioanal. Chem. 2011, 401, 1785–1800. [Google Scholar] [CrossRef] [PubMed]
- Blanco-Zubiaguirre, L.; Ribechini, E.; Degano, I.; La Nasa, J.; Carrero, J.A.; Iñañez, J.; Olivares, M.; Castro, K. GC–MS and HPLC-ESI-QToF characterization of organic lipid residues from ceramic vessels used by Basque whalers from 16th to 17th centuries. Microchem. J. 2018, 137, 190–203. [Google Scholar] [CrossRef]
- Maniccia, E. The Restoration of Superficie 553 by Giuseppe Capogrossi Conserved at National Gallery of Modern and Contemporary Art in Rome. Giuseppe Capogrossi, Investigations on Executive Techniques by Scientific Analysis. Master’s Thesis, University of Palermo, Palermo, Italy, 2019. [Google Scholar]
- Peduzzi, A. Diagnostic Investigations for the Characterization of 20th Century Italian Paintings. Master’s Thesis, University of Rome “La Sapienza”, Rome, Italy, 2018. [Google Scholar]
- De Luca, D.; Carnazza, P. Le Opere di Giuseppe Capogrossi alla Galleria Nazionale di Roma. Indagini Sulle Tecniche Esecutive e Intervento di Restauro; Edizioni TAB: Rome, Italy, 2020; in press. [Google Scholar]
- Di Tullio, V.; Proietti, N. New Insights to Characterize Paint Varnishes and to Study Water in Paintings by Nuclear Magnetic Resonance Spectroscopy (NMR). Magnetochemistry 2020, 6, 21. [Google Scholar] [CrossRef]
- La Nasa, J.; Biale, G.; Ferriani, B.; Colombini, M.P.; Modugno, F. A pyrolysis approach for characterizing and assessing degradation of polyurethane foam in cultural heritage objects. J. Anal. Appl. Pyrolysis 2018, 134, 562–572. [Google Scholar] [CrossRef]
- Carlesi, S.; Ricci, M.; Cucci, C.; Nasa, J.L.; Lofrumento, C.; Picollo, M.; Becucci, M. Multivariate Analysis of Combined Fourier Transform Near-Infrared Spectrometry (FT-NIR) and Raman Datasets for Improved Discrimination of Drying Oils. Appl. Spectrosc. 2015, 69, 865–876. [Google Scholar] [CrossRef]
- Rosi, F.; Miliani, C.; Braun, R.; Harig, R.; Sali, D.; Brunetti, B.G.; Sgamellotti, A. Noninvasive analysis of paintings by mid-infrared hyperspectral imaging. Angew. Chem. Int. Ed. 2013, 52, 5258–5261. [Google Scholar] [CrossRef]
- Rosi, F.; Daveri, A.; Moretti, P.; Brunetti, B.G.; Miliani, C. Interpretation of mid and near-infrared reflection properties of synthetic polymer paints for the non-invasive assessment of binding media in twentieth-century pictorial artworks. Microchem. J. 2016, 124, 898–908. [Google Scholar] [CrossRef]
- Vagnini, M.; Gabrieli, F.; Daveri, A.; Sali, D. Handheld new technology Raman and portable FT-IR spectrometers as complementary tools for the in situ identification of organic materials in modern art. Spectrochim. Acta Part A: Mol. Biomol. Spectrosc. 2017, 176, 174–182. [Google Scholar] [CrossRef]
- Tsuge, S.; Ohtani, H.; Watanabe, C. Pyrolysis-GC/MS Data Book of Synthetic Polymers: Pyrograms, Thermograms and MS of Pyrolyzates; Elsevier: Amsterdam, The Netherlands, 2011. [Google Scholar]
- Bonaduce, I.; Ribechini, E.; Modugno, F.; Colombini, M.P. Analytical approaches based on gas chromatography mass spectrometry (GC/MS) to study organic materials in artworks and archaeological objects. In Analytical Chemistry for Cultural Heritage; Springer: Berlin/Heidelberg, Germany, 2017; pp. 291–327. [Google Scholar]
- Colombini, M.P.; Modugno, F.; Ribechini, E. GC/MS in the characterization of lipids. In Organic Mass Spectrometry in Art and Archaeology; Colombini, M.P., Modugno, F., Eds.; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Workman, J., Jr.; Weyer, L. Practical Guide and Spectral Atlas for Interpretive Near-Infrared Spectroscopy; CRC Press: Boca Raton, FL, USA, 2012. [Google Scholar]
- Monico, L.; Rosi, F.; Miliani, C.; Daveri, A.; Brunetti, B.G. Non-invasive identification of metal-oxalate complexes on polychrome artwork surfaces by reflection mid-infrared spectroscopy. Spectrochim. Acta Part A: Mol. Biomol. Spectrosc. 2013, 116, 270–280. [Google Scholar] [CrossRef]
- Gabrieli, F.; Rosi, F.; Vichi, A.; Cartechini, L.; Pensabene Buemi, L.; Kazarian, S.G.; Miliani, C. Revealing the nature and distribution of metal carboxylates in Jackson Pollock’s Alchemy (1947) by micro-attenuated total reflection FT-IR spectroscopic imaging. Anal. Chem. 2017, 89, 1283–1289. [Google Scholar] [CrossRef]
- Daveri, A.; Malagodi, M.; Vagnini, M. The bone black pigment identification by noninvasive, in situ infrared reflection spectroscopy. J. Anal. Methods Chem. 2018, 2018, 6595643. [Google Scholar] [CrossRef] [PubMed]
- Orsini, S.; Parlanti, F.; Bonaduce, I. Analytical pyrolysis of proteins in samples from artistic and archaeological objects. J. Anal. Appl. Pyrolysis 2017, 124, 643–657. [Google Scholar] [CrossRef]
- Stacey, R.J.; Dyer, J.; Mussell, C.; Lluveras-Tenorio, A.; Colombini, M.; Duce, C.; La Nasa, J.; Cantisani, E.; Prati, S.; Sciutto, G. Ancient encaustic: An experimental exploration of technology, ageing behaviour and approaches to analytical investigation. Microchem. J. 2018, 138, 472–487. [Google Scholar] [CrossRef]
- Miliani, C.; Rosi, F.; Daveri, A.; Brunetti, B.G. Reflection infrared spectroscopy for the non-invasive in situ study of artists’ pigments. Appl. Phys. A 2012, 106, 295–307. [Google Scholar] [CrossRef]
- Mayer, R. The Artist’s Handbook of Materials and Techniques; Viking Press: New York, NY, USA, 1991. [Google Scholar]
- Lee, J.; Bonaduce, I.; Modugno, F.; La Nasa, J.; Ormsby, B.; van den Berg, K.J. Scientific investigation into the water sensitivity of twentieth century oil paints. Microchem. J. 2018, 138, 282–295. [Google Scholar] [CrossRef]
- Caravá, S.; García, C.R.; de Agredos-Pascual, M.L.V.; Mascarós, S.M.; Izzo, F.C. Investigation of modern oil paints through a physico-chemical integrated approach. Emblematic cases from Valencia, Spain. Spectrochim. Acta Part A: Mol. Biomol. Spectrosc. 2020, 118633. [Google Scholar] [CrossRef]
- Samain, L.; Silversmit, G.; Sanyova, J.; Vekemans, B.; Salomon, H.; Gilbert, B.; Grandjean, F.; Long, G.J.; Hermann, R.P.; Vincze, L. Fading of modern Prussian blue pigments in linseed oil medium. J. Anal. At. Spectrom. 2011, 26, 930–941. [Google Scholar] [CrossRef]
- Colombini, M.P.; Modugno, F. Organic Mass Spectrometry in Art and Archaeology; John Wiley & Sons: Hoboken, NJ, USA, 2009. [Google Scholar]
- Vacanti, S. Il Piccolo Trattato di Tecnica Pittorica di Giorgio de Chirico: Teoria e Prassi del “Ritorno al Mestiere” (1919–1928); Nardini: Firenze, Italy, 2014. [Google Scholar]
- Smith, R. The Artist’s Handbook; DK Publishing: London, UK, 2003. [Google Scholar]
- Capogrossi, G.; Morelli, F.R. Giuseppe Capogrossi, Catalogo Ragionato 1920–1949; Skira: Milano, Italy, 2012. [Google Scholar]
- Piva, G. Manuale Pratico di Tecnica Pittorica: Enciclopedia Ricettario per Tutti Gli Artisti, Pittori, Dilettanti, Allievi Delle Accademie di Belle Arti e Delle Scuole Artistiche; Hoepli Editore: Milano, Italy, 1984. [Google Scholar]
- Orlandi, P.A.; Guarienti, P. L’ Abecedario Pittorico; Pasquali: Italy, 1753. [Google Scholar]
- Daudin-Schotte, M.; Bisschoff, M.; Joosten, I.; van Keulen, H. Dry cleaning approaches for unvarnished paint surfaces. In Proceedings of the New Insights into the Cleaning of Paintings, the Cleaning 2010 International Conference, Valencia, Spain, 26–28 May 2010. Universidad Politécnica de Valencia and Museum Conservation Institute. [Google Scholar]
- Cremonesi, P. Proprietà ed Esempi di Utilizzo di Materiali Siliconici nel Restauro di Manufatti Artistici; Il Prato Publishing House srl: Villatora, Italy, 2016. [Google Scholar]
- Cremonesi, P.; Héritier, P.A. Un Approccio Innovativo Alla Pulitura di Superfici Dipinte Sensibili: La Combinazione Simultanea di Erogazione Controllata di Liquido e Micro-Aspirazione; Nardini Editore: Firenze, Italy, 2018. [Google Scholar]










| Samples or Analysed Areas | Py GC/MS Results | FT-IR Results | Elemental Analysis [57] | Identified Materials |
|---|---|---|---|---|
| Preparation layers: | ||||
| Industrial preparation | Ethylacrylate based acryl polymer; egg yolk, paraffin wax | Hydrocerussite [2PbCO3∙Pb(OH)2] probably mixed with a proteinaceous and/or a lipid material | SEM-EDS: Pb | Acryl resin, egg yolk and paraffin |
| Preparation applied by Capogrossi | - | Possible acryl resinin mixture with titanium white | SEM-EDS: Ti XRF: Ti, Zn | Acryl resin, titanium white |
| Pictorial Layers: | ||||
| Colored squares of the background | Acryl resin (ethylacrylate) and styrene resins; alkyd resin | Acryl resin; styrene component | - | Acryl resin in mixture with alkyd resin |
| Black areas | Alkyd resin | Lipid-based medium; ivory black | SEM-EDS: Ca XRF: Fe, Ca | Alkyd resin with ivory black |
| Varnish on the Black Areas | ||||
| - | Acryl resin | - | Only on the black ideograms: a varnish layer based on acryl resin applied by the artist and restoration varnish applied after the vandalization | |
| Samples or Analyzed Areas | HPLC-MS | Py GC/MS Results | FT-IR Results | Elemental Analysis [57] | Identified Materials |
|---|---|---|---|---|---|
| Preparation Layers: | |||||
| Preparation applied by Capogrossi | - | Animal glue (pyrrole, 2-methyl-pyrrole and diketopiperazines) | Gypsum in mixture with a proteinaceous material | XRF: Ca | Gypsum and animal glue |
| Pictorial Layers: | |||||
| Opaque black areas | - | Oil medium | Oil medium; beeswax or paraffin; calcium carbonate | XRF: Ca | Oil medium, beeswax or paraffin and calcium carbonate. |
| Glossy black areas | Safflower oil | Oil medium | Oil medium; beeswax or paraffin; calcium carbonate | Oil medium (safflower oil) and beeswax or paraffin. | |
| Orange area | - | - | Oil medium | - | Oil medium |
| White area | Safflower oil | Oil medium | Titanium and zinc white; oil medium | XRF: Zn, Ti | Titanium and zinc white in oil medium (safflower oil) |
| Samples or Analyzed Areas | Py GC/MS Results | FT-IR Results | Elemental Analysis [57] | Identified Materials |
|---|---|---|---|---|
| Preparation layers: | ||||
| Industrial preparation | Animal glue and oil medium | - | - | Animal glue, oil medium and egg yolk |
| Preparation applied by Capogrossi | - | Oil medium | XRF: Zn, Ti | Zinc and titanium white in oil medium |
| Pictorial layers: | ||||
| White area (background) | - | Oil medium; titanium and zinc white | XRF: Zn, Ti | Zinc and titanium white in oil medium |
| Black area | - | Oil medium; beeswax and ivory black in mixture with Prussian blue | XRF: Ca, Fe | Ivory black in mixture with Prussian blue in oil medium and beeswax |
| Red area | - | Oil medium; beeswax | - | Oil medium and beeswax |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
La Nasa, J.; Moretti, P.; Maniccia, E.; Pizzimenti, S.; Colombini, M.P.; Miliani, C.; Modugno, F.; Carnazza, P.; De Luca, D. Discovering Giuseppe Capogrossi: Study of the Painting Materials in Three Works of Art Stored at Galleria Nazionale (Rome). Heritage 2020, 3, 965-984. https://doi.org/10.3390/heritage3030052
La Nasa J, Moretti P, Maniccia E, Pizzimenti S, Colombini MP, Miliani C, Modugno F, Carnazza P, De Luca D. Discovering Giuseppe Capogrossi: Study of the Painting Materials in Three Works of Art Stored at Galleria Nazionale (Rome). Heritage. 2020; 3(3):965-984. https://doi.org/10.3390/heritage3030052
Chicago/Turabian StyleLa Nasa, Jacopo, Patrizia Moretti, Eleonora Maniccia, Silvia Pizzimenti, Maria Perla Colombini, Costanza Miliani, Francesca Modugno, Paola Carnazza, and Daphne De Luca. 2020. "Discovering Giuseppe Capogrossi: Study of the Painting Materials in Three Works of Art Stored at Galleria Nazionale (Rome)" Heritage 3, no. 3: 965-984. https://doi.org/10.3390/heritage3030052
APA StyleLa Nasa, J., Moretti, P., Maniccia, E., Pizzimenti, S., Colombini, M. P., Miliani, C., Modugno, F., Carnazza, P., & De Luca, D. (2020). Discovering Giuseppe Capogrossi: Study of the Painting Materials in Three Works of Art Stored at Galleria Nazionale (Rome). Heritage, 3(3), 965-984. https://doi.org/10.3390/heritage3030052







