Potential of Calabash (Lagenaria siceraria) and Sweet Potato (Solanum tuberosum) for the Remediation of Dichlorodiphenyltrichloroethane-Contaminated Soils in Tanzania
Abstract
:1. Introduction
2. Materials and Methods
2.1. Description of Study Sites
2.2. Soil Sampling and Analysis
2.3. Pot and Field Experiments
2.4. Plant Sampling and Analysis
2.5. Disposal of Polluted Soils and Plants
2.6. Statistical Analysis
3. Results and Discussion
3.1. POP Concentration in Soils at PPO Tengeru and NHC Morogoro
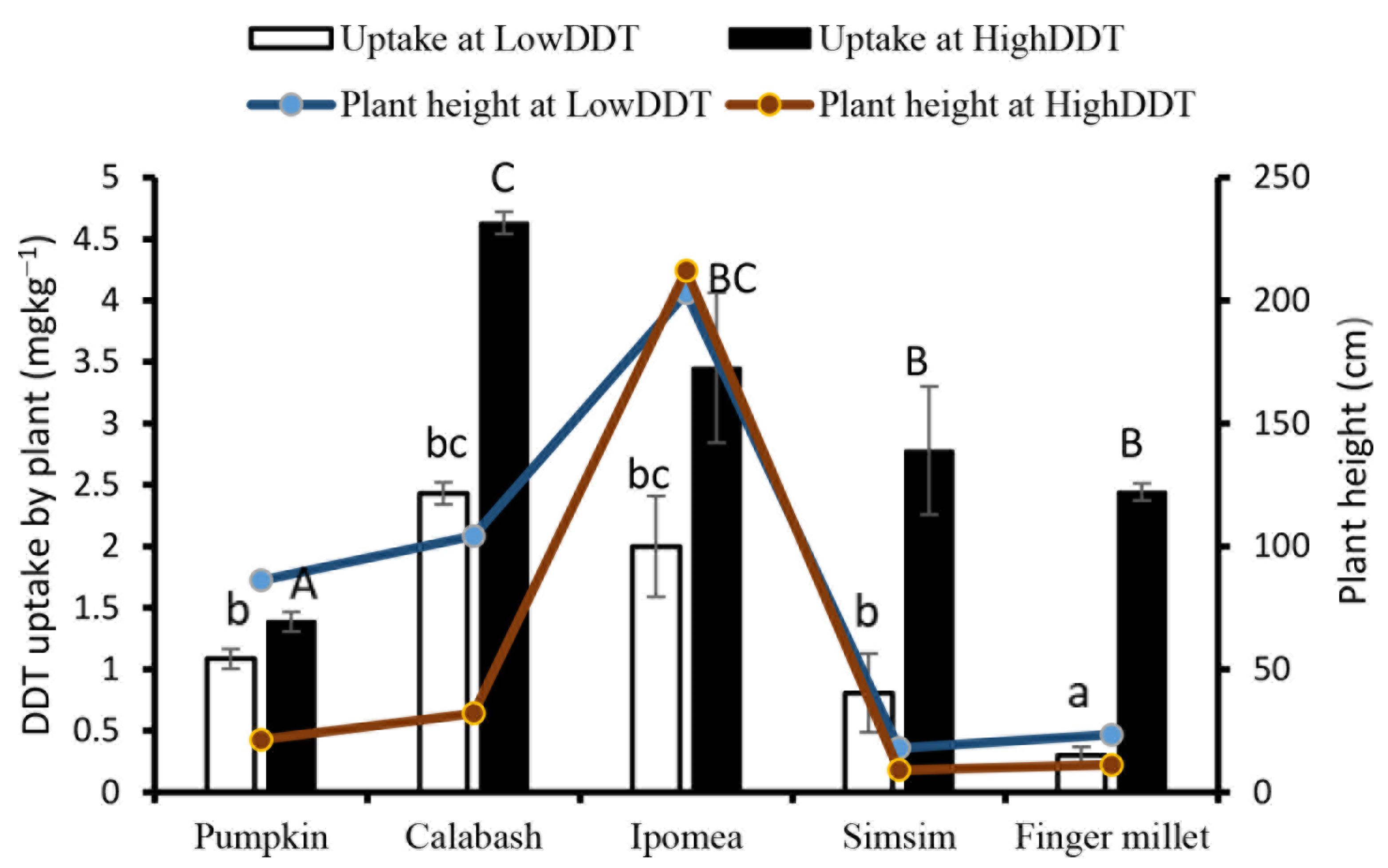
3.2. Effects and Uptake of Residual DDT on Plant Growth under Screenhouse Conditions
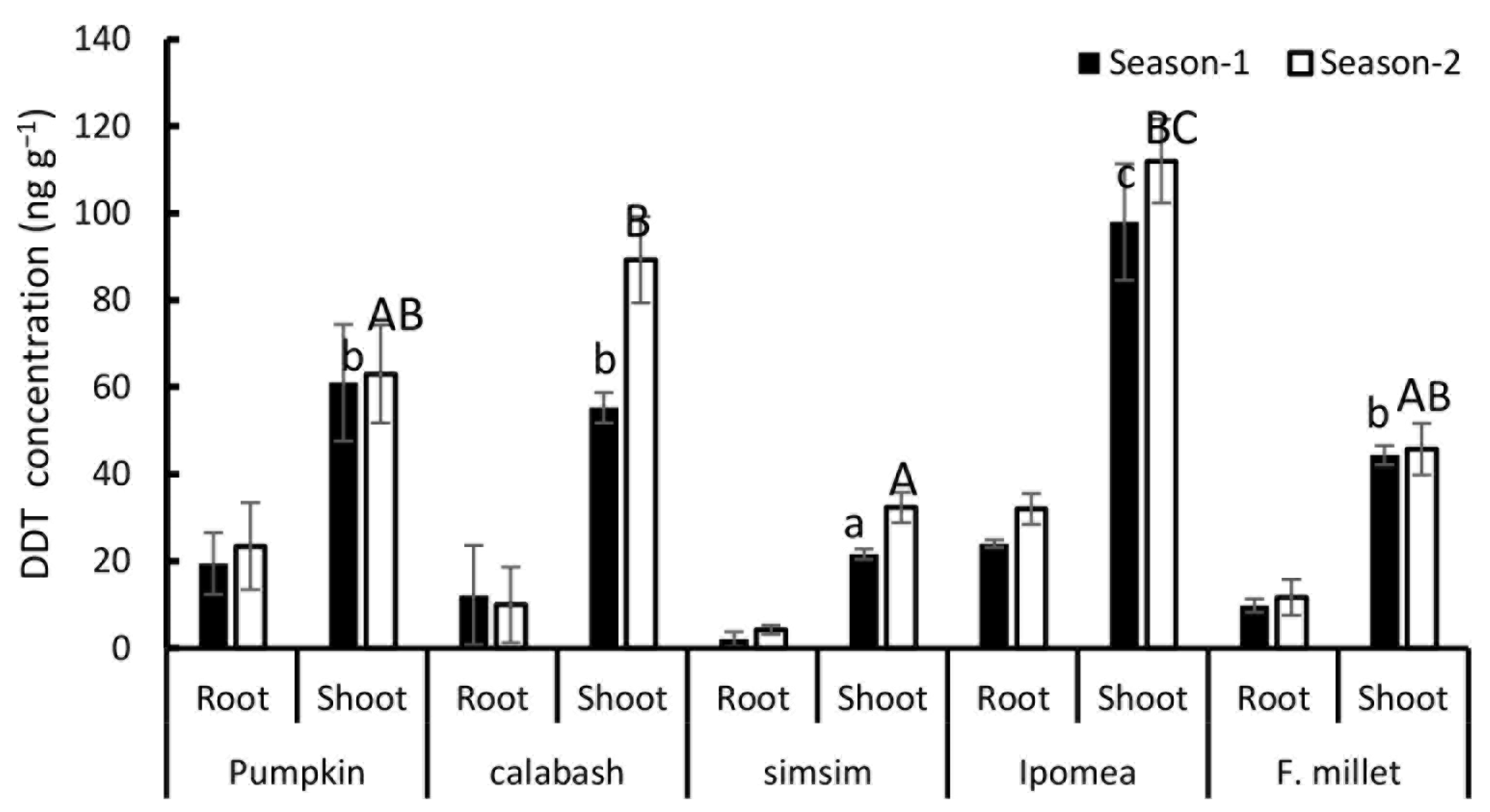
3.3. Plant Uptake of DDT and Metabolites from Contaminated Soils of PPO Tengeru and NHC Morogoro Sites
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Tang, H.P. Recent development in analysis of persistent organic pollutants under the Stockholm Convention. TrAC Trends Anal. Chem. 2013, 45, 48–66. [Google Scholar] [CrossRef]
- Weber, R.; Varbelow, H.G. Dioxin/POPs legacy of pesticide production in Hamburg: Part 1-securing of the production area. Environ. Sci. Pollut. Res. 2013, 20, 1918–1924. [Google Scholar] [CrossRef] [PubMed]
- National Implementation Plan (NIP), Tanzania. The Stockholm Convention on Persistent Organic Pollutants (POPS); UNIDO: Viena, Austria, 2006; p. 31. [Google Scholar]
- Riva, C.; Binelli, A.; Rusconi, F.; Colombo, G.; Pedriali, A.; Zippel, R.; Provini, A. A proteomic study using zebra mussels (D. polymorpha) exposed to benzo(α)pyrene: The role of gender and exposure concentrations. Aqu. Toxicol. 2011, 104, 14–22. [Google Scholar] [CrossRef] [PubMed]
- UK Environmental Agency-UK. Remediation Position Statements. Operational Instructions. 2010. Available online: www.wamitab.org.uk/useruploads/files/waste/.../remediation_position_statements.pdf (accessed on 15 January 2021).
- Lodolo, A.; Gonzales-Valencia, E.; Miertus, S. Technologies for Persistent Toxic Substances. Arch. Ind. Hyg. Toxicol. 2001, 52, 253–280. [Google Scholar]
- Rahuman, M.S.M.; Pistone, L.; Trifirò, F.; Miertus, S. Destruction technologies for polychlorinated biphenyls (pcbs). In Proceedings of Expert Group Meetings on POPs and Pesticides Contamination: Remediation Technologies and on Clean Technologies for the Reduction and Elimination of POPs; ICS-UNIDO Publications: Vienna, Austria, 2000; 55p. [Google Scholar]
- Li, L.Y. Remediation Treatment Technologies: Reference Guide for Developing Countries Facing Persistent Organic Pollutants; UNIDO: Vienna, Austria, 2007; 140p. [Google Scholar]
- Kajiwara, N.; Noma, Y.; Matsukami, H.; Tamiya, M.; Koyama, T.; Terai, T.; Sakai, S. Environmentally sound destruction of hexachlorobutadiene during waste incineration in commercial- and pilot-scale rotary kilns. J. Environ. Chem. Eng. 2019, 7, 103464. [Google Scholar] [CrossRef]
- Yao, Z.; Lib, J.; Xie, H.; Yu, C. Review on remediation technologies of soil contaminated by heavy metals. Proc. Environ. Sci. 2012, 16, 722–729. [Google Scholar] [CrossRef]
- Li, L.Y.; Iwayemi, A.; Li, F.; Comives, T.; Chakrabarti, T. Persistent Organic Pollutants: Contaminated Site Investigation and Management Toolkit; UNIDO: Viena, Austria, 2016; 336p. [Google Scholar]
- Russell, K. The Use and Effectiveness of Phytoremediation to Treat Persistent Organic Pollutants; Environmental Careers Organization: Washington, DC, USA, 2005; pp. 1–49. [Google Scholar]
- Komives, T.; Gullner, G.; Bittsanszky, A.; Pascal, S.; Laurent, F. Phytoremediation of persistent organic pollutants. Cereal Res. Commun. 2009, 37, 537–540. [Google Scholar]
- Lunney, I.A.; Zeeb, A.B.; Reimer, J.K. Uptake of weathered DDT in vascular plants: Potential for phytoremediation. Environ. Sci. Technol. 2004, 38, 6147–6154. [Google Scholar] [CrossRef]
- Suresh, B.; Sherkhane, P.D.; Kale, S.; Eapen, S.; Ravishankar, G.A. Uptake and degradation of DDT by hairy root cultures of Cichorium intybus and Brassica juncea. Chemosphere 2005, 61, 1288–1292. [Google Scholar] [CrossRef]
- Parzych, E.A. Accumulation of chemical elements by organs of Sparganium erectum L. and their potential use in phytoremediation process. J. Ecol. Eng. 2016, 17, 89–100. [Google Scholar] [CrossRef]
- Abhilash, P.C.; Tripathi, V.; Edrisi, S.A.; Dubey, R.K.; Bakshi, M.; Dubey, P.K.; Ebbs, S.D. Sustainability of crop production from polluted lands. Energy Ecol. Environ. 2016, 1, 54–65. [Google Scholar] [CrossRef]
- Khan, S.; Cao, Q.; Zheng, Y.M.; Huang, Y.Z.; Zhu, Y.G. Health risks of heavy metals in contaminated soils and food crops irrigated with wastewater in Beijing, China. Environ. Pollut. 2008, 152, 686–692. [Google Scholar] [CrossRef] [PubMed]
- World Bank. Africa First Stockpiles Program Project: Environmental Assessment (Vol. 6): Country Environmental and Social Assessment (CESA) for Safeguarding and Disposal of Obsolete Pesticides in Tanzania, Inglês. 2012. Available online: https://documents.worldbank.org/pt/publication/documents-reports/documentdetail/191501468008710695/country-environmental-and-social-assessment-cesa-for-safeguarding-and-disposal-of-obsolete-pesticides-in-tanzania (accessed on 4 October 2023).
- Okalebo, J.R.; Gathua, K.W.; Woomer, P.L. Laboratory Methods for Soil and Plant Analysis a Working Manual, 2nd ed.; TSBF TSBT-CIAT and SACRED Africa: Nairobi, Kenya, 2002; p. 8. [Google Scholar]
- UNIDO. Capacity Strengthening and Technical Assistance for the Implementation of the Stockholm Convention (SC) National Implementation Plans (NIPs) in African Least Developed Countries (LDCs) of the SADC and COMESA Sub-Regions. Independent Terminal Evaluation. 2019. Available online: https://www.gefieo.org/sites/default/files/documents/projects/tes/3942-terminal-evaluation.pdf (accessed on 4 October 2023).
- Polder, A.; Müller, M.B.; Lyche, J.L.; Mdegela, R.H.; Nonga, H.E.; Mabiki, F.P.; Mbise, T.J.; Skaare, J.U.; Sandvik, M.; Skjerve, E.; et al. Levels and patterns of persistent organic pollutants (POPs) in tilapia (Oreochromis sp.) from four different lakes in Tanzania: Geographical differences and implications for human health. Sci. Total Environ. 2014, 488–489, 252–260. [Google Scholar] [CrossRef] [PubMed]
- Kariathi, V.; Kassim, N.; Kimanya, M.; Yildiz, F. Pesticide exposure from fresh tomatoes and its relationship with pesticide application practices in Meru district. Cogent Food. Agric. 2016, 2, 12. [Google Scholar] [CrossRef]
- Zhang, W.; Lin, Z.; Pang, S.; Bhatt, P.; Chen, S. Insights into the biodegradation of lindane (γ-hexachlorocyclohexane) using a microbial system. Front. Microbiol. 2020, 11, 522. [Google Scholar] [CrossRef] [PubMed]
- Usmani, Z.; Kulp, M.; Lukk, T. Bioremediation of lindane contaminated soil: Exploring the potential of actinobacterial strains. Chemosphere 2021, 278, 130468. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.; Yu, N.; Wang, L.; Gupta, D.; He, Z.; Wang, K.; Zhu, Z.; Yan, X.; Li, T.; Yang, X. The phytoremediation potential of bioenergy crop Ricinus communis for DDTs and cadmium co-contaminated soil. Biores. Technol. 2011, 102, 11034–11038. [Google Scholar] [CrossRef] [PubMed]
- Neitsch, J.; Schwack, W.; Weller, P. Modern Agrochemicals Influence Bioaccumulation of Incurred DDT Soil Residues in Pumpkins—Residue Risk or a Chance for Phytoremediation? Int. J. Res. Environ. Sci. 2018, 4, 13–23. [Google Scholar]
- Roro, A.G.; Beshir, H.M.; Terfa, M.T.; Tesfaye, B.; Nadew, F.; Olango, T.M. Accumulation of Heavy Metals in Soil and Sweet Potato (Ipomoea batatas) Irrigated with Treated and Untreated Textile Effluents. J. Appl. Sci. 2019, 19, 837–847. [Google Scholar] [CrossRef]
- Ragonezi, C.; Nunes, N.; Oliveira, M.C.O.; de Freitas, J.G.R.; Ganança, J.F.T.; de Carvalho, M.Â.A.P. Sewage Sludge Fertilization—A Case Study of Sweet Potato Yield and Heavy Metal Accumulation. Agronomy 2022, 12, 1902. [Google Scholar] [CrossRef]
- Toishimanov, M.; Abilda, Z.; Daurov, D.; Daurova, A.; Zhapar, K.; Sapakhova, Z.; Kanat, R.; Stamgaliyeva, Z.; Zhambakin, K.; Shamekova, M. Phytoremediation Properties of Sweet Potato for Soils Contaminated by Heavy Metals in South Kazakhstan. Appl. Sci. 2023, 13, 9589. [Google Scholar] [CrossRef]
- Nenman, D.V.; Milam, C.; Michael, D.P.; Silas, K.R. Potential of Sweet Potato (I. Batatas) for Phytoremediation of Heavy Metals and Organochlorine Residues from Abandoned Mine Agricultural Areas of Riyom LGA, Plateau State, Nigeria. Am. J. Appl. Chem. 2022, 10, 104–113. [Google Scholar]
- Huang, F.; Zhou, H.; Gu, J.; Liu, C.; Yang, W.; Liao, B.; Zhou, H. Differences in Absorption of Cadmium and Lead Among Fourteen Sweet Potato Cultivars and Health Risk Assessment. Ecotoxicol. Environ. Saf. 2020, 203, 111012. [Google Scholar] [CrossRef] [PubMed]
- Gupta, A.K.; Verma, S.K.; Khan, K.; Verma, R.K. Phytoremediation Using Aromatic Plants: A Sustainable Approach for Remediation of Heavy Metals Polluted Sites. Environ. Sci. Technol. 2013, 47, 10115–10116. [Google Scholar] [CrossRef]
- Yan, A.; Wang, Y.; Tan, S.N.; Mohd Yusof, M.L.; Ghosh, S.; Chen, Z. Phytoremediation: A Promising Approach for Revegetation of Heavy Metal-Polluted Land. Front. Plant Sci. 2020, 11, 359. [Google Scholar] [CrossRef]
- Mitton, F.M.; Gonzalez, M.; Monserrat, J.M.; Miglioranza, K.S.B. Potential use of edible crops in the phytoremediation of endosulfan residues in soil. Chemosphere 2016, 148, 300–306. [Google Scholar] [CrossRef]
- Mitton, F.M.; Miglioranza, K.S.B.; Gonzalez, M.; Shimabukuro, V.M.; Monserrat, J.M. Assessment of tolerance and efficiency of crop species in the phytoremediation of DDT polluted soils. Ecol. Eng. 2014, 71, 501–508. [Google Scholar] [CrossRef]
- Mo, C.-H.; Cai, Q.Y.; Li, H.Q.; Zeng, Q.Y.; Tang, S.R.; Zhao, Y.C. Potential of different species for use in removal of DDT from the contaminated soils. Chemosphere 2008, 73, 120–125. [Google Scholar] [CrossRef]
- Ilinskiy, A.; Vinogradov, D.; Politaeva, N.; Badenko, V.; Ilin, I. Features of the Phytoremediation by Agricultural Crops of Heavy Metal Contaminated Soils. Agronomy 2023, 13, 127. [Google Scholar] [CrossRef]
- Daryabeigi, Z.A.; Mühling, K.H. Phytoremediation Capability and Copper Uptake of Maize (Zea mays L.) in Copper Contaminated Soils. Pollutants 2022, 2, 53–65. [Google Scholar] [CrossRef]



| Soil Property | Value and Rating | |
|---|---|---|
| NHC Morogoro | PPO Tengeru | |
| pH 1.2.5 H2O | 7.02 n | 6.78 n |
| OC% | 2.36 m | 2.67 n |
| Total N% | 0.53 h | 0.67 h |
| OLSEN available P (mg kg−1) | 2.90 l | |
| Bray 1 Available P (mg kg−1) | 2.19 l | |
| CEC cmol(+)kg−1 | 11.40 l | 10.9 l |
| Na cmol(+)kg−1 | 0.15 l | 1.11 l |
| K+ cmol(+)kg−1 | 2.51 h | 2.71 h |
| Mg++ cmol(+)kg−1 | 2.95 m | 2.90 m |
| Ca++ cmol(+)kg−1 | 3.53 h | 3.83 h |
| Sand% | 56.20 | 28 |
| Silt% | 9.30 | 35 |
| clay% | 32.50 | 37 |
| Textural class | Sandy clay loam | Clay loam |
| Parameters | Optimum Conditions |
|---|---|
| GC parameters | |
| Injection volume | 1 µL |
| Carrier gas (% purity) | Helium (99.999%) |
| Injection mode | Splitless |
| Flow control mode | Linear velocity |
| Injector temperature | 270 °C |
| Linear velocity | 63.5 cm/s |
| Column flow | 1.5 mL/min |
| Purge flow | 3.0 mL/min |
| Equilibrium time | 3.0 min |
| Sampling time | 2.00 min |
| Oven temperature programming | 70 °C held for 1.0 min, ramped @ 25 °C/min to 180 °C, ramped @ 8 °C/min to 300 °C and held for 5 min |
| MS parameters | |
| Ion source temperature | 270 °C |
| Interface temperature | 280 °C |
| Solvent cut time | 2.0 min |
| Acquisition mode | SIM |
| Ionization method | EI |
| Field Sample ID | A2 | A3 | A4 | B1 | B4 | BC2 | C3 | E2 | F2 | NS1 | NS2 | TZ | Canada |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pesticide | Concentration (mg kg−1 Soil) | Permissible Level (mg kg−1) | |||||||||||
| Total Lindane | 12.940 | 2.000 | 0.01 | ||||||||||
| α-Lindane | <0.056 | <0.056 | 5.400 | <0.056 | <0.056 | <0.056 | <0.056 | <0.056 | <0.056 | <0.056 | <0.056 | 2.000 | 0.01 |
| β-Lindane | <0.052 | <0.052 | 5.400 | <0.052 | <0.052 | <0.052 | <0.052 | <0.052 | <0.052 | <0.052 | <0.052 | 2.000 | 0.01 |
| ɣ-Lindane | <0.059 | <0.059 | 2.140 | <0.059 | <0.059 | <0.059 | <0.059 | <0.059 | <0.059 | <0.059 | <0.059 | 2.000 | 0.01 |
| Total DDT | 4.300 | 11.355 | 0.915 | 0.657 | 5.000 | 0.078 | |||||||
| p,p-DDT | <0.004 | <0.004 | 7.355 | <0.004 | <0.004 | <0.004 | 0.822 | 0.456 | <0.004 | <0.004 | <0.004 | 5.000 | 0.078 |
| o,p-DDT | 3.600 | <0.005 | 4.000 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | 5.000 | 0.078 |
| p,p-DDE | <0.006 | <0.006 | <0.006 | <0.006 | <0.006 | <0.006 | <0.006 | <0.006 | <0.006 | <0.006 | <0.006 | 5.000 | 0.05 |
| o,p-DDE | <0.012 | <0.012 | <0.012 | <0.012 | <0.012 | <0.012 | <0.012 | <0.012 | <0.012 | <0.012 | <0.012 | 5.000 | 0.05 |
| p,p-DDD | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | 0.093 | 0.201 | <0.005 | <0.005 | <0.005 | 5.000 | 0.05 |
| op TDE | 0.700 | <0.006 | <0.006 | <0.006 | <0.006 | <0.006 | <0.006 | <0.006 | <0.006 | <0.006 | <0.006 | 5.000 | 0.05 |
| Fenthion | <0.071 | 5.184 | <0.071 | 0.2194 | 0.100 | 0.663 | <0.071 | 0.420 | 0.426 | 5489.24 | 14.998 | ||
| Diazinon | <0.001 | 1.300 | <0.001 | 1.301 | <0.001 | 0.653 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | ||
| Permethrin | <0.015 | <0.015 | <0.015 | <0.015 | <0.015 | <0.015 | <0.015 | <0.015 | <0.015 | <0.015 | <0.015 | ||
| Profile ID | Depth (m) | Pesticide Concentration (mg kg−1) | |||||||
|---|---|---|---|---|---|---|---|---|---|
| p,p-DDT | o,p-DDT | p,p-DDE | o,p-DDE | p,p-DDD | o,p-DDD | p,p-TDE | Total DDT | ||
| P1 | 0.5–1.0 | 0.540 | <0.005 | <0.012 | 0.151 | <0.005 | <0.005 | 2.026 | 2.717 |
| 1.5–2.0 | 0.047 | <0.005 | <0.012 | <0.012 | <0.005 | <0.005 | 0.149 | 0.196 | |
| 2.5–3.0 | <0.004 | <0.005 | <0.012 | <0.012 | <0.005 | <0.005 | <0.006 | <DL | |
| P2 | 0.5–1.0 | <0.004 | <0.005 | <0.012 | <0.012 | <0.005 | <0.005 | <0.006 | <DL |
| 1.5–2.0 | <0.004 | <0.005 | <0.012 | <0.012 | <0.005 | <0.005 | <0.006 | <DL | |
| 2.5–3.0 | <0.004 | <0.005 | <0.012 | <0.012 | <0.005 | <0.005 | <0.006 | <DL | |
| P3 | 0.5–1.0 | 0.639 | <0.005 | 0.284 | <0.012 | 0.743 | <0.005 | 2.843 | 4.509 |
| 1.5–2.0 | <0.004 | <0.005 | <0.012 | <0.012 | 0.009 | <0.005 | <0.006 | <DL | |
| 2.5–3.0 | <0.004 | <0.005 | <0.012 | <0.012 | <0.005 | <0.005 | <0.006 | <DL | |
| P4 | 0.5–1.0 | 0.639 | <0.005 | 0.315 | <0.012 | <0.005 | 0.691 | <0.006 | 1.645 |
| 1.5–2.0 | <0.004 | <0.005 | 0.129 | <0.012 | <0.005 | 0.430 | <0.006 | 0.559 | |
| 2.5–3.0 | <0.004 | <0.005 | <0.012 | <0.012 | 0.110 | <0.005 | <0.006 | <DL | |
| P5 | 0.5–1.0 | <0.004 | <0.005 | <0.012 | <0.012 | <0.005 | <0.005 | <0.006 | <DL |
| 1.5–2.0 | <0.004 | <0.005 | <0.012 | <0.012 | <0.005 | 0.981 | <0.006 | 0.981 | |
| 2.5–3.0 | <0.004 | <0.005 | <0.012 | <0.012 | <0.005 | <0.005 | <0.006 | <DL | |
| Permissible limits | Tanzania | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 |
| Canada | 0.078 | 0.078 | 0.078 | 0.05 | 0.05 | 0.05 | 0.05 | 0.078 | |
| Field Sampling Point ID | A7 | B7 | C7 | D7 | A13 | B13 | C13 | D13 | A25 | B25 | C25 | D25 | TZ | Canada |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pesticide | Concentration (mg kg−1 Soil) | Permissible Level (mg kg−1) | ||||||||||||
| Total Lindane | 13 | 30.11 | 181.02 | 203 | 2.0 | 0.01 | ||||||||
| α-Lindane | ND | 12.01 | 87.11 | 119.50 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | 2.00 | 0.01 |
| β-Lindane | 5.01 | 16.34 | 83.99 | 83.5 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | 2.00 | 0.01 |
| ɣ-Lindane | 8.00 | 1.76 | 9.92 | ND | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | <0.005 | 2.00 | 0.01 |
| Total DDT | 1469.65 | 1591.3 | 319.89 | 889.11 | 8.04 | 313.27 | 1099.49 | 1526.12 | 17.112 | 33.54 | 37.03 | 418.16 | 5.00 | 0.078 |
| p,p-DDT | 8.99 | <0.004 | <0.004 | 5.24 | <0.004 | <0.004 | <0.004 | <0.004 | <0.004 | <0.004 | <0.004 | <0.004 | 5.00 | 0.078 |
| o,p-DDT | 901.89 | 822.05 | 161.11 | 462.6 | <0.005 | 115.55 | 557.52 | 873.79 | 1.06 | 10.35 | 15.25 | 60.43 | 5.00 | 0.078 |
| p,p-DDE | 236.41 | 425.97 | 88 | 39.42 | 4.71 | 114.29 | 299.95 | 286.19 | 6.35 | 18.98 | 10.22 | 229.08 | 5.00 | 0.05 |
| o,p-DDE | 12.98 | 23.29 | 6.79 | <0.0012 | <0.012 | <0.012 | 31.67 | 57.72 | 1.022 | <0.012 | 1.78 | 13.97 | 5.00 | 0.05 |
| p,p-DDD | 309.38 | 316.2 | 63.99 | 108.09 | 3.12 | 83.43 | 195.74 | 303.67 | 8.68 | 4.21 | 9.78 | 112.69 | 5.00 | 0.05 |
| op TDE | <0.005 | <0.005 | <0.005 | 273.76 | 0.21 | <0.005 | 14.61 | 4.75 | <0.005 | <0.005 | <0.005 | 1.99 | 5.00 | 0.05 |
| Aldrin | <0.003 | <0.003 | <0.003 | 17.01 | <0.003 | <0.003 | <0.003 | <0.003 | <0.003 | 0.511 | <0.003 | 1.3 | ||
| Diedrin | <0.003 | 16.63 | 47.51 | 5.65 | <0.003 | 146 | <0.003 | 106.07 | 26.4 | 8.65 | 1.4 | 110.47 | ||
| β-Endosulfan | <0.002 | 51.17 | 14.89 | 667.54 | <0.002 | <0.002 | <0.002 | 5.14 | <0.002 | 54.31 | <0.002 | 27.75 | ||
| α-Endosulfan | <0.001 | <0.001 | 8.45 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | <0.001 | 48.96 | ||
| Simazine | <0.002 | <0.002 | <0.002 | <0.002 | <0.002 | <0.002 | <0.002 | <0.002 | <0.002 | <0.002 | <0.002 | <0.002 | ||
| Profile ID | Depth (m) | Pesticide Concentration (mg kg−1) | ||||||
|---|---|---|---|---|---|---|---|---|
| p,p-DDT | o,p-DDT | p,p-DDE | o,p-DDE | p,p-DDD | o,p-DDD | Total DDT | ||
| P1 | 0.5–1.0 | 1.00 | 10.06 | 0.36 | 3.53 | 1.922 | 10.35 | 27.22 |
| 1.5–2.0 | 1.20 | 6.48 | 1.80 | 0.02 | 6.30 | 0.75 | 23.05 | |
| P2 | 0.5–1.0 | 2.51 | 28.75 | 0.64 | 7.97 | 5.49 | 29.59 | 74.94 |
| 1.5–2.0 | 1.38 | 5.33 | 0.79 | 10.96 | 1.03 | 5. 47 | 24.96 | |
| P3 | 0.5–1.0 | 1.11 | 12.99 | 0.76 | 9.35 | 2.48 | 13.36 | 40.08 |
| 1.5–2.0 | 0.31 | 3.63 | 0.11 | 0.89 | 0.69 | 3.74 | 9.38 | |
| Permissible limits | Tanzania | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 | 5.0 |
| Canada | 0.078 | 0.078 | 0.078 | 0.05 | 0.05 | 0.05 | 0.078 | |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Tindwa, H.J.; Singh, B.R. Potential of Calabash (Lagenaria siceraria) and Sweet Potato (Solanum tuberosum) for the Remediation of Dichlorodiphenyltrichloroethane-Contaminated Soils in Tanzania. Soil Syst. 2024, 8, 1. https://doi.org/10.3390/soilsystems8010001
Tindwa HJ, Singh BR. Potential of Calabash (Lagenaria siceraria) and Sweet Potato (Solanum tuberosum) for the Remediation of Dichlorodiphenyltrichloroethane-Contaminated Soils in Tanzania. Soil Systems. 2024; 8(1):1. https://doi.org/10.3390/soilsystems8010001
Chicago/Turabian StyleTindwa, Hamisi J., and Bal Ram Singh. 2024. "Potential of Calabash (Lagenaria siceraria) and Sweet Potato (Solanum tuberosum) for the Remediation of Dichlorodiphenyltrichloroethane-Contaminated Soils in Tanzania" Soil Systems 8, no. 1: 1. https://doi.org/10.3390/soilsystems8010001
APA StyleTindwa, H. J., & Singh, B. R. (2024). Potential of Calabash (Lagenaria siceraria) and Sweet Potato (Solanum tuberosum) for the Remediation of Dichlorodiphenyltrichloroethane-Contaminated Soils in Tanzania. Soil Systems, 8(1), 1. https://doi.org/10.3390/soilsystems8010001





