Status of Water Quality for Human Consumption in High-Andean Rural Communities: Discrepancies between Techniques for Identifying Trace Metals
Abstract
1. Introduction
2. Materials and Methods
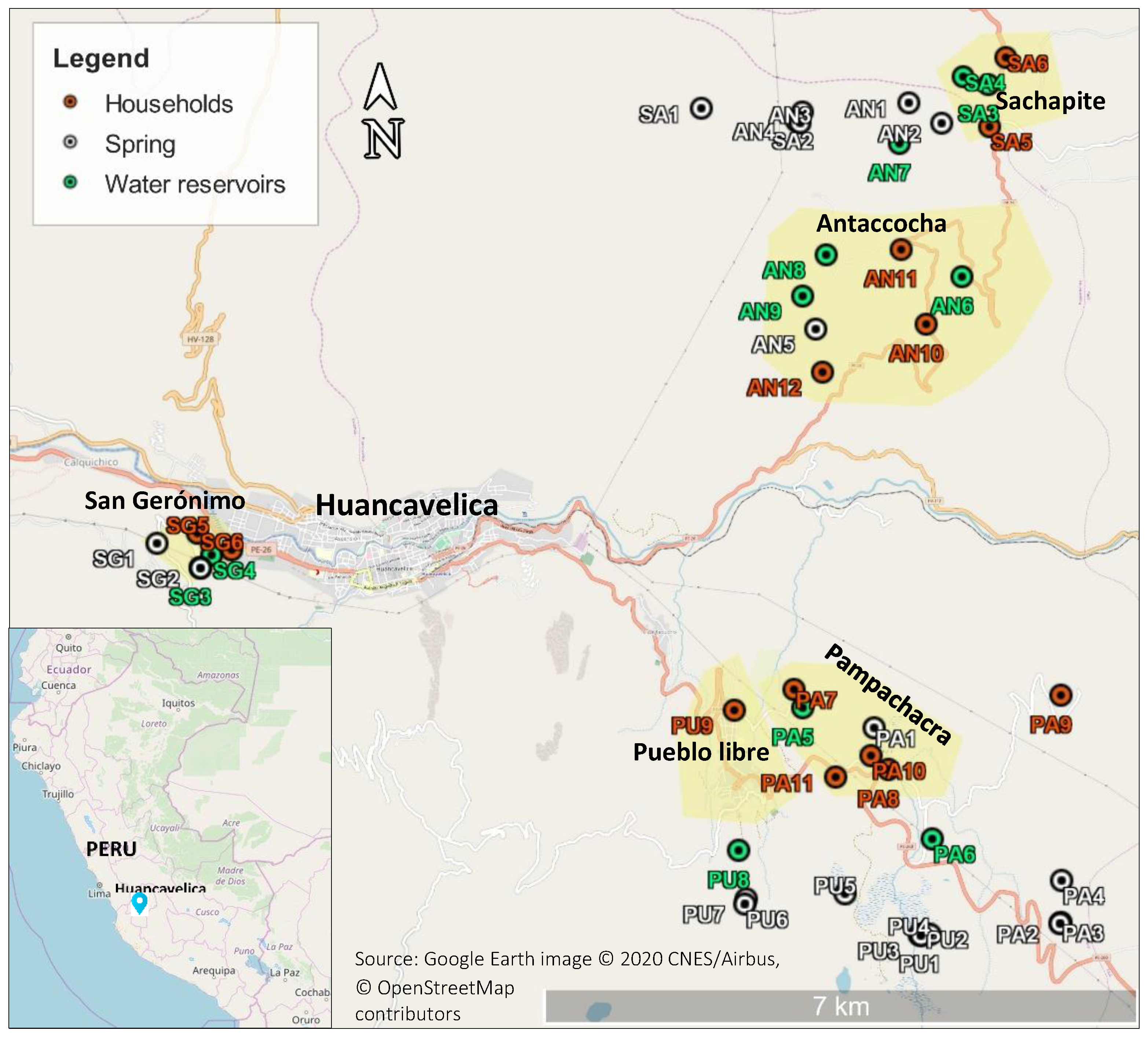
2.1. Description of the Area under Study
2.2. Sampling Points
2.3. Water Analysis Procedures
2.3.1. Determination of Physical–Chemical Parameters
2.3.2. Determination of Total and Thermotolerant Coliforms
2.3.3. Determination of Parasites and Free-Living Organisms
2.3.4. Determination of Trace Metals
3. Results
3.1. Water Quality in High-Andean Rural Communities
3.1.1. Pueblo Libre
3.1.2. San Gerónimo
3.1.3. Sachapite
3.1.4. Pampachacra
3.1.5. Antacoccha
3.2. Comparison of Two Different Techniques to Identify Trace Levels of Metals in Water
4. Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A. Sampling Points
| Community | Description | Label | Elevation (m.a.s.l.) |
|---|---|---|---|
| Pueblo libre | Spring 1 | PU1 | 4188 |
| Pueblo libre | Spring 2 | PU2 | 4181 |
| Pueblo libre | Spring 3 | PU3 | 4184 |
| Pueblo libre | Spring 4 | PU4 | 4179 |
| Pueblo libre | Spring 6 | PU5 | 4175 |
| Pueblo libre | Spring 8 | PU6 | 4170 |
| Pueblo libre | Spring 9 | PU7 | 4154 |
| Pueblo libre | Water reservoir | PU8 | 4072 |
| Pueblo libre | Household | PU9 | 3899 |
| San Gerónimo | Spring Puquiocucho 1 | SG1 | 3893 |
| San Gerónimo | Spring Putaccapuquio | SG2 | 3822 |
| San Gerónimo | Puquiocucho water reservoir | SG3 | 3821 |
| San Gerónimo | Putaccapuquio water reservoir | SG4 | 3757 |
| San Gerónimo | Puquiocucho household | SG5 | 3771 |
| San Gerónimo | Putaccapuquio household | SG6 | 3720 |
| Sachapite | Spring Huariccacca | SA1 | 4328 |
| Sachapite | Spring Picchapuquio | SA2 | 4273 |
| Sachapite | Huariccacca water reservoir | SA3 | 4191 |
| Sachapite | Picchapuquio water reservoir | SA4 | 4206 |
| Sachapite | Huariccacca household | SA5 | 4121 |
| Sachapite | Picchapuquio household | SA6 | 4162 |
| Pampachacra | Spring Barrio Villa Libertad | PA1 | 3957 |
| Pampachacra | Spring Barrio Imperial (Asto1) | PA2 | 4258 |
| Pampachacra | Spring Barrio Imperial (Asto2) | PA3 | 4279 |
| Pampachacra | Spring 1 Barrio Condorsenca | PA4 | 4296 |
| Pampachacra | Barrio Villa Libertad water reservoir | PA5 | 3925 |
| Pampachacra | Barrio Imperial water reservoir | PA6 | 4101 |
| Pampachacra | Barrio Villa Libertad household | PA7 | 3888 |
| Pampachacra | Barrio Imperial household | PA8 | 4037 |
| Pampachacra | Barrio Condorsenca household | PA9 | 4055 |
| Pampachacra | Barrio Centro household | PA10 | 4000 |
| Pampachacra | Barrio Miraflores household | PA11 | 4027 |
| Antaccocha | Spring sector 1 | AN1 | 4176 |
| Antaccocha | Spring sector 2 | AN2 | 4176 |
| Antaccocha | Spring sector 3 and 5 | AN3 | 4246 |
| Antaccocha | Spring sector 4 | AN4 | 4259 |
| Antaccocha | Spring Mulaccarana | AN5 | 3893 |
| Antaccocha | Sector 1 water reservoir | AN6 | 3979 |
| Antaccocha | Sector 2 water reservoir | AN7 | 4108 |
| Antaccocha | Sector 3 and 5 water reservoir | AN8 | 4005 |
| Antaccocha | Sector 4 water reservoir | AN9 | 3991 |
| Antaccocha | Sector 1 household | AN10 | 3839 |
| Antaccocha | Sector 2 household | AN11 | 3911 |
| Antaccocha | Sector 4 household | AN12 | 3780 |
References
- Prüss-Ustün, A.; Bartram, J.; Clasen, T.; Colford, J.M.; Cumming, O.; Curtis, V.; Bonjour, S.; Dangour, A.D.; De France, J.; Fewtrell, L.; et al. Burden of disease from inadequate water, sanitation and hygiene in low- and middle-income settings: A retrospective analysis of data from 145 countries. Trop. Med. Int. Health 2014, 19, 894–905. [Google Scholar] [CrossRef]
- Naghavi, M.; Abajobir, A.A.; Abbafati, C.; Abbas, K.M.; Abd-Allah, F.; Abera, S.F.; Aboyans, V.; Adetokunboh, O.; Ärnlöv, J.; Afshin, A.; et al. Global, regional, and national age-sex specifc mortality for 264 causes of death, 1980-2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1151–1210. [Google Scholar] [CrossRef]
- Troeger, C.; Blacker, B.F.; Khalil, I.A.; Rao, P.C.; Cao, S.; Zimsen, S.R.; Albertson, S.B.; Stanaway, J.D.; Deshpande, A.; Abebe, Z.; et al. Estimates of the global, regional, and national morbidity, mortality, and aetiologies of diarrhoea in 195 countries: A systematic analysis for the Global Burden of Disease Study 2016. Lancet Infect. Dis. 2018, 18, 1211–1228. [Google Scholar] [CrossRef]
- Bartram, J.; Brocklehurst, C.; Fisher, M.; Luyendijk, R.; Hossain, R.; Wardlaw, T.; Gordon, B. Global Monitoring of Water Supply and Sanitation: History, Methods and Future Challenges. Int. J. Environ. Res. Public Health 2014, 11, 8137–8165. [Google Scholar] [CrossRef]
- UNICEF/WHO. Progress on Household Drinking Water, Sanitation and Hygiene 2000-2017: Special Focus on Inequalities; United Nations Children’s Fund (UNICEF) and World Health Organization: New York, NY, USA, 2019; ISBN 978-92-415-1623-5. [Google Scholar]
- WHO/UNICEF Joint Monitoring Programme for Water Supply, Sanitation, and Hygiene: Estimates on the Use of Water, Sanitation and Hygiene in Peru. Available online: https://washdata.org/data (accessed on 12 November 2019).
- Bain, R.E.S.; Wright, J.A.; Christenson, E.; Bartram, J.K. Rural:urban inequalities in post 2015 targets and indicators for drinking-water. Sci. Total Environ. 2014, 490, 509–513. [Google Scholar] [CrossRef] [PubMed]
- Chitonge, H.; Mokoena, A.; Kongo, M. Water and Sanitation Inequality in Africa: Challenges for SDG 6. In Africa and the Sustainable Development Goals; Ramutsindela, M., Mickler, D., Eds.; Springer: Cham, Switzerland, 2020; pp. 207–218. [Google Scholar]
- D.S. N° 004-2017-MINAM Aprueban Estándares de Calidad Ambiental (ECA) Para Agua y Establecen Disposiciones Complementarias. Available online: https://sinia.minam.gob.pe/normas/aprueban-estandares-calidad-ambiental-eca-agua-establecen-disposiciones (accessed on 26 January 2020).
- DIGESA-MINSA Reglamento de la Calidad del Agua Para Consumo Humano: D.S. No 031-2010-SA/Regulation of Water Quality for Human Consumption: DS No 031-2010-SA. Available online: http://bvs.minsa.gob.pe/local/MINSA/1590.pdf (accessed on 21 August 2019).
- George, C.M.; Sima, L.; Arias, M.H.J.; Mihalic, J.; Cabrera, L.Z.; Danz, D.; Checkley, W.; Gilman, R.H. Arsenic exposure in drinking water: An unrecognized health threat in Peru. Bull. World Health Organ. 2014, 92, 565–572. [Google Scholar] [CrossRef]
- Yacoub, C.; Blazquez, N.; Pérez-Foguet, A.; Miralles, N. Spatial and temporal trace metal distribution of a Peruvian basin: Recognizing trace metal sources and assessing the potential risk. Environ. Monit. Assess. 2013, 185, 7961–7978. [Google Scholar] [CrossRef]
- Yacoub, C.; Pérez-Foguet, A.; Valderrama, C.; Miralles, N. Impacts on effluent contaminants from mine sites: Risk assessment, fate, and distribution of pollution at basin scale. Environ. Sci. Pollut. Res. 2014, 21, 5960–5971. [Google Scholar] [CrossRef]
- Yacoub, C.; Pérez-Foguet, A.; Miralles, N. Trace Metal Content of Sediments Close to Mine Sites in the Andean Region. Sci. World J. 2012, 2012, 732519. [Google Scholar] [CrossRef]
- Boehnke, K.F.; Brewster, R.K.; Sánchez, B.N.; Valdivieso, M.; Bussalleu, A.; Guevara, M.; Saenz, C.G.; Alva, S.O.; Gil, E.; Xi, C. An assessment of drinking water contamination with Helicobacter pylori in Lima, Peru. Helicobacter 2018, 23, e12462. [Google Scholar] [CrossRef]
- Quick, R.E.; Montano, S.M.; Mock, C.N.; Tilley, D.H.; Carrasco, A.J.; Cabrera, R.M.; Heitzinger, K.; Rocha, C.A.; Hawes, S.E. “Improved” But Not Necessarily Safe: An Assessment of Fecal Contamination of Household Drinking Water in Rural Peru. Am. J. Trop. Med. Hyg. 2015, 93, 501–508. [Google Scholar] [CrossRef]
- Gil, A.I.; Lanata, C.F.; Hartinger, S.M.; Mäusezahl, D.; Padilla, B.; Ochoa, T.J.; Lozada, M.; Pineda, I.; Verastegui, H. Fecal contamination of food, water, hands, and kitchen utensils at the household level in rural areas of Peru. J. Environ. Health 2014, 76, 102–106. [Google Scholar] [PubMed]
- Faldetta, K.F.; Reighard, D.A.; Dickinson, K.L.; Wang, C.Q.; George, D.R.; Rodriguez Benavides, L.; Strosnider, W.H.J. Assessing domestic water quality in Belén municipality, Iquitos, Peru. J. Water, Sanit. Hyg. Dev. 2014, 4, 391–399. [Google Scholar] [CrossRef]
- Ministerio de Vivienda Construcción y Saneamiento (MVCS). DATASS: Modelo Para la Toma de Decisiones en Saneamiento. Sistema de Diagnóstico sobre Abastecimiento de Agua y Saneamiento en el Ámbito Rural; Biblioteca Nacional del Perú N° 2018-15064: Lima, Peru, 2018. [Google Scholar]
- Miranda, M.; Aramburú, A.; Junco, J.; Campos, M. State of the quality of drinking water in households in children under five years in Peru, 2007-2010. Rev. Peru. Med. Exp. Salud Publica 2010, 27, 506–511. [Google Scholar] [CrossRef]
- Villena Chávez, J.A. Calidad del agua y desarrollo sostenible. Rev. Peru. Med. Exp. Salud Publica 2018, 35, 304. [Google Scholar] [CrossRef]
- Instituto Nacional de Estadístjica e Informática Perú: Formas de Acceso al Agua y Saneamiento Básico. Available online: https://www.inei.gob.pe/media/MenuRecursivo/boletines/boletin_agua_nov2019.pdf (accessed on 10 March 2020).
- Jajda, H.M.; Patel, K.G.N.G.N.; Patel, S.R.; Solanki, V.H.; Patel, K.G.N.G.N.; Singh, S. Comparative efficacy of two standard methods for determination of iron and zinc in fruits, pulses and cereals. J. Food Sci. Technol. 2013, 52, 1096–1102. [Google Scholar] [CrossRef]
- Jarošová, M.; Milde, D.; Kuba, M. Elemental analysis of coffee: A comparison of ICP-MS and AAS methods. Czech J. Food Sci. 2014, 32, 354–359. [Google Scholar] [CrossRef]
- Tyler, G.; Yvon, J. ICP-OES, ICP-MS and AAS Techniques Compared. ICP Opt. Spectrosc. Tech. Note 1995, 1–11. [Google Scholar]
- Instituto Nacional de Estadística e Informática Perú: Perfil Sociodemográfico. Informe Nacional. Censos Nacionales 2017: XII de Población, VII de Vivienda y III de Comunidades Indígenas. Available online: https://www.inei.gob.pe/media/MenuRecursivo/publicaciones_digitales/Est/Lib1539/libro.pdf (accessed on 9 February 2020).
- Instituto Nacional de Estadística e Informática Perú: Encuesta Demográfica y de Salud Familiar 2018—Nacional y Departamental. Available online: https://www.inei.gob.pe/media/MenuRecursivo/publicaciones_digitales/Est/Lib1656/index1.html (accessed on 13 January 2020).
- Accinelli, R.A.; Leon-Abarca, J.A. Age and altitude of residence determine anemia prevalence in Peruvian 6 to 35 months old children. PLoS ONE 2020, 15, e0226846. [Google Scholar] [CrossRef]
- Huicho, L.; Tavera, M.; Huayanay-Espinoza, C.A.; Béjar-Díaz, M.; Rivera-Ch, M.; Tam, Y.; Walker, N.; Black, R.E. Drivers of the progress achieved by Peru in reducing childhood diarrhoea mortality: A country case study. J. Glob. Health 2019, 9. [Google Scholar] [CrossRef]
- Kothari, M.T.; Coile, A.; Huestis, A.; Pullum, T.; Garrett, D.; Engmann, C. Exploring associations between water, sanitation, and anemia through 47 nationally representative demographic and health surveys. Ann. N. Y. Acad. Sci. 2019, 1450, 249. [Google Scholar] [CrossRef] [PubMed]
- Larsen, D.A.; Grisham, T.; Slawsky, E.; Narine, L. An individual-level meta-analysis assessing the impact of community-level sanitation access on child stunting, anemia, and diarrhea: Evidence from DHS and MICS surveys. PLoS Negl. Trop. Dis. 2017, 11. [Google Scholar] [CrossRef] [PubMed]
- Cairncross, S.; Hunt, C.; Boisson, S.; Bostoen, K.; Curtis, V.; Fung, I.C.C.H.; Schmidt, W.-P. Water, sanitation and hygiene for the prevention of diarrhoea. Int. J. Epidemiol. 2010, 39, i193–i205. [Google Scholar] [CrossRef] [PubMed]
- WHO. Guidelines for Drinking-Water Quality: Fourth Edition Incorporating the First Addendum, 4th ed.; WHO: Geneva, Switzerland, 2017; ISBN 978-92-4-154995-0. [Google Scholar]
- Reed, B.; Shaw, R.; Chatterton, K. Technical Notes on Drinking-Water, Sanitation and Hygiene in Emergencies; World Health Organization (WHO); Water, Engineering and Development Centre (WEDC): Loughborough, UK, 2013; pp. 45–48. [Google Scholar]
- Edgell, K.W.; Longbottom, J.E.; Pfaff, J.D. Determination of inorganic anions in water by ion chromatography: A collaborative study. J. AOAC Int. 1994, 77, 1253–1263. [Google Scholar] [CrossRef] [PubMed]
- Oblinger, J.L.; Koburguer, J.A. Understanding and Teaching the Most Probable Number Technique1. J. Milk Food Technol. 1975, 38, 540–545. [Google Scholar] [CrossRef]
- Ayres, R.M.; Mara, D.D. Analysis of Wastewater for Use in Agriculture: A Laboratory Manual of Parasitological and Bacteriological Techniques; World Health Organization: Geneva, Switzerland, 1996; ISBN 9241544848. [Google Scholar]
- Federation, W.E.; American Public Health Association. Standard Methods for the Examination of Water and Wastewater, 21st ed.; American Public Health Association: Washington, DC, USA, 2005; ISBN 0875530478 9780875530475. [Google Scholar]
- ISO—ISO 17294-2:2016—Water Quality—Application of Inductively Coupled Plasma Mass Spectrometry (ICP-MS)—Part 2: Determination of Selected Elements Including Uranium Isotopes. Available online: https://www.iso.org/standard/62962.html (accessed on 10 March 2020).
- Rice, E.W.; Baird, R.B.; Eaton, A.D.; Clesceri, L.S. 3111 Metals by flame atomic absorption spectometry. In Standard Methods for the Examination of Water and Wastewater; American Public Health Association: Washington, DC, USA; American Waterworks Association: Denver, CO, USA; Water Environment Federation: Alexandria, VA, USA, 2012; p. 10. [Google Scholar]
- Piasecki, W.; Goodwin, A.E.; Eiras, J.C.; Nowak, B.F. Importance of copepoda in freshwater aquaculture. Zool. Stud. 2004, 43, 193–205. [Google Scholar]
- Meshitsuka, S.; Aremu, D.A.; Nose, T. A Risk of Alzheimer’s Disease and Aluminum in Drinking Water. Psychogeriatrics 2002, 2, 263–268. [Google Scholar] [CrossRef]
- Wood, D.J.; Cooper, C.; Stevens, J.; Edwardson, J. Bone mass and dementia in hip fracture patients from areas with different aluminium concentrations in water supplies. Age Ageing 1988, 17, 415–418. [Google Scholar] [CrossRef]
- Gauthier, E.; Fortier, I.; Courchesne, F.; Pepin, P.; Mortimer, J.; Gauvreau, D. Aluminum Forms in Drinking Water and Risk of Alzheimer’s Disease. Environ. Res. 2000, 84, 234–246. [Google Scholar] [CrossRef]
- McLachlan, D.R.C.; Bergeron, C.; Smith, J.E.; Boomer, D.; Rifat, S.L. Risk for neuropathologically confirmed Alzheimer’s disease and residual aluminum in municipal drinking water employing weighted residential histories. Neurology 1996, 46, 401–405. [Google Scholar] [CrossRef]
- Rondeau, V.; Commenges, D.; Jacqmin-Gadda, H.; Dartigues, J.F. Relation between aluminum concentrations in drinking water and Alzheimer’s disease: An 8-year follow-up study. Am. J. Epidemiol. 2000, 152, 59–66. [Google Scholar] [CrossRef] [PubMed]
- Suay Llopis, L.; Ballester Díez, F. Revisión de los estudios sobre exposición al aluminio y enfermedad de Alzheimer. Rev. Esp. Salud Publica 2002, 76, 645–658. [Google Scholar] [CrossRef] [PubMed]
- WHO. International Programme on Chemical Safety Aluminnium. Environ. Heal. Criteria 1997, 194. [Google Scholar]
- Nieboer, E.; Gibson, B.L.; Oxman, A.D.; Kramer, J.R. Health effects of aluminum: A critical review with emphasis on aluminum in drinking water. Environ. Rev. 1995, 3, 29–81. [Google Scholar] [CrossRef]
- Gonzales, E.; Huamán-Espino, L.; Gutiérrez Villafuerte, C.; Aparco Balboa, J.; Pillaca, J. Characterization of anemia in children under five years of age from urban areas of Huancavelica and Ucayali, Peru. Rev. Peru. Med. Exp. Salud Publica 2015, 32, 431–439. [Google Scholar] [CrossRef]
- Hunter, P.R.; MacDonald, A.M.; Carter, R.C. Water Supply and Health. PLoS Med. 2010, 7, e1000361. [Google Scholar] [CrossRef]
- Kelly, E.R.; Cronk, R.; Kumpel, E.; Howard, G.; Bartram, J. How we assess water safety: A critical review of sanitary inspection and water quality analysis. Sci. Total Environ. 2020, 718, 137237. [Google Scholar] [CrossRef]
- Andres, L.; Boateng, K.; Borja-Vega, C.; Thomas, E.; Andres, L.; Boateng, K.; Borja-Vega, C.; Thomas, E. A Review of In-Situ and Remote Sensing Technologies to Monitor Water and Sanitation Interventions. Water 2018, 10, 756. [Google Scholar] [CrossRef]
- Grossi, M.; Lazzarini, R.; Lanzoni, M.; Pompei, A.; Matteuzzi, D.; Ricco, B. A portable sensor with disposable electrodes for water bacterial quality assessment. IEEE Sens. J. 2013, 13, 1775–1782. [Google Scholar] [CrossRef]
| Community | Springs | Water Reservoirs | Households |
|---|---|---|---|
| Pueblo Libre | 7 | 1 | 1 |
| San Gerónimo | 2 | 2 | 2 |
| Sachapite | 2 | 2 | 2 |
| Antaccocha | 5 | 4 | 3 |
| Pampachacra | 4 | 2 | 5 |
| Parameter | Analysis Method | Sampling Points |
|---|---|---|
| Physicochemical Parameters | ||
| Turbidity | HI93703 Portable Turbidity Meter (Hanna Instruments, USA) (Environmental Health Unit of the Huancavelica Health Network). Range of detection: 0–1000 NTU | Springs, water reservoirs, and households |
| pH, conductivity | Portable meter HQ40d (Hach, USA) (Environmental Health Unit of the Huancavelica Health Network) Range of detection: pH (0–14), conductivity (0.01 µS/cm–200.0 mS/cm) | |
| Free residual chlorine (FRC) | HI96734 Portable photometer for free and total chlorine (Hanna Instruments, USA) (Environmental Health Unit of the Huancavelica Health Network). Range of detection: 0–10 mg/L | |
| Color | Spectrophotometry (Environmental Health Unit of the Huancavelica Health Network) | |
| Total hardness | SMEWW-APHA-AWWA-WEF Part 2340 C, 23rd Ed.2017. Hardness. EDTA Titrimetric Method. Laboratory CERPER S.A. (Spanish acronym of the laboratory Certifications of Peru S.A.), Lima. (Environmental Health Unit of the Huancavelica Health Network.) | Springs |
| Chlorides | EPA Method 300.0. 1993 Determination of inorganic anions by ion chromatography [35]. Laboratory CERPER S.A., Lima. (Environmental Health Unit of the Huancavelica Health Network.) | |
| Nitrates | ||
| Nitrites | ||
| Sulfates | ||
| Microbiological and Parasitological Parameters | ||
| Total and thermotolerant (fecal) coliforms | Most probable number (MPN) method [36] (Laboratory of Microbiology and Parasitology—UNH) | Springs, water reservoirs and households |
| Parasites and free-living organisms | Protocol to detect protozoa and parasitic helminths in natural water using a Sedgewick Rafter Counting Chambers [37,38] (Environmental Health Unit of the Huancavelica Health Network) | |
| Inorganic Parameters | ||
| Molybdenum (Mo), manganese (Mn), cadmium (Cd), arsenic (As), copper (Cu), chromium (Cr), lead (Pb), zinc (Zn), antimony (Sb), aluminum (Al). | ISO 17294-2. 2016. Water quality—application of inductively coupled plasma mass spectrometry (ICP-MS) [39] Laboratory CERPER S.A., Lima. The limits of detection (mg/L) of the ICP-MS method were: Mo: 0.0002; Mn: 0.00025; Cd: 0.00005; As: 0.0005; Cu: 0.0003; Cr: 0.0005; Zn: 0.0005; Al: 0.0025; Pb: 0.0002; and Sb: 0.0002. (Environmental Health Unit of the Huancavelica Health Network) | Springs |
| Atomic absorption spectroscopy (AAS)—3111 metals by flame atomic absorption spectrometry [40]. The limits of detection (mg/L) of the AAS method were: Mo: 0.03; Mn: 0.006; Cd: 0.0039; As: 0.12; Cu: 0.0099; Cr: 0.015; Zn: 0.003; Al: 0.09; Pb: 0.021; and Sb: 0.069.(Chemistry Laboratory—UNH) | ||
| Community | Springs | Water Reservoirs | Households | |||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P | N | T | NC | %NC | C | P | N | T | NC | %NC | C | P | N | T | NC | %NC | C | |
| Physicochemical parameters | ||||||||||||||||||
| Pueblo Libre | 7 | 10 | 70 | 0 | 0.0% | 70 | 1 | 5 | 5 | 1 | 20.0% | 4 | 1 | 5 | 5 | 1 | 20.0% | 4 |
| San Gerónimo | 2 | 10 | 20 | 0 | 0.0% | 20 | 2 | 5 | 10 | 1 | 10.0% | 9 | 2 | 5 | 10 | 2 | 20.0% | 8 |
| Sachapite | 2 | 10 | 20 | 0 | 0.0% | 20 | 2 | 5 | 10 | 2 | 20.0% | 8 | 2 | 5 | 10 | 2 | 20.0% | 8 |
| Antaccocha | 5 | 10 | 50 | 2 | 4.0% | 48 | 4 | 5 | 20 | 3 | 15.0% | 17 | 3 | 5 | 15 | 4 | 26.7% | 11 |
| Pampachacra | 4 | 10 | 40 | 1 | 2.5% | 39 | 2 | 5 | 10 | 2 | 20.0% | 8 | 5 | 5 | 25 | 6 | 24.0% | 19 |
| Total | 200 | 3 | 1.5% | 197 | 55 | 9 | 16.4% | 46 | 65 | 15 | 23.1% | 50 | ||||||
| Microbiological and parasitological parameters | ||||||||||||||||||
| Pueblo Libre | 7 | 4 | 28 | 4 | 14.3% | 24 | 1 | 4 | 4 | 2 | 50.0% | 2 | 1 | 4 | 4 | 1 | 25.0% | 3 |
| San Gerónimo | 2 | 4 | 8 | 3 | 37.5% | 5 | 2 | 4 | 8 | 2 | 25.0% | 6 | 2 | 4 | 8 | 2 | 25.0% | 6 |
| Sachapite | 2 | 4 | 8 | 2 | 25.0% | 6 | 2 | 4 | 8 | 2 | 25.0% | 6 | 2 | 4 | 8 | 0 | 0.0% | 8 |
| Antaccocha | 5 | - | 16 | 0 | 0.0% | 16 | 4 | 2 | 8 | 0 | 0.0% | 8 | 3 | - | 5 | 2 | 40.0% | 3 |
| Pampachacra | 4 | 4 | 16 | 2 | 12.5% | 14 | 2 | 2 | 4 | 0 | 0.0% | 4 | 5 | 2 | 10 | 0 | 0.0% | 10 |
| Total | 76 | 11 | 14.5% | 65 | 32 | 6 | 18.8% | 26 | 35 | 5 | 14.3% | 30 | ||||||
| Springs | Mo (mg/L) | Mn (mg/L) | Cd (mg/L) | As (mg/L) | ||||
|---|---|---|---|---|---|---|---|---|
| AAS | ICP-MS | AAS | ICP-MS | AAS | ICP-MS | AAS | ICP-MS | |
| AN1 | 0.11 | <0.0002 | <0.006 | 0.00092 | <0.0039 | <0.00005 | <0.12 | <0.0005 |
| AN2 | 0.10 | <0.0002 | <0.006 | 0.00063 | <0.0039 | <0.00005 | <0.12 | <0.0005 |
| AN3 | 0.06 | <0.0002 | <0.006 | 0.00431 | <0.0039 | <0.00005 | <0.12 | <0.0005 |
| AN4 | 0.12 | <0.0002 | <0.006 | 0.00578 | <0.0039 | <0.00005 | <0.12 | <0.0005 |
| AN5 | 0.06 | <0.0002 | 0.05 | 0.02029 | <0.0039 | <0.00005 | <0.12 | <0.0005 |
| PU1 | 0.0599 | <0.0002 | <0.006 | 0.00187 | 0.0042 | <0.00005 | <0.12 | <0.0005 |
| PU2 | 0.0661 | <0.0002 | 0.0456 | 0.04446 | 0.0083 | <0.00005 | <0.12 | <0.0005 |
| PU3 | 0.0743 | <0.0002 | <0.006 | 0.0076 | 0.0111 | <0.00005 | <0.12 | <0.0005 |
| PU4 | 0.0728 | <0.0002 | <0.006 | 0.00197 | 0.0109 | <0.00005 | <0.12 | <0.0005 |
| PU5 | 0.0712 | <0.0002 | <0.006 | 0.08141 | 0.0108 | <0.00005 | <0.12 | <0.0005 |
| PU6 | 0.0711 | <0.0002 | <0.006 | 0.00484 | 0.0113 | <0.00005 | <0.12 | <0.0005 |
| PU7 | 0.0726 | <0.0002 | <0.006 | 0.00782 | 0.0108 | <0.00005 | <0.12 | <0.0005 |
| PA1 | <0.03 | <0.0002 | <0.006 | 0.00743 | <0.0039 | <0.00005 | <0.12 | <0.0005 |
| PA2 | <0.03 | <0.0002 | <0.006 | 0.00082 | <0.0039 | <0.00005 | <0.12 | 0.00755 |
| PA3 | <0.03 | <0.0002 | <0.006 | 0.00282 | <0.0039 | <0.00005 | <0.12 | 0.00225 |
| PA4 | <0.03 | <0.0002 | <0.006 | 0.01908 | <0.0039 | <0.00005 | <0.12 | 0.00241 |
| SG1 | <0.03 | <0.0002 | <0.006 | 0.001 | 0.0104 | <0.00005 | <0.12 | <0.0005 |
| SG2 | <0.03 | 0.00146 | <0.006 | <0.00025 | 0.0103 | <0.00005 | <0.12 | 0.00326 |
| SA1 | <0.03 | <0.0002 | <0.006 | 0.00835 | <0.0039 | <0.00005 | <0.12 | <0.0005 |
| SA2 | <0.03 | <0.0002 | <0.006 | <0.00025 | <0.0039 | <0.00005 | <0.12 | <0.0005 |
| LOD | 0.03 | 0.0002 | 0.006 | 0.00025 | 0.0039 | 0.00005 | 0.12 | 0.0005 |
| ECA 1 | 0.07 | 0.4 | 0.003 | 0.01 | ||||
| WHO 2 | - | - | 0.003 | 0.01 | ||||
| Springs | Cu (mg/L) | Cr (mg/L) | Zn (mg/L) | Al (mg/L) | ||||
|---|---|---|---|---|---|---|---|---|
| AAS | ICP-MS | AAS | ICP-MS | AAS | ICP-MS | AAS | ICP-MS | |
| AN1 | <0.0099 | <0.0003 | <0.015 | <0.0005 | 0.00741 | 0.0021 | <0.09 | 0.01629 |
| AN2 | 0.015 | <0.0003 | <0.015 | <0.0005 | 0.00693 | 0.0064 | <0.09 | 0.01829 |
| AN3 | 0.012 | <0.0003 | <0.015 | <0.0005 | 0.00674 | 0.0037 | 0.12275 | 0.187 |
| AN4 | 0.016 | <0.0003 | <0.015 | <0.0005 | 0.00596 | 0.004 | 0.21218 | 0.2662 |
| AN5 | <0.0099 | 0.00231 | <0.015 | 0.00306 | 0.00863 | 0.0198 | 3.47493 | 0.6782 |
| PU1 | 0.0156 | <0.0003 | <0.015 | <0.0005 | <0.003 | 0.003 | <0.09 | 0.00843 |
| PU2 | 0.0216 | <0.0003 | <0.015 | <0.0005 | <0.003 | 0.0043 | <0.09 | 0.0079 |
| PU3 | 0.0230 | <0.0003 | <0.015 | <0.0005 | 0.00371 | 0.0086 | <0.09 | 0.02019 |
| PU4 | 0.0257 | <0.0003 | <0.015 | <0.0005 | 0.00401 | 0.0073 | <0.09 | 0.03098 |
| PU5 | 0.0275 | <0.0003 | <0.015 | <0.0005 | 0.00359 | 0.0029 | <0.09 | 0.03898 |
| PU6 | 0.0275 | <0.0003 | <0.015 | <0.0005 | 0.00684 | 0.0055 | <0.09 | 0.08187 |
| PU7 | 0.0309 | <0.0003 | <0.015 | <0.0005 | 0.00545 | 0.0046 | <0.09 | 0.02383 |
| PA1 | <0.0099 | <0.0003 | <0.015 | <0.0005 | 0.01327 | <0.0005 | <0.09 | 0.01492 |
| PA2 | <0.0099 | <0.0003 | <0.015 | <0.0005 | 0.00914 | <0.0005 | <0.09 | 0.02241 |
| PA3 | 0.010 | <0.0003 | <0.015 | <0.0005 | 0.00745 | 0.0024 | <0.09 | 0.0739 |
| PA4 | 0.011 | <0.0003 | <0.015 | <0.0005 | 0.01158 | 0.0129 | <0.09 | 0.4514 |
| SG1 | 0.0365 | <0.0003 | <0.015 | <0.0005 | 0.00392 | 0.0031 | <0.09 | 0.01511 |
| SG2 | 0.0356 | <0.0003 | <0.015 | <0.0005 | 0.00405 | 0.009 | <0.09 | 0.01369 |
| SA1 | <0.0099 | <0.0003 | <0.015 | <0.0005 | 0.01442 | 0.004 | <0.09 | 0.05489 |
| SA2 | 0.013 | <0.0003 | <0.015 | <0.0005 | 0.01331 | 0.0019 | <0.09 | 0.02583 |
| LOD | 0.0099 | 0.0003 | 0.015 | 0.0005 | 0.003 | 0.0005 | 0.09 | 0.0025 |
| ECA 1 | 2 | 0.05 | 3 | 0.9 | ||||
| WHO 2 | 2 | 0.05 | - | 0.1 | ||||
| Parameters | Community | Method | Test | |
|---|---|---|---|---|
| AAS 1 | ICP-MS 2 | (p-Value) | ||
| Cu (mg/L) | AN2 | 0.0154 ± 0.0033 (*) | <0.0003 | 0.0078 ** |
| AN3 | 0.0115 ± 0.0033 (*) | <0.0003 | 0.0139 * | |
| AN4 | 0.0158 ± 0.0033 (*) | <0.0003 | 0.0074 ** | |
| PU1 | 0.0156 ± 0.0043 | <0.0003 | 0.0125 * | |
| PU2 | 0.0216 ± 0.0021 | <0.0003 | 0.0016 *** | |
| PU3 | 0.023 ± 0.0009 | <0.0003 | 0.0002 *** | |
| PU4 | 0.0257 ± 0.0013 | <0.0003 | 0.0004 *** | |
| PU5 | 0.0275 ± 0.0042 | <0.0003 | 0.004 *** | |
| PU6 | 0.0275 ± 0.0011 | <0.0003 | 0.0003 *** | |
| PU7 | 0.0309 ± 0.0007 | <0.0003 | 0.0001 *** | |
| PA3 | 0.0104 ± 0.0033 (*) | <0.0003 | 0.0169 * | |
| PA4 | 0.0113 ± 0.0033 (*) | <0.0003 | 0.0144 * | |
| SG1 | 0.0365 ± 0.0074 | <0.0003 | 0.0068 ** | |
| SG2 | 0.0356 ± 0.0014 | <0.0003 | 0.0003 *** | |
| SA2 | 0.0129 ± 0.0033 (*) | <0.0003 | 0.0111 * | |
| Mo (mg/L) | AN1 | 0.1074 ± 0.0107 (*) | <0.0002 | 0.0017 *** |
| AN2 | 0.0964 ± 0.01 (*) | <0.0002 | 0.0018 *** | |
| AN3 | 0.0632 ± 0.01 (*) | <0.0002 | 0.0041 *** | |
| AN4 | 0.1245 ± 0.0125 (*) | <0.0002 | 0.0017 *** | |
| AN5 | 0.0618 ± 0.01 (*) | <0.0002 | 0.0043 *** | |
| PU1 | 0.0599 ± 0.0057 | <0.0002 | 0.0015 *** | |
| PU2 | 0.0661 ± 0.0023 | <0.0002 | 0.0002 *** | |
| PU3 | 0.0743 ± 0.0092 | <0.0002 | 0.0025 *** | |
| PU4 | 0.0728 ± 0.005 | <0.0002 | 0.0008 *** | |
| PU5 | 0.0712 ± 0.0038 | <0.0002 | 0.0005 *** | |
| PU6 | 0.0711 ± 0.0025 | <0.0002 | 0.0002 *** | |
| PU7 | 0.0726 ± 0.003 | <0.0002 | 0.0003 *** | |
| Mn (mg/L) | AN5 | 0.0549 ± 0.0055 (*) | 0.02029 ± 0.0004 | 0.0083 ** |
| PU2 | 0.0456 ± 0.0017 | 0.04446 ± 0.0009 | 0.4357 | |
| PU3 | <0.006 | 0.0076 ± 0.0007 | 0.0936 | |
| PU5 | <0.006 | 0.08141 ± 0.0017 | 0.0051 ** | |
| PU7 | <0.006 | 0.00782 ± 0.0007 | 0.0828 | |
| PA1 | <0.006 | 0.00743 ± 0.0007 | 0.104 | |
| PA4 | <0.006 | 0.01908 ± 0.0006 | 0.0111* | |
| SA1 | <0.006 | 0.00835 ± 0.0007 | 0.0642 | |
| Parameters | Community | Method | Test | |
|---|---|---|---|---|
| AAS 1 | ICP-MS 2 | (p-Value) | ||
| Al (mg/L) | AN3 | 0.1227 ± 0.030 (*) | 0.187 ± 0.0295 | 0.1411 |
| AN4 | 0.2122 ± 0.030 (*) | 0.2662 ± 0.042 | 0.3609 | |
| AN5 | 3.4749 ± 0.3475 (*) | 0.6782 ± 0.107 | 0.0058 ** | |
| PA4 | <0.09 | 0.4514 ± 0.0712 | 0.0441 * | |
| Cd (mg/L) | PU1 | 0.0042 ± 0.0014 | <0.00005 | 0.0173 * |
| PU2 | 0.0083 ± 0.0014 | <0.00005 | 0.0051 ** | |
| PU3 | 0.0111 ± 0.0003 | <0.00005 | 0.0001 *** | |
| PU4 | 0.0109 ± 0.0007 | <0.00005 | 0.0007 *** | |
| PU5 | 0.0108 ± 0.0003 | <0.00005 | 0.0001 *** | |
| PU6 | 0.0113 ± 0.0011 | <0.00005 | 0.0017 * | |
| PU7 | 0.0108 ± 0.0002 | <0.00005 | 0.0001 *** | |
| SG1 | 0.0104 ± 0.0003 | <0.00005 | 0.0002 *** | |
| SG2 | 0.0103 ± 0.0004 | <0.00005 | 0.0002 *** | |
| Zn (mg/L) | AN1 | 0.0074 ± 0.001 (*) | 0.0021 ± 0.0006 | 0.0178 * |
| AN2 | 0.0069 ± 0.001 (*) | 0.0064 ± 0.0006 | 0.5472 | |
| AN3 | 0.0067 ± 0.001 (*) | 0.0037 ± 0.0006 | 0.0499 * | |
| AN4 | 0.006 ± 0.001 (*) | 0.004 ± 0.0006 | 0.1028 | |
| AN5 | 0.0086 ± 0.001 (*) | 0.0198 ± 0.0007 | 0.0046 *** | |
| PU2 | <0.003 | 0.0043 ± 0.0006 | 0.0925 | |
| PU3 | 0.0037 ± 0.001 (*) | 0.0086 ± 0.0006 | 0.0196 * | |
| PU4 | 0.004 ± 0.001 (*) | 0.0073 ± 0.0006 | 0.0417 * | |
| PU5 | 0.0036 ± 0.001 (*) | 0.0029 ± 0.0006 | 0.4205 | |
| PU6 | 0.0068 ± 0.001 (*) | 0.0055 ± 0.0006 | 0.2028 | |
| PU7 | 0.0055 ± 0.001 (*) | 0.0046 ± 0.0006 | 0.3253 | |
| PA1 | 0.0133 ± 0.0013 (*) | <0.0005 | 0.0018 *** | |
| PA2 | 0.0091 ± 0.001 (*) | <0.0005 | 0.0022 *** | |
| PA3 | 0.0074 ± 0.001 (*) | 0.0024 ± 0.0006 | 0.0188 * | |
| PA4 | 0.0116 ± 0.0012 (*) | 0.0129 ± 0.0006 | 0.2427 | |
| SG1 | 0.0039 ± 0.001 (*) | 0.0031 ± 0.0006 | 0.3693 | |
| SG2 | 0.0040 ± 0.001 (*) | 0.009 ± 0.0006 | 0.0188 * | |
| SA1 | 0.0144 ± 0.0014 (*) | 0.004 ± 0.0006 | 0.0077 ** | |
| SA2 | 0.0133 ± 0.0013 (*) | 0.0019 ± 0.0006 | 0.0059 ** | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Quispe-Coica, A.; Fernández, S.; Acharte Lume, L.; Pérez-Foguet, A. Status of Water Quality for Human Consumption in High-Andean Rural Communities: Discrepancies between Techniques for Identifying Trace Metals. J 2020, 3, 162-180. https://doi.org/10.3390/j3020014
Quispe-Coica A, Fernández S, Acharte Lume L, Pérez-Foguet A. Status of Water Quality for Human Consumption in High-Andean Rural Communities: Discrepancies between Techniques for Identifying Trace Metals. J. 2020; 3(2):162-180. https://doi.org/10.3390/j3020014
Chicago/Turabian StyleQuispe-Coica, Alejandro, Sonia Fernández, Luz Acharte Lume, and Agustí Pérez-Foguet. 2020. "Status of Water Quality for Human Consumption in High-Andean Rural Communities: Discrepancies between Techniques for Identifying Trace Metals" J 3, no. 2: 162-180. https://doi.org/10.3390/j3020014
APA StyleQuispe-Coica, A., Fernández, S., Acharte Lume, L., & Pérez-Foguet, A. (2020). Status of Water Quality for Human Consumption in High-Andean Rural Communities: Discrepancies between Techniques for Identifying Trace Metals. J, 3(2), 162-180. https://doi.org/10.3390/j3020014






