Abstract
This article reports the findings of an exploratory study on the complexity and challenges of managing UNESCO World Natural Heritage Sites (WNHS). Despite their protected status, the majority of the WNHS currently face severe anthropogenic stress. While several studies have analyzed this scenario with spatial analysis and review of global trends, this research provides a qualitative analysis of ongoing fragmentation and impact in a specific site by employing the Ecological Integrity (EI) concept to the Shiretoko Peninsula WNHS in Japan. Insights from key scientific literature are synthesized with those from research and monitoring of the Blakiston’s Fish Owl (a keystone species). Findings indicate that ecosystems of Shiretoko WNHS have not sufficiently recovered from the intensive anthropogenic disruption in the 20th century and in some cases face novel stressors from tourism development after the inscription. Besides, ongoing construction of roads and facilities affects fine-scale heterogeneity in the surrounding landscape. The findings highlight the urgent need of protecting lower trophic level species, stream and forest restoration, and restricting infrastructure development. This implies that WNHS managers must implement measures to meaningfully offset anthropogenic stress on EI, and WNHS management should be integrated with the management of its surrounding landscapes.
1. Introduction: The Problem of Managing Natural Heritage on a Changing Planet
This article reports the findings of an exploratory study that sought to understand the complexity and challenges of managing World Natural Heritage Sites from a case study of the Shiretoko Peninsula World Heritage Site in Japan, with a specific focus on one of the keystone species (the Blakiston’s Fish Owl). The article specifically focuses on the fragmentation of landscape level connectivity pathways and the potential of using the Ecological Integrity concept for World Natural Heritage Sites (WNHS) management. World Heritage Sites are known for their Outstanding Universal Value [1], and WNHS, in particular, contain some of the most extraordinarily beautiful landscapes, a high degree of biodiversity and geodiversity, and habitats for many endangered species [2]. These sites therefore are important both for their scientific and social values, the latter mainly rooted in their popularity with tourists and their potential to generate economic benefits [3]. However it has also been noted that currently nearly half of the 229 registered sites face some type of threat such as the deterioration of ecosystems resulting from the expansion of industrial activities and urbanization [4,5,6]; and those threats are amplified by factors such as institutional failure, poor management execution, and conflicts [7,8]. This occurs despite the World Heritage recognition supposedly bringing the highest level of international recognition to the sites [9,10,11]. The problem is also related to the global trend of increasing anthropogenic pressure on Protected Areas (PA) that results in the fragmentation of ecosystems and key interaction pathways between biotic and abiotic nature [12,13]. In their seminal work on systematic quantitative assessment of anthropogenic threats to the WNHS between 1993 and 2009, Allan et al. (2017) found that anthropogenic pressure increased in 63% of the sites during this period: the loss of forest cover and ecological integrity were notable issues in a majority of the sites [14]. They noted that many WNHS are deteriorating rapidly and that changes are currently under-assessed, and mentioned the need for site-level case studies to complement quantitative assessments that aggregate conditions across a large number of sites. This article aims to respond to this need by providing a qualitative analysis of the situation in the Shiretoko Peninsula. The qualitative approach was taken because key indicators are not well identified for the concerned site, as well as for the aim of highlighting the complexity angle.
While quantitative studies such as Allan et al. (2017) take up spatial analysis tools and numerical indicators to describe anthropogenic impact [14], this article presents a holistic analysis through review of literature and empirical data from observing a key species over time; and reflects on the condition of its constituent ecosystem through a discussion of ‘ecological integrity.’
Ecological Integrity (EI) is a management guideline based on the wholeness of natural processes and organisms. Parks Canada (2018) defines EI as “…a condition that is determined to be characteristic of its natural region and likely to persist, including abiotic components and the composition and abundance of native species and biological communities, rates of change and supporting processes [15].” In their article on the topic, Parrish et al. (2003) highlighted the presence (or lack) of functioning ecological processes over appropriate spatial and temporal scales as the key facet of EI [16]. Elsewhere, Miller and Rees (2000) described EI as an ‘umbrella term’ that encompasses both the scientific understanding of wilderness and the social process of valuing it [17]. Westra et al. (2000) pointed out that the concept of integrity is based on ‘wholeness’, and in the case of EI, the wholeness of wild nature (i.e., natural areas and processes that are relatively unmodified by anthropogenic impact) form the most important benchmark [18]. They also observed that for any natural area to retain its integrity, the natural processes and the organisms or agents that partake in them must be functional over time, i.e., an apparently healthy ecosystem observed at a single point in time may not provide a good indication of its ecological integrity. These points have vital ramifications for WNHS management in particular, and for protected area management in general. However it was pointed out by Brown and Williams (2016) that the EI metric to biodiversity is not without its problems, and observed that EI-based assessments may be subject to systematic bias due to the tendency to appropriate diverse data into numerical scores as well as due to observer bias at the ground level [19]. In order to avoid this controversy, this research does not seek to convert the condition of the concerned ecosystem into categorized scores but seeks to present a more holistic and qualitative portrait of the concept of ‘integrity’ in the concerned natural heritage landscape.
The focus of this article, the Shiretoko Peninsula WNHS in Hokkaido Island of Japan comprises of marine and terrestrial ecosystems of outstanding productivity and mutual interactions. But as it is located in a highly industrialized country, the natural landscape and its inhabitants have faced constant threats from development and anthropogenic impact over the last hundred years. Currently there are four WNHS in Japan and anthropogenic pressure on ecosystems can be observed in each of them [20]. Major drivers of anthropogenic disturbance are infrastructure building and land development, extensive deforestation during the last century, tourism and recreational development, and the lack of robust and binding conservation targets [21,22,23,24]. Although Hokkaido is generally seen as the major island least affected by urbanization and development, it nevertheless witnessed widespread land conversion, deforestation, alteration of natural landscapes, and urbanization during the past hundred years [25,26,27,28]. Logging of natural forests and conversion of large areas supporting local flora and fauna into spaces for agricultural and settlement occurred during early-to-mid 20th century in Shiretoko itself [29,30]. Shoyama and Braimoh (2011) drew the important conclusion that despite a major effort of reforesting logged areas, forests of Shiretoko did not recover at patch level—the mean patch size of forest at 2004 was a mere 24% compared to 1947 (the beginning of the rapid land development phase) [31]—this finding provides an important benchmark for understanding the current condition of EI. The area was inscribed onto the World Heritage list in 2005 under Criteria ix, x of the World Heritage Convention; and currently its natural resources are widely seen as significant capital for tourism development. The UNESCO portal on the site mentions several problems such as industrial fisheries, presence of numerous dams on the waterways, and tourism impact as factors that currently affect the integrity of the Shiretoko Peninsula WNHS [32]. In addition, other factors such as climate change effects and over-abundance of particular species (such as the deer species Cervus nippon) are also mentioned as potential problems [32]. A further point of worry here is that protected areas in general are increasingly threatened by land conversion and intensive development in their surrounding landscape [33,34,35,36]. The case of Shiretoko Peninsula, in this sense, is a valuable lesson both in its immediate context as well as for its implications for conservation challenges relating to natural heritage in general.
The article is divided into the following sections. First, the conceptual basis of Ecological Integrity and its fragmentation is explained. Descriptions of the study area and methods used for this research follow. Subsequently, the main problems affecting the EI of the property seen through the lens of the Blakiston’s Fish Owl (Bubo Blakistoni) are outlined. These sections lead to a discussion highlighting the major findings and a brief conclusion summing up the study.
2. Conceptual Basis: Ecological Integrity and Its Fragmentation, and Implications for WNHS
2.1. Ecological Integrity
Ecological Integrity (EI) as a management concept has been widely utilized in the Canadian National Parks from the late twentieth century [37,38]. The concept of EI can encompass a range of assumptions and objectives; but the key premise is that natural ecosystems that evolved over millennia retain the full suite of their (native) species and the biophysical conditions that are necessary for their survival or evolution over time [38]. In this sense EI is a complementary approach to biodiversity and ecosystem health [38,39] and upholds the case for complexity, resilience, and appropriate composition of ecosystems across spatial and temporal scales. EI is has a crucial difference from the concept of Ecological Resilience (ER) [40] as the ER concept emphasizes the functionality of the ecosystem, implying that if key functions are intact, an ecosystem that suffers loss of species can still be seen as resilient, whereas EI holds that a full species suit or a condition similar to it is essential for the integrity of an ecosystem. Translated to management of natural heritage areas, EI is crucially based on the two overarching questions asked by Parrish et al. (2003): (i) ‘Are the most critical threats that confront the biological resources at the park changing in their severity or geographic scope as a result of conservation strategies (or lack thereof)?’ and (ii) ‘Do the ecological systems, communities, and species that are the focus of conservation efforts occur with sufficient size, with appropriately functioning ecological processes, and with sufficiently natural composition, structure, and function to persist over the long term?’ [16; 852]. The most important feature that drives species richness, habitat integrity and wholeness of ecosystems is the availability of material and energy (i.e., throughput for work). That nutrient availability is a key limiting factor for ecosystems is already known for a long time through the seminal works of Likens et al. (1978) [41] and Odum (1985) [42] that highlighted the fact that as ecosystems become stressed they lose the capacity to retain and circulate nutrients and the appropriate structure of food webs. Thus, if there are signs that a natural heritage area (or a protected area with a significant natural ecosystem component) is suffering from nutrient unavailability across trophic levels, it can be said with confidence that the area in question falls short on EI, and appropriate management actions are needed to restore EI. However, here we encounter a familiar problem: most key indicators, whether at species levels or ecosystem-type levels, are poorly understood and insufficiently monitored over time, generating flawed data and much confusion [16,19,43]. It is therefore necessary to complement existing quantitative data with conceptual insights and appreciation of functions and species roles and condition in the ecosystem from a qualitative viewpoint; which can be expounded through relatively descriptive questions of ‘how different species are connected in the food web’ or ‘what type of change has recently happened in key species habitat and foraging areas?’ Such exploratory inquiries are especially helpful where there is lack of data on specific components or a lack of institutional capacity to implement rigorous scientific assessment of ecosystem conditions over time.
WNHS are typified by threatened species and complex ecosystems. Their Outstanding Universal Value (OUV) is derived from aspects such as significant habitats for threatened species and ecosystems, significant examples of ongoing key biological and geological processes and diversity, and landscapes of rare visual beauty or aesthetic appeal [10,11]. Applying the concept of EI for identifying the threats faced by these properties at the site level is therefore appropriate, regardless of the location and jurisdictional structure of the properties. In addition, where the WNHS are located in advanced industrial countries they are likely to be the last remaining refugia for key wild species and loss of integrity of those areas is likely to drive such species and ecosystems to extinction at local or regional scales.
2.2. Fragmentation
Over the years, fragmentation of species habitats and key ecological interaction pathways (inter-species and biotic–abiotic networks) has emerged as the most serious threat to ecological integrity [44,45]. Fragmentation is defined here as the division of formerly larger and more contiguous habitat areas into smaller, isolated remnants [46]. With respect to forest cover, intensification of human impact on the global scale was observed, resulting in the parceling of formerly large forest ecosystems into smaller fragments with reduced ecosystem function [47]. At the ecosystem level, the key problem is that division of formerly larger wilderness areas results in the compromise of ecosystem connectivity: Adhikari and Hansen (2018) showed that land use intensification in the US progressively fragmented wildland ecosystems [48]; Saravia et al. (2018) calculated fragmentation-related global biodiversity loss at 13–75% [49]; and Anderson et al. (2018) documented how existing and proposed dams on Andean watersheds are causing cascading impacts on thousands of species that are sustained by the Andean-Amazon connectivity [50].
The implications from these studies are clear: today, there is scarcely an ecosystem left that is not adversely affected by anthropogenic impact, and at least for terrestrial ecosystems, rapid intensification of human-induced fragmentation has significantly reduced the ecological integrity and functionality of formerly rich ecosystems over a very short period of time. As Allan et al.’s study clarified, even the best measures to protect natural heritage such as the UNESCO World Heritage recognition are inadequate and can be seen as failing in this task so far: a major drawback in this regard is that most World Heritage sites are listed on the basis of their grandeur and exceptionality [2,51], rather than their common plight from anthropogenic change. While there is no denying the importance of most of those sites and the significant conservation benefits garnered by World Heritage listing, such narratives arguably provide a false sense of safety against the backdrop of constant erosion of the key attributes of natural heritage sites at the global level. Related to this is the problem that there are no explicit and concrete mitigation (preservation) measures at many sites to meaningfully offset the expanding anthropogenic impact. Clear indicators of threats and change will be required, but even prior to that, it is necessary to re-conceptualize natural heritage as systems that have been recently fragmented due to anthropogenic impact, and this perspective requires the appreciation of their many interconnected aspects at the qualitative level. A holistic understanding of the ecosystem and its current fragmented state is therefore important for WNHS; especially as many of these sites show a remarkably complex and composite nature of biophysical processes and species, as illustrated by our case study below.
3. Study Area: Shiretoko as Natural Heritage Against the Backdrop of Fragmentation of Ecosystems
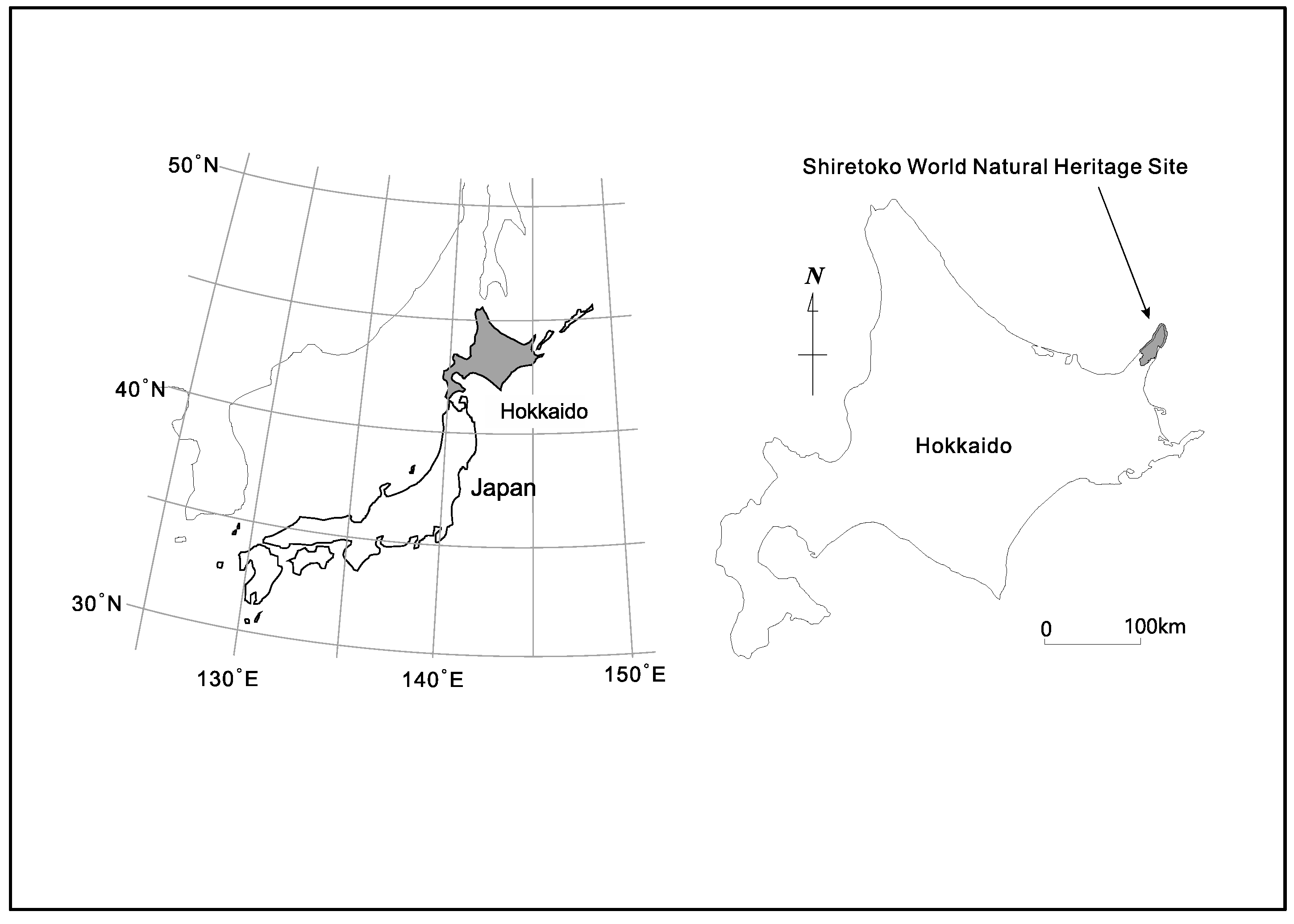
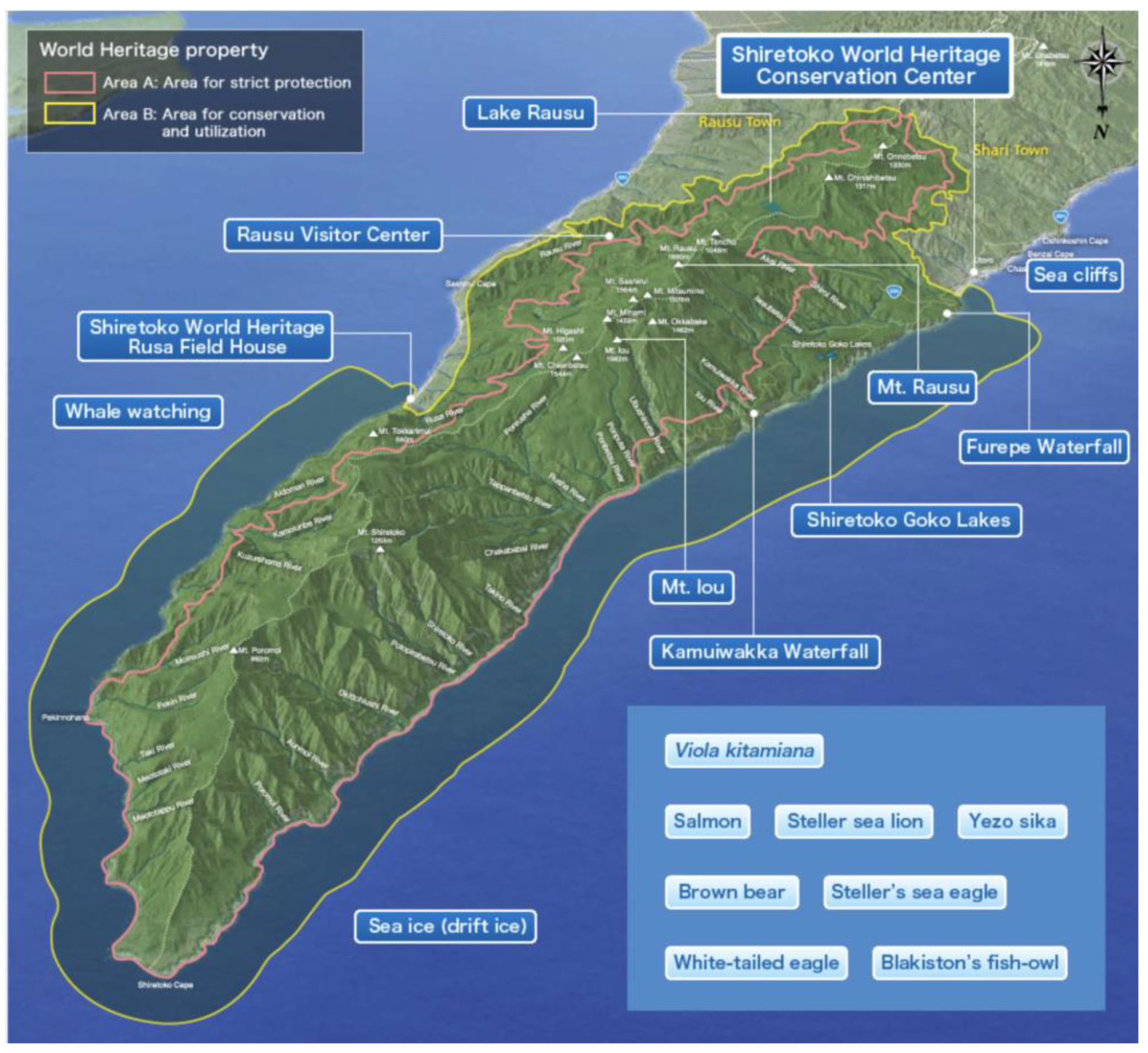
The 71,000 ha. property of Shiretoko Peninsula currently inscribed onto the WNHS list is located in the northeast of Hokkaido Island (see Figure 1 and Figure 2 for an outline of the WNHS property). The Outstanding Universal Value (OUV) of the property is centered on the integration of marine and terrestrial ecosystems. Marine areas surrounding Shiretoko are locations for the formation of sea ice at the most southerly latitude in the Northern Hemisphere. Sea ice formation supports a rich productivity of diatoms that form the base of a vast food web that sustains cetaceans, sea lions, the brown bear, many species of raptors, and migratory birds. The local continental shelf topography, oceanic circulation, and distant factors such as the freshwater pulse provided by the Amur River into the Sea of Okhotsk (crucial for the formation of sea ice), are important biophysical characteristics of the area. Ishikawa (2010) documented 871 plant species in the area of which 97 are endangered [52]; and Shiretoko is either home or seasonal habitat for a number of endangered species such as the Blakiston’s Fish Owl (B. blakistoni), Steller’s Sea Eagle (Haliaeetus pelagicus), Steller’s Sea Lion (Eumetopias jubatus) as well as several cetacean and seal species. In addition, the peninsular landmass provides one of the last remaining contiguous habitat range of the Brown Bear (Ursus arctos) in the Japanese Islands. Chakraborty (2018) provided a detailed analysis of the connectivity between the geological, geomorphological and ecological components of the heritage system—and noted the important role of numerous drainage channels that connect the volcanic upland formations (mountains) to the marine environment [21]. While under natural conditions such drainage systems are able to generate a rich variety of landform and landscape diversity through active erosion, material transport, and habitat provision, currently the rivers in the peninsula are affected by extensive anthropogenic engineering of their watercourses through damming, check weirs, and artificial embankments. These structures and local fishing practices of maximizing catch affect the natural condition of fish migration (notably salmon run consisting of the Chum, Pink, and Masu salmon among others) in such a way that the biomass transportation network throughout the peninsula is now fundamentally altered due to human impact. The WNHS State of Conservation Document of 2017 explicitly mentions check dams and related infrastructure as having considerable negative effect on fish migration and biomass/sediment transport along waterways [32]. The report also notes the continued practice of culling of Steller’s Sea Lions due to the conflict between industrial fisheries (maximal catch) and the species’ food habit—and mentions that the sea lion population at subspecies level had previously witnessed a drastic collapse between 1977 and 2007 due to unknown reasons (unrelated to the current culling practice). Thus, despite the subsequent trend of ‘recovery’, several species remain highly vulnerable. The foregoing discussion also clarifies that despite Shiretoko Peninsula being popularly portrayed as a wild, undisturbed landscape, the heritage property and its constituent parts are under considerable pressure due to anthropogenic fragmentation.
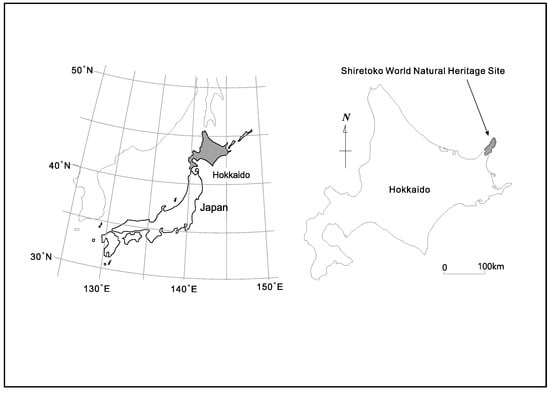
Figure 1.
Location of Shiretoko Peninsula (figure courtesy T. Takenaka).
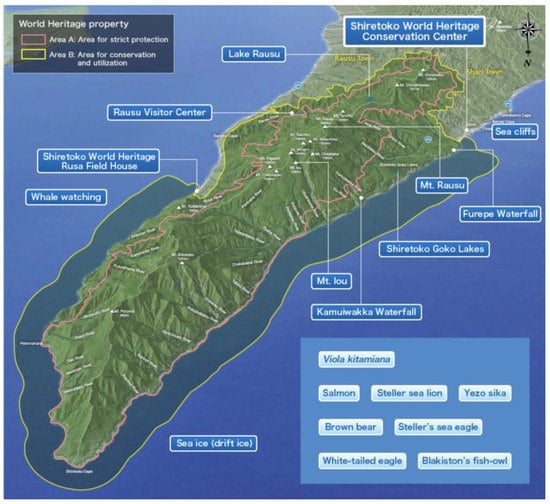
Figure 2.
(Map of Shiretoko Peninsula World Natural Heritage Sites (WNHS) (inverted image of the area compared to Figure 1, see compass bearing top right for reference). Source Ministry of the Environment website (World Natural Heritage in Japan) (http://www.env.go.jp/nature/isan/worldheritage/en/shiretoko/index.html) (Accessed 20 February 2019).
4. A Note on Materials and Methods
This article synthesizes two phases of research: findings of a research project on the habitat and ecology of the Blaksiton’s Fish Owl (B. blakistoni) carried out by the second author for over two decades are combined with the findings from an intensive phase of field visits jointly carried out by the two authors between February 2017 and November 2018. The research project on the fish owl’s habitat and ecology included fish owl census, monitoring of hunting behavior, surveys of stream health (fish species) and observation of forest composition. Field research involved banding of young fish owls and monitoring of owls through installation of CCD cameras in artificial nests and radio transmitters, as well as understanding of owl behavior by following their calls across watersheds [53,54]. Important data such as sex ratio of the owls, life-span, productivity, territories, and food habits, were gained in the process. Those findings were tallied with the four intensive field visits jointly conducted by the two authors during 2017–2018 where key sites were revisited, landscape levels characteristics were observed, and interviews with stakeholders were carried out.
The research adopted a ‘case study’ approach [55,56]. The spatial unit of Shiretoko Peninsula was considered a case for analysis, and the case was analyzed both synchronically through intensive field visits and diachronically from the insights of previous research referred above. During the intensive field visit phase, a total of 18 interviews were conducted with key stakeholders to compare their viewpoints and to identify recent developments in the area. These stakeholders were identified from their affiliation to the WNHS management structure, from their reputation as key informants in the local community, and from their profession (in the case of guides). The interviews were open-ended in consistence with the aim of the overall qualitative framework of the study. Interview data were coded (using open and axial coding) [57,58] to identify relevant information and to tally information with findings from previous research. The article combines findings from these data-gathering phases with instructive literature in the field to present a holistic picture.
Analysis of EI and its anthropogenic fragmentation was based on identification of key spatial and ecological attributes and understanding of key functional aspects of the fish owl’s ecosystem. Key spatial attributes included presence or absence of conditions for feeding and breeding (such as fish and tree species) and the condition of the landscape in the fish owl territory (nature of the forest, whether the forest was degraded compared to its previous state, presence or absence of anthropogenic barriers for fish movement in rivers, and presence or absence of roads, built structures, and tourist facilities in or near the fish owl’s habitat). These attributes in turn provided insights on the functional aspects of the fish owl’s ecosystem. Overdependence on a particular food source indicated depletion of alternative food species; lack of suitable trees for nesting indicated poor breeding success (unless aided by artificial measures) as well as the absence of forest environment necessary for supporting keystone species; presence of artificial barriers on streams indicated poor stream health and fragmentation of biomass circulation as well as cascading effects on the riparian biota; presence of roads and built environments provided evidence of depletion of fine-scale landscape heterogeneity and connectivity; and proliferation of tourists or related anthropogenic disturbances indicated adverse effects on hunting and breeding of an endangered species. These factors are also interrelated and impart a cumulative effect on the ecosystem; therefore, analysis of these aspects required understanding the fish owl’s ecology as a complex and interrelated entity. Such understanding was achieved at times through triangulation of existing literature sources, previous fieldwork findings, and evidence from intensive field visits and interviews.
5. Results: Challenges for EI in a WNHS as Seen Through the Case of the Blakiston’s Fish Owl
The main findings from this research are:
- i.
- Legacies of past land conversion, logging, and stream modification are pervasive and currently limit fish owl survival and breeding in a significant manner, i.e., the EI of the ecosystem has declined over time.
- ii.
- As the fish owl is a keystone species, its current vulnerability shows the declined state of key ecological attributes of the WNHS area: specifically the deterioration of suitable forest composition to maintain rich ecosystems and modification of stream environments resulting in the rupture of upstream-downstream connectivity and a decline of fish availability upstream constitute a deterioration of the material-energy circulation in the system; and hence are major drawbacks for EI.
- iii.
- Recent tourism and related infrastructure development are novel stressors that further impact the EI of the area and the situation is complicated by a low level of awareness in a section of local stakeholders.
These insights are explained in detail below along with descriptions of the ecological characteristics of B. Blakistoni.
5.1. A Brief Note on Blakiston’s Fish Owl as an Endangered Species
The Blakiston’s Fish Owl (B. blakistoni) is the planet’s largest owl species at ~70 cm body length, upto 4.6 kg of body-weight and ~180 cm wingspan [59] (a photo of the owl is provided in Figure 3 above). First described by ornithologist Henry Seebohm based on a specimen collected by the English naturalist Thomas Blakiston in 1883, this raptor finds its habitat range in Russia and northeast Asia. As its name suggests, the fish owl survives on a diet that mainly consists of fish. Globally there are an estimated 1000–1900 individuals, of which around 160–180 individuals are found in Japan. The species was widespread in Hokkaido Island in early 20th century, but its numbers fell drastically during the mid-to-late 20th century due to intense logging, land conversion, river engineering, and urban development. Its habitat steadily diminished throughout this period and currently all fish owl populations exist in fragments of their former habitat range [52]. Fish owl numbers fell to below 100 individuals in the early years of the 1990s and the species became critically endangered. The main threats faced by the species were: drastic reduction of large trees of ~1 m diameter-at-breast-height (DBH) (a crucial requirement for nesting) due to extensive logging of natural forests, fragmentation of their territories and feeding grounds by linear infrastructure intrusions, alteration of riverflow and riverine biomass distribution due to channel engineering, and overharvesting of crucial food resource such as fish. Takenaka (2018) detailed the far-reaching changes to the owl habitats and the ecology of Hokkaido in general: rivers were polluted due to paper mill effluents, potato starch factories, and coalmine tailings; fundamental change in watershed properties resulted from large dams and small check weirs for regulating water and sediment flow that currently number in thousands; and rivers were regulated by channel engineering [53,54]. A conservation-restoration program targeting the critically endangered fish owl began in the 1980s, which resulted in the set-up of 300 artificial nests to facilitate breeding, at present nearly 80% of breeding fish owl pairs of Hokkaido are dependent on nest boxes [53].

Figure 3.
Blakiston’s Fish Owl (photo courtesy T. Takenaka).
Among the breeding pairs, nearly half are located within the Shiretoko Peninsula (both inside the WNHS territory and adjacent areas). There are currently 30 breeding pairs in Shiretoko of which 11 pairs inhabit the WNHS territory, making Shiretoko the most important habitat-area for conserving fish owls in Japan. In 1993 the fish owl was declared a nationally endangered species [53], which aided the subsequent efforts to conserve the species. The second author of this article has led the fish owl conservation/breeding assistance program with support from the Ministry of the Environment and the Forestry Agency. During the stage of preparation of Shiretoko as a World Heritage nomination, the fish owl’s presence was used for appealing to both national and international audience to recognize the value of the natural environment.
5.2. The Ecology of Fish Owls
True to its name, the fish owls persist mainly on fish. In the case of Shiretoko, the species is apparently highly dependent on the Dolly Varden Char (Salvelinus malma)—observation of food mass brought into one nest showed that S. malma occupied 67% of that pair’s diet (a photograph of the Dolly Varden Char is given below in Figure 4). This has implications for EI and ecosystem integrity, as observed in the following section. It appears that though the fish owls of Shiretoko mainly feed on fluvial fish, marine fish is also an important part of their diet—this is why fish owls are often observed flying into the rivermouth or coastal areas where they have higher chances of collision with vehicles and other artificial infrastructure. Fluvial fish are generally caught within a few kilometers of the nest and marine fish predation mainly occurs at late night—perhaps indicating that the owls have adapted to avoid human activities in the coast during earlier hours.

Figure 4.
Dolly Varden Char (S. malma), the mainstay of the fish owl’s diet in the Shiretoko Peninsula (photo courtesy T. Takenaka).
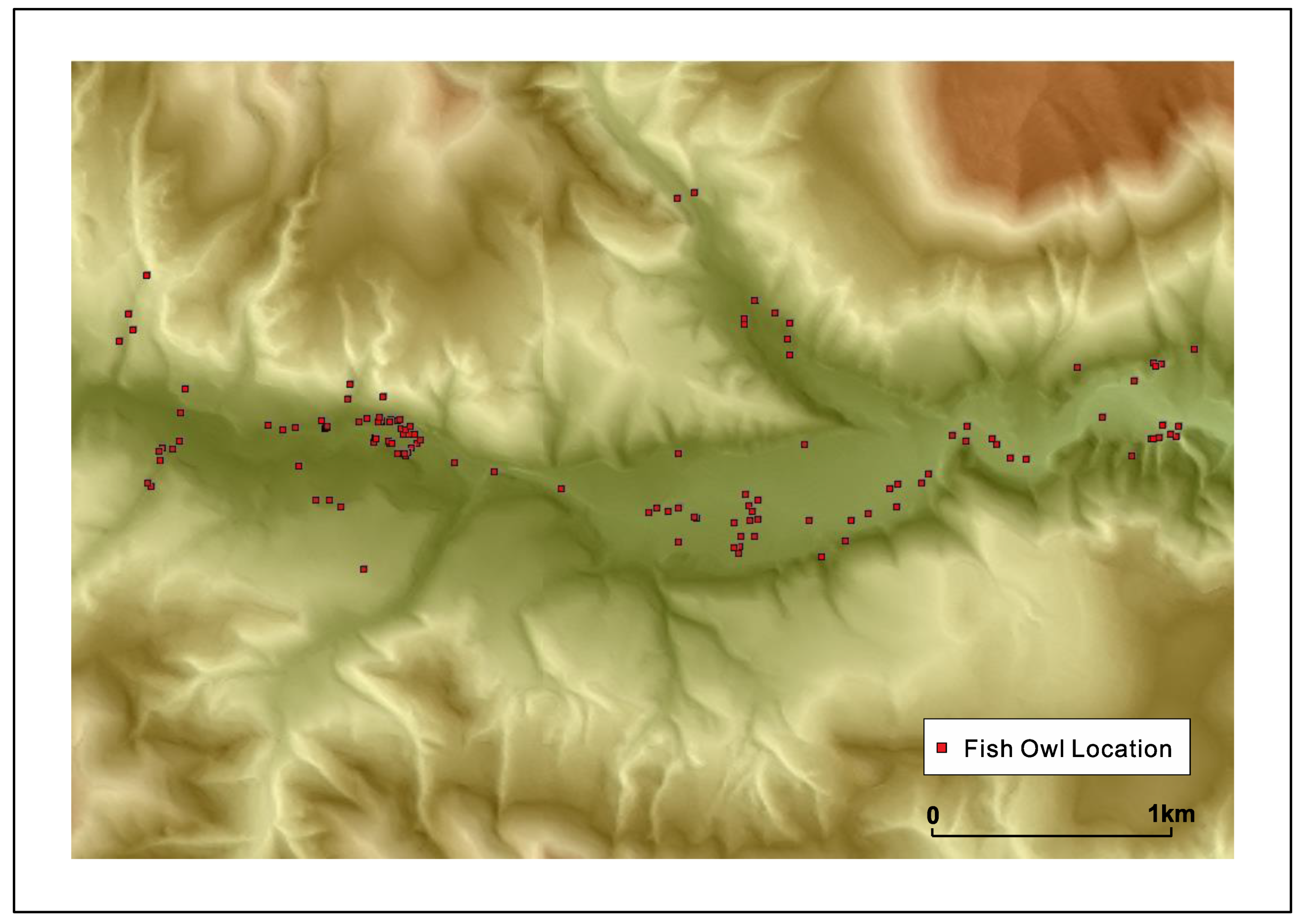
Fish owls nest in pairs and usually the pairing is long-term. A pair establishes and guards its ‘territory’. It was revealed by careful observation and tracking by transmitters that the territory of a pair can extend normally up to 10 km in Hokkaido but the range tends to be a little smaller in Shiretoko, possibly due to landform barriers of between drainage basins and better fish availability; although territory size can be influenced by the density of fish owls in an area as well owing to the highly territorial nature of these raptors. A graphic of a fish owl’s movement across a drainage basin is provided in Figure 5 below in order to illustrate the dimensions of its territory. From the second author’s sampling of fish biomass across 33 river basins in Hokkaido, it was established that fish owls would require presence of 25 salmonid fish/100 m2 over the feeding area, which roughly corresponds to the excess of 1000 gm./100 m2 of fish biomass in the minimum. It was also found that rivers with a high density of S. malma supported a higher density of fish owls compared to streams where Salvelinus leucomaenis (White-spotted Char) and Oncorhynchus masou masou (Masu salmon) were dominant. As Kitano (1995) described, S. malma is a relict species from the last glacial maximum restricted to colder mountainous streams [60], and this raises the possibility that the fish owls of Shiretoko are uniquely adapted to this species. It is also likely that human modification of watercourses that once supported high density of anadromous fish are in a state of higher disturbance and therefore inhibit fish owl survival, leading to the adaptation of the species to colder mountainous stream ecology in remote areas.
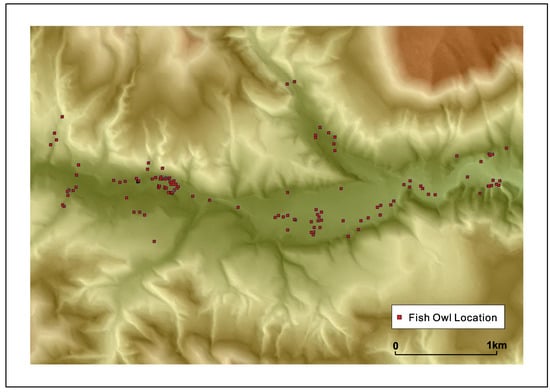
Figure 5.
A fish owl’s movement along a drainage basin showing the broad contours of the species’ territory (figure courtesy T. Takenaka).
Fish owls require large cavities in trees to nest and therefore their intergenerational survival is closely related to the composition of the forest. Large deciduous trees are ideal for the species and from past records and current monitoring it was identified that deciduous varieties such as the Japanese Elm (Ulmus davidiana var. japonica), Mizunara Oak (Quercus crispula Blume), Manchurian Elm (Ulmus laciniata), and Katsura (Cercidiphyllum japonicum) are preferred. Photographs of these key tree species are given in Figure 6 below. As the species nests in large cavities, comparatively older trees are preferred, this implies that old-growth or undisturbed riparian forests were crucial for supporting a high number of fish owls in Hokkaido before the twentieth century economic development and deforestation; and that deforestation, especially logging of older forest tracts, was a main cause behind the swift decline of the fish owl population. Along with the reduction of such forest cover, watershed characteristics were also drastically changed due to human modification of watercourses, and this led a lasting impact on the fish owl’s ecology.

Figure 6.
Large deciduous trees are preferred by the Blakiston’s Fish Owl for nesting: (A) U. davidiana, (B) Q. crispula, (C) U. laciniata, and (D) C. japonicum. (photos courtesy T. Takenaka).
5.3. Current Stressors in the Fish Owl’s Habitat
The biggest current threats for the fish owl ecology are: (i) anthropogenic modification of riparian forests and (ii) the fragmentation of rivers in the WNHS area.
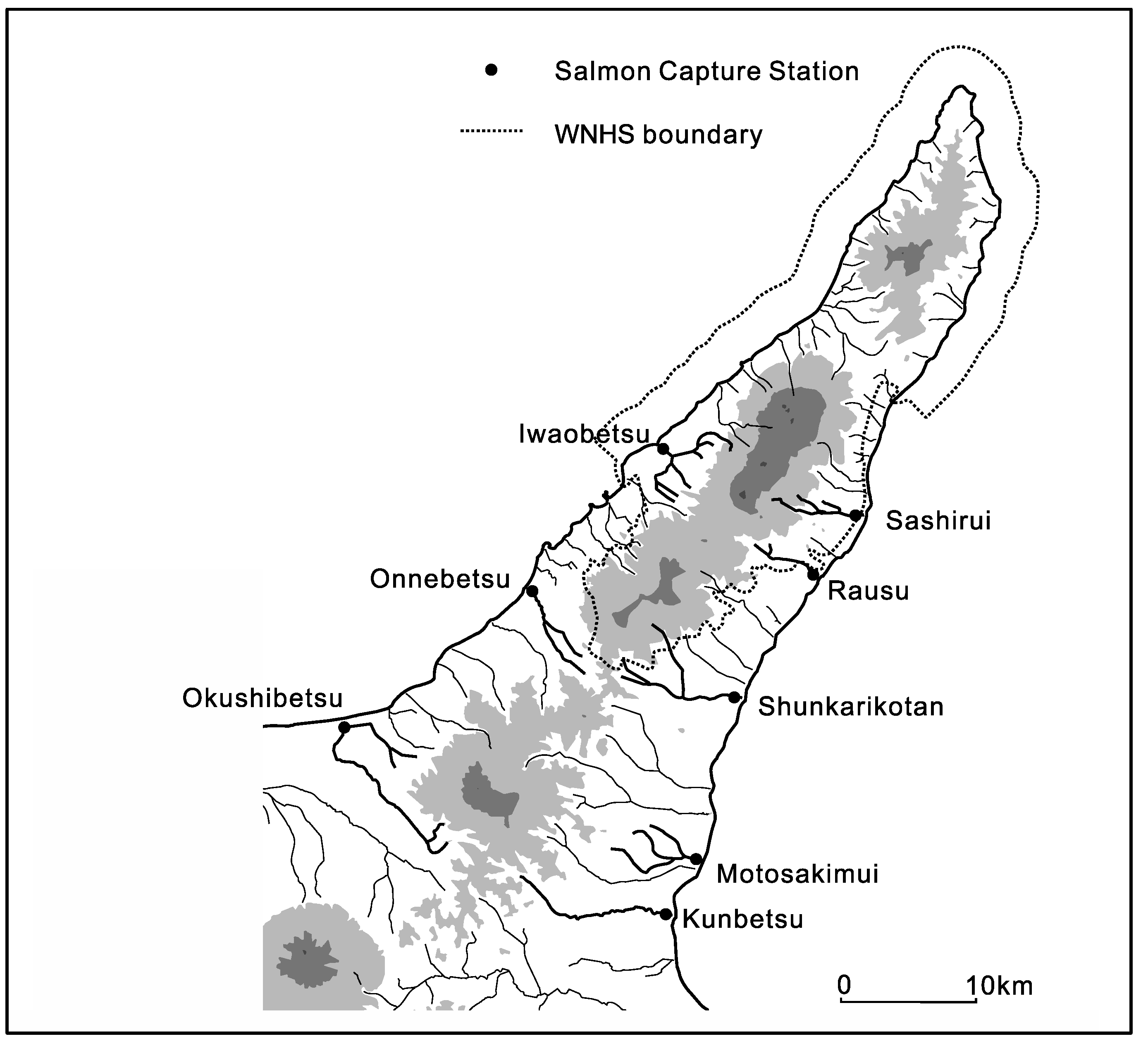
As mentioned above, large deciduous trees are vital for the intergenerational continuity of the species. However, the forests of Shiretoko had experienced logging on an extensive scale merely fifty years ago and have not recovered enough, largely due to the slow natural process of forest maturity. In the WNHS area the key deciduous species of U. davidiana, U. laciniata, Q. crispula, and C. japonicum cover a mere 12–28% of the forested area and trees over 1 m DBH are rare. Nearly all drainage basins have some form of anthropogenic impact and upstream-downstream connectivity is fragmented. A total of 8 artificial salmon capture stations are located in and around the WNHS, of which 3 are located within the WNHS territory (a graphic of their locations is provided in Figure 7 below). Thus the radial drainage that connects ecosystems across the WNHS and trophic levels within ecosystems is constrained and the circulation of material and energy occurs far below the natural level necessary to support these highly complex systems. Without any meaningful protection of prey species and the safeguarding of their migration pathways, conservation of the apex species is likely to remain a highly challenging task. For an instance; the important prey species of S. malma currently faces a novel threat from an increase of sport fishers in the area. Under the Japanese legal system, although national park land is protected, rivers do not have protected status and therefore can be exploited relatively freely. As noted above, S. malma is concentrated in the colder streams of Shiretoko, and an increase of amateur fishing activity is likely to have a strong impact on the species density in the streams. As the size of the streams is small, the effect of fishing can appear suddenly and in a drastic manner—which was observed at several sites. Although specific data is not currently available, based on repeated observations we fear that such change is already underway in some rivers of this area.
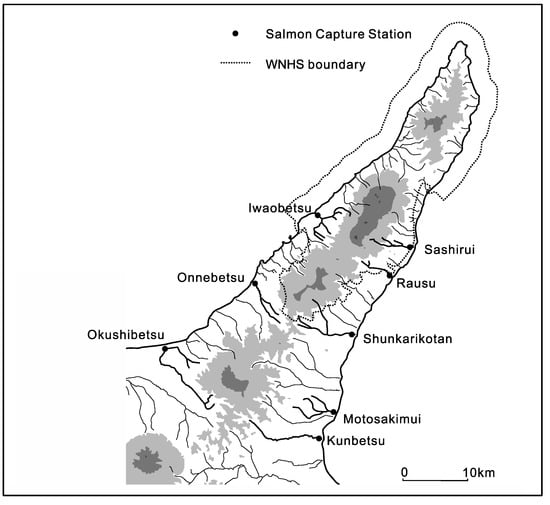
Figure 7.
Locations of salmon capture stations in and around the Shiretoko Peninsula WNHS. Areas marked in grey represent the volcanic mountains of the central highlands, with darker shaded areas representing major elevated peaks. It can be seen how short rivers flowing out of the central highlands form the radial drainage pattern that is crucial for sustaining biomass and ecosystems of the area. The salmon capture stations are significant visible points of anthropogenic alteration of material transport and trophic level connectivity in the area.
While the WNHS itself is protected, its surrounding areas are not; and this poses another significant threat for species like fish owls that locate themselves at the top of the local food chain. Roads connecting Shiretoko to surrounding areas have increasingly expanded in terms of surface area and were straightened to allow speedier traffic. In order to accommodate larger and straighter roads—which are partially in response to facilitate tourist access—coastal landfilling, tunnel construction, stabilization of erosion-prone hillslopes (facing the coast), construction of concrete breakwater, and logging of large trees have taken place since the 2005 inscription. The WNHS Visitor Center and the adjacent tourist facility in Shari Town are located on a large stretch of landfilled site on the bay that erased the fine-scale structure of the coastline of that section. As mentioned earlier the fish owls of Shiretoko also predate on marine fish for a substantial part of their diet and these developments affect their hunting behavior adversely. An increase of roads and vehicular traffic has also brought new risks. In 2018, two fish owls were involved in vehicular collisions; one of the birds died while the other was unable to return to the wild.
In addition to these physical barriers, fish owls and other animal species are also probably affected by tourism related activities. Currently an estimated 2.5 million visits occur in the Shiretoko Peninsula annually. Most tourism activities are concentrated at the peripheral sections of the terrestrial part of the WNHS, but the marine part of the property is widely utilized by sightseeing boats. Visiting the ‘five lakes’ area in Shari, hiking mountains in the central highland, kayaking/canoeing in nearshore waters, and boat cruises that offer glimpses of marine wildlife, are major tourism themes. Owing to heavy snowfall in winter, tourist activities are concentrated in summer to autumn, although drift ice watching tours and snowshoe walks in winter forests are popular as well. Tourism foci are contrasting in the two administrative units of Shari and Rausu Towns: most tourism attractions in Shari are terrestrial but in Rausu—which is also a major local fishing port—tourism packages include marine wildlife watching (cetacean and seal species in summer and birds in winter). Although impact on the natural environment from individual tours is likely to be low, there are worrying aspects such as artificial feeding of fishing eagles in the winter, isolated incidents of tourists throwing food to lure animals for photography, and eagerness of some photographers to approach bears. All of these activities potentially disrupt feeding and movement patterns of animals. In addition, cruise boats often venture close to rocky ledges used by a number of bird species for nesting and feeding, and the existence of a large number of similar tour packages possibly imparts an amount of stress to the local environment. There has been a rapid increase of guided tours and wildlife viewing packages in the area starting around the time when the property was being prepared for WNHS nomination. A number of guides currently engage in searching fish owls with high-powered searchlights in the night—as a part of the nighttime wildlife viewing tours that are gaining popularity. Some guides also take visitors to areas known as fish owl feeding spots and try to locate fish owls with searchlights, this is also likely to disturb the hunting success and feeding behavior of the species. There are even instances of tourists trying to search owls in the night with their own searchlights to avoid paying guiding charge. Apart from these, an inn in Rausu Town engages in baiting a fish owl pair with fish in an artificially lighted location to allow photographing opportunities. Although this practice is currently limited to a single facility, the precedent set in this manner is not ideal for managing an endangered and sensitive species in a WNHS. The interviews revealed a generally low capacity of tourism stakeholders to manage human-wildlife conflict, dependence on middle-aged or older visitors from urban areas, a tendency to facilitate easy access, limited linguistic skills of guides inhibiting meaningful interaction with international visitors (who are coincidentally more likely to approach wildlife), lack of experience in guides in terms of familiarity with other WNHS, and a strong preference of retaining as many tourists as possible. There was also the important revelation that though nominally entrusted with the management of the WNHS, the national park managers are often seen as ‘outsiders’, with local actors such as fishermen and tourism business owners having a strong influence over real management. A table showing information with open and axial coding, along with the interview topic guide, are available as supplementary files.
6. Discussion: Insights from Ecological Integrity for Better Management of WNHS
The concept of EI allows us to think beyond species level to trophic and assemblage levels in ecosystems. Seen from this perspective, the fish owl case study offers several important insights regarding the current level of fragmentation and better management pathways for WNHS.
In this exploratory study, the EI concept was mainly used for describing the state of fragmentation in the trophic levels and species assemblages that are key for the long-term survival of resident keystone species such as fish owls. It was noted that two guiding questions regarding EI are: whether the most critical threats confronting the biological resources are changing severity or geographic scope in response to conservation strategies; and whether the ecosystems, communities, or species that are the foci of conservation occur with sufficient size, with appropriately functioning ecological processes, and with sufficiently natural composition, structure, and function to persist over the long term [16]. From the case study three overarching findings regarding ecosystem integrity can be provided: (i) the reduction of diversity in the prey species over time; (ii) the reduction of ecological functions of streams due to stream engineering; and (iii) the loss of appropriate habitat structure for both the predator and prey species. These conditions have not changed significantly since the WNHS inscription, and in some cases they face further and novel disturbances.
While the current owl population in Hokkaido shows a modest recovery compared to the 1980s, 80% of the owl pairs are dependent on artificial nest-boxes, and in some cases artificial feeding is necessary for increasing the productivity and survival of newborn owls. While artificial feeding is useful for preventing untimely death of owl chicks, it has the detrimental effect of discouraging young owls from dispersing from their natal territories—and over the years this has resulted in a higher ratio of inbreeding in Hokkaido in general. It is also noteworthy that the retraction of fish owl habitat in the twentieth century also contributed to the inbreeding problem. While in Shiretoko better availability of fish in the streams ensured that artificial feeding was not required for owl conservation, artificial nest-boxes had to be installed as deforestation had reduced suitable natural nesting opportunities drastically. The low-productivity of fish owls of Shiretoko reflects this situation. However, the presence of a facility that currently engages in baiting fish owls for facilitating animal photography carries the risk that in the future owls near this location will be able to outperform others in breeding success, resulting in unnatural genetic distribution. The recovery of suitable habitat is an urgent need; but the process to regenerate mature broadleaf forests is a slow one—and is still in its early stages. Similarly, restoration of stream biota and key ecosystem processes such as natural water-sediment transport regime is crucial. Some efforts to create ‘slits’ in existing silt-check dams and construction of fish ladders are aimed at improvement of biomass circulation at the watershed level—but rivers in Shiretoko, and Hokkaido in general, remain far-too-much constrained to allow near-natural biomass circulation. Thus, the two notable aspects of fragmentation that currently affect the EI of fish owl ecosystem—and in turn the terrestrial ecosystem components of the WNHS are:
- (i)
- the fragmentation of the forests and the change in their composition into a state where key species are not able to breed, hunt, and disperse at appropriate scales; and
- (ii)
- the fragmentation of biomass circulation, notably salmonid run in the rivers that support the biodiversity of the WNHS.
As Shoyama and Braimoh (2011) demonstrated; forests of the area still show a legacy of the twentieth century deforestation and land development and occupy considerably smaller patches compared to the pre-cultivation stage [31]. This fragmented state of the habitat poses a fundamental challenge for restoring the ecology of keystone species such as fish owls. It is important to add that as fish owls live for several decades, the species typically shows a ‘time-lag’ in its response to habitat conditions. This was observed in the case of their population shrinkage—while the cultivation of Hokkaido began in the early 20th century, fish owl populations fell to critical levels several decades later in the 1980s–1990s. Similarly, habitat recovery measures will not translate to immediate reduction of threats at the species level. Especially without suitable corridors and habitats for dispersal, the current condition of dependence on artificial feeding and problem of inbreeding will continue. This situation requires the restoration of mature broadleaf forests throughout Hokkaido Island, and especially in and around the Shiretoko Peninsula.
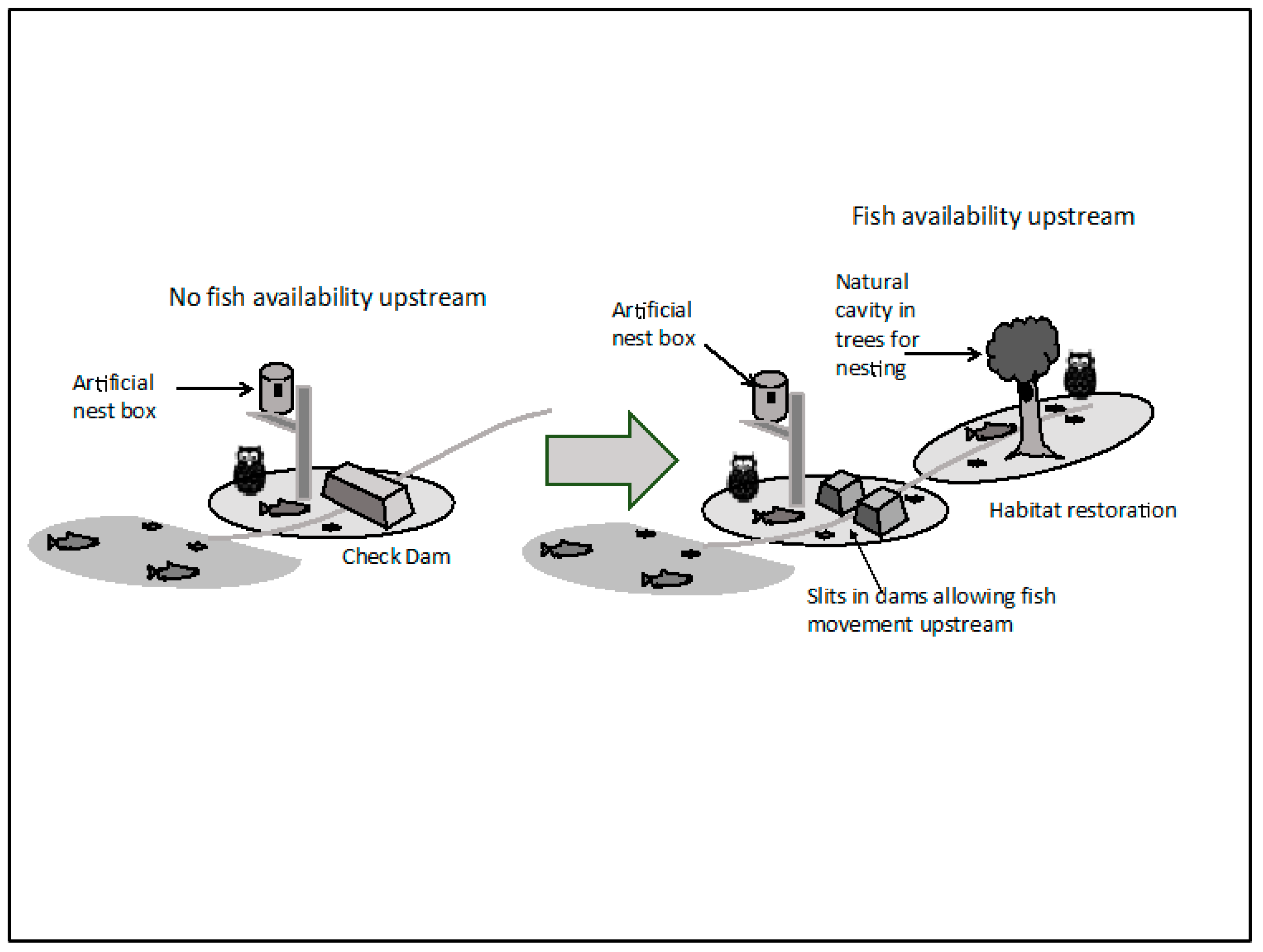
Secondly; most rivers in the WNHS have some type of artificial modification (including dams, check weirs, rivermouth weirs, artificial embankments, and channel engineering). This situation poses a significant barrier for the movement of anadromous fish species. Without the restoration of salmonid migration in the rivers of the WNHS, it is likely that the apex predator species will remain vulnerable. It should also be kept in mind that in the case of Shiretoko, the fish owl’s successful survival is largely due to the availability of fish species such as S. malma—but as this particular fish is adapted to colder stream temperatures, it will likely fare poorly if climate change results in increased warming of the streams. Besides, as reported earlier, the recent tendency of amateur fishers to catch S. malma—which is allowed to continue due to the lack of provisions to stop fishing in rivers—carries the added risk of reducing the species density in Shiretoko’s streams. From this point of view, it is clear that the food source of the fish owl is in a vulnerable state due to over-reliance on a single species (S. malma) which itself is under considerable stress. With reference to the point made by Woodley (2010) [38], it can be said that the trophic levels do not show the desired level of integrity due to unavailability of a diversity of prey species in the rivers; and the implication for the integrity of nutrient cycling is also negative. While the ‘State of Conservation’ document of 2017 mentions river engineering and aquaculture as having negative effect on natural salmonid run in the area [32], in our opinion the problem is under-stated, and swift and effective measures are required to restore the natural fish cycle. Allowing fish and biomass to pass artificial barriers by designing slits in check dams or removing them whenever appropriate will be a meaningful step for restoring the resource flow across drainage basins; this is illustrated in Figure 8 below. There is an urgent need for river management to shift from artificial modification of watercourses in order to safeguard infrastructure, profit, and human interest to a more holistic vision of managing rivers in a world heritage site for their ecological functions.
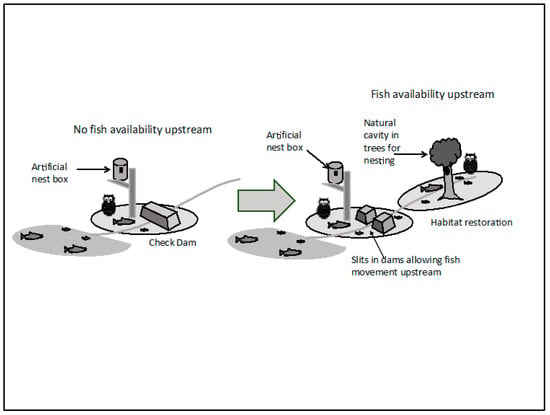
Figure 8.
While current check dams prohibit fish and biomass movement resulting in diminution of the river basin’s capacity to support fish owls (left side of the central arrow), allowing passage of fish and biomass through slits in dams (or removal of those structures) (right side of the central arrow) and restoration of riparian forests will be supportive of accommodating larger number of fish owls per basin (figure courtesy T. Takeneka).
It is also an added cause for concern that the WNHS inscription has apparently led to emergence of some novel stressors in this already fragmented environment. Construction of roads, tunnels, and infrastructure (outside the WNHS area but nevertheless in locations that are used by species such as the fish owl) to accommodate tourist demand; as well as increased pressure on the species from the behavior of tourist and tour guides, are two prominent examples of new stressors. Larger roads bring in speedier traffic, thereby increasing the chance of mortality by accident; and road, tunnel, and buildings erase the fine scale heterogeneity in the landscape that supports the interaction between predator and prey species. The practice of chasing fish owls with searchlight, taking visitors to fish owl feeding areas in the night, and baiting fish owls for photography enthusiasts all constitute worrying signs indicating that this fragile species is being exploited as a tourism resource with potentially adverse impacts on its hunting behavior.
Therefore, from this case study it can be posited that while the fish owl remains a primary conservation focus in this WNHS, the ecological processes required for its long-term survival and the ecosystem within which the species is located do not occur with sufficiently natural composition, structure, and functional attributes; and therefore the ecosystem within which the species is found lacks integrity. While Shiretoko sells ‘connectivity’ between ecosystems as a mainstay for its claim of Outstanding Universal Value, upon close inspection it become clear that connectivities between species, communities, and ecosystems remain in a fragmented and fragile state. The conceptual framework of EI gives us vital insights about the nature of this fragmentation, and is useful even in cases where precise data on the scale and state of fragmentation is not available. The following recommendations are drawn from these insights for the management of this WNHS:
- (i)
- Seen in its role of a ‘keystone’ species, the fish owl is an indicator of the state of the ecosystem it fits into. Habitat shrinkage, dependence on artificial nests, and lack of diversity in food (prey species) are clear signs that the environment in the WNHS (particularly riparian forest composition, stream ecological health, and spatial connectivity) is in a highly fragmented state. This situation requires proactive forest and stream restoration; particularly restoration of forests at suitable patch levels and with suitable tree species, and rehabilitation of natural stream functions.
- (ii)
- As the fish owl is nocturnal and sensitive, it is difficult to monitor the species directly, but it will be useful to monitor the health of its food chain by observing the condition of its prey species and stressors on those species. As mentioned already, the food chain remains disrupted due to artificial alteration of salmonid species’ migration routes and aquaculture. While fishing of salmonid species cannot realistically be prohibited as they bring significant profit; it is evident that overharvesting of these species has resulted in poor stream health and fragmentation of trophic level integrity. Besides, salmonid migration is important for many other species such as U. arctos and fishing eagles. Urgent attention is therefore required to restore salmonid migration in the streams of WNHS.
- (iii)
- It will be necessary to protect prey or lower trophic level species (fish in this case) in order to safeguard the EI of sensitive ecosystems. Currently there is no restriction on fishing in the rivers of the WNHS and its adjacent areas—this situation has resulted in a highly vulnerable situation of trophic level connectivity. It will be required to develop local ‘codes of conduct’ to prohibit overharvesting of crucial prey species, if national level legal protection is not available.
- (iv)
- WNHS managers should proactively push back against development projects in adjacent areas as ecological integrity and species health cannot be managed by solely protecting the WNHS (the ‘kernel’ site) itself but requires protection and rehabilitation of landscapes surrounding the WNHS, particularly in the cases where it is clear that the species that are the foci of conservation use those areas for feeding, migration, or dispersal. In cases such as this particular WNHS this would entail the restriction of coastal development and protection of vegetation at the roadsides that provides crucial cover for species movement. Kormos et al. (2016) and Allan et al. (2017) pointed out the urgent need for utilizing existing mechanisms such as the ‘buffer zone’; and argued that expansion of appropriate buffers can be useful for protecting ecological integrity and promoting connectivity in and between ecosystems [36,61]. Our findings from the Shiretoko WNHS support this argument: the existing buffer zone around the property should be expanded based on ecological principles and its capacity to mediate anthropogenic stress.
- (v)
- It would be necessary to establish corridors for species movement and dispersal in areas around the heritage site. This in turn will require the understanding that the environment constrained within the WNHS cannot retain its integrity over the long term without connection to the broader landscape. Appropriate long-term plans of establishing ecological corridors for species movement as well as for movement of abiotic components such as water or silt that have crucial ecosystem functions are therefore required to address the problem of ecological integrity.
The broader significance of the findings is twofold: they help to generate relevant knowledge for effective safeguarding of natural heritage through the World Heritage Convention, and they provide instructive insights for effective management of natural heritage landscapes in general. Both Kormos et al. (2016) and Allan et al. (2017) noted that currently there are significant gaps in the World Heritage Convention that makes its efficacy to safeguard wilderness a limited one, and that there is a need to upscale heritage conservation by integrating sites [36,61]. EI, as analyzed in this study, can be a pertinent conceptual tool in this regard. Finally, a number of studies have highlighted the potential of EI to inform sustainable management of natural areas and raise social awareness [62,63]; and the insights gained from this WNHS are useful for designing robust natural heritage conservation schemes that can deliver meaningful results against the general backdrop of pervasive anthropogenic fragmentation of the planet’s biophysical heritage.
7. Conclusions
This article presented a qualitative and exploratory analysis of ecological integrity in a WNHS by focusing on the current fragmented condition of the ecosystem of B. blakistoni in Shiretoko Peninsula of Japan. It was described how natural connectivity and integrity of ecosystem components and processes were reduced to a fragmentary state within the last hundred years and how the legacy of this disturbance lingers on at landscape and ecosystem levels. It was argued that the ecosystems of Shiretoko Peninsula have not sufficiently recovered from the intensive development stage in the mid twentieth century, and in some cases they face novel stressors from activities such as tourism and recreation with the WNHS inscription. While stress is present at every level, keystone species are noticeably affected, and where the keystone species also constitute threatened or endangered species—as in the case of B. blakistoni—there is an urgent need of proactive preservation of the species itself, its habitat, and feeding resources. An important conclusion in this regard is that not only the focal species itself, but species at other trophic levels and key landscape level attributes that support interaction within and between ecosystems need to be protected. It was found that even as the forests and streams in the WNHS site remain fragmented, additional stress in the form of development of surrounding landscapes further diminishes ecological functions, fine-scale heterogeneity, and movement options for threatened species. This situation implies that protected areas, especially those that are considered important ‘natural heritage’ landscapes, are under unprecedented threat from intensification of anthropogenic activities in and around those sites; and continue to suffer from loss of species assemblages and loss of ecosystem integrity, the degrees of which are only beginning to be understood. Conserving such natural heritage areas therefore requires proactive and failsafe measures that can meaningfully reduce anthropogenic pressure on the concerned systems. UNESCO WNHS listing should facilitate conservation and provide impetus for rehabilitating ecosystems under stress but there is little visible evidence that this has been realized so far. While World Heritage managers are encouraged to use the WNHS appropriately to frame meaningful conservation goals, international heritage bodies such as UNESCO and the IUCN should also send out unambiguous signals regarding the importance of the preservation of ecological integrity of natural heritage sites.
Author Contributions
A.C. conceptualized and wrote the paper and provided data and analysis for ecological integrity and World Heritage. T.T. contributed data and findings for the sections related to Blakiston’s Fish Owl and provided the photographs/illustrations unless mentioned otherwise.
Funding
This research was partially funded by Wakayama University CTR Research Project Support Program FY 2017–2018.
Acknowledgments
The authors wish to thank the three reviewers for their comments and suggestions on the paper which were useful for improving the initial version.
Conflicts of Interest
The authors declare no conflict of interest.
References
- UNESCO. Convention Concerning the Protection of the World Cultural and Natural Heritage; UNESCO Publishing: Paris, France, 1972. [Google Scholar]
- UNESCO. The World’s Heritage: A Complete Guide to the Most Extraordinary Places; Collins: New York, NY, USA, 2009. [Google Scholar]
- Job, H.; Becken, S.; Lane, B. Protected Areas in a neoliberal world and the role of tourism in supporting conservation and sustainable development: An assessment of strategic planning, zoning, impact monitoring, and tourism management at natural World Heritage Sites. JOST 2017, 25, 1697–1718. [Google Scholar] [CrossRef]
- Osipova, E.; Shadie, P.; Zwahlen, C.; Osti, M.; Shi, Y.; Kormos, C.; Bertzky, B.; Murai, M.; Van Merm, R.; Badman, T. IUCN World Heritage Outlook 2: A conservation Assessment of All Natural World Heritage Sites; IUCN: Gland, Switzerland, 2017. [Google Scholar]
- WWF. Protecting People through Nature: Natural World Heritage Sites as Drivers of Sustainable Development; WWF International: Gland, Switzerland, 2016; Available online: http://awsassets.panda.org/downloads/wwf_dalberg_protecting_people_through_nature_lr_singles.pdf (accessed on 12 January 2019).
- Conniff, R. How Natural World Heritage Sites Are Being Spoiled. Yale Environment 360. 2017. Available online: https://e360.yale.edu/features/how-natural-world-heritage-sites-are-being-spoiled-unesco (accessed on 17 January 2019).
- Wang, Z.; Yang, Z.; Du, X. Analysis on the threats and spatiotemporal distribution pattern of security in World Natural Heritage Sites. Environ. Monit. Assess. 2015, 187, 4143. [Google Scholar] [CrossRef] [PubMed]
- Levin, N.; Ali, S.; Crandall, D.; Kark, S. World Heritage in danger: Big data and remote sensing can help protect sites in conflict zones. Global. Environ. Chang. 2019, 55, 97–104. [Google Scholar] [CrossRef]
- UNESCO. Operational Guidelines for the Implementation of the World Heritage Convention; UNESCO World Heritage Center: Paris, France, 2013; Available online: https://whc.unesco.org/archive/opguide13-en.pdf (accessed on 26 January 2019).
- IUCN. The Benefits of Natural World Heritage: Identifying and Assessing Ecosystem Services and Benefits Provided by the World’s Most Iconic Natural Places; IUCN: Gland, Switzerland, 2014; Available online: https://portals.iucn.org/library/node/44901 (accessed on 15 January 2019).
- IUCN. Issues Brief: Natural World Heritage; IUCN: Gland, Switzerland, 2018; Available online: https://www.iucn.org/sites/dev/files/natural_world_heritage_issues_brief_final.pdf (accessed on 12 January 2019).
- Geldmann, J.; Joppa, L.N.; Burgess, N.D. Mapping change in human pressure globally on land and within protected areas. Conserv. Biol. 2014, 28, 1604–1616. [Google Scholar] [CrossRef] [PubMed]
- Watson, J.E.M.; Darling, E.S.; Venter, O.; Maron, M.; Walston, J.; Possingham, H.P.; Dudley, N.; Hockings, M.; Barnes, M.; Brooks, T.M. Bolder science needed now for protected areas. Conserv. Biol. 2015, 30, 243–248. [Google Scholar] [CrossRef] [PubMed]
- Allan, J.R.; Venter, O.; Maxwell, S.; Bertzky, B.; Jones, K.; Shi, Y.; Watson, J.E.M. Recent increases in human pressure and forest loss threaten many Natural World heritage Sites. Biol. Conserv. 2017, 206, 47–55. [Google Scholar] [CrossRef]
- Parks Canada. Ecological Integrity. Parks Canada, 2018; Available online: https://www.pc.gc.ca/en/nature/science/conservation/ie-ei (accessed on 1 February 2019).
- Parrish, J.D.; Braun, D.P.; Unnasch, R.S. Are we conserving what we say we are?: Measuring ecological integrity within protected areas. Bioscience 2003, 53, 851–860. [Google Scholar] [CrossRef]
- Miller, P.; Rees, W.E. Introduction and outline of the Integrity concept. In Ecological Integrity: Integrating Environment, Conservation, and Health; Pimentel, D., Westra, L., Noss, R.F., Eds.; Island Press: Washington, DC, USA, 2000; pp. 3–18. [Google Scholar]
- Westra, L.; Miller, P.; Karr, J.R.; Rees, W.E.; Ulanowizcz, R.E. Ecological Integrity and the aims of the Global Integrity Project. In Ecological Integrity: Integrating Environment, Conservation, and Health; Pimentel, D., Westra, L., Noss, R.F., Eds.; Island Press: Washington, DC, USA, 2000; pp. 19–44. [Google Scholar]
- Brown, E.D.; Williams, B.K. Ecological Integrity assessment as a metric of biodiversity: Are we measuring what we say we are? Biodiver. Conserv. 2016, 25, 1011–1035. [Google Scholar] [CrossRef]
- Chakraborty, A.; Mokudai, K.; Cooper, M.; Watanabe, M.; Chakraborty, S. (Eds.) Natural Heritage of Japan: Geological, Geomorphological, and Ecological Aspects; Springer: Cham, Switzerland, 2018. [Google Scholar]
- Chakraborty, A. Shiretoko Peninsula: Dynamic interaction between geology, geomorphology, and ecology at the interface of terrestrial and marine systems. In Natural Heritage of Japan: Geological, Geomorphological, and Ecological Aspects; Chakraborty, A., Mokudai, K., Cooper, M., Watanabe, M., Chakraborty, S., Eds.; Springer: Cham, Switzerland, 2018; pp. 31–48. [Google Scholar]
- Chakraborty, A. Shirakami Mountains: Old-growth forests of Siebold’s Beech supporting biodiversity in a dynamic landscape. In Natural Heritage of Japan: Geological, Geomorphological, and Ecological Aspects; Chakraborty, A., Mokudai, K., Cooper, M., Watanabe, M., Chakraborty, S., Eds.; Springer: Cham, Switzerland, 2018; pp. 49–60. [Google Scholar]
- Okano, T.; Matsuda, H. Biocultural diversity of Yakushima Island: Mountains, beaches, and sea. J. Island Mar. Cul. 2013, 2, 69–77. [Google Scholar] [CrossRef]
- Hayward, P.; Kuwahara, S. Divergent trajectories: Environment, heritage and tourism in Tanegashima, Mageshima and Yakushima. J. Island Mar. Cul. 2013, 2, 29–38. [Google Scholar] [CrossRef]
- Tawara, K. Hokkaido: Midori No Kankyo Shi (Hokkaido: An Environmental History of Its Vegetation); Hokkaido University Press: Sapporo, Japan, 2008. (In Japanese) [Google Scholar]
- Enomoto, Y. Kaitaku Shi to Hokkaido (Hokkaido and Its Cultivation); Hokkaido Publication Planning Center: Sapporo, Japan, 2009. (In Japanese) [Google Scholar]
- Ono, Y. Kawa to Tsukiau (Living with Rivers); Iwanami: Tokyo, Japan, 1997. (In Japanese) [Google Scholar]
- Ono, Y. Shizen No Messeji Wo Kiku: Shizukana Daichikara No Sengen (Listening to Nature’s Message: An Agenda from a Tranquil Land); Hokkaido Shimbun Press: Sapporo, Japan, 2007. (In Japanese) [Google Scholar]
- Yagi, K. Kita No Shizen Wo Mamoru: Shiretoko, Chitosegawa, Soshite Horonobe (Conserving Northern Nature: Shiretoko, Chitose River and Horonobe Town); Hokkaido University Press: Sapporo, Japan, 1995. (In Japanese) [Google Scholar]
- Kikuchi, K. Mo Hitotsu No Shiretoko: Sengo Kaitaku Monogatari (Another Shiretoko: Tales of Land Conversion in the Postwar Period); Hokkaido Shimbun Press: Sapporo, Japan, 2005. (In Japanese) [Google Scholar]
- Shoyama, K.; Braimoh, A. Analyzing about sixty years of land-cover change and associated fragmentation in Shiretoko Peninsula, Northern Japan. Landsc. Urban Plan. 2011, 101, 22–29. [Google Scholar] [CrossRef]
- UNESCO. State of Conservation: Shiretoko (Japan); UNESCO: Paris, France, 2017; Available online: https://whc.unesco.org/en/soc/3568 (accessed on 12 February 2019).
- Naughton-Treves, L.; Holland, M.B.; Brandon, K. The role of protected areas in conserving biodiversity and sustaining local livelihoods. Annu. Rev. Environ. Resour. 2005, 30, 219–252. [Google Scholar] [CrossRef]
- Hansen, A.J.; DeFries, R. Ecological mechanisms linking protected areas to surrounding lands. Ecol. Appl. 2007, 17, 974–988. [Google Scholar] [CrossRef]
- Bailey, K.M.; McKleery, R.A.; Bindford, M.W.; Zweig, C. Land-cover change within and around protected areas in a biodiversity hotspot. J. Land Use Sci. 2016, 11, 154–176. [Google Scholar] [CrossRef]
- Kormos, C.F.; Bertzky, B.; Jaeger, T.; Shi, Y.; Badman, T.; Hilty, J.A.; Mackey, B.G.; Mittermeier, R.A.; Locke, H.; Osipova, E.; et al. A wilderness approach under the World Heritage Convention. Conserv. Lett. 2016, 9, 228–235. [Google Scholar] [CrossRef]
- Timko, J.A.; Innes, J.L. Evaluating ecological integrity in national parks: Case studies from Canada and South Africa. Biol. Conserv. 2009, 142, 676–688. [Google Scholar] [CrossRef]
- Woodley, S. Ecological Integrity and Canada’s National Parks. George Wright Forum 2010, 27, 151–160. [Google Scholar]
- Pimentel, D.; Westra, L.; Noss, R.F. (Eds.) Ecological Integrity: Integrating Environment, Conservation, and Health; Island Press: Washington, DC, USA, 2000. [Google Scholar]
- Gunderson, L.H.; Allen, C.R.; Holling, C.S. (Eds.) Foundations of Ecological Resilience; Island Press: Washington, DC, USA, 2009. [Google Scholar]
- Likens, G.E.; Bormann, F.H.; Pierce, R.S.; Reiners, W.A. Recovery of a deforested ecosystem. Science 1978, 199, 492–496. [Google Scholar] [CrossRef]
- Odum, E.P. Trends expected in stressed ecosystems. BioScience 1985, 35, 419–422. [Google Scholar] [CrossRef]
- Woodley, S. Ecological Integrity: A framework for ecosystem-based management. In Beyond Naturalness: Rethinking Park and Wilderness Stewardship in an Era of Rapid Change; Cole, D.N., Yung, L., Eds.; Island Press: Washington, DC, USA, 2010; pp. 106–124. [Google Scholar]
- Kruess, A.; Tscharntke, T. Habitat fragmentation, species loss, and biological control. Science 1994, 264, 1581–1584. [Google Scholar] [CrossRef]
- Debinski, D.M.; Holt, R.D. A survey and overview of habitat fragmentation experiments. Conserv. Biol. 2000, 14, 342–355. [Google Scholar] [CrossRef]
- Didham, R.K. Ecological consequences of habitat fragmentation. Wiley Online Library. 2010. Available online: https://onlinelibrary.wiley.com/doi/10.1002/9780470015902.a0021904 (accessed on 2 February 2019).
- Lewis, S.L.; Edwards, D.P.; Galbraith, D. Increasing human dominance of tropical forests. Science 2015, 349, 827–832. [Google Scholar] [CrossRef]
- Adhikari, A.; Hansen, A.J. Land use change and habitat fragmentation of wildland ecosystems of the North Central United States. Landsc. Urban. Plan. 2018, 177, 196–216. [Google Scholar] [CrossRef]
- Saravia, L.A.; Doyle, S.R.; Bond-Lamberty, B. Power laws and critical fragmentation in global forests. Nat. Sci. Rep. 2018, 8, 1–12. [Google Scholar] [CrossRef]
- Anderson, E.P.; Jenkins, C.N.; Hailpern, S.; Maldonaldo-Ocampo, J.A.; Carvajal-Vallejos, F.M.; Encalada, A.C.; Rivadeneira, J.F.; Hidalgo, M.; Canas, C.M.; Ortega, H.; et al. Fragmentation of Andes-to-Amazon connectivity by hydropower dams. Sci. Adv. 2018, 4, 1–7. [Google Scholar] [CrossRef]
- Galla, A. World Heritage: Benefits beyond Borders; Cambridge University Press: Cambridge, UK, 2012. [Google Scholar]
- Ishikawa, Y. Shokubutsu no genjo to kadai (Current condition ad challenges for vegetation). In Shiretoko No Shizen Hogo (Nature Conservation in Shiretoko); Shiretoko Museum, Ed.; Hokkaido Shimbun Press: Sapporo, Japan, 2010. (In Japanese) [Google Scholar]
- Takenaka, T. Ecology and conservation of Blakiston’s Fish Owl in Japan. In Biodiversity Conservation Using Umbrella Species: Blakiston’s Fish Owl and the Red-Crowned Crane; Nakamura, F., Ed.; Springer: Singapore, 2018; pp. 19–46. [Google Scholar]
- Takenaka, T. Shimafukuro no genjo to kadai (Current conditions and challenges for the Blakiston’s Fish Owl). In Shiretoko No Shizen Hogo (Nature Conservation in Shiretoko); Shiretoko Museum, Ed.; Hokkaido Shimbun Press: Sapporo, Japan, 2010. (In Japanese) [Google Scholar]
- Gerring, J. Case Study Research: Principles and Practices; Cambridge University Press: Cambridge, UK, 2007. [Google Scholar]
- Taylor, L. Case study methodology. In Key Methods in Geography; Clifford, N., Cope, M., Gillespie, T.W., French, S., Eds.; Sage: London, UK, 2016; pp. 581–595. [Google Scholar]
- Bazeley, P. Qualitative Data Analysis Practical Strategies; Sage: London, UK, 2013. [Google Scholar]
- Patton, M.Q. Qualitative Research and Evaluation Methods, 3rd ed.; Sage: Thousand Oaks, CA, USA, 2002. [Google Scholar]
- Slaght, J.C.; Takenaka, T.; Surmach, S.G.; Fujimaki, Y.; Utekhina, I.G.; Potapov, E.R. Global distribution and population estimates of Blakiston’s Fish Owl. In Biodiversity Conservation Using Umbrella Species: Blakiston’s Fish Owl and the Red-Crowned Crane; Nakamura, F., Ed.; Springer: Singapore, 2018; pp. 9–18. [Google Scholar]
- Kitano, F.; Nakano, S.; Maekawa, K.; Ono, Y. Effect of stream temperatures on longitudinal distribution of fluvial Dolly Varden and potential habitat loss due to global warming. Wildl. Conserv. Jpn. 1995, 1, 1–11, (In Japanese with English abstract). [Google Scholar]
- Allan, J.R.; Kormos, C.F.; Jaeger, T.; Venter, O.; Bertzky, B.; Shi, Y.; Mackey, B.; van Merm, R.; Osipova, E.; Watson, J.E.M. Gaps and opportunities for the World Heritage Convention to contribute to global wilderness conservation. Conserv. Biol. 2017, 32, 116–126. [Google Scholar] [CrossRef]
- Asaad, I.; Lundquist, C.; Erdmann, M.; Costello, M. Ecological criteria to identify areas for biodiversity conservation. Biol. Conserv. 2017, 213, 309–316. [Google Scholar] [CrossRef]
- de Juan, S.; Hewitt, J.; Subida, M.D.; Thrush, S. Translating Ecological Integrity terms into operational language to inform societies. J. Environ. Manag. 2018, 228, 319–327. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).